{
"cells": [
{
"cell_type": "markdown",
"metadata": {},
"source": [
"\n",
"*This notebook contains course material from [CBE30338](https://jckantor.github.io/CBE30338)\n",
"by Jeffrey Kantor (jeff at nd.edu); the content is available [on Github](https://github.com/jckantor/CBE30338.git).\n",
"The text is released under the [CC-BY-NC-ND-4.0 license](https://creativecommons.org/licenses/by-nc-nd/4.0/legalcode),\n",
"and code is released under the [MIT license](https://opensource.org/licenses/MIT).*"
]
},
{
"cell_type": "markdown",
"metadata": {},
"source": [
"\n",
"< [PID Control - Laboratory](http://nbviewer.jupyter.org/github/jckantor/CBE30338/blob/master/notebooks/04.10-PID-Control.ipynb) | [Contents](toc.ipynb) | [Interactive PID Control Tuning with Ziegler-Nichols](http://nbviewer.jupyter.org/github/jckantor/CBE30338/blob/master/notebooks/04.12-Interactive-PID-Control-Tuning-with-Ziegler--Nichols.ipynb) >
 "
]
},
{
"cell_type": "markdown",
"metadata": {},
"source": [
"# Implementing PID Control in Nonlinear Simulations\n",
"\n",
"A task common to many control projects is to simulate the PID control of a nonlinear process. This notebook demonstrates the simulation of PID for an exothermic stirred tank reactor where the objective is to control the reactor temperature through manipulation of cooling water through the reactor cooling jacket.\n",
"\n",
"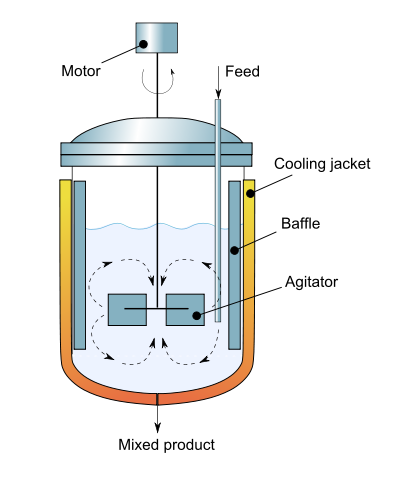\n",
"\n",
"(Diagram By [Daniele Pugliesi](http://commons.wikimedia.org/wiki/User:Daniele_Pugliesi) - Own work, [CC BY-SA 3.0](http://creativecommons.org/licenses/by-sa/3.0), [Link](https://commons.wikimedia.org/w/index.php?curid=6915706))"
]
},
{
"cell_type": "markdown",
"metadata": {},
"source": [
"## Model\n",
"\n",
"The model consists of nonlinear mole and energy balances on the contents of the well-mixed reactor.\n",
"\n",
"\\begin{align*}\n",
"V\\frac{dc}{dt} & = q(c_f - c )-Vk(T)c \\\\\n",
"\\rho C_p V\\frac{dT}{dt} & = wC_p(T_f - T) + (-\\Delta H_R)Vk(T)c + UA(T_c-T)\n",
"\\end{align*}\n",
"\n",
"where $c$ is the reactant concentration, $T$ is the reactor temperature, and $T_c$ is the cooling jacket temperature. The model is adapted from example 2.5 from Seborg, Edgar, Mellichamp and Doyle (SEMD), parameters defined and given in the table below.\n",
"\n",
"The temperature in the cooling jacket is manipulated by the cooling jacket flow, $q_c$, and governed by the energy balance\n",
"\n",
"\\begin{align*}\n",
"\\rho C_p V_c\\frac{dT_c}{dt} & = \\rho C_p q_c(T_{cf}-T_c) + UA(T - T_c)\n",
"\\end{align*}\n",
"\n",
"Normalizing the equations to isolate the time rates of change of $c$, $T$, and $T_c$ give\n",
"\n",
"\\begin{align*}\n",
"\\frac{dc}{dt} & = \\frac{q}{V}(c_f - c)- k(T)c\\\\\n",
"\\frac{dT}{dt} & = \\frac{q}{V}(T_i - T) + \\frac{-\\Delta H_R}{\\rho C_p}k(T)c + \\frac{UA}{\\rho C_pV}(T_c - T)\\\\\n",
"\\frac{dT_c}{dt} & = \\frac{q_c}{V_c}(T_{cf}-T_c) + \\frac{UA}{\\rho C_pV_c}(T - T_c)\n",
"\\end{align*}\n",
"\n",
"These are the equations that will be integrated below.\n",
"\n",
"| Quantity | Symbol | Value | Units | Comments |\n",
"| :------- | :----: | :---: | :---- | |\n",
"| Activation Energy | $E_a$ | 72,750 | J/gmol | |\n",
"| Arrehnius pre-exponential | $k_0$ | 7.2 x 1010 | 1/min | |\n",
"| Gas Constant | $R$ | 8.314 | J/gmol/K | |\n",
"| Reactor Volume | $V$ | 100 | liters | |\n",
"| Density | $\\rho$ | 1000 | g/liter | |\n",
"| Heat Capacity | $C_p$ | 0.239 | J/g/K | |\n",
"| Enthalpy of Reaction | $\\Delta H_r$ | -50,000 | J/gmol | |\n",
"| Heat Transfer Coefficient | $UA$ | 50,000 | J/min/K | |\n",
"| Feed flowrate | $q$ | 100 | liters/min | |\n",
"| Feed concentration | $c_{A,f}$ | 1.0 | gmol/liter | |\n",
"| Feed temperature | $T_f$ | 350 | K | |\n",
"| Initial concentration | $c_{A,0}$ | 0.5 | gmol/liter | |\n",
"| Initial temperature | $T_0$ | 350 | K | |\n",
"| Coolant feed temperature | $T_{cf}$ | 300 | K | |\n",
"| Nominal coolant flowrate | $q_c$ | 50 | L/min | primary manipulated variable |\n",
"| Cooling jacket volume | $V_c$ | 20 | liters | |"
]
},
{
"cell_type": "code",
"execution_count": 1,
"metadata": {
"collapsed": true
},
"outputs": [],
"source": [
"%matplotlib inline\n",
"import matplotlib.pyplot as plt\n",
"import numpy as np\n",
"from scipy.integrate import odeint\n",
"import seaborn as sns\n",
"sns.set_context('talk')\n",
"\n",
"Ea = 72750 # activation energy J/gmol\n",
"R = 8.314 # gas constant J/gmol/K\n",
"k0 = 7.2e10 # Arrhenius rate constant 1/min\n",
"V = 100.0 # Volume [L]\n",
"rho = 1000.0 # Density [g/L]\n",
"Cp = 0.239 # Heat capacity [J/g/K]\n",
"dHr = -5.0e4 # Enthalpy of reaction [J/mol]\n",
"UA = 5.0e4 # Heat transfer [J/min/K]\n",
"q = 100.0 # Flowrate [L/min]\n",
"Cf = 1.0 # Inlet feed concentration [mol/L]\n",
"Tf = 300.0 # Inlet feed temperature [K]\n",
"C0 = 0.5 # Initial concentration [mol/L]\n",
"T0 = 350.0; # Initial temperature [K]\n",
"Tcf = 300.0 # Coolant feed temperature [K]\n",
"qc = 50.0 # Nominal coolant flowrate [L/min]\n",
"Vc = 20.0 # Cooling jacket volume\n",
"\n",
"# Arrhenius rate expression\n",
"def k(T):\n",
" return k0*np.exp(-Ea/R/T)\n",
"\n",
"def deriv(X,t):\n",
" C,T,Tc = X\n",
" dC = (q/V)*(Cf - C) - k(T)*C\n",
" dT = (q/V)*(Tf - T) + (-dHr/rho/Cp)*k(T)*C + (UA/V/rho/Cp)*(Tc - T)\n",
" dTc = (qc/Vc)*(Tcf - Tc) + (UA/Vc/rho/Cp)*(T - Tc)\n",
" return [dC,dT,dTc]"
]
},
{
"cell_type": "markdown",
"metadata": {},
"source": [
"## Simulation 1. Same Initial Condition, different values of $q_c$\n",
"\n",
"The given reaction is highly exothermic. If operated without cooling, the reactor will reach an operating temperature of 500K which leads to significant pressurization, a potentially hazardous condition, and possible product degradation. \n",
"\n",
"The purpose of this first simulation is to determine the cooling water flowrate necessary to maintain the reactor temperature at an acceptable value. This simulation shows the effect of the cooling water flowrate, $q_c$, on the steady state concentration and temperature of the reactor. We use the scipy.integrate function `odeint` to create a solution of the differential equations for entire time period."
]
},
{
"cell_type": "code",
"execution_count": 2,
"metadata": {
"collapsed": true
},
"outputs": [],
"source": [
"# visualization\n",
"def plotReactor(t,X):\n",
" plt.subplot(1,2,1)\n",
" plt.plot(t,X[:,0])\n",
" plt.xlabel('Time [min]')\n",
" plt.ylabel('gmol/liter')\n",
" plt.title('Reactor Concentration')\n",
" plt.ylim(0,1)\n",
"\n",
" plt.subplot(1,2,2)\n",
" plt.plot(t,X[:,1])\n",
" plt.xlabel('Time [min]')\n",
" plt.ylabel('Kelvin');\n",
" plt.title('Reactor Temperature')\n",
" plt.ylim(300,520)"
]
},
{
"cell_type": "markdown",
"metadata": {},
"source": [
"Once the visualization code has been established, the actual simulation is straightforward."
]
},
{
"cell_type": "code",
"execution_count": 3,
"metadata": {},
"outputs": [
{
"data": {
"text/plain": [
""
]
},
"execution_count": 3,
"metadata": {},
"output_type": "execute_result"
},
{
"data": {
"image/png": "iVBORw0KGgoAAAANSUhEUgAAA7gAAAEhCAYAAABVzwz5AAAABHNCSVQICAgIfAhkiAAAAAlwSFlz\nAAALEgAACxIB0t1+/AAAIABJREFUeJzs3Xl8FOX9wPHPzOy9uU+uEM4ESMIlpwJVQFQKAqIVLbWK\nBbFqFUVB26JVi7QieP4qIKiIJxXFiop4gMollygg9xHO3Mkmm73n+f2xyZIlAYKGQOB5v7qdmWee\nmfnOGDL7zTzzPIoQQiBJkiRJkiRJkiRJDZx6rgOQJEmSJEmSJEmSpLogE1xJkiRJkiRJkiTpgiAT\nXEmSJEmSJEmSJOmCIBNcSZIkSZIkSZIk6YIgE1xJkiRJkiRJkiTpgiATXEk6i2Qn5Weurq6ZvPaS\nJElSbcj7Rd2T11Q6lwznOgBJqgt/+MMf+P7778PKjEYjcXFx9O3bl4ceeojo6Oh6i8fr9fLUU08x\ncOBALrvssrNyjF27djF37lxWr15NcXExTZs2ZciQIdx2221YrdazcsyzqaSkhEcffZS//OUvtGrV\n6lft6+2336akpITx48cDwZ+PhIQEZs6cWRehSpIkSb/AxXKvXrt2Lbfccssp64wYMYJp06bV2THP\nJ2vXruX999/n3//+97kORbpIyQRXumBceuml3HvvvaFlt9vNjz/+yEsvvURJSQkvvvhivcWSm5vL\nW2+9xRVXXHFW9v/VV18xYcIEunXrxqRJk4iNjWXLli3Mnj2b1atXM3fuXEwm01k59tmyfft2Pv30\nU+65555fva85c+YwePDg0PKjjz6K0Wj81fuVJEmSfp2L4V6dkZHBu+++G1p+6623WLVqVdi5xcXF\n1ekxzycLFy7k8OHD5zoM6SImE1zpghETE0Pnzp3Dynr16oXT6WTWrFk4nU7sdvs5iq7u5OXlMXny\nZAYPHsxTTz0VKu/duzedO3dm9OjRvPvuu/zhD384h1GeX9q0aXOuQ5AkSZK4OO7VERERYef4xRdf\nYDKZqp23JElnh3wHV7rgRUREAOHvg3z99dcMHz6crKwsBgwYwJtvvhm2jcPh4B//+Af9+vUjMzOT\nPn36MHXqVHw+X6hOYWEhDz74ID179qR79+785S9/IScnh0OHDjFgwAAAxo4dy+TJk4FgU6gXX3yR\nQYMG0bFjR0aMGMHXX38d2t/atWtJT0/nnXfeoXfv3vTp04eysrJq5/PBBx/gcrl48MEHq63r3r07\nd999N40aNQqVHThwgLvvvpuePXvSrVs3JkyYQE5OTmj95MmTmThxIrNnz6Zv37507NiRO+64I6wO\nwPz587nqqqvo1KkTw4YNC4tdCMHs2bPp378/WVlZXHfddaxdu7bauW3cuJHrr7+erKwsrrrqKr74\n4ovQ+srmXIMHD+aFF17g0KFDpKenM3/+fPr160ePHj3Yv38/Xq+XGTNmMHDgQDIzM+nVqxeTJ08O\nXav+/ftz+PBh5syZQ//+/YFgs7gJEyaE4snPz2fy5Mn06dOHzp07M3bsWPbs2RNa/8ILL3DTTTex\naNEiBg4cSFZWFr///e/ZvXt3tWsuSZIk/XoX2r26toQQzJo1K3T/HDlyJOvWrQutX7VqFenp6axf\nv57hw4fTsWNHRo4cye7du/nkk08YOHAgXbt2ZcKECZSXl4dts2rVKgYPHkynTp0YPXo0O3bsCDv2\nhg0bGDVqFFlZWfTr148XX3wRXddD6/v168ezzz7L8OHD6dSpE0uWLAGCT2iHDRtGx44d6dKlC7ff\nfnvoHjpx4kT+97//sXHjRtLT0zl27BgzZ86kX79+Ycf++uuvQ+sBbrrpJp544glGjx5Nx44dmTNn\nDhB8Hev222+nc+fO9OrViyeffBKPx/OLr7d0kRCSdAEYPXq0uPfee4XP5wt9HA6HWL58uejdu7cY\nO3ZsqO6KFStEu3btxMMPPyy++eYb8cILL4j27duLBQsWhOrcfvvtYtCgQeKTTz4Rq1atEs8884xI\nS0sT7777rhBCCJ/PJ4YOHSr69+8vFi9eLL766isxZMgQMWLECOHxeMTnn38u0tLSxKuvvioOHDgg\nhBDinnvuEV26dBGvv/66WLFihZg4caJIT08XX331lRBCiDVr1oi0tDRx1VVXieXLl4tPPvmkxnO9\n9dZbxY033lir63L48GHRo0cPccMNN4hly5aJjz/+WFx55ZVi4MCBorS0VAghxKRJk0TXrl3FjTfe\nKL788kvx0Ucfie7du4t77703tJ85c+aIDh06iJkzZ4qVK1eKRx99VGRkZIiffvpJCCHEM888IzIz\nM8XLL78sVqxYIe6//36RmZkptmzZEnZuV1xxhXjjjTfEypUrxa233iqysrJEUVGRKC0tFQsWLBBp\naWliyZIl4ujRo+LgwYMiLS1NXHbZZWLp0qXigw8+EEIIMWXKFNG7d2+xaNEisWbNGjFnzhyRkZEh\nZsyYIYQQYuvWreKyyy4TDz74oNi6dWvo5+O+++4TQghRWloqBgwYIAYNGiQ+/vhjsWzZMjFy5EjR\no0cPceTIESGEEM8//7zo2rWrGDx4sPjkk0/EsmXLxBVXXCGuv/76Wl13SZIkqbqL6V5d1dNPPy2u\nuOKKGtf961//EllZWWLWrFlixYoVYsKECSIzM1P8/PPPQgghVq5cGbp/Llq0SHz55ZeiT58+on//\n/mLIkCFi2bJlYsGCBSI9PV3MmjUrbJuePXuK2bNniy+//FJcd911omfPnqK4uFgIEbxXZmZmirvu\nukssX75cvPrqq6Jjx45i+vTpodj69u0rMjIyxGuvvSaWLl0qcnJyxOLFi0X79u3FrFmzxJo1a8Si\nRYtEnz59xO9//3shhBAHDhwQY8aMEUOHDhWbNm0SHo9HzJgxQ/Tt2zfsvL/66iuRlpYmjh49KoQQ\nYtSoUaJDhw5ixowZYvny5WL37t3i6NGjokePHuLmm28WX375pXjvvfdEjx49xIQJE057zaWLm0xw\npQvC6NGjRVpaWrVPly5dxOTJk0O/0IUQYuTIkWLMmDFh28+cOVP07NlTeL1e4XK5xK233ipWr14d\nVmfo0KHioYceEkIIsWzZMpGWlha6AQkRvFn0799fZGdnh5KzFStWCCGE+Pnnn0VaWppYvHhx2D5v\nueUWce211wohjt80K2/MJzN48OBa/3L/5z//KXr06BFKZoUQIjs7W3To0EG8+uqrQohggpuZmSlK\nSkpCdZ555hnRuXNnIYQQgUBA9OjRQ/zzn/8M2/fo0aPFq6++KoqKikRmZmboxlrp5ptvFnfeeWfY\nub311luh9Xv37hVpaWnis88+C6uze/duIYQIXcPKxLXSPffcIz766KOwsvHjx4durkIIccUVV4in\nn346LNbKBPe1114TGRkZ4uDBg6H1DodDdO/eXTz55JNCiGCCWzUWIYR45513RFpamigsLBSSJEnS\nmbuY7tVVnSzBLSgoEBkZGeKVV14Jlem6Lm688UZxzz33CCGOJ6tV758zZ84UaWlpYtOmTaGy0aNH\ni7vvvjtsm5deeim0Pi8vT2RlZYl58+YJIYS46667xNChQ0UgEAjVefvtt0VGRkboPte3b9+we6sQ\nQrz00kth91chgn8Eb9euXWj5gQceEKNGjQot1zbBPfEaTZ06VVx66aXC6XSGyr755huRlpYmduzY\nISTpZGQTZemC0adPH/773/+ycOFCpkyZgsVi4brrrmPq1KmhXhnLy8vZsmUL/fr1w+/3hz59+vSh\nqKiIXbt2YbFYePXVV+nZsyfZ2dmsWLGC2bNnU1BQEGr2tGnTJho1akS7du1Cx+/QoQNffvklKSkp\n1WLbsGEDiqJw9dVXh5UPHjyY7du3hzVvatmy5SnPU1VVAoFAra7Jhg0b6NOnT6jpF0BKSgpZWVls\n3Lgx7JhRUVGh5eTkZFwuFwD79u2juLi4Wiccb7zxBrfeeiubN2/G6/XSt2/fate0ajNlgE6dOoXm\nK5tRVx7nZE68Hs8//zxDhw7l6NGjrFy5ktdee409e/aENUk7lQ0bNpCZmUmzZs1CZZGRkfTt25cN\nGzaEyiIiImjduvUZxytJkiSd3MVyr66NH374AZ/PF3b/DAQCp71/JiQkhM6lUmxsLKWlpWHb/Pa3\nvw3bplOnTqF7/7p167jsssvQdT3s+vp8PjZt2nTS8/zzn//MxIkTKS4uZv369SxcuJAVK1ag63qt\nv5ucTIsWLcKW161bR7du3TCZTKEYe/bsidForHZ9JKkq2cmUdMGIiooiKysLgI4dO2K325k0aRKJ\niYnccccdQPB9HSEEU6dOZerUqdX2kZeXBwQ7hPjnP//JkSNHSEhIoHPnzpjN5tC7QSUlJWfUA2JJ\nSQmRkZHVejaOj48HwOl0hspOt98mTZqE3lmpSX5+PjExMRgMBhwOR+gYVcXFxYXdqC0WS9h6VVVD\n51pcXAwEb541qVw/fPjwGtdXTQjNZnPYMYCw931qcuL1WL9+PY8++ii7d+8mJiaGzMxMLBZLrcfc\nO9U12bJlS2j5xGuiKEqt4pUkSZJO7mK5V9dG5f1z6NChNa73er2h+RM73tI07bSjJSQmJoYtx8bG\nUlxcjBCC4uJi5s2bx7x586ptV3l9ofp55uTk8PDDD7Ny5UqsVivp6ek1vj/9S5x4rOLiYrZu3cpn\nn312yhgl6UQywZUuWMOHD2fx4sW8+OKLXH311aSmpoZ+Cd9///1ceuml1bZJTU1l//793Hfffdx8\n882MHTs2dIO44YYbQvUiIyMpKiqqtv2KFStCN+6qoqOjKS0txev1ht2Q8vPzQ+trq3fv3kyfPp3i\n4mJiYmKqrb///vtxOp28//77REVFUVBQUK1OQUEBTZo0qdXxIiMjAaqd77Zt29A0LfTkd+7cuTWe\nR10OV1RaWsqdd95Jnz59mD17Nk2bNgXgvvvu48iRI7XaR1RUFLm5udXKCwoK6nX8RUmSJOnCvVfX\nRuX987XXXgtraVXJYPh1X9NLSkqw2Wyh5cLCQhISElAUhcjISEaMGMG1115bbbvKe2tN7r//fkpL\nS/nwww9JT09HVVXeeOMNvvvuu5NuoyhKtT8OV3aIdSqRkZFcc8013H777dXWnZi8S1JVsomydEF7\n+OGH8fv9PPPMM0Cw2WlaWhqHDx8mKysr9CksLOSFF17A4/Gwbds2fD4fd9xxR+gXaH5+Pjt37gz9\ngu7UqRNHjx4N65Fw586djBs3jr1796JpWlgcXbt2RQhR7a+Qn376Ke3bt6/2tPBUrr32WiwWC9On\nT6+2bvXq1axbt45rrrkmdNzvvvsu7GntwYMH2bJlS62HK6hsvvzNN9+Elf/tb39j/vz5dOzYEYPB\nQElJSdg1XblyJW+//Xa1a3Eytam3d+9eHA4Ht912W+gG7Ha72bhxY9jNs/LpcE26du3Kli1bOHTo\nUKisrKyM7777Tg7hIEmSdA5ciPfq2ujYsSOaplW7f37zzTcsXLjwlPey2lixYkVoPi8vj82bN9O9\ne3cAunTpQnZ2dthxAZ599lkKCwtPus/NmzczYsQI2rdvH4pv5cqVwPEWTideV5vNRnFxMW63O1RW\n9ZWgk+nSpQt79+4lIyMjFGNiYiLPPPMM2dnZtbkE0kVKPsGVLmhpaWkMHz6cRYsWsWnTJrp06cLd\nd9/NhAkTsNvt9OvXj0OHDjF9+nQyMjJITEykXbt2aJrGtGnTGDlyJLm5ubz88st4PJ5Qc9v+/fuT\nlpbGPffcw3333YfZbObZZ5+la9eudO3aNfSXyW+//ZZmzZrRvn17Bg4cyGOPPUZxcTEtW7bk448/\nZu3atWc8qH1cXByPPfYYDz30EDk5OYwcOZKoqCg2bNjAvHnz6NGjB3/84x8BuPXWW/nggw8YM2YM\n48aNw+v18txzz5GcnMzIkSNrdTyj0cif/vQnnn/+eex2O127dmXp0qXs2rWLqVOnEh8fz6hRo3js\nscfIz88nPT2d77//nv/85z/ce++9tT6vyifFX375ZVhT5qpatmyJzWbjueee4/bbb6e0tJR58+aR\nm5sb2h6CfxXfvHkzP/zwQ7WkdeTIkcyfP5/bb7+de++9F5PJxOzZs9F1PXTdJEmSpPpzId6rayMp\nKYkbbriBRx99lNzcXNLS0lizZg0vv/wyDzzwwK/e//Tp0xFCkJSUxEsvvURiYiIjRowAYPz48dxy\nyy088sgjDB48mKKiImbOnElERES1d2GrysjI4J133iElJQWz2cyHH37I8uXLgeArSSaTicjISLKz\ns1m9ejVdu3alb9++PPPMM0yZMoXrrruODRs2sHTp0tPGP2bMGBYvXsxdd93FqFGj8Hq9vPDCCxQX\nF5Oenv6rr4904ZJPcKUL3n333YfFYuHf//43AFdddRUzZsxg1apVjB07lueff57f/va3PP/88wC0\natWKqVOn8sMPPzBu3DheeOEFBgwYwPjx4/npp58IBAKYTCbmzZtHZmYmf//733nkkUdo164dL730\nEqqqEhERwZgxY3j33Xf517/+BcAzzzzD7373O2bPns1dd93Fnj17+M9//sPAgQPP+JyGDBnC66+/\njqZpPPnkk9x5550sXbqU8ePHM2vWLIxGIxBsZvTmm28SGRnJxIkT+cc//kFmZiZvv/12jc2hTuaO\nO+7ggQceYNGiRYwfP56tW7cyZ86cUMcdjzzyCH/84x95/fXXGTt2LEuWLGHSpEmMHz++1sdo27Zt\n6L9DTe8EQTBxfe6558jNzWX8+PFMmzaN9PR0/v73v7N///7Q+0x/+tOf2LFjB2PHjsXv94ftIzIy\nkjfeeIP09HT+9re/MWnSJGJjY3n33XdP2SxLkiRJOnsuxHt1bfz9739n9OjRvPrqq/zpT3/i008/\n5ZFHHmHs2LG/et+TJ0/mtddeY+LEiSQlJfHGG2+EmixfcsklvPLKK+zZs4c777yTp556iu7duzN3\n7txTNo2eNm0aycnJPPjgg0yaNAmn08krr7wCBDvNAhg1ahRms5lx48axY8cO2rdvz6OPPsq6desY\nN24cmzZtYtq0aaeNv3nz5ixYsAC32829997LX//6V1JSUpg/f758pUg6JUX82jfCJUmSJEmSJEk6\nL6xatYrbbruNzz//nNTU1HMdjiTVO/kEV5IkSZIkSZIkSbognJME98cff6RPnz4nXf/xxx8zYMAA\nOnfuzB133BHqvU6SJEmSJEmSJEmSTqZemygLIXj//feZNm0amqbVOEjz9u3b+f3vf8+8efNIT0/n\niSeeIDc3lzlz5tRXmJIkSZIkSZIkSVIDVK9PcF9++WXmz59/yo5n/ve//zFgwAA6deqExWJh4sSJ\nfPvtt/IpriRJkiRJkiRJknRK9Zrgjhw5ksWLF9c4uHalvXv30qZNm9BybGws0dHR7Nu3rz5ClCRJ\nkiRJkiRJkhqoeh0HNykp6bR1XC5XtYG0rVZraEyz0xFCoCjKL4pPkiRJkqS6l5dXWif7URSF+Hg7\nBQVOGtIgEDLu+iXjrl8y7vrXUGOvy7gTEyNPuq5eE9zasFgsuN3usDKXyxUat+t0CgqcqHXwXFpV\nFWJi7BQXO9H1hvODI+OuXzLu+iXjrn8NNfa6jDsurvZjRktnl6oGvyCpKgQC5zqa2pNx1y8Zd/2S\ncde/hhp7fcV93iW4rVu3DmuOXFhYSElJCa1bt67V9kKIOr1gui4IBBrOl7pKMu76JeOuXzLu+tdQ\nY2+ocUuSJEmS9MucdwnukCFDGD16NCNHjiQrK4sZM2bQr18/YmNjz3VokiRdJIQQIAKIig9CRyBA\n6MF5oQfX6QJd96PrOkIPoAd0dD2A0INTXegIPbhN0VEDTqcrVKdyP7quVxxLgBAV+w5O0YPbi4rj\nVl1fdTk4FUDFPAKlYoqA4P9VLHN8GSFA4fi0olwJ1QlSFI43JQptE14nWLGmRFJQZYOKelQ5Tk31\nK+ucoGL/1VeJsMLgKSnYzVm0vPSaGo8iSZIkSdKF6bxIcKdMmQLA448/Tvv27XniiSf461//Sl5e\nHt26deOpp546xxFKklQfhNARuq/i40eIimnFvEIA4VYpLnLg83nR/T78fi96wBf6CN0XTDJFAKEH\ngskjwYSSKlMFHRQ9OEVHUY5/1BoTtV/HfZLyU75RoQBanYdy0SjO2w7IBFeSJEmSLibnJMHt2bNn\n2Bi4jz/+eNj6wYMHM3jw4PoOS5KkX0AIHT3gDn78rtC80L3oAS8Bvxu/143f70H3ewj4vQj9+Ad8\nIHwoih9V+XXvFygVn1DSWK/9xNdM1xWEqPoBXSggFPSKsuDD1OA6RMXz0yoPTIMPYpXjD16Bqg9j\ng3WV4/NV8vOw5YpjhTYPrat6zKrPR5XQ/5/47LSyTKn2PPVknfwpYWtr3rZyRXi5UuWZsoJS9eCh\n64UCilBCB1AUjXbdLz9JLJIkSZIkXajOiye4kiSde0IIRMBNwF9OwO9E95ej+50E/OXo/nICfhd+\nrwu/r7wigfWA8KDgO+NjVSaiofymlh2f67qCrqsEAioBXUUPTbWKqRJMKAOh1sShh7ZCB3RRsRyc\nBpdFxVQPm1f04LIqgkmVoigoqCiqgqpoKKqKqigomoaqqiiKhqppqJqKqhpQDQYMBgOq0YBm1IiI\nsuHx68H6BgOqQUHVNBSDAUVTUTQNVA1F0yrmq5aduF4Nr6sF46ncDkWps97kNU0hLi6CwsKyBvUu\na9W4JUmSJEm6eMgEV5IucLruI+ArDX68pQR8DvRAGcWH3Licxfi8TnRfObruqmiuWzunSp/8fi34\nCQSngcDx5UBAI+BX0X2g+0D4BcInwCvAp4MvgOrzo/gDKH4/qi8QLPMH0AJ+VOFH0wPBqfCj6gEM\nBLCaDGgWM5rVgmqxolqtqJaKeYsF1WxGMZlQTWYUswnVZDq+bKpcNgenZnNovVIH3bI31CRRkiRJ\nkiSpoZEJriQ1YELoBHyl+D1F+L0l+L1FBLwloYTW7y1F6Cd7+zPciQlrIKDg9Rrx+kx4vQZ8PiM+\nvwGfz4DPb0D3a4iAhvABHh1R7gWnG9XlweD3YtQ9GAMuDLoHY8CLUfdh1b3BpFQEqjdsNRhQIyLQ\n7BFodjtaVASqPTY4b7ej2iMwRtqJSY7H6QdMllASq5jMdZKISpIkSZIkSQ2bTHAl6TynB7z4PYX4\nPAX4PQXBRNYTTGT93hI4g6euPp+G22PG4zHh9piCCazXiMcbnEexoiomVN2AUQ9g9Lkwuh0YSgvR\nSnKwekqJ9rswBdyopzuuoqBFRKLFRGGIikeLisIQFYUWHY0hKhotKir4iYwKJrAm02nj1zSFmLgI\ndPkkVJIkSZIkSaqBTHAl6TwghCDgc+Bz5+P3FOBzF+D35ONzFxDwOWq1D6/XQLnLgstlwe0x43YH\nk1hPRUKrGiOx2u1ERJqxW1WirX6EtwCDKw9D4RGUYwfRiwqO9250CorBgCE2HkNsXMUnFmNcxXxc\nHIaYGLTIKPlUVZIkSZKkUxJCEKgYszygCwK6HhzDXBf4dVExnrlesS74CZWJ4HaV9XURnA926Bic\n18WJy8Ex0hUFzBYjTqcnuA8RXCdOso+q64LlwQH4KvcpKsqD51TRt0nlfEUPkWFlFXX1igIdKjqS\nFBXrq9YXoU4mK7dTVJVAIFARJxVDBoZvD8HYKq+zqLIPUaU8FPPx/yrH11eJi9A8oW2q1j1eVuWc\nq/y31lSFW4d0oF9Wo1/2w1JLMsGVpHqmB7z43Hn4XDl43bn4XDn4XDnogVM3Jfb7VZzlVlwuSyiR\nPT41YzRZiIqxBj+xFpJirETaNcyuQrS8I/gO78F75Ajeo0cJlJWG7fuEjndBUTDExWFMTMKYmIgp\nMSk0b4iLR4uMrLNOjCRJkiRJqj+6LvD4Anj9Oj5fAI9fx+sLBD9+Ha9Pxx8IfnwBHb9fxx8QJ5SJ\n0HxAF6iqitPlxeevrF9RLyDw+3UCul4lWRVVklW9Nn9Xly4QAV2w93CJTHAlqSHTdR++8mN4yg/j\nLT+Ct/wofk/BKbcpd5lxOq04nTbKnDbKnFac5VbcbjOqphITZyM23kZ8io22CXaiY4NJrcHnwn1g\nP56D+/DsyMadfYDynBzKT3Hn0KKiMDVqjKlJU0yNG2NKTg4msvEJKAb560GSJEmSzhV/QMftDeD2\n+iumFfOeQLVyl9ePx1s1Sa0y9el4/AF8Ph2vP4D/AnvFR1UUNE1BVRVURUFVQFWDowlUzquKgqIE\n64aWVQWTQUPX9WBdlYptTtzHqfYZXFaqHJuKYwVnldCoEWrFgwG1YmVovVI5Op5SUValnGCBqhC2\nraYq2O1mysu9oWHzKvcf2r7KfKic48erHHav8nmFgsLhg3tY+MaL5Bw5QEJyU343+m5atG4X2r6y\nx5aNa5fz8aLXKXUU0bZ9J35/2wSiYmKprHH8GUiV8wBMJo0eWU0pdZTX7Q/BCeQ3WEmqI0II/J4C\nPM6DeJ1H8JQfxufK5WTvyHp9BhwOO6VlEThK7ZSW2ilz2ggENBQFYuJtJCRF0LZ1BDHxwaQ2KsaC\nqqoIvx/PoUO49/6I67s9HNm7B19e7klj0yKjMDdvjrlJU0yNm2Bp1oTkDm0p9SnyXVZJkiRJOgt0\nXVDu8eN0+yh3V536KXf7TpgGPx5/AKfLh9sbwOevfR8bdcmgqRgNCgZNDc5rKgaDikFTgvMVy8F5\nBaNRI8JmIuAPoKkqBkOVehV1DJqKph5PRA1qcFmtKNMqlkNlqoKmHS/TqpRVJrSVddXj2dQZacgj\nHJyN2D0eD9P++iS33DKGoUOH89lnS3j5P0/w3nuLsdlsoXq7d+/iv2++yIwZL9GmTRtmznyaj955\ngenTn69V3EbD2X99TSa4kvQLCaHjKc8jt/QYBbk7cZceQPfX/Bcpj9dMUVEExSWROEqDCa3HYwIU\nDEaV+KQImrWJICEpgoTkCOIS7BiMWmj7gMuFa9dOCr7cjnvvHtz79yF8NY8/a0xOxpzSHHNKcyzN\nUzGnNEeLjg5rUqxpCsbICJBjhEqSJElSrfj8AUrLfRUfL6XlPhwV09ITpy4fLo//rMRhMqpYTAYs\nJq3ic3wCLl/YAAAgAElEQVTeZNQwGzRMRhWTsWJq0DAZTliuMjUbNYwV640G9YwTxoacKErHbdy4\nHkVRGDHiegCGDBnGe++9xerVKxkw4MpQvc8//5Q+fX5DRkYmAHfeeQ9DhlxJYWEBcXHx5yT2E8kE\nV5JqqfIJrcuxB3fpXjzObETAU62erhtxlEaRn2+juCSSYkckHo85tD423kbLtCiSm0aR3CSK2AQ7\nqhp+M9HdLpw7dlG+fTuuHdtxH9gPevW/5GqRUVhat8baqjWWVq2xtGiBarHW+blLkiRJ0oXK6wtQ\n7PRSXOqhuMxDcZm3YuqpKAsuu72BX30sq1nDZjZitxiwWQzYLcbQNMJmJCHWRsAfwGysnrxWzp/4\nnUFqePwBnUJH7YZxrImqKXh0heKScvTT/FEhLsqCQTv9U9Ps7P20aNEqrKx581Sys/dXq5eR0TG0\nHB0dQ1RUFNnZB2SCK0kNge534y7bF0xqHXsI+Eqq1xEWiktiOHrUTkFRNKWldirfNtA0heSm0TRO\niaZR02iSm0Rithir7UPoOp6D2Th/+hHnlp9w791TPaFVFMzNUrC2TcPSug3WVq0xJCTIzp4kSToj\nc+fOZebMmRiNx38XzZkzh7Zt2/LII4+wZs0aIiMjueuuu7jhhhuA4B/4ZsyYwcKFCwkEAgwbNoyH\nH34YTdNOdhhJOi8IIXCU+8gvcVFQ4qbA4Sa/xB2aLy714HT/sietEVYjkTYjkVYjkXYTkTZTcN5m\nJNJmwm4NT2CtZg3tFKMLyCehFwd/QOeR2WvIL/nlCe6ZSIi2MHVcr9MmuS6XC4vFElZmNltwu90n\n1HPXqt65JBNcSTqB31NMecl2XCU78JRlc0L/wgjMlDoTOXzYTk5uJE6nlcqEVlUVGjeLoknzGJqm\nxpDcJCqsqXFVgXIn5du24vzxR5xbfyJQUj15NqekYE1vhy29Hda26WgREXV9upIkXWS2bdvGhAkT\nuP3228PK//KXv2Cz2Vi1ahU7duxg7NixtG3bls6dO/Pmm2+yfPlyPvroIxRF4Y477mDevHmMHTv2\nHJ2FJB3n8+vkFrvILSwnp8hFbnE5JU4fR/OdFDjcZ/Quq8moEhthJibCTEykmZgIEzERZqIjTETb\nTKFENsJqOGWyKkkNjcViweMJb5no8bixWq0n1DPj8bhrqGfjfCETXOmiJ4TA587FVbyd8pId+FzH\nTqih4BdJ5OXHsHePleKSSI73BwdxCXaatYwlpWUsjVNiMJ4koQXwOxyUbdpI2cb1lG//GQLhzZ0M\nsbHYMrOwZ2ZhS28vE1pJkurczz//zMiRI8PKnE4nX3zxBUuXLsVsNtOxY0eGDBnChx9+SOfOnVm8\neDF//OMfSUpKAuCOO+7gueeekwmuVG90ISgscXOkwMmxQhc5ReWhhLagxE1tnneajCrxURbioy0k\nVEzjIi3BJDYymNRaTJpsGSWdVQZNZeq4Xr+6iXJMtJ3iEmedNVFOTW3J++8vDCvLzj7AlVdeXa1e\ndvaB0HJxcTEOh4MWLVrU/gTOMpngShctn6eQ8sKfcBZtqTZ0j6KacfubcehwDLt3mvH7j/9T0TSF\nJqmxpLaKo0XbeFq1STplcyJfYSFlGzdQtnE9rl07CRvwTdOwtm6DPasT9qwsTE2byRurJElnjcvl\nYt++fcyfP58HH3yQqKgobr/9djp06IDBYCAlJSVUt2XLlnz++ecA7N27lzZt2oSt27dvH0KIWv3O\nqhyC49eqfPewob2DKOOuPSEERaUeDuc7OZzn5HBeGYfynBzJd572HVhNVUiMsdIo3kaz5EgiLYaw\nZDbSZjyv77Hy56R+ncu4NU2jcYL9F2+vqgoxMXashmBv3XWhR48e+HxeFi16lxEjRvLpp59QVFRI\n796XomnHr9FVV13Nn/88lmuvHUa7dh2YPftFeve+lLi42FrFXXV6tsgEV7qoBHxllBdtxVn0E97y\nI2HrVEMEXr05Bw5Es3O7hq4f/zZmMKqkto6ndbtEmreKw2gK/tOp+g8+7Djl5ZRtXI9j9SpcO7aH\nrVNMJuxZHYno2g17Vkc02/nTpEOSpAtbfn4+l1xyCTfddBPPP/88P/74I+PHj+e2226r9k6VxXL8\nnaoT382yWq3ouo7X68VsNnM68fH2Ok0sYmJ++RfDc0nGHS6gCw7nlrLncAm7DxWz51AJ+486cLpq\nHiUAQFUgKc5Gk4QImiTYaZxop2liBE0SIkiKtaLV4knV+U7+nNSvhho31H3sc+e+wmOPPcbLL79E\namoqL7/8Mk2bJjJlyhQAHn/8cXr1uoQnn3ySadOeJC8vj27dujF9+tPExdW+1eHZvuYywZUueELo\nuB27KcvfiMuxi6rv1KoGO7rWhoMH49m2JYDfd3ydyazRok0CrdITSWkZe9J3aUPH8ftxbt2CY/Uq\nnJs3hQ3jo1qt2Dt2JuKSbtgzMlFr8YVQkiSprqWkpLBgwYLQcrdu3Rg2bBjr16+v9u6V2+0OjX14\n4rtZLpcLg8FQq+QWoKDAWWdPcGNi7BQXO+vsqUV9kHEHmxgfyXey94iDA8dK2X+slOycUry+k78f\nmxhjpWminWYVSWyzRDuN4m2YDDXdjwUlJeV1Hnd9knHXr4YaN5y92JOSmvF///dKWFlhYRn33fdQ\naB6gV69+9OrVr1q906nLuE+VUMsEV7pg+b0llBVswlmwiYCvNFSuqCZMEW3JK2jC5rUCR7EXCPag\nqCjQvFU8aZnJtGgTf9qkFsCdk0Pu4k8o/vYbAg7H8eMYDNg7diKq96XYMjuiGqv3nixJklSftm7d\nysqVKxk3blyozOPx0LhxY3w+H0eOHKFJkyYA7Nu3L9QsuXXr1uzbt49OnTqF1rVq1ar6AU5CCHFi\nlwO/iq6LBtnL7MUUt8vjZ+8RR/Dp7OES9hxxnHRcWLNRo3lyBKnJkaQkR9AsMYLG8TYsppq/ptY2\nlovpep8PZNz1r6HGfrbjlgmudEERQuAu3Utp3lrcjt1h68z25viVdmzfYWP3z0XoVcawTWwUQVpm\nI9q0T8JmN53+OH4/ZT9uxvHNcpxbt4S9V2ttm0Zkr0uJ7NYdzd5wm71IknThsdlsvPjiizRv3pxB\ngwaxdu1alixZwoIFCygtLeWZZ57hySefZNeuXXz88cfMnj0bgGuvvZa5c+fSq1cvDAYDs2bNYtiw\nYef4bKTzicPpZXt2ETuyi9l1qITDeWU1dvxkNWukJkfSPDmSFo0iSW0USXKsrcG9vylJ0vlLJrjS\nBUHofpxFP1GauxafOzdUrmpWbLGdyC9qzprvSinIdQLBd8oMRpW0jGQyujQhITmyVscJlJVRvPwr\nipd/RaC4OFSuRUURdVlfovv+BlNFL6OSJEnnm5YtW/Lss88yc+ZMJk+eTHJyMk899RQZGRk88cQT\nPProo/zmN7/BZrPx4IMPhp7Y3nzzzeTn53P99dfj8/kYOnQot9122zk+G+lcKnf72JFdzM8Hitie\nXcShPGeN9RrH22jdJJo2zaJp3TSaxvE21PO4oydJkho+meBKDVrAX05Z3jpK89ej+4/fXE22plhi\nLmHfvmh++uYozrLjQ//EJ9rJ6NqEth2SMZlr90/Ae+woRcs+x7F6JcLrDZXb2ncgZeg1KG07oCun\nb84sSZJ0rvXv35/+/ftXK4+JieG5556rcRtN05gwYQITJkw42+FJ5yldF+w76uDHPQX8tLeAAzml\nYYMCQLAX41ZNokhLiaFN02BCG2GVr+dIklS/ZIIrNUgBXxmO3NWU5a9H6JWdOSnYYtpjsHdl6486\nPy85is97/Clry7QEOnVvRqNm0bXuzbN85w6Kln6Kc/MPoTLFbCG6b19iLh+AtWlj4uIigi/WN8B3\nICRJkiTpZMpcPn7aeIhVmw/z454Cyk7o3VhRoEWjKNqlxtA+NZa2TWMwm+QfeyVJOrdkgis1KH5f\nKaU5qyjL34AQFR1DqSYi4rui2Trz44YStv1wMPTiusGgkt6xER27NSMmrnbD8QghcO3YTsH/FocN\n8WOIjSNmwECi+/0GzSbfrZUkSZIuPIUONxt25LFhRy67DpdUe0qbGGOhY6sEMlrGkZYSg80iv0pK\nknR+kb+VpAYh4C/HcexbSvPXgwh2xaloZiITe6BZu7B5XR7bfvg5lNiaLQY6dmtGRtcmWG2n7zQK\ngolt+batFH78Ea5dO0Pl5pTmxF49mMhLuqEY5D8ZSZIk6cKSW1TOhh15rN+Rx76jjrB1Bk0hvXks\nWS3jyGodT6M4W52OaSxJklTX5Ld16bymB7yU5q3FkbMSoQfffVU1K5FJPTFFduGH73P5af3msMS2\nU48Usi5pWuv3awHKd2wnf9F/ce853vOyuUVL4ocOw96xk7yZS5IkSReU4jIPa7bmsGbbMbJzwsev\ntJoNdG6TQLd2ifTpmoK73NMghyKRJOniJBNc6bwkhE5ZwSZKjq5A9wdvvIpqIiqpN7a4HmzbnM+G\nVZvxuIPNlH9pYus5dJD8Rf/F+ePmUJmlVWvirx2GLSNLJraSJEnSBcPjDbBxZx6rth5j2/7CsObH\nEVYjXdomcEl6Eh1axGLQVDRNwWYx4i73nHynkiRJ5xmZ4ErnHXfpfooOfXZ8uB9FJTKhO5HJl7F/\ndzlrPtxMaUnFUD8GlU49UujcM+WMEltfQQEFixfhWL0qNIatObUFCdddj61DhkxsJUmSpAuCEIJd\nh0r4ZvMRNuzIw+MLhNZZzRqXpCfRs0My7ZrHoKnqOYxUkqRzbfPmH3jxxZlkZ+8nOjqGm2++heHD\nR+JwOHjqqcfZuHEddnsEY8aMZciQ4TXuY+fO7Tz99FT27dtLs2bNmTjxYTIzs+r1PGSCK503vO5i\ncvd8iLNoa6jMFptJTOMrKCkx8vG7uzh6qAQI9tyYntWIHn1bYo801/oYusdD4acfU/TZpwh/8Omv\nMTGRhBHXE9GtO4q8uUuSJEkXAKfbx6qfjrFi8xGO5B8fRk9TFTJbxtE7sxGd2yRgMspejyVJAofD\nweTJ9zNhwkMMHDiIXbt2ct99f6Zp02YsXvw+NpuVjz76nD17djFx4r20aNG6WuLq8XiYNOl+brll\nDEOHDuezz5YwefL9vPfeYmy22nX2Whdkgiudc0L3U5y7hgNHv0WvGPLHZG1MbMo1oDVi7bf7+WnD\noVBTquat4uh1eSvikyJqfwwhKNuwjrz33sFfWAiAFhlJ3NBhxPS7XHYeJUmSJF0Q9hwpYfnGw3y/\nPRefXw+VpyRF0LdjY3p0SCaqlp0vSpJ0dvh1P0Xukl+8vaaB1+SipNxJIHDqurGWaAzq6b/n5uQc\n5dJLL2PQoKsBSE9vR9eul7Bly498++0K3nrrfcxmMx06ZHLllVfx2WdLqiW4GzeuR1EURoy4HoAh\nQ4bx3ntvsXr1SgYMuPKXnewvIL/VS+eUx3mIwuz/4XPnAaAarMQ0HoAtrhO7f85n9VffU+4Mdi4V\nGW2hz5VtaNEm4cyOcfgwuW8vwLX952CBphE78ErihgxDs1rr9HwkSZIkqb7pumDjzjyWrstmz+Hj\nvSAbDSo92idxeeemtGoSJV+/kaTzgF/38/iapylwF9XL8eItsUzp9eBpk9y2bdP5+9+fCC07HA42\nb/6B1q3bomkGmjZtFlrXvHkqK1Z8XW0f2dn7adGiVVhZ8+apZGfv/3UncYZkgiudE3rAS8nRrynN\nWxsqS0zpjTW+L6UOWPLeFg7tD/7D1zSFLr1T6dIzBcMZNKXSPR4KPvqQomVLQQ/+FduWkUnSqJsx\nNW5StyckSZIkSfXM5fHz3Y9HWbb+IPkVfVMANI63cXmXplya2Qi7xXgOI5QkqSEqKytj0qQJpKe3\np2vXbixc+E7YerPZgtvtrrady+XCYrHUqu7ZJBNcqd65HHsoPPgxAW+waYbBkkBii2tp3Kwd33yx\nk1Vf78HnDba3SG0dR58r2xIVc2ZPWst/3kbO/Nfw5QU7qjIkJJB0403YO3eVf8GWJEmSGrTSci+f\nrzvIVxsP4/L4Q+UZLeO4qnsKGS3j5L1Oks5TBtXAlF4P/uomytExdkqK666JcqUjRw7z0EMTaNq0\nKY8//hT79+/H6w3vSd3jcWO1Vn+n1mKx4PHUVLd+W0zKBFeqN7ruo/jwMsry1wcLFJWo5D5EJ/eh\nrNTH6/9ZRfbe4PuxFquRvoPa0rpd4hndpANOJ3kL38Hx3bfBAlUl7urBxA25FtUk3zmSJEmSGi6H\n08vS77P5auPhUG/IBk2hV0YjBnVLodkZ9E0hSdK5Y1ANJNrif/H2mqYQFxGByWut0zGqd+zYzgMP\n3MNVV13DXXfdh6qqpKSk4Pf7OXbsGI0aNQIgO/sALVu2rLZ9ampL3n9/YVhZdvYBrrzy6jqLsTZk\ngivVC2/5EfL3f4DfUwCAydaEuObXYrQksnXTEVZ/tQd/RWcYbTok0WdgG6xn2AlG2aYN5LzxOgFH\n8P0jc2oLkv94G5bmqXV7MpIkSZJUj0rKPHy6Npvlmw7jrbhXmk0a/bs2ZVC3FKIjaj+agCRJUk0K\nCwt44IF7GDXq94wefWuo3Gaz06dPP2bNepFJk/7Gvn17WLZsKU8//Vy1fVxySXd8Pi///e87DB9+\nPZ99toTCwkJ69Ohdj2dSzwnutm3bmDJlCrt37yY1NZV//OMfdO7cuVq9hQsX8vLLL1NcXEzbtm35\n29/+RmZmZn2GKtURIXQcOasoOboc0AGF6Eb9iGrUF1e5ny/+u4UDe4JJb0SUmX6D2pJ6hp1IBVwu\n8t55C8fK4FNbxWQiftgIYgcOQtHk8AeSJElSw+Ty+Pl07QE+X3cQry+Y2FrNGgMuSWFQ9xQirPL9\nWkmS6sbHHy+muLiI11+fy+uvzw2VX3/9KCZN+htPP/0UI0YMxmq18uc//4WMjGBu9vnnnzJ//qss\nWPAeJpOJ6dOfZ/r0p5g16/9o1qwZ06bNuHCbKHs8HsaPH8/48eO54YYbWLx4MXfeeSdffPEFdrs9\nVG/79u1Mnz6dd955h9TUVF555RXuvfdevvzyy/oKVaojfq+DggOL8JRlA2AwxRLfYgRmezMO7Cng\n6yXbcZUHhwVq3S6RETd3xe3xnlFTi/KdOzg2bw7+/HwArG3TSL7tT5iSkur+hCRJkiSpHvj8Oss3\nHeZ/q/ZT5greJ21mA1d2T2Fgt2ay4yhJkurcLbeM4ZZbxpx0/RNPTKuxfNCgaxg06JrQcps2bXn5\n5Xl1Ht+ZqLcEd82aNaiqys033wzA9ddfz+uvv86KFSsYPHhwqN6BAwfQdZ1AIIAQAlVVq/XGJZ3/\n3I695B9YhO4vB8Ae34XYplchhIHvlu3ipw2HATAYVfoMbEtGl8bY7CbcHm+t9q/7fBQs/oCipZ+C\nEKBpJIwYSeygq1FU9aydlyRJkiSdLUII1v6cw6IVe0O9Ihs0lSu7NWNw71SZ2ErSRUKI4MMeQfWH\nPkIIUBT8eoCAHsBfMVLIKR8PiZOvPfVjpZrXnmobcYpjGamflpX1luDu27eP1q1bh5W1bNmSvXv3\nhpX16dOHFi1a8Nvf/hZN07Db7cyfP7++wpR+JSF0HMe+peTYCgAU1UR882uxxXagtMTN5x9uIvdo\nKQBJjSMZeG17omNtZ9SRlDcnh6Oz/g9P9gEATE2b0fhP4zCnNK/7E5IkSZKkepCdU8qby3ay61Cw\nZ1VFgcsyGzO8b0viouQf+hsyIQS60AmIQPCjB+f9enBZFwECQkcoOvm6iaKSMnz+wPH6QidQUbdy\nXzo6uhAIEZwGl/Xj6yvKRGUdKssr6lRsH1w+yT4Qwe0REJonVBZKZBSBZlDx+QIV2xOsj0CI0FxF\n/Zr3IaruHx0E6AgIlVWNpcr2lalWaBKeXIUt11BHUUCvjEGcULFK3eOrRLUa4cvV61SLqYYEsKZE\n9kKkoHBj1lAub9T3rB6n3hLc8vLyau2vLZbq4yJ5PB7atGnDlClTSEtLY86cOdx9990sWbKkVk9y\nFUWhLh7gqaoSNm0ozmXcAZ+T/H0f4HLsAcBkTSap9Q0YLfEc2F3A5x9tw+MKDmfQtXdzev6mJZqm\nnlHcjnXfc3TeXHS3GxSFuEFXkThyJKrx3PSQLH9O6peMu/411NgbatzSxafM5eODb/eyfNPh0Bfs\njq3juf7y1jRLlL0i1xUhBH7dj0f34g0EP56qU92HJ+DFr/vw6X78ur/K1IdfD1RM/Sesr7l+ZRIb\nTGD1c336knReEAhyy/LP+nHqLcG1Wq3Vklm3243NFj6G0osvvkijRo3IysoC4K677uK9995j1apV\n9O/f/7THiY+31+nYbzEx9tNXOg/Vd9zljsPs3vIaPncxAPFNe9C83XBQDKxYuoNvv9gFBIf/GXZT\nZ9IzGtW4n5PFrXu97Hv1dY598hkAxugo0u6/j5jOnc7C2Zw5+XNSv2Tc9a+hxt5Q45YufLoQfPfj\nUf67fE/oPdukWCs3D2xLx9Zn1tnihcyn+3H73bj8Llx+Ny6/u2LZjStwfDlU5nfj1b0E8FPudYcl\nsQ3xKZmqqGiKFvyoKioqqqKgKCqqoqKioCoqSmVZxXKoTkX9YJ0T1yuoVJRX1qlSX1UUFBRQFFQU\nILiNAhXT4PdtTVWxWk143P7QH2nUiu0qax2vX/M+KudDUxSC/6ss54TyYEQolXuA43OE9hm2XHms\nymurqUREmCkr8yB0EapVm21RTliuYe5k+6geX/i+wuopNW+raSpRkRZKS93ouqi2j5qdfO0vyZ1O\njCl8fzWXmzQDnVLTKS4uP+PjnYl6S3BbtWrFggULwsr27dvHkCFDwsqOHDkS9qRXURQ0TUOrZW+4\nBQXOOnuCGxNjp7jYWfGD0zCci7idRT+Tt+8DhO5DUQzEp/6WyITOHDtWxtIPtnJwXxEAiY0iuGZk\nJlExVgoLy2odtzc3h8P/9xLuA8Emybb0djS5Yzx6bGy1/dQ3+XNSv2Tc9a+hxl6XccfFyadoUt06\nVljOa59uZ+fB4B+FTUaVoZe2YFD35hgNF24/Et6AF4e3DKfPSZmvHKfPidNXTtkJU2eVqU/3n/W4\nFBRMmhGTZsKkGjGoRgyqhlE1YlQNGFQDxiplwWVDlanxhGVDRUKqoYUlqFWWK+bVinUGVcNkMJAQ\nF4WjxAW6Gkpcz3eaphAXF0FhYVmdjsl6tjXUuKFK7MaGFbumKaj10FdOvSW4vXv3xuv18sYbbzBq\n1CgWL15Mfn4+ffr0Cat3+eWXM3PmTAYPHkx6ejpvvPEGgUCASy65pFbHEUIQCNRd3LouGtQPTqX6\niFsIgSPn24ohgEAzRpLQ6kbMtibk55Tx6ftbKClyAdChc2MuG9gGg0E7ZVwnxl32wyaOzZ2N7nIF\nmyT/dgjxQ4ejaKfeT32TPyf1S8Zd/xpq7A01bunC5A/ofLL6AIu/248/EGy22i09kVED2jbY92yF\nELj8Loo8JTg8pTi8pZR4HeHz3lIcnlLcAU+dHNOoGrEYzFgNFqyaFavBgsVgqZgGy2MiIwh4wKgE\nE1ezZgqbmtTj80bVcF4kkpqmYDNZcWsBAg3wabMknS/qLcE1mUzMmTOHxx57jBkzZpCamsp//vMf\nbDYbU6ZMAeDxxx/nxhtvxOFwcM899+BwOGjfvj2vvPIKERHyL+jnE133UXhgMeXF2wAw2ZqQ2OpG\nNGMk2XsLWLZ4G15PAFVV6HdVGu07NT6j/Qtdp3DJ/yhY/AEAWmQkjf50B/YMOR6yJEmS1PDsOVTM\nzLc2ciAn2NFiTISJP1yVTpe2iec4slML6AEKXEXklxdR6C6i0F1MkbuIQk9xaN4TqN0ICFUpKNgM\nVuwmG3aDnYiKqd1kI8Jgx260YTfasBmtweQ1lMiaMain/vrakJ/MSZL069VbggvQrl073nnnnWrl\njz/+eGheURTGjRvHuHHj6jM06QwEfE7y9r6Nt/wIALbYLOKbDwVFY/O6g6z+ag9CgMVm5OoRGTRO\niTmj/etuF0fnzsG5aSMA5hYtafLnezDGxdX5uUiSJEnS2aTrgk/WHOCDb/YSqGgu/5vOTbjh8jbY\nLPX6NeykAnqAQncxua588lz55JXnk+cqIM+VT4GrkMAZdJIUYbQTZYokyhRJtDkqOG+uWDZFEmmK\nJMJkx2awoioXbnNsSZLOnfPjN6vUYPg8heTtfhO/N/hebUyTAUQmXYquC775bAfbfzwGQHyinWuu\nzyIy+syaXHmPHePgC8/hPRJMnqMuvYykP/zxnPWSLEmSJEm/VF6xi1c+3hYa+icxxspt17SjXWrs\nOYnHr/vJLc/nqPMYR5w5HHXmcNR5jHxXYa16+rUarMRZYoizxBBrjg1OLTHEWWKJNUcTaYo47dNV\nSZLOf4WFBdxyyygefngKl13WF4fDwVNPPc7Gjeuw2yMYM2YsQ4YMr3HbnTu38/TTU9m3by/NmjVn\n4sSHyczMqtf45W8hqdY8zsPk7X0b3V8OikZ86nDssRl4PX4+//B4Z1It2yYwYGg7jKYz+/Eq2riJ\nff+ege4qB1Ul8Xc3ETNg4HnxXowkSZIk1ZYQglVbjvHmsp24vcGOQQb1TGVkvxYYa9lp5q9V6i0j\nu/QwB0sPcaTsGEecx8gpzztlIqugEGOOJsmWQKItgWRbAq2Tm2EO2Ik2RmM1NMz3hCVJOjPTpj2B\nw1ESWv73v5/EZrPy0Uefs2fPLiZOvJcWLVpXS1w9Hg+TJt3PLbeMYejQ4Xz22RImT76f995bXG3k\nnLNJJrhSrbhKdpK/778I4UfRLCS2/B2WyBaUl3lYsvAn8nOCvRl37plCr8tbnXFSWvjVl+S8uQB0\nHTUigibj78LWrv3ZOBVJkiRJOmtcHj+vf7ad73/OBSDCamTMb9tzZe+WZ+2d0DKfk2zHIbJLD5Nd\neohsxyGKPMUnrW9UDTSyJ9PE3ojG9mSSbIkk2RJIsMRh1IyhevJdVkm6+Hz44X+xWKwkJSUDUF5e\nzuk+gsMAACAASURBVLffruCtt97HbDbToUMmV155FZ99tqRagrtx43oURWHEiOsBGDJkGO+99xar\nV69kwIAr6+0cZIIrnVZZwQ8UZv8PEGjGKBJb34zJmkRRgZMl7/1EaUlwfOO+V7Yl85KmZ7Rvoevk\nvfcOxV98DoC5WTOa3H0vxoTzu9MNSZIkSTrRodwyXvpwCzmFwTEeO7aO57Zr2hF3hq/rnIoQgjxX\nPnuK97O3ZD97SvaTU55XY10FhSRbAs0imtDY3ogmEck0tjciwRon33+VpHNI+P34Cgt/8faaBi5P\nGd4S52lHjzHGxaEYapfyZWcf4J133mT27NcYM2Y0AIcOZaNpBpo2bRaq17x5KitWfF3D9vtp0aJV\nWFnz5v/P3pnHV1He+//9zMzZsyckEBJCIOwg4oKC+wK4gGJdcGtte12rtpfW1tva9tda23qvXuvS\nKthqW1tr3W5LKyiCqIiCKLKHnbAkJJB9OfuZmd8fc3JyTk4ICSSB4PN+vc5rZp555pnvDOGc+cz3\n+3y/Rezbt6dL5+8ppMCVdEpz9Wrqy98BwObMY8Dwm9DsaVSWN/L2GxsJBiKomsKls8YwbFT3RKkR\nCFD5h/l4160FIOO0SeTefhfYZQiURCKRSPoXKzZU8td3txGKGKiK4PoLhzPtzMJjnmZjmiaV3oNs\nq9/Jzobd7GrYQ3O44xrwee4BFKYOZkhqAUNSCyhMzccpw4olkhMKMxKh7Mf/RaSmpk/Op+XkUPzI\no0cUuZFIhF/84qd85zsPkJaWHmv3+wM4HI6Evg6Hk0AgkDSG3+/H6XR2qW9vIgWu5LA0Vn1EY6X1\ndsbuKSB32M0ompM9O2p4d0EpesTA4dS44roJDCxIP8JoiUQa6ql4+kmC+/YCkHHRxYy9/27qG/0y\nDEoikUgk/YZgWOfld7ezYmMlAJmpDu6ZPZ6Swd37XYynMdjEtvqdbK3bwda6HTSGmpL6OFQ7xWlF\nDM8YyrD0oRSlFco5shKJ5Kj5859fYMSIkUyZck5Cu9PpJBRKrGEdDAZwuZLn1DqdToLBjvq6et7g\nTpACV5KEaZo0HniPpkOfAOBIKWbAsDkoqp0dpQd5799bME1ITXdy5Q2nkJndvUnjwf37qHj6SSL1\ndSAEA264kewZMxB9lHhDIpFIJJKeoKrOx7P/2Eh5tReA8cOyuGPmWFLd3cv8b5gG+5rL2VBdysaa\nUg54q5L6pNg8jMgcTkl6McMzhpLvGYiqyN9NiaS/ITSN4kcePeYQ5fR0D409GKL83nvvUltbw7Jl\nSwDwer387Gc/4pZbbiMSiVBVVcXAgQMBK5S5uLg4aYyiomLefPP1hLZ9+/YybdplXbyynkEKXEkC\npmlSX/42LTWfA+BKH0nO0OsQikbpugN8+M52ADJz3MyaMxFPqqOz4ZLwlm7mwO+ewQwGEHY7g+68\nh5RTJ8lMyRKJRCLpV3y29RB/XLSFQEhHCJh93jCunFKE0sXfs7AeZlv9TjbUlLKpppTGUHPCfk3R\nKEkvZnTWCEZnjWBwyiA5b1YiOUkQmoY9N/eoj1dVgSsrBb+j5xLA/e1vbyZsX3fdLObO/QHnnHMe\nO3ZsY/783/Lggz+mrGwXS5Ys5rHHnkoa4/TTzyQcDvHGG39n9uzreOedhdTV1TF58pQesbGrSIEr\niWGaJnX7/o23bh0A7szxZBddjRAq6z7dx8r3dwMwYGAKV95wCq5uvqFuWr2Kqhd+D7qOmp7O4Pvn\n4hw6tKcvQyKRSCSSXiOiG7z2/k6Wfl4OQJrHzl1XjWNMF2rb6obOppptfH5wPRtqNhHUQwn7s51Z\nnDJgLOOzxzAsfSj2uIzGEolEcrx48MEf89hjv+aaa67A5XLxrW99m3HjxgPw7rtv89JLf+Svf30N\nu93O448/zeOP/5r585+loKCARx99QoYoS44PpmlSt/+tmLj1ZE8iq/BKQLB6eRlrPrHmyg4qSOeK\n6ydgd3Szxu3SJVT//WUAbHkDKZj7PZkpWSKRSCTHnWZfCI/L1iXPa11TgOf+uYldB6w5sSMLM7j7\n6nFkpBw+mskwDXbW7WHjrk2s3P8F3rAvYX9RWiGn5IzjlJyxDPLkyYgmiURyQvDGG/+OraelpfOL\nXzzaYb/p0y9n+vTLY9slJSOYN+/FXrevM6TAlbSJ21orm3FK9ulkFl4BwMfv7WTj5xUAFA7LYsY1\n47DZuj7nxzRNav/xJnWL3gLAWTyM/G//J1pqWg9fhUQikUgk3ePjjZW8sHALp5bkcP+1EzoVlxt2\n1fCHt7bQ4g8DcNlZQ7j2gmGoSsdhw3WBelYe+IyVlZ8n1aQtTivijLxTOTV3PBmOo09GJZFIJJJk\npMD9kmOaJvX7F8bErSf7tJi4/ejdHWxeewCAYaNyuPSqsahq1+f/mLrOwb/8iaYVHwHgHjee/Hvu\nQ3HKLI8SiUQiOf58sb0aMNkcXs6jH6/iO2fdituWmDgxEIrw6rKdfLjO+j10OTT+48oxnDYyOQpJ\nN3Q21pTy8YHVbKnbjknb3Lii9MFMGnAKpw2YSLYrq1evSyKRSL7MSIH7JcZKKLWIltovgPiwZFj+\n7g5Ko+J25Pg8LrpiFMph3lJ3hBEMUjn/Wbwb1gOQevYUBn79P7pcaFoikUgkkt6m2R9GOL1oefuo\nCJi8/cZvueKM2bhGjEQ3DD4tPcibH+6mvtkqezEsP407Z40lNzNRBB/yVfPJgc9YVfU5zaG2GrUe\nzc3kQadxbsFZTBhSQl1dzyWEkUgkEknHSLXxJaUtW/IaADxZp5JVOBNIFLejJgzkwstHoShdnxOk\nt7RQ8cyTBHbtBCBz+mXkXHcDohsCWSKRSCSS3iYU1hEOPwAj9waZsLKUvcu28NkF/8GGOpNanxWO\nrCqCq84t5oqzh8RCkkN6mHXVG/nkwGp2NOxOGHdUZglT8yczccB4bIqGqsp5tR1hmiboOqZhgKFj\nRuLWdQNT12Pr1lKPthlt66aBaZhgGGCamKaJIkwMt53mZj+GboBhWvsMw+pvWtsY0XXDjI5jjdH6\nad2Ob29bN8DEarMuBlo99mbbtmlGG2JtcduYUVusdQHUOmwEgyHrmkysKIC4Pm1BAVF7zLZ1iDtH\n/LGt7bHzd/iP0Z1m4gxBCKi0aYRDEet6usrh+nbYfDj7uvnCyEy0u0JTiUR0634fxRhd695B/+7a\n3Q4hoLzV9n70zkyxaYgbroXikb16Hilwv4SYpknDgSWxUkCerIlkDZkFwPLF2yldZxWrHz1hIBd0\nU9yG62qp+M3/Eqq0BHLO9XPImnH5EY6SSCQSiaTvCYUNhBoBIKfBWga0FFr2BykSCi3A2BE5XH9R\nCQOz3Jimyf7mClZWfsbqqrX4I/7YWGn2VM4edAZTB01mgDv7eFxOlzFNEzMSwQyFMEIhzGAQIxTE\nDIasZchaEg4R0AQtDS0Y4bB1TCSMGY60rUci0e3W/ZHEvtH96DqmHrEEYkzUGsf7Vkgkkj6mavES\n8u6WAlfSwzQdXEHzoVUAuDMnHFbcXnjFqG5lcwweqKDiN/9LpL4OVJWBX/8maVPO6fkLkEgkEomk\nBwhFdLDrADhDlthqcOaBUFGBa8d5GTlkEU21hXxSm876ms3U+GtjxwsE47JHMzV/MuOzR6MqXU/C\neDSYpokZDKJ7W9C9Xgy/v+0TsJZ6B22x9ZiYDZ1c4lIIUBTrmUVRQAgURcEUAoRACCXaR1jRZPHt\n0W0R3Y9QENFlrF9rn9Zjo1586xlJgIjaQNwSYa3Gt8c/UwmBaHesUAR2u41QKBI3VrRPXF9B65it\n7Yl9RJwNHfU5/KPdYXYc7oDYpQmcThuBQPjw3sTDjHF4UzrY01N2R1GUNrsNw+ywf7ezmh/hXnWh\nsUsk2d5PUG0aQy6/FN+Rux4TUuB+yWiu/ozGyvcBcKWPIrvoakAcs7j179pJxVO/wfB5EXY7+d+6\nD8/4U3rjEiQSiUTSA9TU1DBr1ix+9atfcdFFF7Fx40ZuuOEGnHGJAO+66y7uvvtuTNPkiSee4PXX\nX0fXda6++mp++MMfoqq9K+h6m1DYAJclJjzCAQQwRNs1Zbl2gxHC2bKLzU0+mtVTSXFfiN1czzmD\nSjh70OnHlAXZNE0Mn49IQwORhnr0xkb0lmb0lhZLxLYkfgxvC2YkcqyXfUSEpiEcDhS7A83lxFRV\nUDUUm83ap2kIzYawaQhVs5atbZqG6LCfCqqKUNTouhK3rlriUVERqmK1ta4r0f2xdQWhapagVdU2\ncRqHqgqyslL63ZxnaXff0l/thv5ru6oKnFkp+Opajtz5GJAC90uEt24j9eVvA+BIGUrO0GvpCXHb\nsmEdlfOexQyFUFJSGPztubiGDe+NS5BIJBJJD/HQQw/R0NBWvmbLli2cf/75zJ8/P6nvyy+/zAcf\nfMC//vUvhBDcddddvPjii9xxxx19aXKPoxsmQlgPh6pp/e6ZWW0Zjm1am5i8wJPBO+bpAKTYp3HZ\n0GIAqhv8PP3GBgZkuLj3K+Njc3SNQCAmXI2mRvwhH00HqtgaqKSCRiZt8yPqGjDD4WO7CFVFcblQ\nXS4UpwvFlfhRXW4UpxPF5ULYHSgOO8JuR7E7otsOFHu0zeFA2O2xnBn99SFaIpF8uZEC90uCr3Eb\ntXv/CYDdnc+AYXNAqIni9hQroVR3xG3TJx9T9acXwDDQsrIpmPs97IPye+UaJBKJRNIzvPLKK7hc\nLgYNGhRrKy0tZfTo0R32X7BgAbfddhu5ubmA5dl96qmn+r3AtcISrVBdNRqxa8vIgahzQVHbwniz\nFBtY0cw0hiKUfrGOgoCX5TtaqKixUVHj5e3HX2Rc0070hgaMQCDpfGEV3vhKDmGbgrfRx7kHE8Wt\nmpKKmpKCmpqK4vFY656UaHt0OyUVxZOC6nGjuNyWt7S7YZQSiURyEiMF7peAQPMeasreAExszgEM\nGH4zQrEfs7itW/w2Na+/CoA9fzCD5z6ALTOzNy5BIpFIJD1EWVkZf/zjH3nttdf4yle+EmvfsmUL\ndrudiy++GMMwuPzyy5k7dy52u53du3dTUlIS61tcXExZWRmmaXbpd0MIQU8k0m9Netid5IedYQlc\nyzOpRCcPmtF5tDlZduyiTYAaJF7Ae3sPMmPhK1TmTIYM68XAR/5MRlRVJc2sU9xuHNlZeHM9hG31\nAKwZ5+Er59yGOysbLSMTLT39hCul19P3u6+Qdvct0u6+p7dsP3ToIP/9379i3bq1eDwebr31a9xw\nw000NTXxy1/+nDVrPiMlJYVvfvNOrrpqdodjbNu2lf/+719RVraLwsIh/OAHP2L8+Am9and7Tqxv\nUkmPE/QdoHr338HUUe0ZDCi5FUV1sWLJjqMWt6ZpUvPGa9QvtsKdncNLGHz/f6KmpPTadUgkEonk\n2IlEIvzgBz/goYceIiMjI2FfZmYmZ511FnPmzKG2tpbvfOc7PP300zzwwAP4/f6EubkulwvDMAiF\nQjgcjiOeNzvb06NexowMT4+Mo0dLygBo0RBl1WYDwJXmISXTTTjUSHlFLu78YMKxlQXDODhqHCKc\nFmurdmTim30bk0qysWdlYs/Kwp6ViRq9R5sPbYf3fxPrXz7CxUXDTu2Ra+lNeup+9zXS7r5F2t33\n9KTtpmly++3f56yzzuL55+exZ88ebrnlFiZPPp0//elPZGSksXLlSrZt28Ydd9zBqaeO59RTE7+/\ngsEgDz74Xe6++26uv/56FixYwIMPfpelS5fi8bTZ2tv3XArck5iwv5rqnS9jGiEULYXckltRtRQ+\nfm8nm75IrHPbZXEbiXDwpT/S9MnHAHhOmcigu76F0oUHHIlEIpEcX5599lnGjBnDBRdckLRv3rx5\nsXW3281dd93FE088wQMPPIDT6SQYbBN4fr8fTdO6JG4Bamu9PebBzcjw0NDgPebMoaZpYhgmSjRE\nWUTnmIaiS8M00Y1oHVxnEfsONEGedaxbD+JTHayadh0Z5X7YVBUb9+36NE4fewq6EPgBvzeM4o+Q\nkeHhUH1dgg3/t/kdJqSPRxEnZp34nrzffYm0u2+RdncfXTdoaQoeueNhEALS0tw0NfmOWAc3Jc2B\nqh75O2bTpo0cPHiQb37zbpqbg2RnD2L+/Bex2x0sXbqUV1/9P7zeMAUFw7j00hm8+urrDBlSkjDG\nypWWPrjssqtobg5y8cWX8cILL7Jw4WIuvXR6j97zrKzDO9akwD1JCQfrObTrrxi6H0V1kltyK5o9\nk5XLdrHx8woARo7L65a4NYJBKuc/i3fDegDSpp5D3te+ccKFVEkkEomkYxYtWkR1dTWLFi0CoKWl\nhe9+13rb3tDQwL333ktKNBonGAzGBOzw4cMpKytj4sSJgBXmPGzYsC6f1zRNdL3nrsMwzGNOeqS3\nlslpDVGOPmy1hiIrisA0LKPHn1ZIpDwDglbd22n25fwrcin14Qjl5VbIsceu4g3p7Kxo5KP1lZwz\nYRDtCUUS59we9FWzpmojp+We2FUHeuJ+Hw+k3X2LtLtr6LrBK8+vprkxeZ5+b5Ca7uSmOycfUeRu\n2bKFoUOH8cwzT7FkyTu43R5uu+2bDB9egqpqDBw4OHafCguH8OGH7yfdt7KyMoqKhiW0DxlSxJ49\nexLaevueS2VyEhIONlG1/S/o4WaEYmPA8JuxOQew6oPdrP+sHICSsblcdOXoLsfA6y0tVDzzJIFd\nOwHIvOwKcq69Xia2kEgkkn7EO++8k7B98cUX85Of/IQLLriA6dOnY5om3/ve9zhw4ADz5s3jhhtu\nAOCqq67ihRde4Oyzz0bTNObPn8/VV199PC6hx2j1HoiYwI22xwSugmlaWZSFYmPQ0CzYZr0gHkAd\nZ6obWG1MJGSz+rtDOjagAXhp4Ra2fLCb7EwXqelO0jNc5A5Mo9JrZa1O0VIoTM1nS/12FuxcxISc\nsdgU+UgmkUiOH01Njaxdu4bTTz+TN998i61bS/ne977NY489mRSt43A4CXSQSK/9dJbO+vYm8tv0\nJEOP+Nmx5iUiwXoQKgOGzcHuHszq5WWs+3Q/AMNHD+CSmV0Xt+Gaaiqe+g2hSiusOef6OWTNuLzX\nrkEikUgkfYuiKMybN49HHnmEs88+G6fTyZw5c7jtttsAuPnmm6mpqeG6664jHA4za9YsvvGNbxxn\nq48NvTU8LipwRXTbjIYLKypgWh5coajocXGA6QMmM6l6FYdEFvWG9TCnZNkoqtdpMg3CQKk3SIk3\nRFV5U+y4ugH7oBiC3gjhtbkwfjs1gTp+9+9XGScm4XTacLo0nC4bDlfbutNlw+G0YXeo8sWyRNLP\nUVWFm+6cfEwhyqoqSE9309joO6IntKshyna7ndTUNL76Veu7fcKEiVx44cW88MJ8QqFEW4PBAC6X\nO2mM9tNZ2vq6jnj+nkQK3JMIQw9RvetvBL1VgCBn6LU4U4fx2UdlfLFyHwDFI3O4ZNYYlC5OhgqU\n7abi6SfRm5tAVRl42zdJm3pOL16FRCKRSPqKZcuWxdZLSkr405/+1GE/VVWZO3cuc+fO7SPLeh+j\nncBtC1GOJptS24SkECp61MOrANmF03GnD2fG/nfZbI4mCDSnODljSpDhNS7+79NGGgBRmE5JipPm\npgABX5i6aNkhYSo4/WlkHRpCXd4+dno2Ym5KwxlI7dRmIYgJXbtDw2ZXsds17A4Vm0PDblex2+PW\no300TUGztS1VTcFmU1A1tV9mkJVI+juqqpCeefSiz6pR7QGl50J9hwwpQtd1dF1HVa1s8rquM2LE\nKNavX0tVVRUDBw4EYN++vRQXFyeNUVRUzJtvvp7Qtm/fXqZNu6xHbOwqUuCeJJhGhJqy1wh6rRDk\nnKFX4c4Yzecf7+Hzj/cCMHRENtOuHtultzgALevWUvn8c5ihEIrLxaB77sMzdlyvXYNEIpFIJH1F\nzINLYpmgmMDV2mrgCqERifZXo4LQlTacIWPuZMDqj2jGRAhYE8ijIOUAY/ObKT2Qymf7G5g4cwyX\nTcwnKyuFV78I8OaOUjLSPcy68RTqmoby19qX8CotVI/bzNn1Mwj7DQL+MAF/hFAwkmCzaRLdlziX\n91hQVJEkgDVNQdUUNE3B6bShGwaKIlAUBUUVqKqCqratd7iM9lUUES0TJRBKu21BclsHfdq3C0Wg\nCAHCEv2tXm3p3ZZIjp4zzzwLp9PJH//4e77+9dvZsmUzy5d/wG9+8zuqqg4wf/5vefDBH1NWtosl\nSxbz2GNPJY1x+ulnEg6HeOONvzN79nW8885C6urqmDx5Sp9eixS4JwGmaVCz5/8INO8GoGDUVdhS\nT+WzFXv47KM9ABQNz2b67HFdFrf1y5ZS/crLYJpomVkM/s5cHAWFvXUJEolEIpH0KTEPbnTybasH\n14zOwdXUtr7xIcpqnIgSiopQU4Am8l0mzUA5+dhHZpLrK+dQg5MXF5YSbtnBrEvOIhINeXbY7RQM\nzaKALL5ZdxPPrPs9jWodvol7uHHUNXE2GgQDEQL+CMGosA0EIoRDEUJBnXBIJxSKEA7qhIIRQiE9\ntq+1PRJpE+od3gfdJKTrhII9mAXsOGL98wiEAoKORHCiIBaCaB/Rdqx1GETbRExMi2h73Hp0O3pk\nwnayXW07k7S4sI7XbCqRiE5i7+SV5HMnnajD87R/CZBsR8fjd4YAbHaNcChCV3yJvfcaonsjCxFn\n9+EM7wVje2JIIQQ2u0o4pGO2Gt8DA18387ssXvZX/v7KKzgcTi457ybKtwnOGHsti5b+mZlXzsBu\nd3D+2deyfyvs37qJjVs+4ePVb3H3bb8C4JrLvs0br/6ZZ3/3WzIzcpl92b18uGgXAKqmcO4lI0hJ\ntx+7sZ0gBW4/xzRN6vb9G3/jVgAyBl1AXtF5LF1YyqcflgEwZFgWM67pmrg1DYPq11+lYcliAByF\nQxj8nbloGZm9dxESiUQikfQx7T24IlYeKNmDi9A6FLgAkehxp+Tlkl+cxYK9BwnhglOG4VpzAL9X\n8NIHYRpq/og/1yoTpBhhTCOCUDRGZ41getFFvLv3fT6qWMnglEGcN/hsq5+i4HLbcbmP/mHQMEz0\niEEkohMJxy8NImE9ttQjRtJ+0zSx2zS83iCRiIGhG+i6aS2N6FI3MHQzcWlYSz1iYpompmFiRJdH\nKmlyrFjjm9Hp0/0vo69EcnzRmDLh6zChraVsRw0Ap42aw2mjkttTtJHMmPrd2Da4uOD0u2P9wi3E\n7bOiYKbNHttbFwBIgduvMU2Thop38dZZZXtSB5xFRv4FrHhvB58ss96UFAzNZMZXxqFqRxa3utdL\n5e/n4du0EQD3+FPIv/seFGffTgyXSCQSiaS3OeIc3LifTSHaPLhaksA1ov0VJuWkMTTVxRtlBylr\n9pN6Wj6RtdWEW8Is2DSSYRM+AxcYwRrKN/wPjtShOFOKmZ43nvLmCkrrtvPa9n+S5cxgXPboHrlO\nRREodhWbXT1y53ZY8/xSqKtr6bF5fqbZKnqtf4PWesSt62bcumEQFcVmQt/WPtZ4AJZwtj4mioCU\nVBfNTX503Yjta+vXJrRj69GliQkd9CdufLDqJFsDJF9f8jV33BBrbv1TFOBy2fH5Q9Z1t9vftpl4\n7uR+8femIzs6P779+EdCKAKn00YgEMY8TnVwj+bFiVDA6bARCIYxOwx06Plr6akRFSFwOGwEg2Hr\nb7EXbntv/EtqqmDKBSVH7nis5+n1M0h6jaaq5TRXfwqAJ+tU0vOn8dlHe1gdDUsuGJrJ5deOR9OO\n/KMWqjxAxW+fInzwIADpF11C7o03I9Tu/yBKJBKJRHKi0ypYRSyLsvWEa3lwTVQ1bg6uEufBVToW\nuLboi+RMh43bRw1mXW0zi/bXwGm51K+vJtwYYl9DKjZXLcIUmGaEQNNOAk1W+b3pqBzSHNREgvx+\n40t8a8LXGNlDIvdEwgoFFqBAbz1h9IYw7wuk3X1Lf7Ub+q/t8Xb3JlLg9lOaDq2isepDAFwZY8gs\nvJLPPtoTy5ZcNDyL6deM65K4bdmwjqrfz8fw+0FVyb3lq2Scf2Fvmi+RSCQSyXGlzYMbzWwc8+Ba\nqGrbQ6MQKhHDSux0uBDlxKzLgkk5aYzO8PD+gTpWqgq1W2rRo/N9yxtzaBl0Ibm2PQS9+9DDzTjQ\nud6j8nKzoMmI8Nz6F7k5K5fi9KHY3YOwuQZidw9C1ZJLc0gkEomkjS4L3EAgkFS4V3J8aKldR0PF\nuwA4U4eRPWQ2qz7Yw/rVVp3bUePyuHjm6A4yByRiGgZ1i96idsE/wDRRU1PJ/9b9uEaM7PVrkEgk\nEonkeJJUBzdhDq6JEidwEUpciHLiOK0eXK2D8nsuTeWKIQM4Lz+Tj4ZksWjtOkwgHIL//WcTp585\nnlumzsKDl0DLPjzefdxi282faw/gM03+VneIa0PNDLGVxsZUtBRszhxszgFxywEomltmEZZIJBK6\nIXBnzZrF008/zZgxY3rTHskR8DVsoW7fvwGwewrIHno9nyzbw8Y1FQAMHz2A624744iFnyNNTVT9\nYT6+0s0AOIYUkX/vt7FlZ/f+RUgkEolEcpxpPwe3LUTZalZEogf3SCHKWnvlG0eGw8Ztpwylojad\ndYcAoWIaJp9/WsGmXbWceXYB5w8ZwbDCU8geIvjPxr38dsMfaQr7eN0b4NqMHIbit+yLtBBsaSHY\nsifhHEJ1otkz0RwZaPaMuPVMNHsGQpFTjiQSyZeDLgvccDh8zG8GS0tL+elPf8rOnTspKiri5z//\nOaeeempSv88//5xf/vKX7Nmzh4KCAn70ox8xZUrf1k86EfE1bqNmz5uAic2VR07xjXy4uIxtG6sA\nKBmby/SrxxwxW7Jv21Yqn5+H3tgAQNq555F7060oDkdvX4JEIpFIJCcESR7c1hDlaHNCCgqhENWx\nySHK0QM68uC2R4uGQ08qyaXcn0ZFeROBmgAr3t7F+pJaCodnMDk3g4lZg/nu6ffz9LrnqQvUkRMC\nqgAAIABJREFU83pDLdeXXMnkzCGEAzWEA9VEAtWEAzXo4WYATD1A2F9J2F/Z4bkVzYNqS0W1paJF\nl4kfD4rqlkJYIpH0e7oscGfOnMk3vvENrrzySgoLC5PClefMmdPp8cFgkLvvvpu7776b66+/ngUL\nFnDPPfewdOlSPB5PrN/Bgwe55557eOSRR5g+fToLFy7k/vvvZ8WKFV/qEGl/43Zqyl4H00BzZJNV\ndCNLFuxiz85aAEaNz+PCK0ajdPIDa+q6FZL8r3+CaSLsdvJuvY20qef01WVIJBKJRHJCkDAH1zQR\nZqLAFUpckinRcR1cIFZnVutCKb7WOripDgcP33I6H26o5NX3dhAM6TRtq2d7lZeqUT7eSbVTnOri\n0qG38uG+VznoO8SrO/7NocJz+UrJTBTRdi5DDxAO1BAJ1hEJ1hMJNUSX9THxC2BEvBgRL2F/Vac2\nCsWBqrlRNDeqzU2TJ42wbkdR3SiqE6E6URR7dN2BojpRVAdCscsQaYlEckLQZYG7aNEiXC4Xy5Yt\nS9onhDiiwF21ahWKonDzzTcDcN111/HnP/+ZDz/8kCuuuCLWb8GCBUydOpUZM2YAlrAuLi7uVLid\n7Pgbd1AdE7dZZBTexNtv7qayvBGAiZMLmHLR8E5/WEJVlVS9+HsCu3cDYM8fzKC778WRn98n1yCR\nSCQSyYlEqwdXCJO4hMkYrZ7ahDJBCpHWObhxIcqmacbG6SxEue2cenQMDSEEF07MZ1JJDi8v2c7n\nWw8RbgxRu/ogrnwPO4als7tZRaiXk+b4gKbgXt7fv4KD3mpuG3cjKTbLOaCoThyeAhyegqTzmUbE\nEryhBvRwc+InFF1GWtodEyQSCkKoHgB/4xEvq+0+qQ4UpVXw2hCKHUWxRdfbPodtExooKkK0fuK3\nNYRQ47a/vM+FEomkc7oscDsStt2hrKyM4cOHJ7QVFxezOyq4Wtm8eTN5eXnce++9fP755wwdOpSH\nHnoIu71rRc6FEPSEFlaiP2CKcnzfRvoad1FT9hqYOpojk/SCm1j4+m5qDlk/SFMvHsaks4fExG17\nu03DoP69pRx643XMUAiAjIsuIm/OTSdUSPKJcr+7i7S7b5F29z391fb+arek74ifg6vE1e6MzcFV\nTCulsrBCdluFbLwHV487rishyhEjEh2jrW+6x863Zo9n/c4a/r5sJwfrfPgPeAkd8uMuTMFVmIqw\nXYrdWEkoXEpp3TZ+tvIJvjJiDpPzRiQI7vYIRYsmoso5bB/TNNDDLRgRH4buQ4/4MSJe9IgPU/ej\nqSH83ib0sBdDD2DoQUwj2PFYehBdD6KHj3gregARFbxanCBWQQiEUKjSNHQdq9gpVlv7dUF0GW2P\nrcf2RdsQsXO2JvAUsXXRaXub86FdOyQdrygCw+fE6wtFX7Qk/9smOzMOsy0Ot5+464kN2rUxO9gl\nsOxWdRe+Zn/sBdHhx+7Ypt4i6VrjUFRBk+nE3xzAOJZSO312OW0nUhRBk+nC3+xv+y7rATaXbuN3\n816kvLySrKwMvnrL9Vxy0Xk0N7fw+G+eZe36TXg8br52y/VcPuOSDsfYsbOMJ5+Zz9695QwePJDv\n3HcnY8dYCWxVzYaZ0fvJbLtVJqi2tpbXX3+dPXv28P3vf59PP/2UESNGMGLEiCMe6/P5cLlcCW1O\np5NAIJDQ1tjYyPLly3nmmWd48sknee2117jzzjtZvHgx6enpRzxPdranR0NkMjI8R+7USzTVbufQ\nrr9jmjp2VxbZQ27l9b9so7HejxAw8/qJTDprSIfHZmR48JVXsOu5+TRtshJJ2bOyKLn/W2SeNqkv\nL6NbHM/7fSxIu/sWaXff019t7692S3qf1pBjhIkS93zY+oAuogK31VPYUYhyONL2NN+dEGVNSX78\nmliSw7jiLJatKWfBx3vwByM0lzURrPAyuCQTW+5UfEo2/uAn+CPNvLzlBf5RdhajsyZTku5mWKqb\nfLcjKQnWkRBCQbOngT0tad/ham2apolphNoEb3RpGMG2bSOM2e7TeVsI6M6DuolpRkCPdHhUn2js\nXqD2eBtwlBw83gYcJZ0H7J/Y9LTthmHyk/+3mG/ccApnTRrP1p21/PLxZxjoLuXlf27GblN59pGL\n2VfRxP889wJpyiZGFGcljBEK6zz08HtcPX0ED31rGitWl/Pjn/6cJ392KU6H9b0XajqDtPwre9j6\nRLoscEtLS/na175GSUkJmzZt4t577+Xjjz/mhz/8IfPmzTtiEiiXy5UkZgOBAG53Yj03u93O+eef\nz7nnngvALbfcwgsvvMAXX3zBRRdddEQ7a2u9PebBzcjw0NDg7dE3I13FW7+VQ7vfsDy39gxCtln8\ned5GwiEdVVOYMXssRSOykgolK4og1aWx6+VXqVm4EDNivS1OO3sKA2/9KqbH0+vFlY+G432/jxZp\nd98i7e57+qvtPWl3VlZKD1klOZGIn4Or6sntsSzKrR7cDgRuvAc3vg7u4WgNUVZFx4mcNFVh+uQh\nTBk/kLdX7WPZF+WEQjplpTW4d2mcMrYEbVAh6+sWEjEa8QVWsf7gHrY3XoCqpGFTBPluB4UeJwUp\nTgo9TjLsWo/PjRVCROff9mwkmGkamKYOho5p6phmJG47Em2L3zaspaGDqWOaBmAgMHC5bfi8AQxD\nB9OM7TNNA0wDExNa100DSzC37ktsxzRpFd9mbN2M20d0vMP0a32ZgtnueCzb4sZThMAwWs+ddIfa\n37AO95vtthNXOz7mcGOaHb4+MBMWYFqe2rjrlPQ/vP4wTS0hdMP6OxUKaJqCogg+31DF//7kYuw2\nlZKhmUw9o4CPVpcnCdzS7TUIIZh2XjEAF04Zwtvv72Ld5oOcfdpgQOD05Pb6tXRZ4P7617/mtttu\n4/7772fSJMsD+Mtf/pLMzEwef/xx3nzzzU6PHzZsGH/9618T2srKypg5c2ZCW3FxMfv27Utos/6j\nd+0/i2maVkhKD2EYZqfldnoDb90GavcuAEw0RxZ1vktZ8VYZpgkuj43Lr51AXn5ah3a1bNzIrr/9\nlUCV9V5Hy8wi9+ZbSJl0OkCfX0t3OR73uyeQdvct0u6+p7/a3l/tlvQ+8XNwE0KUo7pCiSaZavXg\ndjQHt7VEEICtCx5cPebB7TxTcarbzg0XlzBjciFvf7qP99dW4AtGWLW2EmWdYNLoKwgP2sCOls3o\nehVe75s4HJMxbWPZ2xJgb0sg5lLzaCqD3HbyXA4GuuzkuR3kOu3Yu2BvXyNaQ4QV2zGNczjP84nO\nl8Hurj7P9wydn+t43m/T0NHD3Zjg3g5FgYx0Dw2N3uSw8HaotrSuZUc34ZrZHn77pzd57i/rMAyD\nBx98CHf+SDRtOWdc8qtY17E7X2f58vcpnPhQwhCfbHuF4SNEQvuwkc20iBIKJ96Oqgqyc9J73dnW\nZYG7efNmHnnkkaT2OXPmJAnXjpgyZQqhUIi//OUv3HjjjSxYsICampqYp7aVq6++mjlz5vDBBx9w\n/vnn8/LLLxMMBjnrrLO6amq/prn6c+rLFwGgOQawu2IqG9dYYjV7gIfLr5tAanpyNulgRTnVr7+K\nb9NGq0FRyLxkGtlXX4PyJc4+LZFIJBJJR8QeaIVJa8JkkzbhqyitArj9HNy2MSJxIcpd8eBGjuDB\nbU96ioMbLxnBZWcNYdkX5Xyw9gAt/jBrttTDlkLyitIJDlxL0PQTCH5CqrKHkuyLaY5kUukLEjFN\nvBGdnU1+djb5Y+MKIMthI8dpI9tpJ9thI9tpI9thI8NhS8oULZH0FH2babvzc4nofG3r03cC1zR0\nKrc+hx5qOKZxKrrYT7VnkD/m3iOKXMM0cLrc/OIXj3LuuRfw2Wef8vOfP8Sjjz6Bw+FION7pchEI\nBpPGDAQCOJ2uxL5OF8FgCKGoiD7Ki9FlgZuens6BAwcoKipKaN+8eTNZWVmHOaoNu93O73//e372\ns5/xxBNPUFRUxHPPPYfb7eanP/0pAA8//DBjx47lueee4/HHH2fu3LkUFxczb968hFJCJyOmadJU\ntZzGqg8BUB2DWP3FeCrLrSyGRcOzufSqMdgdif9kkcYGahf8g8aPlsdCQ1JHjSTn5luxDe54fq5E\nIpFIJF92dKPVQ9vmwTXjHoiFiAZndhKiHIlPMtUND67azVqzGSkOvnL+cK6cMpSVm6tY8tl+Kmt9\nHNybBhVTcAzdgpJVSbX/ADXlLzM1/0xuGn8ZLbpGhTfIQX+QKn+IKl+QgG5gArXBMLXBMDT6Es6l\nCMiw20i3a2Q6NAamu7HrJmk2lfRou0tVZEkgieQk48MPl1Fauol77/0OAFOnnsvUqefx4ovPEwol\nJpYLBgO4XO6kMZxOJ8FgR31dSX17ky4L3Jtuuomf/vSnPPDAAwBs27YtlgzqG9/4RpfGGD16NH//\n+9+T2h9++OGE7XPPPTfJs3syYxo6dfvfwlu33mpQB/PesuF4W6w/kElThjD5vOKEbKCRxkbqF79N\nwwfLYtmRbTkDyL3+eopmXEx9vbdfhbdIJBKJ5MjU19fz/PPPs2nTJsLh5DQ6Hf3GSjpGj5uDG/Pg\nxmU3bhW4nSWZig9R7lKSqWgWZa2LHtz2OGwqF546mAsm5rN9fwMrNlTy2bZDBHdOREkbjK1oC4rL\ny8cHVrO6ch0XF57P9KHn49SsJJ2madIUjlDlC3HIH6I2GLKEbiBMY8hK1mSYUBcMUxcMU9YM1DQn\n2aEK8GgaKTYVj6bGlh6bSkp06VJVnJqCU1VxqQo2RUhRLJEAQlHJH3MvkWMIUVYVQUaGm4YGX0Iu\ngI7QbOldClE+eLCKUFRTxM6jqowcOZoNG9ZRVVXFwIEDAdi3by/FxcVJYxQVFfPmm68ntO3bt5dp\n0y474vl7ki4L3DvvvBOPx8Ojjz6K3+/nvvvuIycnh7vvvpuvf/3rvWjiyY0RCVBd9hrBlj0A+MND\n+eDdAgwT7A6VS2aOYeiItvT+kcYG6t55m8YP348JW8XtJuvKWWRcfCk2pyy0LpFIJCcr//Vf/8XG\njRu56qqrSEmRya+OhYQQ5ah4NeKEZ2vIYmuIciTaXT3MHFytO0mmuunBbY8QglFDMhk1JJObp43k\ns62H+GRjJTs2ZaHm7UUbvJOwGmLxvqUs2fMRY11ncuXI8ynMSY96YW2MapdhPGwYlrANWOK2MRSh\nKRzBa5jU+II0hyK0Xq1uQlPY2t9VFEFM7DpVJSp+FexK9KMKbHHrdsUSxbFltK8mBKoi0IRAUwSq\nEKiir0NfJZJjQygqNseRI2APh6oKHO4UbIEWlB5yaJ155tnMn/87Fi78F1dcMYt1675g+fIPePrp\n56iqOsD8+b/lwQd/TFnZLpYsWcxjjz2VNMbpp59JOBzijTf+zuzZ1/HOOwupq6tj8uTOkxH3NF0W\nuAcOHOCmm27illtuwefzoes6qamp6LrOxo0bmTBhQm/aeVISDtRSXfYqkUANABUHh7NuXT4gyMlN\nYfo140jPtFz6oapK6pe8S9MnKzCjb+0Vl4uMS6eTeel01JM8hFsikUgksGrVKl566SUmTpx4vE3p\n9+ixekAGaoce3Fi9oGj/aJKpBA/u0YUoa6JbVRo7xeXQOH9iPudPzKehJciabaNZvX0Me8y1qLn7\nMJQgm4Ir2PjFauwNwxibOokxBXmMLMwgN8MVE4Y2RSHP5SDP1ZYZOT4JTyhi0BKO0BiK0BLW8UZ0\nWsJ6dD2CN6LjjW3rSel9DBN8ER1fpAczgcbRXvjaNRVhmlZ7dJ8qQEGgCFBEdIloW09oa93uqM3a\nbv1TEETncxItnYtVf7Vt3doRq3orovshbgyBpgpSwxFaWgKYBknjdVKONnm7neAXh+lHnA1tfTs+\nNqnEbhRVFbSoCo3eQFuZrSPa14EhfYyqCIJ2lQZ/EKN3/ix7DVUVBFsCNPpDRxWx2dHtT8sfwvd/\n8kv+9qff8+RTj5OTm8f93/8x2UUl/Me3H2TeU//D7Gsux+l089U77iV32Eiq/SE+fG8xb77yEk//\n4WUAHvrl/zLvqf9h3vxnGZQ/mP/6+X/TgkqLP4RdU8jsg0RjXf6GveSSS/j444/JyspKKO2zd+9e\nbr31VtavX98rBp6s+Bq2Ubv3n5hGENMUbN4ykr378wCYcMZgzr5gGKqm4NtSSv2SxXg3tN1fxe0m\nc9oMMi65FNUtha1EIpF8WcjOzsbh6NnSLF9W2soEtSWZMmgTqYponaPbSZmgboco94wH93BkpDi4\n5PQCLjm9gCbvGXy6s4zlVcup03YibCHCA7ayTt/Bmk2DiSwbQqqaxYiCdEoGpzMkL5WivBTczo4z\nGKtCxLy/R8I0TUKGiT+iE9AN/LpBUNfxR4zYdiC6L2QYhA2TkGEQ0k3ChkHIMAnF7evK43DENIno\nJrHZf6Gue5clEkmU1EKG3P8wrVl8VgIrN+0FQLvmDk675g4ANgIbo+3kjab4P3/Fb1q3sZF3x0Pk\nRbcWGbAotg8ubchl2sCMXr2MTgXuK6+8wu9+9zvA+rKaNWtW0hshr9fLiBEjes/CkwzTNGis/ICm\ngysACEfsfP7FaOrqM/Ck2LnoytEMHuSiaeVHNCx7j1D5/tixWlYWGRdfSvr5F6K6kyd2SyQSieTk\n5r777uMXv/gFDz30EEVFRdhsiWLDbrcfJ8v6H5EEgRtNMhXnwaWdB7e1TFB8iHJY714W5ViSqaOc\ng9sd0jx2pk0cxbSJo6jx1fGv7ctYV/cFuhpBG7gPbeA+Ak2ZrKsuZM32PDAtmwZkOKNiN5XCvBTG\nDAe7MDhSRtp4hBA4VIGjB0oRmaZJxDQJ6ZYI1k2TiGG16a3Ldm2GAIfTTqM3QDhitLUDhmlan9h6\nuyXt2tpvxx1r0prfs23djG7F1uP2G2Zbpdr4/Wbc8QjrmtvGkkhOLvqiQlmnAve6667D5XJhGAY/\n+tGPuOuuu0hNTY3tF0Lgdrs5++yze93Qk4FIqJHavf8k2GK9xWhoSGXNurEEgg6Gjx7A5DEOgh8t\nYNenn2IGA7HjHEOLyZw+g9TTzkBoPRfWJJFIJJL+xRNPPEFDQwPXXntth/u3bNnSxxb1X6ywPjM6\nB9dqi5+Dq7Sbg9tWJig5RFlVrFDWI9HqwT1SHdyeJsedxTdPvQ5v+Ao+qljJ8vKVNIaaUNPqUdPq\nYaiNSG0eeu0gqhuyqG4IsGZbdex4VRHkZbkZlOVmYLabvEw3OelOctKdZKY5UJXee2IVQmATApsC\nHrp23062erJmVFS3NSQsaJPNybSPBm3fJ8E/njRux9vx45qYqKogI8NDQ4M3IWz/yDYc338bRWmz\nuz/9nQAocffc6Kbtx/NK7ZrC0IEZx7cOrs1mY/bs2QAUFBRw2mmnoUmBdVR46zdRt28hpmEFz+zd\nN4jSrcNxeZycXxQgZcNrHHqrzX2PqpIy6XQyL5mGs6REJk+QSCQSCU888cTxNuGkQTcMiIrYmAc3\nQai1hignZlHWOghR7kp4smmaferB7QiPzc1lQy9h2pALKa3bxscHPmVTzVZMNYyWW46WW45DuEkJ\nFhGozqGu0o1hCHTD5ECNlwM13qQxFSHITLWTne6yBG+qg4wUB+keO2keO+kpdtI9dpx2+fx4tAgh\nEqV90iPh8X1GVFVBmsNGxKahK/1HKKqqIMtlh6Ocx3o8abVd9DPbuxLp0hN0+m3zxBNPcM899+By\nuVixYgUrVqw4bN/vfve7PW7cyYAe9lJfsRhf/SYAQmGNjZtHcPDgAIY56ijcshR1nS82Z8SWN5D0\n8y8gbeo5aKlpx89wiUQikZxwTJ48+XibcNKgG2YsDDlWJkhtC/luzaKcVAdXSfbgdimDshk3X7eP\nPbjtURWVCTljmZAzlvpAA58dXMuag+spbzlA0PQRtG+BwZA+xMHIrJFk6AXYfHnU1UFlrZfqhgAt\nfivhpWGa1DYFqW0Ksn3/4c/psKmkp9hJddvwOG14nJq1dNlwOzVSnDY8LqvN5dBw2lWcdhWHXe1V\nD7FEIjn56FTgrl27lnA4jMvlYu3atYftJ72LyZimibduHfXlSzANK9y4uiaD9ZtG4W5u4syqf5Ea\nqgNAaBopp59J+vkX4Bo5St5PiUQikcS48cYbef7550lLS2POnDmd/kbIOrhdR9fNmAdXjXpwDTX+\nsSjRgxuJhSi39WgtE6R2owauNcaJ483MdGYwvegiphddRJX3EGsOrWftoQ1Ueg8S0INsqN6IlVIG\nBubkUjJiGJdnDKPQPYRIwE5NY4DapgA1jdanoTlIozdIY0uIUKRN1AfDOofq/Ryq93fbRk1VEgSv\n067itKk47Bp2m4JNVbBpClp06bCppKc5CYciKIqI7Y99VAVFEaiKYoWXK9Esy9GPoghUVYmFnqtq\nW3tXQtElEsnxpdNv2L/85S8drks6J+SronrvIvRAOQDhiMq27cUc2uVmVM0Kcnz7EYBz2HDSppxD\n6pmTUWU9Q4lEIpF0wLnnnhtLJnXeeecdZ2tOHiwPbmuIstWW4MFtnfXYSRblSCxEuSvzb+ME7gnq\nkRzoyeXK4mlcWTyNWn8dpfXb2Na4g00HtxI2IlT5DlHlO8SKilUA5LiyKUotYEhOAacWF1CYWoJL\ncwLWi/5ASKfRG6KxJRhdhmjxh/EGwngDEbz+6DIQxusP4wtEOpwfGNENWvxGzGt8PBGCqBBWUJS4\nckDR8kFCHH47qS12bGt/gd2mWqHvAhTa+ibaIBLsgfhSPqJtO64EUXzfWFeSnVTxmyJ+rITxEs8j\nBDgcNkKhcGy+bWvZpD7jKE4mhMBh1wiGIpjdKF3Tt684Oj6bUGiz3eiwS/fO0kcXZdMUZl80gix3\n777k63T0zkKS4xFCcM455/SIQf2ZSKiJ6j1LCLVsjv2hVFblsGNTPoOqtnJ243bMDA/NZ0yiefxQ\nglkpOFQ7zqYtZATTyXUPINOZjiJOzB8+iUQikfQ99913X2x95syZDB069PgZcxJhzcGNemBbMyqr\n8Vmp25cJslo7qoPbnRJBcGJ5cA9HtiuLC1Om8pWJ06mqrmNH3R52NuxmR8Nu9jTtJ2JEqPHXUuOv\nZc2htlKGue4c8j0DGejJY5A7l4GePIoHD8CuHrm8kGGa+IMR/MEIwZBOIKwTCOkEo59AKEIg3Lpu\nfcIR3cqUrJux9bBupU4KBCNxbQbhiBl7KXG0mKb17x7R+1nhVInkBCEQNvjW7HG9eo5Ov2Fvv/32\nLg0ihPhSZ26MhFuo3PkBum8dimIgBHi9TraXFuDZtp/xvrfYPURj1ZlpHBhgA1EBDRXQkDyWS3Mx\nKnM4Y7NHcVruxNibUIlEIpFILr/8csaOHcusWbO44ooryM3NPd4m9Vt0w4zNs1VbPbiaJcIURRAf\nomxGS9FA+zm4XU8yFe/BPd5zcLuLXbUzOmsEo7OsspBhPcyepn2UNe1jX3MF+5rKqQ1Y064O+Wo4\n5KuB6k2x4wWCbGcmOa5ssl1Z5DizyHJlku3MIseVRYrNE/NgWvNzjyyGO6OzLMqGaaLrhlU2yLDK\nCxmGia5b/8bWenS/GW03zFh/a92IrbeOaZpt5X3ab5vREkNJ2yRuC8DptOH1hdANM9qemNo4djVx\nWYyTshTH7TvcsWb7zMtJfdr2m+06tTMJgeXBDQTDlifUpFcz9XbD2dopyV7Q3rG6N0aNeZ+D4R67\nH/H01r+fpgq+cvHIXho97jyd7dyyZYucD9oJoUAj+za8jarsRFUNFAVCIY2yHYNgw0Hszo/YPM7G\nroJMdM26j6n2FFyaE5tiQxUKQT2EL+KnOWSly/ZH/Kyr3sS66k28sf1fTB50OlcMvZR0h0w4JZFI\nJF92li5dyttvv81bb73FY489xmmnncasWbOYMWMG6enpx9u8fkV8iLIaFUFGnMA1oxmPESpG3NNe\nhyHKypGflfQED27/Erjtsak2RmQOZ0Tm8FhbS9jL/qYK9jdXUOk7SJX3IFXeQ4SMMCYmNYE6agJ1\nUN/BeIpGmj2NNHsq6Y5U0uxpsWWaPYVUewpuzY3H5sKpOY8p0k0RAkVTOTYJ3TucbOWNTnT6q93Q\nf22Pt7s36VTgTp06lTPPPJNzzjmHqVOnUlhY2KvG9Beqdm+icucHZGbXY7dZf1ThiErl7kyattRS\nMeALtk7OpMUYhelLxdiZihl0Y4acDBqUwVlj8jj3lEG4HG23P6iHOOSrpqxxL1vqdlBau5WQEWZF\nxSpWV67hymHTubjwPBm+LJFIJF9iBg8ezO23387tt9/O/v37Wbx4MW+++SaPPPII55xzDs8999zx\nNrHfYCWZag1Rttpa5+CqmkLrxDYhFCJxLpKOQpS7lmSqTeD2Nw9uV0ixeRiTPZIx2W3eGcM0qA80\nUuWzxG5toJ5afx21gTpq/XWEDGtObdiIWG1RL3BnCARuzYXb5sJtc+PR3LhtLhyqA4dqx6E6cNkc\nZNWloQdNbKKt3ak5sCkamqJhU2yxdflsJZEkUlq6iR/+8AEWLHgHgKamJn7964f54ovP8HhS+OY3\n72DmTKuUrGmazJ//O956659EIjqXXXYl998/F1VN/p5ramriJz95kJUrVyaN05N0KnAXL17MqlWr\n+Pjjj/nDH/6AYRicc845TJkyhSlTppCRkdHjBp2oHKrcz8YV/yI320t6ZoDsAVZ7KKRRuSuF/Qca\n2JzaQvWQEYjIVESd0wq30U3shoEvaH2J7z7QxO4DTSxYUcbs84q5+LQCFEXgUO0Upg6mMHUw5xdM\npSXk5ZPK1SzZ+wG+iJ9/7FzIltrt/Mf4W3HbXMfxTkgkEonkRCAzM5MBAwaQl5fH9u3bOXTo0PE2\nqV8RXwc35sFVrMciVVMSPLiROBeu1kGIsk3rbhblk0/gdoQiFLJdmWS7MhmXPTphn2matIS91Pjr\naAg20hhqoinYbC1DzbH1lpC3LdQWK7TWG/HhjfjAX9tjdtqioleLCWAtJoBVoaIIBUWbyTXUAAAg\nAElEQVQoqEJFja5bH6uMkRpdt/oo7Y5REEKJJXWykkslL1VFkFLnxOcLgSFAgECxnidjiaKsdSvC\nUiRvR6+powjMjtI+tfWLS1zVvn984ql2YwgEiipICztpbg5gGGaHYxFn/+HsStzV+xGkqgI1hpum\nJh/dnZp9vONbFUVQY7hoavJH73k8R2+daZosX7KMv/3hjyiqSlnjPgCe/tX/YLc7ePqvL7C/bC+P\n/79f4BjgoWT0KJb8exEfrnifXzzzOCB44ue/4tk//ZaZ112TNP5vf/04uVnZLFy4hO3bt/PAA99h\n6NDhjB8/4aht7ohOBW5aWhrTp09n+vTpAJSXl/PJJ5/wzjvv8PDDDzNo0CCmTp0a+5xMvP/eYuoO\n7MYtAuRkRsgc4GdESdsfkLfFSeUelXXNGrV5RQyeNJxxKTqqCl4M6kw7dbS9AMgyTMapDpqrvKze\nchBfMMLflu7gs62HuPeaCaR57AnnT7F7mF50Eefmn8UbO/7Np1Vr2Fq/g9988Rz3nXoH6Y7UPrsX\nEolEIjkxaGlpYenSpbzzzjt88skn5OfnM3PmTObOnUtxcfHxNq9fEV8HtzXJVMyDq8Z5cFGIJNSw\nbXt4DIWtdnuXBG5ciPJJ6MHtLkIIUqPhx51hmAa+iB9f2Ic37McX8eEN+/CF/Xgjvlh7SA8S1EME\n9SBBI8T/Z+/Ow6Mqz8aPf88ye/aFBEhIQlBZXRAVXHCpiCJC9UVttUpbFVFb++K+VIuKfa2KFrUW\ntVqrUrH+aosCRVELbrigouxrIMQQsm+zn+X3x5nMTMgKJAPR53NdrcyZk3OeM+HizH3u+7mfsBHC\nFw4Q1EOtHi50dA7rZ0M9eYmC0OfsXbmT+nWVpJ/Sn8qPSnn0y6fQgxrrVn3G0BvH8sTaZwFwDE/j\nqYVPkXfBUWx9azUZJwzgLyV/B8A43sW/F/+TTUXftTq2HtRY98kqfvnor3A4HAwfPpIJEyaybNmS\nxAa4+8rLy+OSSy7hkksuwTRN1q9fzyeffMKzzz77vQpw//Xa8xxbXE7hsNZPRHRDomavm5IKG2Vy\nGjmBBvKbGznyu/VgptKclYMrS6c4dxf9suvwmU42mcWsZRR+JL4xQ4wekcH/nT6YN1ZsZ9X6vWwt\na+CBv33BrZeNpl9a28ys2+bmyuGXUpxWyMLN/6LcW8Gfv32B/z1uJk7VkaiPRBAEQTgMjBs3jvT0\ndM477zxuuOEGRo3q2S8FPyS6biLJrUuUDVkFM5LBjQRFkqy2yuDa4spZw/uRwY2fg6v+QDK4PUGW\nZJJsHpJsnm7/zL7zE3VDtwJfPUTYCBM2NDRDi/tvbFvL9rARRjN0NCOMbhropo5hGhimgW4aGEY7\n2+L2s15H/mwYmBhWkylaGkOZGJhgmtHXJiayLKHputWoKtrUycCI2weTuNdGpBFUbP/4rkNm9L9t\n52m2tzROe/sJvUFGlrr/d/pgGKaXlqZ5XckY3Z9+4wvw7ox1wg3W+pFkCUdGLE5xZrlp2GBVDQWq\nvTizY9fiyHITrPZhmmarjH3LcYoLB0e3DRpUwMqV/z3QS+tQpwFuSUlJpz/s8XiYMGECEyZM6NFB\nHXKSgpXeNwkGVRpqHVTWqTQ6cphw9nmMSbMaefiDGqs2lPOvtR+RrX7N6WvWYO4u4KusE0nOCHDs\n0ZsZ7V7PcHMrn7insMVn46vqJkwTrpo8nGOGZPH8ko3UNAaZu/Br7vzZ8aQltR+0njLgJFyqixfW\nLWB303e8tPE1rhl5hWgCJgiC8APy7LPPMnbsWPFvfw/Q9LhlgiIlyqasgm4FSKYZC3DDHZQoh8NW\n0NqdADekx9ZwVeTDf5mg7xNFVnDLbtw296EeSqcOx8ZBptm6PLy99xRFIj0jibra5lbLMLXXkZl9\n3o0G4K2C7cRcu6JIpKcnUVe3f5/3wY5ON0ye3riH+lBilppKsytcPyy3VQf4rnyT8jX3//MeHh1/\nH+u+/ZZ7XZt4dPx90feXNS9hyY43eXT8fUycfQa3jv0Vg4uHALCnvJwr/3Qp/zf2buyOWFzTcpxp\nIyZFm0w5HE4CgUAPXWlMp//CnnfeeUiS1OHixy3vfd+WCbrwkp+zdetGdMlk6JgRHf6ldzlUzjpu\nEMcWX8T//SOH10/9gHPX7OaEskq+Ms/lo1XHcdKYDaSl1nNm8A3S03/KZ3UGX9c0keNyMH5YDslu\nO4//4xuq6gPM//c6br3suA4XgB/d72jqj2jgn1vf4puqdXyy53NOGXBSb34UgiAIwiH22muvtXpd\nWlra4b6XXnppbw/neyOsGbBvBleJBLjxGVxJbd1kKj7A3Y8MbkuAKyGJDK7QZ8Q/TGszhzfyUpEl\nVFmxSu/NvtOwS1EkXDYnflVDlxL3QEEzzHbnQ/cWCQmn6upWt/cWdsWOJFnLl6Z4UgmFQrjUWAbX\nCOt43Em4VBdOpxM0Kfa+ZqIoCqme1r2aWo4TLxgM4HL1/IOnTgPc9957r8dP2FcMHTq8222sM1Kc\nzPrxWGb/PcCyEz9k2ooajit/hy/yJ/PZ6uGcMf5bHLZmjg8swZ8xjW9rvbxTVk1xiothBelcPXkY\n8xetZ0tZA0s/LeWCkws7PNeZeaeypW47a6s38MbWJRydNaLL+SuCIAhC3/XMM890az9JkkSAux+s\nADeSgTWsL35GJPBUFRkz0uFXkm2tm0zFfeEPay0BbtcBaygyv9Om2EQGXhB+wFRZYtaoQhpC4a53\n7oCiSKSmemho8HaZfU612/YruN1Xfn4+mqZRUVFBbm4uAKWlu6J9HwoKCikt3cmIESOj7xUWtu0J\n0XKc8vJynM6UNsfpSZ0GuAMHDmyzbcuWLWzbtg3DMBg8eDDDhw/v8UH1RQOyPFwwZgRvbqrk7ZPX\n8bMlNRxR9QWb+43js8+PZPwpazDC9ZyVtoly52CqA2He3FXFtcPyOHFYDpt21bFiTTmLP9nJuBE5\nZKW23ylZkiQuG/o/3LdqBwE9wOKSd/jpURcl+GoFQRCERHn//fcP9RC+l8K6gRQpUVbN1gGu1UW5\n/Tm4rZpMad1vMtWSwbXLh+MKrIIgJJIqS2Q67V3v2AFFkcjwOFCD4V4vZ3e7PZx66nieeeYpbr/9\nt5SUbGf58rd55JF5AEyceB6vvvoyxx9/Aqqq8vLLLzJx4qR2j3Paaaczd+5cbr75DrZta32cntTt\nOoLGxkZmzpzJlClTuO+++5g9ezYXXXQRl112GU1NTT0+sL5owpg8HI1F1CvJfHOUm4GNW0gO1dLU\nnMTeGmtCdbD6U84faHVA3u0NsDaSIZ52RjHJbhthzeCfK3d0ep4UezLnFp4FwCfln1Pj73rdOEEQ\nBOH7oaamhvnz53PHHXdQU1PD0qVL2bp166EeVp8TX6Ks7pPBVZR9S5QjpcwSyO1mcLsT4EYyuCLA\nFQShj7n99t+iaRoXXjiJu+++jeuvvzGasb3wwos59dTTueaa6fzsZ5cwatQxXHrp5QBUVFQwYcJp\nVFRUAHDHHdZxpkxpe5ye1O0A98EHH6SyspIlS5bw2WefsXr1at566y0CgQAPP/xwjw+sL3LaVc4a\nnY+2t5DVw9zoiklR9dcArFnTDyQ7pqmRE1jDUalWvfnKPbWYponbaePC06wg+PONe6ms83V6rjPy\nTiHJ5sEwDVaUfdy7FyYIgiAcFjZs2MDEiRNZsWIFixcvxufz8fHHHzNt2jRWrVq1X8eqrq5m3Lhx\n/Pe/VgfLhoYGbrjhBo4//njOOOMMXn/99ei+pmkyd+5cxo4dywknnMCcOXPQ9cQ0SOktrQNca1tH\nGdyWJlOqJLc9BmBT9iODq4gAVxCEw9vo0WNYsiQ2VTUlJZUHHniI//znfd54YwmTJ0+NvqcoCjNm\nXM+iRctYuvQ9/vd/b0FRrH9Lc3NzWb78w2hpc2pqKvPmzeOdd/7b5jg9qdsB7vvvv8/vfvc7iouL\no9uOOOII7r33Xt55551eGVxfdMrI/ujVAwioNrYMcpLl243H8KJpKvXN1mfXXPM1p+daE68r/CG2\nNVrB7Cmj+pOWZMc04e0vdnd6HptiY3yetTTTJ+WfE9B6vgOZIAiCcHj5v//7P6ZPn87ChQux2axA\n6cEHH+SKK67g0Ucf3a9j3X333dTXx5aCuOeee3C73XzyySc88cQTPProo6xZswaABQsWsGLFCt58\n802WLl3KV199xQsvvNBzF3YIhDU92kVZjcTqRiSAVRQpNgdXipUo7zuPLaxF5vDauv46FdREBlcQ\nBCERuh3gqqqKw9F2CRun00k4fOCTpL9vcjLcFPRLR6/PZn2xEwnIrdsEwPp1VlBraD76GaXkeazP\n86tqq8Tbpsr86Pg8AD5dv5dQuPOn4+MHjkORFAJ6kG+q1vfSFQmCIAiHi/Xr1zNlypQ22y+99FK2\nb9/e7eO8+uqruFwu+vfvD4DX6+Xdd9/lxhtvxOFwcPTRRzN58mT+/e9/A7Bo0SKmT59Ov379yM7O\n5tprr+Vf//pXz1zUIRLWDKSWJlOROWyxDG5sP0mOdVFWpX0DXJHBFQRBONx0O8A9+eSTefjhh1s9\n7a2treWRRx7h5JNP7pXB9VWjBmei1+ZSnm3D65TJabLm1NbX2zAVq3GXr249o7OsDmIb6psJREq9\nTh7ZH0my1tj9emt1p+dJticxPPNIAL7Y+3VvXY4gCIJwmEhNTaW8vLzN9vXr15ORkdGtY5SUlPDX\nv/6V2bNnR7ft2rULVVXJz8+PbisqKmLHDuv+tWPHDoYMGdLqvZKSkg6XEewLwnpcibJmXYeOFeDa\n4nq/SLJKqGU5IKV1gBuMPIi227ruohwUc3AFQRASotsrjd9xxx1Mnz6d008/nbw8K8tYVlZGcXEx\nv//973ttgH3RyKIMFn+WiYnEjjw7o7Z5SaOJepKprc8lM/k7/I3bGTVwCktKIWyYbK73cUxmMunJ\nDkYUZbBuRy2fb9zLScNzOj3XCTnHsbZ6I5tqt9Ic9pKqiCWDBEEQvq9++tOfcu+993LLLbcAsHnz\nZj744AOefPJJfvGLX3T585qmcdttt3H33XeTlhZbo9Dn81lrGcZxOp0EAtb0F7/f3+p9l8uFYRiE\nQqF2q7v2JUkSHSzxvl/kSImwfBBLXrQIa0asRDmSidUjz/2dcZekqDY0rADYocgocUFuIGQFuG6n\n2mp7e+NuaTJlV2yd7ns46cnPO5HEuBNLjDvx+urYEzXubge42dnZvPnmm3zwwQfs2LEDh8PB4MGD\nOfnkk8V6bvsYPCAFFTumL4XdOQFGbQuQVldCffrR7NjuIfNYMI0gSvA7CpJc7Gjys6XByzGZVnfl\n44/MZt2OWjbsrCOsGZ12ZxyROQxFUtBNnU21WzlpwHGJukxBEAQhwWbMmIHH4+Ghhx7C7/fzq1/9\niqysLGbOnMn06dO7/Pmnn36aYcOGcfrpp7fa7nK5CAaDrbYFAgHcbqshotPpbPW+3+/vcOpSezIz\nPT36XSEtzXNQP28YJppuYmspUdbiS5RNkpNViDSeSk9PQ262glOPw0ZGhvUg2TTNaAY3OzMpur0j\nIc0qUU5yubvc93BzsJ/3oSLGnVhi3InXV8fe2+PudoAL1jzcM888k1NPPTW6rWX+rd1+4Gs5fd+o\nikx+v2TKmtL5rl8dAJneMnamH03lXlDs2eihKvyN2zky9fhIgOvDME1kSeLo4ixgM8Gwzpbd9Ywo\n6rjszKk6KE4tZEv9djbUbBYBriAIwveMYRjIcenPyy+/nMsvvxyfz4eu6yQnWw9Hq6qqyM7O7vRY\nS5cupaqqiqVLlwLQ3NzMTTfdxNVXX004HKa8vJwBAwYAVilzS1lycXExJSUlHHPMMdH3Bg8e3O1r\nqKnx9lgGNy3NQ329F8M48PLolsAUxfqvHMnghqzGyehGrHFjY5NGg9cK7mXDpDayvF8gpNFSoa2F\nwtHtHY3bp/kjx1A63fdw0lOfd6KJcSeWGHfi9dWx9+S4O3tQ2O0A9/PPP+e+++5j586dGIYR3W6a\nJpIksXHjxoMa5PdNUf9kSnem4stVqE9WSGmqRpZMDFMipOegUEXIu5sj805jWRl4NZ0Kf4gBbgfp\nyQ4G9UuitLKZtTtqOg1wAYZnHsWW+u1srN3Sp+dDCYIgCG395je/4fHHH0dVW9+yW7KrYAWu9913\nH5999lmnx1q2bFmr12eddRb33HMPZ555Jps2bWLu3LnMmTOHrVu3snjxYp599lkApkyZwvPPP8/Y\nsWNRVZVnnnmGqVO7v7yDaZr05KpChmGi6wd+v/P6I5FsJIOrRALeaNyrGBD5s4mdoO4FwCZJ0fNG\nj4HVZKqr8fjDVtDslJ0HNfZD4WA/70NFjDuxxLgTr6+OvbfH3e0A95577mHIkCHcfvvtbebpCG0V\n5CZjbLCaSO3NUElrCpIi+6nX3dQ3pJKZBEFfOXkOGbcq49MMdjf7GeC2yr2GFqRTWtnMtu8aujzX\nUenWE/bGUBPV/loySe69CxMEQRAS6vPPP+eGG27gySefbFMt1dDQwOzZs/nPf/5z0A0fH3jgAX73\nu99x+umn43a7ufXWW6MZ28suu4zq6mqmTZtGOBzmggsu6Nac38NVIJKqlRQNyTCRI02ktMgXLlXR\nowGuJNsIRt63x3VLbpl/C+B0dP11Khrgqt0r6xYEQRAOTLcD3MrKSubPn09RUVFvjud7o3+GBzPg\nxjRkqtNUjtoVJMVfSb29kPLvXGQeBZg64cBe8j1ONjf42N0c4KR+1s8PGZjKO1/sZldFE6Gw3mmH\nxoFJ/bHJNsJGmJKGUo7KK0jMRQqCIAi97qWXXuLqq6/m2muv5emnn8blcgGwYsUKfvvb3xIMBnng\ngQe4+OKL9/vY77//fvTPaWlpzJs3r939FEVh1qxZzJo168Au4jATCEaCU1lHjQS1JhBZ1hZV1SEE\nkmxHkiRCkco1uxwf4MYyuE57112UfWGrRNmpiiSBIAhCb+r2jJgJEyawcuXK3hzL90puphuQMX1J\nVKVbzxGS6ssAqNijI6tWaVnIv5f8JOvLSqk3NudnSF4qALphsrOiqdNzKbLCoGSrs3VJw64evQ5B\nEATh0DrqqKP4+9//zu7du7nqqqvYu3cvd911FzNnzmTUqFEsXrz4gILbH7JYBlePNpjSpdgzfyUy\nN1eSrYx5KJrBjeugHIxlcF3dCHCjGVxFBLiCIBzeNmxYx9Sp50ZfV1bu5c47b2bSpB8xZcpEHn/8\nYUIhq/meaZrMn/8Ukyefzbnnnskf//goegdzUhobG7nhhhs4++zxXHTR+Sxe/O9eGX+3M7g33XQT\nU6ZMYfHixeTn57dqeAEwd+7cHh9cX5bkspHkshEMeKhOsxpNuYPWGsKhoI5sy8bQdhH2V5KXMhSA\nmkCYkG5gV2TSkqy5uHVNQUr3NnFkflqH5wIoTM1ne0MJOxt39+6FCYIgCAmXn5/PggULuOqqqzjr\nrLNITU3l0UcfZfLkyYd6aH2SPxTL4DpCkQA3bn1aRdbQAVmxAtxAJMB1xpcoRybsKrKEqnSdL/Bp\nokRZEITDm2maLFnyJk899TiKEntwd//99zB4cDH/+tdSmpubuPPOW3jxxb8wY8b1vPHGP1i16iNe\nfHEhkgS33TaLhQtf4fLL23b2f+ihOSQne1iyZDlbtmzhllt+Q2FhMSNHjurR6+h2Bvfuu+9GkiTy\n8vJwOp3Y7fZW/+uODRs2MG3aNI499limTp3KmjVrOt1/1apVDB06FK/X291hHlZyM9yYQTdel4ym\nynhCsfm0mmEFrOHAXvo5rc/PBKoCoeg++f2s7mBlVV1f/0BPfwD2eCtFoylBEITvoZycHBYsWMCI\nESPIzs4+6Dm3P2QtGVxZ1XCEWwLc2DN/WY5keCMZXH+ky7Ir7gtfyzGcdqXLJZAM0yCoWZ2YXaJE\nWRB+8DTdoLLOd8D/21vnY0+1l73d2FfTja4HFPHSSy/w+usLufLKX0a3hcNhXC4X06dfhcPhIDMz\ni3POOZd1674FYNmypVx88U/JysoiMzOLK674Of/5z+I2x/b5fHzwwQpuvPFGHA4Hw4ePZMKEiSxb\ntuTgP9B9dDuDu3r1al555RVGjTqwCDsYDDJz5kxmzpzJxRdfzKJFi7juuut499138XjaroXU0NDA\nXXfd1aeDtew0FyV7XSBJNCYpZNSHcdkM/GEZvz8ZlwxhfyXZNgWHIhPUDfb6Qwz0WDe/gdkevt1e\nQ1lV18sJ9E/KASCgBajx1yEjlm0SBEH4Pvjoo49avb7qqqt44IEHuOKKK7jttttaPWWPX8ZP6Fi0\nQZSs4QhZX/50OXbflIgsC6RYU4j8kXI7l9q2yVR35t8GtNgawqJEWRB+2DTd4K5nP6W6IdD1zj0g\nK9XJ72eM7ValyfnnT+XKK3/J119/Gd1ms9l45JHW/Rk+/vhDhgw5AoDS0p0UFsaWjRs0qIDS0l3R\nlXZalJWVoigq+fn50aXSBg0qYOXK/x7U9bWn2wFuQUFBtNb6QHz66afIssxll10GwLRp0/jb3/7G\nypUrmTRpUpv9Z8+ezaRJk/jLX/5ywOc81DJSHJil1lzbuiSJjHpIkgL4cdPQ4MSVDoYeACNIjtNO\nqTdApT8ug5vdksFtjq6R25Fcdw4SEiYmuxvKKXAU9uq1CYIgCIlx9dVXt7u9urqaa6+9NvpaLNnX\nfYGgDpIBSlyJsjPuYbtpffFUVBe6YRKKrNcYn8H1BawMrqsbHZQDWuyLrEuUKAuCcJjKysrq9H3T\nNJk371F27drJvfc+AEAgEGi1wo7D4cQwDEKhEA5H7N87vz/Q6nXLvoFAzwf63Q5wr7vuOu644w6u\nuOIKBg0a1GY9vq6eGpeUlFBcXNxqW1FRETt27Giz75tvvkljYyM333xznw5w05IcGAErwG3wWDdF\nV7gJcFPfoJKbbu2nherp54oEuHElygOyrJttKGzQ0BwiPbnjm6JdsZHlyqDKX0NZQwUF/Qp75ZoE\nQRCExNq0adOhHsL3TiCkgRIGiJUou6wl9hRVBjOWwfXHNUuJz+A2+6yfT3Z3XTHl0/zRP4suyoLw\nw6YqMr+fMZbaxgMP7GRFIi3VQ32DF6OL9WQzUpzdyt52JRgM8MAD97J9+zaeeupZ0tMzACtIDQaD\nrfZTFKVNMOt0OgmFgm2O6XK56WndDnBblgb4/e9/3+a97jw19vl80aUNWjidbaP28vJy5s2bx9//\n/nfC4XB3h9dqLPLB/w6RZanVfw9EVqoTwg5MU6Ih2QpwHb46cOXQUCchFSmYpo6h1ZPpskqMG0Jh\nlEiXxpyM2C+8ptFPVlrnN8V+7myq/DXs9VYd1LgPhZ74vA8FMe7EEuNOvL469r46biExAiEdSY0E\nuJESZc1hVU05XSpGJCCVVSc+LTZ/zaXGMrhNkYqrJFesOVVHmkKxqUZJtrbTsgRB+GFRFZl+6Qce\n2CmKREaGB4dsoncR4PaExsYGbr7517hcbp555q+kpKRG3ysoKKS0dCcjRowEoLR0F4WFbZeVzc/P\nR9M0ysvLcTpTovv2xhK03Q5wD/YJssvlahPMBgIB3O7YL9cwDG6//XZmzZpFTk4OZWVl+32ezExP\nl80e9kda2oHfiArywoAEYTtNbuva7c014ILmphB2VwZBXxV2xU9eRjLsrqE+pJORYd1kM7CeDDf5\nQvjCZnR7Rwam9WN9zSaqvLUHNe5DSYw7scS4E6uvjhv67tj76riF3uUPatEA1x7J4Gp2N5jgdNkw\n9EiAq7gIxGdwlbYZ3O4EuM0hq1mkS3Wiyt3+6iUIgnDImabJXXfdSkZGJg8++EibKt6JE8/j1Vdf\n5vjjT0BVVV5++UUmTmw7/dTt9nDaaaczd+5cbr75DrZt287y5W+3md/bE7r9r+zQoUM7DBxtNhs5\nOTlMmjSJG2+8sVXDixaDBw/mlVdeabWtpKSk1RIHFRUVfPPNN2zcuJHZs2djRBZWP/3005k/fz5j\nxozpcpw1Nd4ey+CmpXmor/diGAf2ZESOjN8MO/C5rEE5go0ABPxhkJOBKhrq9mJLHw6AX9P5rrIh\n+pQ4K9VBky9ESVk9tcUZnZ7PI1sBcJW35qDGfSj0xOd9KIhxJ5YYd+L11bH35Li7ergo9D1NvjAo\n1hzaljm4YdUF4UiAG83guqIdlCXAER/g+rsf4DaFrQA32S7+LgmC0LesW/cta9Z8hd3u4Lzzzoxu\nP/LIofzpT89x4YUXU1tbyzXXTCccDnPOOedx6aWXA1Zsd8UVF/Pyy6+Tm5vLHXf8lj/+8WGmTJmE\ny+Xi+utvjGZ+e1K3A9z777+fefPm8etf/5pjjz0WgLVr1/LEE09wySWXUFxczNNPPw3EypnjjRs3\njlAoxMsvv8xPfvITFi1aRHV1dau5uwMGDODbb7+Nvi4rK+NHP/oRK1eubLfTcntM06SDtYUPiGEc\neOrfZbc+XjPswOu0bopOLbbkj25a833CwXpSbbGHAjX+MP3d1v5ZqS5K9jRRWefrchwZDisArvLW\noOsGRve7gh82DubzPpTEuBNLjDvx+urY++q4hd7V7A9HM7hJYevhfVhxRgJcFT0ug+vVYh2U45s9\nRgNcd/dLlEV5siAIfcHo0WNYsuQ9AEaNOoaPPlrd4b6KojBjxvXMmHF9m/dyc3NZvvzD6OvU1FTm\nzZtHbW1zr96bux3gPv/88zz44IOcccYZ0W1Dhw4lNzeXBx98kGXLlpGTk8NNN93UboBrt9t57rnn\nmD17No899hgFBQX8+c9/xu12c++99wJWEP19YlNlHHYFI+TAl2wFrHY91mhCN6w5tXq4mWSbiiyB\nYUJ9MEx/tzUxu2XebVU3WolnOq2uVUE9RFPYi0cRN1JBEARB2FeTPww2K0D1RHqehGXrvut0S2Ba\nQa2sumjyW5neZFvrr0wtAW5yt0qUrQBXZHAFQRB6X7cD3MrKSvLz89ts79+/P8rItDwAACAASURB\nVHv27AGsKL2hoaHDYwwdOpSFCxe22d5RYJuXl8fmzZu7O8TDUpLTRkPYjqFIBJ0qjoCGqoCmQzjs\nQAUMrRlZkki1q9QFNeoji8cDpCdZN9z6pmAHZ4jJiAS4ALX+OjxJIsAVBEEQhH01+0JImVaTKLff\nCmbDkhWoul2xBpeKLZmmsPV+fIAb1nS8kWWCkj1dd1FuFAGuIAhCwnR7turo0aN55JFHWgWwjY2N\nzJ07l9GjRwOwfPlyCgsLe3yQfVmS24apWTc/vyvSSVmxaoeDQWu7HvZimibJkUnbzeFYjXVaJMBt\n8IYwzc5T+R6bG1myfqWNoaYevApBEARB+H7QDQNvQEOyWQ+OXZEA169b92i3Oy7AVZNoCrVkcGNT\nieqaY0v6ZXSyhF+Lhkj/jRR78kGOXhAEQehKtzO4DzzwADNmzGD8+PEMGjQIwzAoKyujqKiIp556\nig8++IDHHnuMJ554ojfH2+ckuWzQZD0V9tsl0gCHrONFJhCwYU0tNjB0P57IzbNZi2VwU5OsIDis\nGfiDGm5nx6VQsiSTbE+iIdhIQ1AEuIIgCIKwL2+k5FiyBVF0EzWkYQL+SLMplzMEIZAUB7Jipync\ntkS5Lm79ypYH0Z2pC1jJgQxnWk9dhiAIgtCBbge4AwYMYNGiRaxatYotW7agqipHHHEE48aNA8Dt\ndrNixQoyMjrv9PtDk+yyYdZZH7PPZt08HYQBG16vQmakilgPN5PUEuDGZXBT426c9c2hTgNcgFR7\nMg3BRppEBlcQBEEQ2mjwWtlXyR7EHbAqqsKyI9qY0W4PoYes8mQgrkQ5LoMbmTbkcqi4HJ1/lQob\nGg0hK4ObLgJcQRCEXrdfi7EpisKpp57aqvNxCxHYts/jsoFuBaUBu9V90W4EADfN3liFuKF5SVKt\nRY+98SXKcXN7GpqDDMjqfF5tiiMZmqBBBLiCIAiC0EZdk5V9lWxBPA2RKUNq7N5qUwPoWOXJhmlG\n+2Kk2m1xx7AC3O6VJ8emdokAVxAEoff1wIqxQmecdgVTs26KwUiAq2rWjTEYAEm23jO0+BLlWIBr\ntyk47db2Rl9sXlBHWub3NIoSZUEQBEFoo7YxCJhItiCpXut+G3KnAiBJgGFlW1V7Go0hDT3S/yIz\nroKqZWWDjBRn1+cL1Ef/nOFM7YlLEARBEDohAtxe5rQroFuJ8oDd+riVsLVUUCigISvWzdHQA3El\nylqrY7QsIu8LdB3gtnRobFlzTxAEQRCEmNqmIJI9ABKkNFsBbjA1FwBPsgM9VAeA6sigNhi772Y4\nYgHu3lofADkZri7PV+WrBiDVkYxT7TogFgRBEA6OCHB7mdOuts3ghqwbYzDYOsB1K1aAGzJMdCPW\nMdkTeWrcHGgd+LbHY3MD4Av7eugKBEEQBOH7o64xgOSw7pEpkQxuwG1Ns0pNd6IFawFQHenRANej\nKjiU2FemikiAm5vh7vJ8Fb5KAAak5PbQFQiCIPSuDRvWMXXquW22G4bBr399LU899cfoNtM0mT//\nKSZPPptzzz2TP/7xUXRdb/OzYK3Ac8MNN3D22eO56KLzWbz4370yfhHg9jKHTQFDAVMiGMngygEr\nuxraJ8B1xt08gy3dLgC308oAe/1dZ3DdqvU02av5e+YCBEEQBOF7pLohgOS0AtQMn/Xg2ada03sy\ns2RM03qYbHNkUBMJcDPjsrfBkB6dg5vTjQB3r68KgIHJOT10BYIgCL3DNE0WL17ETTf9Ck1rG3cs\nXPgK33zzdattb7zxD1at+ogXX1zIggWvs3btNyxc+Eq7x3/ooTm43W6WLFnOnDl/4Omnn2TdurU9\nfh0iwO1l1vxZCVNXCdqsG6kSF+BK8QGuGvt1+OPm4XoiJcrebpQou21WgOsLiwBXEARBEPZVUeeL\nZnBTm62HyV7DahaVlhZb31Z1pFMdsF5nxM2/3VsXq5DKTe9GgOsVGVxBEPqGl156gddfX8iVV/6y\nzXvbtm1l6dK3GD/+jFbbly1bysUX/5SsrCwyM7O44oqf85//LG7z8z6fjw8+WMGNN96Iw+Fg+PCR\nTJgwkWXLlvT4dexXF2Vh/7U0iDJ1JZrBVXXrhmmaIEnWTXXfDG5Aj2Vwk6IZ3K5LlN2REuWwESZs\naNhk8SsWBEEQBAB/UKOhOYQ914eqmbibghhIeEPWA2iPxwtBkFUPsuJkj8+6X+e6Yt2Sv6vyAmC3\nyaSndN5FOaiHqAlYc3oHiAyuIAgRmqFF18c+EIoCIbufBp+XDqqBo9KdqajdjAfOP38qV175S77+\n+stW20OhEHPm/I7bbrubt95qXVZcWrqTwsLB0deDBhVQWroL0zSRJCm6vaysFEVRyc/Pp7a2Obrv\nypX/7dbY9oeIfnqZIxLgoquEI5+2asSeEBtYywB1FuDuVwZXjTW88Gt+bJGuyoIgCILwQ9eSfZWc\nXjIaNCTAa08l0igZp62eUBDsrlwCuh6dg9vfHVuyb2eFtUrBoJxk5Lgvb+3Z3fQdJtbBB6fnY4ji\nKkH4wdMMjfs/fST68Ku3ZTrTuXfsrd0KcrOystrd/swzT3HiiWM5+uhj2wS4gUAApzPWQM/hcGIY\nBqFQCIcj9hDQ7w+0et2ybyAQ2J/L6RZRotzLnPbIXyZDIaxGmkzpcQGuEQtwVVnGJlv7+LW4ANfZ\n0kW5OxncWIArypQFQRAEIaai1geSgeTyklVv3VObkwcAYHcoYFgdj+3uXPb6Yvfq/u7Yl7JdFdYy\nQoU5XT9ALm3cDVjr36a5xBJBgiD0PV9++QVffrmaa665rt33HQ4nwWAw+joYDKAoSptg1ul0EgoF\nW20LBgO4XF1P9dhfIoPby1oyuKahEI7MwY3P4OpGZB1c3Xp64VRkwoZOMK7eoKXJlC/YjS7KcRlc\nn2g0JQiCIAhRZZVeJFczkmRGA1xvej4AWf3chP3WfFmbK5fdXuu+nGxTSLJZ92HdMNi11yqtK+zf\ndYC7q6nM2jclr2cvRBCEPkuVVe4de+tBlyinpnloqO/ZEuX2vPvuO3z3XRkXXDABsDK2sixTWrqT\nhx/+IwUFhZSW7mTEiJEAlJbuorCwqM1x8vPz0TSN8vJynM6U6L5FRW33PVgiwO1lzmiJskLYFWky\nZcYCVcNomaNrlUE5FZmmsI4/rkTZEVkfNxjq4m8w4FAdSJKEaZpiqSBBEARBiLOrohHZbWVg+9da\n99kmRwYEYcDAcLSDst3dn5Jq6yFxYVLswfHOiiaCYeteXDyw84ysaZpsrdsOQFFqQc9eiCAIfZoq\nq2S7Mw/45xVFIiMpCXvIha6bXf/AQbj99ru5/fa7o68ffHA2qalp/OpX/wvAxInn8eqrL3P88Seg\nqiovv/wiEydOanMct9vDaaedzty5c7n55jvYtm07y5e/zSOPzOvxMYsS5V7mtMVlcCMlyhLQMt02\nGuBGsrrOyFq4gfYC3LCOaXb+l1iWZFyRheSDerDTfQVBEAThh8I0TXZWNCF7GlB0k+yaELqkUBey\nyugyM63AV7GlINvS2NlkBbhFKbEAd+NOa85cZoqDfmkuOrPHu5eGkDVfd1jmET1+PYIgCIeDCy+8\nmFNPPZ1rrpnOz352CaNGHcOll14OQEVFBRMmnEZFRQUAd9zxWzRNY8qUSdx9921cf/2N0cxvTxIZ\n3F5ma1n6x1BAktBVGUUzUBXQDdB0BTtgRAJch2IFwaH4ADeSBdYNE003samdN7VwKHZ8YT9Bveum\nVIIgCILwQ1DdEMAb0HAk15FTE0YxTGpd/TAiz41djirCGjiSCqgMhKOVVEXJcQHuLivAHVaQ0ao7\naHs21W4BIMnmYWBS/164IkEQhN4xevQYlix5r9337r57dqvXiqIwY8b1zJhxfZt9c3NzWb78w+jr\n1NRU5s2bR21tc69mnkUGt5dJkoSqyKBbQaoWyca2ZHB1LfIHU8c0DWyy9TpkxH7p0TJniJZGdcah\n2iPHCHWxpyAIgiD8MGwurQc1hOxuZmCV9QC4IbMYgIxsJ5rfagjlTCpgU721FFCSqtDPad1TfYEw\nW8vqARhemN7l+dZWbwRgaMYRyJL4uiUIgpAo4l/cBLCpMmakFFmPZHRVyQpgw1rsV2Aa4WgX5bDR\ntkQZuj8PFyCkiwBXEARBEMDKvsrJtQAUlFv3x9okq/nT4OJAbKpQSjHr66xGUsPTk6KZ2q+3VqPp\nJqoicXRx+0tptKgPNrC1fgcAx2WP6vmLEQRBEDokAtwEsCkSmNZHrUdKkBXZCnA1LRa8mkYIezsZ\n3PgAN9CNDK5TiWRwRYArCIIgCJimyabSOpS0KpwBgwHVYfxqEvWa1bMiJ7sKALt7II2Gm3Kf1cNi\nRLoneozPNu4FYGRRZnR1g458ufcbTEycipMRmUN745IEQRCEDogANwFsqgxG6wC3JYOrhWO/AsMI\nR0uUW2Vw7QeWwQ2KAFcQBEEQKKvyUtcUQEmrpKg8iGRCZcpgAFxuCUkvAcCdNozV1dbSHR5VYXCy\ntT5jZZ2P9Tus7O/YETmdnsswDT78bhUAx/YbiU2x9co1CYIgCO0TAW4CqIoczeC2JGxlrAA2HBfg\nmnoIeztNpvZ3Dq5dFRlcQRAEQWixelMlcnIdki3MsJIAJlCRORyAkaP8mHoAkLCnjuDLaqub8vFZ\nKSiRaUPvflmGCaQm2Rl9ZHan51pfs4kqfw0AZ+Sd0luXJAiCIHRABLgJYM3BbQlwIyXKWIFqKK7R\nsWlq0RLlcFyJsqrI0ZtsdzK4LSXKoouyIAiC8ENnmiarN1eiZJWR0qyTvzdMnas/zaYTMMnJ2gWA\nK/VI1jZBU1hHAk7ITgGg0Rviw2/3AHDmcQOth9YdMEyDxTveAeCItMHkJw/s1WsTBEEQ2hIBbgK0\nyuBGPnElksHVw7FlBkxDizaZCsWVKENsHm4grHV5vmiTKdFFWRAEQfiB217eyJ76BpTMCo7e4gOg\nLMtq/HTEkX5MzZpb6846gf+WW2XIozKSyIx0T37z4xKCIR2XQ+HM4zoPWFfvXUNZczkAkwdP7JXr\nEQRBEDonAtwEsObgRpYJinzikmllYrW4hGx8Bje+yRTE5uF2bw6uKFEWBEEQBID3vypDzS7DFdY4\neluABkcWVY4BgMmQwTsBcCQN4ovmVOpDGhJw1oBMAHZVNLFyjRWwThpbQLLb3uF5mkLN/HPrWwCM\nyhrGkLSi3rwsQRAEoQMiwE0AVYkvUba2yZEMra6bIFkbTUNHjWRwtX0D3EgGNxhundltj1MEuIIg\nCIJAXVOQ1Zv3oOaWMHqjD1Uz2ZZ9AgDDhlUjm9UA6Jln8H55HQAnZKfSz2UnrOn8ZfEGdMOkX7qL\ns8fkd3gewzRYsOl1msNeHIqdi4+Y2vsXJwiCILRLBLgJYFNjJcrhyPJAsmmVGuu6iSRbyw2YphYL\ncM3WAW7LnB9N7zrAdShiHVxBEARBWPzJTsjeSVrQx+hNPspTjqDemYPL5ado0DYAbGnH8M89Mppp\nkqQqTMzLxDRNXvzPJr6r9iJJcPX5w1st2dfmPDveYW31RgD+Z8gFZLoyEnF5giAIvWLDhnVMnXpu\n9HU4HObxxx/m/PN/xKRJP+Khhx4gHLZ6/Zimyfz5TzF58tmce+6Z/PGPj6Lr7VecNjY2csMNN3D2\n2eO56KLzWbz4370yfhHgJoBNkcGMBK6RAFcyrF+8rhlIUiTANTTUyILy+j4ZXJsaCXC1bgS4kQxu\nUMzBFQRBEH6g9tb6+GDjdmy52zj7syaCSjLbsk5ElnVOHrsFiTAoblZox7PHF0QCLh6cg1OReeOD\nHaxab83NnXpKEUPyUjs8z9s73+ftXe8DMK7/CZw84MREXJ4gCEKPM02TxYsXcdNNv0LTYs1q589/\nipKSHbz66hssXPhG5M8vA/DGG/9g1aqPePHFhSxY8Dpr137DwoWvtHv8hx6ag9vtZsmS5cyZ8wee\nfvpJ1q1b2+PX0flK5UKPUOPn4Ea6KMuGlcE1dKPDDK5pmkiRgLclwA13I4Nrj6y5F9a7bkglCIIg\nCN83pmny4rINKAVrOWFzE7lVJqvzzkJXFE4cvRGnvQHDlPnC+WPW1vsBOGtABsXJbl57fxvvfLEb\ngBOH9eOCUwrbPYdu6Pxr+xL+u/sjAIamH8FPjrowet8WBEHoiKlphGtrD/jnFQX8wWZCDV46SJZG\n2TIykNTuhXwvvfQC77//Llde+UsWLPgbAJqm8eab/+LZZ18kJcV62Pfggw+jaVacsWzZUi6++Kdk\nZWUBcMUVP+cvf5nP5ZdPb3Vsn8/HBx+s4O2338bhcDB8+EgmTJjIsmVLGDly1P5cfpdEgJsAiixF\n5+Dq0QA3ksHVYxlc4jK4ALppRl/bIj8X7kYGV40EzJohAlxBEAShtaVLl/Lkk09SUVHBgAEDmDVr\nFmeffTZr167lkksuwel0Rve99tprmTlzJqZp8thjj/H666+j6zpTp07lzjvvRFE6Lts9lN75Yjfb\nta8Y1vAdJ6wNsmbABALuFI4/egNZmbWETJWVtqlsb7CqpU7KTuWEtCQe/8ca1u+05uKOOSqbqycP\nbzdgrfRV8fLG19nRsBOAkZlDuXrkFdH7ryAIQkdMTaPkt3egVVcn5HxqVhZFcx7qVpB7/vlTufLK\nX/L1119Gt+3eXYqu62zYsI4777yZQCDAhAnncu21NwBQWrqTwsLB0f0HDSqgtHRXq0QdQFlZKYqi\nkp+fT21tc3TflSv/21OXGiX+JU4ARZaic3D1li7KehjkjufggpXFbfkF2VTrS0R3Mrg2JRLgmiLA\nFQRBEGJKSkq46667eOGFFxg9ejSffPIJM2bM4IMPPmDjxo2MHz+eZ555ps3PLViwgBUrVvDmm28i\nSRLXXnstL7zwAtdcc80huIrObdhZyz/XrmCoup4zPwuzZuA5hNNSGHfcGlJTvOwxs1gpnUl9wLpX\nnpidQnqDxm8XfU6z3yrJO+O4gVw+4QgUufVMLr/m573SD1leuiL6EPms/NP4cfEkFPnwDPYFQRC6\nqyULG6+xsRFNC/Pxxx/y3HMv4fN5ue22WXg8Hn7+86sJBAKtHow6HE4MwyAUCuFwOKLb/f5Aq9ct\n+wYCgR6/DhHgJoAiS9CSwY0EsC3LBFkZ3JYuyhpK3JMOzTAhcr9UIxnc7szBtcm2yM+LAFcQBEGI\nKSoq4uOPP8bj8aBpGtXV1Xg8Hux2Oxs2bGDo0KHt/tyiRYuYPn06/fr1A6zM7rx58w67AHdzaR1P\nrniTEwKrGbkpia8GTiCnsJFhR32JX3GwQj+RTeZgQEIyTYYZNr56bxdlVVY2wWFT+Nk5R3LKqP6t\njlvtr+GT8i/44LtP8GvWl7FUezKXHHUhx2aPTPRlCoLQh0mqStGchw66RDk11UNDD5cot8dut2EY\nBtdccx3JyckkJyfzk59czv/7f6/x859fjcPhJBgMRvcPBgMoitImmHU6nYRCwVbbgsEALpf7gMfW\nERHgJoD1BNgKUCNxrlWirIChtT8HF1ovFbQ/c3BjJcp6m/IAQRAE4YfN4/Gwe/duJk6ciGEYzJ49\nm6SkJDZu3Ijdbuess87CMAzOO+88Zs2ahd1uZ8eOHQwZMiR6jKKiIkpKSrp9j5EkCfkg21q+s+wt\nMs1N1DUonHjB1aR4UqLvGabJ26u3899vX2fyjr2E9aOpGJ3OiUM24fc4+dAYwza9AM2U0bxhlOog\n/gof73tjzRiPPyqby885kswUKxOx11vFuppNrKlcx9a6HdH9bLLK6Xknc37xBFxqLGvRGTlyb5fl\nvnU/FuNOLDHuxDqk41ZsqP1zDvjHZVnCleYh6PJi7NOYticokcSaokgUFBQgyzK6Ho5uN00DMFEU\nicLCQsrKdnH00dY82rKyUgoLi6L7tigoGISmaZSXl+N2W3N5d+8uZfDgtvseLBHgJoCiSGBEAlyp\ndZMpXTfb7aIM1hzcFtFlgrqTwY2UKJuYGKaBIomyKUEQBCGmf//+fPPNN6xevZrrr7+egoIC0tPT\nOemkk7j00kupqanhN7/5DU888QS33HILfr+/VQmay+VqtwStI5mZnoN+2Nq8dxdHjPCRkQ2bVzzL\n9vosTjzzbLaWlbFp9Spyqps4IykL+bj+ePrrVKhOFutnUFPvItwUJtzQQKgmgB5sne44bmg2k8cP\nICk9xKbaNWzfs4ttNSVUemta7ZfiSOLMopM5/6gfkeZM4UCkpXkO+PoPJTHuxBLjTqy+Om7ovbEn\nJ7uQJImMjCQyMpI4++yzef75+Tz22GP4/X7+3/9byAUXXEBGRhIXXXQhr722gLPPPgNVVVmw4G9c\ndNGFZGQktTpmRkYSP/rRj5g7dy5z5sxh69atvPvu2zz77LNt9j1YIsBNAOvJUOsMrqS3BLgGtJQo\nmwZq3CPucHsZ3G6VKMd+rWFDE/OCBEEQhFbUSLnauHHjOOecc3jvvfeYP39+9H232821117LY489\nxi233ILT2boEze/3o6pqt4JbgJoa70FncM+YcjkbVzzPgHwvWTleMrJ9VH7xGt4qGR2VtYUFNNhS\n0IJ29M0mRkjHCO8EDJANJEWDtBCqGsLh1snIkFCcfkpCdTy6pv1l9Tw2N8MyjmRM7jGMyhqGIisY\nPqj1Ne/X2GVZIi3NQ31972RbeosYd2KJcSdWXx039P7Ym5r8mKYZbQZ1222/5cknH+e8884jFAoz\nadJkfvzjS6itbebcc6dQVraH//mf/yEUCnPuuecxderF1NY2U1Gxh8suu5i///11cnP7c8std/LY\nY39g/PjxuFwurr/+RvLzi6Pn2R+dBcUJDXA3bNjAvffey7Zt2ygoKOC+++7j2GOPbbPfP/7xD/7y\nl79QXV1NUVERd955J2PGjEnkUHuUIkuYkSZTRnQObizAbZmDi6l3ncHdjxJlaGk01b0vIIIgCML3\n28qVK/nrX//Kiy++GN0WDocxTZM//OEP3HDDDSQlWV8agsFgNIAtLi6mpKSEY445BrCaVQ0ePLjN\n8TtimmaX88S6kpqSxrHnXMd/3/gLRxQ3Y7dr5AxsJmcgHK3L1NXoNO5pwlcRQvM2otvqCDqDeN0y\nXoeC1y3T7JLxumTCNplqE/C3Pke6I42ClHwKUvIYkjaYwpR8ZCkSmZtW1dXBMAzzoI9xKIhxJ5YY\nd2L11XFD7439mGOOZ8mS96LHdjrd3Hrr3dx6a+v9rPdlrr76Oq6++ro272Vn57J8+YfR18nJKcyb\nN4/a2ubosXtj/AkLcIPBIDNnzmTmzJlcfPHFLFq0iOuuu453330XjyeWXv/000957LHH+Otf/8pR\nRx3FokWLmDlzJsuXLyc9PT1Rw+1RVhflfebgtmRwNQMpcvO0MrhdzMHdjxJl6xii0ZQgCIJgGT58\nOOvWrePf//43U6ZM4cMPP2TlypW89tpr0eWAbr75ZsrLy5k/fz6XXHIJAFOmTOH5559n7NixqKrK\nM888w9SpUxM+fo/Hw5W/uZvtW3fyxbsLyUr3kZbuR1EMsvo1kNUPOAa8PhdV1QOo25uEWh5ioK+O\n5EA1KcEaHLofzaYQTnZhpiShpqXhysgmKTMXT3p/bBk52LKzke32hF+fIAiCcPASFuB++umnyLLM\nZZddBsC0adP429/+xsqVK5k0aVJ0v4qKCq666iqGDRsGwIUXXshDDz3Etm3bOOGEExI13B4VH+Dq\nkfi1pUTZNIH4ADe+i3JcBtem7H+TKbAaTQmCIAgCQHZ2NvPnz+f3v/89999/P4WFhfzpT39iyJAh\nzJ8/nzlz5jB27FicTieXXnop06dPB+Cyyy6jurqaadOmEQ6HueCCC/jFL35xyK4jPTOLH02z1mAM\nBZuo3vUN3trNqMpeFFXD4w7gGbSHwkGgGxK1talUVQ9le1UGRn2YDH8F6f49pO+uwL6zAp1NNAAN\ncedQ0tKwZ/fD1i8He04O9rx8HHl5qOkZonmjIAjCYSxhAW5JSQnFxcWtthUVFbFjx45W23784x+3\nev3ll1/i9Xrb/GxHeqJTI/RsZzWbKsdlcCMlynGZVQlrwJJkYFNj5zOlWBczuy2yzJBhdtppTJal\nVnNwTUnv8c5kvUF04EssMe7E6qvjhr479r467kQYM2YMb7zxRpvtQ4YMaVW6HE9RFGbNmsWsWbN6\neXT7z+5IZsCRpwKnYpoGId93eOu24q3bihHeiyKbZGfVk51Vz/ChO6irT+a78hw2VwwmHLaRqgTI\nMarIaizBWb0LKVJLrdfX46+vx791S6vzyW43jkiw6ywqxlk8BFt2tgh6BUEQDhMJC3B9Ph8ul6vV\nNqez88V9t23bxo033siNN95IRkZGt87TE50a4/VEd7KkJCdE5+Ba21oyuAB2hx0vYLPJZGYmo0gS\numni9jiiE6iTk63ulSZSl53GaiML1QN4ku1kpPVsZ7Le1Fc72YlxJ5YYd+L11bH31XELB0aSZBye\nfByefDLyzkIPewk07cDfuI1A43YM3Ud6WhPpaU0MH7qdir1Z7CwdwJb6PLYk55M00EHhoCQKMw2S\ngzWEqyoJVVYS2lNOeO9eME0Mnw//ls34t2wG3gNASU7BWVyMa8gRuIePwJGXj9QTT9sFQRCE/Zaw\nANflcrUJZgOBAG53+4v7fvTRR8yaNYtf/OIXzJgxo9vn6YlOjdCz3cmCwXDbDK6mxb1vPS0OBYPU\n1jajSKCbUN/op9Zm/YqCAStoDYW0TjuNybKEzRX7tdbUNZFs7H9nskTrq53sxLgTS4w78frq2Hty\n3D29fIGQOIrNgydjFJ6MUZimQaBpB97ab/HXb0KWNQb0r2JA/yqampPZvmMA5RXZrFsXZB2Q2S+Z\no0YewRGTcnB77BjBIKE95QTLygiW7SZYuovAzhLMUAi9qRHvmq/xrvnaOm9yCu7hI/CMHIln5NEo\naQe2rJAgCIKw/xIW4A4ePJhXXnml1baSkhImT57cZt9//vOfPPjgg9x/fznSigAAIABJREFU//3t\nvt+ZnujUGK8nupNJLf9vRpfDRdJjWVajJbtr6Oi6iSxJgElYj527pcxO07sejzOuRDmohftUZ7i+\n2slOjDuxxLgTr6+Ova+OW+h5kiTjShmCK2UIhh7AV7eepqovCAcqSU5q4tijNzNyZBklO/PZti2L\nmkovn7y/nU9X7KB4WDajjs8jp7AIZ2FR9JimphEsK8O/YxuB7dvwbd6EXl+P3tRI02eraPpsFcgy\n7qOGEjz9FJShI5GSUg/hpyAIgvD9l7AAd9y4cYRCIV5++WV+8pOfsGjRIqqrqzn11FNb7bdq1Sru\nu+8+XnjhhT69NFA8pWUOmCnHSpTNuCjcbHnfaiClREqs45cJajmGZuxfkyldNJkSBEEQhFZkxUlS\n1vF4MkcTbN5JU9Vn+Bu2oMpejhi8iSOGJFFVdyRrvk4mFDTZur6Sresr6dc/mVFj8hgyLBtZlpFU\nFWdhIc7CQjjrbEzTJFT+Hb716/CuX4d/8yZMTcO3cQM7Nm4AScJ15FGknnoaSaPHIHdzHWFBEASh\n+xIW4Nrtdp577jlmz57NY489RkFBAX/+859xu93ce++9ANx///0899xzhMNhrrnmmlY/P2/ePMaP\nH5+o4faoWJMTKVqiLJuxQNUk1kUZiHZSNtpZB7c7mQhFVqJ/1k0R4AqCIAhCeyRJwplchDO5iHCg\nioaKD/HVrQejmezUrzh3QhpNodGs+dJGXY2fyj1NvPfWRr74sITR4wo4cmQOiiK3Op5jYB6OgXmk\nn3MuRiCAd923NH+5Gu/abzECAfybN+HfvAl5wcskn3gSKaechnNwsWhSJQiC0EMSFuACDB06lIUL\nF7bZfv/990f//MILLyRySAmhtkwKNuVYiXJ8gGu2vG8Foy27a+0FuN3I4EqShCzJGKYhAlxBEARB\n6AabM5uswosI546PBLrr0MP1uKX3OWN8HmHlJNZ+bbBrew2N9QFW/Gczqz/eyXEnDWLYMf1R1LYN\nQGSnk+QxJ5J20kmkemzsXvkJ9R99FA12Gz5YScMHK3EMKiB9wjkkn3ASkprQr2aCIAhtbNiwjjvv\nvIVFi5YBUF1dxcMP/55vv12Dw2HnvPMuYMaM65FlGdM0eeaZP7F48b/RNJ1zzz2fX/96FoqitDlu\nY2Mj99xzO6tWrcLjSeKXv7yGyZN/3Ga/gyVa/CVAdJkeU0JvaTLVKoNrbTP3LVE22ilR7uZcMkVS\nIscQAa4gCIIgdJfNmUVW4YXkDp2BM9laojDkK8Ns+icnjtnCtOlDGTIsG4DmxiAfLt/Kq899zpZ1\nFZhmx/doxeEgZcwJDPz1/zL4kcfImnYJ9v4DAAiW7qLi+efYcfvN1Ly1CK2xsfcvVBAEYR+mabJ4\n8SJuuulXaFqsX9Djjz9MXl4eixcv57nnXuK995bz9ttLAXjjjX+watVHvPjiQhYseJ21a79h4cJX\n2j3+Qw/Nwe12s2TJcubM+QNPP/0k69at7fHrEAFuAshSSwArRefgyrTN4O4b4MY3/lSVlgC36wwu\nxMqURQZXEARBEPaf3ZVDvyGXk118GTZnPwB89esJVL7ISeOauPTq4zlyZA6SBE0NAd5bvInX/7qa\n0h01nQa6AGpqGhnnTqLg/gfJu+V2PMceB5KE3tBAzaJ/UXL7zVS+uoBwbW0iLlUQBAGAl156gddf\nX8iVV/6y1fbdu0vRdT36b5v8/9u78/io6nvx/69zzmyZ7CFAWAKEgOwSVqGAKCIoikiVulsvt6LF\n1l9dqtZWi+1t9Xettlqvt9bWtu6Pi7auqKACKoqKChQQFQgkkATIvs5yzvl8/5jJJEMCTJTMJPH9\nfDzGOft5zxjmM+/5bLqG2x2awvT111exZMklZGdn06tXNldccRWvvfZKm2s3NjbyzjvruP7663G7\n3YwePZYzz5zP66+/esJfh7SDiYPWNbh2ezW4sQwyZTQnwaFRQVv69R7lnlq4SbOKLSEWQgghRFtJ\nacPwpA6lvuJTqkveRlk+qg+sxunZwszTFjBh2iA+XL+HvV9VUHGogVf/79/0H5TBjDOGkd332FNM\naZqGd+QovCNHETh8iOq336L2vXewm5qofmsNNevXkjZjJllnnYOzd+84vWIhRGezLJv6Wv/XPt8w\nNLA1amqajjs+T0qaO2qsgGM555xFXHnlUj777JOo7ZdeeiX//d+/4YUXnseyLM4++1zmzJkLQFHR\nXoYMGRo5dtCgwRQV7UMpFTW2wP79RRiGg9zc3MiUp4MGDWb9+rUxxdYRkuDGQXPzYmVrkT64ejsJ\nriJU29pegutoldBato2ut23X3ppDC/2vNaWJshBCCPGNaJpOavZkvOmjqC55k4bKLQR9Bzn41d9I\nzipg3qK5HCobxMZ1uynbX0tJUTXP/X0Towr6M3XWEFJSjz9asqt3H/pcdAm9Fi6ieu1bVK15A7u+\nnpr166h59x3Spk0na+EiXL37xOEVCyE6i2XZPPPnj6ir8cXlfqnpHi5ZNjWmJDc7O7vd7Uoprrji\nP7jkkisoLS3h1ltv4IUXnuf88y/A5/Ph8Xgix7rdHmzbJhAI4G41UnxTky9qvflYn+/Evw/SRDkO\njMggU1qraYKOVYMbWm2vBhdi64fbfE9poiyEEEKcGIYzmV6DF9Fn+FWRZssNlZsp/fxhUpP2sujS\nAs767ljSMjwoBTs+K+HpRz5iy8f7sWLtYuT10uuchQy953dkL7kIIy0NbJva9zew9xc/4+BTj2NW\nV3fmyxRCiIjy8nJ+97u7ueyy7+PxeMjLG8rll1/FSy/9CwglqX5/S2203+/DMIw2yazH4yEQiK61\n9vt9JCV5T3jMUoMbB5HKV6W3NFFu3QfXPqIPrt5cg9tyjeY+uBA9+NTRRAaZkgRXCCGEOKE8KYPI\nGXk1dYc/oqZ0HbbZSGXRizRUbmHgoAVc/IOpbN20n0/e30fAb/Lu6q/YuaWU6XPyGTA4M6Z76B4P\nWfPPJuP0M6h57x0qX30Zq6aGmrVvU7vhPTLmzCXrrAUYKcduBi2E6FoMQ+eSZVO/cRPl9HQvNTWN\nJ7SJcnsqKsoxTRPTNHG5XOH7G5FRkgcPHkJR0V7GjBkLQFHRPoYMyWtzndzcXEzTpKSkBI8nLXJs\nXl7bY78pSXDjINJfVmlYzTW4gKaF+9SqcFW6OlYT5dY1uMf/FTgyyJQ0URZCCCFOOE0zSOszHW/G\naKr2v0ZTzZf46/dSuvMR0vrOoGDqTE4a25cP1xfyxb/LOHywnpee2cKQ4b2YccYw0jKSYrqP7nKR\nOWcu6TNmUf32m1S+tgq7sYGq11dRs34tmfPPJnPuPPRWTQSFEF2bYeikZ8b2GdD++RpZWcmgq+Mm\nuN9UXt5Qevfuw0MP/Z6f/OSnVFSU88wzT7Jw4SIA5s8/m2eeeYJJk6bgcDh44om/M3/+gjbX8XqT\nmTVrNvfddx833XQbu3btZs2aN7j33gdOeMzSRDkOopoot+ps3Zz4Hm0U5ahpglrV4MaU4EoNrhBC\nCNHpHK50eg+9mOy872E4U0FZ1Ja9Q+nOP2GoEuacM5IlV01iwKAMAPZ+VcGzj37Eh+v3EAyYMd9H\nd7vJOvsc8u75b7LOXYjmdmM3NVHxwj8p/NktVL25BjsYPP6FhBCiA1wuF/fe+wClpSUsWnQWP/rR\nMubOnceSJZcAsHjxEmbOnM3VV3+fyy//HuPGjeeiiy4DoKysjDPPnEVZWRkAt932C0zT5LzzFvDz\nn9/C8uXXR2p+TySpwY0DLfIzQksfXABdB8tq3Qc3lIzqxxhFGTrYRNmWUZSFEEKIzubNGIknNY+a\n0nXUHf4I01/JoV1P4M0cR/ag+Sz98Uw+eHc377+1m8aGAJ9+UMQX28qYdlo+w0f3iRpt9FgMbzLZ\n519AxulzqXztFWrWrcWqq+Xws09RteZ1shd9l9Rp09F0qcMQQnw9EydO5tVX34qs5+UN5fe//592\njzUMg2XLlrNs2fI2+3Jycliz5t3Ienp6Og888ACVlfWdWvMsn35xYLSqqbVbjYbcnMjadvP+ow8y\n1XoU5VgGmXJE5sGN/ddhIYQQQnx9uuEmc+B8ckb8AJe3PwCNVf9m/7aHqCj5iBFj+3LJsqlMmJaL\nrms01AV46+XPeeGpzRwuq+vQvRzp6fS5+DKG/OYe0mbMAk3DrKig7LFH2bfiDuo/+/S48/EKIURP\nJAluHDQnsqEmyq22N7dcPmIUZcfxanA71ERZanCFEEKIeHJ5+9H3pKVkDjwLTXdhWz727XiO0i/+\nBnYl007L56IfTGFwfi8AyvbX8NzfP2H961/Q1Bjo0L2cvbLJ+Y//ZPBd/0XKhEkABEoOUPI/D1J8\n93/RuPPzE/76hBCiK5MENw5aDzIV1US5uaY2UoN7ZBPllmM7PIpy8zRBMsiUEEIIEXeappPaeyr9\nRi3HmzkaAH99MWU7/0x1yVukZThZsGQcC5aMIz0rNNjMjs2lPP3IR2zdFPu0Qs3c/QfQ/7ofk3v7\nHSSNHAWAb89u9v/u/2f/73+Hb+/eE/r6hBCiq5I+uHFgtEpw0bTQk2pJcJWtNQ+jjFKq1TRB32AU\n5XANrimDTAkhhBAJ43Cl0Td/CYZVzN7tz2MGaqg9uIGGqu1k5S5gcP4wBg7J5N+bDrBpw14CfpMN\nb+5ix+YSZs4dzsAhsU0r1CxpaD65N99Kw47tlD+/Ev++vTRu30bR9m2kTJ5C9vnfxZXTr5NerRBC\nJJ7U4MZB6ybKACqcrDYnuNHjQNmRUZRtdbRRlGOpwZVRlIUQQoiuIr33KAaMWU5qn+8AGlagmsO7\nn6a88DmwGyg4JZdLl01lxLgcAKrKG3n52S28/s9t1FQ1dvh+yaPHMOgXv6TfD6/DmRO6Zv2mj9l7\n5885+PjfCFZWnsiXJ4QQXYbU4MZB6ybKAErXwGqd4Lb8zqBUS4Jrtp4mqNUgUx3pg2tLE2UhhBCi\nS9ANF5kD5pKcNY7K4lcJNOynsXoHTbW7yeg/h5TsScw5ZyRjJvTnvTVfcai0jsIvy9m3q4KR4/sx\n+TuDSU51x3w/TdNInTSFlIKJ1L7/HhUvvYhZVUnNO+upfX8DGXPmkjn/LBzpGZ34qoUQIr6kBjcO\nWpLT6BpcTQslsFE1uMqKjKLcugZX07TIdcyOTBMkg0wJIYQQXYorqS99h/8HmbnnoBkelO2nav9r\nlH3xF5pqd9O3fxrfvXIip58zkuRUF7at2PFZCU8/8iEb1+3B7+vYfLeaYZA+azZDfnsPvb93MXpK\nCso0qVr9OoW3/ZRDTz9BsKKik16tEELEl9TgxkHz3HaqdQ0ubQeZCh3TUoN7ZEtkh6Fj2VZMNbjN\n0wSZtkwTJIQQQnQ1mqaRmj0Jb/oIqg6sprFqG8GmMg7vfgpP6lAy+p/ByHH9GDayN9s+PcCnHxTh\n95l8trGI7Z+VMGFaLmMnDsDljv2rnO50kTnvLNJmzaZq9etUv7kau6mJ6rffonr9OtKmf4ess8/B\n1TenE1+5EEJ0Lklw48A4ooly81y4OuEaXKvV3EFRCW50hhupwY2lD67U4AohhBBdnuFMIXvId/Fl\nT6L6wJsEGg/gq9tD2Rd78GaOJb3faRScMohR4/ux+cNitm7aT8Bv8uH6QjZ/WMy4yQM5efIA3B5n\n7PdMSiJ70WIyz5xPzbq3qVr9BlZ9HbXvvUvthvdInTyFjDPnkzJ8WCe+ciGE6ByS4MZBu31waW6i\nrEU1UVbKOmqC2zxVkGXH0Ae3eZogGWRKCCGE6PI8KYPpe9JSmmq+oLrkLUx/BY1V22is2h5KdHNm\ncsrsoYydNIBP3t/H51tK8ftMNr23ly0fFTN2Yn9OnpKLN9kV8z0Nr5esBeeSccaZ1Ly7nqo3XsOs\nqqLu44+o+/gjkvLzsRYvQh8xBsI/nAsheq4tWzbz0EO/p6hoL+npGVx66ZWcf/4F1NbWcvfdv+LT\nTz8mOTmFpUuv5txzzwdAKcUjj/wPr7zyAqZpcdZZ5/DjH9+AYbT9zKitreWOO27lgw8+aHOdE0kS\n3DhoGUVZDz+F1g3CCe6RNbjtTBMEYBih82OrwQ39r5V5cIUQQojuQdM0vBkjSUo/iYaKz6gpewcr\nWEdj1b9prPo33ozRpOXM4tR5JzFx+mC2fFTMjs0lBAMWn20sZuumA5w0pi/jJg+gV++UmO+ru91k\nzp1H+uzTqf1gA9VrVhMoLaFp926+/N39ODIzyTj9DNJmnYojNa0T3wEhRKLU1tZy2203csMNtzB3\n7jy++upLfvKT5QwYMJAXX3werzeJl15aze7dX3Hzzf8fQ4bkM3bsOP75z//jgw/e4+9/fxZNg1tu\nuYFnn32Syy77fpt73HPPf5Gamsyrr67hyy+/jLrOiSQJbhwcOchUcxNlLdxE2Yzqg2uia6FmRkdr\nohzTKMpSgyuEEEJ0S5qmk5I9ieSs8dRXbKb24AasYA2N1TtorN6BJzWf1D6n8J05+UycPoitm/az\n7ZMDBPwWn28p5fMtpQwYnMG4yQMZnN+rpSXZcehOJxmnnkb6rNk07thO9VtraNi6BbOqivJ/Pkf5\ni/8iZcJE0mfNxjtqNJouY5UK0VMcPFjKd74zg3nzzgJgxIiRTJw4iW3btvLuu+t5+unncbvdjB49\nljPPnM/rr7/K2LHjeP31VSxZcgnZ2dkAXHHFVfzlL39qk+A2NjbyzjvreOONN9q9zokkCW4cHLWJ\ncqQPbsuxyrZwaKHmRUfmsY7mGtwOjaIsCa4QQgjRHWm6g9Tek0npNYGGqq3Ulr2HGajCV7cbX91u\nHO5epPaeypQZ4ymYOoidW0v59ycHqKvxcWBfNQf2VZOW4WHkyf0YMS6HlBinGNI0jeQxY0k7eRye\nxhr2Pv8i1RveQ/n91G/6mPpNH+Po1Yv0maeS9p2ZOHv16uR3QoieRdkWZrDma59v6xr+Rh9BXyPW\ncfIChzMdTT9+F4Phw0dwxx2/jqzX1tayZctm8vOHYxgOBgwYGNk3aNBg1q9fC0BR0V6GDBkata+o\naB9KqchAuwD79xdhGA5yc3OprKxvc50TSRLcOIj8wBkZZCq8PZzgWlbreXBj6IMbQxPl5lGUpYmy\nEEII0b1pukFKrwkkZ42nqXondYc/xN9QjOmvoGr/a1SXvE1y1lhGjilg7KSpFO2uYOumA5QUVVNb\n7eOjdwr5+N1CcodmMXJcP4YM7xXp9nQ83oEDyLniSnp990LqPv6ImnfX49uzB7OigooX/0XFi//C\nM2w4aVNPIWXSFBzp6Z38bgjRvSnbouTz/8EKVH+j6+yP8TjDlUH/UdfFlOQ2q6+v59Zbb2DEiFFM\nnDiZlSufjdrvdnvw+XwA+Hw+PB5P1D7btgkEArjdLT+qNTX5otaPvM6JJAluHOhHTBNka83TBDUn\nuK374FqR49s2UW7ugxtDE2WpwRVCCCF6FE3T8WaOxps5Gn9jCXWHPqSxejvK9lNf/gn15Z/g9PSh\nV68Czl0yjuoqxY7NJXy5/RABv0nR7kqKdlfiSXIwdERv8kf2of+gdPQYmhrrniTSZ80mfdZs/PuL\nqXnvHWo/eB+7oQHfrq/w7fqKQ888hXfkaFKnTiV5/AQcadJfV4jupqTkALfccgMDBgzgV7+6m717\n9xII+KOO8ft9JCV5gVCS6vf7o/YZhtEmmfV4PMe8zokkCW4caJqGptFmmiAtPIWP1bqJckyjKMs0\nQUIIIcS3mdvbH/eQxVjBM2mo3EJ9xWZMfwVB3yGqD6ym+sAaPKlDmDBxDFNPHU/RnkZ2bi1j/94q\nfE0mOzaXsmNzKUleZzjZ7U3OwPSYanbdA3Ppc/FlZF/wPRq3b6Puow+p3/wpKhCg8fPtNH6+HbS/\n4xmaT8r4ApLHT8DVv39Uc0Uhvq003aD/qOu+URNlQ9fIyPBSXX3imigDfPHFTm666cfMn3821133\nE3RdJzc3F9M0KSsrIycnNEd2UdE+8vLyABg8eAhFRXsZM2ZsZN+QIXltrt18nZKSEjyetDbXOZEk\nwY0TQ9faaaIcSj7tIxPc8H77qKMoxzLIVOgP2ZQmykIIIUSPZThTSOs7g9Q+3yHQUEx9xeZwrW4Q\nX10hvrpCQCM9NY/TzhiByUgKd/nZvfMw5QfraWoMsv2zErZ/VoLLbZCbl8Xg/F4Mys86bp9d3ekk\npWACKQUTsP1+GrZuofajjTRu+zcqGMS3exe+3bso/+dzOLN74x03Du/I0XhHjMRIiX2UZyF6Gk03\ncLqzvvb5hqHh9qbg9NWjx9B1MRaVlRXcdNOPufjiy7j88qsi273eZGbOPJVHHnmIW2/9BYWFu1mz\n5g3uvfcBAObPP5tnnnmCSZOm4HA4eOKJvzN//oI21/d6k5k1azb33XcfN910G7t2RV/nRJIEN050\nXcMOj6KstOYa3HATZZvQ/HLKOqIGl6gO2h3pgytNlIUQQohvD03TcKcMwp0yiMyBZ+Gr3UVD9XZ8\nNV+hlImvbg++uj0A5GRkkzdvGBaD2LfPxe7PK6g43EDAb7F752F27zwMQN/+qQwf1ZesPsn06ZeG\n03X0WiDd7SZ1ylRSp0zF9vtp/HwH9Vs+o2HrFqyaGoLlh6lZ+zY1a98GTcOdOwjvqFF4R47Gkz8M\nw3vimykKIWL3yisvUl1dxT/+8Vf+8Y+/RrZfeOHF3HrrL7j33rtZvHgBSUlJLF9+faTGdvHiJVRW\nVnL11d8nGAwyb97ZXHTRZQCUlZVxxRVLeOKJleTk5HDbbb/gD3/4b847r+11TiRJcONE1zTs8Dy4\nzWNKaZEaXBtNM1DKAtvCMFqa8FgKHOHVSB9cuwPTBEkNrhBCCPGtohuuSF9d2wrQVPsVTdWf01S7\nG2X7MX3l1PnKgY308ToYeOpANMcAyivTKNzj4MDeWixLcbCkjoMldaFr6hp9+qXSf1AG/XLT6dMv\nDU+Ss/37u92Rml1l2/j37aV+6xYad2zHV7gHbBt/0T78RfuoeuN10DRc/frjGZpPUn4+nqHDcPXr\nJ9MQCRFHV165lCuvXHrU/b/+9T3tbjcMg2XLlrNs2fI2+3Jycliz5t3Ienp6Og888ACVlfUxVdh9\nXZLgxomha5hHNlEO94+1bRVKcInugwvNzZSlBlcIIYQQHacbLpIzx5CcOQalLPwN+/HVfEVT7S6C\nvkMoZeKv3wvsJUWHccMNJp7cn0Awi4pKLyX73RQXWdg2lB2opexALXwQunZahoc+/dPo0y+Vvv3S\n6NUnpU0tr6brePKG4skbCosWY/uaaPzyS5o+30Hjzh34i4tBKQIlBwiUHKD2vXdCcScl4c4dFH7k\n4s4dhKt/f3SnK75voBCi25EEN070qD640YNM2VYowYW2CW7rgaY60gc3Mk2QJLhCCCGEADTNwJMy\nGE/KYDIGzMUM1OKv34uvfh/++n2Y/kpQFsHGYjSKyU6F7FEwfowHi17U1ady+JCTslIH9Q1eaqt9\n1Fb72LXjUOQeaRkeevVOIat3cuSRkZUUGalZ9ySRcvJ4Uk4eD4BVX49v7x6adu8O9dkt3IPd1ITd\n1ETTl1/Q9OUXLS9A13Hl9MM9YADOnH64cnJw9e2HK6cvuicpru+lEKLrkgQ3ThyGHpkmqHlWoJYa\nXBv01gluy3mtE1xHODE2O1KDG0NzZiGEEEJ8+zhcaTiyTiY562QArGAdvvoi/A3FBBpLCDaVoWwT\nZfvQOUC6F9KHwLAhofMtlUJjUwpVVR6qq500NnpobPKwd1cThV+VR+6j6xqp6R7SM5NIz0wiLfyc\nnplEarqX5LEnkzw2FIOybQJlpfj27MG/vwh/cTH+4iLsxkaw7UhN75GM9AxcOTk4e/fB1Tsbc9AA\n/J4UjMwsHJlZaEbsc4AKIbo3SXDjxOnQobkPbnj+Wy1cu9q6BhdlhUZcDouuwW2eJqgj8+Ca3zz4\nHsxWNqZtYSkTS9nY4YdlNy9b0dtV9HY0RbLPRXVtA6bVajuhAcJsFCiFQqGanyPLtLOt5RkFdngd\nVNRy5Nrha7SmUO0uNy8qFLqm4fE4afIFUOqIY6NOUUduilprubVqZ2/ra7a3v73rtL1ba5qm4XY7\n8PvNNq+7q2gvKk0Dt9uJ3x+ki4YdpfXfja5puNwOAn6zzcjuXZlTN1gw+nQy6ZXoUIToNgxnaqQ5\nM4CuK5Jc9Rwu3Y2v/gDBpkMEfIdQVmguS0OrJ9VbT6oXBg1ouY5SGv5AEvX1bhoa3fj8bvw+F021\nbqoOufH5XQSDDpq7YHlTXKSkuklOdYee09ykZJ1EyuBxpKW48SQ50OprCOwPJbuB0lICZaUEDh5E\n+X0AWDXVNNVU0/TFTgBa0mtA03BkZuLslY2RnoEjPR1HejpGWjqOjPBzejpGapr0+xWiB5AEN05c\nDh2s8NQ9WigB0sKJqmW3aqJsH9lEueUajnAT5Vj64DqN0MAPQdvEVja61j0+sC3bojHoJ2AH8FsB\nAlboOWgF8dst64Hmhx2MrAftIKZthh8WQdvEVC3rLftC25vfGyFEz1QdrOO68f+R6DCE6LY0Tceb\n2p/UYBrezAIg9KOqFawj6DscejQdwvRXYPqrsMz68HkKj7sRj7uR7KP8xmRZOj6/C7/fRSDoJBhw\n4A+4aKhwUFXqJBB0EgiEnoNBBwonniQ3Sd4BJGXkkdTficfrxK1bGL569MZatNpKtNpK9Opy9KrD\nUFcdmpJRKczKSszKyuO9YIyUFPTkZIzkFIzwc2g99NDD2/WkJHRPErrHE3q43WgO+VotRFcQ13+J\nO3bs4M4772TXrl0MHjyYu+66i4KCgjbHvfLKK/z+97+noqKCU045hd/85jdkZ2fHM9QTzukwUL5w\nghuuHdEjNbh2aJog2umD22ryZiPSRPn4SVmSwxNZ9lt+khyd2zfFthX+oEljMECdr5E6fwP1gSYa\ngk00BJpoMhtpDPrwWaGH3/Lht/0ElZ+A8mMSwCSATfercVYq3L9Eqls6AAAW00lEQVS6+YEWrsZr\n6XcdOQ6i90PLMa2OjVwHoo5V7R1L6/Pa2XbcF3C8Y9vZ3+5vLEe5zteISR03JiGOQ+k0+obA+EQH\n0vWsWrWKP/7xj5SVldG/f39uuOEG5s6dS01NDbfffjsbN24kNTWV6667jiVLlgChpOb+++9n5cqV\nWJbFokWL+NnPfoYhzT6/dTRNCzVtdqWRlJYftc+2g5j+KsxAVejZX4UVqMEM1mEFa7HNhsixhmGT\n7PWR7PXFfG/L0jFNg6DpwDSN0CPgwG8amLoDM9XA8rqweudi24OxLB2lDDRNR7c1lAWaaaOCFviD\n0BRA8/vQTRPNNDGUhWGbGA0mRl0QXVWgq0NoykJXdvhhoSsLLbIcetaw0R2OSNKrNSe+zcmvy4Xu\ndKI5XWhOJ5rTie5qXg7tMzwuyEqjwWehDCeawwDDQHM40Awj1MzaCC87DLTwMoYRmVJSCBHHBNfv\n93Pttddy7bXXsmTJEl588UV++MMf8uabb5KcnBw5bufOnfzyl7/kscceY8SIEfz617/mZz/7GY8+\n+mi8Qu0ULocOduiLQEAPJ6hWEAglh7oeGhVQWb6oBNds1STQ5Qyd7w8ef+Co1gltk+kjyZGEUgp/\n0KLRb9IYMGnwmzT6LZoCJo0BiyZfkPqgn8ZAI43BJnxmKBEN2D4CdgBT+TGVH0sLYBNA6UGUHgQj\niGaYoWe985owKiv0HirbCL2XVvOyA2w9vBxqCq7Czx1bb52gaqH9CpTSW21rJ5ntSDIZqyMvqYX+\n0+6dTvTtY75ejAee2MM65e0WPZCuoY/vk+goupzCwkJuv/12HnvsMSZOnMj777/PsmXLeOedd1ix\nYgVer5f333+fL774gquvvprhw4dTUFDAU089xbp163jppZfQNI1rrrmGxx57jKuvvjrRL0l0Ibru\nxJXUB1dS+//2lLKwgvVYwbrQI1CLZTZgm01YViO2GXpY4ecjf001DBvDsHG7g50Sv22Hynzb1rFt\nD0pp2HZomkfb1lC2jh3eplRoGaWhVOiH2VAvolAXI2WHww8/lAqCCqBUQ2hbAPARdQ6tlrVwFycA\nTamW7i0q9B9NNS8qtNDR4a8KLd9LWpLeVuvhAzWlobSWbxZa+HtG8z5arYMWvqwWtS1yvBaanrJ1\npYzWcttW50RtbL4p7R+qEV3ea0csarHd46hbwj/WOA3MoBXdpeu4Z34dJ+g64ctomobDoWOadjtd\ntk7MvTrjNxNNd1Awdz54U0/8xVuJW4K7ceNGdF3n0ksvBeDCCy/kH//4B+vXr2fBggWR415++WXO\nOOMMxo8P/ex+8803M336dMrLy7t1La7HZYAVersbPeGmxlWVkDqE+lo/9bUBnAZUlm6nbncAkoYD\n8PbmTWTbdQQ1jf2HDUDni+Jqfv/yOvwWBE2NoKlhWhA0wbTAMsHCB8NC9779n/+H7fOCHgDDRHME\nwRFEM4Kh5ciz2ZKgGuHHMcSS2ikFWA6U5Qy9fsuJsp0QfmgqtB56dqEpJ+BAwwHKgaY5AAdoDgxN\nBz38Aa2HP2x1LbTuAK2573L4Q7jlM77Vstbqg1zToj4oWj6rW5Yj14m86OgP2LafpdoR60ccd+QH\ndtTnfE/J3LpPP82O0nrwa+tpdGxGeEqB0YkOpUvJy8tjw4YNJCcnY5om5eXlJCcn43K5ePPNN3nj\njTdwu92cfPLJnHvuubzwwgsUFBTw4osv8v3vf58+fUKJyzXXXMMDDzwgCa7oEE0zcLjScbjSj3us\nUgpl+ULJru1HWX7s8EPZfmzLF1q2AqFl249tBbCtAMoyARPLDKCUCQRj+vwOdb+1CTVMkFkohOgM\nm9aWMPGc6zv1HnFLcAsLC8nPj27KkpeXx549e6K27dmzhwkTJkTWMzMzSU9Pp7CwsFsnuL3SPdhF\noVrVOm8owfXUlEH4B4x9+5wMGwoOo4bM1E2kmv2oI4Xtzj5A6AuFP7UJKEcp+Pf24zVTNvAEXWjO\nAEbO7uPlqkentFAyqkJJqKYcaDgxcKLjwMCJoTkwNAdO3cChO3BqDtyGjls3cBkaTkNhaDaGptAI\nPXQUmma3LKPQsNAxI8e07DvyuOjl5nVQaJpqbszb7nNzAaehInmYFvl59Yj9kd9EiTq3+WfWyHWj\nrtOhhrhHrB/tTBV16LGu3/qKWtSG6HtptLwCjnNce3pKKg49OR0/mp70f+/olK1RsrsvtO0F862X\nnJxMcXEx8+fPx7ZtVqxYQVFREQ6Hg9zc3MhxeXl5rF69GgiVzcOGDYvaV1hYiFKqB/04J7oSTdPQ\nHEnoX6OLlWFoZGWlUFlZHxm3RCkrPCJ0EGUHse1gKPlVFsq2UMoKLSu71bKFbVnYtolthR+2hW2Z\nKNvCVhbKtkMPZYdmxVDh9cggk3a4VrZ5W0vVroqq5g0t61p4dg3VeiDJqOpbWlfnalr0eujNa7Xc\nfFyr5ejK0NbntJx3zH/VzTuP/NLRvK51bsl63E+c4x7w9eOTj7tvxrY1kpP7dfp94pbgNjY2kpQU\n/SHl8Xjw+aL7XjQ1NeHxeKK2JSUl0dTUFNN9NE3jRAyAp4drA3X9xPwlnzKmL+9uLcGq7s2egQep\nTGsko/YwGU1lVCflUFTcjwH9D5LkCYSO17ew1p6GhYGmbJyYJGXYBHs7qT8cDDUHcYBhgMMAw6Fw\nGAqnoXDooUegbhg1GTtRmsKwHTiUA4dt4FAGLmXgsnXcSsdt6bhtDbel4zI1nJaO09JxWTqGrYWa\n8Lb6fFVKQ9kq3EzXDje7CUbyMKW05s/pyHkKre3nMrQ6p9XHe1S/1dCzFrlGS71xpEmG0lqOjbpH\nq3vSsu/rJ6BttXutrznSbOy1g9++lKw7kDKva9GUjZlcfMI+w3uafv36sWXLFjZt2sTy5cv5z//8\nzzZlb+sy+siyOSkpCdu2CQQCuN3u496vq5bN8SJxx1f7cYdbhOFp75QuQdc1MjKSqa5uwLa7T1kv\nccdfd429ddydKW4JblJSUptk1ufz4fV6o7YdLek98rijyc5O+WaBHiEjI/n4B8XgO1kp/LMgFzgv\ntOGK0NOsqKOWRJYmARe1d6HzTkg4QgghvsUc4dFep0+fzrx589i2bRt+vz/qmNZltMfjidrf1NSE\nw+GIKbmFrls2x5vEHV8Sd3xJ3PHXXWPv7LjjNnfM0KFDKSwsjNpWWFgY1eQJID8/P+q4yspKampq\n2jRvFkIIIUTHrF+/nquuuipqWzAYZNCgQQSDQUpKSiLbW5fRR5bNhYWFDB06NC4xCyGEEB0RtwR3\n+vTpBAIBnnjiCYLBIM899xzl5eXMnDkz6rhzzz2X1atXs2nTJvx+P/fffz+nnnoqmZmZ8QpVCCGE\n6JFGjx7Ntm3beOGFF7Btm/Xr17N+/XouuugizjjjDO677z6amprYunUrr7zyCgsXLgTgvPPO469/\n/StlZWWUl5fzyCOPsGjRogS/GiGEEKItTbUdW7rT7Ny5kxUrVvDFF18wePBgVqxYQUFBAXfeeScA\nv/rVr4DQHH0PPPAAhw8fZvLkydx999306nWUmcKFEEIIEbNNmzbx29/+lr179zJkyBBuueUWpk2b\nRnV1Nb/85S/54IMP8Hq9/OhHP+LCCy8EwLIsHnzwQZ5//nmCwSALFy6UeXCFEEJ0SXFNcIUQQggh\nhBBCiM4StybKQgghhBBCCCFEZ5IEVwghhBBCCCFEjyAJrhBCCCGEEEKIHkESXCGEEEIIIYQQPYIk\nuO3YsWMHF154IQUFBSxatIjNmzcnOqQO2bp1a5vpl7qyTZs2sWTJEiZNmsTcuXN59tlnEx1STFat\nWsXZZ5/NhAkTOOecc3jzzTcTHVKHlJeXM336dNauXZvoUGLy17/+lbFjxzJhwoTIY9OmTYkO67jK\nysq45pprmDhxIqeeeiqPP/54okOKyUsvvRT1Xk+YMIGRI0dyxx13JDq04/r000/57ne/y8SJE5k/\nfz4vv/xyokMSJ4CUzfElZXNiSNkcH1I2x19cy2Ylovh8PjVr1iz11FNPqUAgoFauXKmmTZum6uvr\nEx3acdm2rVauXKkmTZqkpk6dmuhwYlJdXa2mTJmiXnrpJWVZltq2bZuaMmWK2rBhQ6JDO6Y9e/ao\n8ePHq08++UQppdSGDRvUmDFjVEVFRYIji92yZcvUyJEj1dtvv53oUGJy4403qr/85S+JDqNDbNtW\nixcvVvfcc48KBALqyy+/VFOmTIn83XQnGzZsUDNmzFClpaWJDuWYTNNU06ZNU6+99ppSSqmPP/5Y\njR49WhUXFyc4MvFNSNkcX1I2J46UzZ1Pyub4i3fZLDW4R9i4cSO6rnPppZfidDq58MILyc7OZv36\n9YkO7bj+9Kc/8fjjj3PttdcmOpSYlZSUMHv2bBYuXIiu64wZM4ZTTjmFTz/9NNGhHVNeXh4bNmxg\n4sSJmKZJeXk5ycnJuFyuRIcWk2eeeYakpCT69euX6FBi9vnnnzNq1KhEh9EhW7Zs4dChQ9x88804\nnU6GDx/Os88+S15eXqJD65CGhgZuu+02VqxYQU5OTqLDOaba2loqKyuxLAulFJqm4XQ6Zb7Wbk7K\n5viSsjkxpGyODymb4y/eZbMkuEcoLCwkPz8/alteXh579uxJUESxu+CCC3jxxRcZN25cokOJ2ahR\no7j33nsj6zU1NWzatImRI0cmMKrYJCcnU1xczMknn8wtt9zCDTfcQEpKSqLDOq7CwkL+9re/sWLF\nikSHErOmpiYKCwt5/PHHmTFjBmeffTbPPfdcosM6ru3btzN8+HDuvfdeZsyYwfz589myZQuZmZmJ\nDq1D/vKXv3DSSScxd+7cRIdyXJmZmVx66aXceOONjBkzhssuu4w77rijW31hFG1J2RxfUjbHn5TN\n8SNlc/zFu2x2dMpVu7HGxkaSkpKitnk8Hnw+X4Iiil2fPn0SHcI3UldXx7XXXsuYMWOYM2dOosOJ\nSb9+/diyZQubNm1i+fLlDB48mOnTpyc6rKMyTZNbbrmFn//852RkZCQ6nJiVl5czadIkLrnkEh58\n8EG2bt3KtddeS+/evZk9e3aiwzuqmpoaPvzwQ6ZNm8batWvZtm0bP/jBD8jNzWXy5MmJDi8mDQ0N\nPPnkkzz66KOJDiUmtm3j8Xh44IEHmDNnDu+//z433XQTY8aM6RZfzkX7pGxOHCmbO5+UzfElZXP8\nxbtslhrcIyQlJbUpMH0+H16vN0ERfTsUFxdz8cUXk56ezkMPPYSud48/TYfDgdPpZPr06cybN4+3\n3nor0SEd08MPP8yoUaO6dMHTntzcXJ588klmz56Ny+Vi8uTJLFq0qMu/3y6Xi/T0dK655hpcLldk\nYIWuHndrb775Jv3796egoCDRocRk9erVbN26lbPOOguXy8Vpp53GaaedxgsvvJDo0MQ3IGVzYkjZ\nHB9SNseXlM3xF++yuXt8UsXR0KFDKSwsjNpWWFjIsGHDEhRRz7d9+3a+973vMXPmTB5++GE8Hk+i\nQzqu9evXc9VVV0VtCwaDpKamJiagGK1atYpXX32VyZMnM3nyZEpKSrjxxhv585//nOjQjmn79u1t\nYvT7/V2+X1VeXh6WZWFZVmRbc/+T7mLt2rWcffbZiQ4jZqWlpQQCgahtDodD+uB2c1I2x5+UzfEj\nZXN8Sdkcf3Evmztl6KpuzO/3q5kzZ6rHH388aqTGhoaGRIcWs40bN3abkRoPHz6spk2bph555JFE\nh9Ihhw4dUpMmTVL/+te/lGVZat26dWrixIlq165diQ6tQ04//fRuMVLjnj171Lhx49Rrr72mLMtS\n77//viooKFDbtm1LdGjH1NTUpGbNmqX+8Ic/qGAwqD755BNVUFCgPvvss0SHFrPTTjtNffDBB4kO\nI2Y7d+5UY8aMUc8995yybVt9+OGHasKECWrr1q2JDk18A1I2x5eUzYklZXPnkrI5/uJdNkuC247P\nP/9cXXTRRaqgoEAtWrSoW/3BK9W9CtH//d//VSeddJIqKCiIetx///2JDu24Pv74Y7V48WI1YcIE\ntXjx4m71QdOsuxSiSin11ltvqXPPPVeNHz9ezZs3LzLUfFe3d+9etXTpUjVlyhR1+umnq+eeey7R\nIcXMNE01YsSIbvfl8K233lLnnXeemjBhgjrnnHPU6tWrEx2SOAGkbI4fKZsTS8rmzidlc/zFs2zW\nlOpG9fFCCCGEEEIIIcRRSB9cIYQQQgghhBA9giS4QgghhBBCCCF6BElwhRBCCCGEEEL0CJLgCiGE\nEEIIIYToESTBFUIIIYQQQgjRI0iCK4QQQgghhBCiR5AEV4gEuO222xgxYsRRH3/84x/58MMPGTFi\nBH6/v9Pj2b9/f+Te119//dc+f/fu3cc99uabb47cK5bjhRBCiHiQslnKZtEzyDy4QiRAXV0dPp8P\ngM8++4wf//jHvP3227hcLgC8Xi9Op5Oamhp69+7d6fHs37+fM844g6eeeoqTTjqJtLS0Dp1vWRaV\nlZVkZWVhGMYxj62rq6OwsJAlS5awatUq8vPzv0noQgghxAkhZbOUzaJncCQ6ACG+jVJTU0lNTQUg\nPT0dgOzsbNxud9Rx8ShAW8vMzOxwAQpgGEbMsaamppKVldXhewghhBCdScpmKZtFzyBNlIXooo5s\nBjVixAhWrVrFwoULOfnkk1m6dCmlpaX89Kc/paCggHnz5rFx48bI+QcPHuT6669nwoQJzJo1ixUr\nVtDQ0BDTvZubNa1bt44zzzyT8ePHc8MNN1BSUsKyZcsYP348ixYtYufOnVHHNzdrmjNnDk888QSX\nX34548aNY/78+axfv/4Ev0NCCCFEfEnZLETXJwmuEN3Ifffdx5133smTTz7J9u3bOe+88xg1ahTP\nP/88w4YN46677gJAKcWPfvQjnE4nK1eu5KGHHmLnzp3cfvvtHbrfgw8+yB/+8Acefvhh1qxZwwUX\nXMCCBQtYuXIlHo+H3/3ud8c899JLL+XVV19lxIgR3H777QSDwW/0+oUQQoiuRspmIboWaaIsRDdy\nxRVXMGXKFACmTZtGcXExS5cuBeCiiy7i2muvxbIsPvroIwoLC3n66adxOp0A3H333Zx11lmUlZWR\nk5MT0/1++MMfMmbMGABGjhxJ//79Of/88wFYtGgRjz766FHPXbhwIQsWLABg+fLlLFq0iLKyMnJz\nc7/eixdCCCG6ICmbhehaJMEVohsZNGhQZDkpKSmqQPJ4PNi2jWma7N69m/r6eqZOndrmGoWFhTEX\nokfeb+DAgVH3CwQCRz13yJAhkeWUlBQA+ZVYCCFEjyNlsxBdiyS4QnQjDkf0P1ldb7+XgWmaDBo0\nqN1fcTsyOEas92tP86/Trcmg7UIIIXoaKZuF6FqkD64QPVB+fj5lZWWkpqYyePBgBg8ejGma3HPP\nPdTX1yc6PCGEEOJbR8pmIeJDElwheqAZM2aQn5/PjTfeyPbt29m2bRs//elPqaqqok+fPokOTwgh\nhPjWkbJZiPiQBFeIHkjXdR5++GFSUlK4/PLLWbp0KUOGDOGhhx5KdGhCCCHEt5KUzULEh6ak4b0Q\n33r79+/njDPOYNWqVeTn5/e4+wkhhBDdjZTNQnw9UoMrhIioqqqitra2U+9RV1dHZWVlp95DCCGE\n6CmkbBaiYyTBFUJEXHbZZfziF7/o1HvcddddLFmypFPvIYQQQvQUUjYL0THSRFkIIYQQQgghRI8g\nNbhCCCGEEEIIIXoESXCFEEIIIYQQQvQIkuAKIYQQQgghhOgRJMEVQgghhBBCCNEjSIIrhBBCCCGE\nEKJHkARXCCGEEEIIIUSP8P8AOk99z1wDtEQAAAAASUVORK5CYII=\n",
"text/plain": [
""
]
},
"metadata": {},
"output_type": "display_data"
}
],
"source": [
"IC = [C0,T0,Tcf] # initial condition\n",
"t = np.linspace(0,8.0,2000) # simulation time grid\n",
"\n",
"qList = np.linspace(0,200,11)\n",
"\n",
"plt.figure(figsize=(16,4)) # setup figure \n",
"for qc in qList: # for each flowrate q_c\n",
" X = odeint(deriv,IC,t) # perform simulation\n",
" plotReactor(t,X) # plot the results\n",
" \n",
"plt.legend(qList)"
]
},
{
"cell_type": "markdown",
"metadata": {},
"source": [
"The results clearly show a strongly nonlinear behavior for cooling water flowrates in the range from 140 to 160 liters per minute. Here we expand on that range to better understand what is going on."
]
},
{
"cell_type": "code",
"execution_count": 4,
"metadata": {},
"outputs": [
{
"data": {
"text/plain": [
""
]
},
"execution_count": 4,
"metadata": {},
"output_type": "execute_result"
},
{
"data": {
"image/png": "iVBORw0KGgoAAAANSUhEUgAAA7gAAAEhCAYAAABVzwz5AAAABHNCSVQICAgIfAhkiAAAAAlwSFlz\nAAALEgAACxIB0t1+/AAAIABJREFUeJzs3Xd8FNXawPHf7mx2N70SgkgJJQGS0KS3eymK0hFRVEQB\nQRBRUBDkKnjVi+iVpvgqICBgvd6LoqIigoICgiAiRUIHqenZlO1z3j8WVtYEjRpCe76fz5jdM2fm\nPDOSnX0yZ84xKKUUQgghhBBCCCHEZc54sQMQQgghhBBCCCHKgyS4QgghhBBCCCGuCJLgCiGEEEII\nIYS4IkiCK4QQQgghhBDiiiAJrhBCCCGEEEKIK4IkuEJcQDJI+R9XXudMzr0QQoiykOtF+ZNzKi4m\n08UOQIjycNddd7F58+aAsqCgIGJiYmjfvj2PPvookZGRFRaPy+Xi2WefpUuXLrRt2/aCtLFv3z4W\nLFjAxo0bycvLo2rVqvTo0YPBgwcTHBx8Qdq8kPLz85kyZQoPPvggtWrV+kv7evvtt8nPz2fEiBGA\n799HXFwcM2fOLI9QhRBC/AlXy7V606ZNDBo06Dfr9O3bl2nTppVbm5eSTZs28b///Y/nn3/+Yoci\nrlKS4IorRps2bXjooYf87x0OBz/++CMvv/wy+fn5zJkzp8JiycjI4K233qJjx44XZP9r1qxh7Nix\nNGvWjAkTJhAdHc3OnTuZN28eGzduZMGCBZjN5gvS9oWyZ88ePv30U0aPHv2X9zV//ny6devmfz9l\nyhSCgoL+8n6FEEL8NVfDtTolJYV3333X//6tt95iw4YNAccWExNTrm1eSt577z2OHz9+scMQVzFJ\ncMUVIyoqisaNGweUtWrViqKiIubOnUtRURGhoaEXKbryk5mZycSJE+nWrRvPPvusv7x169Y0btyY\ngQMH8u6773LXXXddxCgvLXXq1LnYIQghhODquFaHhYUFHOMXX3yB2WwucdxCiAtDnsEVV7ywsDAg\n8HmQL7/8kj59+pCWlkbnzp158803A7ax2Wz885//pEOHDqSmptKuXTumTp2K2+3218nJyWH8+PG0\nbNmS5s2b8+CDD3L69GmOHTtG586dARg2bBgTJ04EfF2h5syZww033EDDhg3p27cvX375pX9/mzZt\nIjk5mXfeeYfWrVvTrl07CgsLSxzP+++/j91uZ/z48SXWNW/enAceeICEhAR/2ZEjR3jggQdo2bIl\nzZo1Y+zYsZw+fdq/fuLEiYwbN4558+bRvn17GjZsyH333RdQB2DJkiV07dqVRo0a0bt374DYlVLM\nmzePTp06kZaWxs0338ymTZtKHNv333/PLbfcQlpaGl27duWLL77wrz/bnatbt2689NJLHDt2jOTk\nZJYsWUKHDh1o0aIFhw8fxuVyMWPGDLp06UJqaiqtWrVi4sSJ/nPVqVMnjh8/zvz58+nUqRPg6xY3\nduxYfzxZWVlMnDiRdu3a0bhxY4YNG8aBAwf861966SVuv/12li1bRpcuXUhLS+POO+9k//79Jc65\nEEKIv+5Ku1aXlVKKuXPn+q+f/fr147vvvvOv37BhA8nJyWzZsoU+ffrQsGFD+vXrx/79+/nkk0/o\n0qULTZs2ZezYsRQXFwdss2HDBrp160ajRo0YOHAg6enpAW1v3bqVAQMGkJaWRocOHZgzZw66rvvX\nd+jQgVmzZtGnTx8aNWrEihUrAN8d2t69e9OwYUOaNGnC0KFD/dfQcePG8dFHH/H999+TnJzMqVOn\nmDlzJh06dAho+8svv/SvB7j99tt5+umnGThwIA0bNmT+/PmA73GsoUOH0rhxY1q1asUzzzyD0+n8\n0+dbXCWUEFeAgQMHqoceeki53W7/YrPZ1FdffaVat26thg0b5q+7du1aVa9ePfXYY4+pdevWqZde\neknVr19fvfHGG/46Q4cOVTfccIP65JNP1IYNG9T06dNVUlKSevfdd5VSSrndbtWzZ0/VqVMntXz5\ncrVmzRrVo0cP1bdvX+V0OtXnn3+ukpKS1KJFi9SRI0eUUkqNHj1aNWnSRC1evFitXbtWjRs3TiUn\nJ6s1a9YopZT69ttvVVJSkuratav66quv1CeffFLqsd5zzz3qtttuK9N5OX78uGrRooXq37+/WrVq\nlfr444/V9ddfr7p06aIKCgqUUkpNmDBBNW3aVN12221q9erV6sMPP1TNmzdXDz30kH8/8+fPVw0a\nNFAzZ85U69evV1OmTFEpKSlqx44dSimlpk+frlJTU9Wrr76q1q5dqx5++GGVmpqqdu7cGXBsHTt2\nVEuXLlXr169X99xzj0pLS1O5ubmqoKBAvfHGGyopKUmtWLFCnTx5Uv38888qKSlJtW3bVq1cuVK9\n//77SimlJk+erFq3bq2WLVumvv32WzV//nyVkpKiZsyYoZRSateuXapt27Zq/PjxateuXf5/H2PG\njFFKKVVQUKA6d+6sbrjhBvXxxx+rVatWqX79+qkWLVqoEydOKKWUevHFF1XTpk1Vt27d1CeffKJW\nrVqlOnbsqG655ZYynXchhBAlXU3X6nP9+9//Vh07dix13XPPPafS0tLU3Llz1dq1a9XYsWNVamqq\n+umnn5RSSq1fv95//Vy2bJlavXq1ateunerUqZPq0aOHWrVqlXrjjTdUcnKymjt3bsA2LVu2VPPm\nzVOrV69WN998s2rZsqXKy8tTSvmulampqWrUqFHqq6++UosWLVINGzZUL7zwgj+29u3bq5SUFPX6\n66+rlStXqtOnT6vly5er+vXrq7lz56pvv/1WLVu2TLVr107deeedSimljhw5ooYMGaJ69uyptm3b\nppxOp5oxY4Zq3759wHGvWbNGJSUlqZMnTyqllBowYIBq0KCBmjFjhvrqq6/U/v371cmTJ1WLFi3U\nHXfcoVavXq3+85//qBYtWqixY8f+7jkXVzdJcMUVYeDAgSopKanE0qRJEzVx4kT/B7pSSvXr108N\nGTIkYPuZM2eqli1bKpfLpex2u7rnnnvUxo0bA+r07NlTPfroo0oppVatWqWSkpL8FyClfBeLTp06\nqaNHj/qTs7Vr1yqllPrpp59UUlKSWr58ecA+Bw0apHr16qWU+uWiefbCfD7dunUr84f7v/71L9Wi\nRQt/MquUUkePHlUNGjRQixYtUkr5EtzU1FSVn5/vrzN9+nTVuHFjpZRSXq9XtWjRQv3rX/8K2PfA\ngQPVokWLVG5urkpNTfVfWM+644471MiRIwOO7a233vKvP3jwoEpKSlKfffZZQJ39+/crpZT/HJ5N\nXM8aPXq0+vDDDwPKRowY4b+4KqVUx44d1b///e+AWM8muK+//rpKSUlRP//8s3+9zWZTzZs3V888\n84xSypfgnhuLUkq98847KikpSeXk5CghhBB/3NV0rT7X+RLc7OxslZKSol577TV/ma7r6rbbblOj\nR49WSv2SrJ57/Zw5c6ZKSkpS27Zt85cNHDhQPfDAAwHbvPzyy/71mZmZKi0tTS1cuFAppdSoUaNU\nz549ldfr9dd5++23VUpKiv861759+4Brq1JKvfzyywHXV6V8fwSvV6+e//0jjzyiBgwY4H9f1gT3\n1+do6tSpqk2bNqqoqMhftm7dOpWUlKTS09OVEOcjXZTFFaNdu3b897//5b333mPy5MlYrVZuvvlm\npk6d6h+Vsbi4mJ07d9KhQwc8Ho9/adeuHbm5uezbtw+r1cqiRYto2bIlR48eZe3atcybN4/s7Gx/\nt6dt27aRkJBAvXr1/O03aNCA1atXU61atRKxbd26FYPBwI033hhQ3q1bN/bs2RPQvSkxMfE3j9No\nNOL1est0TrZu3Uq7du38Xb8AqlWrRlpaGt9//31AmxEREf73lStXxm63A3Do0CHy8vJKDMKxdOlS\n7rnnHrZv347L5aJ9+/Ylzum53ZQBGjVq5H99thv12XbO59fn48UXX6Rnz56cPHmS9evX8/rrr3Pg\nwIGALmm/ZevWraSmpnLttdf6y8LDw2nfvj1bt271l4WFhVG7du0/HK8QQojzu1qu1WXxww8/4Ha7\nA66fXq/3d6+fcXFx/mM5Kzo6moKCgoBtunfvHrBNo0aN/Nf+7777jrZt26LresD5dbvdbNu27bzH\nef/99zNu3Djy8vLYsmUL7733HmvXrkXX9TJ/NzmfmjVrBrz/7rvvaNasGWaz2R9jy5YtCQoKKnF+\nhDiXDDIlrhgRERGkpaUB0LBhQ0JDQ5kwYQKVKlXivvvuA3zP6yilmDp1KlOnTi2xj8zMTMA3IMS/\n/vUvTpw4QVxcHI0bN8ZisfifDcrPz/9DIyDm5+cTHh5eYmTj2NhYAIqKivxlv7ffa665xv/MSmmy\nsrKIiorCZDJhs9n8bZwrJiYm4EJttVoD1huNRv+x5uXlAb6LZ2nOru/Tp0+p689NCC0WS0AbQMDz\nPqX59fnYsmULU6ZMYf/+/URFRZGamorVai3znHu/dU527tzpf//rc2IwGMoUrxBCiPO7Wq7VZXH2\n+tmzZ89S17tcLv/rXw+8pWna786WUKlSpYD30dHR5OXloZQiLy+PhQsXsnDhwhLbnT2/UPI4T58+\nzWOPPcb69esJDg4mOTm51Oen/4xft5WXl8euXbv47LPPfjNGIX5NElxxxerTpw/Lly9nzpw53Hjj\njdSoUcP/Ifzwww/Tpk2bEtvUqFGDw4cPM2bMGO644w6GDRvmv0D079/fXy88PJzc3NwS269du9Z/\n4T5XZGQkBQUFuFyugAtSVlaWf31ZtW7dmhdeeIG8vDyioqJKrH/44YcpKirif//7HxEREWRnZ5eo\nk52dzTXXXFOm9sLDwwFKHO/u3bvRNM1/53fBggWlHkd5TldUUFDAyJEjadeuHfPmzaNq1aoAjBkz\nhhMnTpRpHxEREWRkZJQoz87OrtD5F4UQQly51+qyOHv9fP311wN6Wp1lMv21r+n5+fmEhIT43+fk\n5BAXF4fBYCA8PJy+ffvSq1evEtudvbaW5uGHH6agoIAPPviA5ORkjEYjS5cu5ZtvvjnvNgaDocQf\nh88OiPVbwsPDuemmmxg6dGiJdb9O3oU4l3RRFle0xx57DI/Hw/Tp0wFft9OkpCSOHz9OWlqaf8nJ\nyeGll17C6XSye/du3G439913n/8DNCsri7179/o/oBs1asTJkycDRiTcu3cvw4cP5+DBg2iaFhBH\n06ZNUUqV+Cvkp59+Sv369UvcLfwtvXr1wmq18sILL5RYt3HjRr777jtuuukmf7vffPNNwN3an3/+\nmZ07d5Z5uoKz3ZfXrVsXUP7444+zZMkSGjZsiMlkIj8/P+Ccrl+/nrfffrvEuTifstQ7ePAgNpuN\nwYMH+y/ADoeD77//PuDiefbucGmaNm3Kzp07OXbsmL+ssLCQb775RqZwEEKIi+BKvFaXRcOGDdE0\nrcT1c926dbz33nu/eS0ri7Vr1/pfZ2Zmsn37dpo3bw5AkyZNOHr0aEC7ALNmzSInJ+e8+9y+fTt9\n+/alfv36/vjWr18P/NLD6dfnNSQkhLy8PBwOh7/s3EeCzqdJkyYcPHiQlJQUf4yVKlVi+vTpHD16\ntCynQFyl5A6uuKIlJSXRp08fli1bxrZt22jSpAkPPPAAY8eOJTQ0lA4dOnDs2DFeeOEFUlJSqFSp\nEvXq1UPTNKZNm0a/fv3IyMjg1Vdfxel0+rvbdurUiaSkJEaPHs2YMWOwWCzMmjWLpk2b0rRpU/9f\nJr/++muuvfZa6tevT5cuXXjyySfJy8sjMTGRjz/+mE2bNv3hSe1jYmJ48sknefTRRzl9+jT9+vUj\nIiKCrVu3snDhQlq0aMHdd98NwD333MP777/PkCFDGD58OC6Xi9mzZ1O5cmX69etXpvaCgoK49957\nefHFFwkNDaVp06asXLmSffv2MXXqVGJjYxkwYABPPvkkWVlZJCcns3nzZl555RUeeuihMh/X2TvF\nq1evDujKfK7ExERCQkKYPXs2Q4cOpaCggIULF5KRkeHfHnx/Fd++fTs//PBDiaS1X79+LFmyhKFD\nh/LQQw9hNpuZN28euq77z5sQQoiKcyVeq8siPj6e/v37M2XKFDIyMkhKSuLbb7/l1Vdf5ZFHHvnL\n+3/hhRdQShEfH8/LL79MpUqV6Nu3LwAjRoxg0KBBTJo0iW7dupGbm8vMmTMJCwsr8SzsuVJSUnjn\nnXeoVq0aFouFDz74gK+++grwPZJkNpsJDw/n6NGjbNy4kaZNm9K+fXumT5/O5MmTufnmm9m6dSsr\nV6783fiHDBnC8uXLGTVqFAMGDMDlcvHSSy+Rl5dHcnLyXz4/4sold3DFFW/MmDFYrVaef/55ALp2\n7cqMGTPYsGEDw4YN48UXX6R79+68+OKLANSqVYupU6fyww8/MHz4cF566SU6d+7MiBEj2LFjB16v\nF7PZzMKFC0lNTeWJJ55g0qRJ1KtXj5dffhmj0UhYWBhDhgzh3Xff5bnnngNg+vTp3HrrrcybN49R\no0Zx4MABXnnlFbp06fKHj6lHjx4sXrwYTdN45plnGDlyJCtXrmTEiBHMnTuXoKAgwNfN6M033yQ8\nPJxx48bxz3/+k9TUVN5+++1Su0Odz3333ccjjzzCsmXLGDFiBLt27WL+/Pn+gTsmTZrE3XffzeLF\nixk2bBgrVqxgwoQJjBgxosxt1K1b1///obRngsCXuM6ePZuMjAxGjBjBtGnTSE5O5oknnuDw4cP+\n55nuvfde0tPTGTZsGB6PJ2Af4eHhLF26lOTkZB5//HEmTJhAdHQ077777m92yxJCCHHhXInX6rJ4\n4oknGDhwIIsWLeLee+/l008/ZdKkSQwbNuwv73vixIm8/vrrjBs3jvj4eJYuXervsnzdddfx2muv\nceDAAUaOHMmzzz5L8+bNWbBgwW92jZ42bRqVK1dm/PjxTJgwgaKiIl577TXAN2gWwIABA7BYLAwf\nPpz09HTq16/PlClT+O677xg+fDjbtm1j2rRpvxt/9erVeeONN3A4HDz00EP84x//oFq1aixZskQe\nKRK/yaD+6hPhQgghhBBCiEvChg0bGDx4MJ9//jk1atS42OEIUeHkDq4QQgghhBBCiCvCRUlwf/zx\nR9q1a3fe9R9//DGdO3emcePG3Hffff7R64QQQgghhBBCiPOp0C7KSin+97//MW3aNDRNK3WS5j17\n9nDnnXeycOFCkpOTefrpp8nIyGD+/PkVFaYQQgghhBBCiMtQhd7BffXVV1myZMlvDjzz0Ucf0blz\nZxo1aoTVamXcuHF8/fXXchdXCCGEEEIIIcRvqtAEt1+/fixfvrzUybXPOnjwIHXq1PG/j46OJjIy\nkkOHDlVEiEIIIYQQQgghLlMVOg9ufHz879ax2+0lJtIODg72z2n2e5RSGAyGPxWfEEIIIcpfZmZB\nuezHYDAQGxtKdnYRl9MkEBJ3xZK4K5bEXfEu19jLM+5KlcLPu65CE9yysFqtOByOgDK73e6ft+v3\nZGcXYSyH+9JGo4GoqFDy8orQ9cvnH47EXbEk7oolcVe8yzX28ow7Jqbsc0aLC8to9H1BMhrB673Y\n0ZSdxF2xJO6KJXFXvMs19oqK+5JLcGvXrh3QHTknJ4f8/Hxq165dpu2VUuV6wnRd4fVePl/qzpK4\nK5bEXbEk7op3ucZ+ucYthBBCiD/nkktwe/TowcCBA+nXrx9paWnMmDGDDh06EB0dfbFDE0KIq5JS\nCqV0lO5F170opaN7vb5F96LrHnS3G93jRvf6Xns9HrxeN94zZV6vG+X1nKl/ZvGc2Zfu27e/nbNl\nKDj788w6UL56Z8rORHhmOfNaKcBAYsMOVK3T6KKcMyGEEEJcHJdEgjt58mQAnnrqKerXr8/TTz/N\nP/7xDzIzM2nWrBnPPvvsRY5QCCEqjlIKpbtRyoPudeFxO/G4XXjcLrweJx6nE6/LjtflwOt24nW7\n0L1uvF4PuteNrrtRuhfwAvovi0HHgA4GhdGoMBgUBoOOwagwGBVGg8Jg5Ez52QWMxr92B9TAmYuN\ndmapICf3fi4JrhBCCHGVuSgJbsuWLQPmwH3qqacC1nfr1o1u3bpVdFhCCPGHnU1Gdd2J8jrxehw4\n7cW4nA48Lgcetx2vq4hDyo2zuAjd60QpFyg3GDwYDB4MBi8Go47xzKJpZU8oNQNo5gt4gOVIKVDK\n4P8JZ19z5rXBdwMWfnntX3d2mzNlv1r36/e6DpmFsRfhKIUQQghxMV0Sd3CFEOJSoHtd6J4ivJ4i\nPK4inPYinPZC3K5iPK5idFcRutcOyoEBF0bNjaZ5fvcO59lx7yyW8onTqxvQvRperxGvbkTpBnTd\ngPKC0g0oHd973Zfo/fqnruNPAnUddOV7VlVXoCsDulJ41ZlyDCjAiwF1djEYz/lpxHfb14gyaGDQ\nUEYNg8GE0WQCUxCaUQOTCZMpCJPZhGYKQjOZCAoyo5mDMAWZMGkmgjQTmqahGQyYjCZMmkaQ0YhJ\n09CMRkyaEaPRgGYwYDT6Fs1owGg48/PMe4MBTCYjMTFh5OQUls9JF0IIIcRlQRJcIcQVTfe68LoL\n8LpteFwFOIpsOOw2PM5CvJ4ilKcIA3Y0oxOjpp93P6az/ynjp6auG/B4NN/i1fC6jXg9RnSPAd0N\nuhu8HtC9Ct2r8Oq+5049usKjFG5d4VbgUgqPMuDUDTgVOHUjLoMRt0HDjYbXGAQmKwaTBWNQMJrZ\ngiXIjEUzYw0yExxkISTITLD5zGIxYQ4yYjZpmE1GgkxGzEGa76fJSJBJO7Pel1CemyjKYE1CCCGE\nuNRJgiuEuGzpuhuPMxevKx+3Kx97YS4uez4elw3lLcRAEZrRXeq2RsBoAIJK37dS4HabfIsnCI/L\niMdlQHcZ8DpBdyl0l8Lj1vF4Pbi9XlzKgwsPduXFYQSnZsBhMuIwmnAaLbjQcBmsODGjey3gDUJ5\nTFg1K6FBwYSYgwkLNRNiDSLEYiLUaiLaaiLEGkSo1USI1USIxfc62Goi2GwiyFQO86IJIYQQQlwh\nJMEVQlyylNJ9d16dubidebiKszmZnk9xYRbo+RgNjlK30+CXfsHncLs1nC4zLlcQLmcQHocBrwN0\nh44q1vE4PXjcHpxuF07cuM12nBYXxRYvxUFmio1WCrUQ7MZg3FhRmgVUEEqZUboZ5TGDNwiTUSMq\n2EJkmJnwYDNVwszEx4ZiMkBYcBDhIUFEhJgJDzETFhwkSaoQQgghRDmRBFcIcdHpXhceZzZuRxYu\neyb2wgzc9izQ8zAYSnYbNoJvaN4zlAKn04zdYcHpNOO0m/AWg7dIh0IPqtCF2+XEbXTiNtuwW50U\nhXgoCtUoslgpCA3FFhqK8lhRrgiU24pyWVBuC8ppBbvmbzAsOIhKERaiwyxEhf/yMyrMQnS4hagw\nX9JqMPwSoKYZpJuvEEIIIUQFkARXCFFhlPLidmThtp/GVXwae+FJ3I5MDKqoRF2D/z8+TmcQxXYr\nxXYrjiIznkKFbvOCzYXRVgSGYtyWbIpD7BSEKfLDNGxRGvZrw3Br4Xgc4dgLYtCdwShnMMoVjCqw\nggqctybEYiIuykpcbDBxkdYzi+91bKSVYIt8bAohhBBCXKrkm5oQ4oLQvS5cxSdw2U/hKj6Fo/Ak\nXne2bx7Wc5yTw+LxaBQWBVNYFEJRYTBuG+i5bkz5xViLbRiMp3AEF5Ef5iU3UiMvzkReooYrLJgQ\nLRqDqyquIiv5OWZcBVZUZijoJT/mTJqB+OgQEqqHkBBzzhIbQljweR7KFUIIIYS4CuzevZPHHhvH\n8uWfAbBnz26GD78HyznTQdx112AGDRqCzWZj+vRn2bzZNwVsmzZtGTt2AmFhYSX2a7PZeOKJCWzc\nuJHQ0DCGDBlGjx59yj1+SXCFEH+ZUjpuRyauouM4i47jKDyGx5V1ZoKZX5xNZj0eIwUFYdgKQiks\nDMadpzDkOLDk5xPqykFpR/CGO8iN0siJ0chNNJEboYHFQkJoMmZPJN6iMIpyrOQe1HA5TOQGpMo+\nmtFAQqUQqlUKo2qlUK6tFEaV2BDiIoMxGkvWF0IIIYS4WimlWLHiQ+bMmYmm/dLDbe/edFq1asPz\nz88qsc2sWf8GDCxbtgKlFI8/PoFFi+YxevTDJepOm/YM4eGhrFixir179zJu3EPUrFmb1NS0cj0O\nSXCFEH+YrrtxFR3DUXgEZ+ERnEUnQAWOVnw2fSy2W7DZwigoDKUo34Ke5cSUW0iYM48Q91GwFpId\nbSQr1kRmbRNZ0SYcFguR5jiqhV9LjKES4YWhhGWYOZzuJb3YU2pMIRYTNauEU71yuD+hrRIbKgM4\nCSGEEEKUwZIlC1mz5gsGDRrCm28u9pfv3ZtOnTpJpW4zadIUdF3HbDaTmZmB3V5MZGRUiXrFxcWs\nW/cVK1euxGKx0KBBKtdf35XPPlshCa4QouLpXhfOoqM4C4/gKDiCq/gEUHLwJ7fbRF5+uG/JC8Wb\n4SHYlkuEI4tYZzphliJOx5o4USOIU7FBZEWb8GpRhAaFUCO8GinhVbF4YrFlhXD0mJsfj+fjdHkB\nBTj97ZhNRqpXDiexSgSJVXw/46ODAwZ2EkIIIYS4FHm8Ojm20meCKAujZsCpG8jLL0b/ncErYyKs\nmLSy/bG/e/feDBo0hG3btgaU79uXjtlspn//Xni9Xjp1up7hw+/HbDZjMvnSyalT/8mnn35MYmIt\n+vTpV2Lfx44dRdNMVKtWjZycQgCqV6/B2rVflim2P0ISXCFECUop3PbTOAoOYLcdwFl4lNIS2sLC\nYLJzI8nNi6Qw24w5O48oewZRjsMkePI4HadxPMHEntggTsWacVitAMQHx1E7KpHrI2sSbazCiRPw\n08FcVh7Jxe4sAAoC2okMNVO3WhT1qkfRPPUawi0GUJLMCvFnLFiwgJkzZxIU9Mvz5vPnz6du3bpM\nmjSJb7/9lvDwcEaNGkX//v0B32fCjBkzeO+99/B6vfTu3ZvHHnssoAubEEKI3+fx6kya9y1Z+X8+\nwf0j4iKtTB3eqkxJblxcXKnlUVFRNGlyHb179yMnJ5snnpjIggVzGTlytL/OI49M5MEHH2HatKeZ\nNGk8c+bnSuAJAAAgAElEQVTMC9iH3e4IeIYXwGKx4nCU/3mQBFcIAYDudWC37cdh8yW1uqewRB1b\nQQg5uVFk50RSlBlEaH42UfbTVLPvwqIKyIgzc6S6xqbKQZyOjcGr+ZLQqmFVaB1Vm9pRidQIr86J\nUx5+2JfF8rU5ZOTtLdFOXKSV5OpRJFXzLfFRvruzMt2OEH/d7t27GTt2LEOHDg0of/DBBwkJCWHD\nhg2kp6czbNgw6tatS+PGjXnzzTf56quv+PDDDzEYDNx3330sXLiQYcOGXaSjEEIIUVGee26m/3XV\nqtcyaNBg5s59OSDBtVgsWCwW7r//QW69tTc2Wz4REZH+9VarFZfLGbBfp9NBcHBIuccrCa4QVzGP\nq4DC3D3Y8/bgKDwMKvAurcNpJjMrmsysaHIywwizZRNbfIKaxT8S4rZhqxTGvmqwoYqJU7Gx6GcS\n2khzOM1ikqgXU5d6MXUx6VZ+PJjNpo1ZvHbwB+xOb0A7wRaNetWjSU2MISUxhvjo8v+wE0L4/PTT\nT/TrF9h9rKioiC+++ML/bFTDhg3p0aMHH3zwAY0bN2b58uXcfffdxMfHA3Dfffcxe/ZsSXCFEOIP\nMmlGpg5v9Ze7KEdFhpKXX1SuXZRLY7PZWLJkIUOGDCMkJBQAl8uF2ey7Gzt27ChuuWUAbdu2B8Dj\ncaNpGlZrcMB+qlWrhsfj4cSJE1itEQAcPXqExMTEPx3b+UiCK8RVxuPMoyB/Nxn79lKUfzRgna4b\nyMmN9Ce1eraLuOLjJBR/Tz17BiokiOPXhPB1JThaJQ671feBaTQYqR1Zk7S4BtSPSaJKaGWKnR62\npmcy/6v97Dmah1cP/ACuXjmMxnXiSEmMIbFKxF/68BVClI3dbufQoUMsWbKE8ePHExERwdChQ2nQ\noAEmk+/ZqLMSExP5/PPPATh48CB16tQJWHfo0CGUUmV69t1gMGAsh1/xs6OfX26joEvcFUvirlgS\n9x+naRpV4kL/9PZGo4GoqFCCTaDr5d+jTTtzw0LTDERGhvP1119iMCjuv380J0+eYsmShfTufTOa\nZqBevXosWbKAtLQ0NE1jzpxZ3HhjN4KDA7sjh4eH0b7935g+fTrjxz/G/v0HWLVqJTNmzPa3V14k\nwRXiKuD1FFOcu5vi3B04i34OWOfxGMnMiuFURhwZGdGEFuZQqegoqUWbCXEX4IkO50hSKF/ER3Aq\nNghlNABBWDQzTWLr0TCuASmx9QgNCsHh8nU9/u9PO9hxMDsgqdWMBupVj6Jx3Uo0rhNHbKS1gs+C\nECIrK4vrrruO22+/nRdffJEff/yRESNGMHjwYKzWwN9Jq/WXZ6PsdnvA+uDgYHRdx+VylXimqjSx\nsaHlOghcVNSf/2J4MUncFUvirlgSd8W7ULGHh/seDYuJ8c1lO2/ePJ555hluuqkLVquV2267jZEj\nh2EwGHj00XE8//zz3HXXbRiNRm644QbGjRtHSEgIJ06coHv37qxYsYJrrrmG5557lilTptCrVzdC\nQkKYMOFR2rdvXe7xS4IrxBVK193Y89Ipyt2Bw3aAcweJcrlMnM6I5VRGHNlZEUQVniK+cB+1i3/G\n7HXgrRzH4bpRbIoLIjNKA4MOmAk2BdOkUipN4htSN7o2QUYTuq7YfTiHb3Yc5If9Wbjcv7Rj0ow0\nqhNLs+R40mrFEmKVjxwhLqZq1arxxhtv+N83a9aM3r17s2XLFpzOwGejHA4HISG+xwWsVmvAervd\njslkKlNyC5CdXVRud3CjokLJyyu6IHctLhSJu2JJ3BVL4q54Fzr2unVT+PTT1f7RjmNiEpgxY05A\nndzcIv/r++8fw/33j/G/dzh0HI5CrNYIVq/+GoCcnEKMxiBmz54dEPfZNv6os8l3aeTbphBXGJf9\nNIVZ31OUuwPl/eX5Dq/XyKmMWE6cjCczK5rI4gwqF6RTr/AIQboTQ0w0J5tXZ118ISfDPPim5TFh\n1sw0ikvhusqNqB+ThMno+9jIyrPzzY6jfLPjJDm2X774Gg0GGiRG07J+ZZomVSLYIh8zQlwqdu3a\nxfr16xk+fLi/zOl0UqVKFdxuNydOnOCaa64B4NChQ/5uybVr1+bQoUM0atTIv65WrVplblcphdf7\n+/XKStfVZTnQnMRdsSTuiiVxV7zLNfYLHbd88xTiCqB7XRTn7qQw+/szc9T6KAWZWdGcOBnPqYw4\ngh0FJOTvo27hIayeYoxhoRRcV5tvqrj5MTQPDHkAGDBQPzaJVgnNSIurj1kzA76h7Tf/dJp120/w\n0+Fczv1oqnVNBG1TE7iuXjwRIeaKPHwhRBmFhIQwZ84cqlevzg033MCmTZtYsWIFb7zxBgUFBUyf\nPp1nnnmGffv28fHHHzNvnm+ah169erFgwQJatWqFyWRi7ty59O7d+yIfjRBCCFGSJLhCXMY8zlwK\nMjdTmL0Npbv85cXFFn4+nsDPxxPwOk1Uzt9PU9tmwp3ZGAwGgurV5WhqHB+bf6ZYZZ3ZykBccCyt\nqzSnZUJToq1R/v3Zilys/eE4X247Tl7hL+2EBQfRJjWB9g2rULXS+buKCCEuDYmJicyaNYuZM2cy\nceJEKleuzLPPPktKSgpPP/00U6ZM4W9/+xshISGMHz/ef8f2jjvuICsri1tuuQW3203Pnj0ZPHjw\nRT4aIYQQoiRJcIW4zCilcBYeoSBzE/b8dH+5rhs4lRHLz8eqkJUdRZQ3j1pZ31G58DCa8qBFRuFq\n24YN17r53nMEyAMFmkGjaXxD2l7TgjpRtQIGgjl8ysbqLcfY9NNpPOd0JUlNjKFDo2toXDdORj8W\n4jLTqVMnOnXqVKI8KiqK2bNnl7qNpmmMHTuWsWPHXujwhBBCiL9EElwhLhNK6RTn7sKWsQG3/bS/\n3OE0c+RoFY4eq4LHHURC0WFaZH1NuCsHAGtSMtnX1ebTkJ85bt8PHt920cGRtLumJW2qtCTCHH5O\nO4pdh3L45Nsj7Dma5y+3mjXapVWh03XXkhAj89QKIYQQQohLjyS4QlzilO6hKOdHbKfX43Hl+svz\n88M4dKQqJ09VwmzQuTZ7J1Vzf8KsO0HTCG3VmsONqrDSvYtsxxaw+7arFVmTTtXb0qlea2x5dv9D\n/rqu2Lo3k082HuHI6QJ/OwkxIXS+7lrapCbIgFFCCCGEEOKSJt9WhbhE6bqboqzvsWVsxOu2+ctP\nZcRy8NC15OZFEK65qHd6A5VtBzGiYwwNJfzvXdiZFMrnOd9RWHDAv13DuBRuqPF3EiNroGkGTEYN\n8A0ctXHnKT7ZdJTTOcX++rWrRtC9VU0a1onFWI7zVwohhBBCCHGhSIIrxCVG6V4Ks78n/9TX6B7f\n3GBKwYlTlThwsBoFhWFEm+w0PPklcUVHMQBaRASR19/AjqQQVp74hoLTvu2MBiPNKzfh+hp/p0po\n5YB2vLpi485TLFt3kIxcu788tVYM3VvVIKlaVMDzuEIIIYQQQlzqJMEV4hLhe8Z2J3knv8LryjtT\nZuDY8XgOHKpGUXEIlSwO6pxcRXTRcQyAKTqGyK43srtOMAuPryPvSD7gGziq7TUtub7G34ixRv+q\nHcWWPZksX3+Yo6d+6YrcrF483VvVoEZCOEIIIYQQQlyOJMEV4iJTSmHP30v+yTW4HZlnyuD4iXj2\nHqiB3R5MjNVN8ukviC44BoAWGUVMj14cTIri9SOfk33QN6CU0WCkVUIzbqzZmdjg6BJt7TmSy3++\n3M/hcxLbxnXi6NM+keqVJbEVQgghhLja7d69k8ceG8fy5Z8BsGfPboYPvweLxeKvc9ddgxk0aAg2\nm43p059l8+ZNALRp05axYycQFnb+6SPz8/MYPHgQzz03nVq16pR7/JLgCnERuYpPkXt8Jc7CI/6y\nU6djSd9Xk8KiUKKCFck53xCTsx8DYAwJIeam7uQ1S2bh0ZUcTPdtZ8BAs8pN6JbYhfiQuBLtnM4t\n5j9r9rNtX5a/rHHdSvRqW4OaCREX/DiFEEIIIcSlTSnFihUfMmfOTDRN85fv3ZtOq1ZteP75WSW2\nmTXr34CBZctWoJTi8ccnsGjRPEaPfrjUNrZs2cKkSf/g5MnjF+owJMEV4mLwuovIP/klhdnf+8ty\ncqP4Kb0mefkRhIUYaeTcTuz+rRgAg9lMVOfrMXRsw/KTa9myY55/uwaxyfSt3Z1rwhJKtFPscPPh\n+sOs3noMr+4bLblGQji3d65D26bVyckp9I+iLIQQQgghLjyP7iHXkf+nt9c0cJnt5BcX4fX+dt1o\nayQmY9lSviVLFrJmzRcMGjSEN99c7C/fuzedOnWSSt1m0qQp6LqO2WwmMzMDu72YyMioUutu376N\nxx+fyP33P8hTT00uU0x/hiS4QlQgpXspyPqO/FNrUV4nAHZHCDt3J5KRGUNQkEYD83Hid3yBpnyf\nWOGt2xDVuw9rCraz6seXceu+iWyrhFbm5jo9aBCbXKIdXVes3X6C99cdpNDuBiAqzEy/v9WmdWoC\nQSZjBR2xEEIIIYQ4y6N7eOrbf5PtyP39yuUg1hrN5Fbjy5Tkdu/em0GDhrBt29aA8n370jGbzfTv\n3wuv10unTtczfPj9mM1mTCbffqdO/SeffvoxiYm16NOnX6n7T0yszerVqyku9kiCK8SVwFF4lNyf\nV/ifs/XqJvbuq8bhI1XRlZHESCfX7vwIs9M3ArKlZiLxt9/J4SiduXsXkWnPBiAsKJQetbrSpkpz\nNKNWop0jpwpYsjKdQyd9UwuZTUZubFmdm1rWwGIuWV8IIYQQQoi4uJKPuQFERUXRpMl19O7dj5yc\nbJ54YiILFsxl5MjR/jqPPDKRBx98hGnTnmbSpPHMmTOvxH4iIiKwWq0UFxdesGMASXCFuOB0j528\nE6v93ZGVguMnq/BTeg1cLjPxsUHU+fkrgvfvA0CLjCTu5v7oTVN468AKvj/yI+B7zvbv17ale63r\nCTYFl2jH7vTw/rqDrP7+GOpMr+MW9eO5tWMdYiKsFXOwQgghhBDivExGE5Nbjf/LXZQjo0LJzyvf\nLsrn89xzM/2vq1a9lkGDBjN37ssBCa7FYsFisXD//Q9y6629sdnyiYiI/Evt/lmS4ApxgSilKM7d\nRe7xleieIgCK7OH8sL02efkRWCwaTSJOEr3pEwwABgNRnbsQ06sP32T/wEebZ+A40425ZkR1BiT3\npVp41VLb2ZKeyVtf7CW/0AVA5ehgBnZNJqVmTEUdrhBCCCGEKAOT0USlkNg/vb2mGYgJC8PsCr7g\nY6nYbDaWLFnIkCHDCAkJBcDlcmE2+0ZUHjt2FLfcMoC2bdsD4PG40TQNq7XkzZiKIgmuEBeAx2Uj\n5+ePcdj2A6Arjb37qnPwcFWUMlIrwUj1Hz9As/m6HVuqVafy3YOxxYfx4k+LOZB/GIAQUzC9a99E\nm2taYDSUfG42v8jFGyvT2brX1+3ZpBnp3roG3VpVJ8gk3ZGFEEIIIcSfFxYWxrp1X6KUYuTI0Zw6\ndZIlSxbSq1dfAJKS6rF48QIaNEhF0zTmzJlF167dMJvNFy3mCk1wd+/ezeTJk9m/fz81atTgn//8\nJ40bNy5R77333uPVV18lLy+PunXr8vjjj5OamlqRoQrxpyilKMr5kdzjn/kHkcrNj2Pb9kTs9mAi\noyykuPcQ/M1awDc6cmzvvkR27sLaExv5cPNn/kGkmlduQr+6PQk3l5xHTCnFpp9O8+bneyly+OrX\nrxHNoK7JVI4JqaCjFUIIIYQQVzKj0chzz81k1qwX6N69MxaLld69b6Z//9sBGDr0Pv7v/2YzaNBt\nGI1G/va3jowc+SAAp06d4q67+rN06XskJJSc7eNCqbAE1+l0MmLECEaMGEH//v1Zvnw5I0eO5Isv\nviA0NNRfb8+ePbzwwgu888471KhRg9dee42HHnqI1atXV1SoQvwpXnchOUc/xm7bC4CuzPy4I5Hj\nJ+MxGAykJJpJ+PYdKPA9cxGSkkrlu+4mJ1gx84d5HLL55rSNNIdze71+pMU1KLWd/EInSz/fy/dn\n7tpazRoDOtelfcMqGAyGCjhSIYQQQghxpWratBkrVvySeyUm1mL27P8rta7ZbGbMmPGMGTO+xLqE\nhARWrfq61O2++WZL+QRbigpLcL/99luMRiN33HEHALfccguLFy9m7dq1dOvWzV/vyJEj6LqO1+tF\nKYXRaMRqlQFyxKWtMGcn2Uc+QffaAcjNr8TWbbVwOi1ERltppPYStGoNAAaLhUq33k5E+w6sO7GR\nD3au8N+1bZlwHbfU7UlIUOl3YTf/dJqlK9P9d21TE2O4+8Z6xEbK74gQQgghhBAVluAeOnSI2rVr\nB5QlJiZy8ODBgLJ27dpRs2ZNunfvjqZphIaGsmTJkooKU4g/RPc6ObTjI3JOnh0hOYgdu2vx87F4\nwECD2iFcs/ldVK7vWdvgpGQqDx6KMzKEV3e8zq7sPQBEmiO4o14/UuPql9qO3enhrVV7Wb/zlG8/\nFo3bOsldWyGEEEIIIc5VYQlucXExwcGBo2lZrVYcDkdAmdPppE6dOkyePJmkpCTmz5/PAw88wIoV\nK8p0J9dgMGAsORbPH2Y0GgJ+Xi4k7orjLDpBxsH/4nH6JuouLK7Epu8ScTisRERauS4qA+Pni1FK\nYQgKIv6W/kR3uZ5dOXtZvPlVCly+OcCaVm7InfX7EXqeu7YHjufz6vJdZOT67g43qBnNvT0bEPsX\npv65HM83SNwXw+Ua++UatxBCCCH+mgpLcIODg0sksw6Hg5CQwC/1c+bMISEhgbS0NABGjRrFf/7z\nHzZs2ECnTp1+t53Y2NByvaMVFRX6+5UuQRL3haOUzukj6zix71NQOmBk/+HapKcnAAbqN4ilVvqn\nOLbuBiCkZg2SHxmLqWoCb2xfxmf7vgLAYrIwtOlt/K1mq1L/zXp1xX/X7OWtlenoukIzGrjrpvr0\n/XudcvvSfjmc79JI3BXvco39co1bCCGEEH9OhSW4tWrV4o033ggoO3ToED169AgoO3HiRMCdXoPB\ngKZpaFrZpjzJzi4qtzu4UVGh5OUVoesXdn6p8iRxX1gedyFZhz7Abjvge+8N59vNdci3hRNk1miR\nZCL481dwFBcDEN2pM/EDBnDYlcf8z6ZyvNDXxbhmRDWGpN1BfEgcublFJdrJsTmYu3wXe47mAZAQ\nE8KIPikkVokgL69k/T/qcjnfvyZxV7zLNfbyjDsmpuRI5kIIIYS4NFVYgtu6dWtcLhdLly5lwIAB\nLF++nKysLNq1axdQ7+9//zszZ86kW7duJCcns3TpUrxeL9ddd12Z2lFK4fWWX9y6ri74BMoXgsRd\n/hwFh8k6/D90jy/BzMqtypatNfB6TVRKCKOJSsf7wSoUYAwJJWHwEMKaXMeW09t5c897OL0uDBjo\nWqMj3RKvRzNqpR7rrkM5zP1wF4V2NwAdGlXh9s5JWMyl1/8rLuXz/Vsk7op3ucZ+ucYthBBCiD+n\nwhJcs9nM/PnzefLJJ5kxYwY1atTglVdeISQkhMmTJwPw1FNPcdttt2Gz2Rg9ejQ2m4369evz2muv\nERYmf0EXF4dSioKMjeSdWA0oIIhde+pw+EglAFq0qELshndxHfYNmBaclEzCvcMhKpL/7P2Atcc2\nABBuDmNwgztIjqlTaju6rvhow2E+/OYQCt9AUoNvqk+zevEVcJRCCCGEEEJc/ioswQWoV68e77zz\nTonyp556yv/aYDAwfPhwhg8fXpGhCVEq3esk++iH2PN+AsCjR/LNhroUFYVgtmi0axRC0Ecv4Srw\nDRgV06Mnsb36kuPMZ8HWVzhS8DMAdaISGZJyJ5GWiFLbsRW7mP/RbnYdygGgenwYI/umUjm69IGn\nhBBCCCGEECVVaIIrxOXEZc8g69B7eJy+KX7yCqry7aaaeL0asfGhtIw8hfPthXiUwhgSQsK9wwlr\n2JidWT+xePc7FHt8ox7fUKMjPRJvQDOW/hz5/mP5vLJ8J7kFTsDXJfmOLkmYg8r23LkQQgghhBDl\nZffunTz22DiWL/8MgD17djN8+D1YLBZ/nbvuGsygQUOw2WxMn/4smzdvAqBNm7aMHTuh1N63GRmn\nefzxR/nuuy2YTCY6duzMqFFjMJvN5Rq/JLhClKIodyc5Rz9C6W58oyTXJT3dN7dtUv046h7+AseG\nHwAITUwkYcT9GGJi+fTQF3x86HMAQkzBDGpwG2lxDUptQynFF1uO8Z8v9+PVFWaTkbu6JtM2rUoF\nHaUQQgghhBA+SilWrPiQOXNmBgzwu3dvOq1ateH552eV2GbWrH8DBpYtW4FSiscfn8CiRfMYPfrh\nEnWffPIJGjSox4cffkp+vo3HHhvH66+/xvDh95frcUiCK8Q5lFLkn1yD7fR633tC2bwliazscDTN\nQKtmcYStfB1HViYAke3aU//BkZzOL2DRzjfYnrkTgOrhVbk39S5ig2NKbcft8bL4s3Q27PSNqlw5\nJoRRfVK5Nl6eNRdCCCGEEBVvyZKFrFnzBYMGDeHNNxf7y/fuTadOnaRSt5k0aQq6rmM2m8nMzMBu\nLyYyMqpEPbfbTXCwlZEjR6JpFmJj47jhhhtZt+6rcj8OSXCFOEP3usg+8j72/HQAXN7KrF2XiMtl\nJjTcwt9STLjeewmP04HBZKLSHQOJ+fvfyXTl8/zm/+NE0WkAWiQ05fbkfpi1oFLbyS1wMmfZDg6d\ntAHQNKkSQ7vXJ9giv45CCCGEEFc65fHgzsn509trGtidhbjyi3539pigmBgMprJ9x+zevTeDBg1h\n27atAeX79qVjNpvp378XXq+XTp2uZ/jw+zGbzZjO7Hvq1H/y6acfk5hYiz59+pWMIyiI6dNfJCYm\njJwc39g169d/TZ06dcsU2x8h36iFADyufDIPvoPb7ktS8wpqsmHjtShlJL5KOK2iTlH05n9BKbTI\nKK4Z9SDBtWrxU85eFux4kyK3HaPBSN863el4bTsMBkOp7Rw4ns+c93eQX+gCoE+7RHq0rYnxPPWF\nEEIIIcSVQ3k8HHp8Ip6srAppzxQXR+Iz08qU5MbFxZVaHhUVRZMm19G7dz9ycrJ54omJLFgwl5Ej\nR/vrPPLIRB588BGmTXuaSZPGM2fOvPO2o5Ri1qx/c+TIYSZPfvqPH9TvkARXXPWcRT+TefA/Z+a3\nNXDkeD127owDDNStX4l6p7+h6KONAFhqJnLNqAcxRUXxxdG1fLD/ExSK0KAQhqTcSb2Y8/8V6psf\nT7Jk5R48XoXFrDGsRwOaJlWqmIMUQgghhBDiT3juuZn+11WrXsugQYOZO/flgATXYrFgsVi4//4H\nufXW3ths+URERJbYl8Ph4B//mMD+/fuYM2ce0dGlP873V0iCK65qRTk/kn30I1BeMJjZvrMBx475\npvJp3jKBuPXvUnz4EADhLVpS+Z6heDR4fffbbDntG2SqemRVhqXeRYyl9F9Qr67z7pr9fLHlGACV\noqyM7teQayvJ87ZCCCGEEFcTg8lE4jPT/nIX5cjIUPLLuYtyaWw2G0uWLGTIkGGEhIQC4HK5MJt9\nIyqPHTuKW24ZQNu27QHweNxomobVGlxiX/n5+Qwf/hBms4W5cxeVmgCXB0lwxVWp5GBSEXyzMRlb\nfjCmICMdWsZi+uBVXHl5AMT27UdMtx7kOfOZu30xPxccB6Bp5YaMaTeEYpsbr1eVaKfQ7uaVD3by\n05FcABrUjGZE71TCgkt/PlcIIYQQQlzZDCYT5vj4P729phkIjgnDbiks9ftneQoLC2Pdui9RSjFy\n5GhOnTrJkiUL6dWrLwBJSfVYvHgBDRqkomkac+bMomvXbiWm/lFK8dhj44mLi+Opp57FYLhwaagk\nuOKq8+vBpJyeeNauq43bHUR4hIUO9cD55iy8bjcGi4WEocMJb3odh/KPMm/HYmyuAgwY6FGrK91q\ndcJqslCMu0Q7xzILeel/P5KZ5wDg+mbVuLVTbTSjsUKPVwghhBBCiD/DaDTy3HMzmTXrBbp374zF\nYqV375vp3/92AIYOvY//+7/ZDBp0G0ajkb/9rSMjRz4IwKlTp7jrrv4sXfoemZmn2bZtKxaLhRtu\n6Ojff1JSPV5+eX65xiwJrriqeFx5ZB54F7fDN5hUbkFNNp4ZTCrh2ghaWA9T9NaHAJhiY6n6wBgs\n1aqx+dT3vLnnv3h0DxbNzOCUO0iLa3DewaS2pmfy2se7cbq9mDQDg7rWo11Dmd9WCCGEEEJc2po2\nbcaKFav97xMTazF79v+VWtdsNjNmzHjGjPl/9u47TKrybPz495zpM1tme2cLu0sHQZCOwY5SDKJY\nURRFLIkmmhjNmzdF3yS/JMbEboIFuwYNFhALKtI7LizsAtv7zu5O7zPn98dslaVIX3k+18Xl7MyZ\nc+4ZZ3fOfZ77uZ8HD3osNTWVzz77pvP2+vVbO7son8yRZ5HgCmeN7zaTqqgZwO7dSYDEgCFJFFZ+\niWvHFgAMBYWkLboHOTqKZQdW8GnllwAk6OO5c/gtpEel9nqMsKLw4doKlq2JzNuNjdJyz+xh9E8/\nOXMMBEEQBEEQBEHoIhJc4azw3WZSO4oGUVsbiyTBeWNTifvqddw11QDETJpM8g3z8EshXi5aQpGl\nGIB8cy63D51HlNbU6zE8viCLP97DttJmAPLSY7j7x8OIi9admhcpCIIgCIIgCGc5keAKP2iKomCr\n+wJ70zoAwkSzdt0A7HYjWp2KKaPNyO89TcDhAEki6ZprMV90Ca3eNp779mXqXA0ATEg7j7kDrkQt\n9/4r09Tm4Yl3d1Lb7AJg4tBU5l02AI1adWpeqCAIgiAIgiAIIsEVfrgO10wqxqxncp4f72tPEAqF\nkA0G0hbehWnoMPZby/lX0RKcARcSElcVzOBHmRMPOd92Z2kzf1yyCZcniCTB3Kn5XDwm65DbC4Ig\nCMLZKui3Iss6ZPXBS4gci2qnlzcP1DM8PprLshJPyD4FQejbRIIr/CB9t5lUqy2HDRsjzaTS+8Uy\nirzGMBkAACAASURBVBI873wCgCY5hYx7f4o2LZ11dZt5q+Q9QkoIg1rPbUNuZFBCYa/HUBSFlZuq\neeuL/YTDCia9mjuvHMqQnBO/YLUgCIIgnA5ub4A2h48Yo5YaRx3ekI98cy5KMEjLxx9iyC/ANGQo\nAB63n68/KcUcb2Ts+bkHXej1e5poKHkBlTqKtEGLkFXHP4VnVV0LVn+Q1Q1tjE+JJVYrluEThLOd\nSHCFHxyfs4rm8nc7m0mVVw+guDjSTGrQsCTySj7BU1wEgHHQENIWLkIyGVm670NWVUc6vSUbErlz\n+C2kmHpfoywQDLHkkxLW7oqUMGckmbh39jCS44yn4iUKgiAIwkkXDiv89PGvaGr1cO81A3i56hn8\nIT/3nLOAtKJaWj9cBkDeX/+O2hzHvuImykstAOQWJpKSHtNjf177flDChAJ2XK3fEp005rhjDCld\nnVi3Wxz8KF1cZBaEs51YkFP4QXG27KBx/5L25FbL9m+HU1ycjCRJjB+XQvb6V/G2J7fmCy4i476f\n4dereHbnS53J7YC4fB4Yfc8hk9s2h48/vb69M7kdPyyN/7l5tEhuBUHoUywWC+PHj+fLLyNd4ouK\nihg0aBAjR47s/Pfcc88BkYqVv/3tb4wbN44xY8bw6KOPEgqFTmf4winQ0OqmocVNWFF4d8N2/CE/\nACvKv8BXVdW5nfXrrwBwOXyd9xXvqDtof4oS7LzttGxDUY5/mRB/t6VGtrXYT8g+BUHo28QIrvCD\noChhrLWf42jeAEBYieabdQNwOo3o9GqmnGOE954k4HaDSkXy9TdhPv9H1LsaeaHoFZrckSvO52dO\n4Kr8Gajk3ptDHai18dT7RdickS/5KyfnMn/mMKxW10ldz0sQBOFEe+SRR7BarZ0/79mzhylTpvD8\n888ftO3rr7/OV199xQcffIAkSSxcuJAXX3yR22+//VSGLJxiLm+g83aD1YkuJXL7gK0ctyuz8zHb\n11+ScMUM/L6uBHb/niYmXpiPVtd1qqmEuy6KBLyN+N116EwZxxVjoFtCa/EGqHZ5yY0VF5wF4Wwm\nRnCFPi8c8tJ84M3O5NbjT+HzL4fidBqJSzByUYGH8BtPEXa7kaOiyPzZg5jP/xE7mor4y5YnaXJb\nUEkqrh3wY64pvLLX5FZRFL7cXsuf39iGzelHp1Fx94+H8eMpeciyaCYlCELf8uabb2IwGEhLS+u8\nr7i4mIEDB/a6/bJly7j55ptJTk4mKSmJhQsX8v7775+qcIXTJNj9wq0U7vFYi7W+83bIbsexdQvh\nbtsHA2EO7G3uuUOl56i/07L1uGMMf2fEdkOj7bj3KQhC3yZGcIU+LeBtobnsbYK+yAhsc2sOm7dE\nmkn1yzUz3LUdz/tfA6DNyCTjnp+iSkzggwOfsLJyFQAx2mgWDL2J/uacXo/h84dYsnIv63dHGlYl\nmfXcO3s4mclRJ/8FCoIgnGDl5eW89NJLvPPOO8yePbvz/j179qDVarngggsIh8NMmzaN+++/H61W\nS1lZGfn5+Z3b5ubmUl5ejqIoR9UxXpIk5BNwSb3jgmJfu7DYV+MOh7uSR0numeA6Ha10n2Fr+/Jz\nwkOv6rHNrq21DBmZ1vUZ+U6S7LbuJrHfpchq/bHH2J7gJhu0NHn8fNvqYJo/iXj63vvdVz8nIu5T\n72THvnv3Lh566Od8+OFKAPbsKWbBgpvR6boaw82bdyu33HIrdrudv/zlj2zcGBlomjhxEj//+S+I\nioo+aL/79+/jH//4K8XFezCZTFx55Wzmz19wwlceEQmu0Gd57AdoqVhKOOQFZPaVDaB0XxIAw4cn\nkLHlP3iqKgEwjTiHtNsX4lUpvLDzJYpbI0sH5cZks2DYjZh1sb0eo6HVzdPvF3Wub3tOfiK3TR+E\nSS+6NAqC0PcEg0F+8Ytf8Mgjj2A2m3s8FhcXx9ixY5k7dy4tLS389Kc/5Z///CcPPPAAHo8Hvb4r\nCTEYDITDYfx+f48TnkNJSDCd0BMYs9l0wvZ1KvW1uPUNzq4fpK5kVyXJ0D4HO6qgAOe+fXj274f+\nHgASkky0NLuwNDlpaXRTODhS2+xsjFzlMJmzcdtrUcIBgq5dpOb+6NiDbD/BPz87iZVljdj9QTa0\nOMhLNfe597uDiPvU6qtxw4mPXVEUli5dyp/+9CdUKhXx8ZHBnNraikNOYfm///stOp2G1au/RlEU\nfvKTn/D66y/zq1/9qsd24XCYX/7yZ9xyyy28+uqrNDQ0cM011zBy5HAuvPDCE/o6RIIr9DmKomBv\nXIut/ktAQUHH5q0DabbEolJJjB9uwrD8OfwuF0gSCTNmET99JtWuOhZvew2LtxWASeljmVM4C43c\n+6/Blr1NvLh8D15/CEmC2VPymDYuG1msbysIQh/1zDPPMGjQIM4///yDHutoKAVgNBpZuHAhjz/+\nOA888AB6vR6fr6uBkMfjQa1WH1VyC9DS4jphI7hmswmr1dVjdPFM11fjttkjCatWI5OeYqQRkIJ6\nzkkfgCocaU6mGzIEv92Ov7ERZ00dEEVqZiw6g4a6KitffrKXhBQjkiTh9XgBUDASlTACR/NW6stW\noTYNPeZRXH8wMioc8gWYkGLmk2oLq6ssXJybjMYf7FPvd1/9nIi4v79QKIzT7jvyhocgSRATY8Ru\nd3OkvmpRMTpUqqP7A/zyy4v54ovPmDfvVl577WVaWyMXubZv30lOTv/On7v7xS8eIRxW8HrDNDU1\nYbc70OlMvW77xhvvkpqagNXqorKylmAwhCxre932SDqS796IBFfoU8IhLy2Vy/DYIiOw/kAsa9YX\n4vEYiIrWMS6pBeU/LxNWFGSjkdQFCzENG87Xtet4f99HBJUQaknFNQOuZGL62F6P4QuEeGfVfr7c\nXgtAjFHDwplDGCTWtxUEoY9bvnw5zc3NLF++HACn08nPfvYz7rzzTqxWK3fffTdRUZGTBp/P15nA\n9u/fn/LyckaMGAFEypzz8vKO+riKonAimy6Hw0qfbOzX1+IOtCePapXMsP5xNDZGBm7j/QORlEiC\nW+Nqot/Ui2h+63V8rVYwRSFJMGp8P+qqrDTW2tlX3Ez/gUmEO5tMyUQnT8Zp2Uk45KGtYR3mtKnH\nFGNHibKExLikWNY2tOEIhFi6t5ZrcpL71Pvdoa99TjqIuI9OKBTmzRc24bB5T8nxomP1XHfHeUeV\n5E6bNpMbb5zP9u2R+fEd70tJSQlarZbZs2cQCoW44IKLueOOu9BqtUiSGpUK/vCH37JixUfk5uYx\na9bsXt9TnU6PJEnMnj2TurpaLrlkGoMHDz/h779oMiX0GX5PIw0l/+5Mbi2tGXzx1VA8HgMZWdFM\nDG5G+fQ/oCjosrLo9z+/RR5UyL93vcq7pcsIKiES9HH87Ny7DpncVjc5+cMrWzqT2/zMWP53/nki\nuRUE4Qfhk08+YevWrWzZsoUtW7aQnp7O448/zu23385nn33GU089RSAQoLKykueee65zju7MmTNZ\nvHgxDQ0NWCwWnn/+eWbNmnWaX41wsgVDHQmuRFyMNnKnIrN2gxejFPl5l7UUw6QJqBMTUaSueYGZ\nOXFk5sQBsP7LAwSDIZT2JlOSrEKtjSGqfR1cR+N6gr62Y4qxYx1cGdCqZC7OSABga4OVAzb3Me1T\nEM5WiYmJvU4nMZvNTJw4mSVL3ubJJ59n27YtLF7cs1z55z9/iBUrvqRfvxwefvjBwx7njTfe5e23\n/0tJyR5eeulfJ/Q1gBjBFfoIV+u3tFZ9hKIEUZAp2ZfPgbIUQGJ4oZGUta8StEWWu4geP4GUG2+m\nytfIi5tfpKW9JPmcpKHcMPBqjBrDQfsPKwqfb6nhP1/tJxhSkCSYMSGHGRNzUJ2IujpBEIQzmCzL\nPPfcczz66KOMGzcOvV7P3LlzufnmmwG4/vrrsVgszJkzh0AgwIwZM5g/f/5pjlo42Tq6KKtlmWDH\nGrZhmRa7D1VAC7hxhb18WvsNU+fMRflgf+R5liYkqYCJF+bzzoubcdi8bFtXRW5W+wiuFFmtIDZl\nMq7WbwkHXbRWLyep//Xfe652x8CPqv15oxJj2Nhso9bl4z9ljfxkSD+0R1meKQingkolc90d5x1X\nibJKJREba8Rmcx9x9PP7lCgfyp///PfO2xkZmcybN5/nn3+aRYvu7bxfp9Oh0+m4666fcM01s7Db\nbcTE9N7jRqfTkZGRyfXXz+Odd97gttsWHld83yUSXOGMFg75aK1ejrutCIBgyMDGzQOw2mLQ6lSc\nl9CKbsUrhBUFSasl+fobMY2fwPLKVaysXEVYCaOWVMwumMGUjPG9fnFarB5e/mQvxRWRq8cJMXpu\nnzGYwizzQdsKgiD8kKxatarzdn5+Pi+//HKv26lUKu6//37uv//+UxSZcCboGMFVqSRC7eXFMUY9\nzYDPE8QAhGX4rOorRoy6G4wNAHh2FRFyDCc+KZqh52ZQtKWWbesrSU+MnNBL7QmurNYTl3kZLRVL\n8ToO4G4rwhQ//HvF2FGi3NEfQ5Yk5uSl8NSuKlp9AT6rbeGKfknH+1YIwgmlUsnExh084HL0z5eI\njzeBfPLLq+12O0uWvMitt96O0RhpauX3+9FqI1NY7r//bubMuZaJEycDEAwGUKlU6PU9X19bWxsL\nF97C+++/R0cKGggEeu22fLzEJS3hjOVz1dKw94XO5LbNlsCXq0dgtcWQnGxgomc9utXvd5YkZ//P\nb3GPHMBftz3NiorPCSthko2J/Hz03ZyfOeGg5DasKHyxtYb/WbypM7k9b1Ayv7t1jEhuBUEQhLNe\nKNQ1BzfYXl6cEG0gOc6ATOQxky6KsBLmtb3vokpsTyR9HhpffxWAsVPyiI0zoCjQ0uQAuhJcAKN5\nMPqYyBJUrdXLv3epckeC232AKt2k57L+qQCsa7SyX5QqC8Ixi4qKYvXqL1m8+AWCwSA1NdUsWfIi\nV1wxA4DCwoG88spi2trasNvtPPXUE1x66eVotdoe+4mLiyMuLo6///3v7VNhKnjjjSVMn37ip7uI\nBFc44yhKGFvDGhpLXyLob0NRZPaW5rFuw2ACAS1DUoMM3bwYuawYAPOFF5P+0COsDuzjT5v/QbUj\nMn/2/MyJ/GrMffSLzjzoGPUtLv78+jZe/6wUXyBEtFHDnbOGsHDmEIxiCSBBEARB6CxRVqnkzhFc\ntaxm/rSByEp7guvPR0KiztVAWyDSCVUmjHPLJmzffI1Gq2Lq5QMACAUDkR1LXaefkiSR0G8GstqI\nEvZjqVjaOVf3SBRFOahEucMV/VNJM+pQgLfLGrD7g8f0HgjC2U6WZf7857+zf/8+rrjiQu66awFT\np17E1VdfB8Btty1k8OAhzJs3l5tuuobU1DTuv/8XADQ0NHDxxZNpaIhUdzz22J9pbGzkiisu5sEH\nf8rcudczbdr0Ex6zKFEWzih+TzPN5cvwuyNJqtdnYvO2Quz2aEwmNcNd2zCu2QaAymwmZd4ttOQk\n8OLO56h21gEQpzNz46CrGRhfcND+Pb4gH62r4NPN1YTaW8KPG5zCdRcVEG3UHrS9IAiCIJytOr4n\n1bLUOQdXLasY0C+OYpUEISirhGHnjOVb5wZcfg96otGlpEBbEU2vv4quXzZp2TmMGt8PKbgDgDaL\nh7iMruOoNNEk9JtJc9lb+N11WGs/Jy7z0iPGF+52+7tL+GlUMjcWpPGPokpcwRBvlzVw64CMgxJh\nQRAONmrUaD7++IvOn3Nz8/jHP57pdVutVst99z3Iffcd3FgqNTWVzz77pvPnlJRUnnvuOVpbnSe1\ntFokuMIZQVHC1Jetou7Ap9B+5ba6NpXde/oTCqnIMfvJLnoXtc8FQMyEiURfNZuPG79h9ZY3UWhP\nVtNGM6dgBga14Tv7V9hQ3Mg7X+7H5vQDEBet48ZLChlZIObmCIIgCMJ3BbuVKHeM4KrkSHmxRo58\n84aR2fFNPDkTs5GUSPIonTsEdfN2gi0t1D37FNm//i1jJudQsiHy3Ir9VmLTnCQkd61jaYgtJDpp\nLI7mjTiaN6IxpBCVcM5h4wt3WwC0t8Q10aBldk4Kb5U1UO7wsLyqmRnZycf+hgiC0CeIBFc47Xyu\nWtpqluN310d+9uvZWZRPsyUeo15mkGMT5v27AFDFmkm+aR7fJgX4sOhpHP5IOVSKMZnrBvyYgrj+\nB+2/uKKVpV+XUV5vByLLHVx6Xj+mj89Bp1UdtL0gCIIgCD2XCeqYg6uW2k8d2xc21hu0+AJhmnYO\nJFuJXEBea9nG7Qvm0/a3JwhaLNQ98yQZ9z9AjFlLyAfBICz/TxGzbxqFKVrXeTxzxkX4PY34nBW0\nVn+MRpeALirrkPGFuiW4h5pzNzwhmiqXl3WNVtY32UjQa5mQIvpsCMIPmUhwhdMmFHRjrVuFq2Vb\n532V1WnsLcklGFKTrWkhp/gT1OEASBLmCy6iefJQ/ln7BXUlkVp+tazmsuwLuSj7fDRyz4/zgVob\n760uY09lV8OK4f0TuO6iAlLijKfmRQqCIAhCH9XbHNyOEVylPcGdPimP4i1+2trC5Kr0ALjCLhZb\nv2DBjTfR8vJLeEpLaHzp38iTIQQgqXDafSx/t4hZN5yDVhf5/pYkFYm5c2gsWUzQ30Zz+dukFMxH\no0/oNb5wtwrHw5UeT8tKpMXrp8Tm5uOqZhJ0GgaYTcfz1giCcAY76iZTXq/3ZMYhnEUUJYSjeTP1\nxU91JrdOl5ENm4ezq7gAkxzm3NpPyN/zIepwAH3/fPjpAt4c4OSZ0tepc0WS23OTR/CbsQ8wLffC\nzuRWURR2l7fyt7e289irWzuT29y0GH5+7Tncd/UIkdwKgiAIwlHo7KIsSwQ7mkxJKhRFgXDksZTE\naBZMHwxA+yYokkKVo5a3ovZjnhnpkOrYtJGgPbJefeGQNAAsTU4+XVbceRwAldpIUt61SCod4aCb\npv2vEfTbe4+v+wiufOgEVyVJXNs/jVSDFgV440A9lQ7P9307BEHoI446wZ0xYwZ79uw5mbEIP3CK\nouBq20X9nmdpq1lBOOQlGFRRvDeP1WtH4bDFMKB1M+fueROzpwF1XBzha2aw9JJ4/tG0jFLrAQDy\nYrN54Nx7uHXoDSQY4oFIGdX63Q387qXN/O3tHexuX/YnI8nEvbOH8et55zIkJ/60vXZBEARB6Gt6\njOC2N5lSyarO8mQASaVi9MBkrr0gn44UMyaQA0Bxawn/zbETPXESACFvZLmehJQYJl0cWRqouqyV\nz76T5GoMSSTlXYckqQkFbDQdeI1Q8OClfnrOwT38a9GpZOYVpBOrURMIK7y8r45alxi8EYQfoqMu\nUQ4EAgetI/p9FRcX85vf/Ib9+/eTnZ3N7373O8455+AGAlu2bOGxxx6joqKCzMxMHn74YcaPH39c\nxxZOH0VR8Nr3Ya3/ioCnof0+qK1PZm9pLgGflkx7CTmW7WjDPmSjEefkkXyW6aLCsxFskf1kRWcw\nLecihicO7vws1re4WL2zjrVFDTg9gc5j5qXHcNl5/RhVmHTYq7qCIAiCIPSu+xzcUPcR3G4JLqpI\nyfIl5/Wj7KtylLBCU3U0qYkjaNDsZKdlN7pzR3CRayRBuQUAd9Euhk0djccVYOu6SspLLXzx4R4u\nmjkIWY6Mveij+pGYezXNZW8T9Fpo2v8ayfk3olJ3VWGFjrJEuYNZp+G2gRm8sKcGZzDES6W1LBiQ\nSapRd8TnCoLQdxx1gjt9+nTmz5/PFVdcQVZWFnq9vsfjc+fOPezzfT4fd955J3feeSdXX301y5Yt\nY9GiRXz++eeYTF3zIBobG1m0aBGPPvool1xyCR9//DH33nsva9asOeiYwplNUcK423Zjb1xLwNvU\neX9jUzwl+3JwOE2kOQ6Q27IdQ9CFbNBjGTmE5dlOWqR90F49lB2TxeU5FzEkYSCSJGF3+dla2syG\n3Q3sq7F17lcCRuQnctnYfhRkxh73BRlBEARBOJsFO5YJUsn4lY45uGqUUNeaspKqq1mjJIFC5F/5\n9lQyRwRo0RWzybKT8KRhXOiLfGc7Nm9B40lk9GWXEwqF2bGxmgN7m5EkiQumD0SliiS5htgCErKv\npKXyfQKeBpr2LYkkuZpI9+XuI7jfXSboUBL1Wm4dkMG/9tbgDob5d0kN8wszyDCJc0xB+KE46gR3\n+fLlGAwGVq1addBjkiQdMcHdsGEDsixz/fXXAzBnzhxeeeUVvv76ay6//PLO7ZYtW8aECRO49NLI\n+mfTp08nNze384qecOYLB704W3fiaN5AyN+VgLa0xlKyLwdrWzQpjjKGtBVhCtgImwyUjO7Hqkw3\nfm0zABISwxMHc37mRArj+mNz+fl6Rx2b9zaxt6qNbt9pxEXrmDQsjcnD00g0G74bjiAIgiAIxyDU\nbQTXc4gRXEnV1QMj3D6kOjw/kdX7LdTszCJ1WAiboYQtrUVMMUejQYGwguW9/xByOhl71dWEQwrf\nbqlh/54m/L4gl/x4CBpNJHE2xQ8FiCS53iYa971Ccv5NqLUxPebgHqlEubtUo475AzJ4qaS2Pcmt\nZV5BOrnR4hxCEH4IjjrB7S2x/T7Ky8vp37/nEi65ubmUlZX1uG/37t2kpKRw9913s2XLFnJycnjk\nkUfQarVHdRxJkjgRuXBHWWtfK289XXErioLfXYe9eQuull0oStfV3YamBMrKM7G1msiwlzLYuht9\n0IUrMYqv88wU5WgIqb2AjEljZEL6GCalj6PVIvNtUStvHNhMdZOzx/G0GpkR+YlMGpbGsP7xqE7T\nBRDxOTm1RNynXl+Nva/GLQhnks45uLJM8DBzcCFyHtDhwnMziU4y8fH6ShqKckgZpMIeXQyEAQlt\nVjbe/cW0ffoJIaeD8Tfdgkots31DFVVlrXz01k4uv3oYOr0GiCS5kqzCUrGUoK+FxtIXSep/HSGl\na7mfox3B7ZBp0nP7wExeLKnFGQzxcmkt1/VPY6DoriwIFBfv4le/eoBlyz4BYO/eYu644xZ0uq5y\n/ptums+8ebdit9v529/+yKZNGwGYMGEi99//S6KionrdN0A4HObee+9kwIBB3HPPfSc8/u+1TFBL\nSwvvvvsuFRUVPPjgg2zcuJGCggIKCgqO+Fy3243B0PPKmF6vP6g7s81mY/Xq1Tz55JM88cQTvPPO\nO9xxxx2sXLmS2NjYIx4nIcF0QktTzX30D92pitvvtdHWsIOWum14nHWd94dCMnUNSZSVZxJqC5Jh\nL2WYfT8qyc++LB07C8zUJ2pAklBJMqNShpKtH0ywNZG9262sXLYLnz/U41hajYoxg1OYPCKDcwcm\no9edOatcic/JqSXiPvX6aux9NW5BOBOEDjkHt6shVMcc3HC3CbEqlcxV5/dHr1Wx9OsyGvf0w5wr\noTJXAPB5XpCp7tEENm7Bvm4tQZuNMQsXoTdoWP/lARpq7bz/6namzRlKbPvKB0bzIJJyr8FS/h9C\nATuNpS/hT5vTdcxjOPdLNepYOCiTxSW1WP1BXt1Xx+VZiUxIMYtpTsJZSVEUPv74A5566u+ouk0/\nKC0tYdy4Cfy///fEQc954om/ABLvvfcxiqLw61//kpdeeoF77/3ZIY/zxhuvsXPndgYMGHQyXsbR\nJ7jFxcXMmzeP/Px8du3axd13383atWv51a9+xXPPPXfEJlAGg+GgZNbr9WI09lyyRavVMmXKFCZN\ninTcu+GGG1i8eDHbtm1j6tSpR4yzpcV1wkZwzWYTVquLcPeF1s5wpyLuUNCNq20PDsu3+FxVdP8K\ncDoNVNakUVebiLmtngL7N8R5GqhL0vDNuXr29YvGq5ORkUlRZ2LwZeCsS2LdJj9rFStg7XGs9EQT\nw/snMCwvnsJ+ZrTqyC+b2+XF7TopL+97EZ+TU0vEfer11dhPZNzx8Ye+Ci0IP2Qdc3BVKplgjzm4\nB4/gdv89k9vrha8Yn4M5SsfLK/ZiK89ELqwEoCXg4JlCLwv045G+Xo979y6qHvsDg+/9KforBvL1\nihLaWtwsfWUbl1w5hMycOAAMsYUkF9xMc9lbhIMuWmo+Ay6KHPMY89EEvZaFgzJZsq+eerePj6st\nNHsDzOiXhEpUgAhnmSVLXmTVqs+ZN+9WXn/9lc77S0tLyM8v7PU5Dz/8v4TDYbRaLc3NTXg8bmJj\nzb1uC7B3714+/vgDpkz50YkOv9NRJ7h//OMfufnmm7n33nsZOXIkAI899hhxcXH89a9/ZenSpYd9\nfl5eHq+99lqP+8rLy5k+fXqP+3Jzc6mqqupxXzgc7lH6cjiKonSvnDlu4bBCKNR3Tuo6nMi4FUUh\n6LXgtpXibN1L0FuHJEX2LQGBgIqGxkRq6pJRat2kOCsY51xNqznMnkIdJdkJOKJUyIoGlSsZf00C\nIWsSrpCm/Qj+zmOlJ5rIz4ilIDOWQdlxxMf0bPpwpv6/EJ+TU0vEfer11dj7atyCcCbotYuyrIIe\nTaYiowo91rJVdY00TByWRmKsnmff39n1HDS4Q16eyiznhukTiV+xgUBjA1WP/Z7M2+9k+tzhrHx/\nNz5vkI/e3smEC/IZNjoDSZLQmTJILbyNprI3CHdbylZSgkDXiNP3EavVcMfATN4pa2CP1cWmZhvN\nXj9z81KJ0Z451WLCD4cSDhEM2I684SGEZQmf20vA6yZ0hIu4ak0sknx0vxtXXDGLefNuZfv2rT3u\n37evBK1Wy9VXzyQUCnHBBRdzxx13odVqUasjvyP/93+/Y8WKj8jNzePKK6/qdf9+v59f/vKXPPTQ\nr1m27P2jiulYHPVv7e7du3n00UcPun/u3LkHJa69GT9+PH6/n1dffZVrr72WZcuWYbFYOkdqO8ya\nNYu5c+fy1VdfMWXKFF5//XV8Ph9jx4492lCFEyAUcOB1VuFoKcfr2IeMo/MxSYqUIDc1x1NXl4i/\n2k+Co5ZC9w6akkLsHaBjRUY0Tr2asNNM2JpAuCqBsDOW7ksvxxg19EuJJjs1msIsM2OGpRPwN/Fu\n9wAAIABJREFU+sXJqCAIgiCcAboSXJlQxwjuQcsERU4le4zgfmfkc0C/OB668Rz8VasBcOwbjLbf\nAfwqG6/G7GPSlUMZs/IAYaeTun/+nbhLLuOqG6ez4r/FtFncrP1iP7VVbUy9fCB6gwa1zkxqwa3U\n7f+MjtMTy75XSM6bg0Z3bGve61QyN+Sn8Um1hTWNVsodHp7cXcXc/qnkxxiPvANBOEpKOETdnqcJ\n+a1H3vgwao5yO5XWTPqgu48qyU1MTOz1frPZzMiR5zJr1lW0trbwP//zEIsXP8+iRfd2bvPznz/E\nT37yc/70pz/w8MMP8tRTLxy0n2effYpJkyYxYsQ5Z0aCGxsbS11dHdnZ2T3u3717N/HxR/5jotVq\n+de//sVvf/tbHn/8cbKzs3n22WcxGo385je/AeD3v/89gwcP5tlnn+Wvf/0r999/P7m5uTz33HM9\nlhISTqyOEVqPswpHSxl+VzUquaupU0dK6vNpaGqOp6UxGqXSSZyjFrOuiPoUia0FGmrjYvH74iJJ\nbbWZsCMOwmqQwGjSEp2hwxSrw2TWY4jRIetkQgo0KApNXjcbd5QTDoUACRnQyjJ6lYxOFfmvQS0T\no1UTo1Fj1mqI0apQi+7agiAIgnBSdDSZUqtkgt1GcJVgLyXKoUMnuABJsVpq22+HfAZs28cQVbiH\nUEwta3T1VExL4qoNMVBdR9unn6ArLWH6LbezdnMbZSXNVOxr4d2XtnDRjEGkZZmR1XqiU6eCoz4S\nq6eBhr0vEJd5KTFJI4/p9cqSxOX9ksg06XmvohFXMMRLJbX8KC2eqenxqEXJsnCW+vOf/955OyMj\nk3nz5vP880/3SHB1Oh06nY677voJ11wzC7vdRkxMV/+krVs3s3XrZt57bylOp5+T6agT3Ouuu47f\n/OY3PPDAAwCUlJR0NoOaP3/+Ue1j4MCBvPXWWwfd//vf/77Hz5MmTTpoZFc4cZRwEL+7DkdrOc6W\nAyihRlSqQOfjHZVF4bCEzRZNa0s0nuowqvo2FM0B3GYnNfkaaqLj8PgHRRLaZjPUR6E2alEb1agT\nNaj7qVEbNaiMaqT2LwV3+z8CfggcFNr3IgMJeg0pBh0pBi3Z0Qb6mfRoVSLpFQThh6mtrY0XXniB\nXbt2EQgc/Ee0t+9YQThWHaWPKlkiFOoYwT3UHNyuEmW5t+9hpes5l4/P4+XPW3DuHYoqyYwuZy81\nGhdPTlCYW5ZP8ub9+CrKqf/j7xgz+2oyLxnE2lUHcNp9LHtjB8PHZDFmcg4dR5QBlcZIOOimtepD\nvPZSYqLmAseWkA5PiCbdpOON/fU0ePx8Wd/KXpuLObkppBl1R96BIByGJKtIH3T3cZUoq2QJs9mI\n1XpiS5R7Y7fbWbLkRW699XaMxsiAo9/vR6uN/C7cf//dzJlzLRMnTgYgGAygUqnQ63s2F/7880+p\nra1hwoQJKIqC1+tFlmWqqip6bV51PI46wb3jjjswmUz86U9/wuPxcM8995CYmMidd97JLbfcckKD\nEk6sUNCDx15JW30pPkcFao0NWY78MsjQOWUlEFDTZo3B2mrE1xjG32rHZ2iiNaaS+ngzzUkJhLzD\nkEJmZH8cao8Wg1GDOl6NyqhB1spIkoREGC1BdFIIvexHJ/vQqSR0Kg0atQ6NWo9GpUUjy6hkCVmS\nkCTQ6TW4PX5CYQVFAV84jC8UxhuK/NcdDGHzB/G2l0yFgWZvgGZvgF1tkdegkiKt/wfHRTEsLgqz\nTnPQ+yEIgtBXPfTQQxQVFTFz5szDLsEgCCdC9y7KQV9k3q36EMsEHWkEV1G6EuAxg9LIyshl8UfF\nVDVl4XHGYizcRVhn580CO8Pis7hgTQuKy43lzdeIzi9g5qwb+GptE20WNzs3VVOxz0LW1BwgcrKf\nNvBOWqs+wmMvxW0toXjd3zBnXITBPPyYOiIn6rUsGpzFJ9UW1jfZqHf7eKa4iqnp8UxJjRMVZMJx\nkWTVMZfTA6hUEjpjFBqvE/kkT+2Liopi9eovURSFRYvupaGhniVLXmTmzB8DUFg4kFdeWczgwUNR\nqVQ89dQTXHrp5Qct8frLXz7Cww//mvj4KFpbnfz+9/9LbKz59C4TVFdXx3XXXccNN9yA2+0mFAoR\nHR1NKBSiqKiIYcOGnfDghO9PURS8LguNB7bTUlcMigWDsat7tbbbhUe3W0dbWwwuixqPxY/DZ8dm\naqTBqKM5NolQbD9U8khU+igMRolMU5BYox+TyoeRNgySFyNeDHgxSl70+NDhR02QHt8lChBs/+eL\n3CWrTehMGWiNmRhi8tFHpZKQEE1rq/OIc3B9oTB2f5AWn59Gj58Gt586t5dmb4CQApVOL5VOLyuq\nLeREG5iQbGZwnOl7r5EnCIJwptmwYQNLlixhxIgRpzsU4SzQMQdXddg5uJEEN9RjmaDeEtxuz5Fk\nspKj+PXNo/loXQUfr6/E9e041OkH0KSXUZTgY/+lRq7cHUNySQPe/fvw/eNRpl48jbLcYezcUoet\nzUP9mgoYFt8+ghtFYt5cXK07aKtZSTDgwlKxDJ1pG3FZV6A1JH/v16+RZWZkJzMkLoqlFY20+YJ8\nXtvKdouDGdlJFMaK6XPCD58sy/z5z3/niSf+yhVXXIhOp2fWrNlcffV1ANx220KeeeYfzJs3F1mW\nOf/8qSxa9BMAGhoauOmmq3n11XdJTU09ZTEfdYJ74YUXsnbtWuLj43ss7VNZWcmNN97Izp07D/Ns\n4WRRlDCOlipq923G56rFZHKj00eusnZfdlhRwO6IwtZiwG1RsNu8tEpemkx2mqJTkbJSidGYiI8K\nkWvwco7kQx/0oQlUovEFUQISgWY1waCaUEgmHJYJKxKOsAG7Evk8yHIYWVKQ5DBqVRitNkCsWSIm\nFgwGPzIOwsFIN4hw0IXHVorHVoqtfhVqbSyetBFooocjaw5/RUunkkkyaEkyaBnYrQu5IxCkwuGh\n1OZmd5sTbyhMhcNDhcNDvE7DJRkJDIuPEmvbCYLQZyUkJKDTiRJJ4dQ45BzcYylRDnd7jqTq3O+V\nk/MYWZDEkpV7Ka8tINSWjC5vNx6jnTfPDVOQnsAlWzyoHW5sKz4k2fwNl108h021OlxS5JgBX4ht\n6ysZPiaTqISRGGPzcDZ+jrVpNz5XNQ17nycqaQyxKZNRab5/UpoXY+QnQ7JZWWNhY5ONFl+Al0vr\nGGQ2cXlWIgl67ZF3Igh9yKhRo/n44y86f87NzeMf/3im1221Wi333fcg99334EGPpaam8tln3/T6\nvEce+e0JibU3h01w33zzTZ5++mkgMjI4Y8aMg5IDl8tFQUHBSQtQ6Mnv91C1ayPN9Xsw6JzExPpQ\nq8OY9GDqtqJOMChjtUbjalFjbwvR6A5gMUjYE4zoUs0kpsskhLyku9zo7E7CbishT4CQNwBKCEUO\nEgoHkcJ+FCWMFAZdMEyMX0EdgrCkJihrIv9UOrxqIz61CbcmCpchkQAHlwabonUMGJpA4SANasmC\nz1WDz1lBKOAg6LfRWLkaWI0+Oo/YtPPRmbK+13sTrVEzLD6aYfHRzMpOZp/NxfomG/vtblp9Ad4q\na2Bdk57ZOSkkG8SXkSAIfc8999zDH/7wBx555BGys7PRaHr+rf1uSZggHI8eywT1GMFtL8eSJKT2\nUt3DdVGGniO4HQluh+zUaB6ZN5o139bzn68O4Nw1HlVSNdqsfexLg8rLjEzao2XoHjshq5XQu/9m\nTL8c5HGzaAWkkMLGteUU76jn3AnZDBqRSv9zbqGmbBuWqhWE/FaczZtwtewgJnk80cnjkVXf73dF\np5KZmZ3M6MQYPqhqpsrpZY/VRYnNxejEGKamxxOrFdOiBOFMcNgEd86cORgMBsLhMA8//DALFy4k\nOjq683FJkjAajYwbN+6kB3q2ctla2LH+U8KeRsyxfqJjvWhlyEjvuZ3Xq8HeasTRChYHNCgyTnM0\nMVojiSYnsWEran+I6CYLAaMNv9FEnS6GcGwScnwGMipQJIJB8AeD+IIBXH4vDq+XoORF0viQtD4k\nvRuN1k5UwI/Z4cLsCJFgDZFmCZDYHERSIhXJPpURhzEZZ84IWjTJ2OwBXA4f29bXsWOjROHQFEZP\nnEZCto6Apx6PvQR3604CPjteRxleRxmG2AHEZV6KWnvoxaIPRS1LDIqLYlBcFPVuHytrLJTa3FQ5\nvTxdXMUVWUmMSYoRo7mCIPQpjz/+OFarlauu6n2NwT179pziiIQfso6yY1mGcPscWrWshpAb6Bq9\nhe/MwT1CifJ3E1yIdDCeMiKdUYVJLFtTzlfbZTytqWgy90NyNauGy2zNNXPBTj/9qpwEqirQmVbD\nlMvRqCJLGDpsXr5aUcL2DVX86NIBZOTmkzZoEY6m9dgb16GE/dgavsZh2UJM8jiiEs9FVukPiuVw\n0k16Fg7MZHuLg5U1FhyBEJua7WyzOBibHMvk1Dixdq4gnGaH/Q3UaDRceeWVAGRmZjJq1KjOxXyF\nk6Ohupxdmz/DqHJiNgeIivGRlXLwdk6HHnurBqtVotGrwqbTEaXWYgz60KUlUpCeQUpyJunmOOKj\nDWjUx9YMQVEUXN4gDS1uKhsdVDY4KC5vodFjxxLdhmxuQc62IOu8aANhshoCDKwNklfjJ8lRQVJR\nBbmSBGOm0txvNKWlbXjdAfZ+28D+PU2MnpjD8DGZxGdk0H/IFdSUb6WtdjUBbyMeWwleRzlxmZdh\nih9xzMlomlHHLYUZlFhdvFfRiCMQ4r+VTTR5/VyelSjm5gqC0Gc8/vjjpzsE4SzSMYIryd3m10pd\nywRJ3SoIepQo99aAqccc3EN3dI0yaLjh4kIuGZPFB2vKWbdbS7AhB3XGfmwJdbw/SU1Go5pJu7yE\n2zvDal1WJvk2UJk+niqLgq3Nw7K3dhAdq2fYuRkMHD6BqMTR2Bu+wWHZQjjowlr3BbaGNUQnjSY6\naSwqzdE3bZMkiVGJMQyLj2JDo42vG1pxB8OsbbSyocnKiIRoJqXEkSo6LgvCaXHYbPXxxx9n0aJF\nGAwG1qxZw5o1aw657c9+9rMTHtzZwNpiYd2q94hRO4mPjyS0hbk9t1EUcNgM2FpVNDtVNIV0qLRG\nUuLiyZ86gqlpaejarxaqVFJnd7IjNWs6GpIkEWXQkJ8ZS35mbHs8CvUtbjbvbWLNt/W0VHiQo9oI\nJtVSllnPgawwmoCB82rVjC7xQUsbbFpFyu6NDL5+HtVyNlvXV+J1B9jwVRkV+y1cNnso8fFRmOIG\no4seiKv1W6y1nxIOeWit+gC/u564zEuRpGPvWjjAbOInQ7L5T3kDJTY36xqtuAIhrs5LEUmuIAh9\nwnnnnXe6QxDOIsHOEdyu84nIOriRXh+Squs0MnTELsqHH8H9riSzgdumD+aycdl8tK6CzXtMBOvy\nUGfspya5gbcv0JDsiySlcjiEtmovBVV7yYjLpDp3CjV2LQ6bl3WrDrB5TQUDh6UyZNQk0gePxd64\nHlfLdpSwD3vjWuxN6zHGDiIq8Vx0UdlHfUFdI8tMTotjTHIMaxusrGu04gmF2WZxsM3ioDDWyHlJ\nsQyINaESa+gKwilz2AR3+/btBAIBDAYD27dvP+R2oszz6Pn9fr5c8S4afzMJcQFi47wMze+ZiIZC\nEg6rAWubimaXCitGcvLyGX3pecRGGQ+x51NHkiTSE03MmpTLjIk5bC9tZtmaCmrK4wnUFGDoV04g\noZK1OWE2ZGm4rm0MSd/sIuxy0fyvZ0m75DKuve3HbFxdwZ6d9TTU2Hln8RZuvHMcWr0qklQnjMAQ\n05+WymV4HQdwWjYTCthIzLn6uNbyMmlU3FiQzgeVTWxutrOz1YFRLTO9X5L4HAuCcEa69tpreeGF\nF4iJiWHu3LmH/Vsl1sEVTpSwohBW2s9P5K7R2cgIbvsazOqDS5R766AMB3dRPloZiSYWzhzCVVPy\n+HRLNd/sjMVX40CdUoE1U4cBsJsk9mXpyK/xYWyrYUDbG2To4mjImUCNkkTAH6Joay1FW2tJyYhh\n4LAR5BZMwGfbisOyGSXkw23djdu6G7UukajEUZjihqDSRB8xPgC9SsWFGQlMTo1ji8XO2oY22vxB\nSm1uSm1uojUqRiXGMDoxRjSkEoRT4LAJ7quvvtrrbeH7qaouY8c3y0mO8pGQ7GNQv2CPxyMjtHra\nWtU0OdX4DAlMmTSV4enph9jjmUOWJM4dkMzIwiRW76jj3a/24zkwCLk+ncRhJThUrbyWWMmUG8cx\nYXU9npK9tH36CYHWVs6/fSFZuXF8ubwEt8vPkmfWM/O6ESQkR67IqjRRJPW/jrbaT3E2b8JjK6Wl\n8r8k5Pz4uEZyVZLEldnJSEhsaraxvslGkkHLuOTvP9dXEAThZJs0aVJnM6nJkyef5miEs0X3Edke\nJcpyV4myrD64RLnXDsoAHevgSvKxrUtrNnD9RYXMnJjL2qJ6Vu9MxN4gQS74ZYnlk2OJcYYYUeJm\ncLmXKF8b+SUf00/WUZc4lFrzILxhNY21dhpr7az9XCY7P4W8whtJSq7Dbd1OwNNA0GfBWvsp1trP\n0EVlY4obitE8CFltOGKMWpXMhBQzY5NjKW5zsrHJRpnDgyMQ4uv6Nr6ubyPTpIs0xIyLwqwTTakE\n4WQ4bIJ7uJLk7iRJYuLEiSckoB+K1V+txNOwl6S4AHEJHs4Z1HOU1uPW0mbR0mxX4dEncPm0Kxka\ndfTzP840siTxo5EZDM2N58n3iqhugqbNo8g7r5L68D5Wu3bjuXQ4l6WkYl/9Fc4tm2jUaMibfxsx\nZgMfvf0tXk+AD9/eyeybRhFjjnyRSJJMfOZlyLIGe+PayNXV+njM6VOPK15JkpiZnYQ9EGSv1cXy\nKgvZUQbSxHwZQRDOMPfcc0/n7enTp5OTk3P6ghHOGh3zbwEkqeu2WlKhhNov1HcbwQ0d5Qju0ZQn\nH06UQcOl5/XjkjFZvL2njm9dbhRXNL7S82hNqmH1yAbWnRNFXo2PwWVesut95DRtJbtpG63GNOrN\nA2k2ZhIMwoG9zRzY24xKLZOVO47+BRBrOoDfWYIS9uNzVuBzVtBasxydKRtDbAGG2EI0usMvZ6iS\npM5VHVq8frY029lqseMMhqhx+ahx+VhRbSHLpGeQ2URhrLH9/ENUkgnCiXDYBHfBggVHtRNJkkTn\nRuCb1V/gri0iNSlAdrwHKbbrMUUBW5uBZoualmAUky6cwYCJp27B41Ml0Wzg4RvP5en/FrGrrJWy\nDXkMm2xiv28Hmy3fYh5/PhO0l2D9/FPs69eiSU0l6YoZXHnDObz/2nY8rgArlu7iqptHoe72xRmb\ndgGhoBtXy3bsjd+gi8rCEJN/XLHKksTVuSk8ubsKqz/I0vJG7hqcJebjCoJwxpo2bRqDBw9mxowZ\nXH755SQnJ5/ukIQfqFC3ZX8kqfsIrhol0Msc3GAkCVYdYgS3s0T5OBPcrpgkYqN14HJTmBnL8IzJ\nbN7bxLc7GwjENLI3vp7S8y1Ee4IUVnrJr/aR1lJHgruOgKylMSqXpqhsrIY0QsEwFftaqdgHEEdi\n8o/IL/CQEFcHwQpQQvic5fic5VhrP0WtS0Af0x99VDa6qGxU6kNPH0vQa7k0K5GLMhI44HBT1Opk\nd5sTbyhMtctLtcvLp7UtRGtUFJpNjPLGkyzLmFQn5n0ShLPRYRPcPXv2iHmJR7Bx01pa928hLTFA\nVoIbeWDXY8GgTGuzgcY2Nar4HC6bNuv0BXoK6bQq7p09jH/+51t2V7RRvDaVcy4YQbF9J59Vf03u\nBTeR7HTg2LCelv++hyGvP4lDh3DVTefy5r830trsYuPX5Uy8sCuBlSSJ+MxpBNwN+D31tFZ9SNqg\nu5BVxzfialCrmJObwr9Laqlz+9jcbGdscuyRnygIgnAafP7556xYsYKPPvqIv/zlL4waNYoZM2Zw\n6aWXEhsr/nYJJ073EVyl2xxctawi1D6C272Lcih0+BJlJXxiRnC7C7XPEdaoZMYWpjB2cAoe3yCK\nylrYW21j854aWvW1bEppZmuhhWifj/41vkiy21xCpr0Ev6zDYsqiKSqbNkMaYVmNpcmLpUkCMtBo\nUsjr7yEtpQ2jvh4JH0FfC87mFpzNmyLH1yeji8pGZ8pAa0xHrUs46PxZJUsUxpoojDUxKzuZA3Y3\nu9uclNpc2AMhHIEQW5vtbG22AxCv05AdpScn2kB2lIFEvUZcgBeEo3TYBHfChAmMGTOGiRMnMmHC\nBLKysk5VXGe0HTu2UrN7LekJflKSPKR1Kz8OBmVaGg3UWbUUnHsho8cMPo2Rnj4atYpFVw7lD0u2\n0tjqpnxTP3LPs1Jur+T1kqU8PPcefDU1+GuqaXzlJUyPPkb+wGRGju/HtnVVfLu5hv4DkkjN7Dph\nk2Q1CTk/pn7v84QCDmz1XxGXeelxx5oXY2R4fBTftjr5tMbCOQnR6A41h0gQBOE0ysjIYMGCBSxY\nsIDq6mpWrlzJ0qVLefTRR5k4cSLPPvvs6Q5R+IHonuAi9WwyFezsoty9RLl9BPdQyxKeoBLl7jpG\nmVXdEj+DTs34oalcMSUfi6WQ/bU2ig60sLuihUpHNVuim9g+zoJOayOzyU+/Bj/Z9eWcU7+fkKTC\npk+mxZhOqzEDpy6eQEBNyd5oSvZGA1nEme2kp1lJSrJj1FuRJIWAt4mAtwmnZXPkNcpatMY0tMZ0\ntIZUNPpE1PpEZDlyQUAtSwwwmxhgNqEoCg0eP6U2F/tsbqpcXoJhhVZfgFZfgO0tDgA0skSaQUe6\nSUe6UUeaUUeSXotWnK8IJ0Fx8S5+9asHWLbsEwD27i3mjjtuQafrGli66ab5zJt3K3a7nb/97Y9s\n2rQRgAkTJnL//b8kqpepl3v2FLNgwc297udEOmyCu3LlSjZs2MDatWv597//TTgcZuLEiYwfP57x\n48djNp89TXmKdu+gcsdqUuMCJCR7iB/c9cc+FJJobTZS16Ihe8T5jJk+4jRGeuYw6jXc/eOh/O6l\nzbTaAwxqHUe9rhFXwM1/qz7jutvuoPIP/0vA0ozlww9IvGM+Y6fkUrGvhdZmF2u/2M/seaN6XAXV\n6BOJSZmIvWE1jubNRCedh1oXd9yxTstKZHebC08ozKYmG5PTjn+fgiAIJ1NcXBxJSUmkpKRQWlpK\nU1PT6Q5J+AEJdmsypdDVAVkja/B2JLjqrtPII3dRbn+OfNhTz++lYwRXdYiRTVmW6J8eS//02P/P\n3nnHR13ff/z5Hfe9leQyScIOQ9G6iloBLSoWFQfYKmitYtVWQH8OtI7iLmqdOGvFFlSsq7ZVrOLC\ngVuLIg62hJ2QnUty67t+f3zvLgm5SCAXYuLn+fDM977fz33ufcfdfb+vz3txys+HENNHUloWZM3m\nOlZtraI0uok1xVXIw2vJkmvoWxOhuLKOvlWVDN3yOYbsod7ThzpPAfWePjS486mtC1BbF4CVoCgm\nOdlB8nLqyM2tJ5DVhKKY8fzdjUQbN7ayR9VyUD35uDz5uNy5KFo2qjuHIk+AYl8u4/rnkRnw8dXm\nKtbXh9nQGGZjY4SoaaFbNpuaImxqirSaM1tTKfBo5Hs0Crwu8j0aOZpKQHOhitZEgl3Etm1eeeUl\nHnroXpQWC1hr1qxm1Kgx3HnnfW0ec999dwES//nPK9i2zXXXXc1jjz3KxRe3bSG7Zs0qxo4dy5//\nfE9a2pm2x/f+ymRlZXHsscdy7LHHArBlyxY++ugjXnvtNf70pz9RXFzMmDFjkrfexDtvvU7NtvX4\n5Qi5AZPcgjAHtBC1liVRU+WlrMpF4YhRHDLhMA7uRnt/qPQvyODkMYN58YNSPvqinkmnjOP1bYv4\n3/ZljO0/hpxjj6f2tUXUvP460VMnoSgahx8zjP8+u5yKsga+W1XJsH1a55hlFR5OY9UXWEYj9ds/\nIG/gyZ22M6C5ODg/k88qg3ywvZbRhQHUVI3qBQKBoBtpbGxk8eLFvPbaa3z00Uf07duXk046iZkz\nZ1JSUrLzCQSCDqIbLYpMtQhRdslqcx9cdRdycJMhyntO4O6I5lLYe2AOew/M4WRKsOxDqKwLs7G8\ngdLyetbHtvG+XEasXy2au46iaDUFwRoK6ioYULucnHKLsJpHgzuXRncODe5cavQcqqoTi+I2Gf4Q\ngUAD2VmNBAINZGY0oarOe2PEajFitUSCa9vYJquZuDw5+DPzCVgeRqp+Ds3PRCryUWdlsV1XKQtb\nbAtF2RaKEol7zOtiBnUxg7XBUKv5JCDTpRDQXGS7VXI0F1maSoaqkOFSyHCpZLoUPMruVbUW9E4W\nLJjP228vZurU83jqqSeS+9esWc2wYXulfMysWTdiWRaaplFZWUE4HCIQSO0EXbNmNSNGjEh5LJ3s\n0q9M//79mTJlClOmTMG2bb799ls++ugjHn300V4lcF94bh4HDd3G4B0qH1sW1FX7KK9yERh4IKOP\nO6p7DOxhnDB6EB98XUZVfYQN32bTt38R25rKWVT6JjNO/A317y/Bampi679fIPu00+k/OIeBQ3LZ\ntL6GZZ9sYuiI1j1qZdlFVuHh1G19nabq5QSKjkTVsjpt59iiXP5XGaRBN1lZ18T+uR3rfycQCAR7\nitGjR5OTk8OECRO46KKL2H///bvbJEEvpaXAteMhyhKS0ybITCFwzZ0VmUq/B9dICNzdXI+WJYnC\nHB+FOT5+tk8h4FzAN4RilFWHKKtuYmNNJavqt/NBtIqwWUuOUUueXk1OeAs5DSbFDRbesBfTDhB2\nZRGqz6KpKotKV19iqg+w8XiiZGaEyPCHyIj/9fkieNyxpC2W0UC0sYFo46aUtuYBubbMvrYbW/IQ\ncgWol3MISlnU2T7qbC81potG03kzbCComwR1k81N7b8HiiQlRa9XlfEoLf4qMh5Vxqs4QtijyGiK\njCZLuGQZTXH+KqL68y5jWDb1MX23H68oEkZTlPpIbKee0F3x5p944iSmTj2PZcs+b7WSAw2kAAAg\nAElEQVR/7drVaJrG5MkTMU2TcePGc8EFF6JpGmr8d+C2227m1VdfpqRkCKeccmrK+desWc2WLZtY\nuPDkNvOkk+/9lSktLf3eB/v9fsaPH8/48ePTalS3Iyk4a1820ahKfbWbqnqVjAH7ccSxx3S3dT0O\nVZE55ecl/P3llXy5toYzD/g5LzQ9z8qaNWw1qsk59niqX/g35W+8SebxJ4LXz09HDWTT+hqqtjey\ndWMd/Qe3DhnOyB9JsHwJlhmhqeZLAkVjO21nrsfFsCwfa4MhPq8KCoErEAh+cDz66KOMGjVKeFwE\nXY5uNIcl25Kz7YqL02QV5VQCt50cXNuKP6aLc3DTQaZPI9OnsdeAbKBfcr9l29Q3xqgORqisa2Jr\nfTUrGqupDdcSjdbgCtXgjWwmMxYmMxolN2rjjnhRa71g+TAkH5Wqn82uIqKKD11z4/ab+LwRfN4I\nXm8EjyeKxx3DrcVwu2O4XM3/DpJkIUlhIEymXUumuaG14RLoikIjfhptHw34abD9NOKnwfYRwksY\nDzotioPZNvW6QX3833R3kG0LxTZRWv21kLCRsZFs56+MjWzbyICMtcN9nLGAhI2EhBTfn7wvOYss\nsgSSbSNJEnJyP4CNZEtJuS3LzoHEHufK3nk8koQs2Xg8LqIRPdmhSWoh1pu37OS2FLdwx+Nyi0dJ\nzZutRiW2LRveIZMQe6ZStg+L4+QQbbIH4u/DjmzasoW169ZhGCZffPmlM1SSGTR4KGeedR4NDUHm\nz/srVdVVTDplcvJxvzj2RI4adyxPP/U4l1x6IZdedlWbuRXVxbhx4zj22JOorKzi+uuvYd68ucyY\ncXEaX/FOBO6ECROQJAnbTr0ykDjW29oE/XLKb1m7diWmZDPikJ90aYz4j4VR+xbx4vulVNVH2PJd\nBoXZBWwPVfL2pvc5++hJ1LzyX+xYjPqPPiRwzLEUDwjQpziTirIGVny5rY3AlWUX/twDaaj8lMaq\nL8gqPAJJ6nxI8cH5WawNhlhbH6I+phPQRBN2gUDQvTz33HOt7m/alNrDA3D66ad3tTmCHwl6yyrK\nSYHrnBNTenCNjuXg0o0hyp1FliRyMt3kZLoZ1i8A9G0zxrZtwlGD+qYY9Y1RKhuD1ISD1EcbaArV\nYTTWQrgWObQFNRLCjOpEGi0MU6XRVJFtF5LlxsYHdjYoCqpHRvFJKF6QNeemahaaS8flMuK3+LZq\nElAayJGD7b4O3VYI4yGMh5DtSW5HbY0YLqJoRNGI2S22cdFen15LkrEkmd33R3YjUbqu/fAPRD6E\nkHnBalvw6fuoM71EgX/pfgByz7+GGuC/AIFcMo/5FR++8hyxE85p/UAFXCdOZd0tl/J0rYXL3/p5\ns8+5gqraNXi9Xvr168/Uqecyd+5f9qzAfeutt9L6ZD2JESP2JTc3g5qaxu42pVcgyxLjRvbnn++s\n45Nvt3Pqr0bxYul/+bLya6bsdQpZh42i/v33qH3nHbLGjUeSJPY5sJiKsgZK11YRjei4Pa3FZkbe\nSBoqP8XUg0QbN+LJ7Hz+2T45ftyyTNSyWFnXxKg+P55CagKB4IfJ3LlzOzROkiQhcAVpIxGirCoy\nZjx/Vk2EFxsp+uDutE1Q1xWZUn9AEQ2SJOHzuPB5XBTn+RlBbscfK0NGhpey7fU0hKM0xiI0RSOE\nwiEiTQ1Ew41EwyFMPYIRiWA2SVgxFUk3sA0XGDYYNpJhg22jyKAoIMtOmyJFjW8rNori5FbLcgiv\nHMIv20gyyArI8W3nvo0s2yDbWLKKIcuYkoopy5iSgikrzt/4zZAUTJxtS5KwkFv8dfy2FjImMiYK\nlt36vo2EFffZ7rjt3Jd3uN/sURV0DXqokc1vvsjA405F9XgBsAwdOd4m7Ku/3ka/sceT95ORgLMA\nJskKitudcp6isYcl98ViMTStcy0/U/G9vzL9+vVrs2/NmjWsW7cOy7IYMmQI++7742yDI9h1jjig\nmBfeX09Mt5DqBqLKKrpl8HnFcg476ijq33+PWHkZ0Y0b8QwezNARffhg8TpMw+K7VZXse1DrlVKX\ntwCXtxA9vJ1Q3aq0CFyXLLNXwMfXtY2srBUCVyAQdD9vv/12d5sg+BGSELiaS0a3HN9cQuBaunNf\ncu1CiHIX5ODq8RBlVy8pCilLEh63SiDDTYZXA3pGqpSiSEmnUCLq0TJNbNvC1HUs28IyLExsTN3E\nsm0sw8TGwrZtTNME244vpNhYtg2miQXYtoVtW1iWhW1Z8c+R7USX2haS5Wxb4DwPjuPUspzPo41N\n/D+wnHGJ/bIEmttFJKIT/yhht7hB877v2075N/E/u/Xo9py6tp1KpO84c/z14HxWNE0lGjPaibSV\nkmN3im23sNlhg17JWtvi6NhWbNniwa8+wTIbGDvhROpra3nmjec54mejGBXbitk3nw2vP8eEYj+y\nLPPCC89wwE9HMs6qhFiLp5EtHvr6E9yD8jAMna1by1iwYD4TJ/6yY3buAh3+lQkGg1x11VW8++67\nBAIBTNOksbGRkSNHMnfuXDIze8aXUNB9ZHhdHDAkj8/XVLJ8dT0H7LcvX1R8xZcVX3Pkwb/H3acP\n0YoKGr9YimfwYNwelYFDcildU8WGtVVtBC6ALzCC+vB2wvWrsPsfn5a8tBHZfr6ubWR9Q5ioaYme\nuAKB4AdFdXU1zz//PBs2bODKK6/k008/Zfjw4QwfPry7TRP0IpICV1XQ497X5hzcuMBt4XmxEgK3\nnWI2XVFFuVngCg/eDw1ZUQAFRf3hpnqlEuY9ha62/QtvBi+6nmH84UcBMLz/YO67727uvfVPuN0e\nJk36FeeddwGSJHHkoWN4+OH7mffQA8iyzJFHHs2MGZfg9XopLy/n7LMn8+STz1NUVMQ+gwbz4INz\nOP74Y5LzTJ7867Tb3+Er91tvvZWKigpeeeUVPv30U5YuXcp///tfIpEId955Z9oNE/RODo23/Fm5\nsY69s5wy4WvqviNiRMgbMwqAhs+XJscPHpYHwJaNdei6yY54s505TL0BPbw9LTbune3kG5i2zabG\ncFrmFAgEgnSwYsUKjjvuON59911efvllQqEQH374Iaeddhoff/zxLs1VVVXF6NGjeeeddwCor6/n\noosu4uCDD+aoo47i+eefT461bZt77rmHUaNGceihh3LLLbc4XhdBryWRg+tyyRg7CtyY45aRW1Q+\nTebgtuPBxU5/kamEwBX9XgWC9DJy5CG88kpzqmpJyRDuv/9hXn99CS+99Drnnz8t6VTSNI3LLruS\n//73DRYufI3LL78ar9cJZS4qKuLNN9+nqKgoOc/jjz/O4sXvtZknnXRY4L799tvceOONDB06NLlv\n+PDh3HDDDbzxxhtpN0zQOzlwaD6qImHZNnawD7IkY9kW31SvJu+wnwGgby9Hr6oEYOBQR+CahsW2\njXVt5nN5+iCrjiCNNG5Ii40+VaGP1zlpb2iI7GS0QCAQ7Dn+/Oc/c8455/Dss8/iiuc/3XrrrZx9\n9tncfffduzTXtddeS11d8+/q9ddfj8/n46OPPuKBBx7g7rvv5st4Bc2nnnqKd999l5deeolFixbx\nxRdfMH/+/PS9MMEPjmYPbssQZeczZ0WjAEgtBK4Rr7qsqqkFbFfk4OrxMFThwRUIBC3psMBVVRW3\nu20SsMfjQdd7ZN00QTfg1pR45UFYu6mJYdlDAFhVvZaMvYYjxT9jodWrAPD5NfILnQps2za3FbiS\nJOHJGARANE0CF2BwhrPytEF4cAUCwQ+Ib7/9lokTJ7bZf/rpp/Pdd991eJ5nnnkGr9dLcXExAE1N\nTSxevJhLLrkEt9vNAQccwEknncSLL74IwMKFCznnnHPo06cPBQUFTJs2jRdeeCE9L0rwg8SIC1yX\nqmAki0w54jURotzSg6vr8aJUrp3k4HZJiLJIJRIIBM10+BdhzJgx3Hnnna1We2tqarjrrrsYM2ZM\nlxgn6J3sO9ipKLhiQw3DAoMBWFdXiqyq+IY7TdbDq1Ylxxf3dwRx+Zb6lPO5M505Io0b221ptasM\nzvQAsLkxgmH1rLwMgUDQewkEAmzbtq3N/m+//Zbc3I5Vay0tLeWxxx7jpptuSu7buHEjqqoyYMCA\n5L6SkhLWr18PwPr16xk2bFirY6WlpWn7zRX88GhZZCpmOiHJLiXuwY2l8ODG04hUV3se3Hjebho9\nuIYIURYIBCno8K/MNddcwznnnMORRx5J//79AdiyZQtDhw7ltttu6zIDBb2PfQfn8p/31lMTjFLg\ncj5L20OV1EWC+EaMoOmbr5MeXIDiAQG+/nwrFeUNGIbZJvzJ7R8IgG1GMaLVuDz5nbZxoN/x4Bq2\nTWUkRrEv/SXMBQKBYFf59a9/zQ033MAf/vAHAFavXs17773Hgw8+yLnnnrvTxxuGwVVXXcW1115L\ndnZzlfhQKITH42k11uPxEIk4aRrhcLjVca/Xi2VZxGKxlNFdOyJJEulwsslxISP3MEHTE+2OxUOO\nPZpK1HQErVf1oChSMgdX9biTfW8THl+3W0nZCzchcBVVa7dX7q6i2/HnVOVWc/bE9xuE3Xuanmo3\n9Fzb95TdHRa4BQUFvPTSS7z33nusX78et9vNkCFDGDNmTJckBwt6LwMLM1AVGcO0MBqyUCQF0zZZ\nVbmO/vEqoEZNNUYwiJqVRVE8pNkybSrLG5Me3QQuTz6SpGLbBrFQeVoEbrZbTfbDLQtFhcAVCAQ/\nCC644AL8fj+333474XCY//u//yM/P5/p06dzzjnn7PTxDz/8MPvssw9HHnlkq/1er5doPK8yQSQS\nwefzAY7YbXk8HA63m7qUirw8f1qvFbLjxQB7Gj3JbltyViS8bhVTdsRuwOcnNzcjGaKcmZtFbq6T\nRmTFBW4g25fc15JtkhOinJGZlfL4rmJYdrK1S14g9XP2pPe7JcLuPUtPtRt6ru1dbfcuxYmoqsrR\nRx/NEUcckdyXyL/VWoSpCATfh6rIDCrM4LttQTaVhemXVcymhi2sr93E0AGHgySBbRPdtAF1vwPw\nZ7rxZ2o0NcSo3t5W4EqSjMtbSCy0lVi4DD/7ddpGWZIo8mlsbIxQHoru/AECgUDQRViWhdzC/fmb\n3/yG3/zmN4RCIUzTTLbpq6yspKCg4HvnWrRoEZWVlSxatAiAxsZGLr/8cn73u9+h6zrbtm2jb1+n\nJVtpaWkyLHno0KGUlpZy4IEHJo8NGTKkw6+huropbR7c7Gw/dXVNWD0ofaQn2l0XL7Lodas0hkPO\nTlOmurohWWQqFLOoqWkEIBp1BGw0ZiT3tcSIOTUtwhE75fFdJWI0V/EON0WpkZvn7InvNwi79zQ9\n1W7ouban0+7vWyjrsMD97LPPuPnmm9mwYUOyeTI4rQMkSWLlypWdMlLw46KkOIvvtgUpLQsysG9f\nNjVsYWPdFpT+XlyFhejl5UQ2bsS/3wEA5BVk0NRQQ3Vl6pOi5uvrCNxQWdpsLPK52dgYoSwsBK5A\nIOg+Lr30Uu69915UtfUpO+FdBUe43nzzzXz66affO9drr73W6v64ceO4/vrrOfroo1m1ahX33HMP\nt9xyC2vXruXll1/m0UcfBWDixInMmzePUaNGoaoqc+fOZdKkSR1+DbZtk86uQpZl97i+ldCz7A5H\nHMHqdatUGo7Y1WQ3RlSHeO61rbiSryfRyk9R5JSv0bKcsGYkLS3vQcRovhZVbNp5zp7zfrdE2L1n\n6al2Q8+1vavt7rDAvf766xk2bBhXX311mzwdgWBXGVTkeBy2VDYxOsOp4rmhbgsAnoGD0cvLiW7a\nmByfW+Bn0/oaqiuaUs7n8jheCyNanTYbC+OtgqrCokq4QCDoPj777DMuuugiHnzwwTbRUvX19dx0\n0028+uqrnS74OHv2bG688UaOPPJIfD4fV155ZdJje+aZZ1JVVcVpp52GruucfPLJHcr5FfRcIrFm\ngZvIwXUrGnakuX2e7HHqVViWhRW/WE1VRdm27RZFplxpsS9mNgtctyKqKAsEgmY6LHArKip45JFH\nKCkp6Up7BD8S+uY7sffhqEFAcZo/14braYg1osXD42Ll5cnxeX2cMITqysZk1EBLXJ54v1y9AcuM\nIiudz5nNczsn4XrdQLcs0YZAIBB0CwsWLOB3v/sd06ZN4+GHH8brdUTFu+++y3XXXUc0GmX27NlM\nnjx5l+d+++23k9vZ2dncf//9KccpisLMmTOZOXPm7r0IQY8jEnM8sl6PSjTseF89ihsz3Nw+T45/\nFg29WWy6UlRRthPeW0CW01PTIiIErkAgaIcO/yKMHz+eJUuWdKUtgh8RRbnNoXV2uDmGvrypAq3I\nEbx6xXbseDh8Tp4z3tAtmhpj7Ijqzktup8uLm+tuXmWuiQovrkAg6B723ntvnn76aTZv3sz555/P\n9u3bmTVrFtOnT2f//ffn5Zdf3i1xKxB8H0mB61aJGAkPrhsrFEqOkX2OwI3F828BNHdb34nVQuBK\nSnpqtkSFwBUIuowVK75h0qTjk/dXrVrB2LE/Y/z4nydvCxbMByAYDHLjjX9kwoRxTJgwjtmzr6ex\nMXVKoa7rzJ49m+OPH8cJJxzD7bfPTtZzSicd9uBefvnlTJw4kZdffpkBAwa0KngBcM8996TdOEHv\nxetWyctyUx2MUlNr4nf5aNJDVIar6VfohCzbuo5RU40rv4CsbG/yscG6MBmZrVeAFVcmkuzCtnT0\nSA2ar2+nbcx2u5AlsGxH4BZ6RSVlgUDQPQwYMICnnnqK888/n3HjxhEIBLj77rs56aSTuts0QS8l\nqjcL3GSIsurGqmsWuIrXWXyORloIXE/bS0vbbK5lIctpErjxBXBZAlV08xAI0oJt27zyyks89NC9\nKEpzNMaaNasZNWoMd955X5vH3HffXYDEf/7zCrZtc911V/PYY49y8cWXtxn7178+yPr163juuRew\nLJsrr7yMZ555kqlTz0vr6+iwwL322muRJIn+/fvvdg7uihUruOGGG1i3bh2DBg3i5ptv5qCDDmp3\n/Mcff8y5557L559/jt/fM8tgC9qnOM9PdTBKWXWIPvn5lNZvojJUjavfAckxsfJyXPkFuD0qHq9K\nJGwQrA3Td0B2q7kkSUJ156GHy9PmwVUkiWzNRU1UpzoiPLgCgaB7KSws5KmnnuL3v/890Wi00zm3\nAsH3kcjBdbtkdMvZdkKUgwBILhdSvPBZtIUH153Cg2u38uCmZ7E44cF1y7JoVynocRimRU0wsvOB\n7SArElFLoq4+lMx/b4/cLA9qB6McFiyYz9tvL2bq1PN46qknkvvXrFnNsGF7pXzMrFk3YlkWmqZR\nWVlBOBwiEMhuM84wDBYufIHnn3+eQCCAadrceuudGIaRYtbO0WGBu3TpUv7xj3+w//7779YTRaNR\npk+fzvTp05k8eTILFy5kxowZLF68OKV4ra+vZ9asWdh2z6sMJugY+XGvbHUwQv6AvLjArULWNNTc\nPIyaavSK7YDzmcvK9hIJNxCsS/2DoGoBR+DqwbTZmOt2BG5tNP1fPoFAIOgIH3zwQav7559/PrNn\nz+bss8/mqquuarXK3rKNn0Cwu9i2nQxRVlzN5a/dioYVbxmUyL8FiMU9uLIspSwyZZktc3DTFaJs\nx20S4cmCnoVhWsx69BOq6ndf4O4K+QEPt10wqkMi98QTJzF16nksW/Z5q/1r165G0zQmT56IaZqM\nGzeeCy64EE3TkhX+b7vtZl599WVKSoZwyimntpl78+ZNGIbJ8uXLefjh6UQiEcaPP55p0y5Kzwtt\nQYcF7qBBg4jF2uY+dpRPPvkEWZY588wzATjttNN44oknWLJkCSeccEKb8TfddBMnnHACf//733f7\nOQU/bPKynFXc6voIe/mcHNrKsON9deXFBW5tbXJ8VraXirIGgnXhtpMBiisLADOWPoEb0JyvSFAX\nAlcgEHQPv/vd71Lur6qqYtq0acn7omWfIF3EdCvRCQhJbeGdVdxY8SJTsre5lkbCg6u51ZTeVMtK\nhCjLILUtQrU7RON9p4TAFQjSR35+fsr92dnZ/PSnBzNp0qnU1FRz/fXXMG/eXGbMuDg55oorruGS\nS67g9ttnM2vWlTz00KOt5ggGgxiGzjvvvMP8+U/S0NDIVVfNxO/389vfpj7P7S4dFrgzZszgmmuu\n4eyzz2bgwIFt+vHtbNW4tLSUoUOHttpXUlLC+vXr24x96aWXCAaDXHHFFULg9mLyAk6oe1UwQp4n\n19kO1wCg5uQAYNTWJMdnZTvjg+2seKlaXOCm0YOb5XI+5w0xIXAFAkH3sGrVqu42QfAjo6llWo7a\nvJ2h+dGTAretB9edIv8WwIr30ZVVT9rCiUPxPrg+NT2CWSDYU6iKzG0XjOp0iHJ2wE9dfVNaQ5Tb\n44477k1u9+vXn6lTz2Xu3L+0Erhutxu3282FF17ClCmTCAbrycoKJI9rmgvLsrjsssvIzMzE58vg\njDN+w7/+9Vz3CdxEa4DbbrutzbGOrBqHQqFka4MEHo+HSKT1P+62bdu4//77efrpp3erqpYkSaSj\nm4ssS63+9hR6kt19cpzPQzRm4lecvrhNeggLA1euI3jN2loUxXktmVmOwA01xpL7WuJyO18iUw+m\nPL47BNzNHtxUc/ak97slwu49S0+1G3qu7T3VboHgh0BDqMX1l9JcIMqv+qiJV1FW2vHgpsIy42HN\nqi/l8d0hZDgeXJ8qPLiCnoeqyPTJ2f3vg6JI5Ob6ccs25k4EbmcJBoMsWDCf8877PT6fk1Yai8XQ\nNCcSc+bMizjttDM4/PCfA2AYOoqi4PG01n39+w9EluVWEcGmaXZJOmqHBW5nV5C9Xm8bMRuJRPD5\nmv9xLcvi6quvZubMmRQWFrJly5Zdfp68PH9aiw1kZ/fM4lY9we5hLfLGvK7mFR68BoH+RdQAVn0d\nublOG6HCYsdDG2qMkZPT9t/ZRSGVpWCZUQJZKoq6e8XQWtJPN2BDBQ26SXaOH7mdz1ZPeL9TIeze\ns/RUu6Hn2t5T7RYIupPGcLPANSXnYtSrelFkBTMucBMtgqAjHty4KFa6QuAKD65A0JVkZGTw3nvv\nYNs2M2ZcTHl5GQsWzGfixF8CsNdeI3jiiXnsu+9+KIrCQw/dx3HHnYCmtc63z8zMZOzYo5gzZw7X\nXz+bpqYQ//zn0xx77IS029xhgTtixIh2haPL5aKwsJATTjiBSy65pFXBiwRDhgzhH//4R6t9paWl\nrVoclJeXs3z5clauXMlNN92EFS8Bf+SRR/LII49wyCGH7NTO6uqmtHlws7P91NU1YVk9p9BVT7Lb\ntm0UWcK0bOqqmvvZbdheRqHbEbXRqmqqqxuQJAkLZ4xpWpRtq8PjdbWaT48236/cXo7mTZ1HsCvI\n8VVp07bZUlFPhqv1V6Ynvd8tEXbvWXqq3dBzbU+n3YlFNoHgx0JD2BG1fo9Kk97kbLsccWo2OGlA\nSlZWcnyiTVB7HlzTiIc1p9GD2yQErkCwR5BlmTvuuJf77rubE088Brfbw6RJv2Ly5F8DcP7503j4\n4fuZOvV0ZFnmyCOPZsaMSwBH25199mSefPJ5ioqKuO66m3j00Yc488zTiMV0Jkw4kTPOOCvtNndY\n4P7pT3/i/vvv5+KLL0629vn666954IEHmDJlCkOHDuXhhx8GmsOZWzJ69GhisRhPPvkkZ5xxBgsX\nLqSqqqpV7m7fvn356quvkve3bNnCMcccw5IlSzrcJsi2bUxz5+M6imV1veu/K+gpdmf5NWobooRC\n4FHdRIwoNeF6igNODq5t6Oj1DSiZmXi8zStBwfoILq31x1eSmy9C9WgQRcvrtH3eFqslwaiBV059\nIu0p7/eOCLv3LD3Vbui5tvdUuwWC7iQRopzhc9EQa3S2Xc51mBF0BK6a2SxwQyFHEPv8rReeEyQ8\nuLLqTXl8dxA5uAJB1zFy5CG88spbyfslJUO4//6HU47VNI3LLruSyy67ss2xoqIi3nzz/eR9v9/P\n7NmzmTnz6i49N3dY4M6bN49bb72Vo446KrlvxIgRFBUVceutt/Laa69RWFjI5ZdfnlLgaprG3/72\nN2666SbmzJnDoEGD+Otf/4rP5+OGG24AHBEt+HGR5XMEbrApRq43m20N26mPBlGzBybHGPV1KJmZ\n+DKaBW6oMUZeQeu5JFlFkjVsK4ZphEgHLU+ciZOpQCAQCAS9mca4wM30ajREnWKPGQkPblzgKi0F\nbmNC4KZuAZTIwVW6IAfXLwSuQCDYgQ4L3IqKCgYMGNBmf3FxMWVlZYCj0uvr69udY8SIETz77LNt\n9rcnbPv378/q1as7aqKgB5IZX+0NNsUIFGayrWE7jXoTSkazN9ZsdFaPFUXG7VGJRgzCodQFyGTV\nhxmLJVeLO4tLllAlCcO2kydTgUAgEAh6M4kc3Ayvi2DUOQf7XX4nSi5FiHK4KZ6n257ATYQopykH\n17JtwqLIlEAgaIcO/yqMHDmSu+66q5WADQaD3HPPPYwcORKAN998k8GDB6fdSEHvJcvnnAyDoRgZ\nmhP+1KQ3IWsaktupzpYQuNBcwCIaTi1wE6vDlpm6V+6uIklS8uQpBK5AIBAIfgzUNTqVk7MzNWrC\ndQAE3FlYkQh2vMOFGhe4tm0nF53b8+AaegMAiis9Rd9ChkkipmrH2hgCgUDQ4V+F2bNnc8EFFzB2\n7FgGDhyIZVls2bKFkpISHnroId577z3mzJnDAw880JX2CnoZWfGTYX1TjKHxwlJNejyUyZ+BEY1i\nNjYkx3u8LoJ1ESLtCFxZcfJ70hWiDE6YclA3hcAVCAQCwY+CmgZH4OZmelgfqgUg2x3ADDY7ORIh\nypGwnizk1jKVKIFlxrBNp4uG4spqc3x3qG/Rmz6gCYErEAha0+Ffhb59+7Jw4UI+/vhj1qxZg6qq\nDB8+nNGjRwPg8/l49913yY33LxUIOkLSg9sUIzPuwW2MV2xUMjIwaqpbeXATlXA+nEQAACAASURB\nVJMTFRt3JFGhMV0hytCchysErkAgEAh+DNQGHUGanalRU+2I2mx3AKOqJjlGzXGKQSbybyG1B9fU\ng8ltRUuPwA3GBa4siRxcgUDQll1a9lIUhSOOOKJV5eMEQtgKdge/1/kINoUNMnf04GZmAjuEKMfH\nt+fBVbpU4IoiUwKBQCDo3eiGRTAecuz1mZiVzuJujjuAXr0ecM7PcjyNKFjniGFZlvBluNvM10rg\nujLTYmO97gjcLJfabn96gUDw40Vk5gu6FZ/b8ciGogZZcYHb0oMLtA5R9jjjI+16cJ0Q5URBi3Tg\njefghoUHVyAQCAS9nNp4/i2A7IkktwPuAHp1NQBqXnOf+WC9c77NyHIjy23FphFzzuGy4kWWU7cR\n2lUSHtwskX8rEAhSIASuoFvxuR3vqG5YeOP5s016CNu2UeK9j83GpuT4ZIhyOx5cSXZWjy0rmvL4\n7uCO98KNWcKDKxAIBILeTUVtcwSULjsRVJrsIkvLwKiuAsCV19xnviHuwc3KTt3j1og5Yc2Klp02\nG2uizjVAtlsIXIFA0BbxyyDoVnye5tVc2XZydyzbImbpyB7nZGlHm1eQdxaiLCtxgWvGUh7fHTTF\nEbjRLmxILRAIBALBD4Hyakfg5mW5qYk6HttCfwGSJKFXVgLgaunBTQpcT8r5jIgjcF2evJTHd4eq\niHMNkO9JXbVZIBB0jhUrvuGPf/wDCxe+BsCqVSu44ILf4nY3pyGcffa5TJ16Xryrzp/57LNPARgz\n5nBmzryajBYtPwHKy8s5++zJSJKEbTvX1LFYjOLifjz77H/Sar8QuIJuxetuLg5hm83bESOK7HFO\nllakOdxYi1dL1GOpw4Vl2TnZ2en04CYErvDgCgQCgaCXs73GOecW5vooD20CoI+vANu2iW7bCoDW\nt29yfCJEuT0Prh4XyS53egSubdtURZxF7HxPekKeBQKBg23bvPLKSzz00L0oSvN1+Zo1qxk1agx3\n3nlfm8fcd99dgMR//vMKtm1z3XVX89hjj3LxxZe3GldUVMTbb39Abm4GNTWNVFRUct55Z3HZZX9I\n++sQAlfQrbT04JpG8xcpakbQvPF82kizB9elxUOa9dQCV1ISAlfHti0kqfNR+MkQZVMIXIFAIBD0\nbsprnLSgolwf20KOx7bQV4DZ0IDV5BzTih2Ba5oW9bXtC1zbtjHiAldNk8Bt0E1i8bZEwoMrEKSX\nBQvm8/bbi5k69TyeeuqJ5P41a1YzbNheKR8za9aNWJaFpmlUVlYQDocIBHaeknD33X9m3LjxjBo1\nJm32JxACV9CttPTgmrFmMRoxo3gSHtxwW4Fr6BaWZbcpaJEIUQawrRiSkjpkaldIenCFwBUIBAJB\nL6esxglR7pPjZVmTI3D7+PKJxb23AFpxMQB1NSGsePpOXh9/m7lMvQHbcsKJ0xWinPDeAuS7hQdX\n0DMxLIPaSP3OB7aDokBMC1MfasLcSQ3UHE8AVe6Y5DvxxElMnXoey5Z93mr/2rWr0TSNyZMnYpom\n48aN54ILLkTTNFTVmfu2227m1VdfpqRkCKeccur3Ps/SpZ/x9dfLuf762R2ya1cRAlfQrSiyjNul\nENVNTL2FB7edEGWXq3mMoZtoOxSYSBSZAicPV06LwHVEdNSysG0bSbQkEAgEAkEvJBiKURN0UnwC\nOTqRBmd7QGZfYt9+A4ASyEbxOWK2usLx6KoumUBOWw+uHi6Pb0lp8+CWhRybslwqHtEDV9ADMSyD\nP31yF9WR2j3yfHmeHG4YdWWHRG5+fn7K/dnZ2fz0pwczadKp1NRUc/311zBv3lxmzLg4OeaKK67h\nkkuu4PbbZzNr1pU89NCj7T7PggWPc/rpZ+Hz+Xb9BXUAUUVZ0O34PM4XLhyxkeMhxREz2lxkyjCw\nDaclQMKDC6nzcGWlOVzJNtOTh6vFQ5QtGwxbFJoSCAQCQe9kY7nT0kcCTE8dAJriosjfh8iGUgA8\nAwcmx1dXOFWW8woyUi7+RkPbAHB5ClqdnzvD1rjA7edv23NXIBB0DXfccS9nnHEWXq+Xfv36M3Xq\nubz33jutxrjdbjIyMrjwwkv48ssvCAZTe6jLyspYtuxzTj75lC6zV3hwBd2OJy5aIzETj+ImZITj\nRaYCyTFWJIKSkdFa4KbIw5VbenDTVGgqEaIMTpiySxbrQgKBQCDofWwoCwJQlOejLOyI08HZA1Bk\nhcj69QB4hgxNjq8ocwRxXmEGqYiFygDQfMVps3FrkxC4gp6NKqvcMOrKTocoB7L91NelN0Q5FcFg\nkAUL5nPeeb/HF4/eiMViaJrzHZw58yJOO+0MDj/85wAYho6iKHg8qQvPvfPOO4wceTDZ2elrHbYj\nQuAKuh0tHnYcjRl41LjANZtDlMEJU24jcFN4cKWWObhpahXUUuAmClsIBAKBQNDbWLfVEbiDi7Io\nrV8KwNDcQZihJmLljlhNCFzTtNi+zRlf3D/QZi7btomF4lWX0yRwI4aZzMHt5+t8CpJA0F2oskqB\nb/fD9hVFIjcjAy3mxeziNpYZGRm899472LbNjBkXU15exoIF85k48ZcA7LXXCJ54Yh777rsfiqLw\n0EP3cdxxJ6BpqaM2li9fzn77HdClNgtXlKDbcbtae3ABomYUqcXKT6KScssc3JQCV5KRJGfdxrLS\nI3A1ubUHVyAQCASC3oZuWKze7OQEDh3gY0NwMwD7Fe5NaNVqZ5Ak4SkpAaCyvAHTcM6JqQSuHt6O\nZTgFq9wZg9JiY2ljGBsnhHpghhC4AsGeQJZl7rjjXtatW8uJJx7DhRf+jqOP/gWTJ/8agPPPn8a+\n+/6EqVNP5+yzp1BUVMzMmVcBTu/b8eN/Tnl5eXK+rVu3kpeXOtc3XQgPrqDbaRa4Bm6vI3AjRhTF\n28KDG6+krKgykgS23X4vXEnRnLzdNAnclh5cXfTCFQgEAkEvZP22emK6c45z59Ri19hISPykYC9K\nn58HON7bRIGpLRscMZyR5SYz0FZsRhqcnF1Z9ePy9EmPjUGn6GRfnxuvKDAlEHQZI0cewiuvvJW8\nX1IyhPvvfzjlWE3TuOyyK7nssivbHCsqKuLNN99vte8f//gHNTWNXep5Fh5cQbejueJteHb04Got\n8mljTs6NJEk774Ub9+AmWhN0FrVF4QxdhCgLBAKBoBfyTWkN4PS/3RzZAMDgwAC8Lg9NX38NgH+/\n/ZPjN6ytAmDQsNRhlpGG7wDwZA5JS/cB27ZZXe9UbR6S1TWVVwUCQe9ACFxBt+NO5uCaaPEqizFL\nR5JlpHhvLVtvFqtJgdueB1d2+uLZlpEW+2TJCYcCMITAFQgEAkEvw7Zt/reyAoD9h+bwZYUjaPfP\n34em0g3oVY6Y9f3EEbiNDVEqy50KyoOHtQ01NI0wkYYNAHizhrY5vjtURGJURZxrgX2z2/bcFQgE\nggRC4Aq6nZY5uK54lTfddE5ikisuVmPN4caqa2cCNy6K7fQIXEmSUGVH4goPrkAgEAh6GxvKG6io\nc8J/iwaGadAd8Xpw4YFUvrsEADU/H8/gwQCsW7EdcBac+w1sWwk1XLcSsJAkFW9g77TY+E2NY1Om\nS2GAyL8VCATfgxC4gm6nZQ6uS3EErW61FriW3kLgxnNizXYKPjUXmUpPiDKAKy5wDVvk4AoEAoGg\nd/H+V06F5D7ZXkqj3wIwMLMffdw5VL7n5M9ljRqNJMvYts3Kr5yCMcP26YOitr2UbKpZDoAnMBxZ\n6Xw7H8u2+aLaqdj8k5wM5DSEPAsEgt6LELiCbqdlDq5LTghcx/sqxUuM27FmsZo4mbYrcBO9vtIU\nogygSs5zihBlgUAgEPQmmiI6H33jCNyfHZDFskonPPnwvocR/OxT9No6kCSyRh8OwLZNddRVO9WR\n9zmwbfufWKiMaJNTgTkj98C02Li2PkRt1DmnH1rQtmKzQCAQtEQIXEG344oL1phhoiUEbjxEWXbF\nBW6LHFwl4cE1duLBTVOIMiBClAUCgUDQK1m8dAsx3UJzyURz1mHZFj7VyyGFP6X61UUAZPz0p2iF\nRQB8/tFGAAqKMulTnNlmvmDFxwCo7lw8WcPTYuP75U7F5kEZHop9nfcICwSC3o0QuIJuxxUv9a8b\nFqoSz8H9nhDlpAe3PYEbF8np9OA2hygLgSsQCASC3kF9U4zXPtsEwKiDsvik/BMAjhpwBLHPvyC6\nZQsAecefADje260b6wA4eMygNtWRY6EyQrXfAJDZZ3Raqid/FwyxvsHJDx5blNPp+QQCQe9HCFxB\nt+NS4t5Rw2r24O4YopxC4Bo7CVG20hqiHBe4woMrEAgEgl7C8++sIxoz8bplQnlfYdgmGS4/Rxcc\nStW//glAzqEH4xs+HNO0eO+NNQDkF2YweHjr9kC2bVO79Q3A8d5m5B3UaftMy2bRZqeCc3+/mxGi\nerJAIOgAQuAKup1E0SjdaJmDmwhRTlRRbg5RVnfmwU30wbXTV2SqOURZFJkSCAQCQc9n2dpKPvrG\nKRZ18M90vq1ZAcCkoRMI/vNfGLW1oCiUnPtbAL74aCO1VU7u7c+PHd7GO9tY9T+ijU74cnbfXyBJ\nSqdt/GB7LWWhKAATBhSkxSMsEAh6P0LgCrqdRA6ubli4lNRtglqFKO8sBzfNfXChRYiy8OAKBAKB\noIdTXhNi3ssrARg82OYbw2kFtE/uXvykNErwow8AKPjlqXj79WXT+hqWfuiI15/8tC9F/VoXeoqG\ntlG3dTEAvuyf4Mse0WkbNzSEeXNrNQCHFmRRkunt9JwCgaBjrFjxDZMmHZ+8v2rVCsaO/Rnjx/88\neVuwYD4AwWCQG2/8IxMmjGPChHHMnn09jY2NKeetrKxk+vTpHHvsUUyadByPPPIQVhc4j4TAFXQ7\nzR5ca5eqKBvtCtz4qnE6i0zFqyjrIgdXIBAIBD2Y2oYo9z2/nFDUwJ+lE+n/KVEzRpaWyWR7P7Yv\neBwA74h9yJswgW2b63j1305ebV4fP2PGDW01nxGto+q7Z7FtA8WVRc6ACZ22sToS45nvyrBsyPe4\nmDAgv9NzCgSCnWPbNi+/vJDLL/8/DKP52nvNmtWMGjWGN998P3mbOvU8AO677y5A4j//eYV///tl\namvreOyxR1POP2fOnQwcOJBFixbzt78t4K233uT11xel/XWoaZ9RINhF1BYeXG2HEGVJjYcbm81i\nVdlpH1xH4Npp7Fkbf0osIXAFAoFA0EOpqAtzz7PLqKyLoHib8O23nPpYEJes8nvpZwQfnQemiVbc\nl77TL6J8WwOLnv8aPWbi9bs47pf7obqaQ4/1SDUV657ENBqRZBcFQ85AUX2dsrE6EuPvq7bSoJto\nssRvhhXjUTof7iwQ/JCwDQO9pma3H68oEI42EqtvwjS/f6wrNzd5Pb0zFiyYz9tvL2bq1PN46qkn\nkvvXrFnNsGF7pXzMrFk3YlkWmqZRWVlBOBwiEMhOOXbTpo30798XO349LcsSbrenQ7btCkLgCrod\nV0KwWjZyXJyatvNtTQpco/nbu7MqyiQF7k6+8btAoqm8KQSuQCDo4SxatIgHH3yQ8vJy+vbty8yZ\nM/nFL37B119/zZQpU/B4mi82pk2bxvTp07Ftmzlz5vD8889jmiaTJk3ij3/8I4oQHj2GL9dV8ff/\nriAUNXDlVeAd9g2NRgwNlWmVwzHeeBZsGzU/n76XXs7a9Q289/paTNPC7VE5+fQDCeQ0hwmH69dS\nvfEFLDMSF7eno/mKOmXjd8EQT68rI2xauGSJc/bqR6FXtAUS9C5sw6D0umswqqr2yPOp+fmU3HJ7\nh0TuiSdOYurU81i27PNW+9euXY2maUyePBHTNBk3bjwXXHAhmqahxue97babefXVlykpGcIpp5ya\ncv6zzjqH22+/hWeeeQbTNJkw4STGjftF51/kDogQZUG3k8jBBbDtuJC04gI3fvHU0oObLDK1Ew8u\nVvoErhIXuKLGlEAg6MmUlpYya9Ysbr31VpYtW8a1117LZZddRk1NDStXrmTs2LEsW7YseZs+fToA\nTz31FO+++y4vvfQSixYt4osvvmD+/Pnd/GoEHSEYivH3l1fwwL++ImQ24R3+NerQL9DtGAMbVGZ8\nrCK/vgRsG/eAgeT931W8/V457yxajWlaZOd6OXXqSPL6ZABgmRFqNr9K5fpnsMwIsuKhYOiZeDKH\n7LaNumXx2uYq5q/eSti08Coy5wzvK/JuBYI9TH5+fspibtnZ2Rx++M9ZsOA5HnxwLl98sZR58+a2\nGnPFFdfw6qvvMHDgYGbNujLl/LZtM23aNBYvXsKTT/6T5cuX8eKL/0776xAeXEG3k8jBBZDseH6t\nbWLbNlK86BRGs8CV422F2q+i3AUhypLogysQCHo+JSUlfPjhh/j9fgzDoKqqCr/fj6ZprFixghEj\nUhcHWrhwIeeccw59+vQBHM/u/fffz+9///s9ab5gF2gM67zxv00sXrqFiBVG7b8BrWgTtmyQHTQ4\nap3MoNVlED+veQ4fx9aBo3nj2VXJGheDhuVx2lkHE9V1dD1GU9UX1G//EMtwCsi4vMUUlExGdacO\nR9wZlm3zVU0Di7fWUBN1UpP6eDTOHl5MnkdLw7sgEPzwkFSVkltu73SIciDgpz7NIcrtcccd9ya3\n+/Xrz9Sp5zJ37l+YMePi5H63243b7ebCCy9hypRJBIP1ZGU1F6SrqqrizjtvY+nSpTQ2xigpGcJZ\nZ/2WF1/8d7se391FCFxBt6MqzStFCQ8ugGVbLXJwW4QoxwWxZbYjNuMFodIZopwwUeTgCgSCno7f\n72fz5s0cd9xxWJbFTTfdREZGBitXrkTTNMaNG4dlWUyYMIGZM2eiaRrr169n2LBhyTlKSkooLS11\nFiI70LpFkiTkTsaMvfHaf8mzV1Fbr/Czk39Hlj+rcxPuQeR4Jf7E367CtCxWbazj/eXbWLqqAtNX\nhdJ/K57cclTLpGRLlJ9s0Bm0NYxkgw00FY+geu+xlJYZmNu3AuDxuhh99BD2G9kXRWmgdsOHNFQu\nwzSaAGchObt4LIGiw5sLO+4CIcNkWWWQj7fXURmJ19wAjuybwy/65+Hq5IdlT73f6UbYvWfpVrsV\nF2px4W4/XJYlvNl+ot4mrC7o8KHEL3wVRSIYDPL44/M4//wL8PudXtSGEcPtdqMoEpdeeiGTJ5/B\nEUeMBcCyDBRFwe/3JecBqKurwjAMdF1Pvucul4qqKq3GpQMhcAXdTqsQZav5A27aprNEhZOvkCDx\npWjvC50MURY5uAKBQJCS4uJili9fztKlS7nwwgsZNGgQOTk5HHbYYZx++ulUV1dz6aWX8sADD/CH\nP/yBcDjcKjfX6/ViWRaxmHORszPy8vyd7mHauH0jw38SIrcAVi+ZS4U+mLPPPbdTc+5psrP9aZ3P\ntm3KqptYtaGGpau288V3m4i4KpADVSgHVJIbjTCgPMbAVTEGl8XQdBtd1qjyDaAuZwhVWSWEYsAW\npxWf5lY49PBBjDzURzi4lvLVrxIKbml+Qkkmr/hgioccg9uXt0u2NsQMvq6oZ9n2Or6tDKK3OIcf\n2CfAKXsV0z+rcwWqdiTd7/eeQti9Z+mpdkPX2Z6Z6UWSJHJzM8jO9vHBB0vweFxcccUVbNu2jSef\nfJwpU6aQm5vBgQcewD/+8ThHHDEKRVF45JEHmTRpEkVFua3mPPjgAykqKuKOO+7guuuuo6Kiguee\ne4rJkyeTm5uRVvuFwBV0O64WIcrYzduGZab04HZU4KbXg5sQuGmbUiAQCLqNRFGQ0aNHc+yxx/LW\nW2/xyCOPJI/7fD6mTZvGnDlz+MMf/oDH4yEajSaPh8NhVFXtkLgFqK5u6rQH96iJv2Hlu/PpO6CR\n/D4h8lnB4mduoKZWJRh1YbkzGT7iQIYN3yvpZfihIMsS2dl+6up2z9ti2zYNIZ3ymhDbqprYXF3L\n5trtbG7YTkwO4nHVkmPXMsQXpqDWoGCzQX6tgRZz06hl0+TOYV1ODg3uPBrdzRedsmGRmRGmsMhk\n8BCLzIwgevhD1n8Za/X8iiuDjLwDySw4GJc7h6YINEVS97kEJ6e2MqxTFoqyoSHMhoYwFeHWc6qS\nxEH5mYwuzKZ/hgcMi5qa9ufcFTr7fncXwu49S0+1G7re9oaGMLZtJ7+Td9wxhzlz7uKwww7D7fZw\nyim/4uSTT6WmppGzzjqPurogJ5xwIooic+SR47jookuoqWmkvLyMM8+czNNPP09RUTF3330/Dz10\nL4cffjg+n4+TTz6Fk0761W59979PFO9RgbtixQpuuOEG1q1bx6BBg7j55ps56KCD2oz75z//yd//\n/neqqqooKSnhj3/8I4cccsieNFWwB1FaClyredu0zeYiUylycK12ikx1RRVlRXhwBQJBL2DJkiU8\n9thjPP7448l9uq5j2zZ33HEHF110ERkZzkVDNBpNCtihQ4dSWlrKgQceCDjFqoYM6XhRIdu2d5on\ntjMCWdkcdOx03vr339hrSAi3RycnL0xO0pFYA8ZGyr6R0WMKuq5gxGQMQ8I0wTQlDAMMS8K0JHRT\nwrTAsCVMW0ZHwpJULFkBRUNyudG8fvweD363G7/Xj8+fgd/nx+X1oKgqsqwgyxIyErIkI0kSiiQh\nSTIJh7URP1dFTJvK6iDhmIFu6uimSdQwCEd1GmMRGiNhwuFGopEmYuEmouEmjEgTZqwJyQjjNmN4\nTQOvYeLRJfbWXewXc6HFXEi2hi73Jap6iah+Kjw+yoa4UV0WqmridsfwuGNkuevxuCvweGJkZERx\na2FaOtZjTc3bipaNLzCcwgEHYEh9seIRVoZhEbNsQoZJg25QFzWojxnUxQzqYjoV4Rg1UZ1UZ0tV\nkhiW5WOfHD/75WTgVeOdE7po9diy7C6buysRdu9Zeqrd0HW2H3jgwbzyylvJuQcOLOG++x7e4bkB\nbBTFxaWXXsmll7YuLGWaNgUFRbz55vvJ+4MHlzB//nxqahqTc9t2+n8D9pjAjUajTJ8+nenTpzN5\n8mQWLlzIjBkzWLx4cauV1k8++YQ5c+bw2GOPsffee7Nw4UKmT5/Om2++SU5Ozp4yV7AHaRV3b7cO\nUU7twY3n4O40RDn9RaZEDq5AIOjJ7LvvvnzzzTe8+OKLTJw4kffff58lS5bw3HPPJdsBJULQHnnk\nEaZMmQLAxIkTmTdvHqNGjUJVVebOncukSZP2uP1+v5/fXnYdX321kqVv/YeCLJ1AjoHXF016iBXF\nQvFaeLx6p5/PNGVMQ8YwZcwGGbNWotaUMA0J0wDDdMSzYYJhObeYKWHYEjEbYrZEVJKdGwq66sJQ\nFWzFhSSrqLKCioxLlnEh4ZIlXJKN3+0i05uNIgWQZZAkkCWQJAtZspHjf5EsDMlCli1UycajGOQp\nISCMjRS/gYWEjYyFQhM+GvFTiYRtS1i2hIkLW83CdgWw1SwsxY8puTGiNpTKNES20KRbhA2TsGl2\nOJpJkyX6+z0MyvQyOMPDwAwvbkU08BAIBF3LHhO4n3zyCbIsc+aZZwJw2mmn8cQTT7BkyRJOOOGE\n5Ljy8nLOP/989tlnHwB++ctfcvvtt7Nu3ToOPfTQPWWuYA+itkzu3yFEOdljMUUOrtmewJW7og+u\n81d4cAUCQU+moKCARx55hNtuu40//elPDB48mL/85S8MGzaMRx55hFtuuYVRo0bh8Xg4/fTTOeec\ncwA488wzqaqq4rTTTkPXdU4++WTO7cb81/79B1B81iXJ+5VVlfzvw7eJNdUi2zqaZOFSbTTFQlFA\nVW0UBRTVRlFsFNVCUWxU1URRLWQ59W+7olgoikU66/kahoxpKhiGjB5TMWIyekxGj0kYUTB0CyNm\noesm0ZhJKGYQky2iqk3IJRHS3IRVLyHVR0z2gu0GXEi4kWw3ku1BkhUkRUJSZSRVQlZlJJeMrMhI\nrvh9VXbGJFy4sfit7Z3/b+/Ow6Os7sb/v+9l1uwLWSAJhLAjEhYVBMQdxQV9lNraalta17b+Hq1t\n1adatb8+tYtb9bG1Lr2K+tTvV21d0bohOyqKILuQARJIAtm3We/7fP+YLBNIJEjIJPHzuq5cM3PP\nvXwy1yRnPnPO+Zwv5dQ1UpwmKU4HKU6TIW4HWR4X2R4nKU6zvYaFEEL0lT5LcH0+H0VFRZ22FRYW\nUlJS0mnbJZdc0unxJ598QnNz82HHdqc3KjWCVITrS05HRwVGLXZpZs1GdzqA6Dq4bT29pqO1SrKt\nuqy6phsdCW5vVWVrmydsq849zgPx9QaJu68N1Lhh4MY+UOPuC9OnT+ef//znYdtHjRrVaehyLMMw\nuPnmm7n55puPc3RfzZDMIcxfcMVXPj4cDNBQd5D62loaG6pprq8nFGjGCgfADqMpCw0r2lOqtybJ\nbT9m7K2NYUaHBXeX15mmjWnauFxAQrDrnQ4RDDoIBFz4A07CfoNIM0QaQlj1LYSbgthaC2FXM35P\nC00J0OB20aC7qdMSCUY8qBYXKuRBBT2ooBcV9ES/UNbAdBqYLgOn28TlMnC5Hbg9Jl6vg6QkJ1mZ\nibhNDY9h4DENvIaO1zRwmzrJDhO3oR9zATEhhOhNfZbgtrS04PF0XrDb7XYTCAS6PWbnzp3cdNNN\n3HTTTaSnp3e7X6zeqNQYa6BWVhtIcUdi5tLGvkcSkpw4kxOoBDSl2ieTJyc3ANEhyl1NMDfsBA4A\nKJu0tN55PyTVtU5M0rUurzmQXu9YEnffGqhxw8CNfaDGLfqWw+UmIzufjOz8XjmfUgplRwiHAgSb\nG7ECTZiEqa+uJRz0Ew61EIn4sW0/SgXRtBCaHsEwIhiOCA6H1alX2eUK43KFSUnp6lrg93toakmn\nsdGLv9YkfMBGr2shKVCHrpUS8DZRm6xTm2JSnWNwINUkpKKJrvInEW5JJNSSREN1IliOw67hdhpk\nprjJTPGQk+5l2JAE8oYkkpZhSnIrhOh3+izB9Xg8hyWzgUAAr7frkvAr+mi4zgAAIABJREFUV67k\n5ptv5vvf/z7XXnttj6/TG5UaYeBWVhuIcauYYb8tzR1zpmrqGkkORocmW6Fwe4W1lpboN96W1XXF\nRX9T27AqRU1NI5p27G+IYGv1x1DE6nTNgfh6g8Td1wZq3DBwY+/NuHt7+QIx+GmahmY4cHkcuDxJ\nGEb0y9HYwipfJpogB7HCjYQC9QRb6gi21BEK1BEJ1oNdj2k0o2kKTQOvN4DXGyArsxYKo+eIRHQa\nGhOpq8+hqSYJVWEzdHc94wMHSAlU0OKt5UCGg8p0k/JMB1XDTZSuYUS8aIFUQvXJhOtTsJuTCYSg\n7GAzZQebO8WpaZCV6iE/O4miockUDU1heE4iDvPo18YVQoje0mcJ7siRI3n22Wc7bfP5fFx44YWH\n7fvSSy/xm9/8hnvvvbfL579Mb1RqjDVQK6sNtLgNXcOyFbbV8U1wKGKhtI4qyu2/T1vBJ6vr39GO\nnccbiaDrh38b/VVZ3byuA+31biNx962BGjcM3NgHatzi6y2aILvRDTcO9xASUg/fRymbSKiOSKCa\nUKAaf2MloZZKVOQgmmZhmjbpaQ2kpzUwckT0mIbGBGpqc9l1cAJNlSYpTQcZd7CcWev3YetB9mU7\nKM1uoSy7geqhBq5hGoZmkG5mkWwPxWzJprEqgf1VLfiDFkpBZa2fylo/67YdAKLteUF2EuMKUplQ\nmM6YvBRJeIUQfarPEtyZM2cSCoV45pln+OY3v8krr7xCVVUVs2fP7rTfmjVruOeee3j66adlaaCv\nkbYEN7YuVLSKcmuCa3UUmYqdA2vb6rA5du1VlKMHAsee4JqyTJAQQoh+RNN0HK50HK50PCmjScmO\nblfKJhKsJtRSQbBlP/76PURClWgokpOaSU5qZkTBfixLp6o6lQMHi9h14CRcDQ1kNpcx7fNS5n5S\nQ3Oiyc6hDkryXOzL2s9BvRxc4C5wceLkIkYmjCbZyqe6RrG7vIFd+xuobQxi2QpfeQO+8gbe/HAv\nDlNnTH4qkwrTmTpmCJmpni//xYQQ4hj1WYLrdDp54oknuPvuu3nggQcYPnw4f/7zn/F6vdx1110A\n3HvvvTzxxBOEw2GuueaaTsc//PDDnHbaaX0VruhjpqETitioTlWUI2hG61u0i2WCAGzbRtc7fzMc\nm+Aq24Je+OJYb09wj/1cQgghxPGiaToO95Boz2/6JMgD2woSbC4l2LQXf0MJYf9+DMMmO6uG7Kwa\nTpgANbUp7CvPZX3lJMzmFnKafIzx7aJ4Rx2Wy2RvfgKf5WuUZis2Vm1hY9UWdE1nTGoRxSdP4qoh\nkwkHTEr2N/BFWT1b9tSw72Az4YjNZl8Nm301PP/+TobnJDF97BCmjc0iJ73raWpCCHEs+izBBRg3\nbhzPP//8Ydvvvffe9vtPP/10X4Yk+om2XlnLUmhoKBSWsjt6cCMRlFKtVbJjenAtdfi7OGbOraJ3\n1sI1pAdXCCHEAKUbLjzJo/AkjyJ16JlY4Sb8DTvxN3xBoGEX2CEy0uvJSK/nhPE7OViVxt6yfNZW\nTSbRX01O4y7ySnZRuDOEleRl36gM1uQGqEi12Vb7Bdtqv+D/7niZiRljmZF7EgvHjMPUR1PbGGTL\n7ho2767h813VNAci7KloZE9FIy8tK6EwN4lZk3I5ZUI2Ce7em04khDg2W7Zs4vbbb+WVV94CYNu2\nLVx77fdwuVzt+1x11fe5+upFNDQ0cP/9v+Wjjz4E4NRTZ3Hzzb8gMfHw+hX79pXx85//kfXr15Oa\nmsZ3v/sDzj//6Kaj9kSfJrhCdMdoTVptBYamE1EWlh2B2N5ZpUDT0A8ZonyozkOUeyfBlXVwhRBC\nDBaGI5HEjGISM4pRdgR/wxc0127CX78DXbfae3b9fiel+3IoLStmV2A62Y0+htVvJ399KQXrwcrL\nYeeEdJal1+HXI3xetZXPq7aS6Ejg5JypzB56CrMm5TJrUi4Ry2bb3lo+2X6Q9TsO0tASxlfeiK+8\nkeff28nUMZnMOXEoE0akSWVmIeJEKcUbb7zKo48+iGF0fJ7esWM7M2acyu9//9Bhxzz00B8AjX/+\n8w2UUvzyl7/gb3/7Kz/5yS2d9rMsi1/84qdMnnwir732FuXlldxyy49JTU1l5szZh533WEiCK/oF\no3XYsWUpdE0HZWErG83o+EZXWRE03dm5B7er6qixPbiqdyqOtfXgSn4rhBBiMNF0E2/qeLyp47Gt\nIC1122iu+Yxg0x48nhBjRu1ldNFeKg9kULI7j3V1RSRZDQyr3kTOvhLGllUwPiGBluIxfFio8Zld\nRlO4mfdLV/B+6QompI/l9PxZjE8fwwmFGZxQmMFV545l695aVn1ezifbDxKO2Hy09QAfbT1AboaX\nM6fmceoJOXhc8jFViL60ePHTvP/+u1x99SKee+7v7dt37NjOqFFjujzmjjt+hW3bOJ1ODh48gN/f\nQkrK4ZXxSkv34vOV8OKLLxAI2BQUDOfSSy/n9ddfkQRXDE5ma69sxLIxdAPscLTIlO7u2MmywXHI\nHFzr8B7aTssC9VoPrgxRFkIIMbjphovEjMkkZkwmHKiiueZTmms2YoVbyMmuJie7mtq6JEp257Hd\nPBVf9skMq/qcvPptuFetZ+5qjXOKT2RncQ5LKaEuWM+Wmu1sqdlOlieTuXmzmDn0JFyGk4kj0pk4\nIp2WcyJ8vK2SFRvLKdnfQHl1C8+9s4N/Lt/FrEm5nD09nywpTCUGGcuyaWoIfuXjDUMDW6O+3n/E\nlQISk10YRs+WzLzgggVcffUi1q//pNP2L77YjtPpZOHCi7EsizPPPIdrr70Rp9OJaUbTyf/+73t4\n883XKSwcySWXXHbYuW3bwjAMnE5n+9KxmqZTVlbao9iOhiS4ol9on4Nrt/bgApaywTikYBQccYgy\nxPbg9tYc3NbzAbZS7QmvEEIIMRg53Jlk5M+j6ISLKS35iPqKtYT9FaSlNjKteCstLW5KduexWytm\nT+YUhjWXkHdgPazfQMH6DVw3dhy1M2fxnnsfuxp2c8BfxQtfvMKS3e9wRt5s5uaditfhxes2mVs8\njLnFw/CVN/DeJ2V8tLUSf9Di3XVlvPdJGSePz2b+jOHkZ8ma1GLgsyybf/z1IxrrA31yvaQUN9+6\n9uQeJbmZmZldbk9NTWXKlGksWHAZNTXV3HnnbTz11OPccMNP2vf56U9v46abfsp99/2aO+74GY8+\n+tdO5ygoGEFu7lDuv/9+rrrqB5SXV/Dqq/86tl+uG5Lgin6hfYiyrTBa59BaykaL+WNUrZWUY4co\nd/Wt1fHswYXWYcqS3wohhPga0A0HSRmT8aRMItjko6FyDYHGXXi9AU6YsJNRRXvZVZLP3rIRlCYU\nMSxURsG+tbB9G57t27gsPx/77LNYkVrLugMbaA638Lrvbd7Z+wGnDTuVM/LnkOJKAqAwN5kfXjiB\nb5wximWf7eP99fuobwrx4ZZKPtxSyeSiDC6YOYJReSlxflWE+Hr53e8ebL8/bFgeV1/9fR5//H86\nJbgulwuXy8WNN97EN76xgIaGepKTO/5WTdPkd7+7n0ceeYAFC+YzYkQh8+adz+rVK3o9XklwRb/Q\nVmTKsuz2HlxbWWgxPbi0DkeOTXBVl3NwY3p9e6mKcmyCaymFIRmuEEKIrxFN03AnjcSdNJKQ/wCN\nB9bQXLMRtyvExPG7GFVUyq6SPPaW5bKv8HKGqUry96yE0lL42z84bVge5553Hqsy6lm5/0OCVoh3\n9n7A0rKVnJp7MvNGnEGqK/phODnByUWzCjnvlOGs2lTOW2v3cqDOz4Zd1WzYVc3Y/FQunl3I+OFp\ncX5VhDh6hqHzrWtPPuYhyikpXurrW3p1iHJXGhoaWLz4aRYtugavNwGAUCiE0xmtqHzzzT/i8su/\nyaxZcwCIRMIYhoHb3XlqgW3b+P1+nnzyyfah1X/+8yOMHj32K8fWHUlwRb9gtv7hRSwVnYMLWLbd\nqYpyew+u0Xkd3EMdjx7cmFHR2Mg8XCGEEF9fTk8WGcMXkJJzGvWVK2mu3oDLGWLCuBJGjSxlly+P\nPaVD2TfiMoZpVeT7lsG+MkJP/Y1pw/I4bf75fJjZxAf71uCP+Fm+bzWryz9iztAZnDP8jPYeXYep\nc3rxMOacmMvH2w6wZM0eyg42s720jj/8Yz3jClJZMLuQsQWS6IqBxTB0UtK++txyw9BIT08AXR0x\nwT1WiYmJLF++FKUUN9zwEyoqylm8+GkuvvhSAMaMGcff//4UEyacgGEYPProQ8ybNx+n09npPLqu\nc+edt3PNNT/knHMuYP36T3nttZd54IFHez1mSXBFv9AxB9fGMNt6cO1OPbjtc3DjUEVZj+mx7XLa\nrxBCCPE1Y7rSyCi4iJTsOTRUrqSp5jOczjDjx/ooGlnGrpLWRHf4ZQzTYxLdJ57khGF5zJx/Pp/k\nB3m3dDnNkRaWlq1k5f4POW3YTM4ZfjpJzuicW0PXmTEhh1PGZ7NhVzWvrfLhK29k2946tv3vesYP\nT+PSOSNl6LIQx4Gu6/zudw/y0EN/5IILzsLlcrNgwX+wcOG3APjBD67jscce5uqrr0DXdebOPYMb\nbrgJgIqKCq66aiHPPPMCOTk5/PrXv+WBB37H73//e7KycvjFL37JuHHjez1mSXBFv9A+RNlW6O1z\ncK1Oc3DpYg6u3WdzcGPCkErKQgghRDvTlUp6wYUk58yhoXIVTdXrcTq6SHTbenRLoolu9RNPMCYv\nn2kXzOfjzGbeLV2BP+LnvdLlrNi/ltPzZnFWwWkkOqLDIjVNo3hUJpOLMtiwq5qXV5Swt7KJrXtq\n2brnE04oTGfBnELG5B++RIkQouemTp3OG2+81/64sHAkDz/8WJf7Op1O/vM/f8Z//ufPDnsuJyeH\nd97pmGM7fvwEXnrpJWpqmo5rz7MkuKJfiF0H19A6enAxOt6iqm0ObhyqKMfOwZUeXCGEEOJwpjOF\n9Pz5JGfPak10P+1IdAv3sbMkjz2luewrvIw8rYr8kg+grJTqxx9nfH4BJ10wn7Vp9bxftoqAFeDt\nPUtZXraa0/Nnc1b+HLwOL9A50V3/RRUvr/BRdrCJTb4aNvlqmFyUwXcvmkhGgiO+L4gQIi4kwRX9\nQqd1cNuXCbLQ9NgqyhHgkHVwu5yDqxEtc6yOSxVlW3pwhRBCiG51SnQrVtJUsx5n6xzdjh7dXMoK\nLyefg+T5lkHpXoJ/+QsnFAznlAsvYE1yDUv3rSJgBXlr93t8ULqKM/Jnc2b+HLyO6NxFTdOYOmYI\nxaMz+XT7QV5Z6WNfVTMbdlVzy0PLKR6VyUWzRlCYmxznV0QI0ZckwRX9Quw6uJ2XCYqpomwfXkW5\n2+ENmg7K6rUqyp2KTEmCK4QQQhyR6UwhveACknNaE93qz9qLURWNLGNnSR57S3MpLbycfCrJ9y2H\nvXsIPvZnThxRyMz5F7EyqYpl+1YTsAK8uftdPihbyRl5szkjJtHVNY3p47KYOnYI67Yd4JWVPsqr\nW/hsZxWf7ayieFQmF88ewYgcSXSF+DqQBFf0C7Hr4LYvE2RbYBxeRRmiw5RtS3UzRDk6D1cp67j0\n4B7nYnVCCCHEoGI6W+foZs+mvnJFe9XlieNKGFVYxs6SfPaW5VJWuJB8VUHe7uWw20fwsT8zdeRI\nZs9fwPKESpbvW4s/EmDJ7ndZWraSM/LncGb+bDxmR6J78vhsTpmQzea99Tz31lZJdIX4GpIEV/QL\n7T24lt25B7fTEOWYBFdvTXC/rAeX4zUHVzJcIYQQ4miZrtTWqsuzW5cX+gxX6zq6bUOX95blUjry\nG+Tb5eT5VkBJCYFHH2N60SjmzL+EFZ4Klu9vTXR977C0dCVn5c/h9PxZHYmurjF3ah4TC1JYs7mC\n11btPizRXTC7kOE5SXF+RYQQx4MkuKJfMPWOdXD12Dm4sUOUD0lwoes5uACaZkRXqz0e6+BKgiuE\nEEJ8ZR3LC82mvmIFzTUbcLuC7YnuzpI8SstyKS36BvnWfvJ3r4BdOwk8spOTRo9hzvmXsty9nxX7\n1+KP+Hnd9zbvl67gzPzTmJt3KklGtBiVrmvMmJDDyeOy+WhbZZeJrvToCjH4SIIr+oVO6+DqHT24\nnYYoxySz0UJTFqrbksZtPbi9tA6uVFEWQgghepXpSiNj+MUk58ymoWIFzTUbcbuCnDB+F6NGlrFz\nVz6lZTmUFl1BgbWPYbtXwRc78H+xg5NGjWb2WReyIuUgK/d/SEvEz+u+f/PO3qXMyZvBZSeeh0a0\ninKnRHdrJa+u2k1FTUeiO354GufPKGDiiPTWQpVCiIFMElzRL5ht6+BaCjNmmSBN00DXwbYPm4ML\n3S0TFLMWrqyDK4QQQvRrDlc6GcMXRNfRrVhOc83n0UR3wk5GFZXi2z2UvWU57Bn5DXKtSvLKPoSd\nXxDY+QXTc3KZdeZcVuf4WXFgHUErxLt7lrO0dBUn50zhrPy55CZkA62J7sQcTh7fOdGNrqNbS35W\nIuefUsBJ47Paa4MIIQYeSXBFv2AYrUOUbYW3bQ6uHU1oNV2P9t52OUT5CHNwe62KckwPbq+cUQgh\nhBCxoonuJSRnz6G+YgUttdFEd/xYH6NH7aW0LJvde4axv+BiMrR68vZ9REbFPkL/+zzFycmcevpc\nPitysrTmE5rDLazZv441+9dxQsZ4ziqYw+jUIjRN60h0J2Tz2RdVvPnhHnbta6D0QBN/fW0LLy0r\n4dyT8pl9Yi4el3xUFmKgkb9a0S8YemyRqY4e3OiTJkQiKLuLBLebIlO93oOLFJkSQggh+oLDnUHm\niEsI58yh8eCHNFd/hmlEKBy+nxEF+6k8kIFvzzA2qLNJ0oMMrfyMnIZdWK++xiink+KTT6Z82lD+\n1biBmkAtm6q3sql6KzkJ2cwdNpOTc6biNt3orevoTh0zhC/K6nhz7V4+21lFdUOAf7z3Bf9cUcKp\nE3M4Y+ow8oYkxvtlEaLPbNmyidtvv5VXXnkLgG3btnDttd/D5XK173PVVd/n6qsXdTruySf/wpo1\nq3jqqWe6PG9DQwN33vkL1qxZQ0JCIosWXcOFF17S6/FLgiv6BbN1yHHEVujtVZRbe3ANHcWhywS1\nLSvUTQLb61WUO+7LEGUhhBDi+HO4M0jPn09K7uk0VX1C08GPsSJN5GRXk5NdTVOTh71luZQY09iZ\ndTI5DbsYVrsVtXIliSvhmsJCaqdM5u20A+z2l1PRXMn/2fEyr+x6k5NzpjE3byY5rcOXR+elMvry\nVPZXNfPWR3tZu7mSYMhi6fp9LF2/jzH5qZw5dRhTxwzBNGT4shiclFK88carPProgxgxdXB27NjO\njBmn8vvfP9TtsZs2fc5zz/2dkSNHdbvPfff9/yQlJfDGG++wY8cObr31/2PEiCJOOGFSr/4ekuCK\nfqF9HVxLtd9v68Ftr6RsxRaZOsIcXFqP6aUEV9M0dKLDk6XIlBBCCNF3DNNLSs4ckrNOpaVuMw0H\n1hL2V5CY6GfCuBLGjvZRUTmEvWU5fJQ0ihSrnqFVm8navRuPz8elCQkw9QTW5+usxEfACrJ832qW\n71vNyJQRzMidxtSsE/GYHoZmJrBo/ngWnl7Eyo3lLF2/j6r6ADtK69hRWkdygpMZE7KZPSmXvCzp\n1RWDy+LFT/P+++9y9dWLeO65v7dv37FjO6NGjen2uJaWFu67714uvfRyNmz4rNt9li//gH//+9+4\nXC4mTDiBc86Zx1tvvSEJrhicOlVRjlkmCIDWqsqHroML3Q9R7ujB7Z0qyhCtpGwrJUOUhRBCiDjQ\ndIOE9BPxpk0i1LKfpupPaandhEGYYUMPMGzoAZpb3Owvz2Jv8hS2N88ks3EPuY27SF/xEZNRTBua\nS+X4PN7PrGO/0URJ/W5K6nfzwo5XKR5yAjNypzMmrYgkr5PzZwxn3skFbCyp5v1Py9hUUkNDc4i3\nPy7l7Y9LGZ6dxKxJOZwyIZskrzPeL48YQJRtEQnXf+XjbV0j2BIgHGjBOkLPi+lIQdONL92nzQUX\nLODqqxexfv0nnbZ/8cV2nE4nCxdejGVZnHnmOVx77Y04ndH3/SOPPMC8efPJyMjsNsEtK9uLYZjk\n5+dTU9MEQEHBcJYtW9qj2I6GJLiiX2ibgxuxFEYXQ5SBrufgdltFuXVMcS/14ELrMGUlQ5SFEEKI\neNI0DVfCMFwJw0gbdg7NNZtoqv6EsL+SBG+A0UV7GV20l/r6RPaVZ7GtYg7KD1kNu8iu8ZH+3qcs\n1DTsUcPZOdzDipQamlxhPq5cz8eV60lxJlOcNYmpWScyMmU4xaMyKR6VyYE6P6s/L2fV5xVUNwTY\nU9nInspG/s/7O5kwIp3p44YwZfQQEj2OeL9Eoh9TtsX+rf+DFao7pvOU9XA/w5nK0PE/6lGSm5mZ\n2eX21NRUpkyZxoIFl1FTU82dd97GU089zg03/ISVK5exe3cJt956O//+95Juz+33BzrN4QVwudwE\nAoEe/iY9Jwmu6BfMmDm1ensP7qFDlLtaJqjrBFZrTZJ7q4oytK2Fq2SIshBCCNFP6IabpCHTScyc\nRthfQXPN57TUbcIKN5GSEv2ZMK6E2rokKg5ksq3yDMKNGkOa9jCkbA+jvtjDGF0jODyHrcMMPh7S\nQj0NLCtbxbKyVaQ4kyjOmsSUISdSlDqCS+aM5OLZhWzfW8fqz8v5ePsBQmGbz0uq+bykmsX6dsYP\nT2P6uCymjM6Unl0xKPzudw+23x82LI+rr/4+jz/+P1xxxZU8/PD9PPjg/3Sas9sVt9tNKBTstC0Y\nDODxeHs9XklwRb9gxKyD29aD2568tg9Rjp2D2zpP9whDlHu3B7c1qZYeXCGEEKJf0TQNpzcXpzeX\njIJzcFBJ+e6PaK7dgrJDpKU2kpbayPgxPpqaPFQcyGTPwRlsrnGS0bSfjIP7mLB3P8V2EP/QdHZl\nG2zKDFOZ0cCystUsK1tNgullfMYYJmaMY0LuWH4wfAJXnjOGz76oYt32A3xeUkPEstnkq2GTr4a/\nvwUjhyZz4sgMTizKJD87sf2zhPj60nSDoeN/dExDlA1dIzXVS11d7w5R7kpDQwOLFz/NokXX4PUm\nABAKhXA6XXz88YfU1tbwwx9eBUA4HCYSiXDeeafz1lsfdDpPfn4+kUiE/fv343YnA7B37x4KCwu/\ncmzdkQRX9AtGew+uiunBbRui3JbgRtr3bxuirLr9o+7dKsoArZ3GkuAKIYQQ/Zim6SSnjyai5ZKW\nN59AYwkt9dvx12/HjrSQmOhnVGIpo0aWEo4YVNekUlVdxO6q6eg1zaS3lDNsyz4mBOqxvQ725brZ\nmqUoy7ZYF/mMdZWfoaExIjmf8eljGDOsiOvHjycS0diwq4p12w7yeUk14YjNrn0N7NrXwL9W+EhO\ncDJpZDoThqcztiCV9GR3vF8qESeabuBwpX/l4w1Dw+VNxBFoQu+us6eXJCYmsnz5UpRS3HDDT6io\nKGfx4qe5+OJLmTdvPvPmzW/fd8mS13jppf/b5TJBXm8Cc+bM5f777+enP72NnTt38c47/+YPf3i4\n12OWBFf0Cx1zcG0MvW0ObtsQ5dbe2JjhyB1DlPtmHVyI6cHttTMKIYQQ4njSdBNPyhg8KWNQ6gJC\nzWWtye4OIsFqHKZFTlY1OVnVALT4XdTWplBVN5md1QnoVc2k1h7gpP2VnB04SCjZZE+mRukQk31Z\nPpbU72HJ7ncxdZPC5AJGp47k3DOK+O75o9hZ1sTnu6rZWFJNTUOQhuYQqz6vYNXnFQBkpXoYW5DK\n2IJUxhWkMSTNE8+XSogu6brO7373IA899EcuuOAsXC43Cxb8BwsXfuuIx1ZUVHDVVQt55pkXyMnJ\n4bbbfslDD/2eiy+ej8fj4cYbb2LixBN6PWZJcEW/YLZXUe7owW1bJggj+jbtqopyt8MyenkdXACj\nNcGVIlNCCCHEwKNpOq7EAlyJBaQNO4dIsI5AYwmBxhL8jSUoK4DXE8TriVZkBgiHDWrrkqmtG8e+\n+ulYB8N462uZdKCKUz+tRjMbKM/QqUw3qczYxrvpO1ni1NE1nWGJuYwoLOCyE/PwWEPZX6axyVfD\nrv0NhCM2B+r8HKjzs2JjOQCpiS7GF6aTl+llRHYSI3KT8bjko7roe1OnTueNN95rf1xYOJKHH37s\niMfNn38R8+df1P44JyeHd95Z0f44JSWFhx9+mJqaJqzj2PMsfzWiX2gfotxVFeXW+badE9y2Objd\nFZk6Hj240VspMiWEEEIMfKYrlUTXVBIzp6KUTailnGDTboJNpQSa9qLsAA6HRdaQWrKG1LYf5w84\nqW9Ip6ahgMZ6N3Z1iCH7GigsqSUxVEvIWU91OhxMa6Q0pYTPUk0avTpu003+uGGcMTUHZySFploP\n5ft0SspaCEds6pqCrPm8vP06GpCT4SU/K5H8rESGDUkkf0gi6cmujtUihBCHkQRX9AvtRaZi1sG1\nD62ifFRDlI9DFWWkyJQQQggxGGma3r70ENmglCISqCLYXEqweS+BpjIioRo0wOMO4XF3DGsGsCyd\npmYvTU2ZNDV50Ro0MmtC5O5rxhuox7QbCXhqqEs5SG2KSV2SQW2SQWOGQU5BJilmBkY4iXCzl8py\nqK4yUWEn5dUtlFe38NHWA+3X8rhM8oYkMCwzgex0L9lpXrLTPQxJ9bSvSiHE15kkuKJfiF0HV2/r\nwW1b97a9yFTP18HtqKJsdf38V4lxgBWZspXCshRhK0I4YkdvLYtwOIytIpTXOairaSAUDBIOR7DD\nYcKRCFY4jB2xsCKR6K0VvVWWBXYElB0d+q1sUAqFQlMKZduggYZq7znXUIBqv227r6JPgta6VdNQ\n0YNR6ChNi341oWnYmoZCb7+PptN+9phvsJXWdtK2+wB69L4WjQBi99E6H6dFo6M1StqWUu64c5j2\n57S2Jw//Rr39GQ2U6ryPat1+2Pm6OL6D1uXd7o7vDVqnOHr4/o+7DKVkAAAag0lEQVRz74JmKyZl\nZjBz2ilxjUMIMTBpmobDMwSHZwiJmVMBsK0gIX8l4ZZygi3lBJr2Y4Wr0VAYhk1KchMpyU2dzqMU\nBAJOWvzDwO8mtdlBYiPkHQyj7QpgNjWhtAaCnoM0e0M0JNqYCTopeQaBBBe2Jw0VSSLY5KSp3sQO\nuQkEPXxR7ueLsjpiGwJNg4xkN9lpHrLSvWQku0lPdpGe5CYj2U1qkhNDlwRYDH6S4Ip+IfYbx7YE\nN9I2RNmMLpiuQqGOfdoS3G7G77cNUVZ27yW4evsc3KM/1rJtAiELfyBCUzBIY8BPU9BPSyhAwN9E\nKNBMOOAnEmjGCvixgwEIRlDhCIQttLBCtxSaBZqto6FhGqAbOg5TwzA0NFPD1EEzotX1dIPWHxW9\nNUEzbAwDdNOmVldohkLXFQ5doTSFoRtYmo7tMLBcBhHNwNIMIppJmOhPBJOwchHBwMLAQm+9jbmv\nDCIY2Oit+3XsY6NjIw2sOP6amyuYGe8ghBCDhm64cCcW4E4sIKl1m7IjhIPVhANVhP0H8DcdIOw/\niLLq0DQbTQOPJ4THEyKDw5eFUQqCoSQCgQwCQRehgEmkBexGBZURaAlihRoIWSECRpigO0TAHcKf\nEsHvchAwPTTgoVlLIGy7qQ27qG10srXGhQo7UWEXRByAhqZF5/m2Jb3JCU6SE5ykJDhJ9jpbHztI\nSXDiML/6sjJCxJskuKJfMIyObyAdWjShDVnRhFZ3RRdJt2MWh+7owe1mDq4ePUbZ4V6Jz1aqveOq\nqsHPhpaD1LW0UBdsImAHqa6vIdzSgO2vRws2YwT8mKEIZkhhWuCIaJi2gWEZ6LYDTRkYDhPdaaK5\nNZxuHbcLDBcYToUjUcdM1TFNA9ME04xgmjamGcFhWhhG17+3UhDGJICLIE4CqvU25nEAZ8z96PYw\nJjbdNGZtna/9lKY6eoY7+l9VawdxNPDYW03F3G/fHnOcarsfvT3seodtObRvOHbfL3vhYo47bLfu\nzxn7/Jf1kX75tY9RT08dx05cXdkMa/7qawwKIURPaLqJ05ON05MNaRNJbd2ulEUkWEckVEskWEug\nuZpgSw2RUB3Y9eha9POJpoHbFcLtCgFN3V4HwLJchEIJhMIOQiEHkaCOFdRQYYUK2dihIFbET8Sy\niVgWIWURMiIEjAgBFCEDAppGsFmjPODCV+UiRPQnbLtQthMiDpRl4jacJDg9JDjdJLidrT8mXrej\n9dYkwe3A6zZJ9DrwWxBoCeIwdFwOo/1zmhDx0KcJ7pYtW7jrrrvYuXMnw4cP55577qG4uPiw/V5/\n/XUefPBBqqurOeWUU/jNb35DZmZmX4Yq+pgZM2TGoUWT04AVTWh1V3SdODsQm+C2FZnqpge3NcG1\n7dBhz0Usm2Z/mCZ/mMaWEPUtAWpaGqgLNNEQaCLQVE/EXweBJvRAM0YohCNkUzPpDEjPofTzz1Bb\ntqHbDlBODN1BrssEj4nmcqO73ZjJNg5HBKcjjNMRweEIRx87wzgdQRyOyGFxHcpSOgGiCWkLnk4J\nacCKSVyVs32/IM7uE9WvSEfh0MCpgVMHp67h0HUcuobZet80NExNx9F6a+odjw1dx6HpmHrbj4au\ntf4QrU4dfRztJTda7xuHbgMcpk5mRiJ1tc3YlhowRTYMQyM9PfG4Vw08HgZq7LFxi86WLFnCI488\nQkVFBUOHDuXmm2/m7LPPpr6+njvuuIO1a9eSlJTEj370IxYuXAhE5yM+8MADvPDCC1iWxYIFC7j9\n9tsxDOnlEaIrmmbgcGfgcGcAkDSk4zmlFLYVwAo3YIUbCQfrCTTXEQ7Uo6xmQsF6NLsZXQ92mu1h\nGHZ7b/DRMbFtsCyj9UfHsnRsC6xIGGVFsCPN2JaGbSlsK/r5yrIUylZYQYUdUFgqOootrKDGhgMK\nLKURsaPbQ0ojgk7YBkvTsTBRhokyHGCaGA43DtPEdDjRTRdOhwPDdGA6HDhMBw6nE6dp4jRMnKaB\n0zBxORy4TBOnw8Bh6hi6jt76+cPQNXRdwzB0jNbHhh4d1aZr2oD5jCB6X58luMFgkOuvv57rr7+e\nhQsX8sorr3DDDTfw7rvvkpCQ0L7ftm3b+NWvfsXTTz/N2LFj+fWvf83tt9/OE0880VehijiI7cE1\nYnpwbWWjuVwAqJgeXKc7+qEq4A9TVeenKRCmvsVPdXMjdf5m0qxyitxQUVXOc2v/ghZswgz4MUNh\nnBFwWjoOy8CwHNEeVduJUzkYYjrB40Bzm2judHR3Oo5kG6czTMhj4AcSh3qYkA0ORwtORz2G8eUf\n+m2lEcRBEBeNJEYTUjuakMb2sAaUmyAugrgI4CDSS4mqy9DxGjpe08BrGnhMnQSHQUaSBy1s4dZ1\n3IaO09Bx6jpOQ8Oldzw2+9G3sIahYeo6uqbFzLMVQvSUz+fjjjvu4Omnn2bq1KmsXr2aa6+9luXL\nl3P33Xfj9XpZvXo127dv55prrmH06NEUFxfz3HPP8cEHH/Dqq6+iaRrXXXcdTz/9NNdcc028fyUh\nBhxN0zBMD4bpAU82HiB5yOFfKCplY1sB7EgzVqSFcLCJYEsToUAj4VAzVrgZO9yCbQXQVAhND6Pr\nEUzz8OlZug66buFw9N7UraOlVPQzEUrDVhrKbhuNp6GUBhaoiIZSoFT0ltb7ARv8rfdjzxd7i2od\nu6RiBhqpLvbrdEzbmC3VcXxX56abmhwah2870uvQfqetjkcXx2udr334BTuO66oKSE/DiX09j+pj\nVdtLcdTfe2uMKJ7LyLGTj/bAo9JnCe7atWvRdZ0rr7wSgMsvv5y///3vLFu2jPnz57fv99prr3HW\nWWcxeXL0F7/11luZOXMmVVVV0os7iCV5He339+4LtN//++rlDK06SCFQtX0rLzz9IEawBbMlExjL\ngfJa3nn0tzgsE8N2oFlOdOXCURSGKZBshiisTUJzp2AkgcNp4XSEcThbe1edYRyOME5nM05HGF3v\n/i91n5VDpRpCs9NLwHBRr5KiPah2W4LqbE1O3QRwE8RJUDkJ9tKfmalprUmqjqc1WfWaOl7D6GJ7\nNJH1GkZ7Aa9YA7VXTghxbAoLC1m1ahUJCQlEIhGqqqpISEjA6XTy7rvv8u9//xuXy8WJJ57IhRde\nyMsvv0xxcTGvvPIK3/3ud8nKygLguuuu4+GHH5YEV4jjSNN0DNOLYXpxAO5ESMo48nFKKZQdxLZa\nhyyHAoQCLYRCfiKhIFYoiBXyY4X82FYI2wpFp3SpCIoIEEHTLDTdbp1HrNB0G11XaFq0dseXfV7q\n/vcBQ4tmrTL24+vr4BfvDJ4E1+fzUVRU1GlbYWEhJSUlnbaVlJQwZcqU9sdpaWmkpKTg8/kkwR3E\nUhNdOEydcMTm9Q8O4pkW3b4uuISxiQEKAU8wxPzVGwA46A2wcehYwKTOPPOwd3LErqGATRimYtqc\nXV8pJqUAzYVmeNAND2mWC4JwkAz+ac37yr+rDq2JqN6aiHZOVGOfa09cDQOnlP4XQvSChIQESktL\nmTdvHrZtc/fdd7N3715M0yQ/P799v8LCQt5++20g2jaPGjWq03M+nw+lBs5UASG+LjRNQzPc6IYb\n05mCywsJRz7sqCilQFkoFUHDIinJSXVVPeFQGCsSIRIKRBPpcBgViWBbYWwrgm1FV2mwrDC2HcG2\nLWzbQtkWtrLAjq7UoGJWbFAq2putWldpULHdsq0rM2golAKtU1GLmPtaa40NreO5Q/91RR+39tNq\nsds6znXEf3exvbHdL3rQxca22hptJzikW/cI1+3pv2Gti2sek6P8969sjRYr69ivewR9luC2tLTg\n8Xg6bXO73QQCgU7b/H4/bre70zaPx4Pf7+/RdTRNozcqoLdNjh9ok+QHbtw63zp3LIuXbAXLiVU5\nHCN7DwC+oS4OpDnIqo0WZIgYGl6tisRwNU2O6FeZGjYO3cbpALdLx2OkE/Cn4PZEi8woNDTNhaZ7\n0E0vpjMBh9OL4fCiG223HgzTi976baluuturMQMkBkJs2lxKY7hjeI/H0El0mbh1HU/bMGBHTO9q\na5Ka0Najahq4DT3uHwgH7vtE4u5rAzX2gRp3X8nNzWXDhg2sW7eOG2+8kR/84AeHtb2xbfShbbPH\n48G2bUKhEK7WaSRfRtpmibsvSdx9QSP6lb0DXdfwJCSQGHZ1v3xjP6TrGqmpCdTVNQ+ouGHgxh4b\n9/HUZwmux+M5LJkNBAJ4vd5O27pLeg/drzuZmYnHFughUlN7+zuvvjEQ41541hgWnjWm9dGCzk9e\nffj+5x7xjBcde1Ax0oEHhqb36jnjbSC+T0DijoeBGvtAjft4M81o8z9z5kzOPfdcNm3aRDAY7LRP\nbBvtdrs7Pe/3+zFNs0fJLUjb3Ebi7lsSd9+SuPveQI39eMfdZ2MeR44cic/n67TN5/N1GvIEUFRU\n1Gm/mpoa6uvrDxveLIQQQoijs2zZMr73ve912hYOhykoKCAcDrN///727bFt9KFts8/nY+TIkX0S\nsxBCCHE0+izBnTlzJqFQiGeeeYZwOMyLL75IVVUVs2fP7rTfhRdeyNtvv826desIBoM88MADnHba\naaSlpfVVqEIIIcSgNGHCBDZt2sTLL7+MbdssW7aMZcuWccUVV3DWWWdx//334/f72bhxI6+//joX\nXRQdCXPxxRfz1FNPUVFRQVVVFY8//jgLFiw4wtWEEEKIvqcpdfQFnr+qbdu2cffdd7N9+3aGDx/O\n3XffTXFxMXfddRcA9957LxBdo+/hhx/m4MGDTJ8+nd/+9rdkZPSgbJwQQgghvtS6dev47//+b3bv\n3s2IESP4+c9/zowZM6irq+NXv/oVa9aswev18uMf/5jLL78cAMuy+NOf/sRLL71EOBzmoosuknVw\nhRBC9Et9muAKIYQQQgghhBDHi6w7IoQQQgghhBBiUJAEVwghhBBCCCHEoCAJrhBCCCGEEEKIQUES\nXCGEEEIIIYQQg4IkuF3YsmULl19+OcXFxSxYsIDPPvss3iEdlY0bNx62/FJ/tm7dOhYuXMi0adM4\n++yzef755+MdUo8sWbKE888/nylTpnDBBRfw7rvvxjuko1JVVcXMmTNZunRpvEPpkaeeeooTTjiB\nKVOmtP+sW7cu3mEdUUVFBddddx1Tp07ltNNOY/HixfEOqUdeffXVTq/1lClTGDduHHfeeWe8Qzui\nTz/9lP/4j/9g6tSpzJs3j9deey3eIYleIG1z35K2OT6kbe4b0jb3vT5tm5XoJBAIqDlz5qjnnntO\nhUIh9cILL6gZM2aopqameId2RLZtqxdeeEFNmzZNnXzyyfEOp0fq6urUSSedpF599VVlWZbatGmT\nOumkk9SqVaviHdqXKikpUZMnT1affPKJUkqpVatWqYkTJ6rq6uo4R9Zz1157rRo3bpx6//334x1K\nj9xyyy3qySefjHcYR8W2bXXppZeq++67T4VCIbVjxw510kkntb9vBpJVq1apWbNmqfLy8niH8qUi\nkYiaMWOGevPNN5VSSn388cdqwoQJqrS0NM6RiWMhbXPfkrY5fqRtPv6kbe57fd02Sw/uIdauXYuu\n61x55ZU4HA4uv/xyMjMzWbZsWbxDO6K//OUvLF68mOuvvz7eofTY/v37mTt3LhdddBG6rjNx4kRO\nOeUUPv3003iH9qUKCwtZtWoVU6dOJRKJUFVVRUJCAk6nM96h9cg//vEPPB4Pubm58Q6lx7Zu3cr4\n8ePjHcZR2bBhAwcOHODWW2/F4XAwevRonn/+eQoLC+Md2lFpbm7mtttu4+677yYnJyfe4XyphoYG\nampqsCwLpRSapuFwOGS91gFO2ua+JW1zfEjb3Dekbe57fd02S4J7CJ/PR1FRUadthYWFlJSUxCmi\nnrvssst45ZVXmDRpUrxD6bHx48fzhz/8of1xfX0969atY9y4cXGMqmcSEhIoLS3lxBNP5Oc//zk3\n33wziYmJ8Q7riHw+H3/729+4++674x1Kj/n9fnw+H4sXL2bWrFmcf/75vPjii/EO64g2b97M6NGj\n+cMf/sCsWbOYN28eGzZsIC0tLd6hHZUnn3ySMWPGcPbZZ8c7lCNKS0vjyiuv5JZbbmHixIl8+9vf\n5s477xxQHxjF4aRt7lvSNvc9aZv7jrTNfa+v22bzuJx1AGtpacHj8XTa5na7CQQCcYqo57KysuId\nwjFpbGzk+uuvZ+LEiZx55pnxDqdHcnNz2bBhA+vWrePGG29k+PDhzJw5M95hdSsSifDzn/+c//qv\n/yI1NTXe4fRYVVUV06ZN41vf+hZ/+tOf2LhxI9dffz1Dhgxh7ty58Q6vW/X19Xz44YfMmDGDpUuX\nsmnTJn74wx+Sn5/P9OnT4x1ejzQ3N/Pss8/yxBNPxDuUHrFtG7fbzcMPP8yZZ57J6tWr+elPf8rE\niRMHxIdz0TVpm+NH2ubjT9rmviVtc9/r67ZZenAP4fF4DmswA4EAXq83ThF9PZSWlvLNb36TlJQU\nHn30UXR9YLw1TdPE4XAwc+ZMzj33XN577714h/SlHnvsMcaPH9+vG56u5Ofn8+yzzzJ37lycTifT\np09nwYIF/f71djqdpKSkcN111+F0OtsLK/T3uGO9++67DB06lOLi4niH0iNvv/02Gzdu5LzzzsPp\ndHL66adz+umn8/LLL8c7NHEMpG2OD2mb+4a0zX1L2ua+19dt88D4T9WHRo4cic/n67TN5/MxatSo\nOEU0+G3evJlvfOMbzJ49m8ceewy32x3vkI5o2bJlfO973+u0LRwOk5SUFJ+AemjJkiW88cYbTJ8+\nnenTp7N//35uueUW/vrXv8Y7tC+1efPmw2IMBoP9fl5VYWEhlmVhWVb7trb5JwPF0qVLOf/88+Md\nRo+Vl5cTCoU6bTNNU+bgDnDSNvc9aZv7jrTNfUva5r7X523zcSldNYAFg0E1e/ZstXjx4k6VGpub\nm+MdWo+tXbt2wFRqPHjwoJoxY4Z6/PHH4x3KUTlw4ICaNm2a+te//qUsy1IffPCBmjp1qtq5c2e8\nQzsqZ5xxxoCo1FhSUqImTZqk3nzzTWVZllq9erUqLi5WmzZtindoX8rv96s5c+aohx56SIXDYfXJ\nJ5+o4uJitX79+niH1mOnn366WrNmTbzD6LFt27apiRMnqhdffFHZtq0+/PBDNWXKFLVx48Z4hyaO\ngbTNfUva5viStvn4kra57/V12ywJbhe2bt2qrrjiClVcXKwWLFgwoN7wSg2sRvTPf/6zGjNmjCou\nLu7088ADD8Q7tCP6+OOP1aWXXqqmTJmiLr300gH1j6bNQGlElVLqvffeUxdeeKGaPHmyOvfcc9tL\nzfd3u3fvVosWLVInnXSSOuOMM9SLL74Y75B6LBKJqLFjxw64D4fvvfeeuvjii9WUKVPUBRdcoN5+\n++14hyR6gbTNfUfa5viStvn4k7a57/Vl26wpNYD644UQQgghhBBCiG7IHFwhhBBCCCGEEIOCJLhC\nCCGEEEIIIQYFSXCFEEIIIYQQQgwKkuAKIYQQQgghhBgUJMEVQgghhBBCCDEoSIIrhBBCCCGEEGJQ\nkARXiDi47bbbGDt2bLc/jzzyCB9++CFjx44lGAwe93jKysrar33TTTd95eN37dp1xH1vvfXW9mv1\nZH8hhBCiL0jbLG2zGBxkHVwh4qCxsZFAIADA+vXr+clPfsL777+P0+kEwOv14nA4qK+vZ8iQIcc9\nnrKyMs466yyee+45xowZQ3Jy8lEdb1kWNTU1pKenYxjGl+7b2NiIz+dj4cKFLFmyhKKiomMJXQgh\nhOgV0jZL2ywGBzPeAQjxdZSUlERSUhIAKSkpAGRmZuJyuTrt1xcNaKy0tLSjbkABDMPocaxJSUmk\np6cf9TWEEEKI40naZmmbxeAgQ5SF6KcOHQY1duxYlixZwkUXXcSJJ57IokWLKC8v52c/+xnFxcWc\ne+65rF27tv34yspKbrrpJqZMmcKcOXO4++67aW5u7tG124Y1ffDBB5xzzjlMnjyZm2++mf3793Pt\ntdcyefJkFixYwLZt2zrt3zas6cwzz+SZZ57hO9/5DpMmTWLevHksW7asl18hIYQQom9J2yxE/ycJ\nrhADyP33389dd93Fs88+y+bNm7n44osZP348L730EqNGjeKee+4BQCnFj3/8YxwOBy+88AKPPvoo\n27Zt44477jiq6/3pT3/ioYce4rHHHuOdd97hsssuY/78+bzwwgu43W7++Mc/fumxV155JW+88QZj\nx47ljjvuIBwOH9PvL4QQQvQ30jYL0b/IEGUhBpCrrrqKk046CYAZM2ZQWlrKokWLALjiiiu4/vrr\nsSyLjz76CJ/Px//+7//icDgA+O1vf8t5551HRUUFOTk5PbreDTfcwMSJEwEYN24cQ4cO5ZJLLgFg\nwYIFPPHEE90ee9FFFzF//nwAbrzxRhYsWEBFRQX5+flf7ZcXQggh+iFpm4XoXyTBFWIAKSgoaL/v\n8Xg6NUhutxvbtolEIuzatYumpiZOPvnkw87h8/l63Igeer28vLxO1wuFQt0eO2LEiPb7iYmJAPIt\nsRBCiEFH2mYh+hdJcIUYQEyz85+srnc9yyASiVBQUNDlt7hHUxyjp9frStu307GkaLsQQojBRtpm\nIfoXmYMrxCBUVFRERUUFSUlJDB8+nOHDhxOJRLjvvvtoamqKd3hCCCHE1460zUL0DUlwhRiEZs2a\nRVFREbfccgubN29m06ZN/OxnP6O2tpasrKx4hyeEEEJ87UjbLETfkARXiEFI13Uee+wxEhMT+c53\nvsOiRYsYMWIEjz76aLxDE0IIIb6WpG0Wom9oSgbeC/G1V1ZWxllnncWSJUsoKioadNcTQgghBhpp\nm4X4aqQHVwjRrra2loaGhuN6jcbGRmpqao7rNYQQQojBQtpmIY6OJLhCiHbf/va3+eUvf3lcr3HP\nPfewcOHC43oNIYQQYrCQtlmIoyNDlIUQQgghhBBCDArSgyuEEEIIIYQQYlCQBFcIIYQQQgghxKAg\nCa4QQgghhBBCiEFBElwhhBBCCCGEEIOCJLhCCCGEEEIIIQYFSXCFEEIIIYQQQgwK/w8Q2qPqU0au\nyAAAAABJRU5ErkJggg==\n",
"text/plain": [
""
]
},
"metadata": {},
"output_type": "display_data"
}
],
"source": [
"IC = [C0,T0,Tcf] # initial condition\n",
"t = np.linspace(0,8.0,2000) # simulation time grid\n",
"\n",
"qList = np.linspace(153,154,11)\n",
"\n",
"plt.figure(figsize=(16,4)) # setup figure \n",
"for qc in qList: # for each flowrate q_c\n",
" X = odeint(deriv,IC,t) # perform simulation\n",
" plotReactor(t,X) # plot the results\n",
" \n",
"plt.legend(qList)"
]
},
{
"cell_type": "markdown",
"metadata": {},
"source": [
"There's a clear bifurcation when operated without feedback control. At cooling flowrates less than 153.7 liters/minute, the reactor goes to a high conversion steady state with greater than 95% conversion and a reactor temperature higher than about 410K. Coolant flowrates less than 153.8 liters/minute result in uneconomic operation at low conversion.\n",
"\n",
"For the remainder of this notebook, our objective will be to achieve stable operation of the reactor at a high conversion steady state but with an operating temperature below 400 K, an operating condition that does not appear to be possible without feedback control."
]
},
{
"cell_type": "markdown",
"metadata": {},
"source": [
"## Simulation 2. Fixed Time Grid\n",
"\n",
"Introducing feedback control requires a change in the simulation strategy. \n",
"\n",
"The new approach will be to break the simulation interval up into small time steps of length $dt$. At each breakpoint a PID control calculation will be performed, the coolant flow updated, then `odeint` will be used to simulate the reactor up to the next breakpoint.\n",
"\n",
"The following cell demontrates the simulation strategy assuming a constant coolant flowrate. Note the use of Python lists to log simulation values for later plotting."
]
},
{
"cell_type": "code",
"execution_count": 5,
"metadata": {},
"outputs": [
{
"data": {
"image/png": "iVBORw0KGgoAAAANSUhEUgAABGwAAAEMCAYAAACRPCChAAAABHNCSVQICAgIfAhkiAAAAAlwSFlz\nAAALEgAACxIB0t1+/AAAIABJREFUeJzs3Xd8Tff/wPHXHUlu9iKxySBWECORSvhWlQqlarS0qBmj\nFFVq71UVJEFQu9r6US1VSmlRexatLalNlkR2bnLv74/UrdtcJMgo7+fjkcfNPfeMz33n5nPPeZ/P\nUOj1ej1CCCGEEEIIIYQQothQFnUBhBBCCCGEEEIIIYQxSdgIIYQQQgghhBBCFDOSsBFCCCGEEEII\nIYQoZiRhI4QQQgghhBBCCFHMSMJGCCGEEEIIIYQQopiRhI0Qz9HznHRNJnATQogXl9TxQgjx3yF1\ntmkSl4InCRtRpH744Qfef/99GjRogK+vL127dmXPnj1FXayncuzYMT755JNn3k9iYiJDhgwhKioK\ngBs3buDl5cXevXufed9CCFFcfPrpp3h5eT325/Dhw0VdzAIxb948Nm3aVNTFEEK8RArrnPvw4cN4\neXlx5coVALp27crQoUOf+3EeOHfuHF5eXuzcudNo+e3bt/Hy8qJhw4a5kgqzZ8+mfv36ZGdn5+kY\nP//8M9OnT39uZX7Y474DFy5cCBR8DJ9WaGgoGzduLOpivPDURV0A8XLS6/WMHDmSHTt28N5779G3\nb1+ys7PZsmULffv2ZdKkSbz77rtFXcx82bBhA9euXXvm/Zw/f55t27YxaNAgAFxcXFi3bh0eHh7P\nvG8hhCguBgwYYFTPBwcH06xZMzp27GhY5unpWRRFK1BZWVksWrSIqVOnFnVRhBAvgaI+554wYQJm\nZmYFtn8vLy/s7Ow4ffo0zZo1Myw/ePAg9vb23Lt3j3PnzlG9enXDaydOnKBBgwaoVKo8HWP16tWU\nKFHiuZf9gd69e/P666/nWl66dOkCO+bzsGDBAiZOnFjUxXjhScJGFIl169bxww8/sGLFCho2bGhY\n/uqrr2Jubs706dNp3rw5Tk5ORVjK4sHc3Jw6deoUdTGEEOK5qlChAhUqVDA8V6vVlCpVSuo7IYR4\njor6nLugE+9KpZJ69erx+++/Gy0/ePAgr776KkePHuXAgQOGhE1mZiZ//vknH3/8cYGWKz/KlSsn\n333ikaRLlCgSq1atolmzZkZfHA98+OGHvPvuu6SkpACwdetW2rVrR+3atXnttddYunSpUdNGLy8v\nNm3axIcffkidOnVo1KgR4eHhRvuMj4/nk08+wc/PjwYNGjB48GDu3r1reP3u3bsMGTKEevXqUa9e\nPT755BPu3btneP3TTz9l+PDhLFmyhMDAQGrVqkVwcLBhH59++infffcdp06dwsvLixs3bhAWFkbn\nzp2ZMGECPj4+9OnTB4DLly8zcOBAfH19qVmzJi1atGD9+vVATjPSbt26ARAUFERYWJjJLlEHDx7k\n3XffxcfHh8DAQGbPnk1mZqbh9aZNm7Jy5UpGjRpFvXr18PX1ZcqUKWi12qf7gwkhRBG5c+cOgwcP\npm7dutSvX5+RI0eSmJhoeH348OGMHDmS8PBwXnnlFXx8fBg/fjzJyclMmDCBunXr0qRJE0M9+2Cb\noUOHMn/+fHx9ffHz82PKlClG9aher2fx4sU0bdoUb29v2rdvz9GjRw2vHzhwAC8vL9atW0fDhg1p\n3LgxqampJCQkMGHCBBo3bkyNGjUIDAxk1qxZZGVlkZWVRY0aNQAYO3YsH3zwAQCNGzdm7ty5Ru97\n8ODBhtevXr2Kl5cXa9asITAwEF9fX65fvw7A+vXradGiBTVr1qRVq1bs2LHjucZfCPHflp9zbnjy\neXdmZibh4eE0b96cWrVq0a5dO3799ddHHv/h7jwPukudOHGCDh064O3tTYsWLXJ1Z9q/fz/t2rWj\nVq1avP322/z888+G82tT/Pz8+OOPP9DpdIZlhw4dokGDBvj5+XHw4EHD8rNnz5KRkYGfnx+QU9d/\n8cUXBAUFUbNmTerXr8+HH35oOMfv2rUrR44cYevWrXh5eRn2c/LkSTp37kytWrUIDAwkLCzM6PhN\nmzZl7ty5hlg+z26wsbGxfPrppwQEBFCnTh369Olj6IL2IFaxsbGG9QcOHIiPjw9ZWVmGZe3atWPu\n3LmGv8k333yDv78/AQEBJCcnc//+fSZNmkTjxo2pWbMmAQEBTJ8+3XAt8SAWEydOpGvXrob9fvvt\nt7zxxhvUrFmToKAgtm3b9tze98tKEjai0N29e5fIyEgCAwNNvl62bFlGjx5N+fLl+fLLLxk2bBi+\nvr4sWLCAdu3aMX/+fGbPnm20zdSpU6lQoQKLFi0yJDoe9MvNysrigw8+4MSJE4wZM4bPPvuMqKgo\n+vfvD0BqairdunXj4sWLTJs2jSlTpnDy5EmCg4ON+rb++uuv/PLLL0yaNIlp06Zx8uRJZsyYAeQ0\n7W/SpAmVK1dm3bp1uLi4AHD69Glu3brFwoUL6dmzJ8nJyXTr1o2MjAw+//xzFi5ciLu7O+PGjSMq\nKooaNWowfvx4AObOnWvUNeCBX375hR49elCxYkVCQ0Pp3bs3X331Va7xc8LCwlAoFISFhdGrVy++\n/PJLowsWIYQo7pKTk3n//feJjIxkxowZTJo0iWPHjhEcHGx0Yvzzzz9z9OhRPvvsM/r06cO6det4\n++23yc7OZsGCBdSsWZNJkyZx+/ZtwzZ79+5l+/btTJ8+ncGDB/Ptt98yadIkw+uzZ89mwYIFvPvu\nuyxYsICKFSvSs2dPzp8/b1TG1atXM2vWLEaPHo2VlRVDhw7l8OHDjBo1ii+++IK2bduyfPlyNm3a\nhFqt5quvvgJyuoCNGzcuX/FYsmQJ48ePZ8yYMZQvX561a9cyfvx4mjdvzqJFi2jYsCEfffTRf3Ys\nOCHE85Wfc24gT+fdw4cPZ/ny5bz//vuEh4fj6elJ//79H5u0+bfhw4fz1ltvsXjxYsqUKcOwYcNI\nSEgAcoYGCA4OpmLFioSHhxMYGMjw4cMfu78GDRqQkpLC5cuXAbhy5QrR0dH4+/vj5+fH8ePHDQn5\nkydP4uTkZEg4LF26lPDwcN577z2WL1/OsGHDOHToEJ999hmQ06WrevXqvPLKK6xbt85Qxu7du+Pg\n4EBYWBh9+vRh2bJlua5Pli1bRtu2bfn8889NJswe0Ol0hqT+g5+Hv+MelpyczLvvvsvJkycZNWoU\nn3/+Offu3aNLly7cvn0bf39/1Go1R44cAXISUseOHSM1NZVz584BOTeyz507Z/S5WLlyJTNnzmTM\nmDHY2NgwbNgwDhw4wKhRo1i6dClvv/02q1at4rvvvgMwxKJ3795MmDDBsGzs2LE0bdqURYsW0ahR\nI4YOHcovv/zy2L+feDzpEiUK3YOM9ZP6ZWZnZxMWFkaHDh0YNWoUAAEBASgUChYtWkTv3r0NzTcD\nAgIYMWIEAA0bNuSnn35i7969NGnShN27d3PhwgU2bdpE1apVAXB1dWXQoEFcv36dvXv3cvPmTbZv\n307ZsmUBqF69Oi1btuTXX3819IfNzMxkyZIl2NnZAXDp0iXWrFkD5DTtd3JyIiEhwahJY1ZWFmPG\njKFSpUoAnDlzBjc3N+bOnYutrS0AtWvXxtfXlxMnTtC+fXtD01EvLy9KlSqV625CaGgo/v7+zJo1\nC4DAwEDs7e0ZOXIk586do1q1agB4eHgYBkh75ZVX+PXXX9m7dy9dunTJ419KCCGK1saNG4mOjmbH\njh2UKlUKgKpVq9KqVSv27NnDq6++CuTUtWFhYdjZ2REQEMD//d//oVKpmDJlCgqFggoVKrBz507O\nnTtn+O5JT09n8eLFhguVrKwsZs6caWgmv3r1aoYOHUqvXr2AnLq2c+fOLFy4kNDQUEMZe/bsSZMm\nTYCcGwB6vZ6pU6dSv359APz9/fn11185fvw47du3p3bt2gCUL18+32OTdezY0TDOQVZWFuHh4XTu\n3NlQ5sDAQO7du8f8+fMNZRJCvLzyes4NeTvvjo6OZvv27cyePZs2bdoAOS0Eo6OjmTdvnqFOfpI+\nffrQuXNnQ9neeOMNDh8+TIsWLVi6dCkeHh7MnTsXhUJB48aNSU5O5ssvv3zk/qpVq4atrS2nTp2i\nSpUqHDx4kAoVKlC2bFnUajVpaWmcPHkSPz8/Tp48ia+vLwqFAoDo6GgGDx7Me++9B4Cvry+RkZHs\n2rULyOnSZWNjg4ODg+Ecf9GiRZQvX57w8HDDODiWlpZMmjSJ3r174+zsDEDNmjUNLSUfZ/LkyUye\nPNloWVBQUK6Wl5DTguXOnTv89NNPlCtXDshpYfTaa6+xfPlyxowZQ+3atTl69ChBQUFcunSJtLQ0\n3NzcOHHiBN7e3hw8eBAbGxvq1KnD8ePHAePvsvT0dLKzs5k0aZIh0eTv78/u3bs5fvw4nTp1MsSi\nXLlyeHp6otPpCA0NpWPHjoZrssDAQBISEpg/fz5NmzZ9YhyEadLCRhS6BxXbozLHD0RGRpKQkMAb\nb7xhtDwoKAitVsupU6cMyx6cAAMoFApcXV1JTU0FcjLppUqVMiRrICchs2vXLsqXL8/Ro0epXLky\nrq6uhqx2uXLlqFChAocOHTJs4+bmZkjWQE7SJy0t7bHvQa1WGy4GALy9vVm7di0WFhZcuHCB7du3\ns2TJEoA8dVdKSUnh/PnzJmOiUCgMlS5ArVq1jNZ5OCZCCPFfcPToUapUqUKJEiUM9XPFihUpV66c\n0QxSlSpVMqqfnZycqF69uuGE3MHBAYCkpCTDOrVq1TKqn1977TV0Oh2///47v//+O1qtlsDAQMNx\ns7OzCQgIyDVzlZubm+F3KysrVq5cSb169bh27Rp79uxh8eLFxMfHP5cuqQ8f68qVK8THxxuVMSsr\ni8DAQM6ePWv0XoUQL6e8nnND3s67jx8/jkKhMLnO+fPnSU5OzlO5Hj5vf5CMf3BOfeTIEV599VVD\n/Q3kOt6/qVQq6tWrZ7g2OHjwoCHR4Orqipubm6HufpC4eWDs2LH07NmT2NhYDh8+zNq1azlx4sRj\n6+yjR4/SqFEj9Hq9Ud2r1Wo5ceKEYb2H6+zHCQ4OZsOGDUY/w4YNM7nu8ePHqVmzpiFZA2Bra0tg\nYKDhOqBRo0aGLrxHjx6lVq1a1K1b11C2AwcO8Morr6BW/9N24+GyajQaVqxYgZ+fn+G7bMmSJcTF\nxT0yLlFRUcTGxtK4ceNc30nnz583tKAS+SctbEShe5Dlf7hp+r/duXPHMEbBv0dlf5C1fvhLwcLC\nwmgdpVJp6G+bmJj42IHUEhISOHv2rGFcgYc9nOTRaDSPPMajODg45BqBPjw8nGXLlpGWlkb58uXx\n9fUFeOK+IOdiQ6/XG2LwgLm5OTY2NkYxeZryCiFEcZKQkMCZM2dM1s8xMTGG362trXO9/u868N9K\nlixp9PzB98T9+/cNFzdvvvmmyW0fHuvm3/Xx9u3bmTFjBrdv36ZkyZLUqVMHjUbzXOrfh4/14OS3\nX79+JteNjY01tOQUQryc8nrOXapUqTyddycmJmJra4u5ubnJdR4eC+dxHj5vVypz2g88qHfv3buX\n67z93/WsKQ0aNGDTpk3odDqOHDli1MW1YcOGHD9+nJs3bxIdHW3UPeny5cuMGTOG33//HWtra2rU\nqIGFhcVj6+yEhARWrVrFqlWrcr0WHR2dr3JDzt/J29s7T+vev3/f5H6dnJz4448/AAxj6sTHx3P0\n6FHq1atHhQoVmDdvHpCTsBkwYECu7R+2c+dOpk2bxq1btyhRogR16tR5bFwejP05cOBAk6/HxsYa\nbp6I/JGEjSh0Tk5OVK1alQMHDpicRvDmzZs0bdrUMJbLw4NmPfzc3t4+T8eztbU1GkD4gT179uDt\n7Y2dnR0+Pj6MGTMm1zp5PUZeff/99yxcuJBp06bRvHlzrK2tSU9PZ8OGDXna3sbGBoVCQVxcnNHy\njIwMkpKSnnt5hRCiKNna2lKvXj1D8/yHPeuJ38MDFwOGetXR0dFwB3HlypXY2Njk2vbhu5IPu3Ll\nCh9//DFdu3alV69ehguft99++7FlUSgUue6AP+nC50GLolmzZpnsWlWmTJnHbi+EePHl55z7wQ3E\nx513379/n6SkJDIzM42SNvk9N38cFxeXXOe58fHxT9zO19eXkJAQTpw4QVJSklFSpmHDhmzatIkT\nJ07g4uKCu7s7kJMk6t+/P6VKlWLbtm24ubmhUCiYPXs2165de+SxbG1tad26NW+99Vau1wq67rWz\nszNKCj0QFxdniH/NmjWxt7fnyJEjHD9+nLfffpuKFSsSHR3Nvn37uHXr1iPHNQL466+/GDJkCF26\ndKFPnz6GGxymxtZ8uFwAM2bMoHLlyrlef7hFkMgf6RIlisT7779vGCTy38LCwjAzM6NZs2Y4ODjw\n008/Gb2+bds2VCpVri4/j1K7dm1u377NhQsXDMsuXrxI3759iYyMxMfHh6tXr+Lm5oa3tzfe3t5U\nrlyZsLAwzpw5k+f39OAOweOcPHmSSpUq0a5dO8Md4f379wP/3Fn4d4uch9nY2ODl5WUyJgA+Pj55\nLq8QQhR3Pj4+XLt2DXd3d0P97OHhQWhoqOFO4tM6ffq0UTJ/165dmJmZ4ePjQ61atVCpVCQmJhqO\n6+3tzd69e1m/fv0j6/s///wTrVZLcHCwIVkTHR3NpUuXDHclTW1rZWXFnTt3DM8zMjL4888/H1t+\nDw8PbG1tiYuLMyrjuXPnWLp0qVF3AiHEyyuv59zu7u5PPO+uW7cuer3e5DrVqlV7YsvGvKhXr16u\nAYx37979xO1q1KiBRqPhq6++onLlykYtRvz8/EhLS2Pr1q1G3aHi4+O5du0anTt3xt3d3ZA8P3jw\noFES/d/1to+PD3/99ZdR3atWq5k3b16uZNPzVrduXf744w+jMS6Tk5PZt2+fYVwZpVKJv78/3333\nHbGxsdStW5eKFStSsmRJwsLCqFy5sqErmilnz541fJc9SNbExsZy8eLFR8blwecnNjbWKC4XLlxg\nyZIlebpOEqZJCxtRJNq3b8+uXbvo06cP3bp1w8/Pj5SUFL777jt++eUXpk6diqurKwMGDGDGjBlY\nW1vTuHFjfv/9dxYtWkTXrl3zfHe1adOmVKlShUGDBjFkyBAsLCyYN28edevWpW7dulSpUoVVq1bR\nu3dvevXqhbm5OcuXL+f06dN8+umneX5PdnZ2XLt2jYMHDz4ycVKzZk3WrVvHkiVLqFOnDmfPniU8\nPByFQmHou/ugCfuuXbtydfUCGDRoEAMHDmTkyJG0bt2aqKgo5s2bx+uvv27UhUsIIf7rOnbsyOrV\nqw31s5mZGV988QVnz55l7Nixz7TvtLQ0+vfvT79+/bh27Rrz5s2ja9eu2NnZYWdnR8eOHZkwYQLR\n0dFUqVKFQ4cOERERYRjg15Tq1aujVCqZOXMmb731Fnfu3CEiIgKtVmsYQ0ypVGJjY8OhQ4fw9vam\natWqBAQEsG7dOnx9fSldujQrVqx4Yhcqc3Nz+vbtS2hoKJmZmfj4+HDu3DlCQkJo3759ri4LQoiX\nU17PuYEnnnc7ODjQrFkzJk6cSEJCAm5ubmzZsoXDhw8THh7+XMrbt29f2rVrx7Bhw2jXrh1//vkn\nq1evBh5/c1SlUlG3bl127NiRa4INR0dHvLy8+O2335g4caJhubOzM6VLl2bZsmVYW1uj0+n4+uuv\nOXv2rFHyyc7OjgsXLnD48GF8fX3p168f7733HqNGjSIoKIjExETmzp2LpaVlnseteVrt27dn9erV\n9OrVi48++ghzc3OWLFmCTqeje/fuhvUCAgIYM2YM1atXN7QUrV+/Ptu2baNnz56PPUbVqlVRqVTM\nnDmT9u3bEx0dTUREBBkZGUbjd9rZ2XHkyBF8fHyoWrUqwcHBzJs3D61WS7169Th//jxz586lTZs2\n8p30DCRhI4qEUqkkPDycL7/8ku+//561a9eiVqvx8vJi+fLlNGrUCIDu3btjYWHBihUrWLt2LaVL\nl2bo0KFPrGge9iABM2PGDMaNG4daraZx48aMGjUKpVKJnZ0da9as4bPPPjMkaGrWrMmqVasMTSbz\nolOnTuzcuZO+ffua7NMKOZVsZGQkq1atYtGiRVSoUIHRo0ezefNmw0BplStXplWrVoSGhnLnzp1c\n77VZs2aEh4cTHh7Ojz/+iLOzM++//z4ffvhhnssqhBD/BQ4ODnz11VfMnDmTESNGoFAo8Pb2ZuXK\nlVSsWPGZ9l2nTh38/Pz4+OOPsbS0pHfv3kZ9+seNG0eJEiVYsWIFMTExhulvu3Xr9sh9enp6Mm3a\nNBYuXMjWrVtxdXUlKCgIvV7Pxo0b0el0KJVKBg4cSFhYGFeuXOH7779n0KBBxMfHM336dDQaDe++\n+y5eXl6cPXv2se+hb9++WFlZsWbNGhYsWICLiwt9+vTJNTaBEOLllddzbsjbefecOXOYN28eS5Ys\n4f79+1SpUoVFixbleYaoJ6lcuTILFixg9uzZ9O/fHy8vL4YNG8aMGTOwtLR87La+vr789ttvJqfQ\n9vf35/z580avKRQKQkNDmTJlCoMHD8bOzo4GDRoQEhLC0KFDuXDhAl5eXnTv3p0hQ4bQt29ftm/f\nTp06dVi2bBnz5s1j4MCBWFtbExAQwCeffIKZmdlzicOj2NrasmbNGmbNmsXYsWNRKBTUr1+fdevW\nGWa7hZyEDeS0WHqgQYMGbNu27bHdoSCntcz06dNZsGAB27dvx9XVlTfeeIPmzZuzbt06srOzUalU\n9O/fn/nz53P58mV++OEHevbsiUajMVznuLi40KNHj0eOayPyRqGXUUiFEEII8RIZPnw4N2/e5Ouv\nvy7qogghhHjI/v37sbOzMxqEd926dcyZM4fDhw9Ld0/x0pEWNkIIIYQQQgghitzJkydZs2YNI0aM\noEKFCkRFRRESEkKnTp0kWSNeSpKwEUIIIYQQQghR5IKDg0lJSSE8PJyYmBhcXV354IMP6Nu3b1EX\nTYgiUSRdok6fPs2AAQPYt2+fyde3bNnC3LlziYuLw8/Pj2nTphlmWxBCCCGEEEIIIYR40RXq/Fp6\nvZ4NGzbQs2dPtFqtyXXOnz/PhAkTCAkJ4dChQ5QoUYJRo0YVZjGFEEIIIYQQQgghilShJmwiIiJY\nvXo1/fr1e+Q6P/zwA6+99hq1a9dGo9EwfPhwfvvtN2JjYwuxpEIIIYQQQgghhBBFp1DHsGnfvj39\n+vXjyJEjj1wnMjISHx8fw3NHR0fs7e2JiorKU7eo2NhklPlOQylwdLTi3r1UQCbN+ofExTSJS24S\nE9OePi5OTjYFU6QXWExMUr7WVygUODtbExeXgkyY+A+Ji2kSF9MkLrk9S0xKlrQtoFK9uPJb94N8\nbk2RmJgmcTFN4mJaQdT/hZqwcXFxeeI6aWlpaDQao2WWlpakpaXl6RjOztZPPYK4k5P1U233opO4\nmCZxyU1iYprEpXhSKnO+WJVKyM4u6tIUHxIX0yQupklccpOYFH/yN8pNYmKaxMU0iYtpBRGXYjdL\nlEajIT093WhZWloaVlZWedo+Li4l3y1slEoFDg7WJCSkoNNJhvABiYtpEpfcJCamPUtcpIWNEEII\nIYQQL7dil7Dx8PAgKirK8Dw+Pp7ExEQ8PDzytL1er3/qbJZOpyc7Wy42/03iYprEJTeJiWkSFyGE\nEEIIIUR+Feqgw3nRunVrduzYwbFjx8jIyCAkJITGjRvj6OhY1EUTQgghhBBCCCGEKBTFImEzfvx4\nxo8fD0C1atWYMmUKY8aMwd/fn+joaGbMmFHEJRRCCJFXW7dupWXLlvj4+NCqVSt27twJwJkzZ6hW\nrRo+Pj6Gn4iICCCndeScOXNo2LAhDRo0YOrUqWRLp2ghhBBCCPESK5IuUX5+fhw+fNjwfPLkyUav\nBwUFERQUVNjFEkII8YyioqIYPXo0y5cvp27duhw4cIC+ffuyd+9ezp07R+PGjVm8eHGu7dauXcvu\n3bvZvHkzCoWC4OBgli9fTp8+fYrgXQghhBBCCFH0it0YNkIIUdj0ej1Z2Tq0WToys3IeH/7JzMo2\n/J6VrSMrW0+27sGjnuzsnOXZOr3Razq9HvdyjgTWfPIMeS8KNzc39u/fj7W1NVlZWcTGxmJtbY25\nuTlnz56latWqJrfbtGkT3bt3N8wmGBwczPz58ws0YROTkMbWw9d5pYYLtpbmBXYcIYQQQgghnoYk\nbIQQ/yl6vZ60jGxS0rWkpmeRlpFFemY26dqcx4zM7JznmVkP/f73c202GVodmdpstNk6tFpdzmOW\nrsDK++uJm3iWboirY95munsRWFtbc/36dVq0aIFOp2PixInY2Nhw7tw5zM3Nadq0KTqdjpYtWzJ0\n6FDMzc2JjIzE09PTsA83NzeioqLQ6/UoFIo8HffBNIp5tfXQVX45fpOb0Un0ebN6ft/mC0upVBg9\nihwSF9MkLrlJTIQQQjwvkrARQhQZnV5PSpqW+ymZ3E998JhJUmomKWlZpKRrSUnPIvXvx5Q0LakZ\nWeiLYMIlhQLM1SrUKgVqlRKVSoFamfOoUipRqxSGZTm/K1GrlFSu4EgpZyv0BZcTKpZKly7NqVOn\nOHbsGAMGDKBixYo4Ojri5+fHO++8Q1xcHB999BGhoaEMHz6ctLQ0NBqNYXtLS0t0Oh2ZmZlYWFjk\n6ZjOztZ5Tu4AuJV1gOM3OX0lDnsHa1RycWXEwcG6qItQLElcTJO45CYxEUII8awkYSOEKBAZmdnE\nJ6UTfz+D+PvpxCflPN5LyiAhOScpk5SqRfccsi8qpQKNuQqNuQoLc3XOo5nKsExjrsbCXIXGTIWF\nuQpztRK1WomZWom5WvX349/LVErMzVSYqZSYmT14rkSVn6YbD8qlUuDkZEN8fDLZvFzTeqvVOV8v\n/v7+NG/enF27dhkGGAawsrIiODiYkJAQhg8fjkajISMjw/B6WloaarU6z8kagLi4lHy1sKlS1g6A\n+ymZnPjzFh5l7fO+8QtMqVTg4GBNQkIKOt3L9bl9HImLaRKX3J4lJk5ONgVUKiGEEP9FkrARQjwV\nnU5P3P2M5criAAAgAElEQVR0ou+lEZuYRmJaFtduJxJ9L517SemkpGfle5+WFirsrMyxtTLHxtIM\na40aK82DRzXW/1pmrTHD0kKNmbpYTHgngD179rBixQpWrlxpWKbVatHr9cyaNYuBAwdiY5NzQZKR\nkWFIyHh4eBAVFUXt2rWBnMGL3d3d83VsvV5PfiaWcnW0xNXJirvxqZy8GEulUnb5Ot6LTqfTk50t\nF+D/JnEx7VnjotfruXPnNqVLl3mOpSpa8lkRQgjxrCRhI4R4LG2WjjvxqdyMSeZmbAo3Y1K4HZ9K\nbEIa2Xm8c2hnZYajnQYnWwucbDU42JrnJGaszbG3/vt3KzPMzVQF/G5EQatevTp//PEH33//PW3a\ntOG3335jz549rFu3jn79+qHX6/n444+5desWERERdOrUCYA2bdqwbNkyGjZsiFqtZvHixbRt27ZA\ny6pQKGhQzZUt+6M4HRlHu8b5SxAJ8SIKCKiPhYUFyr+bq+n1ekqUKMn773endeu3Cuy4CxbMB+DD\nD4cU2DGEEEKI/xpJ2AghDJLTtPx15z5Rt5O4fjeJm7Ep3I1Pe2K3JUdbC8q62OBkY0EJew1OdjmJ\nGSc7CxxtLTBTSyLmZVGyZEkiIiKYPn06kydPplKlSixYsABPT08iIiKYOnUqDRs2RKPR8M4779C9\ne3cAunTpQmxsLB06dECr1fLmm2/So0ePAi9vvb8TNlfvJJGYnIG9Td67YAnxolq6dBXu7jmDgGdn\nZ7Nr1w6mTp1AzZq1qVTJrUCOmZiYgL29Q4HsWwghhPivkoSNEC+pTG02f91JIur2faJu3+ev20lE\nJ6Q9cn1zMyVlS1hT5u8fV0crXBwtKelgiZVG/c9YLdL8+6VXv359Nm7cmGu5p6enUVeph6lUKoYO\nHcrQoUMLuHTGvD1LYK5WkpmlY/uR63Rq6vnkjYR4DrKydcTfTy+UYznZaVCrnq7rqEqlonnzloSG\nziEq6gqVKrlx/34i8+d/zuHDh9BoNLz3Xhfat+8M5CRe5s37nDNnTnHvXjzlypXn448/pVatOgB8\n990GvvpqDYmJCVSrVp0RI8bw22+72bFjGwqFgjt3bjN16iyOHj1ERMQCrl+/RpkyZQkOHoC/fwCQ\n0wqoXbuO/PzzT7z3Xje6di345K4QQghRFCRhI8RLIi0jiys3E7lwPYEL1xOIunX/kV2aXJ2sqFTK\nlnIlrSlbwoYyJa0pYa9BmY8ZeIT4L7AwU9HctzxbDlxl+9Fr1KtaEo8yMviwKFhZ2TpGLzlEbGLh\nJGxK2GuY3rfhUyVttFot33//LRkZGdSo4Q3AlCnjsbOzZ/36zSQlJTBy5DA0GhtatnyThQtDAVi7\ndj1KpYr58+cQERHOwoVfcOjQAZYsWcicOaF4eVVj8eIFTJkynoiI5Vy5chl7ewc+/HAIkZFXGDny\nYyZMmEKjRo05evQQ48Z9yuLFK/HwyEmqZmZm8MMPO9BqM59foIQQQohiRhI2QrygdDo9UXfu80dk\nPH9ExhF1O8lk1yYnOwvcStlRqbQtbqXtqFTKFiuNWRGUWIii0TbQjeMXYrgdl8rSH84ytGNtXJ2s\nirpYQhSZfv16oVQqyMzUAnr8/PyZN28RLi6uxMXFcujQAbZs+RlLS0tsbKzo1asXX375FS1bvknf\nvgPQaDSoVGpu376Fra0tMTExAOzcuZ2WLVtRvXpNAHr06MPVq1G5jr9r1w7q129AkyZNAfD3D6BR\no8bs2LGN/v0HAdCsWXPMzMwwM5PvKyGEEC8uSdgI8QJJSddy6nIsZyLj+TMqnuQ0ba51ypa0xqu8\nA14VHKlczh4HGbNDvOTM1Sp6tqrGjDUniL6XxsSVR3k70J3A2qXRmMvXpHj+1Col0/s2LLZdoiIi\nluHu7smtWzcZPfoT7O0dqFEjJ8ly9+4d9Ho977zzzwDEer0eW9ucWdZiY2OZP/9z/voriooVK2Jr\na49erwMgPj4eD4/Khu0sLS2pWrV6ruPfuxdPqVKljZaVKlWamJjof96TU4k8vx8hhBDiv0rORIX4\nj7ufmsnJizEcvxDDuav3cnVzKmGvwdvdmeqVnPCq4ICNpdyNFOLfPMrYM/Sd2iz94Sz3UzL5etcl\nvt8Xibe7M7U8nKnp5oydtXlRF1O8QNQqJS6OxbslV5kyZZk5cw4ffNCF0qXL0L17L5ydS6BSqdi8\neQfm5uaoVApUqmxu3sxJpkyYMJq2bd9mwYKlKBQKtm3bQmTkZSBnUPLY2H+SLikpySxfvpR+/T40\nOq6rayn+/PMPo2W3b9+iZEkXw3PpoSuEEOJlIAkbIf6D0jKyOHY+mkNn73L+2j0e7umkVimoWsER\nb3dnvD2ccXW0RCFntkI8UY1KTkzq0YANe65w+Oxd0jKyOXIumiPnolEA5Vxs8Chrj0cZO9zL2FHK\nyUr+t8QLr1Sp0gwePIzPPptGo0aN8fSsTO3aPixaFEa/fgPRajOZOHE0Dg5OjB07mdTUFCwtNSgU\nCv76K4qvvlpNVlYWAM2bt2T8+E9p0SIId3dPVq9ewdmzfxi6NqWkpADw2mvNWbNmBXv37qZRo0CO\nHDnEvn17WbBgSVGGQgghhCh0krAR4j9Cp9dz4eo99p25w/GL0WRqdYbXzNVKvN2dqedVktqeJbC0\nkH9tIZ6GvY0FvVpVp8P/PDly7i5nIuM4fzWBrGwd16OTuR6dzO6TNwGw1qhxK2NHRVdbKrjaUsHV\nhpIOljI4t3jhBAW9yc8//8SMGZNZsmQlEydOY/78OXTo0Ibs7Gz+978mDB78MQAjRowmNDSEhQvD\nKFmyJK1atWHJkoUkJiZQr14D+vcfzPjxo7l3Lw5v79pMnDgNgFdfbcb48aO4e/c2ISHhzJjxOYsW\nhTFlynhKlSrFhAlTqVatRlGGQQghhCh0Cr3exCik/2ExMUn53kalUsiUxCZIXEwr7LjcT8lkz+83\n2XvqFnH3M/4ph1JBbc8SNKzuire7MxbmqgIvy6PIZ8W0Z4lLyZK2BVSqF1d+6/+8/n0ytNlcuHaP\nSzcSuXIzkajbSWRos02uqzFXUcHF5u8ETk4Sp0wJ66eeUrkoyP+zaRIX0yQuuUndX7jk3P/5kJiY\nJnExTeJiWkHU/3IbXohi6uqdJHYeu87hc3fJeugfvqKrLY28S+FX3RVbKxlTQ4iCZmGmopZHCWp5\n5AxyqtPpuRGTzJVb97l65z5X7yZzMyaZrGw96ZnZXLyRyMUbiYbt1SoFZUpY5yRw/k7mlHexkZZw\nQoj/lNOnTzNgwAD27dsHQGZmJrNmzWLLli0ANGvWjAkTJmBubo5eryckJIT169eTnZ1N27ZtGTVq\nFCpV7ptLiYmJjB49mkOHDmFra8vAgQPp2LFjob43IYQoruRsUYhiRKfXc/JiDDuOXufSQxd8GnMV\nAd6lCaxdhvIuNkVYQiGEUqkwtKCBsgBkZeu4HZfKtbtJXL2bxLW7yVyPTiItI5usbD3X7iZz7W6y\n0X5cHC0fao2T8yiztgkhihu9Xs+3337LzJkzjRIuISEhXLp0ie3btwMQHBzM8uXL6devH2vXrmX3\n7t1s3rwZhUJheK1Pnz659j9u3DisrKw4cOAAFy5coE+fPlSuXJk6deoU2nsUQojiShI2QhQDOr2e\nY+ej+eHAX9yMSTEsd3W05LV65WjkXVruxgtRjKlVSsq72FDexYZG3jnTEev0emIT0nKSNdFJfydt\nkkhIzgQg+l4a0ffSOHYhxrAfO2tzKpWyxbOsPVXKO+BW2hYzddF1dxRCiIiICLZt20a/fv1YunQp\nAFqtlnXr1rF+/XocHBwACA0NNQwwvWnTJrp3746LS87MXsHBwcyfPz9XwiYlJYWdO3eyfft2LCws\nqFWrFq1bt+b777+XhI0QQiAJGyGKlE6n5+jfiZpbsf8kaqpXcqR5g/LUdHeWAUyF+I9SKhS4OFrh\n4mhF/ar/TEd8PyWTa9FJXL+bzNW7SVyPTuZOXCr6v187fSWO01figJzuVJVK2VG5vD013ZypXM7+\nPzUejhDiv699+/b069ePI0eOGJZdvXqV7OxsTp06xYABA0hLS6N169YMGzYMgMjISDw9PQ3ru7m5\nERUVhV6vN5pd7+rVq6jVasqXL2+07o4dO/JcPoVCgTKf1aJSqTB6FBKTR5G4mCZxMa0g4iIJGyGK\ngF6v59TlODbsuWKUqKnp7kSbRm54lrUvwtIJIQqSnbU5Nd2cqenmbFiWkZnNjZicFjhXbt3n0o0E\nYhLSycrWc/lmIpdvJrLt0DU05iqqV3LCp3IJ6lYpKS3vhBAF7kErmYclJCSg1Wr59ddf2bBhAykp\nKQQHB2Nra2tI4Gg0GsP6lpaW6HQ6MjMzsbD4p+tnamqq0XoAGo2G9PT0PJfP2dnaKAmUHw4O1k+1\n3YtMYmKaxMU0iYtpzzMucqYnRCG7eieJdb9c4vy1BMOyWh7OtGnkhnsZuyIsmRCiqFiYq/Aoa49H\nWXterZuz7F5SBpdvJnLpegJnr97jVmwK6ZnZnLgYw4mLMazefoHaniXwr+5KLU9nVPm9xSyEEE/J\n3NwcnU7HkCFDsLOzw87Ojh49erBmzRoGDBiARqMhI+OfmS3T0tJQq9VGyRrISeQ8vB5Aeno6VlZW\neS5LXFzKU7WwcXCwJiEhBZ1OZrgBicmjSFxMk7iY9ixxcXIyPU6pJGyEKCT3UzPZ8OsV9p25bVjm\nVd6Bjq96SqJGCJGLo60FDaq60ODv7lRxiemciYzj98ux/BkVjzZLx7Hz0Rw7H42znQWv1StP49ql\nsdKYFXHJhRAvukqVKqFUKsnMzDQsy87ONvzu4eFBVFQUtWvXBiAqKgp3d/dc+6lYsSJarZZbt25R\npkwZw7oPd6d6Er1ez0OHzhedTi9TEv+LxMQ0iYtpEhfTnmdcJGEjRAHT6fTsPXWLb/dcISU9ZzC+\nUk5WdHrVk9qezk/djFcI8XJxttfwP5+y/M+nLMlpWo5fiObQn3e5cD2BuPsZ/N+vl9m0L4r/+ZSh\nlX8lbCwlcSOEKBh2dnY0a9aMkJAQQkJCSEtLY9WqVbz55psAtGnThmXLltGwYUPUajWLFy+mbdu2\nufZjY2PDa6+9xpw5c5g6dSqXLl1iy5YtLFmypLDfkhBCFEuSsBGiAN2ISWbF1vNE3b4P5EzP/Vag\nO03rlpWBQ4UQT83G0owmdcrSpE5ZbsamsOvYdQ78cYcMbTbbj1xn3+nbvPlKJV6tWw4ztdQ1QhS2\n69evY23tWNTFKFAzZsxg1qxZBAUFodVqeeutt+jZsycAXbp0ITY2lg4dOqDVannzzTfp0aMHALdu\n3aJVq1b8+OOPlClThilTpjBhwgSaNGmClZUVn3zyiaFljhBCvOwUer3+hWrDFBOTlO9tVCoFTk42\nxMcnS5Ouh0hcTMtLXLKydWw7fI3N+6LI/rv/om81F95pWhlHWwuT2/yXyWfFtGeJS8mStgVUqhdX\nfuv/F+1zm5ymZeex6/x05BqZWh0ALg6WdH3DixqVnPK8nxctLs/LyxSXQ4cO8PXXa7h06SJ6vZ5q\n1arTt+8Aqlatnmvd/MRl2bLFREVdYerUz1i9ejlXr0YxbtyUAnkPqampNG/emPXrN1O6dJmn2seJ\nE8cYN24kP/64K1/bXb58gU8+GcLmzT9J3V8I5Nz/+ZCYmCZxMU3iYlpBnPtLCxshnrObMcl8seUc\nV+/mnEA421nQvWVVoxlhhBDiebOxNOOtQHea1CnLpn2R/Hb6NtEJacz55nf+V6cM77xWGQszVVEX\nUxRzmzd/xxdfRDBy5Fj8/PzJzs5m48b1DB7cn4iI5bi7ezyX43Tr1vO57Kc4SklJRqvVFnUxhBBC\nvAAkYSPEc6LX69n9+y2+2XUJbVbO3e3/+ZSl4/88ZOpdIUShcbS14IOW1XitXnlWbD3HX3eS2P37\nLS7dSKT/WzUpU0Km4CwOsnRZ3EtPLJRjOWrsUSuf/D2Unp5OePg8JkyYSqNGgQCo1Wo6d36fhIR7\nXL0ahbu7B/HxcYSGhnD06CEsLDS8+WZrunXrjUplRmpqKosWhbFnzy8AvPJKAB9+OBQbG+PZLx5u\nbTNt2kSsra25ePECly5doEKFSowYMQYvr6ro9XpWrFjKxo3rMTc3p2PHzkREhPHNN9/lq+XMsWNH\nWLYsguvXr5GZqcXXtyFjx05Co9GQlJTEvHmfsX//b6jVZgQFtaZ//8FG26ekJPPRRwOoXLkKI0aM\nITMzg0WLwti9+xf0ej2vv/4GwcEDSU5OYujQwWRmZtC0aQAbNvyAvb1DnssphBBCPEyuIoV4DpLT\ntKzadp7jF2OAnAumXq2qUT0f3RCEEOJ5Ku9iw5hu9fjx4FU27YviZmwKU1cfo1/bGtTyKFHUxXup\nZemymHxoNnHp9wrleM4aR8Y3/OSJSZszZ34nOzsLPz//XK/17z/I8Pvo0Z9QunQZ1q/fTFpaKmPH\njiAzM4vg4EF89tk07t2LZ9Wqb1CpVEydOp7Zs6cxadKMxx77p5+2Eha2mAoVKjB9+iQWLw4nJCSc\nH3/czNatP7Bo0TKcnJyYMmW80WxEeZGWlsaYMZ8wbtxkAgKaEB19lwEDerNz50+0bv0Wn38+nbS0\ndP7v/zaTnZ3FoEH9KFu2POXLVwByElkjRgzF07MyI0aMQaFQEB4+nxs3rrFq1dfodHrGjRvJ6tXL\n6dUrmLlzQxkzZiTbtu2SrgJCCCGeSaGORHj27Fk6dOhAnTp1aNu2Lb///rvJ9VatWkXTpk2pX78+\ngwYNIjY2tjCLKUS+RN2+z8QVRwzJmrpVSjKpp68ka4QQRU6lVNKmkRsjOvtgZ21OemY28zec5teT\nN4u6aKIYSkhIwNbWDrX60Ymdmzdv8Mcfp/noo+FYWVnj4uLCRx99xI8/biEjI53du3fRv/8gHB0d\nsbOz48MPh/DLLzvJyEh/7LEDAhpTuXIVLCw0NG3anOvXrwOwY8c2OnXqTPnyFbC2tmHAgMGP3Y8p\n5ubmLF++loCAJiQnJxMbG4O9vQMxMTFkZmayd+9u+vTpj52dHY6OTsyaFYK/fyMAsrN1jB49HIVC\nwciRY1EoFOj1erZu3Uz//oOwt3fA0dGRXr2C2bz5u3yXTQghhHicQmthk5GRQb9+/ejXrx8dO3Zk\n06ZN9O/fn507d2Jt/U/z7K1bt7JgwQIWL15MzZo1WbhwIf3792f9+vWFVVQh8uy3U7dYs+MiWdk6\nzNRK3n2tMv+rU0am6hZCFCteFRwZ160+od+e5np0Mmu2XyA1XUsr/0pFXbSXklqpZnzDT4pdlyhn\n5xLcv3+frKysXEmb+/fvY2Vlxb178VhaWuLg8E83nzJlyhAfH0dCQgJZWVmUKvVPVyVX19Lo9Xpi\nYmIee2wHh39mVFKr1ej1OV2LY2NjcHFxNdpffqlUKvbv38u6dV8B4OlZmfT0NHQ6HUlJ99Fqtbi4\nuBjWL1u2HAA3blwnOTkJnU7H+fNnuXHjOuXLVyAh4R4ZGRkMGhRs+L7X6/VotVlkZGTku3xCCCHE\noxRawubQoUMolUq6dOkCQIcOHVi1ahV79uwhKCjIsN6OHTvo1KkTPj4+AAwaNIiVK1dy4cIFvLy8\nnngchUKBMp/thpRKhdGjyCFxMU2pVKDN0rHqp/P8cjznLnVJBw2DO9SiguvLObuDfFZMk7iI4sTZ\nXsOn79Ul7NvTnL+WwLd7IgEkaVNE1Eo1Ja2K12D0NWp4Y2am5tCh/QQENDF6bebMKVhZWREcPJC0\ntDQSExMMY7PcuHEDOzt7SpZ0wdzcnDt3bhsSOrdv30KpVBolZPLDxcWVu3fvGJ7HxEQ/ct1p0ybS\ntWsPKlSoSFZWFgAWFhacOXOK5cuXsnTpKkM3p8GD+wE5iSIzMzNiYmIM7+fw4YMkJiZSokQJbGxs\nCQkJ5/PPZzBr1lTCwhZjZ2ePmZkZy5evNSR30tLSiI+Pw8LixZsJUgghRNEptIRNVFQUHh7GMwu4\nubkRGRlptEyn06HRaAzPFQoFCoWCq1ev5ilh4+xs/dStGxwcZCBGUyQuxpJSMxm3+AB/RsYBUNfL\nheHv18PWyryIS1b05LNimsRFFBeWFmqGdKzNwu//4PSVOL7dE4mFmYpm9csXddFEMWBhYUFw8Id8\n9tl0lEoVvr4NyczMYN26rzh27AgREcsoWdKFevV8CQ2dw8cfjyItLYXQ0FBatHgDpVJJ8+YtiYgI\nY+LE6SiVShYunI+/f6Ncgw7nVVDQmyxduohGjRrj7FyCJUsWPnLdyMgr/PzzT3zwQW9+/nkbzs4l\ncHR04uLFC6hUSiwsLMjOzmbHjm2cOnWSGjW8UalUNG36OsuWRTB27CTS09NZsGAeHTt2BkClUqJU\nKunXbxDvvdeBTZs28tZb7Xn99TeIiAhnxIgxqNVqZs+ezp07t1m48AvMzMzJzMxEq9WizEPLJiGE\nEOJRCu1bJDU1FUtLS6NlGo2G9HTjPs1NmzYlJCSEZs2a4e7uzpIlS0hPT89zE9O4uJSnamHj4GBN\nQkIKOp0MDveAxCW3u/dSCfnmFHfiUwFo/UpF2jfxQJueSXx6ZhGXrujIZ8W0Z4mLk9PTXdwI8STm\nZioGtqvJvPWnOXf1Hl/tvISDjQX1q7o8eWPxwnv77Y7Y2NiyYsUSJk8eh1KppHr1GoSFLcbd3ROA\nCROmMH/+53Tq1AaAtm3b0qNHMACDBw9j4cJQund/h4yMTAIDmzB48MdPXZ7XX3+DqKhI+vTpjqWl\nJS1a5LTKNjMzy7Xu8OGfMnv2dL7+eg2lSpVm/PgpKBQKfH0b8uqrzejW7V1UKiVeXtVp2bI1V6/+\nBcDQoSMIDZ3Du+++jUKhoG3bt3nzzbc4ceKYYd92dnYMGjSUOXNm0qhRIEOGDGfhwjC6du1Eeno6\ntWvXYfLknIGVPT0r4+npyRtvNGX58rWUKycJUSGEEE9HodfrC+XqasWKFezfv58vvvjCsGzw4MFU\nrVqVAQMGGK27ZMkSvvnmG7RaLR06dGDnzp0MHz6cJk2a/Hu3ucTEJOW7bCqVAicnG+Ljk2U0/4dI\nXIxdvplI6IbTJKdpUasU9GxVjYbVSxV1sYoF+ayY9ixxKVny5exe9yzyW/+/7J/b9MwsZn99kqjb\nSZiplYzsUhf3MnYvfVweReJiWkHH5dKlizg6OlGiRM7MZn/9FUW3bu/w8897sbDQPGHroiF1f+GS\nc//nQ2JimsTFNImLaQVR/xfaLFHu7u5ERUUZLYuKisLT09NoWXR0NEFBQfzyyy/89ttv9OjRg6tX\nr1KtWrXCKqoQuZy6HMvsr0+SnKbFSqNmct9XaOSd/4EPhXgZbN26lZYtW+Lj40OrVq3YuXOn0es6\nnY6uXbsya9YswzK9Xs+cOXNo2LAhDRo0YOrUqfmeulfkj8ZczeD2tXC2s0CbpSN842kSU17eloKi\neDp06ABTpownNTWVjIx01q5dRZ06dYttskYIIYR4ngotYePv709mZiZr1qxBq9WyYcMGYmNjCQgI\nMFrvwIEDBAcHEx8fT3JyMlOnTqVRo0ZGo/cLUZiOnLtL+MYzaLN0ONtpGNe9Pt6eJYq6WEIUS1FR\nUYwePZpp06Zx8uRJxowZw5AhQ4iPjzess3z5co4dO2a03dq1a9m9ezebN29m69atnDhxguXLlxd2\n8V869jYWfNShNuZmShKSM1m86Q+ydbqiLpYQBu+804XSpUvTqVMb2rRpQVLSfcaNm1zUxRJCCCEK\nRaGNYWNubs7SpUuZOHEiISEhVKxYkUWLFmFlZcX48eMBmDx5Mm3btuXChQsEBQWh0+lo0qQJn332\nWWEVUwgje0/dYtW28+iB0s5WDH/XhxIOcldPiEdxc3Nj//79WFtbk5WVRWxsLNbW1pib5wzKff78\neTZu3Mjrr79utN2mTZvo3r27ITkfHBzM/Pnz6dOnT56Pnd9ZAmUWrxwVS9vSI6gaizf9yflrCWze\n9xe93qr10sfl3+TzYlpBx8XS0oIxY8YXyL4LinxWhBBCPC+FOnR91apV+eabb3Itnzz5nzslCoWC\nkSNHMnLkyMIsmhC5/HzsOl/vvARARVdbhr1TW2aCEiIPrK2tuX79Oi1atECn0zFx4kRsbGzIzMxk\n5MiRTJkyhfXr1xttExkZadRF1s3NjaioKPR6fZ5n/nvaWQJlFi9o3diTazEpbDvwF5v3RdGoTjmq\nVnIq6mIVS/J5MU3ikpvERAghxLOSuQaFMOGXEzcMyZrK5ez5qENtrDTy7yJEXpUuXZpTp05x7Ngx\nBgwYQMWKFdm9ezcBAQHUq1cvV8ImLS0Njeaf1muWlpbodDoyMzOxsLDI0zHzO0ugzG5m7O2ASpy6\nGM2t2FTmfHWcKb19MVerirpYxYZ8XkyTuOQmMwQKIYR4XuQKVIh/2XvqFl/uuAhAlfIODO1YGwtz\nuWgRIj/U6pyvF39/f5o3b8706dNRKpW5EjUPaDQaMjIyDM/T0tJQq9V5TtZAzsDFTzNOsU6nlxkO\nAJVSSZ/WNZi6+hh34lL5v18u0/m1KkVdrGJHPi+mSVxyk5gIIYR4VoU26LAQ/wX7z9xm1bbzAHiW\nteejDrUkWSNEPuzZs4cPPvjAaJlWq6VOnTpcu3aNV155hfr167Nlyxa+/PJLgoODAfDw8DCaSTAq\nKgp3d/fCLLoAKpaypU1AJQB2Hr3BlZuJRVsgIYQQQoiXmCRshPjb8QsxLN96Dj3gVtqOIR1rY2kh\njdCEyI/q1avzxx9/8P3336PT6dizZ48hiXPy5EmOHTvGsWPHaN26Ne+//z6LFy8GoE2bNixbtow7\nd+4QGxvL4sWLadu2bRG/m5dT61cqUaGULXpg5bbzZGXLrFFCCCGEEEVBEjZCABevJ7B485/o9VDB\nxYZh78iYNUI8jZIlSxIREcHq1aupX78+8+fPZ8GCBXh4eDx2uy5dutC0aVM6dOhAq1atqFu3Lj16\n9OUMlrYAACAASURBVCikUouHqVVKBnWsgwK4GZvCruM3irpIQgghhBAvJbkiFS+9GzHJhG44TVa2\njhL2GoZ2qo21xqyoiyXEf1b9+vXZuHHjY9eZOXOm0XOVSsXQoUMZOnRoQRZN5FHVSk4E1i7D3lO3\n2LQviobVXbG3yft4QkIIIYQQ4tlJCxvxUotLTGfu/50iNSMLG0szPn6njlyUCCEE0PFVDywt1KRn\nZvPtnsiiLo4QooidPn2agIAAw/MzZ85QrVo1fHx8DD8RERH/z96dh0Vdrg0c/84wDMuAbK4gIJv7\nvqPmnuZeuVCWRz2ZoGa+2GKnTr6mWZ1KezU74ZJZasf0aFkumbsGLpE7ixuICG4gi7INzMz7BzlF\njDAgMAj357q4hN/8nmfuueUaZu55fvcDwLBhw4ocb9OmDc2aNePmzZvF5i1pHiGEqO1khY2otXLy\nCljy39Ok3c3DxtqK0HHtaOBqb+mwhBCiWqijUfNkLx/+s/ci4WevM7irJx71ZMthIWobg8HA5s2b\n+eCDD7Cy+mMjhpiYGHr37m3sRfZn27dvN36v1+uZNGkSHTp0oEGDBsXOLWkeIYSo7WSFjaiV9AYD\nK3+M5trtLBQKmPZka3wa1bF0WEIIUa306+hBXSdbDMCWQ7LKRoja6H5fspCQkCLHo6Ojad68eanj\nv/76a+7du8fLL79s8nZz5xFCiNpIVtiIWmnzwcucupQCwDP9A2jr52bhiIQQovpRWSl56jFfVm6L\n5uTFFC4nZeDn4WTpsIQQVWj06NGEhIRw/PjxIsdjYmJQq9X0798fvV7PkCFDCA0NRa1WG8/JyMhg\n2bJlfPHFF0VW55R1npIoFAqUZfwIWqlUFPlXSE4eRPJimuTFtMrIixRsRK0TfvY6O49eBaB3O3cG\ndm5s4YiEEKL66tayATuOJZB0O4vNBy/z2rMdUCjkBZoQtUX9+vVNHndxcaFbt24EBQWRmprKrFmz\nWLp0Ka+++qrxnG+++YZ27drRrl27B85vzjwlcXPTlPs5ydlZU65xNZnkxDTJi2mSF9MqMi9SsBG1\nSvz1TL76KRaAZp7OPD+oqbzxEEKIEiiVCkb39mPp5jPEXk0n6sodWvvIqkQhars/Nwa2t7cnODiY\nxYsXFym0bNmyhTlz5jz0PCVJTc0q1wobZ2cN6elZ6PWGsg2uoSQnpkleTJO8mPYweXF1Nd0nUAo2\nota4l5PPv787R4HOQF0nW2Y83QaVlbRxEkKI0rTzd8PPow6XkzLZfDCOlk1cUUqxW4haKyMjg7Cw\nMGbMmIGDQ+GbjLy8PGxs/thp8/Lly6SkpNC7d++Hmqc0BoMBna58j0OvN6DTyZvNP5OcmCZ5MU3y\nYlpF5kXerYpa4X6T4dTMXFRWSmY81QYHO2tLhyWEEI8EhULBmD5+ACTcuMupiykWjkgIYUmOjo7s\n3r2bZcuWkZ+fT0JCAmFhYTz99NPGc06dOkWrVq1K7EVjzjxCCFGbScFG1ArbIq5wNi4VgOcHNcW7\noaOFIxJCiEdLMy8XWni7ALD9SAIGg3yiJkRtpVQqCQsLIzY2lu7duzN+/HieeOIJJk6caDwnKSmJ\nevXqFRubnJxMhw4dSE5ONmseIYSozeSSKFHjRcXfYevheAB6tWnEY20bWTgiIYR4NA0L9CYmIY34\n65nEXk03FnCEEDVft27dOHbsmPFnf39/1qxZ88DzH7SNt7u7OydPnjR7HiGEqM1khY2o0TKztKzc\nFo0B8KzvIE2GhRDiIbTwdsGnUeEKxR1Hrlg0FiGEEEKImk4KNqLGMhgMrN4RQ2aWFhtrK6Y92Rq1\ntZWlwxJCiEeWQqFgaHdvAKKupHHlRqaFIxJCCCGEqLmkYCNqrH0nkjhzubBvzfiBATR0tbdwREII\n8ejr0LSe8fl0x5EEC0cjhBBCCFFzScFG1EjXbt3j232XAOjcvD69pG+NEEJUCKVCwZDuXgD8dv42\nN+5kWzgiIYQQQoiaSQo2osbJL9Cx/McoCnR6XOvYMPGJZtK3RgghKlBgq4a4ONpgAHYelVU2Qggh\nhBCVQQo2osb5/pd4km5noQBeHN4Sja21pUMSQogaRWWlZHAXTwCORN0kM1tr4YiEEEIIIWoesws2\nubm5lRmHEBXicnIGPx27CsDgbl4085ItZ4UQojL0auuOjdqKAp2eg6eSLR2OEEIIIUSNY3bBZsSI\nEcTExFRmLEI8lPwCHau3x2AwQCM3e556zMfSIQkhRI1lb6uiV5vC/mD7T1yjQKe3cERCCCGEEDWL\n2QWb/Px86QMiqrXvDsdzPTUbhQL+PqwF1irZwlsIISrTwE6NUQDp97REnr9l6XCEEEIIIWoUlbkn\nDh8+nMmTJzNs2DA8PT2xtbUtcntQUFCFByeEuS4lZbDreOGlUE9088LP3cnCEQkhRM3XwNWeNn5u\nnLmcyp7Ia3Rv2dDSIQkhhBBC1BhmF2x27NiBnZ0d+/btK3abQqGQgo2wmAKdnjU7YzEYwL2uhid7\nyaVQQghRVR7v7MmZy6nEJWdyOSkDPw8pmAshhBBCVASzCzamCjVlFR0dzdy5c7l06RLe3t688847\ntG/fvth5mzZtIiwsjPT0dAICAvjnP/9J69atH/r+Rc2089hVklMKd4WaPKS5XAolhBBVqGUTF9zr\nakhOyWJ3ZKIUbISoZtLT0zl//jwFBQUYDIYit/Xq1ctCUQkhhDCH2QUbgNTUVDZt2sSVK1d47bXX\nOHbsGAEBAQQEBJQ6Ni8vj5CQEEJCQhg7dixbt25l2rRp7NmzB41GYzwvNjaWjz/+mA0bNuDt7c2q\nVauYNWsWe/fuLfujEzXezTvZ/Bh+BYC+HT3kjYIQQlQxhULBwE6N+XrXeX47f5u0u3m4ONpYOiwh\nBLBlyxbmzZuHVqstdptCoZANRYQQopozu+lwdHQ0gwcP5sCBA2zbto3s7GzCw8MZM2YMR44cKXX8\n0aNHUSqVjB8/Hmtra8aMGUPdunU5ePBgkfMSEhLQ6/XodDoMBgNKpbJYvxwhAAwGA1/vOk+BTo+T\ng5rRvf0sHZIQQtRKga0borFVodMbOHgqydLhCCF+t3TpUsaNG0dkZCSxsbFFvqRYI4QQ1Z/ZK2ze\nf/99Jk6cyMyZM+nQoQMACxcuxMXFhY8//pjNmzeXOD4+Ph4/v6JvqH18fIiLiytyrFevXjRp0oRh\nw4ZhZWWFRqPh66+/NjdMFAoFSrPLUIWUSkWRf0Wh6p6XX87cICYhDYAJg5vhqLGukvut7nmxBMmJ\naZIXUVvYWFvRs00jfv41kUOnkxneowkqqzL+MRZCVLi0tDQmTZqEg4ODpUMRQghRDmYXbKKionj3\n3XeLHQ8KCmLdunWljs/OzsbOzq7IMVtbW3Jzc4scy8vLw9/fn7lz59K0aVNWrlzJSy+9xPbt281a\naePmpin39uPOzprST6qFqmNe7mZr2bD3EgBdWzZkUKBPlW87Xx3zYmmSE9NqW1527NjBp59+yo0b\nN3B3dyc0NJSBAwcSGxvLggULiImJwcHBgaCgIKZPn45CocBgMLB48WI2bdqETqdj1KhR/OMf/8DK\nSnpSPSr6tHfn518TSb+n5fSlVDo1q2fpkISo9Xr06EFERATjxo2zdChCCCHKweyCjZOTE8nJyXh7\nexc5HhUVhaura6nj7ezsihVncnNzsbe3L3Js2bJlNGzYkDZt2gAwY8YMNm7cSEREBP379y/1flJT\ns8q1wsbZWUN6ehZ6vaH0AbVEdc7L1z+d5262FrW1kmf6+5GWllVl912d82IpkhPTHiYvrq6P5qeh\n8fHxvPnmm6xevZqOHTsSERHB1KlTOXjwINOmTWPSpEmsXbuWGzduMG7cOJo3b86AAQNYv349Bw4c\n4IcffkChUBAcHMzq1at58cUXLf2QhJkauWlo4e1CTEIaB05ek4KNENVAq1atWLhwIfv27cPHxwdr\n66KrkWfPnm2hyIQQQpjD7ILNs88+y9y5c3n11VcBOH/+PIcOHeLTTz9l8uTJpY739fUtthInPj6e\n4cOHFzmWnJxcZCWOQqHAysrK7E9ZDQYDOp1Zpxaj1xvQ6eTN5l9Vt7wk3rrHvhPXABge2ARnBxuL\nxFfd8lIdSE5Mq0158fHxITw8HI1GQ0FBASkpKWg0GmxsbNi+fbvx+T0tLQ29Xo+TU2Gj8K1btzJx\n4kTq168PQHBwMEuWLJGCzSOmXwcPYhLSiLqSxs072TRwtS99kBCi0hw7doy2bduSlZXFuXPnitxW\n1SuThRBClJ3ZBZupU6ei0Wj44IMPyMnJ4aWXXqJu3bqEhIQwadKkUscHBgai1WpZu3YtzzzzDFu3\nbiUlJaXYdoJ9+/blk08+YejQoTRr1oy1a9ei0+no1KlTmR+cqHkMBgPrd1/AYID6znYM7upp6ZCE\nEH+h0WhITExk8ODB6PV65s2bV6R/woABA7h27RojRoygY8eOAMTFxeHv7288x8fHh/j4eAwGg9lv\nKsraw0x6DJn2MHnp1Lwezg5q0u9pOXg6mWcHlr6L5KNCfl9Mk7wUV51ysnbtWkuHIIQQ4iGYXbBJ\nTk7m2Wef5bnnniM7OxudToejoyM6nY6zZ88aL2F6ELVazcqVK5k3bx6LFy/G29ubzz//HHt7e+bO\nnQvA/PnzCQoKIjMzk5kzZ5KZmUmLFi1YtWqVNEsTAPwae4sLiekAPDMgAGuV9LcQojpq1KgRp0+f\nJjIykunTp+Pt7U1gYCBQ2OPm5s2bBAcH89lnnzFz5kxycnKK9Cmzs7NDr9ej1WqxsTFvi+jy9jCr\nbT2GzFXevAwObMK3uy8QfvY6U55qi411zXqelt8X0yQvxVkqJ7/88gvdu3dHpVLxyy+/PPA8hUJB\nz549zZ73zJkzTJ8+3Tjn2bNnGTduXJHn7uDgYEJCQjAYDHTq1AmD4Y/VpZ06dWLVqlXF5s3IyODN\nN9/k6NGjODo6MmPGDMaOHWt2XEIIUZOZXbAZMGAA4eHhuLq6Fuk7k5CQwPPPP8/p06dLnaN58+Zs\n2LCh2PH58+cbv1coFEydOpWpU6eaG5qoJfK0Or7dV9houLWvK+383SwckRDiQVSqwj8vgYGBDBo0\niL179xoLNjY2Nnh5eTFlyhTWrFnDzJkzsbW1JS8vzzg+JycHlUpldrEGyt7DTHovmfaweenWvB4b\n91zgbnY+u8Lj6NW2USVEWfXk98U0yUtxlu5fNmXKFMLDw3Fzc2PKlCkPPE+hUJi1tbfBYGDz5s18\n8MEHRVoUxMTE0Lt3b5YvX15sTEJCAgAnTpwotZD+9ttvY29vT0REBOfPn+fFF18kICCA9u3blxqb\nEELUdCUWbP7zn//w2WefAYVP1iNGjCj2pJuVlUVAQM1Z8iyqr+1Hr5B2Nw8rpYJnBwTItddCVJK0\ntDRWrFjBuXPnyM/PL3a7qcL7fQcPHuTLL79kzZo1xmP5+fnk5+czYMAANm/ejLOzs/F4nTp1APDz\n8yM+Pp527doBhT3OfH19yxR3eXuY1aYeQ2VR3rw4a2xo71+XkxdT2PfbNQJbNayE6CxHfl9Mk7wU\nZ6mcxMbGmvy+vMLCwti5cychISGsXLnSeDw6OprmzZubHBMdHU2zZs1Kfa2WlZXFnj172LVrFzY2\nNrRt25bhw4fz/fffS8FGCCEopWAzZswY47L0N998k+DgYBwdHY23KxQK7O3t6d69e6UHKmq3W2nZ\n/HTsKgCPd/GkkZssvRaisrzxxhucPXuWkSNHlvly1JYtW3Lu3Dm+//57Ro4cyeHDhzl48CAbN24k\nJiaGTz75hLfeeovExERWrVrFjBkzABg5ciRffPGFcRn/8uXLGTVqVGU8PFEF+nXw4OTFFC4nZ3L1\n5l28GjiWPkgIUWkMBoPJArxarS517OjRowkJCeH48eNFjsfExKBWq+nfvz96vZ4hQ4YQGhqKWq0m\nJiaGe/fuMWrUKG7dukWXLl146623aNCgQZE5EhISUKlUeHr+0ZPQx8eHn3/+2ezHVtb+ZVC9+gxV\nF5IT0yQvpkleTKuMvJRYsLG2tubJJ58EoHHjxnTs2NG4zF2IqvTtvksU6Aw4adSM6NHE0uEIUaMd\nPXqUr7/+2rjapSzq1atHWFgY7733HvPnz6dJkyZ89tln+Pn5sWTJEt555x169uyJk5MTkyZN4qmn\nngJg/PjxpKSkMGbMGPLz8xkxYoRZOxCK6qmljyv1ne24lZ7DgZNJ/O0J05/CCyEq17Fjx5g/fz5X\nrlxBr9cbj99v6G7OJVH3d+/7KxcXF7p160ZQUBCpqanMmjWLpUuX8uqrr6JWq2nfvj2zZs3CxsaG\nhQsXMnPmTDZu3Fhkjuzs7CI9cABsbW3Jzc01+zGWt38ZSO8lUyQnpkleTJO8mFaReSmx+rJ48WKm\nTZuGnZ0dv/zyS4mNy2bPnl1hQQnxZ1FX7nDyYgoAY/v5YWcjRUMhKpObm1uZesf8VefOndmyZUux\n440aNSIsLMzkGCsrK0JDQwkNDS33/YrqQ6lQ0KeDO5v2X+ZI1E3G9vOX524hLGDu3Ln4+/szZ86c\nYoWRh/Xn53N7e3uCg4NZvHgxr776KjNnzixy7pw5c+jevTu3bt0qUgCys7Mr0r8MIDc3t0i/zNKU\ntX8ZSO8lUyQnpkleTJO8mFYZPcxKfPV08uRJ8vPzsbOz4+TJkw88T3qJiMqiNxj47/7LAPi616F7\nDeuFIER19NJLL7FgwQLeeustvL29sba2LnK7OUvohejVphHfHYojL1/Hkagb9O/Y2NIhCVHr3Lp1\ni7CwMHx8fCp03oyMDMLCwpgxY4bx0tm8vDxjsX/FihX07NmTVq1aAaDVagGKfRjg7e1Nfn4+ycnJ\nuLu7A4U9zPz9/c2Opbz9y0B6L5kiOTFN8mKa5MW0isxLiQWbtWvXmvxeiKoSGXuLhJt3ARjb1w+l\nFAeFqHSLFy8mPT2d0aNHm7zdnCX0Qjjaq+ncvD5Ho26y/2QS/Tp4yAc8QlSxxx9/nIMHD1Z4wcbR\n0ZHdu3djMBh45ZVXSE5OJiwsjHHjxgEQFxfH4cOHWbp0KSqVioULFzJgwACcnJyKzOPg4MCAAQNY\ntGgR7777LhcvXmTbtm2sWLGiQuMVQohHVYkFm5IugfozhUJBz549KyQgIe4r0OnZcigOgDa+bjTz\ncrFwRELUDosXL7Z0CKKG6NfBg6NRN0m6ncXFaxk09XS2dEhC1CqzZ89m5MiRbNu2DU9PT5R/uXZo\n0aJF5ZpXqVQSFhbGu+++S/fu3bG1tSUoKIiJEycC8M9//pOFCxcyZMgQ8vPz6du3LwsWLAAgOTmZ\nYcOGsX37dtzd3VmwYAH/+7//S58+fbC3t+e1114rVw81IYSoiUos2EyZMsWsScxtWiZEWRw+ncyt\ntBwUwOg+ZdveVwhRfl27drV0CKKG8PdwonE9DdduZ3HgZJIUbISoYm+99RYKhYLGjRs/dA+bbt26\ncezYMePP/v7+rFmzxuS5Dg4OvP/++yZvc3d3L9JqwdnZmSVLljxUbEIIUVOVWLCJiYmR5cvCIvK0\nOn4IvwJAt1YNZEtYISrZM888w4oVK6hTpw5BQUElPvdv2LChCiOzHL1Bz683TnI2JZpbOSkU6HXY\nqmyws7LFTmWLs40TzrZOuNg44WzjXPizTR2slFaWDr3aUCgU9OvgwdqfLxB5/hbPZAdQx156IAlR\nVSIjI1m3bh1t2rSxdChCCCHKocSCTY8ePejSpQs9e/akR48eeHp6VlVcopb7OTKRjCwtVkoFTz0m\nq2uEqGy9evUyNhd+7LHHLByN5aXkpLImagPxmQllGqdAQR21A862zr8XcpxwsS0s5rjU0qJO91YN\n2XjgMnlaHeFnrjOku7elQxKi1vD29jY2/BVCCPHoKbFgs2vXLo4ePUp4eDirVq1Cr9fTs2dPAgMD\nCQwMxNlZljaLincvJ5+fjhW+SerXwYN6znYWjkiImu+ll14yfj98+HCaNGliuWAsTG/Qs+rsWhLv\nJQPQ0q0ZvnWaYGNlTY4uj7yCPLIKsknPzSA9L4O0vHTydIVviAwYyNDeJUN7lwQSTc5vLOrYOONi\n6/R7EeevhZ2aU9Sxs1ER2KohB04mceBUEoO7eUkDeSGqyLRp03jjjTeYMGECXl5eqFRFX/r36tXL\nQpEJIYQwR4kFmzp16jBo0CAGDRoEwLVr14iIiOCnn35i/vz5NGrUiB49ehi/hKgI2yKukJOnw0Zt\nxfAeTSwdjhC1zpAhQ2jZsiUjRoxg6NCh1K9f39IhVanIm6eMxZoXWj9Px/ptSx2TU5BDWm4GaXkZ\npOelk3a/mJObXnJR5+6DizqOagdcbJypa+dKfft61LevSz27ujSwr4u9tX3FPeAq0Le9OwdOJnE7\nPZeo+Du08XWzdEhC1AqhoaEAvPfee8Vukx6UQghR/ZVYsPmrxo0bM27cOMaNG4fBYCAqKoqIiAhW\nrFghBRtRIVIycth34hoAT3T1oo5Geh0IUdX27NnDzp072bZtGx999BEdO3ZkxIgRDB48uNiWrDVN\nvi6fHy7/BEDbuq3MKtYA2KnssHOww92h4QPPeVBR535h569FnUztXTIfUNRxUjvi6ehBY0cPPB3c\naezogZutS7XtO+fVwBE/jzpcTspk/4kkKdgIUUViY2MtHYIQQoiHUKaCzZ07d7C3t8fW1pbo6GgO\nHjxI69atmTp1amXFJ2qZrYfjKdAZcLS3ZlAX6ZkkhCV4eHgwZcoUpkyZQmJiIrt27WLz5s28++67\n9OzZk88//9zSIVaaX2+eJC0vHaVCySi/IRU6d3mKOndy07mdncLtnBRuZaeSq8sFKFyhkxrLudQ/\n3ow5WGsIcPYlwMWPpi5+NLSvX60KOP06eHA5KZPTl1NIzcjFzenhdqwRQpg2atQoevToQc+ePena\ntStqtXz4JYQQjyqzCzZ79uxh9uzZhIWF4eHhwfPPP4+7uzurVq1i9uzZTJgwoTLjFLXAtdv3iDh3\nA4ARPZpgZ1OmeqIQohK4uLhQr149GjRowIULF7h165alQ6pUlzOuANDcNYCGmqq/FKykoo7BYOBe\nfhY3s29z7V4y1+4mc+1uEslZN9EZdNzLz+Lk7bOcvH0WADdbV9rVa0X7em3wcfJCqVBW9cMpokvz\n+mzYe4l7OfkcOp3MU72lobwQleHDDz8kIiKCNWvWMHv2bJo3b27sQdmmTZtqVci1lAKdnuspWaRn\nZKPXGSwdTrWgtFKQp1dITv5C8mKa5MU0pZUCxzoVe9m62e+IlyxZwsyZM+nRoweLFi3C3d2d7du3\ns2/fPhYuXCgFG/HQthyMwwDUdbKlbwcPS4cjRK1179499uzZw08//URERATu7u4MHz6c0NBQfHx8\nLB1epbqaWXhJprdj9Vvhp1AU9rVxVDvg7/zH/0OBvoDkrBvEpSdwMf0yF9PiyCrIJjX3DvsSD7Mv\n8TDONk70aNSFHu5dcbG1zIYB1iorerVpxE/Hr3LoTDIjejZBZWXZIpIQNVGzZs1o1qwZkydPRqvV\ncvLkSSIiIpg/fz7Xrl2ja9euBAYG0qNHD7y9a9+ubQU6PW+uOEpKRq6lQxFC1ED1Xe15f2o3FFRM\ncdzsgs2VK1cYPnw4APv372fAgAFA4R+FlJSUCglG1F4XEtM5danw9+ip3r7yIl4ICwoMDMTFxYUh\nQ4YwY8YM2rRpY+mQqoRWp+VGduEKIu86jS0cjflUShVejo3xcmxMX8+e6A16Eu8mcfp2FKdun+Nm\n9i3S8zLYcWUPO6/spXXdFgzy7ouvU5Mqj7VPB3d+On6VjHtaTl1MoXPz2tXQWoiqplar6datG926\ndSM0NJSMjAzjDrCrV69m9+7dlg5RCCFECcwu2DRo0IDo6GjS0tK4dOkS77zzDgAHDhygceNH54Wt\nqH4MBgP/PXgZAM/6DnRr2cDCEQlRu61YsYLu3bvXumXz1+5dR2/QA+Dp+Oiu8lMqlHjX8cS7jicj\n/Z4g+d4Njlz/laPXI8kuyOFsSjRnU6Jp6dqMYb6P06SOV5XF1sDFnlY+rkTF32H/ySQp2AhRBfR6\nPQaDASsrK7Kzs8nJyeH5559n/vz5lg7NIlRWSv41LRCdwor0jCy5nON3SisFzk4ayclfSF5Mk7yY\nprRS4O9dl7uZ2egqKC9mF2z+/ve/M2vWLADat29Pp06dWLZsGWFhYXz44YcVEoyonU5fSuXStQwA\nRvfxQ1nL3iQKUR18++23RX6+evXqA88NCgqq7HAs4urdwsuh6qgdcbapObthuTs0ZHTACEb4PsHJ\nW2fYm3iIpHvXib5znug752lXrzVjA0ZW2aVSfdt7EBV/h5iENK6nZtHITVMl9ytEbRQZGcnLL7/M\nokWL8PPzY8yYMWi1WnJycvjwww8ZOnSopUO0CJWVkvquGmyUhgp7U/Wos7JS4Co5KUbyYprkxTQr\nKwXWqoq9UsTsgs348eNp3749ycnJ9OrVC4BevXoxcOBAmjdvXqFBidpDrzew+ffVNc08nWnj62rh\niISonZYvX27WeQqFosYWbBIzkwDweoRX15REbWVNt0ad6NKwA6dun2N7/G5uZN3k9O1zxN65wEjf\nIfRuHIgVVpUaR/sAN1wcbUi7m8eBk8k8OzCgUu9PiNrsX//6F0OHDqV9+/Z8+eWX2NjYsH//frZt\n28ayZctqbcFGCCEeFWXahqdly5bk5uayfft2Bg8ejEajqZXNykTFORJ1g6SULADG9PWrdZdgCFFd\n7Nu3z9IhWNz9FTZejjX7Ml+lQknH+m1pX681x26c4LtL28jKz2bTxa0cv3mCya2fwdXVodLu30qp\npHc7d7b+Ek/42euM7uOL2rpyi0RC1Fbnz59nyZIl2NnZsW/fPgYOHGjsazNv3jxLhyeEEKIUZq/X\nuXPnDkFBQfztb3/j7bffJi0tjUWLFjFs2DASExMrM0ZRQ+UX6Pj+cBwAnZrWw8+j5lyCIMSjF75g\n0gAAIABJREFULjU1lbCwMN544w1SU1PZsWMHFy9etHRYlUar03I96yYAXo9Qw+GHoVQoCWzUmbe7\nvUqXBh0BSMhM5INjSzmaeKJS77t3O3eUCgXZeQUci7lZqfclRG3m7OxMUlISiYmJREVF0adPHwDO\nnDlD/frSQ0oIIao7sws2CxcuxM3NjWPHjmFjYwMULrP08vJi4cKFlRagqLn2n0giNTMPhQKe7uNr\n6XCEEL+Ljo5m8ODBHDhwgG3btpGdnU14eDhjxozhyJEjlg6vUtzIvoWBwmuwGzu4WziaquWodmBS\nq2eY0e4FHK0dyNXlsThiJZsvbEOn11XKfbo42tChaV0A9kZew2CQ69+FqAxjx45l+vTpBAUF4e3t\nTWBgIGvXrmXOnDlMnDjR0uEJIYQohdkFm4iICP7nf/4HjeaP5oBOTk688cYbREZGVkpwoubKzi1g\n25EEAB5r20iaTgpRjbz//vtMnDiRDRs2YG1tDRQW7SdMmMDHH39s4egqxz1tlvH7OmpHC0ZiOS3d\nmvFG11n4OhVe6rw74SDLTn9BTkFupdzfwE6FK5mu3rrHhcT0SrkPIWq7mTNn8v777zN16lTWrVuH\nUqnEy8uLpUuXMmHCBEuHJ4QQohRmF2x0Oh16vb7Y8bt372JlJdeei7L56XgC93LysVYpGdVLVtcI\nUZ1ERUUxcuTIYseDgoK4fPmyBSKqfFn52QDYqeywUtbev2nONk7M7hzCkIB+AFxIu8SSk8u5q71X\n4ffV1NMZr/qFvXL2/HatwucXQsC4cePw8vJi0qRJ1K1buKqtT58+9O3b17KBCSGEMIvZBZuBAwfy\n0UcfcefOHWNj2EuXLrFgwQIGDBhQaQGKmif9Xh4//1rY92hgp8a4ONpYOCIhxJ85OTmRnJxc7HhU\nVBSurjVzJ7d7+YUrbDTW9haOxPJUShWTO47juRajUaAg8W4Sn5z4nLTcil0Fo1AoGNjZE4ATF26T\nkpFTofMLIeDatWuoVGXaY0QIIUQ1YnbB5s0338TBwYGePXuSnZ3NiBEjGDFiBI0aNeLNN9+szBhF\nDfNj+BW0+XrsbVQMDZRdxoSobp599lnmzp3Lrl27gMJdRtavX8+8efNq7Jbe91fYOFjL5Zn3Pda4\nO5NbPYuVwoqb2bdZ9Nu/uZWdUqH30a1lfRztrTEYYN+JpAqdWwgBzzzzDDNmzGDNmjXs2bOHX375\npciXEEKI6s3skruDgwNLliwhMTGRy5cvU1BQgJ+fHz4+PmbfWXR0NHPnzuXSpUt4e3vzzjvv0L59\n+yLnzJ07lx9//NH4s8FgICcnh48//pgRI0aYfV+ierqZls2h04Wf3A8N9EZja23hiIQQfzV16lQ0\nGg0ffPABOTk5vPTSS9StW5eQkJAa26TyfsFGVtgU1alBe2xVdqw8+zVpeel8emolr3SajrNNxezq\nZ62yok97D7ZFXOHQqWRG9fTBRl17L0kToqL9+9//BuCDDz4odptCoSAmJsbsuc6cOcP06dONhZ6z\nZ88ybtw4bG1tjecEBwcTEhKCVqvlo48+YufOnWi1Wjp27MjcuXNxdy/e1L2keYQQorYrsWATHx9v\n8ri3t3exc0or3OTl5RESEkJISAhjx45l69atTJs2jT179hRpZDx//nzmz59v/HnJkiVERkbyxBNP\nlP5oRLX33aE4dHoDzg5qBnSqHVvnCvEo0Ov1KJV/LLp87rnneO6558jOzkan0+HoWNiI9/bt29Sr\nV89SYVaarN8viZIVNsW1cmvGjHYv8NnpVdzJTePTU6sI7RhSYbnq18GDnUcTyM4r4EjUDfp28KiQ\neYUQEBsb+9BzGAwGNm/ezAcffFCkb2VMTAy9e/dm+fLlxcYsX76cs2fP8v333+Po6MjChQt55ZVX\n+M9//lPs3JLmEUKI2q7Egs2QIUNQKBQP3G7z/m3mVOiPHj2KUqlk/PjxAIwZM4avvvqKgwcPMnTo\nUJNjzp07x9q1a/nxxx+NO5WIR9eVG5kcj7kFwKhePthYy6eoQlQXs2bN4pNPPinW68De/o8VJzt2\n7OCdd97h2LFjVR1epZMVNiULcPFlSusJLD/7FTeybvLv06t5uf2L2KpsSx9cChdHGzo3r8+x6Jvs\njkykd3t3lL/3yhNCPDytVsuuXbu4cuUKEyZM4Pz58/j5+RmbEJcmLCyMnTt3EhISwsqVK43Ho6Oj\nad68uckx2dnZTJ8+3Xgfzz33HE8//XSxDwdKm0cIIWq7Egs2e/furbA7io+Px8/Pr8gxHx8f4uLi\nHjjm/jaEjRo1Mvt+FAoFSrM78xRSKhVF/hWFKjovmw8W/l83crOnTwd3rB7RfMvvS3GSE9Mepbwc\nP36cGTNm8Omnn6JWq4vclpGRwbx589i5cyc9evQoda4dO3bw6aefcuPGDdzd3QkNDWXgwIHcuHGD\n+fPn89tvv6FSqXjiiSeYM2cOarUag8HA4sWL2bRpEzqdjlGjRvGPf/yjynYhlKbDpWtdtwUTWozj\nq+gNJGQmsvLsWqa3+3uF7Kr1eGdPjkXf5HpqNmcvp9LO37w3kkKIkiUmJjJx4kR0Oh0pKSk8+eST\nrF+/nmPHjvHll1/SsmXLUucYPXo0ISEhHD9+vMjxmJgY1Go1/fv3R6/XM2TIEEJDQ1Gr1cyZM6fI\nufv27SMgIKBYsaa0ecwhr/0rhuTENMmLaZIX0yojLyUWbDw8ii9LvnDhApcuXUKv1+Pr62vWEz0U\nVtrt7OyKHLO1tSU3N9fk+b/99huXLl1ixYoVZs1/n5ubxriLVVk5O8tSeFMqIi+nL9wmKv4OABOH\nt6Je3ToPPaelye9LcZIT0x6FvHz99ddMmTKF4OBg/v3vfxufrw8cOMA///lP8vLyWLBgAWPHji1x\nnvj4eN58801Wr15Nx44diYiIYOrUqRw6dIjXXnuNgIAADh06RGZmJjNmzOCzzz4jNDSU9evXc+DA\nAX744QcUCgXBwcGsXr2aF198sSoe/p9W2FT//ytL6tqwI9kFOWy6sJXYtIv89+IPBDV76qHn9XWv\nQ1NPZy4kprPz2FUp2AhRQRYuXEivXr2YN28enTp1AmDx4sW89dZbvP/++6xdu7bUOerXr2/yuIuL\nC926dSMoKIjU1FRmzZrF0qVLefXVV4uct2PHDpYvX/7A1/TmzvMg8tq/YklOTJO8mCZ5Ma0i82J2\n0+HMzExef/11Dhw4gJOTEzqdjnv37tGxY0eWL19u7G/wIHZ2dsWKM7m5uUWW2//Zli1bGDlyZJH+\nNuZITc0qV5Xd2VlDenoWer3py79qo4rKi95g4IsfzgKFL8qbezhy5869igqzysnvS3GSE9MeJi+u\nrg6VFJVpzZo145tvvmHy5Mm88MILfPLJJyxZsoQtW7bQr18/5s2bR4MGDUqdx8fHh/DwcDQaDQUF\nBaSkpKDRaFCr1djZ2TFt2jRsbGyoV68eI0aMYPfu3QBs3bqViRMnGt8YBAcHs2TJkios2MgKG3P1\nbdyT1Jw77Es8zKGkI7g7NOQxj8CHnndINy8uJKZzITGdy8kZ+LlXTGNjIWqz3377jW+//bbIyhaV\nSsW0adN46qmHK7aGhYUZv7e3tyc4OJjFixcXKbSsWLGC5cuXs3TpUrp27VrueUoir/0rhuTENMmL\naZIX0yrjtb/ZBZuFCxdy69Yttm/fbry06eLFi8yZM4cPP/yQBQsWlDje19eXdevWFTkWHx/P8OHD\nTZ6/f/9+li1bZm54RgaDAZ2uzMMA0OsN6HTyC/dXD5uXo9E3iL9+F4AxffzQ6wEe/TzL70txkhPT\nHpW8eHp6sn79el544QX69++Pk5MTH3/88QOfpx9Eo9GQmJjI4MGD0ev1zJs3DwcHh2Kfru7fv9/Y\ntyAuLg5/f3/jbT4+PsTHxxv7pJmjrMvi7y9XLTAUoNXnA1DHxgErq9q9vNec5bxjmg3nZvYtolLP\ns/HCVho5NKCZq98DzzdH+6Z18ainIel2FruOX2Xm6LYPNV9Fk+XfpkleiqtOOVGr1WRmZhY7fu3a\ntTJ/KPpnGRkZhIWFMWPGDBwcCt9k5OXlYWNjAxQ2sp87dy7h4eGsX7/+gT1qSpvHHPLav2JJTkyT\nvJgmeTGtIvNidsFm3759rFq1qkgfmoCAAObOnUtwcHCpBZvAwEC0Wi1r167lmWeeYevWraSkpNCr\nV69i5yYmJpKZmUnr1q3L8FBEdZRfoGfL771r2vq50dzbxcIRCSFK0qBBA9avX8+LL75IXl6eWT1r\nTGnUqBGnT58mMjKS6dOn4+3tTWBg4SoMg8HAwoULiYuL46OPPgIgJyenyJaudnZ26PV6tFqt2S/c\ny7ss3srujz+oHnXr4upctaubqqvSlvO+2nsqb+35kOS7N1l1di3vP/4G9R0e7lKmsQOa8n8bTvLb\n+dvk6sC9XvX7v5Dl36ZJXoqrDjkZOXIkCxYs4J133gEKCyRxcXG88847ZS7G/5mjoyO7d+/GYDDw\nyiuvkJycTFhYGOPGjQNg2bJlHDlyhI0bN5a4s2Bp8wghRG1ndsFGpVKZfNFsa2tLfn5+qePVajUr\nV65k3rx5LF68GG9vbz7//HPs7e2ZO3cugHE776SkJJycnMxuNiaqr30nrpGSkYtCAWP7+Zc+QAhh\nEb/88kuRn1944QUWLFjAhAkTeP3114s0/zVVaP+r+7tNBQYGMmjQIPbu3UtgYCC5ubm8/vrrnD9/\nnrVr1+Lm5gYU/i3Jy8szjs/JyXng350HKeuy+PvLVpNTbhuP6XIU3NE/updsVoSyLOcNbjORfx3/\nlLvaLD48FMZrXWZgbVX+XR3bNHHGxdGGtLt5bNgVy6Sh1WfnGFn+bZrkpbjqdDnsK6+8wuLFixk/\nfjxarZYxY8agUql49tlnmT17drnnVSqVhIWF8e6779K9e3dsbW0JCgpi4sSJFBQUsHr1agoKChg0\naFCRceHh4aSnpzNs2DC2b9+Ou7v7A+cRQghRhoJNjx49+PDDD1m8eDHOzs4A3Llzh48++sjsT2Cb\nN2/Ohg0bih2/X6i5r3v37oSHh5sbmqimsnLz2RZxBYDH2rrjUdfynzQJIUybMmWKyeMpKSkEBwcb\nf1YoFMTExDxwnoMHD/Lll1+yZs0a47H8/HwcHR1JT09nypQp2Nvb8+233xr/lgD4+fkRHx9Pu3bt\ngMJLZn19fcv0GMq7LP5uXpbxe1ulnSzt/Z05y3nr2tZlUqvx/Pv0F1y9m8Sm8z8+VBNiBQoe7+zJ\nxv2XOHzmOiN7+eCkqV4f3sjyb9MkL8VVh5yoVCpef/11Zs2axdWrV9HpdHh5eWFvb09qaqqxaG6O\nbt26cezYMePP/v7+RZ7r/3yfp06deuA89vb2nDx5stR5hBBClKFg88YbbzBx4kT69OlD48aNgcLr\nX/38/HjvvfcqLUDx6NoWcYWs3AJsrK148jEfS4cjhChBbGxshczTsmVLzp07x/fff8/IkSM5fPgw\nBw8eZOPGjcycOZO6devy6aefYm1ddBXGyJEj+eKLL+jevTsqlYrly5czatSoCompNPd+3yHKxkqN\nSmn2n0Xxu1ZuzXjCuz8/JezjUNIR/Jx96Nygfbnn69PenR8jrpCTV8DPv15lbF9ZnSlEebVo0YLw\n8HBcXV0JCAgwHk9MTGTkyJFFCidCCCGqH7NfmdarV48ffviBQ4cOERcXh42NDb6+vvTo0aPcW+mJ\nmut2eg57f7sGwOCunjg7mH9ZgxDi0VWvXj3CwsJ47733mD9/Pk2aNOGzzz4jIyOD48ePY2NjU2Sn\nkJYtW7J+/XrGjx9PSkoKY8aMIT8/nxEjRjB58uQqifne7ztEOciW3uU21OdxLmdc4WJ6HN/E/hdP\nB3caaExvBVwaOxsVAzp5sC0igX2/JfFEVy8c7avXKhshqrPNmzezadMmoHDl4YsvvlisSH779u0H\nbtcthBCi+ijTR4kqlYp+/foV6V9wv3+N9JsRf7blUBwFOgNOGjVPdPOydDhCiCrUuXNntmzZUuz4\n+fPnHzjGysqK0NBQQkNDKzM0k2RL74dnpbRicqvxvP/r/3FXe49V59bxeueZ5e5nM6iLF3sir5Gr\n1fHzr4mM7vNwO1AJUZsMGTKE69evA3Dq1Cm6du1abEcojUZTrL+MEEKI6sfsgs3x48d55513uHLl\nCvrCfZkBjFuultTTQNQu8dczORZ9E4BRj/lgq5ZLDIQQ1dc9beElURpZYfNQnGzq8PdW41l6ciXJ\nWTfYenknY5qOLNdcDnbWDOjUmO1HEtjz2zUGd/XCwa78zYyFqE3s7e156aWXAPDw8GDYsGHywaoQ\nQjyizH4n/fbbb+Pv78+cOXOKbL0qxJ8ZDAY27rsEQCM3ex5r28jCEQkhRMlkhU3FaerizyDvfuxK\n2Mf+a7/Qwq0ZrdyalWuuwV292PPbNfK0OnYdvyqrbIQw07fffstTTz2FWq1Gq9Xy3XffPfDcoKCg\nKoxMCCFEWZldsLl16xZhYWH4+EjzWPFgJy6kcD4xHYCxff2xKsseu0IIYQFZ+bLCpiIN83mc2DsX\nSbibyNqYb3mr62wc1WXfqtjBzpqBv6+y2SurbIQw2/Llyxk0aBBqtZrly5c/8DyFQiEFGyGEqObM\nLtg8/vjjHDx4UAo24oG0+To27L0IQAtvF9r5m79VpBBCWMofTYdlhU1FsFJaManVM7z/6xLuau+x\nLmYTIW0nlWuDgkFdPIv0snm6d9m2eheiNlq1ahXp6emkp6fzxRdfWDocIYQQD8Hsgs3s2bMZOXIk\n27Ztw9PTE+VfVk4sWrSowoMTj5YdRxNIzczFSqlg/ONNZfcwIcQjQVbYVLz69vUYGzCK9bGbOJca\nw+GkI/Ru3KPM8zjaqxnQqTE7jiawOzKRgZ0aU0cjvTiEKMnQoUNRKBTGPpOmSA9KIYR4NJhdsHnr\nrbdQKBQ0btxYetiIYm6l57Dj6FUABnZujEddeeMjhHg0GAs2KjsLR1KzBDbqTHRqLCdvn2XLpW0E\nuPjRSNOgzPM80c2L/SeTyMkr4MfwKzw3qGklRCtEzbF3715LhyCEEKKCmF2wiYyMZN26dbRp06Yy\n4xGPqG/3XqRAp8dJo2ZkT7lsTgjx6MjXFwCUewtqYZpCoeDZ5qOJz7xKel4GX0Z9w2udZ2KtLNvO\ngQ521gzt7sXmg3EcOJXEwM6NaeAql68J8SAeHh6WDkEIIUQFMbsjrLe3N1qttjJjEY+oM5dTOXkx\nBYCx/fyws5FtvIUQjwaDwUDB7wUblVIKNhVNY23PxJZBKFCQdO86P1zeWa55Hu/siYujDTq9gc2H\n4io4SiGEEEKI6snsd9bTpk3jjTfeYMKECXh5eaFSFR3aq1evCg9OVH95Wh3rfj4PgH9jJwJbNbRw\nREIIYb77xRoAa6WVBSOpuZq6+DPQqw+7rx5gX+JhWro1o4Vr2S5rUltb8WQvH77cGUtk7C0uJ2fg\n5+5USRELIYQQQlQPZhdsQkNDAXjvvfeK3SZNy2qvLYfiSMkobDQ8YVAzaTQshHik5P+pYCMrbCrP\ncN9BxKZdJPFuEl9HF2717aAuW6+znm0a8fOviSSlZLFp/2XmjO8gf3OEEEIIUaOZfUlUbGzsA7+k\nWFM7XU7KYE9kIgBDu3vjWd/BwhEJIUTZFOj+XLCRFTaVRaVUMbnls6iV1mRq77IudhMGg6FMcyiV\nCsb09QPgQmI6p36/FFcIIYQQoqYyu2AjxJ/lF+hZvSMGA+BeV8PwHk0sHZIQQpSZVp9v/F6lkP5b\nlamBpj5jAkYCcDYlmsNJR8s8R1s/N5p7OQPwn70X0ebrKjRGIYQQQojqRAo2oly2RVzhemo2CmDy\nkOZYq+RXSQjx6PnzChtruSSq0vVw70q7eq0B2HLpR65n3SzTeIVCwfjHm6JUKEjJyGXH0YTKCFMI\nIYQQolqQd9mizK7cyDS+SH68iyd+HtL4UQjxaNLq/rTCRi6JqnQKhYLxzUfjbONEvr6AL6O+KdJH\nyByN6znweJfGAOw4epWbadmVEaoQQgghhMVJwUaUSU5eAWFbo9DpDdR3tuOpx3wtHZIQQpRb0V2i\nZIVNVXCw1jz0Vt8je/rg7KCmQKfnm90Xy9wPRwghhBDiUSAFG1Em634+z620HKyUCoJHtcJGLZ9I\nCyEeXUV3iZLns6pyf6tvgH2Jh4lJvVCm8XY2KoL6BwBwNi5VGhALIYQQokaSgo0wW/jZ6xyJKuw3\nMLqPHz6N6lg4IiGEeDj5RS6JkqbDVWm47yC8HD0A+CpmA5nau2Ua37VFfWMD4vV7LpCdW7ZLq4QQ\nQgghqjsp2Aiz3LyTzbqfCz8Bbe3jyqCunhaOSAghHt79FTYKFFgpZIVNVVIpVUxqNR61lZq72nt8\nFbUBvUFv9niFQsHzg5qhslJwJzOPjfsvVmK0QogzZ87Qq1cv489nz56lRYsWdOjQwfgVFhYGgMFg\nYNGiRXTv3p0uXbrw7rvvotOZ3tUtIyODGTNm0KlTJ/r27cumTZuq5PEIIcSjQAo2olQ5eQUs23KW\nvHwddTRqXhjeEqVCYemwhBDioeX/vkuUSmmFQp7XqlwD+3o82+xpAGLTLrLryv4yjXevq+HJ33up\nHTp9nbNxqRUeoxC1ncFg4L///S9///vfyc//Y1ViTEwMvXv35uTJk8avkJAQANavX8+BAwf44Ycf\n2LFjBydOnGD16tUm53/77bext7cnIiKCpUuX8vHHH3Pq1KkqeWxCCFHdyfpvUSKd3sDn350jKSUL\npULB1BEtcdKoLR2WEEJUiHx94ZsPlTQctpiuDTtyMe0yEdd/ZXv8z/g5N6Gpi5/Z4wd39eTEhdvE\nJWeyZmcsC17oir2t/H8KUVHCwsLYuXMnISEhrFy50ng8Ojqa5s2bmxyzdetWJk6cSP369QEIDg5m\nyZIlvPjii0XOy8rKYs+ePezatQsbGxvatm3L8OHD+f7772nfvr1Z8SkUCpRl/AhaqVQU+VdITh5E\n8mKa5MW0ysiLFGxEib78MYrTlws/sRz/eAAtm7haOCIhhKg4f15hIyxnbNNRXMlMJDnrBl9GfcM/\nuv4PddSOZo21Uip5YVgL/nf1r6TdzWPD3kv8fViLSo5YiNpj9OjRhISEcPz48SLHY2JiUKvV9O/f\nH71ez5AhQwgNDUWtVhMXF4e/v7/xXB8fH+Lj4zEYDEVWMyYkJKBSqfD09Cxy7s8//2x2fG5umnKv\nkHR21pRrXE0mOTFN8mKa5MW0isyLFGzEAx04mcTWQ5cB6N/Rg/4dG1s4IiGEqFjGgo1C/hxaktpK\nzQutn+dfkUvJ1N7ly3Pf8FL7KViZWUhr5Kbh6d6+bNx/iV/OXqd9QF06Nq1XyVELUTvcXyXzVy4u\nLnTr1o2goCBSU1OZNWsWS5cu5dVXXyUnJwdbW1vjuXZ2duj1erRaLTY2Nsbj2dnZRc4DsLW1JTc3\n1+z4UlOzyrXCxtlZQ3p6Fnq9oWyDayjJiWmSF9MkL6Y9TF5cXR1MHpdXqMKkkxdu8/VP5wFo7evK\nswMDLByREEJUvPuXRFlbyZ9DS2uoqc+zzZ7mq+gNXEi/zPeXdzA6YITZ4wd18eTExdtcupbB6u0x\neNV3oK6zXSVGLETtdr/BMIC9vT3BwcEsXryYV199FVtbW/Ly8oy35+TkoFKpihRroLCQ8+fzAHJz\nc7G3tzc7DoPBwAP6GZdKrzeg08mbzT+TnJgmeTFN8mJaReZFmg6LYk5fSuHf359Dpzfg2cCR6U+1\nxqqsH10IIcQjQFbYVC9dG3akX+PCXWj2JR7m1xsnzR6rVCoIHtEKja2K7LwCPt8aRYHO/F2nhBDm\ny8jI4F//+hf37t0zHsvLyzMWZPz8/IiPjzfeFh8fj6+vb7F5vL29yc/PJzk5uci5f76cSgghajN5\nFy6KOBeXymffnUWnN9DQ1Z53Q3qgkeaNQoga6v623tbSdLjaeMp/GAHOhW/s1sf+l8S7yaWM+IOb\nky1ThrcEIP56Jhv3X6qUGIWo7RwdHdm9ezfLli0jPz+fhIQEwsLCePrpwl3fRo4cyRdffMGNGzdI\nSUlh+fLljBo1qtg8Dg4ODBgwgEWLFpGTk8OZM2fYtm0bI0aYv7pOCCFqsiot2ERHRzNmzBjat2/P\nqFGjHrhlX2RkJE899RQdOnRgxIgRHDlypCrDrLXOxafy6ZazFOgM1He2Y85zHXCtY1v6QCGEeEQV\nGHeJkqbD1YWV0ooXWj+Ps40T+fp8Vp79irvae6UP/F07/7oM6eYFwJ7Ia/x2/lZlhSpEraVUKgkL\nCyM2Npbu3bszfvx4nnjiCSZOnAjA+PHj6d+/P2PGjGHYsGF07NiRyZMnA5CcnEyHDh2Mq2oWLFhA\nQUEBffr04eWXX+a1116jXbt2FntsQghRnVRZwSYvL4+QkBCefvppfv31VyZMmMC0adPIysoqct7N\nmzeZNm0aISEhnDhxguDgYGbOnFmm5mOi7A6fSWbJpjPkF+ip62TLa89KsUYIUT47duxgyJAhdOjQ\ngWHDhrFnz54it6elpTFgwAAuXLhgPGYwGFi0aBHdu3enS5cuvPvuu+jK25SgDLTGXaLkkqjqxFHt\nwNQ2f0OlVJGam8byM1+h1eWbPf6p3r74ezgBsGp7DIm3zC/4CCFM69atG8eOHTP+7O/vz5o1a/jt\nt98IDw/n5ZdfNu7WZGVlRWhoKL/88gvHjh3jn//8J1ZWhYVxd3d3Tp48ibu7OwDOzs4sWbKE48eP\nc+DAAcaMGVP1D04IIaqpKnuFevToUZRKJePHjwdgzJgxfPXVVxw8eJChQ4caz9u6dSs9evRg8ODB\nAAwfPhwfHx+UZvZQUSgU5eoU/+d/axODwcB3h+LY+ssVANzr2vPKM+2p62RXq/NSEslLcZIT02pj\nXuLj43nzzTdZvXo1HTt2JCIigqlTp3Lo0CFcXV2JjIzk7bff5tq1a0XGrV+/ngMHDvBxCk8gAAAg\nAElEQVTDDz+gUCgIDg5m9erVvPjii5Uab4Hu/iVRUrCpbrzreDKhxTi+jPqG+MwE1sZ8y+RW41Eq\nSv8jr7JSEjKqFQu+iiQjS8uS/57m7b91xsnBptSxQgghhBDVRZW9Qo2Pj8fPz6/IMR8fH+Li4ooc\ni4qKokGDBsyYMYPIyEiaNGnCW2+9hVqtNut+3Nw0xup+WdW2feRztQV89t/THPit8I1TG7+6vDmp\nCw72RXNd2/JiLslLcZIT02pTXnx8fAgPD0ej0VBQUEBKSgoajQa1Wk1kZCSzZs3itddeY86cOUXG\nbd26lYkTJxq3jw0ODmbJkiWVXrDRGi+JkoJNddS5QXtScu7wY9xPnLh1hrp2bozyG2LWWNc6trw8\npi3/Wn+CO5l5LN18htfHd8TGWi5/E0IIIcSjocpeoWZnZ2NnV3R7TVtb22KXOmVkZHDo0CE+/fRT\n/u///o+NGzcydepUdu3ahZOTU6n3k5qaVa4VNrVtH/lrt+/x2ZZzJKcUXpLWo3VD/j6sBdpcLXdy\ntUDtzIs5JC/FSU5Me5i8uLo6VFJUlU+j0ZCYmMjgwYPR6/XMmzcPBwcHAgIC2Lt3L7a2tsUKNnFx\ncUV2BfHx8SE+Ph6DwWB2Eb6sKyyVSsUfK2ysrLGyqj0roUpS3VaGDfXtT2puKhHJv/Jzwn7q2rvQ\nu3GgWWP9GzsRPKoVn24+S/z1u6zeHsP0p1ujLMcHO9UtL9WF5KU4yYkQQoiKUmUFGzs7u2LFmdzc\nXOzt7YscU6vV9O7dm169Crf1fO655/jiiy84ceIE/fr1K/V+DAYD5W17UBv2kTcYDBw6ncx/9lxE\nW6BHoYAnH/NleKA3CoXC5OOvDXkpD8lLcZIT02pjXho1asTp06eJjIxk+vTpeHt7Exj44DfZOTk5\n2Nr+0TfLzs4OvV6PVqs1bhNbmvKssLy/S5TGzvaRLpJVhuq0MuylHn8j81Am526d5z8x31Hf2YUe\nXp3NGjuohwOZuTq+2h7Nr7G32HggjmlPt5XVuBVM8lKc5EQIIcTDqrKCja+vL+vWrStyLD4+nuHD\nhxc55uPjw9WrV4sc0+v1GAy1681OZbiTmcu6ny9w6lIKAC6ONgSPbEVTT2cLRyaEqGlUqsI/L4GB\ngQwaNIi9e/eWWLCxtbUlLy/P+HNOTg4qlcrsYg2UfYWlUqkg//dGtvp8A3fuSGNaqL4r5l5o+Ryf\n5Czn6t0klh79kv9v797joqrz/4G/ztyYGe4gqCh3FQQVxBvmrVDTSmUrL/2s/XY1qHbtZ5tb23fX\nNd3t2++x39q2NcV1y76a1X410zbtYqV0MU1MRVRQYVIEkcsAcpvrOb8/gIFpBgEVZmBez11jzmfm\nzHnPh/E9H9/z+ZxjbpIwOmRkl/a9LWkQLpTW4MCxUnxy8CdIVhH3zRzWraKNu/aLq7FfHHnq7Eoi\nIrr5eq1gM3nyZJhMJmzduhX33Xcfdu/ejcrKSttMmlbp6elYsmQJDhw4gOnTp2Pbtm0wGo2YNGlS\nb4Xa74iihP3HSrAjuxBGU/P0o6TYYDw6LwE+GqWLoyOi/iQ7OxubN2/G22+/bWszm83w9fW95n6x\nsbHQ6XS2S7nqdDrExMR069jXM8OydYaNXFB43CyozrjbzDCVTI0nkx7FX3/MwpXGcvwjdwueSnoU\nwwNjO98ZwAOz42AwWXHo1BV8evgiFHIZ7pnevfcY4H794i7YL47YJ0REdKN67bLeKpUKmzZtwp49\nezBx4kS888472LBhA7RaLVatWoVVq1YBABISErBhwwa89tprGDduHD788ENkZWXB25vTSq/HuUs1\n+PPWHGzbdxZGkxXeagUevWskli8cw2INEd10CQkJyMvLw65duyCKIrKzs5Gdne0wm/LnFixYgDff\nfBNlZWWorKzExo0bkZ6e3uPxts6wUQg86XBf4KvywfKxyxCsDoRZtGBD7macr9F1aV+ZTMCjd43E\nuLgQAMDHB3/CR9/qOIOXiIiI3FavjlDj4+Px/vvvO7SvWbPGbnvq1KkOM2+oe8qrG7H9QCGOFlTY\n2lITB+K+mcPhp+3aFbeIiLorJCQEWVlZeOmll7BmzRpERUXhjTfecLhK4M8tXboUlZWVWLhwIcxm\nM+bPn4+HH364x+NtnWHDy3r3HQFe/vh18uP464/rUWuqwxvH/4mMMQ8hPmh4p/vKZTJkLEjEup0n\nkVtYhV3f6lBvMOO+mcOv60TERERERD2JI9R+Rn/VgD2HLuDr46WwtqybHhrigyUzhyExKsjF0RGR\nJxg/fjx27tx5zccUFBTYbcvlcqxYsQIrVqzoydActF4lipf17ltCtMH4vymZeP3YJlQba7AhdzOW\njfolRg3o/Jw2CrkMT909Clm7T+HYuUp8kXMJ9Y1mPHLXSCjkvTbxmIiIiKhTHJn0E/qrBmz9vADP\nb/we+38sgVWU4O+jwsN3xmP1wxNYrCEicsIkNi+J4gybvidUG4IVKZkYoA6CRbTgHye34OiVE13a\nV6mQ48m7R2HamMEAgEOnr+D1HbloMlp6MmQiIiKibuEItY8rLq/HZz9cxOHTV2wzanw0StwxKQJp\nKUPhpZK7OEIiIvfFGTZ9W7AmCCvGPYHXj/0DVxor8NapbdAbqjErYkanV4CSy2R46I54+HmrsOf7\nC8jT6fGnLTn41T2jMTiY580jIiIi1+MItQ+SJAmnL1Tjs8MXkafT29pbCzW3pQyBWsVfLRFRZ1pn\n2LBg03cFePljRcoTyMp9Gz9dvYhdhXtR0VSFJSN+Abns2l9aCIKAe2fEItDXC+/uO4fLVY3405Yc\nLJuXiOThA3rpFRARERE5xxFqH2KxijiSX47PDl/ExfJ6W3togAa3TwzHlFGDOaOGiKgbOMOmf/BV\n+eDpsY/jf06/j+MVefiu9DD0hmo8lPh/4KPsfLZMWspQDBngjfW78lDXaMbrH+Ri3i2RWDAlmue1\nISIiIpfhCLUPaDJakH28FPtyilFdZ7S1x4T5Ye7ECKSMCIFMxqtbEBF1V+tVoliw6ftUchUeHfUA\ndp3fiy+Lv8YZ/Vn8vyOv47FRDyDSL7zT/eMiAvHHhyZg3c6T+KmsDh8fvIC8Ij2WzU/gEikiIiJy\nCY5Q3Vh1nRH7coqRfbwETUYrAEAAkDx8AOZOisCwIf6drtEnIqKOma086XB/IhNkuGf4PAzyDsW/\nzu6C3lCNV4+ux8IR6ZgaNqnTz8wgPzV+90AK/nd/Ib48egk/ldXhxc1HsDhtGGaOH9pLr4KIiIio\nGUeobqiksgGfHr6AQ6faTiSskMswdfQgzJ4Qzm/6iIhuAlESYZVEAJxh09/cEjYRQ33D8M+T76DK\noMf7BTtRUH0e9424Gz6qa3+GKhVy3D97BJJig/Hm3jOorTfhnc/P4tCpK/j1krEI0HDpMREREfUO\njlDdyNniGnx6+CKOn6+0tflolEhLGYK0lKHw81a5MDoiov7FIrZdwlkh8OOwv4nwHYrnJyzH/5z+\nF/KqzuBYeS7O1xTh/viFGD0godP9R8UEY+2jk7D1swIcyS/H+ZJarPjrAcwcPxTpU2KgVfM9Q0RE\nRD2Low0XEyUJJ85V4pPDF3G+pNbWHuynxpyJ4Zg2JownEiYi6gHmdgUbpZwfh/2RVqlF5piH8E3J\n9/jw/B7UmeqRlfs2UgePx92xd3U628ZHo8QTvxiFqUVV2LbvLMqrm7DvyCUcPFmGuyZHIS1lCFRK\nfkYTERFRz+AI1UUkScKPZyux+1sdLlW0XfEpPNQHd6RGYEJ8KOQyXpmCiKincIaNZxAEAdOH3oL4\noBHYeuZfKKq9gEOXc3Cy4jQWxM7FLWETIROu/Xk7OiYYf358EvYfL8P2L8+iwWDB/+4/j305xZg/\nJQpTRg2GUsHPbCIiIrq5OELtZZIk4fi55kJN+0tzj4wMxB2pEUiMCuKJhImIeoHdDBuew6bfC9UO\nwIqUJ7C/+Fvs0X2OBksj3ivYiYOXj2Dh8PmI8Y+65v4qhRz/5/Y4TBgRjF3f6PDNicuorjNiy6cF\n2P2tDrePD8eM5CFcKkX9Vm5uLp588kl8++23du2iKOLBBx/EqFGj8NxzzwEA7rrrLpSWltoeY7FY\nYDKZ8PXXX2PgwIF2+588eRKLFy+GWq22tWVkZCAzM7MHXw0RUd/AUUUvyiuqwgdfF+FCWZ2tLSEq\nEL+YGoNhQ/1dGBkRkeexm2HDgo1HkAkyzIyYjnEDk7Dz3Mc4Wn4CF64W45Wj6zF6wEjMj5mLIT6D\nr/kcQX5qPDg3HnMmRmDXN0U4cqYctfUmbD9QiH8f/AlTRw/GjOQwDAnx6aVXRdSzJEnCBx98gJdf\nfhlyueMSwLfeegs5OTkYNWqUrW3Pnj2226Io4qGHHsLYsWMdijUAcObMGUyfPh0bN27smRdARNSH\ncYTaCy5V1ON/vzqPPJ3e1jYyMhDpU6MxIjzAhZEREXkuFmw8V4CXPx4ZdT9u0U/EzvMfo6T+Mk5W\nnkFeZT7GDUzC7ZG3dVq4GRSkRWb6KNw9vRGf/1CMb09ehsFkxRdHL+GLo5cwbIg/pieFYVxcCDRe\nfH9R35WVlYVPPvkEmZmZ2LRpk919+fn52LlzJ2bPnt3h/lu2bEF9fT2WL1/u9P7Tp08jPj7+psZM\nRNRfcATRg2rrjfjwGx2+yS2F1Hx1bgwb4o97Z8QgLiLQtcEREXk4Lomi+KDheH7C0zh65QQ+LvoM\nlQY9cq4cR86V40gIisPsyFsxPCDmmkuVBwZq8cs5cUifGo0Dx0vwzYlSVF014nxJLc6X1GLLZwVI\nig3GxISBGB0TBLWK7zXqW+69915kZmbihx9+sGs3mUx47rnnsHbtWmzfvt3pvrW1tVi3bh3efPNN\np7NzgOYZNiqVCmlpaRBFEXfccQdWrFgBlaprV0cVBAHdPe2jTCbY/ST2SUfYL86xX5zriX7hqKEH\nWKwiPvvhIj7+/gKMJisAICRAjUW3DsO4uBCeo4aIyA1whg0BzcukJgwai7Gho/H95Rx8ceEAKg16\nnNYX4LS+AGHegzBtSCpSh4wD0PEyJz9vFRZMica8yVE49ZMeB46VILewChariKNnK3D0bAUUcgFx\n4QEYHTsAY2KDMTBQwzEBub3Q0FCn7a+88gqmTp2KcePGdViweffdd5GUlISkpKQOnz8wMBCTJk3C\nkiVLUFVVhaeffhqvv/46nn322S7FFxzsfd1/jwICrn2lOE/EPnGO/eIc+8W5m9kvHKHeZOcu1WDL\npwUoqWwAAGi9FJh3SxRmjhvKK0gQEbkRMws21I5CpsC0IamYEjYRx8pPYt/FAyiuK0FpQxn+dXYX\ndhXuxS0R45EUNBqxftEdXllKJhMwOiYYo2OC0WAw48eCChw+cwVnLlTDYpVw6qdqnPqpGu9/eQ6h\nARokRAVi2FB/DBsagBB/NQs41Cd8//33OHToUIeFmlY7d+60nYi4I1lZWbbbWq0WGRkZePXVV7tc\nsKmqariuGTYBAd6oqWmAKErd27mfYp84x35xjv3i3I30S1CQ8y+FOEK9SRoMZuw4UIjs481nxBcA\n3Dp2CO6eHgMfjdK1wRERkYPWGTYyQdbpZZ3Jc8gEGcYNTEJK6BgU1v6Eb0sO41j5CRitJuzXHcR+\n3UH4q/wwbmASkkJGIdovAnKZ86Ue3molpiWFYVpSGOqbzMgrqkJuURXyivSobzKjvKYJ5cebcKBl\n7ODvo8LwIf6ICfNHxEAfRAz05RiC3NLevXtx8eJF3HLLLQAAg8EAQRBQVFRkO3lwYWEhKisrMX36\n9A6fp7a2FllZWXjqqafg49P8jxWj0QgvL68uxyJJEqzW63sdoijBauU/NttjnzjHfnGO/eLczewX\nFmxukCRJOJJfjne/OIerDSYAwNAQbzw4Nx6xQ3jlJyIid9U6w4bnryFnBEHAsIBoDAuIxsLh83Gk\n/CiOlp+ArqYYtaar+Kr4G3xV/A28FVokBMdh1ICRSAiKg1apcfp8PholUhMHITVxEERRgu7yVZws\nqsLZ4hoUlV6FySKitt6EnIIK5BRU2PYL9PVCeKgPwkN9MChIi0FBWgwM0rKQQy61du1arF271rb9\n/PPPIzAw0G42zfHjx5GYmHjNc9H4+vpi3759kCQJv/nNb1BaWoqsrCwsXry4R+MnIuorOEq9AfVN\nZmz5rAA5+eUAAJVChvSp0Zg9IRwKOb+tJSJyZ60zbLgcijrjo/LGrMgZWDz2Lpy6WIjDl4/hWHku\nrjRWoMHSiCNXjuHIlWOQCTJE+A61FXpi/aOgVWodnk8mExA7xN/2xY7FKqK4vB7nLtXi/KUa/FRW\nh8paAwCgus6I6jojcgur7GPSKDEwSINgPzWC/dQI8lMjyM/LdttbreDyKnKpkpIShISEOLSXlpbi\nrrvuwp49exAWFoasrCz86U9/QmpqKtRqNZYsWYIHH3zQBRETEbkfQZKkfjWHqaKirtv7yOUCgoJ8\noNfXd3nq0smiKry19wxq65tn1SRGBeKXc+MRGuD8m7W+6Hr6xROwXxyxT5y7kX4JCfHtoaj6r+7m\n/0NlR7D19Hb4e/nhpSm/76Go+h7+fXbOWb+UN1Yir+oM8irP4FxNEURJtNtHgIAwn0GI9otAuO8Q\nhPsOQZj3ICjlnc+OaTSYUVxej4vl9Si+Uo+SynqU6ZvQZLR0ui/Q/CWSn7cKvloV/LRK+GpV8PVW\nwk+rgq9WCa2XElq1AhovBTRecmi9lFB7ySHrZpGH7xdHzP29q7fG/v0d+8Q59otz7BfneiL/82vF\nbrJYRew4UIjPjxQDaB4QLU4bhtvGDuE3WUREfQiXRNGNCtUOQJp2GtLCp6HJ0oSC6kKcrynC+Rod\nLtWVQoKEkvrLKKm/bNtHJsgw2HsghvgMxiBtKAZ6h2KQNgQDNMF2s720aiXiIgIRFxFoa5MkCXWN\nZpTpG3FF34gr1U3Q1xmgrzVA3zITx9pykkOTRURlrcE2U6crBABqLwW0XnKovRRQKeTwUsqgUsqh\nUsrhpWi9LYNXS5taJYe/nwYGgwkyQYBCLoNCJkAul0Ehb96WywUoZD/blssgEwQIQvOMI5kgOGwL\nAji2IiIij8ZRajfU1BuxYVcezl2qBQBED/bFY/MSMDiYlzMjIupruCSKbiaNQoPkkFFIDhkFAGiy\nNKGo9gLO1+hQXFeCi3WX0GBuhCiJDkUcoLmQE6wORLA6CEHqwJY/AbbbAV5+kMvk8PNWwc9bhRHh\nAQ4xiKKE2gYT9HUGVF81oq7RhKuNZlxtNKGuofl2XaMJdY1mNBkttuJOKwlAk9HSMovH2FNd1S2C\ngJbijQCZrN1tJ4Wd1tqO0PJf27atXUDL/wFBgNDuvuYmoeW+1udoe5629rY72/Zv95iWOwYEaLEk\nLRYB3l0/eS4REdHPcZTaRQUXq7Fh9ynbiYVvnxCOhbfG8lw1RER9lIUzbKgHaRQaJAbHIzE4HkDz\n7JgaYy0u1pWguK4Elxuu4EpjOcobK2GVrBAlERVNVahoqnL6fAIE+Ci94avygZ/KF74qH9ttH5UP\ntAoNtAo1tEotAoPUGBLqBy+5V4czVCRJgsksotFoQWNLkabRYLEVbJpMFpjMIkxmK0xmEUaL1Xbb\nZGlpM1thslghSYDJbIXFKsFiFWGxSrBaRdyMSfKSBFglCYAEXOeVgFyhsOQqxsQEYXLiIFeHQkRE\nfRhHqZ2QJAmfHynG9v2FECUJXio5HrlzJCbEh7o6NCIiugFmzrChXiQIAgLVAQhUByApJNHWbhWt\nqDLocaWxAuWNldAbqqE31LT8rEajpQkAIEFCnbkedeZ6lDaUdemYMkEGjUINrUIDtdwLKrkKKrkK\nXi0/VXIVvGTt2rxUUGmU8JYp4CeTQylTQC7IoZApoJCpoBAUkMtatlvaVQoFQoP9UVtrgGRtPqYg\nCJAkCaIk2Yo3lpZLnDYXdJqLOqIoQYIEUQREqWVbkiBKzduSKLW0t2y33mdrlyBJbfc191Pzf5p/\ntrW1nrFRkqS2QlK7x7W22R5nu7N1f8l2f9v+kv3xbL9rYHCoL1Jig7r0eyIiIuoIR6nX0GS0YPMn\n+barQA0O1uKpu0cjbACXQBER9XVcEkXuQC6TI1QbglCt49V0AKDJYoDeUI0aYy2umupRZ6pDnake\nV011tu16UwOaLE2wSPZTUERJRIO5EQ3mxt54KTYCBMiE1nPUyCCDrOUcNc3FnObttrbmdhlk7fZr\nfhbBtgzJfpmT/Xa7rbalTq37Cu3vc3wuATK7JVJtz/bzF2Xf+vPHtN9LEACZOggmaQY0cLxKGBER\nUVdxlNqB0soGvPHhSVyuah7kjI8PxcN3xEPjxS4jIuoPeNJh6gs0CjWG+AzGEJ/B13ycJEkwixY0\nWhrRZDGg0dyEJksTGi1NaDQ3wWA1wmQ1wSSaYLKaYLSaYLKa226LzT/NVhMsohUWyQKLaIVV6v46\nJAkSrJIVHn3hkCtAiDIEkwaNd3UkRETUh/XqKPX06dNYtWoVzp8/j8jISLz44otITk52eFxGRga+\n//57yOVyW9uxY8d6Lc6c/HK8ufcMjCYrZIKAxbfFYvaEcF6pgIioC/bu3Yu///3vKCsrQ1hYGFas\nWIFZs2ahtrYWL7zwAg4dOgRfX1889dRTWLRoEYDmf2y++uqr2L59O6xWK9LT0/G73/3O7nPgZuMM\nG+pPBEGASq6ESu6PAC//m/a8kiTBIllhFS32hRzRAotkhQQrtL4q6GvqYLGKEKXmPxKkltvNPyVJ\nhFUSIUKCJIlt90FsWeoktvvTfM6a5v+h3ZIl+21bq9R2b+tSJrs9JLQ9F6SWZU/tnkP6+X12PeDk\nVuu25OxhEGRAiF8QkkITQUREdCN6bZRqNBqRmZmJzMxMLFq0CLt378YTTzyBL774At7e9kuMTp8+\njW3btmH06NG9FR4AwGCy4N1955B9vBQA4O+tQmZ6ot0lNYmIqGM6nQ4vvPAC3nrrLaSkpODgwYN4\n/PHH8fXXX2P16tXQarU4ePAgCgoKsGzZMgwfPhzJycnYtm0bDhw4gI8++giCICAjIwNvvfUWli1b\n1mOxsmBD1DlBEKAUFB3ORJPLBQQF+UAvq4fVo6fUtLH1iZ59QkREN6bXLnF06NAhyGQyLF26FEql\nEgsXLsSAAQOQnZ1t97iqqiro9XqMGDGit0KD/qoB/9ydh9+u/95WrBk+1B9/fHgCizVERN0QHR2N\n7777DikpKbBYLKisrIS3tzdUKhW++OILLF++HF5eXhgzZgzmzZuHXbt2AQB2796NBx98EKGhoQgJ\nCUFGRgY+/PDDHo2VS6KIiIiIyJ312ihVp9MhNjbWri06OhpFRUV2badPn4a3tzcyMjKQn5+PqKgo\nPPfccxg7dmyXjiMIAmTdLENt2JWHs8W1AAC5TMDd02Nw5+QIyLv7RP2MTCbY/aRm7BdH7BPnPLVf\nvL29UVxcjDlz5kAURaxevRoXL16EQqFAeHi47XHR0dH4/PPPAQBFRUUYNmyY3X06nQ6SJHV5OWp3\n83/rcgYvuQpyuWf9jq7FU9+3nWG/OMd+ccQ+ISKim6XXCjaNjY3QaDR2bWq1GgaDwa7NaDQiOTkZ\nK1euRGRkJHbs2IFly5bhk08+QUiI8ysotBcc7N3tc81MTR4KQZAhOS4EaePCERbi0639+7uAAF4V\nyxn2iyP2iXOe2C+DBw/GiRMnkJOTgyeffBKPPvoo1Gq13WPafwY0NTXZ3a/RaCCKIkwmE7y8vLp0\nzO7m/zlx03DVfBW3x01DUBDz/s954vu2K9gvzrFfHLFPiIjoRvVawUaj0TgUZwwGA7Ra+8sdzpo1\nC7NmzbJtL126FO+99x4OHz6MefPmdXqcqqqGbs+wuS15MO6+dRhqahogihL0+vruPUE/JZMJCAjw\ntvULNWO/OGKfOHcj/dLXCwgKRfPHy+TJk3H77bcjLy8PRqPR7jHtPwPUarXd/U1NTVAoFF0u1gDd\nz/+R6kj8adZK1NQ0MO+3w7/PzrFfnGO/OPLk3E9ERDdXrxVsYmJi8M4779i16XQ6hyLMp59+ClEU\nceedd9rajEZjlwftkiTB2v0rUAIARFHiyeGcYL84x35xxD5xzpP6JTs7G5s3b8bbb79tazObzYiI\niMDXX3+N0tJShIWFAWj+DGhdBhUbGwudToekpCTbfTExMd069vXmf0/6/XQH+8U59otz7BdH7BMi\nIrpRvXaSlsmTJ8NkMmHr1q0wm83YsWMHKisrMXXqVLvHNTY24s9//jPOnz8Ps9mMf/7znzAYDJgy\nZUpvhUpERNcpISEBeXl52LVrF0RRRHZ2NrKzs7FkyRLMnDkTr7zyCpqampCbm4uPP/4Y8+fPBwAs\nWLAAb775JsrKylBZWYmNGzciPT3dxa+GiIiIiMh1em2GjUqlwqZNm7B69Wq8+uqriIyMxIYNG6DV\narFq1SoAwJo1a3DPPfegoqICjz32GGpqapCQkIBNmzY5LJ0iIiL3ExISgqysLLz00ktYs2YNoqKi\n8MYbbyA2NhZr167FH//4R8yYMQNarRYrV660zahZunQpKisrsXDhQpjNZsyfPx8PP/ywi18NERER\nEZHrCJIk9au5mhUVdd3eRy4XEBTkA72+nlNX22G/OMd+ccQ+ce5G+iUkxLeHouq/upv/+b51jv3i\nHPvFOfaLI+b+3sWx/83BPnGO/eIc+8W5nsj/nn3daiIiIiIiIiIiN8SCDRERERERERGRm2HBhoiI\niIiIiIjIzfS7c9gQEREREREREfV1nGFDRERERERERORmWLAhIiIiIiIiInIzLNgQEREREREREbkZ\nFmyIiIiIiIiIiNwMCzZERERERERERG6GBRsiIiIiIiIiIjfDgg0RERERERERkZthwYaIiIiIiIiI\nyM2wYENERERERERE5GY8vmBz+vRpLFy4EMnJyUhPT8fx48ddHZJbyMnJwaJFixPSbP0AAAoUSURB\nVDBu3DjMmjUL77//vqtDchuVlZWYPHky9u/f7+pQ3EJZWRkyMjKQkpKC6dOnY8uWLa4OyS38+OOP\nuOeee5CSkoI5c+bg3//+t6tDonaY+51j7u8Yc7895n7nmPvdH/O/I+b+a2P+t8f876hHc7/kwQwG\ngzRt2jRp27ZtkslkkrZv3y6lpqZK9fX1rg7NpWpqaqQJEyZIH330kWS1WqW8vDxpwoQJ0nfffefq\n0NzC448/LsXHx0tfffWVq0NxOVEUpbvvvlt6+eWXJZPJJJ09e1aaMGGCdPToUVeH5lIWi0VKTU2V\nPvnkE0mSJOnIkSNSQkKCVFxc7OLISJKY+zvC3H9tzP1tmPudY+53f8z/jpj7O8f834b531FP536P\nnmFz6NAhyGQyLF26FEqlEgsXLsSAAQOQnZ3t6tBcqrS0FDNmzMD8+fMhk8mQmJiISZMm4ccff3R1\naC733nvvQaPRYPDgwa4OxS2cOHEC5eXlePbZZ6FUKjF8+HC8//77iI6OdnVoLnX16lXo9XpYrVZI\nkgRBEKBUKiGXy10dGoG5vyPM/R1j7rfH3O8cc7/7Y/53xNx/bcz/9pj/HfV07vfogo1Op0NsbKxd\nW3R0NIqKilwUkXsYOXIk/vKXv9i2a2trkZOTg/j4eBdG5Xo6nQ6bN2/G6tWrXR2K2zh16hSGDx+O\nv/zlL5gyZQrmzJmDEydOIDAw0NWhuVRgYCCWLl2KZ555BomJibj//vvxhz/8gR/2boK53znmfueY\n+x0x9zvH3O/+mP8dMfd3jPnfEfO/o57O/Yqb8ix9VGNjIzQajV2bWq2GwWBwUUTup66uDpmZmUhM\nTERaWpqrw3EZi8WC3/72t/jP//xPBAQEuDoct1FbW4vDhw8jNTUV+/fvR15eHh577DGEh4dj/Pjx\nrg7PZURRhFqtxt/+9jekpaXh4MGD+M1vfoPExEQOgNwAc3/nmPubMfc7x9zvHHO/+2P+vzbm/jbM\n/84x/zvq6dzv0TNsNBqNQ4I2GAzQarUuisi9FBcX47777oO/vz/WrVsHmcxz3y7r16/HyJEjMWPG\nDFeH4lZUKhX8/f2RkZEBlUplO9HWl19+6erQXOrzzz9Hbm4u5s6dC5VKhVtvvRW33nordu3a5erQ\nCMz9nWHub8Pc7xxzv3PM/e6P+b9jzP32mP+dY/531NO536P/JsbExECn09m16XQ6DBs2zEURuY9T\np05h8eLFmDp1KtavXw+1Wu3qkFxq79692LNnD8aPH4/x48ejtLQUzzzzDP7xj3+4OjSXio6OhtVq\nhdVqtbW1rt/0ZJcvX4bJZLJrUygUPI+Bm2Du7xhzvz3mfueY+51j7nd/zP/OMfc7Yv53jvnfUY/n\n/pty6uI+ymg0SlOnTpW2bNlid6b4hoYGV4fmUhUVFVJqaqq0ceNGV4fitm677TaeKV6SpKamJmna\ntGnSa6+9JpnNZuno0aNScnKydOzYMVeH5lL5+flSYmKitGPHDkkURenw4cPS2LFjpdzcXFeHRhJz\nf0eY+zvH3N+Mud855n73x/zviLm/a5j/mzH/O+rp3C9IkgeXwwDk5+dj9erVKCgoQGRkJFavXo3k\n5GRXh+VSWVlZ+Otf/+owPfQ//uM/sGLFChdF5V7S0tLwhz/8AbfddpurQ3G5CxcuYM2aNTh58iR8\nfHzw1FNP4d5773V1WC731Vdf4W9/+xuKi4sRFhaGp59+GrNnz3Z1WNSCud8Rc3/nmPvbMPc7x9zv\n/pj/7TH3dw3zfxvmf0c9mfs9vmBDRERERERERORuPPocNkRERERERERE7ogFGyIiIiIiIiIiN8OC\nDRERERERERGRm2HBhoiIiIiIiIjIzbBgQ0RERERERETkZliwISIiIiIiIiJyMyzYUJ/y/PPPIy4u\nrsM/f//733H48GHExcXBaDT2eDyXLl2yHXv58uXXvX9hYWGnj3322Wdtx+rK44mI+gvmfuZ+IvJM\nzP/M/55OkCRJcnUQRF1VV1cHg8EAADh27Bh+/etf46uvvoJKpQIAaLVaKJVK1NbWIiQkpMfjuXTp\nEmbOnIlt27ZhxIgR8PPz69b+VqsVer0eQUFBkMvl13xsXV0ddDodFi1ahL179yI2NvZGQici6jOY\n+5n7icgzMf8z/3s6hasDIOoOX19f+Pr6AgD8/f0BAAMGDICXl5fd43ojYbcXGBjY7YQNAHK5vMux\n+vr6IigoqNvHICLq65j7mfuJyDMx/zP/ezouiaJ+5+fTIuPi4rB3717Mnz8fY8aMwSOPPILLly9j\n5cqVSE5Oxu23345Dhw7Z9r9y5QqWL1+OsWPHYtq0aVi9ejUaGhq6dOzWaY4HDhzA7NmzkZSUhBUr\nVqC0tBSPP/44kpKSkJ6ejvz8fLvHt05zTEtLw9atW/HAAw9g9OjRmDNnDrKzs29yDxER9T/M/URE\nnon5n/ozFmzII7zyyitYtWoV3nnnHZw6dQoLFizAyJEj8cEHH2DYsGF48cUXAQCSJOFXv/oVlEol\ntm/fjnXr1iE/Px8vvPBCt473+uuv47XXXsP69euxb98+3Hvvvbjzzjuxfft2qNVq/Pd///c19126\ndCn27NmDuLg4vPDCCzCbzTf0+omIPBFzPxGRZ2L+p/6CS6LII/zyl7/EhAkTAACpqakoLi7GI488\nAgBYsmQJMjMzYbVa8cMPP0Cn0+Hdd9+FUqkEAPzXf/0X5s6di7KyMgwaNKhLx3viiSeQmJgIAIiP\nj0dYWBh+8YtfAADS09OxadOmDvedP38+7rzzTgDAk08+ifT0dJSVlSE8PPz6XjwRkYdi7ici8kzM\n/9RfsGBDHiEiIsJ2W6PR2CVAtVoNURRhsVhQWFiI+vp6TJw40eE5dDpdl5P2z483dOhQu+OZTKYO\n942KirLd9vHxAQBW2YmIrgNzPxGRZ2L+p/6CBRvyCAqF/VtdJnO+GtBisSAiIsJpFbw7JzPr6vGc\naa3ut8eLuRERdR9zPxGRZ2L+p/6C57Ahaic2NhZlZWXw9fVFZGQkIiMjYbFY8PLLL6O+vt7V4RER\nUQ9g7ici8kzM/+TuWLAhamfKlCmIjY3FM888g1OnTiEvLw8rV65EdXU1QkNDXR0eERH1AOZ+IiLP\nxPxP7o4FG6J2ZDIZ1q9fDx8fHzzwwAN45JFHEBUVhXXr1rk6NCIi6iHM/UREnon5n9ydIHGBHNF1\nu3TpEmbOnIm9e/ciNja23x2PiIgcMfcTEXkm5n/qbZxhQ3QTVFdX4+rVqz16jLq6Ouj1+h49BhER\ndR1zPxGRZ2L+p97Cgg3RTXD//ffj97//fY8e48UXX8SiRYt69BhERNR1zP1ERJ6J+Z96C5dEERER\nERERERG5Gc6wISIiIiIiIiJyMyzYEBERERERERG5GRZsiIiIiIiIiIjcDAs2RERERERERERuhgUb\nIiIiIiIiIiI38/8BIsntY+5UdXEAAAAASUVORK5CYII=\n",
"text/plain": [
""
]
},
"metadata": {},
"output_type": "display_data"
}
],
"source": [
"# set initial conditions and cooling flow\n",
"IC = [C0,T0,Tcf]\n",
"\n",
"# do simulation at fixed time steps dt\n",
"dt = 0.05\n",
"ti = 0.0\n",
"tf = 8.0\n",
"\n",
"# create python list to log results\n",
"log = []\n",
"\n",
"# start simulation\n",
"c,T,Tc = IC\n",
"qc = 153.8\n",
"for t in np.linspace(ti,tf,int((tf-ti)/dt)+1):\n",
" log.append([t,c,T,Tc,qc]) # log data for later plotting\n",
" c,T,Tc = odeint(deriv,[c,T,Tc],[t,t+dt])[-1] # start at t, find state at t + dt\n",
"\n",
"def qplot(log):\n",
" log = np.asarray(log).T\n",
" plt.figure(figsize=(16,4))\n",
" plt.subplot(1,3,1)\n",
" plt.plot(log[0],log[1])\n",
" plt.title('Concentration')\n",
" plt.ylabel('moles/liter')\n",
" plt.xlabel('Time [min]')\n",
"\n",
" plt.subplot(1,3,2)\n",
" plt.plot(log[0],log[2],log[0],log[3])\n",
" if 'Tsp' in globals():\n",
" plt.plot(plt.xlim(),[Tsp,Tsp],'r:')\n",
" plt.title('Temperature')\n",
" plt.ylabel('Kelvin')\n",
" plt.xlabel('Time [min]')\n",
" plt.legend(['Reactor','Cooling Jacket'])\n",
"\n",
" plt.subplot(1,3,3)\n",
" plt.plot(log[0],log[4])\n",
" plt.title('Cooling Water Flowrate')\n",
" plt.ylabel('liters/min')\n",
" plt.xlabel('Time [min]')\n",
" plt.tight_layout()\n",
"\n",
"SS = log[-1]\n",
"qplot(log)\n"
]
},
{
"cell_type": "markdown",
"metadata": {},
"source": [
"## Simulation 3. Proportional-Integral-Derivative Control\n",
"\n",
"Proportional-Integral-Derivative (PID) control is the workhorse of the process control industry. In standard form, the PID algorithm would be written\n",
"\n",
"$$q_c(t) = \\bar{q}_c - K_c\\left[(T_{sp}-T) + \\frac{1}{\\tau_I}\\int_0^t (T_{sp}-T)dt' + \\tau_D\\frac{d(T_{sp}-T)}{dt} \\right]$$\n",
"\n",
"For the control reactor temperature, note the controller is 'direct-acting' such that a positive excursion of the reactor temperature $T$ above the setpoint $T_{sp}$ is compensated by an increase in coolant flow, and vice-versa. Thus a negative sign appears before the term $K_c$ contrary to the usual textbook convention for negative feedback control.\n",
"\n",
"The practical implementation of PID control is generally facilitated by a number of modifications."
]
},
{
"cell_type": "markdown",
"metadata": {},
"source": [
"### Independent Parameters\n",
"\n",
"A common practice is to introduce an independent parameterization for each fo the P, I, and D terms. Rewriting, the control equation becomes\n",
"\n",
"$$q_c(t) = \\bar{q}_c - \\left[k_P(T_{sp}-T) + k_I\\int_0^t (T_{sp}-T)dt' + k_D\\frac{d(T_{sp}-T)}{dt} \\right]$$\n",
"\n",
"where\n",
"\n",
"\\begin{align*}\n",
"k_P & = K_c \\\\\n",
"k_I & = \\frac{K_c}{\\tau_I} \\\\\n",
"k_D & = K_c\\tau_D\n",
"\\end{align*}"
]
},
{
"cell_type": "markdown",
"metadata": {},
"source": [
"### Setpoint Weighting\n",
"\n",
"Step changes in setpoint $T_{sp}$ can produce undesired 'kicks' and 'bumps' if PID control is implemented directly using in standard form. It is common practice to introduce setpoint weighting factors for the proportional and derivative terms. This can be written as \n",
"\n",
"$$q_c(t) = \\bar{q}_c - \\left[k_Pe_P(t) + k_I\\int_0^t e_I(t')dt' + k_D\\frac{e_D(t)}{dt} \\right]$$\n",
"\n",
"where\n",
"\n",
"\\begin{align*}\n",
"e_P(t) & = \\beta T_{sp}(t) - T(t) \\\\\n",
"e_I(t) & = T_{sp}(t) - T(t) \\\\\n",
"e_D(t) & = \\gamma T_{sp}(t) - T(t)\n",
"\\end{align*}\n",
"\n",
"Common practice is to set $\\gamma = 0$ which eliminates derivative action based on change in the setpoint. This feature is sometimes called 'derivative on output'. This almost always a good idea in process control since it avoids the 'derivative kick' associated with a change in setpoint.\n",
"\n",
"In practice, the term $\\beta$ is generally tuned to meet the specific application requirements. In this case, where setpoint tracking is not a high priority, setting $\\beta = 0$ is a reasonable starting point."
]
},
{
"cell_type": "markdown",
"metadata": {},
"source": [
"### Discrete Time Implementation\n",
"\n",
"The simulation strategy adopted here requires a discrete time implementation of PID control. For a sampling time $dt$, the PID algorithm becomes\n",
"\n",
"$$q_c(t_k) = \\bar{q}_c - \\left[k_Pe_P(t_k) + k_Idt\\sum_0^{t_k} e_I(t_{k'}) + k_D\\frac{e_D(t_k)-e_D(t_{k-1})}{dt} \\right]$$\n",
"\n",
"Implementation is further streamlined by computing changes is $q_c(t_k)$\n",
"\n",
"$$\\Delta q_c(t_k) = q_c(t_k) - q_c(t_{k-1})$$\n",
"\n",
"Computing the differences\n",
"\n",
"$$\\Delta q_c(t_k) = -\\left[k_P(e_P(t_k)-e_P(t_{k-1})) + k_I\\ dt\\ e_I(t_k) + k_D\\frac{e_D(t_k) - 2e_D(t_{k-1}) + e_D(t_{k-2})}{dt}\\right]$$"
]
},
{
"cell_type": "markdown",
"metadata": {},
"source": [
"### Bounded Control\n",
"\n",
"A final consideration is that the coolant flows have lower and upper bounds of practical operation. \n",
"\n",
"$$q_c = \\max(q_{c,min},\\max(q_{c,max},q_c)) $$"
]
},
{
"cell_type": "code",
"execution_count": 6,
"metadata": {
"scrolled": false
},
"outputs": [
{
"data": {
"image/png": "iVBORw0KGgoAAAANSUhEUgAABGwAAAEMCAYAAACRPCChAAAABHNCSVQICAgIfAhkiAAAAAlwSFlz\nAAALEgAACxIB0t1+/AAAIABJREFUeJzs3XdcleX7wPHPGewpDsSNirgFB0iCppmlVqammZV+zYHm\nyL1wz8xyoqLmzvqq/Sqrr6aZOco9KhU3uAcge3M45/fHkSeODAERUK/36+WLc57zjPtcyH3ucz33\nUBkMBgNCCCGEEEIIIYQQosRQF3cBhBBCCCGEEEIIIYQpSdgIIYQQQgghhBBClDCSsBFCCCGEEEII\nIYQoYSRhI4QQQgghhBBCCFHCSMJGCCGEEEIIIYQQooSRhI0QT1FhLsImC7oJIcTzQ+p0IYQo2aSe\nzp7EpWhJwkaUKD/99BMffPABzZo1w8vLiw8//JD9+/cXd7EK5MSJE4wZM+aJzxMTE8Pw4cMJDQ0F\n4NatW7i7u3PgwIEnPrcQQhSX8ePH4+7unuu/o0ePFncxn4pFixaxffv24i6GEOI5V1Tt6qNHj+Lu\n7s7Vq1cB+PDDDxkxYkShXyfD+fPncXd3Z8+ePSbb7969i7u7O82bN8+SVJg/fz5NmzYlPT09T9f4\n9ddfmTNnTqGVObPcPveWL18OPP0YFtSSJUv47rvvirsYLxRtcRdACDBmaseNG8fu3bt5//33GTBg\nAOnp6fz8888MGDCA6dOn06NHj+IuZr58++233Lhx44nPc+HCBXbu3MnQoUMBKFeuHFu2bKFGjRpP\nfG4hhCguH3/8sUm97u/vT9u2benWrZuyrWbNmsVRtKdKp9OxYsUKZs2aVdxFEUI8p4q7XT116lTM\nzMye2vnd3d2xt7fnn3/+oW3btsr2w4cP4+DgQFRUFOfPn6du3brKa6dOnaJZs2ZoNJo8XWPjxo2U\nKVOm0MueoV+/frz66qtZtru4uDy1axaGZcuWMW3atOIuxgtFEjaiRNiyZQs//fQT69ato3nz5sr2\n1q1bY25uzpw5c2jXrh1OTk7FWMqSwdzcHA8Pj+IuhhBCPJEqVapQpUoV5blWq6V8+fJSvwkhxBMq\n7nb10062q9VqmjRpwl9//WWy/fDhw7Ru3Zrjx49z6NAhJWGTmprKuXPnGDVq1FMtV35UqlRJPu9E\nnsiQKFEibNiwgbZt25p8qGQYMmQIPXr0ICEhAYAdO3bQuXNnGjVqxCuvvMLq1atNuj26u7uzfft2\nhgwZgoeHBy1atCAwMNDknJGRkYwZMwZvb2+aNWvGsGHDuH//vvL6/fv3GT58OE2aNKFJkyaMGTOG\nqKgo5fXx48czevRoVq1ahZ+fHw0bNsTf3185x/jx4/n+++/5+++/cXd359atWyxdupT33nuPqVOn\n4unpSf/+/QG4cuUKgwcPxsvLi/r16/Paa6+xbds2wNjFtFevXgB06NCBpUuXZjsk6vDhw/To0QNP\nT0/8/PyYP38+qampyutt2rRh/fr1TJgwgSZNmuDl5cXMmTNJS0sr2C9MCCGKyL179xg2bBiNGzem\nadOmjBs3jpiYGOX10aNHM27cOAIDA3nppZfw9PRkypQpxMfHM3XqVBo3bkyrVq2UejXjmBEjRrB4\n8WK8vLzw9vZm5syZJvWmwWBg5cqVtGnThgYNGtC1a1eOHz+uvH7o0CHc3d3ZsmULzZs3p2XLliQm\nJhIdHc3UqVNp2bIl9erVw8/Pj3nz5qHT6dDpdNSrVw+ASZMm8Z///AeAli1bsnDhQpP3PWzYMOX1\n69ev4+7uzqZNm/Dz88PLy4ubN28CsG3bNl577TXq169Px44d2b17d6HGXwjx7MlPuxoe37ZOTU0l\nMDCQdu3a0bBhQzp37szvv/+e4/UzD+fJGC516tQp3nnnHRo0aMBrr72WZTjTn3/+SefOnWnYsCFd\nunTh119/VdrQ2fH29ubs2bPo9Xpl25EjR2jWrBne3t4cPnxY2R4cHExKSgre3t6AsX7/8ssv6dCh\nA/Xr16dp06YMGTJEacd/+OGHHDt2jB07duDu7q6c5/Tp07z33ns0bNgQPz8/li5danL9Nm3asHDh\nQiWWhTn0NSIigvHjx+Pr64uHhwf9+/dXhqBlxCoiIkLZf/DgwXh6eqLT6ZRtnTt3ZuHChcrv5L//\n/S8+Pj74+voSHx9PbGws06dPp2XLltSvXx9fX1/mzJmjfF/IiMW0adP48MMPlfP+3//9H6+//jr1\n69enQ4cO7Ny5s9Det5CEjSgB7t+/T0hICH5+ftm+XrFiRSZOnEjlypX56quvGDlyJF5eXixbtozO\nnTuzePFi5s+fb3LMrFmzqFKlCitWrFASHRljdnU6Hf/5z384deoUAQEBfPbZZ4SGhjJo0CAAEhMT\n6dWrF5cuXWL27NnMnDmT06dP4+/vbzLu9ffff2fv3r1Mnz6d2bNnc/r0aebOnQsYu/q3atUKNzc3\ntmzZQrly5QD4559/uHPnDsuXL+ejjz4iPj6eXr16kZKSwueff87y5cupXr06kydPJjQ0lHr16jFl\nyhQAFi5caDJUIMPevXvp06cPVatWZcmSJfTr14+vv/46y/w5S5cuRaVSsXTpUvr27ctXX31l8gVG\nCCFKmvj4eD744ANCQkKYO3cu06dP58SJE/j7+5s0kn/99VeOHz/OZ599Rv/+/dmyZQtdunQhPT2d\nZcuWUb9+faZPn87du3eVYw4cOMCuXbuYM2cOw4YN4//+7/+YPn268vr8+fNZtmwZPXr0YNmyZVSt\nWpWPPvqICxcumJRx48aNzJs3j4kTJ2Jtbc2IESM4evQoEyZM4Msvv6RTp06sXbuW7du3o9Vq+frr\nrwHjELDJkyfnKx6rVq1iypQpBAQEULlyZTZv3syUKVNo164dK1asoHnz5nzyySfP7NxvQognl592\nNZCntvXo0aNZu3YtH3zwAYGBgdSsWZNBgwblmrR51OjRo3n77bdZuXIlFSpUYOTIkURHRwPG4f/+\n/v5UrVqVwMBA/Pz8GD16dK7na9asGQkJCVy5cgWAq1evEhYWho+PD97e3pw8eVJJwp8+fRonJycl\n4bB69WoCAwN5//33Wbt2LSNHjuTIkSN89tlngHFIV926dXnppZfYsmWLUsbevXvj6OjI0qVL6d+/\nP2vWrMnyHWTNmjV06tSJzz//PNuEWQa9Xq8k8jP+Zf5cyyw+Pp4ePXpw+vRpJkyYwOeff05UVBQ9\ne/bk7t27+Pj4oNVqOXbsGGBMSJ04cYLExETOnz8PGG9Wnz9/3uT/xfr16/n0008JCAjA1taWkSNH\ncujQISZMmMDq1avp0qULGzZs4PvvvwdQYtGvXz+mTp2qbJs0aRJt2rRhxYoVtGjRghEjRrB3795c\nf38i72RIlCh2Gdnsx43ZTE9PZ+nSpbzzzjtMmDABAF9fX1QqFStWrKBfv35K105fX1/Gjh0LQPPm\nzfnll184cOAArVq1Yt++fVy8eJHt27dTu3ZtAJydnRk6dCg3b97kwIED3L59m127dlGxYkUA6tat\nS/v27fn999+VsbKpqamsWrUKe3t7AC5fvsymTZsAY1d/JycnoqOjTbo76nQ6AgICqFatGgBnzpzB\n1dWVhQsXYmdnB0CjRo3w8vLi1KlTdO3aVelW6u7uTvny5bPcaViyZAk+Pj7MmzcPAD8/PxwcHBg3\nbhznz5+nTp06ANSoUUOZPO2ll17i999/58CBA/Ts2TOPvykhhCha3333HWFhYezevZvy5csDULt2\nbTp27Mj+/ftp3bo1YKxbly5dir29Pb6+vmzduhWNRsPMmTNRqVRUqVKFPXv2cP78eeWzJjk5mZUr\nVypfWnQ6HZ9++qnSZX7jxo2MGDGCvn37Asa69b333mP58uUsWbJEKeNHH31Eq1atAGPC32AwMGvW\nLJo2bQqAj48Pv//+OydPnqRr1640atQIgMqVK+d7LrJu3bopcx7odDoCAwN57733lDL7+fkRFRXF\n4sWLlTIJIV4seW1XQ97a1mFhYezatYv58+fz1ltvAcZegWFhYSxatEiphx+nf//+vPfee0rZXn/9\ndY4ePcprr73G6tWrqVGjBgsXLkSlUtGyZUvi4+P56quvcjxfnTp1sLOz4++//6ZWrVocPnyYKlWq\nULFiRbRaLUlJSZw+fRpvb29Onz6Nl5cXKpUKgLCwMIYNG8b7778PgJeXFyEhIfz222+AcUiXra0t\njo6OSjt+xYoVVK5cmcDAQGUeHCsrK6ZPn06/fv0oXbo0APXr11d6R+ZmxowZzJgxw2Rbhw4dsvS2\nBGMPlnv37vHLL79QqVIlwNjD6JVXXmHt2rUEBATQqFEjjh8/TocOHbh8+TJJSUm4urpy6tQpGjRo\nwOHDh7G1tcXDw4OTJ08Cpp9fycnJpKenM336dCXR5OPjw759+zh58iTdu3dXYlGpUiVq1qyJXq9n\nyZIldOvWTfne5efnR3R0NIsXL6ZNmzaPjYN4POlhI4pdRqWXU1Y5Q0hICNHR0bz++usm2zt06EBa\nWhp///23si2jQQygUqlwdnYmMTERMGbZy5cvryRrwJiQ+e2336hcuTLHjx/Hzc0NZ2dnJeNdqVIl\nqlSpwpEjR5RjXF1dlWQNGJM+SUlJub4HrVarfDkAaNCgAZs3b8bCwoKLFy+ya9cuVq1aBZCn4UoJ\nCQlcuHAh25ioVCqlQgZo2LChyT6ZYyKEECXR8ePHqVWrFmXKlFHq46pVq1KpUiWTFaSqVatmUh87\nOTlRt25dpXHu6OgIQFxcnLJPw4YNTerjV155Bb1ez19//cVff/1FWloafn5+ynXT09Px9fXNsnKV\nq6ur8tja2pr169fTpEkTbty4wf79+1m5ciWRkZGFMgQ187WuXr1KZGSkSRl1Oh1+fn4EBwebvFch\nxIsjr+1qyFvb+uTJk6hUqmz3uXDhAvHx8XkqV+a2eUYCPqPdfOzYMVq3bq3U2UCW6z1Ko9HQpEkT\npf1/+PBhJdHg7OyMq6urUl9nJG4yTJo0iY8++oiIiAiOHj3K5s2bOXXqVK719PHjx2nRogUGg8Gk\nvk1LS+PUqVPKfpnr6dz4+/vz7bffmvwbOXJktvuePHmS+vXrK8kaADs7O/z8/JS2fosWLZRhu8eP\nH6dhw4Y0btxYKduhQ4d46aWX0Gr/7a+RuayWlpasW7cOb29v5fNr1apVPHjwIMe4hIaGEhERQcuW\nLbN8Dl24cEHpQSWejPSwEcUu4w5A5q7qj7p3754yZ8GjM7ZnZLQzf2BYWFiY7KNWq5WxuDExMblO\nshYdHU1wcLAyz0BmmZM8lpaWOV4jJ46Ojllmpw8MDGTNmjUkJSVRuXJlvLy8AB57LjB++TAYDEoM\nMpibm2Nra2sSk4KUVwghilN0dDRnzpzJtj4ODw9XHtvY2GR5/dE671Fly5Y1eZ7xuRAbG6t80Xnz\nzTezPTbzXDeP1r+7du1i7ty53L17l7Jly+Lh4YGlpWWh1LeZr5XREB44cGC2+0ZERCg9N4UQL468\ntqvLly+fp7Z1TEwMdnZ2mJubZ7tP5rlwcpO5ba5WG/sMZNS1UVFRWdrmj9at2WnWrBnbt29Hr9dz\n7Ngxk2GtzZs35+TJk9y+fZuwsDCT4UlXrlwhICCAv/76CxsbG+rVq4eFhUWu9XR0dDQbNmxgw4YN\nWV4LCwvLV7nB+Htq0KBBnvaNjY3N9rxOTk6cPXsWQJlTJzIykuPHj9OkSROqVKnCokWLAGPC5uOP\nP85yfGZ79uxh9uzZ3LlzhzJlyuDh4ZFrXDLm9xw8eHC2r0dERCg3TETBScJGFDsnJydq167NoUOH\nsl1i8Pbt27Rp00aZyyXzhFqZnzs4OOTpenZ2diYTCGfYv38/DRo0wN7eHk9PTwICArLsk9dr5NUP\nP/zA8uXLmT17Nu3atcPGxobk5GS+/fbbPB1va2uLSqXiwYMHJttTUlKIi4sr9PIKIURRsrOzo0mT\nJkpX/cyetBGYeeJiQKlHS5UqpdxNXL9+Pba2tlmOzXyHMrOrV68yatQoPvzwQ/r27at8CerSpUuu\nZVGpVFnuhj/uS1BGj6J58+ZlO7SqQoUKuR4vhHg+5addnXGTMLe2dWxsLHFxcaSmppokbfLb/s5N\nuXLlsrRlIyMjH3ucl5cXCxYs4NSpU8TFxZkkZZo3b8727ds5deoU5cqVo3r16oAxSTRo0CDKly/P\nzp07cXV1RaVSMX/+fG7cuJHjtezs7HjjjTd4++23s7z2tOtbe3t7k6RQhgcPHijxr1+/Pg4ODhw7\ndoyTJ0/SpUsXqlatSlhYGH/88Qd37tzJcV4jgGvXrjF8+HB69uxJ//79lZsa2c2fmblcAHPnzsXN\nzS3L65l7BImCkyFRokT44IMPlEkjH7V06VLMzMxo27Ytjo6O/PLLLyav79y5E41Gk2XIT04aNWrE\n3bt3uXjxorLt0qVLDBgwgJCQEDw9Pbl+/Tqurq40aNCABg0a4ObmxtKlSzlz5kye31PG3YPcnD59\nmmrVqtG5c2flDvGff/4J/HvX4dEeOZnZ2tri7u6ebUwAPD0981xeIYQoaTw9Pblx4wbVq1dX6uMa\nNWqwZMkS5a5iQf3zzz8myfvffvsNMzMzPD09adiwIRqNhpiYGOW6DRo04MCBA2zbti3H+v3cuXOk\npaXh7++vJGvCwsK4fPmycocyu2Otra25d++e8jwlJYVz587lWv4aNWpgZ2fHgwcPTMp4/vx5Vq9e\nbTK0QAjxYslru7p69eqPbVs3btwYg8GQ7T516tR5bG/GvGjSpEmWCYz37dv32OPq1auHpaUlX3/9\nNW5ubiY9Rry9vUlKSmLHjh0mw6EiIyO5ceMG7733HtWrV1cS5ocPHzZJnD9aV3t6enLt2jWT+lar\n1bJo0aIsyabC1rhxY86ePWsyj2V8fDx//PGHMq+MWq3Gx8eH77//noiICBo3bkzVqlUpW7YsS5cu\nxc3NTRmKlp3g4GDl8ysjWRMREcGlS5dyjEvG/5+IiAiTuFy8eJFVq1bl6buQeDzpYSNKhK5du/Lb\nb7/Rv39/evXqhbe3NwkJCXz//ffs3buXWbNm4ezszMcff8zcuXOxsbGhZcuW/PXXX6xYsYIPP/ww\nz3db27RpQ61atRg6dCjDhw/HwsKCRYsW0bhxYxo3bkytWrXYsGED/fr1o2/fvpibm7N27Vr++ecf\nxo8fn+f3ZG9vz40bNzh8+HCOiZP69euzZcsWVq1ahYeHB8HBwQQGBqJSqZRxvRld2n/77bcsQ70A\nhg4dyuDBgxk3bhxvvPEGoaGhLFq0iFdffdVkCJcQQjxrunXrxsaNG5X62MzMjC+//JLg4GAmTZr0\nROdOSkpi0KBBDBw4kBs3brBo0SI+/PBD7O3tsbe3p1u3bkydOpWwsDBq1arFkSNHCAoKUib4zU7d\nunVRq9V8+umnvP3229y7d4+goCDS0tKUOcPUajW2trYcOXKEBg0aULt2bXx9fdmyZQteXl64uLiw\nbt26xw6hMjc3Z8CAASxZsoTU1FQ8PT05f/48CxYsoGvXrlmGLwghXhx5bVcDj21bOzo60rZtW6ZN\nm0Z0dDSurq78/PPPHD16lMDAwEIp74ABA+jcuTMjR46kc+fOnDt3jo0bNwK53wDVaDQ0btyY3bt3\nZ1lEo1SpUri7u3Pw4EGmTZumbC9dujQuLi6sWbMGGxsb9Ho933zzDcHBwSbJJ3t7ey5evMjRo0fx\n8vJi4MCBvP/++0yYMIEOHToQExPDwoULsbKyyvO8NQXVtWtXNm7cSN++ffnkk08wNzdn1apV6PV6\nevfurezn6+tLQEAAdevWVXqHNm3alJ07d/LRRx/leo3atWuj0Wj49NNP6dq1K2FhYQQFBZGSkmIy\nR6e9vT3Hjh3D09OT2rVr4+/vz6JFi0hLS6NJkyZcuHCBhQsX8tZbb8nnUCGRhI0oEdRqNYGBgXz1\n1Vf88MMPbN68Ga1Wi7u7O2vXrqVFixYA9O7dGwsLC9atW8fmzZtxcXFhxIgRj62EMstIwMydO5fJ\nkyej1Wpp2bIlEyZMQK1WY29vz6ZNm/jss8+UBE39+vXZsGGD0p0yL7p3786ePXsYMGBAtuNdwVgB\nh4SEsGHDBlasWEGVKlWYOHEiP/74ozKJmpubGx07dmTJkiXcu3cvy3tt27YtgYGBBAYG8r///Y/S\npUvzwQcfMGTIkDyXVQghSiJHR0e+/vprPv30U8aOHYtKpaJBgwasX7+eqlWrPtG5PTw88Pb2ZtSo\nUVhZWdGvXz+T8f2TJ0+mTJkyrFu3jvDwcGUp3F69euV4zpo1azJ79myWL1/Ojh07cHZ2pkOHDhgM\nBr777jv0ej1qtZrBgwezdOlSrl69yg8//MDQoUOJjIxkzpw5WFpa0qNHD9zd3QkODs71PQwYMABr\na2s2bdrEsmXLKFeuHP37988yT4EQ4sWS13Y15K1t/cUXX7Bo0SJWrVpFbGwstWrVYsWKFXleIepx\n3NzcWLZsGfPnz2fQoEG4u7szcuRI5s6di5WVVa7Henl5cfDgwWyX0Pbx8eHChQsmr6lUKpYsWcLM\nmTMZNmwY9vb2NGvWjAULFjBixAguXryIu7s7vXv3Zvjw4QwYMIBdu3bh4eHBmjVrWLRoEYMHD8bG\nxgZfX1/GjBmDmZlZocQhJ3Z2dmzatIl58+YxadIkVCoVTZs2ZcuWLcqKtmBM2ICxx1KGZs2asXPn\nzlyHQ4Gxt8ycOXNYtmwZu3btwtnZmddff5127dqxZcsW0tPT0Wg0DBo0iMWLF3PlyhV++uknPvro\nIywtLZXvMuXKlaNPnz45zmsj8k9lkFlHhRBCCPECGT16NLdv3+abb74p7qIIIcQL788//8Te3t5k\nEt4tW7bwxRdfcPToURniKV5o0sNGCCGEEEIIIUSxOH36NJs2bWLs2LFUqVKF0NBQFixYQPfu3SVZ\nI154krARQgghhBBCCFEs/P39SUhIIDAwkPDwcJydnfnPf/7DgAEDirtoQhQ7GRIlhBBCCCGEEEII\nUcLIWltCCCGEEEIIIYQQJYwkbIQQQgghhBBCCCFKmOduDpvw8LgCHadSqShd2oYHDxKQUWKmJDY5\nk9jkTuKTsyeJTdmydk+pVM+Wgtb3+SX/jyUGIDEAiQEUfQykvjcqSH0v/19zJrHJncQnZxKbnD1p\nbHKq76WHzUNqtTHIaolIFhKbnElscifxyZnE5tkhvyuJAUgMQGIAEoNnifyuciaxyZ3EJ2cSm5w9\nrdhIqIUQQgghhBBCCCFKGEnYCCGEEEIIIYQQQpQwkrARQghRKCIiIvDx8eH3338H4MyZM9SpUwdP\nT0/lX1BQEAAGg4EvvviC5s2b06xZM2bNmkV6enpxFl8IIYQQQogSRRI2QgghCkVAQADR0dHK8/Pn\nz9OyZUtOnz6t/Bs4cCAAmzdvZt++ffz444/s2LGDU6dOsXbt2uIquhBCiDzYsWMH7du3x9PTk44d\nO7Jnzx4AYmJiGDx4ME2aNOHll19m27ZtyjGSoBdCiIJ77laJEkIIUfS++eYbrKyscHFxUbYFBwdT\nu3btbPffvn07vXv3ply5cgD4+/uzePFi+vfvn+drFtWkd2q1yuTni0hiIDEAiQG82DEIDQ1l4sSJ\nrF27lsaNG3Po0CEGDBjAgQMHmDZtGtbW1hw6dIiLFy/Sv39/3Nzc8PDwMEnQq1Qq/P39Wbt2bb7q\neyGEeFEVacImODiYKVOmcOXKFapWrcr06dPx8PDIsp+/vz+HDx9Go9Eo206fPl1k5dQbDFy7G8c/\nVyPQG8C1vB31qzthptU8/mAhhHjBhIaGsm7dOrZu3UqXLl2U7efPn8fc3Jw2bdqg1+tp3749I0aM\nwNzcnJCQEGrWrKns6+rqSmhoKAaDAZUqb1+ESpe2yfO+hcHR0aZAx12/G8vBv27zVssa2NuYF3Kp\nilZBY/A8kRhIDODFjIGrqyt//vknNjY26HQ6IiIisLGxwdzcnD179rBr1y4sLCxo2LAhb7zxBj/8\n8AMeHh6FkqAvSqcvhfP31QfKc41GRcuGFahaXpZYF0IUvSJL2KSkpDBw4EAGDhxIt27d2L59O4MG\nDWLPnj3Y2Jh+6AUHB7N582YaNGhQVMUDQK83cDT4Pt8fCOHug0ST1yqVtWX8+55YW5oVaZmEEKIk\n0+l0jB07loCAABwdHU1eK1WqFN7e3rz77rs8ePCATz75hCVLljB69GiSkpKwtLRU9rWyskKv15Oa\nmoqFhUWerv3gQUKR9bBxdLQhOjoBvd6Q7+OnrDxEZFwK50IiGPOe51Mo4dP3pDF4HkgMJAZQ9DFw\ncrJ96tfIDxsbG27evMlrr72GXq9n2rRp3LhxA61WS+XKlZX9XF1d2b17N0ChJOgL0qOyIL2hdOl6\nVv54jlSd3mT7zbB4Jvdumr8ClGAvck+xvJD45Exik7OnFZsiS9gcOXIEtVpNz549AXjnnXfYsGED\n+/fvp0OHDsp+Dx48IDIyklq1ahVV0UhMTuP7g6GcvhxOZGyKsr2soyWW5lpuhcVzKzyexd/+w6h3\nPTA3k542QggBsHz5curUqUOrVq2yvJYxwTCAtbU1/v7+LFiwgNGjR2NpaUlKyr/1bVJSElqtNs/J\nGjDOi1CU0yDo9QbS0/P/BS0yzvg+z4ZEFuj4kqSgMXieSAwkBvBix8DFxYW///6bEydO8PHHH9O3\nb1+TBDyApaUlycnJAIWSoH+SHpX56Q0VE5+iJGsauZUhISmNK7diiElILXHJs8LwIvYUyw+JT84k\nNjkr7NgUWcImNDSUGjVqmGxzdXUlJCTEZFtwcDA2Njb4+/tz4cIFqlWrxrhx4/D0zNtdyYJk4Pf9\ndYffTt5SnjesUZq3/VypXsEelUrFgb/usOZ/57l8K4Yf/gjlvbZu+bvAM04yqTmT2ORO4pOz5yU2\nO3bsIDw8nB07dgAQHx/PyJEjGThwINHR0QwePBhbW2MjNyUlRWmc16hRg9DQUBo1agQYPyOqV69e\nPG+iCKXr9WiKoluQEEI8JVqt8euDj48P7dq14+zZsyYJeIDk5GSsra0BCiVBX5AelQXpDRUWlaQ8\n7vmKGzfzoWREAAAgAElEQVTC4rhyK4a4hFQiI+PzV4ASTHrL5U7ikzOJTc6eNDY5JYWLLGGTmJiI\nlZWVybbM2fcMKSkpeHh4MGbMGKpWrcq3335L//792blzJ2XLln3sdQqSgW/fojqR8alUKGtD8/ou\nVC1vb/L6221qEZmQxvf7rvDnmbsM6NLwhZzPRjKpOZPY5E7ik7NnPTa//PKLyfM2bdowefJkWrVq\nRbt27TAYDIwaNYo7d+4QFBRE9+7dAXjrrbdYs2YNzZs3R6vVsnLlSjp16lQcb6FI3Q5PoIqzzIMg\nxOMYDAbu3buLi0uF4i6KeGj//v2sW7eO9evXK9vS0tKoUqUKBw4c4M6dO1SoYPx9hYaGKsOgCiNB\n/yQ9KvPTGyoxWac81mrUWJkbvyolp6aTkpqOVvN8Jdxf5J5ieSHxyZnEJmeFHZsiS9hYWVllSc5k\nzr5naNu2LW3btlWe9+zZk2+++YajR4/yxhtvPPY6BcnAa4E+7d2VjFh2GXS/+s78sO8KcYlp7D12\nnWa1y+XvIs8wyaTmTGKTO4lPzp4kNs9Ct2y1Wk1QUBCzZs2iefPmWFpa8u6779K7d2/AWLdHRETw\nzjvvkJaWxptvvkmfPn2KudSFT5duOg9CyN1YSdiIZ4qvb1MsLCxQZ2pclS5dhg8+6M0bb7z91K67\nbNliAIYMGf7UriHyp27dupw9e5YffviBt956i4MHD7J//362bt3K3bt3+eKLL5g1axaXL1/m559/\nZtWqVcCzlaBPTv03YWNprsEm09yVCck6HJ7xieOFEM+eIkvYVK9ena+++spkW2hoaJYkzC+//IJe\nrzeZ1yZzN/rHedI5DXLKiDnaWlCnWimCr0Vx4K87NHZ7fG+f541kUnMmscmdxCdnz1ts9u7dqzyu\nWbOmyZ3YzDQaDSNGjGDEiBFFVLLikZiiM3keeieWlz0qFlNphCiY1as3UL16TTQaFQ4OVmzd+n/M\nmDGV+vUbUa2a61O5ZkxMNA4Ojo/fURSZsmXLEhQUxJw5c5gxYwbVqlVj2bJl1KhRg5kzZzJ16lRa\ntWqFtbU1Y8aMUXrUPEsJ+uTUf79EWJhrsLH696tSYnKaJGyEEEWuyBI2Pj4+pKamsmnTJnr06MH2\n7duJiIjA19fXZL/ExES++OILatWqRdWqVdmwYQPJycm0aNGiqIqaI98GLgRfi+JMyAOi41NwtM37\n2FshhBAvnszd68HYw0aIR+nS9UTGJj9+x0LgZG/5RMM6NBoNr73WgUWLviA09CrVqrkSGxvD4sWf\nc/ToESwtLenUqSsffNAblUpFTEw0ixZ9zpkzfxMVFUmlSpUZNWo8DRt6APD999/y9debiImJpk6d\nuowdG8DBg/vYvXsnKpWKe/fuMmvWPI4fP0JQ0DJu3rxBhQoV8ff/GB8fYxvS17cpnTt349dff+H9\n93vx4YclMxnwPGjatCnfffddlu2Ojo4sXrw422OepQR9RsLGwlyDWqUy7WGTpMvpMCGEeGqKLGFj\nbm7O6tWrmTZtGgsWLKBq1aqsWLECa2trpkyZAsCMGTPo0qUL4eHh9OvXj+joaOrWrcvq1auzDJ0q\nDo1rlcXKQkNSSjqnL0fQ2lPukgohhMhZQnKayfM74Qkkpeiwsiiyj19RwunS9UxcdYSImKJJ2JRx\nsGTOgOYFTtqkpqaydes3pKSkUK9eAwBmzpyCvb0D27b9SHR0FGPHDsfJyYmOHd9i+fIlAGzevA21\nWsPixV8QFBTI8uVfcuTIIVatWs4XXyzB3b0OK1cuY+bMKQQFreXq1Ss4ODgyZMhwQkKuMm7cKKZO\nnUmLFi05fvwIkyePZ+XK9dSoUfNhuVL46afdpKWlFk6gxAspY0iU5cMVYS0fJm70BgPxj9TnQghR\nFIq0xVi7dm3++9//Ztk+Y8YMk+f+/v74+/sXVbHyzNxMQ40KDpwNjeRm2PMzU7wQQoin49EeNgbg\n6u0Y6lcvXTwFEqIABg7si1qtIjU1DTDQvLkPixatoFw5Zx48iODIkUP8/POvWFlZYWVlRc+evdi+\n/Ts6dnyLAQM+xtLSEo1Gy927d7CzsyM8PByAPXt20b59R+rWrQ9Anz79uX49NMv1f/ttN02bNqNV\nqzYA+Pj40qJFS3bv3smgQUMBaNu2HWZmZpiZmWU5Xoi8yuhhY2luTNioVCpsrLTEJaaRkCQJGyFE\n0ZNbfPlUqawtZ0MjuSUJGyGEEI+R0cPG2kKLk70lt8LjOX89ShI2QqHVqJkzoHmJHhIVFLSG6tVr\ncv/+HQICxuLg4Ei9esYky/379zAYDLz77r8TEOv1BuztjStuRkREsHjx51y7FkrVqlWxs3PAYDBO\nxh0ZGUmNGm7KcVZWVtSuXTfL9aOiIilf3sVkW/nyLoSHh/37vpzK5Os9CZGdfxM2/35FsrE0MyZs\nkmVIlBCi6EnCJp8qlTMuwXsrPB6DwZDvJcSFEEK8ODJ62FhbaqlTtZSSsBEiM61GTblSxT/0+3Eq\nVKjI8uXL6dSpE+XLV6B3776ULl0GjUbDjz/uxtzcOCFrbGwsiYmJAEydOpFOnbqwbNlqVCoVO3f+\nTEjIFcA4iW1ExL9Jl4SEeNauXc3AgUNMruvsXJ5z586abLt79w5ly/67Yqc0x0RhUIZEPexhAygT\nDyfKkCghRDEo+KxzL6hKZY3L6SanpvOgiMabCyGEeDZl3JG1sTSjTrVSAFy/F5dlbhshnhUVK1bk\nk09Gsm7daq5cuYyzc3kaNfJkxYqlpKQkExsbw+TJ41i9ehkAiYkJWFlZolKpuHYtlK+/3ohOZ/y7\naNeuPb/88j8uXbqATqdj48Z1BAefVYY2JSQkAPDKK+04deo4Bw7sIz09ncOH/+SPPw7Qtm27YouD\neD6lPDIkClAmHpZJh4UQxUESNvnkUtoG9cPbOLfCE4q5NEIIIUqyjDkPrC21uFd2RK1SYQAu3ogu\n3oIJ8QQ6dnwLT88mzJ07g/T0dKZNm01k5APeeectevToQpkyZRg5chwAY8dO5OuvN9GuXSsCAsbQ\nvv0bREdHERMTTZMmzRg0aBhTpkykY8dXuHr1MtOmzQagdeu27Nv3GyNHDqFSpcrMnfs569ev5vXX\nW7N8+WKmTp1FnTr1ijMM4jmUeZWoDDaWxh42kmgXQhQHGRKVT2ZaNeVLW3MnIoGb4fF4uMmYaSGE\nENlLVHrYaLGy0OLqYsfVO7Gcvx5F41pli7l0QjzeH3+cyHb7woXLlMdOTqWZPn1Otvv5+rbC17eV\nybaePXspj994oxNvvNEpy3HNmnmzc+feTM+b06xZ83yVUYj8ymkOG0BWiRJCFAvpYVMAlcoa57G5\nHS4TDwshhMiZMunwwwZ/7arGYVFnQh5gMBiKrVxCCCGyyn4OGxkSJYQoPpKwKYCMeWxkaW8hhBC5\nydzDBsCjprFXZlhUEnceJBZbuYQQQmSV/Rw2MumwEKL4SMKmADISNvcjk0jTpRdzaYQQQpRUCZlW\niQJwrWCPg41xJZ3Tl8KLrVxCCCGyym1IlCzrLYQoDpKwKYCMIVF6g4F7kUnFXBohhBAlVWKK8Y5s\nRoNfrVLh+XDus9OXJWEjhBAlSW7Leickp6GXoaxCiCImCZsCcLSzUB7HJqYWY0mEEEKUZI/2sAHw\nfDjZcOjdOCJjk4ulXEIIIbJKzmVZb4MBklOkl40QomhJwqYAtBo1VhbGxnd8ooxnFUIIkZUuXa/M\nh5DR4AeoXaWU8mXgpAyLEkKIEiPbIVFW/9bf8TIsSghRxCRhU0B2Dyvv+CRJ2AghhMgqMdOd2Mw9\nbMy0ajzdjL1sjpy7V+TlEkIIkVWaTk+63jjkySKbSYdBJh4WQhQ9SdgUkK21MWETJ0OihBBCZCMx\n053YzHdoAXzqOwPGYVF3HyQUabmEEEJklTF/DZgOicqccJelvYUQRU0SNgVk+7DxHSc9bIQQQmQj\nIdOd2Mx3aAHqVC2lrBZ15Nz9Ii2XEKLo3blzu7iLIB4jYzgUmCZsNGo1VhbG5wnSw0YIUcS0j99F\nZEcZEiVz2AghhMhGRg8bFSjznmXQqNV413Vm9/GbHD53j05+rqhVqmIopRCPd+TIIb75ZhOXL19C\npYLatevQv//H1K5d94nOu2bNSkJDrzJr1mds3LiW69dDmTx5ZiGV2lRiYiLt2rVk27YfcXGpUKBz\nnDp1gsmTx3Hs2LF8HXfp0gXGjPmE7dt3Fei6omikmCRsTOtsG0szklLSSZAbtc8tvd6ALv3fYXFq\nsxSi41LQpRswGAzo9Qb0gEFvQG8woDeAwWDAYDAea8CAXm/cpn+4/dHjDBgnrzYyKI8zbcKA6cac\njsl0CIZHNhpyOM+/j7M5Tx4XQFOrwcbGkoSEZPT6zOUohBXUnvAUxb2Gm0oF1SqVomoZq0I9ryRs\nCihjSJTMYSOEECI7GXdirSy02SZjXqpfnt3HbxIRk8z5a1HUc3Uq6iIK8Vg//vg9X34ZxLhxk3jp\nJR8cHKxZvXotw4YNIihoLdWr1yiU6/Tq9VGhnKckio+PR6eToTQlXU49bMCYsImISc510uH9f91m\nz4lb2FmbUc3Fnk4tXE3mwhH5k6bTk5SqIynl4b9kHYkp6SSn6khNSyclTU+aLp1UnZ7UND2punTl\nZ5pOT2qa8acu3UC6/tGfBtLT9ej0BtLTjY+L+8u+eH5M+KAxbpUcC+18krApIGVIlPSwEUIIkY3U\nNOOtp5wa7FWc7XB1sSf0biy/n74tCZsXmE6vIyo5pkiuVcrSAa06b82/5ORkAgMXMXXqLFq08EOj\nUWFhYUHPnh8SGRnF9euhVK9eg8jIByxZsoDjx49gYWFJ27bt6NdvEObm5iQmJrJixVL2798LwEsv\n+TJkyAhsbW1NrpW5t83s2dOwsbHh0qWLXL58kSpVqjF2bADu7rUxGAysW7ea777bhrm5Od26vUdQ\n0FL++9/v89Vz5sSJY6xZE8TNmzdITU3Dy6s5kyZNx9LSkri4OBYt+ow//zyIVmtGhw5vMGjQMJPj\nExLiGTJkEG5utRg7NoDU1BRWrFjKvn17MRgMvPrq6/j7DyY+Po7Roz8hNTWFV1/149tvf8LBofAa\n8qLwZJ7D5tF628bK+DeTUw+bC9ej2LjrotJL4cKNaEJuxzC8e6MsvXVeVInJOqLikomKTyEuIY24\nxFTikh7+TEwz/ktKIyk5jcSUdHTp+sef9BmlyvRA9fBZ5vs6xsem2zNtMh6Tabsq8wmzbANVpicm\n53u44ZHDH1t2tVr1sFdRDu/rCaiKubfxk16+fGkbKpSxKZzCPCQ1SAHZWRvnHohPkkmHhRACICIi\ngjfffJM5c+bQunVr7t27x4wZMzh58iRarZbXX3+dcePGYW5ujsFgoEmTJibdeJs0acKXX35ZjO+g\ncGU0NrWanD/9W3tWJPRuLH9djiAqLoVSdhZFVTxRQuj0OmYcmc+D5KgiuV5py1JMaT4mT0mbM2f+\nIj1dh7e3T5bXBg0aqjyeOHEMLi4V2LbtRxISEpg4cTRr1qxk0KChfPbZbKKiItmw4b9oNBpmzZrC\n/PmzmT59bq7X/uWXHSxdupIqVaowZ850Vq4MZMGCQP73vx/ZseMnVqxYg5OTEzNnTiE9PT3Xcz0q\nKSmJgIAxTJ48A1/fVoSF3efjj/uxZ88vvPHG23z++RySkpLZuvVH0tN1DB06kIoVK1O5chXl+NGj\nh1OzphtjxwagUqkIDFzMrVs32LDhG/R6A5Mnj2PjxrX07evP558vZvLkcfzvf7/lq5yiaGX0sLEw\n02TpFWn/cM6xqLiULMfFxKew8sdzGAzgUtoat0oOHPj7LpduxbBo69+M6uGJmfb5njbUYDAQn5TG\n/agk7kcmEh6dRGRcClFxKUTGJhMVl2LSg6mgzLRqLM01WJhpMDfTYK5VG/+ZaTDTqh9uV2OmffhT\no0arUaPRqNCqH/7UqNGoVSbbNGo1Wo3q4XbjOUuVsiE2NhGDAdQqFSrVw59qFWqMSQtV5u2qh9v4\n97FaZUxAZLxe3MmIwqDRqHBysiUyMp70dOmXlNnTio0kbArINtOy3gaD4bn4AxRCiCcREBBAdHS0\n8nzMmDG4ublx4MABYmNjGTx4MMuWLWPEiBFcv34dgFOnTj239adOl5Gwybmh7lWnHFv2XiYhWcf+\nv27ztl/1oiqeEI8VHR2NnZ09Wm3OzcXbt29x9uw/fPrpAqytbbC2tqFfv0HMnj2Njz7qz759vxEU\ntJZSpUoBMGTIcN5/vxsTJ07N9dq+vi1xc6sFQJs27QgMXATA7t076d79PSV58vHHw/jjjwP5el/m\n5uasXbuZihUrER8fT0REOA4OjoSHh5OamsqBA/tYtWoD9vb2AMybtwAzMzNu3bqJXq9nyJAhqFRq\nxo2bhEqlwmAwsGPHj6xYsUbpPdO3rz/TpgXQt69/vspW0p04cYJ58+YREhJCqVKl6NevHz169ODM\nmTN0794dS0tLZV9/f38GDhyIwWBgwYIFbNu2jfT0dDp16sSECRPQaErWcKGMhMKjw6EAXJysAbJd\n1e+nQ9eISUjFwkzDkC4NcCltQ+Vydmz+9RKXbsXw1e6L/Kd97efisy5dr+d+ZBK3wuO5E5FAWFQS\n96MSuR+ZRGJK3of92VhqsbU2x87aDDsrM+wyHlubY2OpxdJci7WFBitLLVYWD/+Za4ss8aV88bbS\nSFJCFDtJ2BSQ3cM5bHTpBpJT07NMKCmEEC+Sb775BisrK1xcXABITU3FysqKQYMGYWFhQdmyZXnz\nzTf59ddfAQgODsbd3f2JGrAqlQp1EbTd1GqVyc+8Sn/Ye8hMa7yDlx0rjRbfhi7sOnaTA3/fpZOf\na64JnuJS0Bg8T55WDDQaM6a3GEtUcvTjdy4EpSwd8zwkqmzZMsTGxmIw6NBqzUxiEBsbi7W1NTEx\nUVhZWVG6dCnluAoVXIiMfEBcXAw6nY6KFSsofwMVKlTAYDDw4EGEcodao1GZPFapwNGxlHKMubkW\ng0GPRqMiIiKc8uXLm5wPjF+wcvo7y3g946e5uZZDhw6yZctmAGrWrEVychJgIDExjrS0NFxcnJVj\nqlSpDMDdu7eIi4tDr9dz/vw57t69ReXKVYiMjCIlJYWhQ/2VOs1gMJCWpkOnSzW59rMsJiaGjz/+\nmMmTJ9OxY0fOnz9Pnz59qFKlCrdu3aJly5asXLkyy3GbN29m3759/Pjjj6hUKvz9/Vm7di39+/cv\nhneRs4whUdkNY80Y4nAvMpF0vR7Nww+fNJ2eo8HGlf46+FTFpbRxv1eaVCI2IZWfDl3j4D93qeZi\nT2vPikXxNgqNLl3PzbB4rtyK4cb9OG6Gx3MnIvGxQ5WsLbSUK2VFaXtLStlZ4PTwp/GxBY62FiXy\nc06IkkqyDAWU0cMGjL1sJGEjhHhRhYaGsm7dOrZu3UqXLl0A4x3sVatWmez3+++/U7t2bQDOnz9P\nfHw8nTp1IiwsjGbNmhEQEICzs3Oer1u6tE2R3rF0dMzfmGQzM+PngqWFFicn2xz3e7u1G7uO3SQ6\nPoWr9+LxaVCwFWyKQn5j8Dx6WjEoR8mb16Rly5cwNzfjzJlTvPLKK8p2R0cbJk0ah42NDSNHjiQp\nKQmVKk3pRXPmTCSOjo64u1d/OI9NDNWrG5MeV6/eR61WU716JayszDE3N/59ZH5sYWGGlZW58ndj\nZ2eFRqPGycmWSpUqEhcXpbx2/foDABwcrLP8nY0fPx5/f39cXV3RaIy9J5ydnbh27RLr1q1m27Zt\nVKtWDYBevXphZWVO9eqVMTMzIyUlHienSgAcPHiQ6OhoypUrh729PWvWrGHatGnMnz+HTZs24eBg\nhZmZGdu3b6dyZeP7TExMJCIiAheX0ty4YYVKpcq1HngW3Llzh1atWvHmm28CUK9ePby9vTl16hQR\nERFK/f6o7du307t3b8qVKwcYe94sXrw4XwmbgiTo85tkTX3YK9LKXJsluVapnPF3p0s3EBmbQvnS\nxh43py5FkJCsQwX4NXIxOa7Ly9W5cT+Ov68+YPPuS5R1tKRRzTL5exNPSXaxSUzWcfV2DJduRnP5\nVgxX78Qoc7E9ykyrpkJpG8qXtsK5lDXOTtaUd7LG2ckKWyuzZ743kdykyJnEJmdPKzaSZSigjDls\nwJiwKetYuMt3CSHEs0Cn0zF27FgCAgJwdMz+C6fBYGD27NmEhIQwf/58wJjQ8fDw4JNPPsHCwoLZ\ns2czdOhQtm7dmudrP3iQUGQ9bBwdbYiOTkCvz3vX6Nj4ZOMDA0RGxue4n7VWRd1qpQi+FsX2/Vdx\nr2j/pEUudAWNwfPkRY3BwIGDmTRpMomJqfj4vISlpYagoNX8+echVq1ai5mZDU2bejFt2gzGjJlA\nfHw8Cxcu4tVXXyM6OpHXXmvPp59+xowZs1GrNcyaNYeXXvIlLU1FUlIqqak6IiPjTR6npKSRlJSq\n/N3ExSWRnq4nMjKedu3as3LlCjw9vSlTpgzz5hnrlJiYRKytTf/Ozp+/wNat/8dHH/Xnu+++pUyZ\nMqhUFty9G4FKpSI5OZ3w8Bh27drJiRMnqF27HjExSbzySju++GIBU6bMIDk5mTlz5vLuu+9hZWWH\nSqVGrVbTr98gunfvwtq1G+ncuSvt2rVnzpxPGT9+ElqtlnnzZnPv3l2CgtaQnJxOSkoK9+9HYWZm\nliXGuSlJSZ46deoodTgYe9ycOHGCTp06sXr1aszNzWnTpg16vZ727dszYsQIzM3NCQkJoWbNmspx\nrq6uhIaG5mtKgSdJ0Oc5yfrwA8XWxjxL3O0drNGoVaTrDcQkp1P34etHL5wFoKFbGdyqZU3GjO/j\nzfjAg1y/F8ey788yy/8lalcrGRPMh0UlEhwaxfnQB5y/Fsm1u7HZLu3sZG+BW+VSVHOxp1oFe6q5\n2ONSxhbNC/CFXW5S5Exik7PCjo0kbArI2kKLSmVcs15WihJCvKiWL19OnTp1aNWqVbavJycnM3bs\nWC5evMimTZsoXbo0AEOHDjXZb9y4cTRv3pywsDDlLuzjGAwG8jnX6BPRP1z+M68y7kxqNarHHvey\nR0WCr0VxLjSS2+EJlH84X0JJk98YPI9etBi8/XY3rK3tWLNmFdOnT0aj0VCnTj2WLl1JtWo1SE83\nMGXKTBYv/pyuXY09L9q1a4+//xDS0w0MHTqS5cuX8MEH75KSkoqfXyuGDRtFerrBuMqIwZDlscFg\nbF9lxFn/8CZ/erqBV155natXQ+jbtxdWVla89loHANRqbZbfy6hR45k/fw5ff72J8uVdmDx5Jno9\nNG3qTevWbXn//XfRaNS4u9elffs3CA0NJT3dwPDhY1iy5Au6deuMSqWiU6cudOz4NqdOnYCH66LY\n2toxdOgIvvjiU3x8fPnkk1EsX76Unj27kZycTKNGHkyfPpf0dAOurjWpVq06r73WhnXrNlOpUuWi\n+eU9RXFxcQwcOJB69erRpk0bvv32W7y9vXn33Xd58OABn3zyCUuWLGH06NEkJSWZzG1jZWWFXq8n\nNTUVC4u8TbRekAR9fpOs0bFJAGjV2SfZnZ2suRORwKVrD3CvaEdMfAonz4cB4F2nXI6J+eHdGjJz\n/Qki41KYuPxP+r5RB5/65fP3Zp6QXm/gZli80nvm8q1oImOzTqAMULGsDW6VHKhV2RG3So6UdbTM\nkiyLic46l8/z5EVN0OeFxCZnTxqbnBL0KoMhu1zqsys8PK5AxxVkVudhiw8Sn5RG3451aNHApUDX\nfRbIbOA5k9jkTuKTsyeJTdmydk+pVPn3+uuvEx4erjTm4uPjlblrunfvTr9+/bC2tmbJkiUmPXBW\nrVpFixYtqFevHgD379+nZcuWHDt2DAcHhzxdu6D1fX4V9Hf11e6L7D11m4Y1SjO8W6Nc99Wl6xkX\ndJiouBRaeVSg9+vZDy0oLvK3LDGAkhGDy5cvUaqUE2XKGHszXLsWSq9e7/LrrwewsLB8zNFPrqhj\nUJLq+ww3b95k4MCBVK5cmUWLFpkkYzLs2rWLBQsWsGvXLho3bsy6deto1MhYD166dInOnTtz7ty5\nPF+zIPV9fn9Xa34O5s+z9/CqU46BnepneX3592c4cTEcn3rO9H+zHruP3eC/e69gYa5h0RDfbOe+\nyXAnIoFF2/4mIsbY89LTrQyd/aorQ60KW3KqjpA7sVx5mJy5eic221WazDRqXF3scKvsSM2KDtSo\n6GAy7cOLqiTUdSWVxCZnTxqbnOp76WHzBOyszYhPSiM+SXrYCCFeTL/88ovJ8zZt2jB58mRefvll\nevXqRZkyZVi6dGmWYQAhISEcPHiQJUuWoNVqmT17Nq+88kqekzXPgoyJGc3yMLmiVqPm1aaV2fr7\nFf48c5dOvq442soS30I86siRQ5w4cYy5cz9Ho1GzefMGPDwaF0myRsC5c+fo168fb731FuPGjUOt\nVhMTE0NQUBCDBw/G1taYgEhJSVF6z9SoUYPQ0FAlYRMaGkr16iVvRbzcVomChxMPXwznTkQiAH+e\nvQdAU/eyuSZrMo6d1Lspy78/y6Wb0Zy+HMHpyxFUKmtDfdfSVHG2pbKzHS5O1vme/8JgMBAWnUTo\n3VhCbsdy+XYMN+/Ho8/mnrytlZmx90wVR5rWdaGUtTbLEuZCiJKlSBM2wcHBTJkyhStXrlC1alWm\nT5+Oh4dHjvsfPnyYPn36cPLkSWxsSt44ucxLewshhPjX6dOnOXbsGBYWFnh5eSnb69aty+bNm5k0\naRKzZ8+mffv2pKWl8fLLLzNz5sxiLHHh0z28u6LN4zKkrTwq8POhaySm6Pj1+E26ta75+IOEeMG8\n+25Pbt++Sffub5GWloanZxMmT55R3MV6IURERNCvXz/69OnDgAEDlO12dnb8+uuvGAwGRo0axZ07\ndwgKCqJ79+4AvPXWW6xZs4bmzZuj1WpZuXIlnTp1Kq63kaOMVaIszbP/epSxAtTdyASu34vjZphx\nCHf/oE0AACAASURBVFSL+nnrZW9vbc7Y9zw5Gnyf7w+GEBGTzK3wBG6F/zu8yEyrpqyjFeUcrSjr\naEUpOwusLDRYW5phaW5cYjoxJY0HMcmExyQTEZ3EzbB4EpKzX1Lb2ckat0oOuFV0wK2yI86lrJTV\n2KSXhBDPhiJL2KSkpDBw4EAGDhxIt27d2L59O4MGDWLPnj3ZJmNiYmKYOHEiJXnEVkbCRuawEUII\no7179yqPL168mON+tra2zJ07tyiKVGwyetho87iUr5WFljZNKvHzoWvs++s2HX2qYW0pHWGFyMzc\n3Jzx4ycDk4u7KC+cb7/9lsjISFasWMGKFSuU7b169SIoKIhZs2bRvHlzLC0teffdd+nduzcAPXv2\nJCIignfeeYe0tDTefPNN+vTpU1xvI0fJaXnoYYNxfrKfDl0DoLS9JbWq5H2FN7VahU/98njXdeby\nrWhOXgon9E4sN8PjSU3Tk6bTcycigTsR+Z8jxkyrpqqzHTUrOuBWyYEalRywz7RIihDi2VRkLcEj\nR46gVqvp2bMnAO+88w4bNmxg//79dOjQIcv+06ZNo0OHDnz55ZdFVcR8s7OWHjZCCCGyl6bL+5Co\nDG2bVmL3sRskpaTz++lbdPSp9pRKJ4QQ+ZNx4zUn69evz3a7RqNhxIgRjBgx4imVrHCkpBrrbHOz\n7BM25Z2slAVHTl0KB+Cl+uULNKRIrVbhXqUU7lVKAcZJge9HJXIrPIGwqETCo5MIi0oiLjGNxBQd\niSk6UlLT0ahVWJhpKO1gSRkHS8o4WOFSxprqLvZUKGODNh+fN0KIZ0ORJWxCQ0OpUaOGyTZXV1dC\nQkKy7Pvjjz8SGxvLqFGj8p2wUalUBVrmtSDrptvbGLPW8UlpaPJ4B/VZ9LTWlH8eSGxyJ/HJmcTm\n+ZcxJEqTjwa0vbU5fg0r8NupW/x64hbtmlXGTJv73AhCCCGeXMacL9ocPpfNtBpqVHTgyq0Y436a\n/2fvvsOjqtIHjn+nZJKZJJBCEnoIRTpEULqigChKWZWirIgoQgDRDRZc+IlIscMusK4BFF2BtaLi\nInYFFAREmpKAlAAJoSUhIZmUaff3x2SGjClM6swk7+d58pice+/cM2/inct7z3mPiv7dqmfREbVa\nRZPwQOe0q9JUZBl0IUTdUWsJm7y8PPR6vUtbQEAABQUFLm1paWksW7aM//73v5jNFR+5Eh4eWKWL\nWUXWTY9qZC+slm+ylLkMV11S3WvK1yUSm/JJfMomsam7KlJ0uLhbe7Xgh31nuGw08ePBswzq0bwm\nuieEEKIYa9E1u7wHKfFjunM0NYsCk5Um4YFEhujL3Le6SbJGiPqp1hI2er2+RHKmoKAAg8Hg/Nlm\nszF79mzi4+OJiooiNTW1wufJyDBWeoRNRddNVxdl4rNyCsnMzK34SX1EVdeUr8skNuWT+JStKrGp\nDwniusDsqGGjrdhNdqMQPb07RfHzoXP8b8dJ+ndtgn8ZQ/SFEEJUD2vRZ7GmnISN3l9LtzaNaqtL\nQghRewmb1q1bs27dOpe25ORkhg8f7vz53LlzHDhwgKSkJObPn4/NZr/ZHThwIAkJCVx33XVXPY+i\nKFitle+nzaa4XS1dX1RFPq/AUi8qrFckNvWNxKZ8Ep+ySWzqLovFUXS44k8RRt0Qw+6k82Tnmvju\n11Ru7xNd3d0TQghRjGNKlExVFkJ4k1qrTNW3b19MJhNr167FbDbz0UcfkZ6ezoABA5z7NG3alIMH\nD7Jnzx727NnDZ599BsDWrVvdStbUNsfKH1ab4tWrWQkhhKh9lZ0SBRAZomdgbFMANv98SorbCyFE\nDXOMsJGEjRDCm9Rawkan07F69Wo+//xzevXqxbp163j99dcxGAzMmzePefPm1VZXqk3xIZNWme4h\nhBCiGEfR4cqu2jGiXyv8/TTkFVr45MeSBfqFEEJUH8f0ZG1laisIIUQNqbUpUQAdOnTgvffeK9G+\nYMGCUvdv3rw5R44cqeluVVrxlT+sNgVZyEMIIYSDxVnDpnI3/w2D/BneL5oNW0+wZd8ZBnZvSsuo\n4OrsohBCiCI2GWEjhPBCkkKuguIXdKlBIYQQojhn0eEq3PwPvb4lkSF6FAXWff2Hs8aCEEKI6uVO\n0WEhhKhtkrCpAtcpUTYP9kQIIYS3cRYdruQIGwA/rZrxt1wDwLEz2fyw90y19E0IIYQrGWEjhPBG\nkrCpAqlhI4QQoiyOGjaVKTpcXLc24fTpHAXAR1uOczErv8p9E0II4UqKDgshvJEkbKrApYaNTIkS\nQghRjLOGTRUTNgD3Dm5HsMGPQrOVVf87JKM6hRCiGimKIlOihBBeSRI2VaCVKVFCCCFKYSt286/V\nVv3mP9ig44FhHQA4fuYyG386WeXXFEIIYVe8PJiMsBFCeBNJ2FSBTIkSQghRGkf9Gqj6lCiHa9tF\ncHOPZgB8vuMkB46lV8vrCiFEfVf8Pl6jkoSNEMJ7SMKmCmRKlBBCiNJYin0mVMeUKIdxN7clOioY\nBVj1v0OczTBW22sLIUR9ZSuesNFIwkYI4T0kYVMFMsJGCCFEaRz1a6B6EzY6Pw2P3NWVIL0f+YVW\nln14kMtGU7W9vhBC1EfF7+NlSpQQwptIwqYKimfgLVLDRgghRBHXhE313vyHNwxgxp1d0GpUXMjK\nZ9lHB8gvtFTrOYQQoj6xFStiI0WHhRDeRBI2VeAywkamRAkhhChiLp6w0Vb/R237lqFMHt4JgOSz\nOSx5fz95BeZqP48QQtQHLiNspIaNEMKLSMKmCjTqYjVsZEqUEEKIIjVRdPjPenWM4v7b2gNwIu0y\nr7y3n9x8SdoIIURFudSwkRE2QggvIgmbKig+JUqW9RZCCOFQU0WH/+ym2GZMGtYBFXDqXA6vvLuP\nbKlpI4QQFWItNipSatgIIbyJJGyqQK1S4Rg1KVOihBD1XXp6On379uWHH34AIDs7mxkzZtCzZ09u\nuukmPvzwQ+e+iqKwZMkS+vTpw/XXX8+iRYuwWq2e6nq1M9dQ0eHS3NC9KZOHd0KlgpQLuSx4+xeS\nz16u0XMKIeqnPXv2MGbMGHr27MmQIUN47733AN+/3lulho0QwktJwqaKHNOiZEqUEKK+mzt3LllZ\nWc6fn3nmGQwGAzt27GD58uW8+uqr7N+/H4D169ezZcsWPvvsMzZv3szevXtZs2aNp7pe7Yo/rfXT\n1vzNf98ujZk2qgs6rZpLOYW8sG4v2387W+PnFULUH9nZ2UyfPp3777+fX375hWXLlrF06VJ27Njh\n89d7m6wSJYTwUlpPd8DXaTQqLFZJ2Agh6rd3330XvV5PkyZNADAajXz77bd89dVX+Pv7061bN4YP\nH86nn35KbGwsGzduZOLEiURGRgIwdepUli1bxsMPP+z2OVUqFepaeOzguHmvyE188ae1Op2mVopY\n9u4cReNwA8s/Okh6dgFvfp7EsTPZ3DukHXr/qn3cVyYGdY3EQGIA9TsGaWlpDBw4kBEjRgDQuXNn\nevfuzd69e2v8el/TrC41bOR5thDCe7h9B1dQUEBAQEBN9sUnadUqCnF9miqEEPVJcnIyb731Fh98\n8AF33XUXAKdOnUKr1dKiRQvnfjExMXz99dcAnDhxgrZt27psS05ORlEUVG4mN8LDA93etzqEhAS6\nva/+bC5gH1rfKDy4prpUQlhYEP+cFcbLa/dw8Fg6W/encejkJWaOiaVHh8gqv35FYlBXSQwkBlA/\nY9CxY0deeeUV58/Z2dns2bOH9u3b1/j1vjIJ+som13R+apc6lXVRfU48ukPiUzaJTdlqKjZuJ2xG\njBjB8uXL6dixY7V2wNc55rnKCBshRH1ksVh46qmnmDt3LiEhIc72vLy8Ekn+gIAACgoKAMjPz3fZ\nrtfrsdlsmEwm/P393Tp3Roax1kbYhIQEkpVldBk2X55LWUbAXr8mMzO3JrtXqr+N6coXO0/zybYT\npGfl8+zqn+nXpTF339SaRg31FX69ysSgrpEYSAyg9mMQFhZU4+eojJycHOLi4pyjbN555x2X7dV9\nva9Kgt6d5FqG8coKe+HhQQQbdJU6l6+pj4nHipD4lE1iU7bqjo3bCRuz2VyrTzJ9hUYjNWyEEPXX\nv//9bzp27MjAgQNd2vV6PYWFhS5tBQUFGAwGwH4zX3x7fn4+Wq3W7Zt3sBeyrM26lTab4naB+UKz\nfdSlVqPyUFF6FcN6R9OtTSPe2pzEibTL7Pj9HLuTzjOoR3OG92tFkN6vwq9akRjUVRIDiQHU7xik\npKQQFxdHixYt+Oc//8nx48dr/HpfmQR9RZJrl7LynN9fzs7DXFC3V9uT5Gv5JD5lk9iUraqxKStB\n73bCZvjw4UyaNIk77riDFi1alHhyOm7cuAp3qi5wjrCRKVFCiHpo8+bNXLx4kc2bNwOQm5vLrFmz\nmDx5MmazmbS0NJo2bQrYp045hsW3adOG5ORkunfv7tzWunVrz7yJGmCxFCVstJ6thdCsUSBz7uvJ\nlv1n+OynZC7nmfn6lxS2Hkjjhm5NGHJdCyJDKj7iRghRPx06dIjJkyczcuRIZs+ejVqtJjo6usav\n91VJ0LuTXDObr9zHK0r9Wf21Pice3SHxKZvEpmzVHRu3EzabN29Gr9fz/fffl9imUqnqfcLGIhlG\nIUQ99OWXX7r8PGjQIJ555hluvvlmDh8+zJIlS1i0aBFHjx5l06ZNrFq1CoCRI0fy5ptv0qdPH7Ra\nLStXrmTUqFGeeAs1wlKUxPer4SW93aFWqxjUozl9Ozfm619S+HL3aQpNVr7dk8p3e1KJbdeIG7o3\npUtMWI0vQS6E8F3p6elMnjyZSZMmMWXKFGd7UFAQgwcP9unrvWvRYZlRIITwHm4nbEpL1IhiU6Ik\nwyiEEC4WLlzIs88+y8CBAzEYDDz55JPOJ6zjx48nPT2d0aNHYzabGTFiBJMmTfJwj6uPuegzwZsS\nIHp/LaMGxDCoRzO27DvD93vPkG00se9oOvuOphNs8KNXxyiuax9B2+YNZaUUIeqorKwsjhw5gsVi\nQVFc718HDBhQ5nEfffQRmZmZvP7667z++uvO9vvvv9/nr/eyrLcQwltVaJ3PjIwMPvzwQ06ePMmT\nTz7Jrl27aNeuHe3ataup/nm9K0WHZUqUEEIUT+6HhISwbNmyUvfTaDTEx8cTHx9fW12rVY5pst6U\nsHEINugY0T+G23pHszvpPNsOpHE0NZucPDPf/ZrKd7+mEhigpWvrcDrHhNGuRQiNw2TalBB1wccf\nf8z8+fMxmUrWaFGpVCQlJZV5bFxcHHFxcWVu9+XrvWOEjQpQS81OIYQXcTthk5iYyP3330/btm35\n/fffmTFjBtu3b+fvf/87CQkJ9O3btyb76bVklSghhBB/ZnZMidJ6742/n1ZN/65N6N+1CRey8tl5\n6By7Es9zNiMPY4GFnYnn2Zl4HoCQIH+6tm1EdGQg0VHBNIsIJEBXoWc+QggvsHz5csaOHcvf/vY3\ngoK8cwUqT3CMsJHRNUIIb+P23dYLL7zAxIkTmTlzJtdeey0AixcvJjQ0lFdffZUNGzbUWCe9mUbj\nKDosCRshhBB2jho2Gi8cYVOayBA9I/vHMLJ/DBcu5XHgWAYHj6dz9Ew2JrONrNxCftx/hh//dEyz\niECaRwTROMxARIieiFA9DQx+sqqkEF7q0qVLPPDAA5Ks+RPHg1fHfb0QQngLtxM2hw4dYtGiRSXa\nx40bx7p166q1U77EMcdfRtgIIYRwsFjsnwneUHS4oiJDDdxyvYFbrm+BxWoj5UIux85kc/J8LodO\npHPZaAbgQlY+F7Ly2Xc03eV4fz8NjUICCA3yJyTIn4ZBOkKC/AkJ0tEwyJ9ggx+BAX4Y/LXyNFuI\nWtavXz927NjB2LFjPd0Vr2IrquUjBYeFEN7G7YRNw4YNSUtLIzo62qX90KFDhIWFVXvHfIXUsBFC\nCN+iTj4BgBISghIadtU2VXgohAW57tewIUpYeJlthrTTNM46S3DklY9Z9clkUBTXY8tra9AQJbwa\n2k6dBJvN/bbgBiiNGgGgSz1NG5uNdm0aEDqsExkZOeQc+oNz6UZO56s4btJx5qIR5eRJTIUm8nQG\nLhsacuaiEfOxE6QrNmcbQGT2edTF2vT+GlrmZRCoU6NqEAwRkQT4a4jIOo9ODargYGwREfj7aQhJ\nP4u/xt6mjopCq1VjOJeKRlHsx0ZGolWr8D97Bq1iRd2gAaqoSDRqFZrUFLBYUAKDUCIj7e835XSF\n2lQNgq/8HaSmgNnsul9F2wyBKFFRZbedSQWTqUptGAzYohpXvS3tDBQWogoKhLC2Lm2l7Ydej61x\nk4q1nU2DgoKqtQUEYGvStGJt585Cfr7bbSp9AIS1d93P3x9b02bltjn66mmdO3dm8eLFfP/998TE\nxODn5+eyfdasWR7qmWc57uOlfo0Qwtu4nbC59957mTdvHk888QQAR44cYdu2baxYscLtSu+JiYnM\nmzePY8eOER0dzXPPPUdsbKzLPoqisHz5cj788EOMRiNdunRh3rx5XlvYWKZECSGEbwnvbf/cMT4+\nm7zZc13bZj1F3tP/B0BY3x6obDbyH38KXn3J3tavJyqrFWP8E+T9fZ69rf91qCwW8h57HOPcZwG4\n/8nRTLJa2HbrffDgjfb9buiFqrCQvJnxGJ95zt52Y29UBQXkzXgM47ML7W0D+6DKzydv+qMY59tH\ntobd1A9VnpG8aTMxPrcYgNCb+6M25pIX9wjGBc/b2wYNQJ2bQ97U6RgXvnilLecyeVOmYVxkfx+h\nQ25EnZ1F3sNxGBe/bG+75UbUWVnkPzSF3BdetbcNHYj60iUKHpoCb6xEpVLR8s6htMrMpPukyeS+\ntNQev47jUGekc37M/ey+5//IyC5gzIQHCbycyY6+I1h92wyyjSZefXc2oXlZfNl1KK/dMp38Qit/\nfyOecOMlvux6C6/dMgOAt1c+SLgxk6+7DGHF0EcAeGvVQzTKzeDrzoNZcetMANaseoiI3Ay+6TyY\n5UVtb65+mMici3zbeRDLbn3U3vbGFCIvX2Brl0G8PnIWKpWKZf96kMjs8/zUbTCr73oCtUrFS/+c\nROSls/zcfTBvj3salQoWvDqRyIw0frl2MO9PnIfVamPOC/cRkX6GvT0G898JzwAw+/n7iLiYyr5r\nB/HuA/a/gycWTyDywmkO9ChqU6mYteg+Is+f5uC1N/PuQ8+hQsXfFt1H5LlT/HbtTbw/2f53MHPR\n/USdTebQtTfx/hT738GMhfcTlXaCxGsH8sHU50EF0xbcT9SZ4yTF3sgH019EBUxZcD+NU49xuPsN\nfDDD/jt/eOFEmqQc5Uj3Abw/4+WitgdokvIHf3Trz3uPvALA5EWTaHr6CH907cd7M+1/B5MXP0jT\nU4c52rUvq59cjslkZtKih2h2MomjXfry3mNLAHjw+ck0S07kWOfevPu3fwAw6fmHaZ58iOOdevHf\n+H8C8MCLU2hx/HeOd7qe/8YvK2qbSovjv3Gi43Wsn7UcgIkvxdHy2EGSO/Rk3eMritqm0fLYAZLb\n92DdE/+y///2ynSi/9jPyfY9WFvUNuGVGbT6Yx+nroll7VP2FY3ue2UGrY7s5XS7WN6ZbW/766uP\nEHP4V0636847sxPsbUtmEpO0h5S23fjP0ysBGL/kUVon/cKZtl3J+GEbYQYtwTPj0G39AXPP68n6\n4jsAgh+bju6H7zD3vI6sL+xF2IP/NoPs9z7GG+zatYtu3bphNBr5/fffXbbV56mMjho2MsJGCOFt\n3E7YTJkyhcDAQF588UXy8/N55JFHaNSoEXFxcTzwwANXPb6wsNBZXX7MmDFs3LiRadOm8e233xIY\nGOjc76OPPuKrr75iw4YNREREsGLFCp566ik++eSTSr3BmqZ1TIlSJGEjhBDCVb25+S96mw0C/ejT\nyT7aQq/TAHBtuwhentYPRVFo8JYO8qDHNRHMGtedvAILhre1YIQWEUHc0K0JBSYrflr7Z2uwQUez\niEAKTVYcoaxMRBXsn9EWm4KxwGJvK/rcNpmtZF4uBK48ZS8wWUhLN9qPsdjbcvMtHDl9CQBzUVuO\n0cSRlKyi17G3Xc4zk3TqkvO1AbKNJg6dtLcVOvYzFvL7icyi8xXtl2viwPEMl7as3ELntLP8Qnvf\ns3IK+fWPiwDkFWvbc/gCABOK3mNWbiG/FLX9tajtUq7pSlthyf3GF5idfXG03VvUlpVr4sf9ZwAY\nm28p2q+Q3UkXitquHOtoG+NoM15pG51nLorBlba780tpK2W/u/JNzjg72u40ltzvL3mmot+RmV1F\nxbNHGh3HmpxtI4racoq1DXe2XTn2DsexRhMbvz/KlOEd8UVr1671dBe8klWKDgshvJRKUdzLNKSl\npdG4cWPUajV5eXlYrVaCg4OxWq0kJibStWvXco/funUrzz77LFu2bHG2jRgxgmnTpnH77bc72xRF\nIT8/H4PBQG5uLv/85z85ceIEa9ascesNXbyY49Z+f6bRqAgLCyIzM7dCo2Ve+/g3fv3jIjd2b8oD\nwzpU6tzerrKxqQ8kNuWT+JStKrGJiAiuoV75lspe7ys6JUodHkpom5ZkZuaiHDtu3+8qU6LeX/0l\nB45n0Dm2NX+9x76Koi9OiXK0qUMaEHpNjD0GJ5JL7nf6FFitVWsLCkaJiLhqmy0wCHNYOBarAidP\nYjVbMOsNmEIbYbHaUKekYDWbMAcEUtAwDItNQXsmBcVkxhRgoCA0HMUG/udSwWLBHGAgPyQcRYGA\ns6lgtWD2N5DXMBwFBf25M6gsVsx6AzRujDGvEP35NFQWCyb/omNRCEo/i9pswVT0eigQmJ6Guth+\nAIEXz6KxWjD5B5DX0B6DoPRzqK0WzLoAjI79Ms6hsTja7PsFp59DZTFj9g/AWHRscMY51BYzZp2e\n3KJjgzPOo7GaXY+tapvFjMU/AEtUEwoLzQQVtbnsl1m0n58/uaERbrXlhBbF4NIFtGaTy34Vb9OR\nG2qfehaUWUpbaftduojWXOh2GwH+DB19AyF6DcqZtBqfElUd1/uffvqJPn36oNVq+emnn8rcT6VS\n0b9//yqfryZU5npfkc/ZLfvP8M6XRwhv4M8r070zBtVJ7s/KJ/Epm8SmbFWNTVnXe7dH2AwePJjt\n27cTFhaGwWBwtp86dYr77ruPAwcOlHt8cnIybdq0cWmLiYnhxIkTLm0qlQqDwcDHH3/MnDlzCAoK\ncjtZ4zheXYkaj46MekUz69qiJVttilJnK8tXNjb1gcSmfBKfsklsPMcW07pCbapi13Z3j00Pb8a5\nDB3XNAy9sl+rmJLH1kZbdKsqt7nEoLT9WkbXapsf4KcF2rctsR8RpYx8aB1esq1L45Jt1zYr2Yb9\nvK43YiV/51BKX0ptK22K9zWltLV3s620h0Wljf6oeptrDKr7HJ081FYxxWNQWhLG3bbaNHnyZLZv\n3054eDiTJ08ucz+VSkVSUlIt9sx7yLLeQghvVW7C5t133+W1114D7CNfRowYUWJ+q9FodKu+TF5e\nHnq93qUtICCAgoKCUvcfPnw4w4cPZ+3atUyePJmvv/6akJCQq54nPDywSnNwQ0ICr75TMXq9DgCt\nVkNYWN1eIrGisalPJDblk/iUzRtic+nSJVatWsXvv/+O2Wwusf29997zQK98m7loWW9tHU3kCyF8\nx+HDh0v9XlxxZUqU763sJ4So28pN2IwePRq9Xo/NZmPOnDlMnTqV4OArQ3Uco2H69Olz1RPp9foS\nyZmCggKX0TrF6XT2RMhDDz3E+vXr2b17N0OHDr3qeTIyjJUeYRMSEkhWltGZZXeHtWh+el6+iczM\n3Iqf2AdUNjb1gcSmfBKfslUlNtWdHH766af57bffGDlyJEFBdTvxXFsszoSN3PwLIbyPoiilJugd\n99/1jeNzWCsjbIQQXqbchI2fnx9/+ctfAGjevDk9evRAq3V7FpWL1q1bs27dOpe25ORkhg8f7tK2\nfPlyrFYr8fHxgP0DxWQyuSSKyqMoClZrpboI2C/YFZlz5hg6abFW7DhfVNHY1CcSm/JJfMrmDbHZ\nuXMn77zzDt27d/doP+oSR6FaR/FcIYTwBrt27WLBggWcPHkSW1GRbbDfP8uUKJkSJYTwPuVmX5Yu\nXcq0adPQ6/X89NNP5RYqmzVrVrkn6tu3LyaTibVr13LPPfewceNG0tPTGTBggMt+3bt358knn2TY\nsGG0bt2alStXEhQURI8ePSrwtmqPYwUQq9V2lT2FEMI7hYeH4+/v7+lu1CnmoiScjLARQniTefPm\n0bZtW2bPnk1AQICnu+M1ZJUoIYS3Kjdhs2/fPsxmM3q9nn379pW5nzs1Y3Q6HatXr2b+/PksXbqU\n6OhoXn/9dQwGA/PmzQNgwYIFDBw4kFmzZjFjxgxycnK49tpreeONN7z2HxOOC7tVpnsIIXzUI488\nwsKFC5k7dy7R0dH4+fm5bK+vQ+SrwiI1bIQQXujChQskJCQQE1OyQHl95hhho5GEjRDCy5SbsFm7\ndm2p31dWhw4dSi1euWDBApef77nnHu65554qn682aIsK5kjCRgjhq5YuXUpWVhZ33313qdvr6xD5\nqnAkbPxkhI0QwovccsstbN26VRI2fyIjbIQQ3qrchE15U6CKU6lU9O/fv1o65GscS3nLlCghhK9a\nunSpp7tQ55iLathopYaNEMKLzJo1i5EjR7Jp0yZatGhRYlWkJUuWeKhnnuVI2GiqsNKsEELUhHIT\nNpMnT3brRepzkTKNTIkSQvi4Xr16eboLdY7jM0Fq2AghvMncuXNRqVQ0b95catgUI0WHhRDeqtyE\nTVJSklv1aeozR8LGIgkbIYQPueeee1i1ahUNGjRg3Lhx5V7rS5vKKsrnHGEjNWyEEF5kz549rFu3\njq5du3q6K17FKjVshBBeqtyETb9+/bj++uvp378//fr1o0WLFrXVL5+hKXp66ulleYUQoiIGEFhD\nBgAAIABJREFUDBjgLC58ww03eLg3dc+VosMywkYI4T2io6MxmUye7obXkRE2QghvVW7C5quvvmLn\nzp1s376dN954A5vNRv/+/enbty99+/YlJCSktvrpta5MiZIaNkII3/HII484vx8+fDitWrXyXGfq\nICk6LITwRtOmTePpp59mwoQJtGzZEq3W9Z8CAwYM8FDPPMuqyAgbIYR3Kjdh06BBA4YOHcrQoUMB\nSE1NZceOHXz55ZcsWLCAJk2a0K9fP+dXfSQ1bIQQvm7YsGF06tSJESNGcPvttxMZGenpLvk0s8WG\npWjUpU6n8XBvhBDiivj4eACef/75Etvqc01KW9GDV0nYCCG8TbkJmz9r3rw5Y8eOZezYsSiKwqFD\nh9ixYwerVq2qvwkbmRIlhPBx3377LV988QWbNm3ilVdeoUePHowYMYJbb72Vhg0buvUamzdvZsWK\nFZw7d46mTZsSHx9Pp06duOOOO1z2M5lMNG/enK+++gpFUejZsyeKcuX62bNnT954441qfX+1LSu3\n0Pl9SJC/B3sihBCuDh8+XC2vc/DgQaZPn+5cUfa3335j7NixLoWMp06dSlxcHIqisHTpUj788EOs\nViujRo3i73//OxqN9yS0ZVlvIYS3qlDCJjMzE4PBQEBAAImJiWzdupUuXbowZcqUmuqf15MpUUII\nX9esWTMmT57M5MmTSUlJ4auvvmLDhg0sWrSI/v378/rrr5d7fHJyMnPmzGHNmjX06NGDHTt2MGXK\nFLZt28a+ffuc+128eJE777yTuXPnAnDq1CkA9u7dW6cK3F/KuZKwCZWEjRDCw0aNGkW/fv3o378/\nvXr1QqfTVfq1FEVhw4YNvPjiiy4Jl6SkJG688UZWrlxZ4pj169ezZcsWPvvsM1QqFVOnTmXNmjU8\n/PDDle5HdbNJ0WEhhJdye3L9t99+y0033cTevXs5deoU9913H5s3b+Zvf/sba9eurck+ejWZEiWE\nqEtCQ0OJiIggKioKjUbDhQsXrnpMTEwM27dvp0ePHlgsFtLT0wkMDCzxj4Jnn32WYcOGceONNwKQ\nmJhI+/bt61SyBq6MsPH306D3954nyEKI+unll18mMjKSt99+mwEDBnD//fezcuVKDh486DLC0R0J\nCQm88847xMXFubQnJibSoUOHUo/ZuHEjEydOJDIykoiICKZOnconn3xS6fdTE2SEjRDCW7k9wmbZ\nsmXMnDmTfv36sWTJEpo2bcrnn3/O999/z+LFi5kwYUJN9tNraYqWbJUpUUIIX5Wbm8u3337Ll19+\nyY4dO2jatCnDhw8nPj6emJgYt14jMDCQlJQUbr31Vmw2G/PnzycoKMi5/eeff2bv3r288sorzrak\npCRyc3MZNWoUFy5c4Prrr2fu3LlERUW5dU6VSoW6Fmr6Om7g3b2RzzbaV2AJDfZHq60bRYcrGoO6\nSGIgMQDfjEH79u1p3749kyZNwmQysW/fPnbs2MGCBQtITU2lV69e9O3bl379+hEdHV3ua919993E\nxcWxe/dul/akpCR0Oh2DBg3CZrMxbNgw4uPj0el0nDhxgrZt2zr3jYmJITk5GUVR3E7YV+Z6X5Hf\nla0ocaXVqJ339nWZL/4d1yaJT9kkNmWrqdi4nbA5efIkw4cPB+CHH35g8ODBgP1DID09vVo75Uu0\nRZ8eMsJGCOGr+vbtS2hoKMOGDWPGjBl07dq1Uq/TpEkTDhw4wJ49e5g+fTrR0dH07dsXgFWrVvHg\ngw8SGBjo3F+n0xEbG8tjjz2Gv78/ixcvZubMmXzwwQdunS88PLBWR+eEhARefScg32yfIhsZZiAs\nLOgqe/sWd2NQl0kMJAbguzHQ6XT07t2b3r17Ex8fT3Z2tnNF2DVr1vDNN9+Ue3xZRelDQ0Pp3bs3\n48aNIyMjg8cee4zly5fzxBNPkJ+f71LbRq/XY7PZMJlM+Pu7N220Ktd7d35XjuldBr2uzl23y+Or\nf8e1ReJTNolN2ao7Nm4nbKKiokhMTOTSpUscO3aM5557DoAtW7bQvHnzau2UL5EaNkIIX7dq1Sr6\n9OlT5eSHY3nYvn37MnToUL777jv69u3L2bNn+eWXX1iyZInL/jNnznT5efbs2fTp04cLFy64tVJV\nRoax1kbYhIQEkpVldNY5KM/ZizkABOm1ZGbm1nT3akVFY1AXSQwkBlD7MaiJ5IHNZkNRFDQaDXl5\neeTn53PfffexYMGCSr9mQkKC83uDwcDUqVNZunQpTzzxBAEBARQWXqntlZ+fj1ardTtZA5W73lfk\nd1VQaAbAbLbUmet2eeT/5fJJfMomsSlbVWNT1vXe7YTNgw8+yGOPPQZAbGwsPXv25F//+hcJCQm8\n/PLLFe5QXSFTooQQvuj99993+fn06dNl7jtu3LhyX2vr1q289dZbvP322842s9lMcHAwYB+V2atX\nL8LCwlyOW7VqFf3796dz586AfQUpwO2beEVRsFrd2rVa2GyKW9f6zMv2f5iEBPrXuc8Gd2NQl0kM\nJAbguzHYs2cPjz76KEuWLKFNmzaMHj0ak8lEfn4+L7/8MrfffnuFXzM7O5uEhARmzJjhnApbWFjo\nvJa3adOG5ORkunfvDtgL1bdu3bpC56jK9d6d35Vju1ql8snfa2X56t9xbZH4lE1iU7bqjo3bCZvx\n48cTGxtLWloaAwYMAGDAgAEMGTKkzCJj9YGmKN2vYP/lyHw+IYQvKG0lj9KoVKqrJmw6derE77//\nzqeffsrIkSP58ccf2bp1q3Nq04EDB4iNjS1x3IkTJ/jxxx9Zvnw5Wq2WxYsXM3jwYLeXEvdWjlWi\nQoNlhSghhHd56aWXuP3224mNjeWtt97C39+fH374gU2bNvGvf/2rUgmb4OBgvvnmGxRF4fHHHyct\nLY2EhATGjh0LwMiRI3nzzTfp06cPWq2WlStXMmrUqOp+a1UiRYeFEN6qQst6d+rUiYKCAj7//HNu\nvfVWAgMDr1qcrK4rvvyf1WZDrZYVQYQQ3u/777+vtteKiIggISGB559/ngULFtCqVStee+012rRp\nA8CZM2dKTdj83//9H4sXL2bYsGGYzWZuuukmFi5cWG398gRFUcjKtY8UCpElvYUQXubIkSMsW7YM\nvV7P999/z5AhQ5x1bebPn1+p11Sr1SQkJLBo0SL69OlDQEAA48aNY+LEiYD9oW96ejqjR4/GbDYz\nYsQIJk2aVI3vqupkWW8hhLdyO2GTmZnJtGnTOHToEDabjV69erFkyRKOHz/OmjVraNGiRU3202sV\nryRvsSr4VSgFJoQQ3iEjI4MPP/yQkydP8uSTT7Jr1y7atWtHu3bt3Dr+uuuu4+OPPy5127p160pt\nDwoK4oUXXqh0n71Rbr4Zi9Ve00xG2AghvE1ISAhnzpzBarVy6NAh4uPjATh48KBbtcMcevfuza5d\nu5w/t23b1mVabHEajYb4+HjnubyRVbkyJUoIIbyJ2+W7Fi9eTHh4OLt27XLOSX3ppZdo2bIlixcv\nrrEOejtNsQposlKUEMIXJSYmcuutt7JlyxY2bdpEXl4e27dvZ/To0fz888+e7p5PcUyHAknYCCG8\nz5gxY5g+fTrjxo1zruS3du1aZs+e7RwRUx/JCBshhLdyezzIjh07+M9//uOyJGvDhg15+umnuffe\ne2ukc77AdUqUJGyEEL7nhRdeYOLEicycOZNrr70WsCfpQ0NDefXVV9mwYYOHe+g7snLtCRuVChoE\n+nm4N0II4WrmzJl07NiR1NRUhg8fjlqtpmXLlixfvpybbrrJ093zGGfRYUnYCCG8jNsjbKxWK7ZS\nlq7OyclBo6m/dVuKT4myWmVpbyGE7zl06BAjR44s0T5u3DiOHz/ugR75LscIm4aBOpcRmEII4Q3G\njh1Ly5YteeCBB2jUqBEAAwcOrNfJGrjy0FVG2AghvI3bd5NDhgzhlVdeITMzE1XR/M5jx46xcOFC\nBg8eXGMd9HYywkYI4esaNmxIWlpaifZDhw6VWIpblE9WiBJCeLPU1FS0Wim4+Gc2RUbYCCG8k9tX\n7Dlz5jB37lz69++PoiiMGDGCwsJCbr75ZubMmVOTffRqGo3UsBFC+LZ7772XefPm8cQTTwD2VUS2\nbdvGihUrvG4lD2+XcbkAgNDgAA/3RAghSrrnnnuYMWMG48aNo3nz5gQEuF6rBgwY4KGeeZYs6y2E\n8FZuJ2yCgoJYtmwZKSkpHD9+HIvFQps2bYiJianJ/nk9rVqmRAkhfNuUKVMIDAzkxRdfJD8/n0ce\neYRGjRoRFxdXr4tQVkbKhVwAmjYyeLgnQghR0r///W8AXnzxxRLbVCoVSUlJtd0lr+Ao+6CVhI0Q\nwsuUm7BJTk4utT06OrrEPvU1cSNTooQQvshms6EuVmPlr3/9K3/961/Jy8vDarUSHBwMwMWLF4mI\niPBUN32KxWojLd0IQMvIYA/3RgghSjp8+LCnu+CVbDLCRgjhpcpN2AwbNgyVSoWilJ6IcGyrzxl5\nmRIlhPBFjz32GP/4xz9K1DIwGK6MDNm8eTPPPfccu3btqu3u+aS0dCOWopVGWjYuP2GTkpPGqcun\nub5xD/w1utronhBCAGAymfjqq684efIkEyZM4MiRI7Rp08ZZhLg+kilRQghvVW7C5rvvvqutfvgs\nlxE2VknYCCF8w+7du5kxYwYrVqxAp3NNGGRnZzN//ny++OIL+vXr56Ee+p7T5+3TofT+WiIall3D\nZkfabt478glWxcrXp7ZwX8cxXBPapra6KYSox1JSUpg4cSJWq5X09HT+8pe/sH79enbt2sVbb71F\np06dPN1Fj7DJKlFCCC9V7ipRzZo1K/FlNBo5cOAA+/btIzs729leX7lOiZIaNkII3/DOO++QmJjI\n1KlTyc/Pd7Zv2bKFO+64g59++omFCxeyZs0aD/bSt5w+nwNAy8gg52qKf7b73F7WH/4Iq2IFIKMg\nk38feJMLeRdrrZ9CiPpr8eLFDBgwgB9++MGZrF+6dCk33XQTL7zwgod75zkywkYI4a3cXtb78uXL\nxMXFMXLkSJ577jnmz5/PXXfdxfjx48nJyXHrNRITExk9ejSxsbGMGjWK/fv3l7rfBx98wNChQ+nR\nowd33303e/bscbebtU6juXJht8iUKCGEj2jfvj3//e9/SUlJ4aGHHuL8+fPMmTOHuLg4unbtyqZN\nmxgzZoynu+lTHAmbFlFBpW632qxsOvEVAG0atiK+xzQa6oIx2yy8e+STMqcfCyFEdfn111954IEH\nXGqYabVapk2bxu+//+7BnnmW1TnCxu1/GgkhRK1w+6q0ePFiLly4wOeff86uXbvYs2cP//vf/ygo\nKODll1++6vGFhYXExcVx11138csvvzBhwgSmTZuG0Wh02W/nzp0sXbqUZcuWsWfPHu677z7i4uK4\ndOlSxd9dLVCrZEqUEMI3tWjRgvXr13P58mUGDRrEli1bePXVV3n99deJiorydPd8ik1ROF20QlR0\nVOn1a3af30dGgf2z7J72d9E2JIYx1/wFgD8uHWPnuV9rp7NCiHpLp9Nx+fLlEu2pqakEBgZ6oEfe\nwVl0uIzRkUII4SluJ2y+//57nn32Wdq0uTLPvl27dsybN4+vv/76qsfv3LkTtVrN+PHj8fPzY/To\n0TRq1IitW7e67Hfu3DkeeughOnbsiFqt5s4770Sj0XDs2LEKvK3ao1KpnNOiZEqUEMLXREVFsX79\nejp37kxERITUrKmki1n5FJjs05xalpKwsSk2vj75PQCxEV1oGtTY+X23Rp0B+Oz4FxRYCmqpx0KI\n+mjkyJEsXLjQOZomOzubrVu3Mm/ePIYPH+7h3nmOVWrYCCG8VLlFh1121Grx9/cv0R4QEIDZbL7q\n8cnJyS7JHrAvBX7ixAmXtr/85S8uP//6668YjcYSx5ZFpVJRmdGMjjmrlZm7qtGosNoUFFynSNUV\nVYlNXSexKZ/Ep2yejs1PP/3k8vNDDz3EwoULmTBhAk899RQajca5bcCAAbXdPZ+TdNI+csZfp6FJ\nuKHE9r3nD3AhPx2A21oNdrarVCrubjecxIzDXDbl8M2pLYxoc1vtdFoIUe88/vjjLF26lPHjx2My\nmRg9ejRarZZ7772XWbNmebp7HuMsOlwH7+OFEL7N7YRNv379ePnll1m6dCkhISEAZGZm8sorr7j1\nRDYvLw+9Xu/SFhAQQEFB2U8Tjx07xqOPPsqjjz5KWFiYW/0MDw8ss9ijO0JCKj4c1E+jxmS2odfr\nCAsrvXZBXVCZ2NQXEpvySXzK5qnYTJ48udT29PR0pk6d6vxZpVKRlJRUW93yWfuP2ZMxXVqFodW4\nPjWwKTa+PGUfXdMlvCMtgl0L9TfSh3Nzixv45vQWvkvZRt+mvWikd+8zTwghKkKr1fLUU0/x2GOP\ncfr0aaxWKy1btsRgMJCRkUF4eLinu1jrbIr9oSvIAyYhhPdxO2Hz9NNPM3HiRAYOHEjz5s0B+3zX\nNm3a8Pzzz1/1eL1eXyI5U1BQgMFQ8kkk2J/+xsfHM2nSJKZMmeJuN8nIMFZ6hE1ISCBZWUZnlt1d\njgRR9uV8MjNzK35yL1eV2NR1EpvySXzKVpXYVEdi+PDhw1V+DWFXaLaSdMo+wqZ720Ylth+4eIiz\nxvMADIsZXGI7wK2tbmbn2T3kmHNZf/gjZsZORq2S4pdCiOrVsWNHtm/fTlhYGO3atXO2p6SkMHLk\nSPbt2+fB3nlG8c9gjdSwEUJ4GbcTNhEREXz22Wds27aNEydO4O/vT+vWrenXr59bI1pat27NunXr\nXNqSk5NLnS+7YcMGFi9ezIIFCyo8n1ZRFKzWCh3iwmZTKlw82DHf1Wy21enCw5WJTX0hsSmfxKds\nEhvfl3gyE7PFhgro1qbk0+kdZ3cD0CG0Ha0atCz1NfRaPePa38kbv6/lj0vH+OnMTm5sLvWEhBBV\nt2HDBj788EPAfp/88MMP4+fn57LPxYsXiYyM9ET3PM5aLGEjI2yEEN7G7YQN2IdR3nzzzS71DBz1\na3Q6XbnH9u3bF5PJxNq1a7nnnnvYuHEj6enpJWoj/Pzzzzz33HOsWbOG6667riLd8xjHfFerjCAQ\nQoh650DRdKjWzRrQILDkZ2FmQRYAHcLaldhW3LWRXekR2Y29Fw7y0dH/EWmIuOoxQghxNcOGDePs\n2bMA7N+/n169epVYESowMJChQ4d6onse5zLCRhI2Qggv43bCZvfu3Tz33HOcPHkSW7HVkBRFcavG\ngU6nY/Xq1cyfP5+lS5cSHR3N66+/jsFgYN68eQAsWLCA1atXYzabefjhh12OX7ZsGTfeeGNF3lut\nubJKlCRshBCiPik0Wfn1yEUAYkuZDgWQY8oBoIGu9OW+ixvX/k5Sc9K4kJ/Oqt/+w7Ruk2gX6l7R\nfSGEKI3BYOCRRx4BoFmzZtxxxx1XfdBan8gIGyGEN3M7YfPMM8/Qtm1bZs+eTUBAQKVO1qFDB957\n770S7QsWLHB+v2bNmkq9tidpiormWK2yrLcQQtQnPx5Mw1hgQaNW0a9LkxLbrTYrRnMeAMG6q9ce\nCvIL5JHYySz59TWyTTks37+aka1vY1CLG9CoNVc9Xggh/uz999/nzjvvRKfTYTKZ+OSTT8rcd9y4\ncbXYM+9glRE2Qggv5nbC5sKFCyQkJBATE1OT/fFJzilRioywEULUT5s3b2bFihWcO3eOpk2bEh8f\nz5AhQ/jtt98YO3asS6J/6tSpxMXFoSgKS5cu5cMPP8RqtTJq1Cj+/ve/uywp7s2sNhtf/5ICQJ/O\nUYQG+5fYJ8d8pRB9sBsjbADC9WE8du1UVv32DufyLvDp8c3sPPcrI1vfRtdGHaUYsRCiQlauXMnQ\noUPR6XSsXLmyzP1UKpXbCZuDBw8yffp0fvrpJwCys7OZM2cOO3fuJDg4mBkzZjBmzBgAr7/W22SE\njRDCi7mdsLnlllvYunWrJGxK4ZwSJYVDhRD1UHJyMnPmzGHNmjX06NGDHTt2MGXKFLZt20ZSUhI3\n3nhjqf9IWL9+PVu2bOGzzz5DpVIxdepU1qxZU2JKrLfaeeg86dn21Q9vub45G49/AcCgFjc4R9Pk\nmIzO/YN17i/hHhUYyZPXzeTDoxvZeXYP54znWfXbf2gSGMXQ6JvpGdldRtwIIdzyxhtvkJWVRVZW\nFm+++WaVXktRFDZs2MCLL77oknB55plnMBgM7NixgyNHjvDwww/Trl07YmNjvf5aby1W6kFG2Agh\nvI3bCZtZs2YxcuRINm3aRIsWLVD/ae3sJUuWVHvnfIVzSpTUsBFC1EMxMTFs376dwMBALBYL6enp\nBAYGotPpSExMpEOHDqUet3HjRiZOnOhcmWTq1KksW7bMa27iHY6dySbraAbXtgkF7DfzGdkF/Pfb\no4C9ds1F5QRfn/oBgC2p23mw83i6NurkrF8DEOxXseXYA7T+TOg4lhua9WHj8S/549IxzhrP85/E\n99h04muGtBxI3ybX4afxu/qLCSHqrdtvvx2VSuWsO1kad2tSJiQk8MUXXxAXF8fq1asBMBqNfPvt\nt3z11Vf4+/vTrVs3hg8fzqeffkpsbKzXX+ul6LAQwpu5nbCZO3cuKpWK5s2bV7qGTV11ZZUoqWEj\nhKifAgMDSUlJ4dZbb8VmszF//nyCgoJISkpCp9MxaNAgbDYbw4YNIz4+Hp1Ox4kTJ2jbtq3zNWJi\nYkhOTi73HxXFqVQq1LUwO2j1Z4mcy8zj5h7NmHhbe/IKLazedIj8QguBAVomDmvPeyfede5vsprY\nnraL2KjOGC32KVGBfgZ0fhVamNGpTWg0s66bSnL2ab5M/p4DFw+RUZDJ+398whcnv2FQyxu4oXkf\nAv0M1fJ+S+OYJlCfpwtIDCQG4Jsx+O6776rtte6++27i4uLYvXu3s+3UqVNotVpatGjhbIuJieHr\nr78GqPK1Hip3vXf7d1Vss5+f2nlfX5f54t9xbZL4lE1iU7aaio3bd4979uxh3bp1dO3atVo7UBdo\nZUqUEELQpEkTDhw4wJ49e5g+fTrR0dGEhobSu3dvxo0bR0ZGBo899hjLly/niSeeID8/3+UBgF6v\nx2azYTKZ8PcvWQ/mz8LDA92+2a+KG3s054Nv/+CHvWfIybdw+txlLlzKB2Dm2Gtp3ETPoV+OANA0\nOIq0nPNkmy8TFhaE5YIJgFB9Q8LCKjbC5s/CwjrRM6YTKdlpbEz6mp9O/8JlUy6fHvuCL5K/48ZW\nvRl2zc00b1Cy+HF1CQlxf1pXXSUxkBiAb8WgWbNm1fZajlEyxeXl5ZV4mBsQEEBBgX3KaFWv9VC1\n6/3Vflc5pisPXMPDgghrqK/UeXyRL/0de4LEp2wSm7JVd2zcTthER0djMpmq9eR1hSzrLYQQoNXa\nP1L69u3L0KFD+e6770hISHBuNxgMTJ06laVLl/LEE08QEBBAYWGhc3t+fj5ardbtG/iMDGOtjLC5\no08LUi/ksOPgWfYknQdAq1Exfsg1dGzRgO+O/IzVZsVPreWmZv357+GPuWDMIDMzl3NZGQAEagxk\nZuaWdxq3BdKA8deMZmiLQXxzcgs/p+2h0Grim+M/8s3xH2kT0oo+Ta7juqhu6P2q5x8earWKkJBA\nsrKMLtMH6hOJgcQAaj8GVU301ga9Xu9yLQcoKCjAYLCP+qvqtR4qd71393d16dKVWmOXs/NQWa0V\nO5EPkv+XyyfxKZvEpmxVjU1Z13u3EzbTpk3j6aefZsKECbRs2dJ5Y+4wYMCACneqrtBoHMt6yx+t\nEKL+2bp1K2+99RZvv/22s81sNqMoCi+99BIzZswgKMj+IVRYWOi8SW/Tpg3Jycl0794dsBcvbt26\ntdvnVRSF2riv1mhUzBrfk2bhSaReyEWjVjO4Z3OiGwdjtSrsPrsPgC6NOtHY0BiAAksBOQV5XC60\nJ2mC/IKq/TMiVBfK2GvuZHjMrWxP283W1B1cKszieNZJjmed5IMjn9IprD1dIzrTNbwjQRUoelwW\nm02p9591EgOJAUgMiouOjsZsNpOWlkbTpk0B+/XcMQ2qqtd6qNr1/mq/K5O5eEkDVb36vcrfcfkk\nPmWT2JStumPjdsImPj4egOeff77ENneKlNVlV0bYSA0bIUT906lTJ37//Xc+/fRTRo4cyY8//sjW\nrVt5//33nct3P/7446SlpZGQkMDYsWMBGDlyJG+++SZ9+vRBq9WycuVKRo0a5eF3Uzp/Pw3D+7Uq\n8QF8qSCLo1knALg+KpawgBDntsyCS+SY7AmbBm4u6V0ZBj8Dt0TfxKAWN5CU+Qc7z/3KbxcPYbZZ\nOJB+iAPph1ChonXDVnSL6ESnsPY0CYyqlelkQoi6LygoiMGDB7NkyRIWLVrE0aNH2bRpE6tWrQK8\n/1rvsqy3XBeFEF7G7YTN4cOHa7IfPs2RsLHIsDAhRD0UERFBQkICzz//PAsWLKBVq1a89tprtG3b\nloSEBBYtWkSfPn0ICAhg3LhxTJw4EYDx48eTnp7O6NGjMZvNjBgxgkmTJnn43VTMnvP7AdBr9XQK\n74BGpUatUmNTbGQWXOJy0SpRjmW+a5JGraFLo450adSRPHMe+y7+xsGLiRy5dBSzzcLx7GSOZyfz\nCZ8TrAuifWhb2oe2o31oW8L1oTXePyFE3bVw4UKeffZZBg4ciMFg4Mknn3SOqPH2a71VVokSQnix\nyi1ZIVzIlCghRH133XXX8fHHH5dob9u2rctUqeI0Gg3x8fHOEZy+6Jfz9ulQPSK74qe2f6SG+oeQ\nUZBJZkGWc4RNbSRsijP4GejftDf9m/am0GoiKfMPDl48xKGMw+SajeSYctlzfr8z4RTi35CYhtG0\nbtCSmIbRNA9u5nw/QgjxZ71792bXrl3On0NCQli2bFmp+3r7td5lhI0kbIQQXkbuxqqBTIkSQoj6\nJy33HGdyzwJwXdS1zvawAHvCJqMgk1yzvZhlbSdsivPX6IiN6EJsRBdsio2zxvMczjzc5VojAAAg\nAElEQVTK4UtHOZaVjMlqIqswm30XDrLvwkEANCoNjQMjaRbUhGZBTWjRoCmd9W1QlFqo8iyEELXI\nqhQbYVMPlvQWQvgWSdhUA1klSggh6p99F38D7KNT2obEONvDAuzTi1JyzmBT7In8mqxhUxFqldqZ\nhBnc8kYsNgunc86QnH2K5OxTnMg+RbbpMlbFypncs86EFAB7wV/jT4Q+3P5laESEvhGRhkaE+jek\noX8DtDIqRwjhYxwjbFRIDRshhPeRO6tqIFOihBCi/knKOAJAt0adUKuujDxxJGxO5aQ62zw5wqY8\nWrWW1g2jad0wGrCvxHKpMIvTl1OdCZvU3LNkFGQCUGgtJDU3jdTctFJfL9gviBD/BoQENKShf0Ma\n6oIJ9Ask0M9w5UsbSJAuEJ3aTwofCyE8zvHAVaZDCSG8kSRsqoGMsBFCiPol12zk5OUUADqFt3fZ\n5kjYmKwmZ1uwn3cmbP5MpVIRFhBKWEAosZFdne0mpZACrZFjZ09zzniRi/kZXMxL52J+hnPaF0CO\nOZcccy4pZSR0itOqtRi0egI0/vhrdPhr/fEv+t7e5l/UpsNP7YdWrUWr1uKn0ji/v/Klse9TbJu6\nqAC08wsVapUalUrlkmATQtRvjhE2UnBYCOGNJGFTDaSGjRBC1C9HMo+ioKBRaWgX0sZlW/GlvQH0\n2gD8NH612b1qp9cG0CysEaGElxhNmm/JJ6vwMlkF2WQVun7lmIzkmo0YzXkUWAtcjrPYLFw25XCZ\nnNp8KwCoUDkTN45EjuNLpVKhxvGzChUqKPqvVqMuVqBUhQqKtjl/chk1pPrTPs4WVbFtqLAf4vqz\n46fKvr+KH3T1Y1Qq8NNqMFusOMp+VLaXVXl/FT5XNY7k8tNoubvrbURpmlTbawrPsskIGyGEF5OE\nTTVwFCiTKVFCCFE/JGb8AUCbkBgCtP4u2xwjbBx6RHartX55gl6rR6/V0yQwqtz9rDYrRkseuSZ7\nAsdoySPPnE+htZBCq6nov4UUWkwUOL63FlJgKcRis2C2WbAoFiw2KxabBYvNgkLlPncVFBRFcdYY\nEqIiAo/qmdjhHk93Q1QTS9EDVxlhI4TwRpKwqQZ+RTVsLFa58RNCiLpOURQSM+31azqFXVNie2ix\nETah/iGMaTeq1vrmzTRqDQ10wdVagNlqs2JRriRwnF+KFatiRVEUrIoNm2JDUWzYFAUbRf9VrNgU\nxd6OPXlz5cveblVsUJQWUqnAYNBhzCvEZlPsrYp9myNxpBRbbca+vSg55GhRHFuK2pRi24r9XJlE\nVPFz1wQFBZUK9Hod+fkmrna6Cr+HCu9e8++3NDqNH8M6DQRzjZ5e1CIZYSOE8GaSsKkGflp7wsZk\nkYSNEELUdRfy07lssk/j6VBKwsZPreXudiM4ffkMo9uN8PnpUN5Mo9agQYO/Rlfz59KoCAsLIjMz\nt96OqJUYFMUg2B4DUTdI0WEhhDeThE010PlpADCZrR7uiRBCiJqWmnMGsCdmmpYxDWhQixtqs0tC\nCCEqyTHCRisJGyGEF5JlEqqBvyNhIyNshBCizkvJsa+A1DSwCRq1xsO9EUIIURUyJUoI4c0kYVMN\nnFOiZISNEELUealFS1Y3D27q4Z4IIYSoqitTouSfRUII7yNXpmqg08oIGyGEqA8URSGlaEpUC0nY\nCCGEz3OMsJFVooQQ3kgSNtVA52cPo9liX1lCCCFE3ZRtukyu2QhA86BmHu6NEEKIqrIW3burVZKw\nEUJ4H0nYVAOd9koYzTLKRggh6izH6BoVKpoFNfZwb4QQQlSVY8UzGWEjhPBGkrCpBo5VokDq2Agh\nRF2WWlRwOCowEl0tLCUthBCiZknRYSGEN5OETTWQETZCCFE/pOaeBaBFkNSvEUKIusAqNWyEEF6s\nVhM2iYmJjB49mtjYWEaNGsX+/fvL3f/tt9/m0UcfraXeVV7xETaFMsJGCCHqrIz8DACiDBEe7okQ\nQojq4Kg/KSNshBDeqNYSNoWFhcTFxXHXXXfxyy+/MGHCBKZNm4bRaCyxb15eHi+//DIvvvhibXWv\nSmSEjRBC1A+ZhVkAhAWEergnQgghqoOMsBFCeDNtbZ1o586dqNVqxo8fD8Do0aP5z3/+w9atW7n9\n9ttd9n3kkUfQ6/WMGzeOS5cuVeg8KpUKdSXSUI6semWy6/qAK2G02GxoNHXrgl+V2NR1EpvySXzK\nJrHxPYVWE0ZzHgChASEe7o0QQojqIMt6CyG8Wa0lbJKTk2nTpo1LW0xMDCdOnCix7wsvvEBUVBQr\nVqyocMImPDwQVRWW5QsJCazwMRbrlVE1AQH+hIUFVfr83qwysakvJDblk/iUTWLjOy4VZDm/D5OE\njRBC1AlWKToshPBitZawycvLQ6/Xu7QFBARQUFBQYt+oqKhKnycjw1jpETYhIYFkZRmdmfaK0KhV\nWG0K6Zm5ZGbqr36AD6lqbOoyiU35JD5lq0psvDEpvHnzZlasWMG5c+do2rQp8fHxDBkyhHPnzrFg\nwQJ+/fVXtFott912G7Nnz0an06EoCj179kRRrrz/nj178sYbb3jwnZQts+DKA4SG/g092BMhhPBO\nb775Jv/4xz/w8/Nztq1evZp27doxZ84cdu7cSXBwMDNmzGDMmDEe7OkVMsJGCOHNai1ho9frSyRn\nCgoKMBgM1XoeRVGwVqHur82mYLVW/B+WOj81+YVWCkzWSh3vCyobm/pAYlM+iU/Z6kJskpOTmTNn\nDmvWrKFHjx7s2LGDKVOmsG3bNp588knatWvHtm3buHz5MjNmzOC1114jPj6eU6dOAbB3794qjYys\nLZn59hE2DXTB+Klr7eNTCCF8RmJiIvHx8Tz00EMu7Y8++igGg4EdO3Zw5MgRHn74Ydq1a0dsbKyH\nenqFuWikvEYji+cKIbxPrV2ZWrduTXJysktbcvL/t3fvUVHW+R/A33O/AAICKiQigoGwKSIq/NRU\nLC1XZXfTXVfb3ZOVt1Y7utqWlaGuq+e4Xigz05O2XnbdbPenpdSaadRPV4syDQq6MJsYkgJyZ+7P\n749hRkYdGGCGeWDer3M8Mzw8M8+XD+PnO3zm83wfHeLj47tqCF6lkNuuFGU0cdFhIvIvsbGxOHPm\nDFJTU2E2m1FRUYGAgAAolUpoNBosWrQIKpUKERERmD59Oi5cuADA9sY+ISGhWxRrAKCq+ZQorl9D\nRHRnX331FYYMGeK0raGhASdPnsTSpUuhUqkwdOhQTJs2DUeOHPHRKJ3pDWYAgEYpa2NPIqKu12Uf\nEWZkZMBoNGL//v2YPXs2jh49ioqKCowdO7arhuBV9itFGc28rDcR+Z+AgACUlpZiypQpsFqtyM7O\nRmBgIHbt2uW03+nTp5GYmAjA9sa+vr4eWVlZuHbtGkaOHIlnn33W7dNiO7rIfHvZ1zW40XyFqDB1\naI9bXL4tXCSbMQAYA4AxaE1TUxN0Oh327duHlStXolevXnj00UeRlJQEuVyO6Ohox76xsbE4ceKE\n28/dkXzv7u9Kb7K9d9eq5X6T2/k6bh3j4xpj45q3YtNlBRulUondu3cjOzsbW7ZsQUxMDF555RVo\ntVqsXr0aALB27dquGo7HKRXssCEi/xYZGYmLFy8iPz8fixcvRkxMDDIyMgDYTlddv349SkpKsGnT\nJgC2eSElJQVPPvkkVCoV1q9fjyVLluCNN95w63idXWS+verMdQCAqJAIUa4j1BW4SDZjADAGAGNw\nJxUVFRgxYgR+/etf48UXX8SlS5ewcOFCPPLII1Cr1U77ulrH0pXO5Pu2flem5tOSe4do/S6383Xc\nOsbHNcbGNU/HpktPwk9MTMShQ4du2+6qULNkyRJvD8lj2GFDRP5OLrdNKRkZGZg8eTLef/99ZGRk\nQK/X46mnnkJxcTH279+PsLAwALfn+D/+8Y9IT0/HtWvX0KdPnzaP19FF5tvLvkD0tfpKAIBGEoCq\nqnrvH1hEuIA4YwAwBkDXx6A7FRCio6Nx4MABx9dpaWnIyspCfn4+DAaD077tXceyI/ne3d9VXYPR\ndsdq9Zvczv/LrWN8XGNsXOtsbFzle66a6CHssCEif5WXl4e9e/fi9ddfd2wzmUwICgpCdXU1Hnvs\nMWi1WvzjH/9ASMjN9V927dqFMWPGIDk5GQBgNNreNKtUKreO29lF5tvDKlgda9iEKEO6/ULRHdUT\nFsnuLMaAMQAYgzspLCzEmTNnMH/+fMc2g8GAyMhImEwmlJWVISoqCkD717HsTL5v63fV1LyGjUoh\n87vfKV/HrWN8XGNsXPN0bLgcuoeww4aI/FVSUhIKCgpw5MgRWK1W5OXlIS8vD9OmTcOSJUsQHh6O\n1157zalYAwAlJSXYuHEjbty4gbq6Oqxfvx6TJk1CcLD4Lplda6iH2Wp7U99bxUWHiYhupdVqsX37\ndrz77ruwWq34z3/+g+PHj2Pu3LmYNGkSNm/ejKamJly6dAnHjh3D9OnTfT1kAIDeaMvtai46TEQi\nxA4bD2GHjTgJggCLtfmfxQpzc8XTbLHCYm2+tQgwW62wWgUIAppvBVgFNN+2uG+9uU0QAEgArVaF\n+no9zGYrBDg/3ioIQHOBVbg5KMd9oUXxVcDt+wotdmjxVE7P0fIxt+5/89vC7cfzYFG8xWicSCUS\nqNUK6PUmWyzcf0KP8Wjt34NPJpECacmRiO/XfdrdXYmIiMDOnTvx5z//GWvXrsXAgQPx8ssvo6am\nBh9//DFUKhVGjRrl2D8pKQkHDx7Ec889h/Xr1+PBBx+EyWTChAkTsG7dOh/+JK5VNFQ57vMqUURE\nt4uNjcW2bduwdetWPP300+jbty82bNiA5ORkrFu3Di+88ALGjx8PrVaLlStXYtiwYb4eMqyCAL3B\n9mGrRsU/i4hIfJiZPMTeYWNih027CYIAk9mKRoMZTQYzGvVmNDbfNhls9/VGM4wmK4wmCwwmK4xm\ni+Nr+32DyQKj2WorzFgEWKy2WyKxeu+TK9i5Yjyk3eSy1q1JS0vDv/71r9u2FxcXu3xMYGAgNmzY\n4M1heUxl0w0AgFwqR6CCC+0REd1JZmYmMjMzb9seEhKCnJwcH4yodQajxfFZjEbJP4uISHyYmTxE\nqWg+JYodNg4mswWVtQZU1uhRXW9AbaMRtQ1G1DaYUNtoRF2DETWNRjQ0mURdWJHAtoiURGLrGLFf\nWlICCaRSie0SkAJs35dKmrfbLkEpkUggge17Ts/ZvN1+AEmLo9n3df6+5LZ9bftJWtxv/spxX9Li\nfsvnk9zc5uU6gUQigUIhg8lkcer+cfexYuSpYUkkEqQl9YNCLuU5wN1AncG2EGWQIlC0r00iImof\nvfHmB61qFU+JIiLxYcHGQ5RyW5I3+FGHjSAIqG004WpFA65WNeJ6dRMqa/SorNWjskaPGvuq+x2k\nUcmhVcmgUSmgVsmgUsiglEtttwoplHIZlC3uqxRSKOQyyOUSyKVSyGQSyGVSyKUSyGTNXzdvl0lt\n33PcyiSQSmwFGKnE9se0rTjTeuFAJpOgd+9AVFXV84/uO2B8XGsZGxK/emMjAECr0Ph4JERE5CmN\nzQsOA+ywISJxYmbyEEVzh42ph3bYGEwWfPtDDUp+qMXlH+twtbIRVysb0KA3t/1gAIEaBYIDlOgV\noESQVoFeAUr00tq+DtQoEKCWNxdo5NCq5VAr5ZBK+Sk2EYlDg71gI2fBhoiop9C3LNhwDRsiEiFm\nJg9RNXfY9ISrRAmCgPKqRhSXVqPkh1r898c6lFW0fj354EAl+oZoEBasRliwBuHBaoT1Utu+7qWC\nQs42UyLqvupZsCEi6nGajDcLNrxKFBGJEQs2HmLvsDGau1+Hjb1AU3S5GsWXb6D4crXL05l6BSgx\nsF8QosIDEBmmRVSY7VarVnTxqImIuo6jw0ah9fFIiIjIU+xXiFIpZezsJiJRYsHGQ+xr2BhN3aPD\nxmyx4pvSanz+bSUufluBa9VNt+2jUckQf1cIBkUF4Z7BfRAWqEAvrZILbhKR32kwNQBghw0RUU/S\n1HxKlIbdNUQkUizYeIiyG3TYmMxWXPquAp8UXcMXJVWOScpOq5Lj7ugQJAwIQeKAUET3CXRcBYkL\nxxKRP6s3cNFhIqKepqn5KlFcv4aIxIrZyUNudtiIq2BjFQR8U1qN/xT+iPyia06r4QNA/4gADIsP\nR0p8OGIje7EdlIjoDupNXMOGiKinsS86rOYVoohIpJidPMTRYSOSU6Iqa/TIu/gDzhaUo6rW4Ngu\nAZAwIAQjEvpgWFwYwkP4xwcRUVu4hg0RUc9jX3RYo+IpUUQkTizYeIi9w8ZiFWCxWiGTSrt8DFZB\nwJe6Kpz67Adc/K4CQouzl/pHBCLjJ30xekhf9O6l7vKxERF1V1bBikaTbZ0vdtgQEfUcTc2LDmvY\nYUNEIsXs5CH2DhvAdlqURtV1BZtGvRkfXSrD6Qs/4NqNm4sHB2oUGHtPJDJ+0g/RfQK7bDxERD2J\nvVgDcA0bIqKeRN/cYaNmhw0RiRQLNh5i77ABbIv7alTeP2Z1vQHv5Zfigws/OD4hAIC4u3ohc3h/\npCVGQCHnBERE1BmN5hYFGzlPiSIi6inYYUNEYsfs5CHOHTbeXcemvKoR757/HmcLymFuvmqTXCbF\n//ykLyYO74+YfkFePT4RkT9hhw0RUc/U5Oiw4Z9ERCROzE4e0rLDxuClS3uXlNXinXPf47Ovr8O+\nPI1WJUfmiLswaUQ0ggOUXjkuEZE/azQ3Ou5zDRsiop7DfpUoLjpMRGLFgo2HKFp02JjMnuuwEQQB\nX5RU4d3z36PocrVje2iQClNGRmPcsCho+KkAEZHXNDR32KjlakglXb+gPBEReUeTo2DD99JEJE7M\nTh6iatFhYzR1vsPGbLHik6JreOfcZVy5Xu/YHhUegAdHD8DopL6Qy/iHAxGRtzWabB02AeyuISLq\nUbiGDRGJHbOTh7TssDF2osOmyWDG/126ihOflKKyVu/YPrh/MB5Mj8HQuDBIJZJOjZWIiNxnX3RY\nq+CCw0REPYUgCI41bHhKFBGJFQs2HiKVSCCXSWG2WDvUYVNZo8f7n15B3kXnKz6lxIdjanoM4vsH\ne3K4RETkJvuiw1xwmIio5zCarBCaF4VUs8OGiESK2cmDVAp7wca9DhtBEFBSVov38kuRX3QdVsF+\nxScJ0pP6YcroAbgrPMCbQyYiojY0NJ8SxQWHiYh6Dnt3DcA1bIhIvJidPEght50WZWzjKlE36gw4\nV1iO//viKq5W3rz6SKBGgczUuzAxtT+v+EREJBL2U6ICeEoUEVGPYV9wGAA0Sp4SRUTixIKNBykV\ntmR/pw6bipomXPimAp9/U4GiyzccLZgAEBmmxeSR0chI7ud4DiKi7iQ3NxcvvfQSysvLERUVhWXL\nluG+++5DTU0NVq1ahXPnziEoKAhPPPEEZs2aBcDWZbhlyxYcPnwYFosFWVlZeOaZZyCTiSsP8pQo\nIqLO+/LLL7F69Wp8++23iImJwZo1a5CSkuKz8eiNN9+vq9lhQ0Qi1aXZyd1EfezYMWzduhWVlZUY\nPXo01q9fj/Dw8K4caocomztsTGbbaVHfXKnBl/+tQqGuCpev1Tvtq1HJMGpIX4y5JxJxUb0g4ULC\nRNRN6XQ6rFq1Cnv27EFqairOnj2L+fPn48MPP0R2dja0Wi3Onj2L4uJiPP744xg8eDBSUlJw8OBB\nfPDBB3jrrbcgkUiwYMEC7NmzB48//rivfyQnPCWKiKhzDAYDFi5ciIULF2LWrFk4evQoFi1ahJMn\nTyIgwDen/7fssFGzw4aIRKrLCjbuJuqioiK88MIL2LNnDxISErBu3To888wz2L17d1cNtcPs3THv\n5Zfifz/SwWxxPjVKo5JjWFwYht8dgWFxYeymIaIeITY2FmfOnEFAQADMZjMqKioQEBAApVKJkydP\n4t///jdUKhWGDh2KadOm4ciRI0hJScHRo0fxu9/9Dn369AEALFiwADk5OaIr2Ny8ShQLNkREHXHu\n3DlIpVLMmTMHADBz5kz89a9/RV5eHqZOneqVY3713yrUFVegocHgWCeypdLmD1OVcinkMult3yci\nEoMuK9i4m6jffvttTJo0CcOGDQMArFixAhkZGaioqBB9l42quQBTXW90bOsfEYCkgb1xT1wYEqJD\nOCEQUY8UEBCA0tJSTJkyBVarFdnZ2bh8+TLkcjmio6Md+8XGxuLEiRMAgJKSEsTHxzt9T6fTQRAE\nt7oOJRIJpF2QUo0WW04PUgZAJvPPbkipVOJ0648YA8YAYAw6SqfTIS4uzmlbbGwsSkpK3Hp8e/N9\n6bV6bDx4wa19NSq53+V2vo5bx/i4xti45q3YdFnBxt1EXVJSguHDhzu+Dg0NRXBwMHQ6nVsFm46+\ngfdEgCem3oW6JhMG9gtCcmxvJA0MRUigqsPPJxb8j+kaY9M6xse1nhibyMhIXLx4Efn5+Vi8eDEe\nffRRqNVqp33UajX0ej0AoKmpyen7Go0GVqsVRqMRKlXbuTMsLKBLTif9acIkfHX9a/xPXCo0CnXb\nD+jBQkJ45ULGgDEAGIP2amxshEbj3KXYcj5oS3vzvSZAjWGDw1FW0dDqflKJBNPHDULv3oFuP3dP\nwtdx6xgf1xgb1zwdmy4r2LibqG99Aw/Y3sQ3NTW5dZzOvoHvTIAfHBuIB8fGtb1jN8X/mK4xNq1j\nfFzrSbGRy21TSkZGBiZPnoyCggIYDAanffR6PbRa29WW1Gq10/ebmpogl8vdKtYAQGVlQ5d02Nx3\n13jMTJ6K6uoGNFnr235ADySVShASEoDq6gZYrbefWuAPGAPGAOj6GPSUQoJGo7ntPX/L+aAtHcn3\nK2anuP27qqryr9zO/8utY3xcY2xc62xsXOX7LivYuJuoXRVxvJnQAb74WsPYuMbYtI7xca0zsRHb\nG/i8vDzs3bsXr7/+umObyWTCgAED8OGHH6KsrAxRUVEAbN2W9tOg4uLioNPpHKfA6nQ6DBo0yO3j\nCoIAy+0X5fMaq1WAxeLfr2PGgDEAGAOAMWivQYMG4cCBA07bdDodpk2b5tbjO5Pv+btyjbFpHePj\nGmPjmqdj02UFG3cTtf0NvF1VVRVqampuO53Klc6+geeLzzXGxjXGpnWMj2s9ITZJSUkoKCjAkSNH\nMGPGDHz00UfIy8vDG2+8gatXr2Lz5s3405/+hG+++QbHjh3Drl27AAAzZszAa6+9hvT0dMjlcrz6\n6qvIysry8U9DRESelpGRAaPRiP3792P27Nk4evQoKioqMHbsWF8PjYhI1LpsBdyWidpkMuHNN9+8\nY6KeNm0aTpw4gfz8fBgMBmzZsgX33nsvQkNDu2qoRETUDhEREdi5cyf27duHtLQ05OTk4OWXX0Zc\nXBzWrVsHs9mM8ePHY+nSpVi5cqWjo2bOnDnIzMzEzJkz8dOf/hSpqal45JFHfPzTEBGRpymVSuze\nvRvHjx/HqFGjcODAAbzyyitud9ATEfkriSDc4Tp3XlJUVITs7GwUFxcjJiYG2dnZSElJwerVqwEA\na9euBQDk5uYiJycH169fR1paGjZs2ICwsDC3jnH9el2HxiaTSdC7dyCqquq7/afdnsbYuMbYtI7x\nca0zsYmICPLSqLqXjub79uLrmDEAGAOAMQC6PgbM9zYdyfd8vbrG2LSO8XGNsXGts7Fxle+77JQo\nAEhMTMShQ4du224v1NhNnTrV6VLfRERERERERET+pMtOiSIiIiIiIiIiIvewYENEREREREREJDJd\nuoYNERERERERERG1jR02REREREREREQiw4INEREREREREZHIsGBDRERERERERCQyLNgQERERERER\nEYkMCzZERERERERERCLDgg0RERERERERkciwYENEREREREREJDIs2BARERERERERiQwLNkRERERE\nREREIuP3BZsvv/wSM2fOREpKCrKysvD555/7ekiikp+fj1mzZmHEiBG47777cOjQIV8PSXQqKiqQ\nkZGB06dP+3ooolFeXo4FCxYgNTUV9957L/bt2+frIYnKZ599hl/84hdITU3FlClT8Pbbb/t6SOQC\n5wjOA3b+nuuZ15m7uxPmbteY09vm7/neFc4Drnl1fhD8mF6vF8aNGyccPHhQMBqNwuHDh4X09HSh\nvr7e10MTherqamHkyJHCW2+9JVgsFqGgoEAYOXKkcObMGV8PTVTmz58vJCYmCqdOnfL1UETBarUK\nP//5z4WNGzcKRqNR+Prrr4WRI0cKn376qa+HJgpms1lIT08X3nnnHUEQBOGTTz4RkpKShNLSUh+P\njG7FOYLzQEv+nOuZ15m7uxPmbteY093jz/neFc4Drnl7fvDrDptz585BKpVizpw5UCgUmDlzJsLD\nw5GXl+froYlCWVkZxo8fj+nTp0MqlSI5ORmjR4/GZ5995uuhicbf//53aDQaREZG+nooonHx4kVc\nu3YNK1asgEKhwODBg3Ho0CHExsb6emiiUFtbi6qqKlgsFgiCAIlEAoVCAZlM5uuh0S04R3AesPP3\nXM+8ztzdnTB3u8ac3jZ/z/eucB5wzdvzg18XbHQ6HeLi4py2xcbGoqSkxEcjEpchQ4Zg06ZNjq9r\namqQn5+PxMREH45KPHQ6Hfbu3Yvs7GxfD0VUCgsLMXjwYGzatAljxozBlClTcPHiRYSGhvp6aKIQ\nGhqKOXPmYPny5UhOTsbcuXPx/PPP842BCHGO4DwAMNcDzOsAc3d3wtztGnN665jvXeM84Jq35we5\nR56lm2psbIRGo3HaplarodfrfTQi8aqrq8PChQuRnJyMzMxMXw/H58xmM5566ik8++yzCAkJ8fVw\nRKWmpgbnz59Heno6Tp8+jYKCAjz22GOIjo5GWlqar4fnc1arFWq1Gjk5OcjMzMTZs2fxhz/8AcnJ\nyXzDJDKcI5z54zzAXG/DvM7c3Z0wd7vHH3N6a5jvW8d5wDVvzw9+3WGj0WhuS7GzLA0AAAcXSURB\nVN56vR5ardZHIxKn0tJSzJ49G8HBwdi+fTukUr9+2QAAduzYgSFDhmD8+PG+HoroKJVKBAcHY8GC\nBVAqlY7Ft95//31fD00UTpw4gUuXLuGBBx6AUqnEhAkTMGHCBBw5csTXQ6NbcI64yV/nAeZ6G+Z1\n5u7uhLm7bf6a01vDfN86zgOueXt+8Ov/nYMGDYJOp3PaptPpEB8f76MRiU9hYSF++ctfYuzYsdix\nYwfUarWvhyQKubm5OH78ONLS0pCWloaysjIsX74cu3bt8vXQfC42NhYWiwUWi8WxzX5OJwFXr16F\n0Wh02iaXy7kOgghxjrDx53mAud6GeZ25uzth7m6dP+f01jDft47zgGtenx88snRxN2UwGISxY8cK\n+/btc1pFvqGhwddDE4Xr168L6enpwquvvurroYjexIkTuZJ8s6amJmHcuHHCtm3bBJPJJHz66adC\nSkqKcOHCBV8PTRSKioqE5ORk4c033xSsVqtw/vx5Yfjw4cKlS5d8PTS6BecIzgO38tdcz7zO3N2d\nMHe7xpzuPn/N965wHnDN2/ODRBD8uyxWVFSE7OxsFBcXIyYmBtnZ2UhJSfH1sERh586d2Lp1620t\npL/97W+xbNkyH41KnDIzM/H8889j4sSJvh6KKHz//fdYu3YtvvjiCwQGBuKJJ57AQw895Othicap\nU6eQk5OD0tJSREVF4cknn8T999/v62HRHfj7HMF5wJk/53rmdebu7sTfc7crzOnu8+d87wrnAde8\nOT/4fcGGiIiIiIiIiEhs/HoNGyIiIiIiIiIiMWLBhoiIiIiIiIhIZFiwISIiIiIiIiISGRZsiIiI\niIiIiIhEhgUbIiIiIiIiIiKRYcGGiIiIiIiIiEhkWLChbu3pp59GQkKCy38vvfQSzp8/j4SEBBgM\nBq+P58qVK45jL126tMOP/+6779rcd8WKFY5jubM/EVF3xVzPXE9E/oH5nvmenEkEQRB8PQiijqqr\nq4NerwcAXLhwAUuWLMGpU6egVCoBAFqtFgqFAjU1NYiIiPD6eK5cuYJJkybh4MGDuPvuu9GrV692\nPd5isaCqqgq9e/eGTCZrdd+6ujrodDrMmjULubm5iIuL68zQiYhEi7meuZ6I/APzPfM9OZP7egBE\nnREUFISgoCAAQHBwMAAgPDwcKpXKab+uSOgthYaGtjuhA4BMJnN7rEFBQejdu3e7j0FE1N0w1zPX\nE5F/YL5nvidnPCWKerxb2yYTEhKQm5uL6dOnY+jQoZg3bx6uXr2KlStXIiUlBZMnT8a5c+ccj//x\nxx+xdOlSDB8+HOPGjUN2djYaGhrcOra9DfKDDz7A/fffj2HDhmHZsmUoKyvD/PnzMWzYMGRlZaGo\nqMhpf3sbZGZmJvbv34+HH34Y99xzD6ZMmYK8vDwPR4iIqPtjrici8g/M9+RPWLAhv7R582asXr0a\nBw4cQGFhIWbMmIEhQ4bgn//8J+Lj47FmzRoAgCAI+P3vfw+FQoHDhw9j+/btKCoqwqpVq9p1vBdf\nfBHbtm3Djh078N577+Ghhx7C1KlTcfjwYajVavzlL39p9bFz5szB8ePHkZCQgFWrVsFkMnXq5yci\n8gfM9URE/oH5nnoqnhJFfuk3v/kNRo4cCQBIT09HaWkp5s2bBwD41a9+hYULF8JiseDjjz+GTqfD\n3/72NygUCgDAhg0b8MADD6C8vBz9+vVz63iLFi1CcnIyACAxMRFRUVH42c9+BgDIysrC7t27XT52\n+vTpmDp1KgBg8eLFyMrKQnl5OaKjozv2wxMR+QnmeiIi/8B8Tz0VCzbklwYMGOC4r9FonBKkWq2G\n1WqF2WzGd999h/r6eowaNeq259DpdG4n9VuP179/f6fjGY1Gl48dOHCg435gYCAAsApPROQG5noi\nIv/AfE89FQs25JfkcueXvlR657MDzWYzBgwYcMcqeXsWO3P3eHdir/63xIu7ERG1jbmeiMg/MN9T\nT8U1bIhaERcXh/LycgQFBSEmJgYxMTEwm83YuHEj6uvrfT08IiLyAOZ6IiL/wHxP3Q0LNkStGDNm\nDOLi4rB8+XIUFhaioKAAK1euxI0bN9CnTx9fD4+IiDyAuZ6IyD8w31N3w4INUSukUil27NiBwMBA\nPPzww5g3bx4GDhyI7du3+3poRETkIcz1RET+gfmeuhuJwBPmiDzmypUrmDRpEnJzcxEXF9fjjkdE\nRMz1RET+gvmefI0dNkRecOPGDdTW1nr1GHV1daiqqvLqMYiIyDXmeiIi/8B8T77Cgg2RF8ydOxfP\nPfecV4+xZs0azJo1y6vHICIi15jriYj8A/M9+QpPiSIiIiIiIiIiEhl22BARERERERERiQwLNkRE\nREREREREIsOCDRERERERERGRyLBgQ0REREREREQkMizYEBERERERERGJzP8DD1rEo6WqsowAAAAA\nSUVORK5CYII=\n",
"text/plain": [
""
]
},
"metadata": {},
"output_type": "display_data"
}
],
"source": [
"# setpoint\n",
"Tsp = 390\n",
"\n",
"# set initial conditions and cooling flow\n",
"IC = [C0,T0,Tcf]\n",
"\n",
"# do simulation at fixed time steps dt\n",
"dt = 0.05\n",
"ti = 0.0\n",
"tf = 8.0\n",
"\n",
"# control saturation\n",
"qc_min = 0 # minimum possible coolant flowrate\n",
"qc_max = 300 # maximum possible coolant flowrate\n",
"def sat(qc): # function to return feasible value of qc\n",
" return max(qc_min,min(qc_max,qc))\n",
"\n",
"# control parameters\n",
"kp = 40\n",
"ki = 80\n",
"kd = 0\n",
"beta = 0\n",
"gamma = 0\n",
"\n",
"# create python list to log results\n",
"log = []\n",
"\n",
"# start simulation\n",
"c,T,Tc = IC\n",
"qc = 150\n",
"\n",
"eP_ = beta*Tsp - T\n",
"eD_ = gamma*Tsp - T\n",
"eD__ = eD_\n",
"\n",
"for t in np.linspace(ti,tf,int((tf-ti)/dt)+1):\n",
" # PID control calculations\n",
" eP = beta*Tsp - T\n",
" eI = Tsp - T\n",
" eD = gamma*Tsp - T\n",
" qc -= kp*(eP - eP_) + ki*dt*eI + kd*(eD - 2*eD_ + eD__)/dt\n",
" qc = sat(qc)\n",
" \n",
" # log data and update state\n",
" log.append([t,c,T,Tc,qc])\n",
" c,T,Tc = odeint(deriv,[c,T,Tc],[t,t+dt])[-1] # start at t, find state at t + dt\n",
" \n",
" # save data for PID calculations\n",
" eD__,eD_,eP_ = eD_,eD,eP\n",
"\n",
"qplot(log)"
]
},
{
"cell_type": "markdown",
"metadata": {},
"source": [
"## Simulation 4. Interactive Controller Tuning\n",
"\n",
"In this example, PID control is used to stabilize an otherwise unstable steady state, thereby allowing the reactor to operate at temperature and conversion that would not be possible without control. The acheivable operating conditions are limited by the controller tuning and the limits on the available control action.\n",
"\n",
"The following simulation provides for the interactive adjustment of reactor setpoint temperature and the proportional, integral, and derivative control gains. Adjust these in order to answer the following questions:\n",
"\n",
"1. What is the minimum achieveable temperature setpoint for the reactor with conversion greater than 80%? What limits the ability to reduce the temperature setpoint even further?\n",
"\n",
"2. Adjust the temperature setpoint to 420K. Adjust the controller gains for satisfactory closed-loop performance. Repeat the exercise for a setpoint of 390K. How is the behavior different? What happens when when the proportional gain is set too small? Explain what you see."
]
},
{
"cell_type": "code",
"execution_count": 7,
"metadata": {},
"outputs": [
{
"data": {
"image/png": "iVBORw0KGgoAAAANSUhEUgAABGwAAAEMCAYAAACRPCChAAAABHNCSVQICAgIfAhkiAAAAAlwSFlz\nAAALEgAACxIB0t1+/AAAIABJREFUeJzs3XdcleX7wPHPGRz2EPcCceEWF0iKfjXTstLUNLXULBVN\nszQ3uVdlTlARUzMzv5bfyurnzFm5RzlwQ5oKIiJ7nvH743gO58gQUYH0er9evoRn3ucGnvOc67nu\n61YYDAYDQgghhBBCCCGEEKLEUBZ3A4QQQgghhBBCCCGENQnYCCGEEEIIIYQQQpQwErARQgghhBBC\nCCGEKGEkYCOEEEIIIYQQQghRwkjARgghhBBCCCGEEKKEkYCNEE/Q45yETSZ0E0KIp4dc04UQomST\n63TupF+KlgRsRIny888/89Zbb9GiRQt8fX3p168f+/btK+5mFcqxY8cYO3bsIx8nISGBDz/8kMjI\nSACuX7+Ot7c3+/fvf+RjCyFEcZkwYQLe3t75/jt8+HBxN/OJWLRoEZs3by7uZgghnnJFdV99+PBh\nvL29uXLlCgD9+vVj1KhRj/08JufOncPb25tff/3VanlUVBTe3t60bNkyR1Bh3rx5NG/eHJ1OV6Bz\n7Ny5kzlz5jy2NlvK731v2bJlwJPvw8JasmQJ33//fXE345miLu4GCAHGSO348ePZsWMHb775JkOG\nDEGn0/HLL78wZMgQpk+fTu/evYu7mQ9l06ZNXLt27ZGPc/78ebZu3cr7778PQLly5di4cSM1atR4\n5GMLIURxee+996yu64GBgXTo0IGePXual9WsWbM4mvZEabVali9fzqxZs4q7KUKIp1Rx31dPnToV\nGxubJ3Z8b29vXFxcOHXqFB06dDAvP3jwIK6urty9e5dz585Rr14987oTJ07QokULVCpVgc7x1Vdf\nUaZMmcfedpNBgwbxwgsv5FhesWLFJ3bOx2Hp0qVMmzatuJvxTJGAjSgRNm7cyM8//8yaNWto2bKl\neXm7du3QaDTMmTOHjh074u7uXoytLBk0Gg0+Pj7F3QwhhHgkHh4eeHh4mL9Xq9VUqFBBrm9CCPGI\nivu++kkH25VKJc2aNePPP/+0Wn7w4EHatWvH0aNHOXDggDlgk5mZydmzZ/noo4+eaLseRpUqVeT9\nThSIDIkSJcLatWvp0KGD1ZuKyYgRI+jduzcpKSkAbNmyhW7dutG4cWOef/55Vq5caZX26O3tzebN\nmxkxYgQ+Pj60atWKkJAQq2PGxcUxduxY/Pz8aNGiBSNHjuTWrVvm9bdu3eLDDz+kWbNmNGvWjLFj\nx3L37l3z+gkTJjBmzBjCwsIICAigUaNGBAYGmo8xYcIEfvjhB/766y+8vb25fv06wcHB9OnTh6lT\np9KkSRMGDx4MwOXLlxk+fDi+vr40aNCATp068d133wHGFNP+/fsD0LlzZ4KDg3MdEnXw4EF69+5N\nkyZNCAgIYN68eWRmZprXt2/fni+//JKJEyfSrFkzfH19mTlzJllZWYX7gQkhRBGJjo5m5MiRNG3a\nlObNmzN+/HgSEhLM68eMGcP48eMJCQnhueeeo0mTJkyZMoXk5GSmTp1K06ZNadu2rfm6atpn1KhR\nLF68GF9fX/z8/Jg5c6bVddNgMLBixQrat29Pw4YN6dGjB0ePHjWvP3DgAN7e3mzcuJGWLVvSpk0b\nUlNTiY+PZ+rUqbRp04b69esTEBDAp59+ilarRavVUr9+fQA+/vhj3n77bQDatGnDwoULrV73yJEj\nzeuvXr2Kt7c369atIyAgAF9fX/755x8AvvvuOzp16kSDBg14+eWX2bFjx2PtfyHEv8/D3FfDg++t\nMzMzCQkJoWPHjjRq1Ihu3bqxZ8+ePM9vOZzHNFzqxIkTvP766zRs2JBOnTrlGM70xx9/0K1bNxo1\nakT37t3ZuXOn+R46N35+fpw5cwa9Xm9edujQIVq0aIGfnx8HDx40Lw8PDycjIwM/Pz/AeH3/4osv\n6Ny5Mw0aNKB58+aMGDHCfB/fr18/jhw5wpYtW/D29jYf5+TJk/Tp04dGjRoREBBAcHCw1fnbt2/P\nwoULzX35OIe+xsbGMmHCBFq3bo2Pjw+DBw82D0Ez9VVsbKx5++HDh9OkSRO0Wq15Wbdu3Vi4cKH5\nZ/Lf//4Xf39/WrduTXJyMomJiUyfPp02bdrQoEEDWrduzZw5c8yfF0x9MW3aNPr162c+7v/+9z9e\nfPFFGjRoQOfOndm6detje91CAjaiBLh16xYREREEBATkur5y5cpMmjSJqlWr8vXXXzN69Gh8fX1Z\nunQp3bp1Y/HixcybN89qn1mzZuHh4cHy5cvNgQ7TmF2tVsvbb7/NiRMnCAoK4rPPPiMyMpJhw4YB\nkJqaSv/+/bl48SKzZ89m5syZnDx5ksDAQKtxr3v27GH37t1Mnz6d2bNnc/LkSebOnQsYU/3btm1L\nrVq12LhxI+XKlQPg1KlT3Lx5k2XLlvHOO++QnJxM//79ycjI4PPPP2fZsmVUr16dyZMnExkZSf36\n9ZkyZQoACxcutBoqYLJ7924GDhyIp6cnS5YsYdCgQXzzzTc56ucEBwejUCgIDg7m3Xff5euvv7b6\nACOEECVNcnIyb731FhEREcydO5fp06dz7NgxAgMDrW6Sd+7cydGjR/nss88YPHgwGzdupHv37uh0\nOpYuXUqDBg2YPn06UVFR5n3279/P9u3bmTNnDiNHjuR///sf06dPN6+fN28eS5cupXfv3ixduhRP\nT0/eeecdzp8/b9XGr776ik8//ZRJkybh4ODAqFGjOHz4MBMnTuSLL76ga9eurF69ms2bN6NWq/nm\nm28A4xCwyZMnP1R/hIWFMWXKFIKCgqhatSrr169nypQpdOzYkeXLl9OyZUs++OCDf23tNyHEo3uY\n+2qgQPfWY8aMYfXq1bz11luEhIRQs2ZNhg0blm/Q5n5jxozhtddeY8WKFVSqVInRo0cTHx8PGIf/\nBwYG4unpSUhICAEBAYwZMybf47Vo0YKUlBQuX74MwJUrV4iJicHf3x8/Pz+OHz9uDsKfPHkSd3d3\nc8Bh5cqVhISE8Oabb7J69WpGjx7NoUOH+OyzzwDjkK569erx3HPPsXHjRnMbBwwYgJubG8HBwQwe\nPJhVq1bl+AyyatUqunbtyueff55rwMxEr9ebA/mmf5bva5aSk5Pp3bs3J0+eZOLEiXz++efcvXuX\nvn37EhUVhb+/P2q1miNHjgDGgNSxY8dITU3l3LlzgPFh9blz56x+L7788ks++eQTgoKCcHJyYvTo\n0Rw4cICJEyeycuVKunfvztq1a/nhhx8AzH0xaNAgpk6dal728ccf0759e5YvX06rVq0YNWoUu3fv\nzvfnJwpOhkSJYmeKZj9ozKZOpyM4OJjXX3+diRMnAtC6dWsUCgXLly9n0KBB5tTO1q1bM27cOABa\ntmzJtm3b2L9/P23btmXv3r1cuHCBzZs3U6dOHQDKly/P+++/zz///MP+/fu5ceMG27dvp3LlygDU\nq1ePl156iT179pjHymZmZhIWFoaLiwsAly5dYt26dYAx1d/d3Z34+HirdEetVktQUBDVqlUD4PTp\n03h5ebFw4UKcnZ0BaNy4Mb6+vpw4cYIePXqY00q9vb2pUKFCjicNS5Yswd/fn08//RSAgIAAXF1d\nGT9+POfOnaNu3boA1KhRw1w87bnnnmPPnj3s37+fvn37FvAnJYQQRev7778nJiaGHTt2UKFCBQDq\n1KnDyy+/zL59+2jXrh1gvLYGBwfj4uJC69at+fbbb1GpVMycOROFQoGHhwe//vor586dM7/XpKen\ns2LFCvOHFq1WyyeffGJOmf/qq68YNWoU7777LmC8tvbp04dly5axZMkScxvfeecd2rZtCxgD/gaD\ngVmzZtG8eXMA/P392bNnD8ePH6dHjx40btwYgKpVqz50LbKePXuaax5otVpCQkLo06ePuc0BAQHc\nvXuXxYsXm9skhHi2FPS+Ggp2bx0TE8P27duZN28eXbp0AYxZgTExMSxatMh8HX6QwYMH06dPH3Pb\nXnzxRQ4fPkynTp1YuXIlNWrUYOHChSgUCtq0aUNycjJff/11nserW7cuzs7O/PXXX9SuXZuDBw/i\n4eFB5cqVUavVpKWlcfLkSfz8/Dh58iS+vr4oFAoAYmJiGDlyJG+++SYAvr6+REREsGvXLsA4pMvJ\nyQk3Nzfzffzy5cupWrUqISEh5jo49vb2TJ8+nUGDBlG6dGkAGjRoYM6OzM+MGTOYMWOG1bLOnTvn\nyLYEYwZLdHQ027Zto0qVKoAxw+j5559n9erVBAUF0bhxY44ePUrnzp25dOkSaWlpeHl5ceLECRo2\nbMjBgwdxcnLCx8eH48ePA9bvX+np6eh0OqZPn24ONPn7+7N3716OHz9Or169zH1RpUoVatasiV6v\nZ8mSJfTs2dP8uSsgIID4+HgWL15M+/btH9gP4sEkw0YUO9NFL6+osklERATx8fG8+OKLVss7d+5M\nVlYWf/31l3mZ6YYYQKFQUL58eVJTUwFjlL1ChQrmYA0YAzK7du2iatWqHD16lFq1alG+fHlzxLtK\nlSp4eHhw6NAh8z5eXl7mYA0Ygz5paWn5vga1Wm3+cADQsGFD1q9fj62tLRcuXGD79u2EhYUBFGi4\nUkpKCufPn8+1TxQKhfmCDNCoUSOrbSz7RAghSqKjR49Su3ZtypQpY74ee3p6UqVKFasZpKpVq2Z1\nPXZ3d6devXrmm3M3NzcAkpKSzNs0atTI6nr8/PPPo9fr+fPPP/nzzz/JysoiICDAfF6dTkfr1q1z\nzFzl5eVl/trBwYEvv/ySZs2ace3aNfbt28eKFSuIi4t7LENQLc915coV4uLirNqo1WoJCAggPDzc\n6rUKIZ4dBb2vhoLdWx8/fhyFQpHrNufPnyc5OblA7bK8NzcF4E33zUeOHKFdu3bmazaQ43z3U6lU\nNGvWzHz/f/DgQXOgoXz58nh5eZmv16bAjcnHH3/MO++8Q2xsLIcPH2b9+vWcOHEi3+v00aNHadWq\nFQaDwep6m5WVxYkTJ8zbWV6n8xMYGMimTZus/o0ePTrXbY8fP06DBg3MwRoAZ2dnAgICzPf6rVq1\nMg/bPXr0KI0aNaJp06bmth04cIDnnnsOtTo7X8OyrXZ2dqxZswY/Pz/z+1dYWBh37tzJs18iIyOJ\njY2lTZs2Od6Hzp8/b86gEo9GMmxEsTM9AbBMVb9fdHS0uWbB/RXbTRFtyzcMW1tbq22USqV5LG5C\nQkK+Rdbi4+MJDw831xmwZBnksbOzy/MceXFzc8tRnT4kJIRVq1aRlpZG1apV8fX1BXjgscD44cNg\nMJj7wESj0eDk5GTVJ4VprxBCFKf4+HhOnz6d6/X49u3b5q8dHR1zrL//mne/smXLWn1vel9ITEw0\nf9B59dVXc93XstbN/dff7du3M3fuXKKioihbtiw+Pj7Y2dk9luut5blMN8JDhw7NddvY2Fhz5qYQ\n4tlR0PvqChUqFOjeOiEhAWdnZzQaTa7bWNbCyY/lvblSacwZMF1r7969m+Pe/P5ra25atGjB5s2b\n0ev1HDlyxGpYa8uWLTl+/Dg3btwgJibGanjS5cuXCQoK4s8//8TR0ZH69etja2ub73U6Pj6etWvX\nsnbt2hzrYmJiHqrdYPw5NWzYsEDbJiYm5npcd3d3zpw5A2CuqRMXF8fRo0dp1qwZHh4eLFq0CDAG\nbN57770c+1v69ddfmT17Njdv3qRMmTL4+Pjk2y+m+p7Dhw/PdX1sbKz5gYkoPAnYiGLn7u5OnTp1\nOHDgQK5TDN64cYP27duba7lYFtSy/N7V1bVA53N2drYqIGyyb98+GjZsiIuLC02aNCEoKCjHNgU9\nR0H9+OOPLFu2jNmzZ9OxY0ccHR1JT09n06ZNBdrfyckJhULBnTt3rJZnZGSQlJT02NsrhBBFydnZ\nmWbNmplT9S096k2gZeFiwHwdLVWqlPlp4pdffomTk1OOfS2fUFq6cuUKH330Ef369ePdd981fwjq\n3r17vm1RKBQ5noY/6EOQKaPo008/zXVoVaVKlfLdXwjxdHqY+2rTQ8L87q0TExNJSkoiMzPTKmjz\nsPff+SlXrlyOe9m4uLgH7ufr68uCBQs4ceIESUlJVkGZli1bsnnzZk6cOEG5cuWoXr06YAwSDRs2\njAoVKrB161a8vLxQKBTMmzePa9eu5XkuZ2dnXnnlFV577bUc65709dbFxcUqKGRy584dc/83aNAA\nV1dXjhw5wvHjx+nevTuenp7ExMTw+++/c/PmzTzrGgH8/ffffPjhh/Tt25fBgwebH2rkVj/Tsl0A\nc+fOpVatWjnWW2YEicKTIVGiRHjrrbfMRSPvFxwcjI2NDR06dMDNzY1t27ZZrd+6dSsqlSrHkJ+8\nNG7cmKioKC5cuGBedvHiRYYMGUJERARNmjTh6tWreHl50bBhQxo2bEitWrUIDg7m9OnTBX5NpqcH\n+Tl58iTVqlWjW7du5ifEf/zxB5D91OH+jBxLTk5OeHt759onAE2aNClwe4UQoqRp0qQJ165do3r1\n6ubrcY0aNViyZIn5qWJhnTp1yip4v2vXLmxsbGjSpAmNGjVCpVKRkJBgPm/Dhg3Zv38/3333XZ7X\n97Nnz5KVlUVgYKA5WBMTE8OlS5fMTyhz29fBwYHo6Gjz9xkZGZw9ezbf9teoUQNnZ2fu3Llj1cZz\n586xcuVKq6EFQohnS0Hvq6tXr/7Ae+umTZtiMBhy3aZu3boPzGYsiGbNmuUoYLx3794H7le/fn3s\n7Oz45ptvqFWrllXGiJ+fH2lpaWzZssVqOFRcXBzXrl2jT58+VK9e3RwwP3jwoFXg/P5rdZMmTfj7\n77+trrdqtZpFixblCDY9bk2bNuXMmTNWdSyTk5P5/fffzXVllEol/v7+/PDDD8TGxtK0aVM8PT0p\nW7YswcHB1KpVyzwULTfh4eHm9y9TsCY2NpaLFy/m2S+m35/Y2Firfrlw4QJhYWEF+iwkHkwybESJ\n0KNHD3bt2sXgwYPp378/fn5+pKSk8MMPP7B7925mzZpF+fLlee+995g7dy6Ojo60adOGP//8k+XL\nl9OvX78CP21t3749tWvX5v333+fDDz/E1taWRYsW0bRpU5o2bUrt2rVZu3YtgwYN4t1330Wj0bB6\n9WpOnTrFhAkTCvyaXFxcuHbtGgcPHswzcNKgQQM2btxIWFgYPj4+hIeHExISgkKhMI/rNaW079q1\nK8dQL4D333+f4cOHM378eF555RUiIyNZtGgRL7zwgtUQLiGE+Lfp2bMnX331lfl6bGNjwxdffEF4\neDgff/zxIx07LS2NYcOGMXToUK5du8aiRYvo168fLi4uuLi40LNnT6ZOnUpMTAy1a9fm0KFDhIaG\nmgv85qZevXoolUo++eQTXnvtNaKjowkNDSUrK8tcM0ypVOLk5MShQ4do2LAhderUoXXr1mzcuBFf\nX18qVqzImjVrHjiESqPRMGTIEJYsWUJmZiZNmjTh3LlzLFiwgB49euQYviCEeHYU9L4aeOC9tZub\nGx06dGDatGnEx8fj5eXFL7/8wuHDhwkJCXks7R0yZAjdunVj9OjRdOvWjbNnz/LVV18B+T8AValU\nNG3alB07duSYRKNUqVJ4e3vz22+/MW3aNPPy0qVLU7FiRVatWoWjoyN6vZ4NGzYQHh5uFXxycXHh\nwoULHD58GF9fX4YOHcqbb77JxIkT6dy5MwkJCSxcuBB7e/sC160prB49evDVV1/x7rvv8sEHH6DR\naAgLC0Ov1zNgwADzdq1btyYoKIh69eqZs0ObN2/O1q1beeedd/I9R506dVCpVHzyySf06NGDmJgY\nQkNDycjIsKrR6eLiwpEjR2jSpAl16tQhMDCQRYsWkZWVRbNmzTh//jwLFy6kS5cu8j70mEjARpQI\nSqWSkJAQvv76a3788UfWr1+PWq3G29ub1atX06pVKwAGDBiAra0ta9asYf369VSsWJFRo0Y98CJk\nyRSAmTt3LpMnT0atVtOmTRsmTpyIUqnExcWFdevW8dlnn5kDNA0aNGDt2rXmdMqC6NWrF7/++itD\nhgzJdbwrGC/AERERrF27luXLl+Ph4cGkSZP46aefzEXUatWqxcsvv8ySJUuIjo7O8Vo7dOhASEgI\nISEh/N///R+lS5fmrbfeYsSIEQVuqxBClERubm588803fPLJJ4wbNw6FQkHDhg358ssv8fT0fKRj\n+/j44Ofnx0cffYS9vT2DBg2yGt8/efJkypQpw5o1a7h9+7Z5Ktz+/fvnecyaNWsye/Zsli1bxpYt\nWyhfvjydO3fGYDDw/fffo9frUSqVDB8+nODgYK5cucKPP/7I+++/T1xcHHPmzMHOzo7evXvj7e1N\neHh4vq9hyJAhODg4sG7dOpYuXUq5cuUYPHhwjjoFQohnS0Hvq6Fg99bz589n0aJFhIWFkZiYSO3a\ntVm+fHmBZ4h6kFq1arF06VLmzZvHsGHD8Pb2ZvTo0cydOxd7e/t89/X19eW3337LdQptf39/zp8/\nb7VOoVCwZMkSZs6cyciRI3FxcaFFixYsWLCAUaNGceHCBby9vRkwYAAffvghQ4YMYfv27fj4+LBq\n1SoWLVrE8OHDcXR0pHXr1owdOxYbG5vH0g95cXZ2Zt26dXz66ad8/PHHKBQKmjdvzsaNG80z2oIx\nYAPGjCWTFi1asHXr1nyHQ4ExW2bOnDksXbqU7du3U758eV588UU6duzIxo0b0el0qFQqhg0bxuLF\ni7l8+TI///wz77zzDnZ2dubPMuXKlWPgwIF51rURD09hkKqjQgghhHiGjBkzhhs3brBhw4bibooQ\nQjzz/vjjD1xcXKyK8G7cuJH58+dz+PBhGeIpnmmSYSOEEEIIIYQQolicPHmSdevWMW7cODw8PIiM\njGTBggX06tVLgjXimScBGyGEEEIIIYQQxSIwMJCUlBRCQkK4ffs25cuX5+2332bIkCHF3TQhip0M\niRJCCCGEEEIIIYQoYWSuLSGEEEIIIYQQQogSRgI2QgghhBBCCCGEECXMU1fD5vbtpELtp1AoKF3a\nkTt3UpBRYtakb/ImfZM/6Z+8PUrflC3r/IRa9e9S2Ov9w5LfY+kDkD4A6QMo+j6Q671RYa738vua\nN+mb/En/5E36Jm+P2jd5Xe8lw+YepdLYyUrpkRykb/ImfZM/6Z+8PY19Exsbi7+/P3v27AHg9OnT\n1K1blyZNmpj/hYaGAmAwGJg/fz4tW7akRYsWzJo1C51OV5zNz9PT+LN6WNIH0gcgfQDSB/8m8rPK\nm/RN/qR/8iZ9k7cn1TdPXYaNEEKI4hEUFER8fLz5+3PnztGmTRtWrFiRY9v169ezd+9efvrpJxQK\nBYGBgaxevZrBgwcXZZOFEEIIIYQosSQ2JoQQ4pFt2LABe3t7KlasaF4WHh5OnTp1ct1+8+bNDBgw\ngHLlylG2bFkCAwP54Ycfiqq5QgghhBBClHiSYSOEEOKRREZGsmbNGr799lu6d+9uXn7u3Dk0Gg3t\n27dHr9fz0ksvMWrUKDQaDREREdSsWdO8rZeXF5GRkRgMBhQKRYHOW1QpuUqlwur/Z5H0gfQBSB+A\n9IEQQoiiJQEbIYQQhabVahk3bhxBQUG4ublZrStVqhR+fn688cYb3Llzhw8++IAlS5YwZswY0tLS\nsLOzM29rb2+PXq8nMzMTW1vbAp27dGnHAgd3Hgc3N8ciO1dJJX0gfQDSB/Ds9sGWLVsIDg4mOjqa\nSpUqMWrUKDp06EBCQgKTJk3i0KFDODs7M3z4cHr27AkYa5YtWLCA7777Dp1OR9euXZk4cSIqlaqY\nX40QQpR8ErARQghRaMuWLaNu3bq0bds2xzpTgWEABwcHAgMDWbBgAWPGjMHOzo6MjAzz+rS0NNRq\ndYGDNQB37qQUWYaNm5sj8fEp6PXP5owI0gfSByB9AEXfB+7uTk/8HAUVGRnJpEmTWL16NU2bNuXA\ngQMMGTKE/fv3M23aNBwcHDhw4AAXLlxg8ODB1KpVCx8fH6lZJoQQj6BIAzbh4eFMmTKFy5cv4+np\nyfTp0/Hx8cmxXWBgIAcPHrSKvJ88ebLI2qk3GPg7KolTV2LRG8CrgjMNqrtjo5YnAUIIYWnLli3c\nvn2bLVu2AJCcnMzo0aMZOnQo8fHxDB8+HCcn4weOjIwMc0CmRo0aREZG0rhxY8D4QaB69eoPdW6D\nwUBRTiyl1xvQ6R7+A9r128kcDr9FJ18PnOxtnkDLik5h++BpIn0gfQDPZh94eXnxxx9/4OjoiFar\nJTY2FkdHRzQaDb/++ivbt2/H1taWRo0a8corr/Djjz/i4+NjVbMMjPf5ixcvLnEBm6vRSew/dTPH\nz1WlUtCmUSU8K2RPuXvwTDRanZ6AxpVyHOd0xB2u306mk68HyiLMAhVCPJ2KLGCTkZHB0KFDGTp0\nKD179mTz5s0MGzaMX3/9FUdH67TS8PBw1q9fT8OGDYuqeYDxzffIuVt8vy+CqDupVuuqlHVi/JtN\ncLT7d99sCyHE47Rt2zar79u3b8/kyZNp27YtHTt2xGAw8NFHH3Hz5k1CQ0Pp1asXAF26dGHVqlW0\nbNkStVrNihUr6Nq1a3G8hCduzZbzREYlciYijqkDWxR3c4QQotAcHR35559/6NSpE3q9nmnTpnHt\n2jXUajVVq1Y1b+fl5cWOHTsAiq1m2cPWG/rm14tcup6Q67obt1MI6t8MgLjEdFb+Eg6AZwVnvCq5\nmLczGAys+OksqelaalRyoW4194drdBGRWkz5k/7Jm/RN3p5U3xRZwObQoUMolUr69u0LwOuvv87a\ntWvZt28fnTt3Nm93584d4uLiqF27dlE1jdR0LT/+HsGJi7HEJaabl5d1s8Neo+afmGSu305m8aZT\nfPSGD7Y2kmkjhBD5USqVhIaGMmvWLFq2bImdnR1vvPEGAwYMAKBv377Exsby+uuvk5WVxauvvsrA\ngQOLudVPRmRUIgBXbyWRmaVDI+8hQoh/sYoVK/LXX39x7Ngx3nvvPd59912rmmQAdnZ2pKcb76mL\nu2ZZQeuxGgPtAAAgAElEQVQNpWZoAfCq5ELFMsZ9bt5O4e+oRDK1evPwtORMvXmf20mZNLMYtpae\nqSU13XgcnUJZooa05eZZrcVUUNI/eZO+ydvj7psiC9hERkZSo0YNq2VeXl5ERERYLQsPD8fR0ZHA\nwEDOnz9PtWrVGD9+PE2aNCnQeQoTgd/75w1+PXbd/H3D6qXp1saL6pVcUCgU/PbXTb745RyXryfw\n428R9H2h6IJJJYFEUvMmfZM/6Z+8Pa19s3v3bvPXNWvW5Msvv8x1O5VKxahRoxg1alQRtaz4lHK2\n5W6SsV7PyUux+NUrX8wtEkKIwlOrjR8f/P396dixI2fOnLGqSQaQnp6Og4MDQLHVLHvYekPpGcYx\ntm0bV6Jd08oAbDt8zRiwydIRF5cMQGpK9muJuZNsXg4Qn5S9LjYuxWpdSSL1qPIn/ZM36Zu8PWrf\n5BXgLbKATWpqKvb29lbLLKPvJhkZGfj4+DB27Fg8PT3ZtGkTgwcPZuvWrZQtW/aB5ylMBP7FVtW5\nk5RBpbJO+DeoiGdFF6v1XdvVJj5Vy6bdl/j9VBSDXmuEne2zV69ZIql5k77Jn/RP3qRvnn5O9jbm\ngM0fp6MkYCNEARgMBqKjo6hYMWeNEFE89u3bx5o1a6wC8VlZWXh4eLB//35u3rxJpUrGn1dkZKR5\nGFRx1ywraL2hLK3xBCqlInt703+G7GNotdkZNkmpWVbHTkrNMn+dlqGzWqfXG0rcQ5pnsRbTw5D+\nyZv0Td4ed98UWdTB3t4+R3DGMvpu0qFDBzp06GD+vm/fvmzYsIHDhw/zyiuvPPA8hYnA2wDvdK5j\njojlFg0PaFieH/ZeJiVdy7Y/Imjj8+zcQEgkNW/SN/mT/snbo/RNSU+xFta0uuyb+7N/x3E3KYNS\nzgV/sixEcWrdujm2trYoLW6uSpcuw1tvDeCVV157YuddunQxACNGfPjEziEeTr169Thz5gw//vgj\nXbp04bfffmPfvn18++23REVFMX/+fGbNmsWlS5f45ZdfCAsLA/49Ncuy7l2rbdTZv+umZ8CW79N6\nQ/bXKWnZARqAtHvDqgAyMrO/PnAmii+3nqdPh9q0a1L5sbZbCPF0K7KATfXq1fn666+tlkVGRuYI\nwmzbtg29Xm9V18ZyZpEHedRZQ/KKiDnba/CpVYbjF26z+8QNWjWsWPiT/EtJJDVv0jf5k/7Jm/TN\n009ncaNvMMDR8zF0bFE1nz2EKFlWrlxL9eo1UakUuLra8+23/2PGjKk0aNCYatW8nsg5ExLicXV1\neyLHFoVTtmxZQkNDmTNnDjNmzKBatWosXbqUGjVqMHPmTKZOnUrbtm1xcHBg7Nix5oyaf0vNsqx7\nmTMai1lhTRkxFjEaLJ+xpKRnB2Uguw4OQHpW9geSr3dcRKszsG77Bf7jU6nQ9XiEEM+eIgvY+Pv7\nk5mZybp16+jduzebN28mNjaW1q1bW22XmprK/PnzqV27Np6enqxdu5b09HRatWpVVE3N0398KnP8\nwm0ioxK5disJj/LOD95JCCHEM01nkWEDcDj8lgRshBWtTm816cGT5O5ih1r1kKnIFlQqFZ06dWbR\novlERl6hWjUvEhMTWLz4cw4fPoSdnR1du/bgrbcGoFAoSEiIZ9Gizzl9+i/u3o2jSpWqfPTRBBo1\n8gHghx828c0360hIiKdu3XqMGxfEb7/tZceOrSgUCqKjo5g161OOHj1EaOhS/vnnGpUqVSYw8D38\n/Y33kK1bN6dbt57s3LmNN9/sT79+JS8Y8LRo3rw533//fY7lbm5uLF68ONd9/g01y/QGA9p7D08s\nM2xM03JbZtUY9AXNsMkO2KgshkL9HZ2E133lF4QQIi9FFrDRaDSsXLmSadOmsWDBAjw9PVm+fDkO\nDg5MmTIFgBkzZtC9e3du377NoEGDiI+Pp169eqxcuTLH0KniULdaKUq72HEnMZ2/LsdKwEYIIcQD\nmT4E+NYtx5FzMURGJRJzN5VypYr/fU0UP61Oz6SwQ8QmFE3ApoyrHXOGtCx00CYzM5Nvv91ARkYG\n9es3BGDmzCm4uLjy3Xc/ER9/l3HjPsTd3Z2XX+7CsmVLAFi//juUShWLF88nNDSEZcu+4NChA4SF\nLWP+/CV4e9dlxYqlzJw5hdDQ1Vy5chlXVzdGjPiQiIgrjB//EVOnzqRVqzYcPXqIyZMnsGLFl9So\nUfNeuzL4+ecdZGVlPp6OEs8Uy7o0uQ6JMuQ+JCo53Tpgk5qee8CmtKsdKenGkguHw29JwEYIUWBF\nWjm3Tp06/Pe//82xfMaMGVbfBwYGEhgYWFTNKjClQoG3hxsHzkQTcTOxuJsjhBDiX8A0JKqBV2nC\n/75LcloWv52KokfbGg/YU4iSYejQd1EqFWRmZgEGWrb0Z9Gi5ZQrV547d2I5dOgAv/yyE3t7e+zt\n7enbtz+bN3/Pyy93YciQ97Czs0OlUhMVdRNnZ2du374NwK+/buell16mXr0GAAwcOJirVyNznH/X\nrh00b96Ctm3bA+Dv35pWrdqwY8dWhg17H4AOHTpiY2ODjY1N0XSKeKpk5hGwMWXYWA6Jsvw6vwwb\nyyFRKWnZy4+ej6FX+5rmYwshRH6evamOHlGNSi4cOBPNlZuJGAwGGYMqhBAiX6aiwxobJS3qlGPP\nyRv838GrXLuVzJAu9XC0kw+YzzK1SsmcIS1L9JCo0NBVVK9ek1u3bhIUNA5XVzfq1zcGWW7disZg\nMPDGG9kFiPV6Ay4uxgyC2NhYFi/+nL//jsTT0xNnZ1cMBuPfRFxcHDVq1DLvZ29vT5069XKc/+7d\nOCpUsK4dWKFCRW7fjsl+Xe5lHuo1CWEpyzJgo7LMsLk3JCqvosP51LCxzLBJscjEuZuUwaV/4vH2\nKAUY3yOOnY/B26OUFKQXQuQgAZuHVL2SKwDJaVncjk+TlHYhhBD5MmXYqFVKugZ48U9MMpdvJHA6\n4g7//fUS776S8wOqeLaoVcp/xf1EpUqVWbZsGV27dqVChUoMGPAupUuXQaVS8dNPO9BoNAAkJiaS\nmpoKwNSpk+jatTtLl65EoVCwdesvRERcBoxFbGNjs4MuKSnJrF69kqFDR1idt3z5Cpw9e8ZqWVTU\nTcqWLWf+Xp6fiUeRpcsjw+belwbLIVEWwZssrd7qAW5uRYe1Oj3pmdYzopyJjDMHbL7cep4DZ6Lx\nrODMlAHN5WGwEMJK4avOPaOqlHNEc+9CfkWGRQkhhHgAU4aNSqnAxUHDxLea0i3AOLPOH2eiOXf1\nbnE2T4iHUrlyZT74YDRr1qzk8uVLlC9fgcaNm7B8eTAZGekkJiYwefJ4Vq5cCkBqagr29nYoFAr+\n/juSb775Cq3W+KG2Y8eX2Lbt/7h48TxarZavvlpDePgZ89CmlJQUAJ5/viMnThxl//696HQ6Dh78\ng99/30+HDh2LrR/E0yUrzxo2pqLD2dsa7pvYMS0jOxiTlksNG8ssnOqVjJlnl68nAPDH6SgOnIkG\n4Gp0EuevxT/KyxBCPIUkYPOQVEol1e4VCou4IQEbIYQQedPrDeabe9MwFIVCwcv+1fCsYCxcv277\nBasUeyFKupdf7kKTJs2YO3cGOp2OadNmExd3h9df70Lv3t0pU6YMo0ePB2DcuEl88806OnZsS1DQ\nWF566RXi4++SkBBPs2YtGDZsJFOmTOLll5/nypVLTJs2G4B27Tqwd+8uRo8eQZUqVZk793O+/HIl\nL77YjmXLFjN16izq1q1fnN0gniKWRYfVudawyX1IFEBSWnah69yGRFnWuWlc0zh0LzIqkZj4NNbt\nuGB1rB1HrhX6NQghnk4yJKoQalRy4eI/8Vy5mVDcTRFCCFGC6fTZHwIsp3VVKhX06+jNrK+OER2X\nalXPQIiS5Pffj+W6fOHCpeav3d1LM336nFy3a926La1bt7Va1rdvf/PXr7zSlVde6ZpjvxYt/Ni6\ndbfF9y1p0aLlQ7VRiILKu4aN8X99HkOiAJJSsyh/7/Kd25CoZIuAjU/NMvywP4JMrZ5vdl4kM0uP\nk70Nrz5XjQ27LvHXlTtE3UmhYmnHx/bahBD/bpJhUwimdMZ/YpLJzNI9YGshhBDPKtOU3kCOQq/V\nK7lQpazxpvxw+K0ibZcQQohsWdrs+3mNTc4MG+shUfcHbLIzbNJyy7C5V3BYpVRQpawjrk7GWk+n\nrtwBoGX98rRrWtlccPjH33LOlCaEeHZJwKYQTIWHdXoDN2JTirk1QgghSiqdxV2+SpWzkKRfvfKA\ncZpXrUXRSyGEEEXHVHRYqVCgUuasYWOwmiXKet+k1OwMmlTLGjZZOvQGg3lKb0d7GxQKBbUqu1rt\n/1yDCqhVSl5tVQ0wvh+cjrjz6C9KCPFUkIBNIbg5acyFh+8kFM00nEIIIf59LIMwlkOiTPzqGgM2\nKelazkbGFVm7hBBCZDMNibIsOAzZs0Tp86lhYznkyTLDBiAzS2de72hnrERRs4qbeX3F0g54ljfW\nM2vTuBI1Khuz+Ndtv8C5q3ethtUKIZ5NErApBIVCQWlXOwBiJWAjhBAiDzqdZYZNzrfcMm721Kxi\nfNr626moImuXEEKIbJl5BGzMGTYFGBKl1xtyTN+dkakzD4lytLcBoFaV7Awb//oVzOdQKhQM6FQH\nlVJBbEI68zacZOKKQ0TIrLRCPNMkYFNI2QGbtGJuiRBCiJJKa/F0VJ3LkCiANo0qAXDy4m1uxaUW\nSbuEEEJkyzPDxlTDxnJI1H1JL6YhUWmZ1tk1YCw8bJrW28nOGLCpWs6JKmWdcHXU0KphRavtq5Rz\nYkiX+ub6ZrEJ6cz9+jj7/7pZ2JcmhPiXk1miCqmMqz0gGTZCCCHyZplho1bm/oykZf3yfL//CvHJ\nmWw/+g/9O3kXVfOEEEJgEbBR5T4kyoAxs0ahUOSc1vtewMayfo1JRqbOPK23o73xY5dapWTqwObo\n9QZs1Koc+7SoU44Wdcrxd3QioT+eJSY+jbXbzlPOzZ46njKboBDPGsmwKaQy9zJs7iRKwEYIIUTu\nrGrY5JFho1YpeaFFVQB+PxVFQkpmrtsJIYR4MvLKsDENV4LsYVF5DYm6v34NQHqmZQ0bG/NylVKZ\na7DGUrUKLkx5uwWVyzpiMMCKn87K+4MQzyAJ2BRSaZfsGjb3X7iFEEIIsJ4l6v5pvS21bVwZe1sV\nWp2e7UeuFUXThBBC3GOaJUqdx5AoyC42nNcsUblm2GRl17BxsrfJsf5BHOzUDOvaAFsbFQkpmQT/\n71SugSEhxNNLAjaFZMqwMRYTkwunEEKInKyKDucyS5SJg52aF5obs2x2n7hOojxFFeKpcvPmjeJu\ngsiHKcNGkyNgk/216QGt/r6IjSkgYwqkqFVKbDXG7BnjkKjsab0Lo1IZR95+qQ4AETcTWbxJgjZC\nPEukhk0hmQI2YCw8XJiouRBCiKebrgBFh01eaFGVncf+IS1Dx7bD1+jVvuaTbp4QBXLo0AE2bFjH\npUsXUSigTp26DB78HnXq1Huk465atYLIyCvMmvUZX321mqtXI5k8eeZjarW11NRUOnZsw3ff/UTF\nipUKdYwTJ44xefJ4jhw58lD7Xbx4nrFjP2Dz5u2FOq948rQFGBKlz2NIlPZeYD71XhDFwVaFQqEg\nI1NnHBKVbj2td2H41StPanoW63Zc5OI/8UxaeYg+z9eiRZ1yVm38N9HrDej0BnR6PXq9Aa3eYFym\nM6AzGNDp9BbbGNfpDQYMBovgmbG4EHqAe8vvLbL42gAG08/v3v73lltuh8WPVaEEJyc7kpPTMejB\nQN6jKQo70CK//fI7XyFXPTYKBTg62pGSkl7o1/60UiigWpVSeJaxf6zHlYBNIbk4arBRK8nS6rmT\nkE61Ci7F3SQhhChWsbGxvPrqq8yZM4d27doRHR3NjBkzOH78OGq1mhdffJHx48ej0WgwGAw0a9bM\n6sa3WbNmfPHFF8X4Ch4/rVWGTf5JrY52NnRoVpWfD/zNnj9v8Gqratjbytu0KF4//fQDX3wRyvjx\nH/Pcc/64ujqwcuVqRo4cRmjoaqpXr/FYztO//zuP5TglUXJyMlqtZESUZHkWHbYM2OhNQQLrT6mm\nwLwpYGNvZ2N8b0sxZt9k3Jvqu7AZNibtmlYBYMOuyyQkZxK6+Sy/n4rizY61KV/K4ZGO/TD0BgOJ\nKZncTcogJT2L1HQtKWlZpKRrSUk3/p+WriVDqyMzS0/Wvf8zsnRkafVk3vted//YMiGeEhPfakqt\nKm6P7XhyJ1hICoUCdxc7bsWlykxRQggBBAUFER8fb/5+7Nix1KpVi/3795OYmMjw4cNZunQpo0aN\n4urVqwCcOHHiX/t0sCBMN/IKBSjzGRJl8nyzKmw5dJWMTB2Hw2/xnyaVn3QTRQmg1Wu5m55QJOcq\nZeeKWlmw27/09HRCQhYxdeosWrUKQKVSYGtrS9++/YiLu8vVq5FUr16DuLg7LFmygKNHD2Fra0eH\nDh0ZNGgYGo2G1NRUli8PZt++3QA891xrRowYhZOTk9W5LLNtZs+ehqOjIxcvXuDSpQt4eFRj3Lgg\nvL3rYDAYWLNmJd9//x0ajYaePfsQGhrMf//7w0Nlzhw7doRVq0L5559rZGZm4evbko8/no6dnR1J\nSUksWvQZf/zxG2q1DZ07v8KwYSOt9k9JSWbEiGHUqlWbceOCyMzMYPnyYPbu3Y3BYOCFF14kMHA4\nyclJjBnzAZmZGbzwQgCbNv2Mq+vju5EXj0em1hhUyZFhY/GtKU5z/7TeBoMxmJOWnp1hYxoOG5eY\nYd7Oye7Rs/HbNa1CPS93vt5xkbORcZyJjGPyF4fp0LwqL/t7WhU2LiyDwUBSWhax8enEJqQRm5Bu\n/Bef/bVlQf2SSIHxs5pCYXz/BYVxeJsCFGQvN30N1tlUxvUKcxaOaVl+58t7Zd5r89+vcOd7lHuq\ngu6qVCrR3/+HIACoUNqRSmUcH+sxJWDzCMq4SsBGCCEANmzYgL29PRUrVgQgMzMTe3t7hg0bhq2t\nLWXLluXVV19l586dAISHh+Pt7f2INxYKHpC08liYAi0FCbjcz/QAUa1S5jlLlKVSLra0qFuOQ2dv\nsffkDdo3q1wiAlqP0gdPiyfVB1q9lhmH5nEn/e5jPW5eStuVYnqrcQUK2pw9+xc6nZbnnnsOlUph\n1QcjRmQHMCZNGkulSpX43/9+JiUlhQkTxrBmzQree28k8+bNJi4ujq+/3ohKpWL69Ml8/vkcZs6c\ni1KpQKFQmI9t+lqhgO3bt7B0aRhVq3owe/Z0wsJCWLRoKT///BNbtvxMWNhq3N3dmT59CjqdDpVK\nke/fmGmdSqUgMzOdoKCxTJ06k4CAtsTE3CIw8F127dpOly6vMX/+XNLS0vjf/35Gq9UyYkQgVat6\n4OHhAShIS0tjzJgPqVWrFhMnfoxCoWDhwsVcv36Nr7/+L3q9gaCg8axbt5rBg4eycOESJk0ax7Zt\nux/thyeeGFPR4fsDNrkVHc5tshGdXp+dYWOrNmfsWM4ma5rW+1GVL+XA6F6NOXo+hg27LpGQnMm2\nw9fYffw6vnXL06hGaapXcsHN2daq/ZavIyUli7up8Vy5FkfM3bQcwZmMLF2B22Nvq8LB1gZHezWO\ndjY42qlxsFOjsVGhUavQ2Chz/d9GrUR9729fpVSiUpq+zv6nVCpQqe6tu/eer1Ao7gVksoMTSmP0\nxRyoeVQqlQJ3dyfi4pKtatEJ6Zv8PKm+kYDNIzBP7S0BGyHEMywyMpI1a9bw7bff0r17dwA0Gg1h\nYWFW2+3Zs4c6dYyFE8+dO0dycjJdu3YlJiaGFi1aEBQURPny5Qt83tKlHYs0mOHm9vBPTOzsjVkT\napUSd3enB2xt9Np/anHo7C2uxSRzOymLOtXcH/q8T0ph+uBp87j7QKvTosxnBrHHTalS4l7KCbXq\nwbeAWm06rq6ulCtnnRFi2QfXrl3jzJlTrFy5And34+/qRx+NYsKECYwZM5q9e3ezYcMGatQwFtWe\nPDmIzp07M3/+POztNWg0atzdnay+trW14fnnn8fPrykAr73WhU8++QR3dyd27drOwIFv07ixsX7O\npEkTeOmlfbi6OuT7N2Zra7xWuLo6UKGCOz/++CMeHh4kJSWRmZlC6dLuJCfH4+SkYd++PWzatIlq\n1YwZO2FhK9BoNFy9ehWDQc+IESPQaGyYN+9T81P4LVt+ZsOGDXh5GYetjB79IaNHj2b8+DE4O9sb\nM7MLeA0oyY4dO8ann35KREQEpUqVYtCgQfTu3ZvTp0/Tq1cv7OyyazwGBgYydOhQDAYDCxYs4Lvv\nvkOn09G1a1cmTpyISpX/tNZFKa9pvXOfJSrnBzGtzmBRw0ZNutIY8LAK2DyG7BcThUJhDs5sOXSN\nHUeukanV8/vpKH4/HQUY33dcHTXY26pRqRTodAaS0zJJTMnK9TXkRqNWUsbNnjKudvf+Gb8u62ZP\nKWdbHO3VDxzuK4R4NBKweQSmgI1k2AghnlVarZZx48YRFBSEm1vuaf4Gg4HZs2cTERHBvHnzAGNA\nx8fHhw8++ABbW1tmz57N+++/z7ffflvgc9+5k1JkGTZubo7Ex6fkmB3kQRIS0wDjDFFxcckF2qeC\nq4bKZR25cTuFH/ZeIrBL/Ydu8+P2KH3wtHiSfTC15Rjupsc/eMPHoJSdG4kFvG+xtXUkPj6BmJi7\nqNU2Vn0QH5+Ag4MDkZHXsbe3BzTm33FHRzdiY2OJjLxOVlYWjo5u5nX29q4YDAYuXIgkLS2TzEwt\ncXHJVl9nZGTh6pq9T1paFlqtjri4ZKKionF2LmV1PICEhFQcHPL+G0tNTbXYLo1fftnGxo3rAahZ\nszbJySmkpmZw9epNsrKysLV1Mp/DyckYiEpKSiMxMRG9Xs+pU6c4deocVat6EBcXR3p6Om+99ZY5\niGwwGMjK0hIVdYekpDQMBkOBrwGWSlKQJyEhgffee4/Jkyfz8ssvc+7cOQYOHIiHhwfXr1+nTZs2\nrFixIsd+69evZ+/evfz0008oFAoCAwNZvXo1gwcPLoZXkbvsGjbWQSTLZwLmIVG5/Pnr9AbzzE2W\ntcfi7gVsVEoFdprHH6Cy06jp3qY6HVtU5cDpKI6cj+FqdBI6vQGtTm8VMMqNWqWgtIsdZdzsKetq\nR+l7wZjSrnaUdbXH2cGmRGR5CvEsk4DNIyjtci/D5t4NuRBCPGuWLVtG3bp1adu2ba7r09PTGTdu\nHBcuXGDdunWULl0agPfff99qu/Hjx9OyZUtiYmIoV65cgc5tMBjQFTxr+5GZZq94GJn30spVSsVD\n7fsfn8qs33mRI+Ex9G5fq8TMRFiYPnjaPIk+UKDC3bb0Yz1mngwUuP116zbExkbNH3/8QevW2X/j\ner2B2bNn4ODgQGDgcNLS0oiLu2uuzXL9+g1cXFxxdy+LRqPhxo0onJ2z1ymVSlxc3NDrDff+jg1W\nXxtnbslup6lUgk5noFy58kRFRZnXRUffMq+7/3XNnj2Nfv0G4uHhSUaGcaYetVrDn3/+yapVYaxc\nuZaqVT0AGDlyKHq9AWdnN2xsbIiOjsHJyRgMOnz4IAkJCZQpUwZnZ2dWrVrFhAlBzJkzk+DgFTg5\nuWBjY8Pq1eupXNmYYWPskzuo1Rpzu/7tfzs3b96kbdu2vPrqqwDUr18fPz8/Tpw4QWxsrDmD8n6b\nN29mwIAB5mt7YGAgixcvfqiATWGGwD7MMEZTTRaNxnr4qtoi40ahMA55yP1o2QEbR3sb82w9SanG\n3zsnexurYz1urk4aXvL35CV/TzKzdNyMTSE2IZ3ElEzSMrToDcb2O9nb4OZki5uzLZ6V3VDodUUz\ntdC/jAwDzpv0Td6eVN9IwOYRODtqAEjL0KHV6VEXYUqzEEKUBFu2bOH27dts2bIFMM6GMnr0aIYN\nG0avXr0YNGgQDg4ObNy40SoDJywsjFatWlG/vjF7JDMzEwBbW9uifxFPkGkWjAdN6X0///oV+G7v\nZTKz9Px+KooX/TyeRPOEyJetrS2BgSP47LM5KJUq/P39SU5OZtWqMI4dO0Jo6CrKli1Hs2a+LFky\nn48+mkhKSjKrVq2gY8cXUSqVdOz4EqGhwUybNgelUsmyZYvx92+Vo+hwQXXu/CorVy6nVas2lC5d\nhrCwZXluGxFxhZ07t/H224PYuXMrpUuXoVQpdy5evIBKpcTW1hadTseOHVv566+T1K/fEJVKRfv2\nL7BqVSgffzyd9PR0li5dRM+efQBjsU2lUsl7771P79492Lz5e157rQcvvPAioaEhjBsXhFqtZt68\nOURHR7Fs2RdoNBoyMzPJysrCxqZkBF8Lo27duuYsSTBm3Bw7doyuXbuycuVKNBoN7du3R6/X89JL\nLzFq1Cg0Gg0RERHUrFnTvJ+XlxeRkZEYDIYCZ288yhDYggxjNNwLw7g42VllNSVnZhdWdXV1wN3N\nHjt7TY79nZwdzJk3ri52qNXW2TRl3fMfsve4VSjvWmTneprJMOC8Sd/k7XH3jQRsHoGDRcpjeqYO\nJ3sJ2Aghni3btm2z+r59+/ZMnjyZ//znP/Tv358yZcoQHByc40NKREQEv/32G0uWLEGtVjN79mye\nf/55XF2frptM0xP1hx3j72CnpmW9Cuz/6yZ7/7xBR9+quRaPFOJJ6969J05OzqxZE8bMmZNRqVTU\nrVuf4OAVVK9u/BA+depMFi/+nF69ugDQseNLBAaOAGDkyNEsW7aEAQPeICMjk4CAtowc+VGh2/PC\nCy8SGRnB4MEDsLe3p1OnzgC5BkLGjJnAvHlz2LBhHRUqVGTKlJnG2h++LWnXrgP9+/dGpVLi7V2P\nl156hatX/wZg1KhxLFkyn969u6NQKOjatTuvvvoaJ04cMx/bxcWF998fxfz5n9CqVQAffjiGZcuC\n6devF+np6TRu7MOMGXMBqFGjFtWqVadz5+dZs2Y9VapULfTrLymSkpIYOnQo9evXp3379mzatAk/\nP/kVEQ8AACAASURBVD/eeOMN7ty5wwcffMCSJUsYM2YMaWlpVrVt7O3t0ev1ZGZmFjhIX5ghsA8z\njDEt3ZgJo7s39M4k0SKLPu5uMkq9juQU4zAjpUJhrgVzJy6Z9HsZNtpMLQaD9Qw6HmUdCzUk7kmR\nYa75k/7Jm/RN3h61b/IK6ioMuZU6/xe7fTupUPsVpqpzdFwqk8IOAfDJUH/KudkX6twlnVQDz5v0\nTf6kf/L2KH1TtqzzE2rVozMFbFxdXenTpw+2trZWhSXr1avH+vXrSU5OZvbs2ezZs4esrCz+85//\nMGXKlIcK2BT2ev+wHuVntePINf67+zIVSzswe3DLh9r3anQS0788CsCI7g1pWrvsQ+3/OMnfsvQB\nlIw+uHTpIqVKuVOmTBkA/v47kv7932Dnzv3Y2to9YO9HV9R9UBKv9//88w9Dhw6latWqLFq0yCoY\nY7J9+3YWLFjA9u3badq0KWvWrKFx48YAXLx4kW7dunH27NkCn7Mw1/uH+VlN/uIwN2JTeKN9TTr5\nZmc03opLZeK9e/1Ph/pT1s2e7UeusXH3ZWxtVObZlGYP9mP5j2e5fjuZnv+pgVZv4If9EebjDOxc\nh4BGBZ92/kkrCX/LJZn0T96kb/L2qH2T1/VeMmwegWVRsbR0bTG2RAghSobdu7Onrb1w4UKe2zk5\nOTF37tyiaFKx0uoLl2ED4FnBmXrVShH+911+PvA3TWqVkeKP4pl36NABjh07wty5n6NSKVm/fi0+\nPk2LJFgj4OzZswwaNIguXbowfvx4lEolCQkJhIaGMnz4cPNQt4yMDHP2TI0aNYiMjDQHbCIjI6le\nvXqxvYbc5DVLlOUl9/5ZomzUSnPARqc3oLtXbEmlUnLfiCiqV3R5Es0WQjwDZAzPI3Cwzb4amwqN\nCSGEECa6e4UsH7aGjcmrz1UDjNk2ZyLjHlezhPjXeuONvlSsWJFevbrQpUsnkpISmTx5RnE365kQ\nGxvLoEGDGDhwIBMnTkR5LxDt7OzMzp07CQkJISsri6tXrxIaGkr37t0B6NKlC6tWrSI6OprY2FhW\nrFhB165di/Ol5JCpNQZebFR5T+ttniVKnx2wMdHpDObCxWqVAluLGaFsbVRULC31PoQQhVOkGTbh\n4eFMmTKFy5cv4+npyfTp0/Hx8clz+4MHDzJw4ECOHz+Oo2PJu9DZqFWoVQq0OgOpErARQghxH63O\nVHS4cM9Hald1o1YVVy5dT+DnP/6mgZe7ZNmIZ5pGo2HChMnA5OJuyjNn06ZNxMXFsXz5cpYvX25e\n3r9/f0JDQ5k1axYtW7bEzs6ON954gwEDBgDQt29fYmNjef3118nK+n/27jy+qSpt4PjvJmnaJi3d\nKDuUssgmgohAgREVxQFZxhFBGR1kBCngMsUdRkQWd5gXHIeCiBuMOoyOOIjLIIICAjLiwqpIWQuU\n7nuz3fePLE3aJk3bdH++H/k0Pbn35uQgNzfPfc5zzIwbN45p06bV19uokPcMm9LzrTNQ4yxN4R7c\nsdhsHud79yW849qEy4o6Qohqq7OATUlJCYmJiSQmJnLbbbexadMmZs2axdatWysMxuTk5DBv3jwa\neomd0GAdeYVmybARQghRjsWZIl/Ni3VFURg3rDPL3/uB4+dyOHo6m15xUYHsohBC+MV5He/NG2+8\nUWG7VqslKSmJpKSkWupZzZmtFQds3AMtzqlQqlpxho3VNQVWITioNGAT37bh1SESQjQedRaw2bNn\nDxqNhilTpgAwceJE3nzzTXbs2MGYMWPKbb9w4ULGjBnD2rVrq/Q6iqJUuYo8VH/ddIMjYFNstqKt\nZsp7Q1dba8o3BTI2vsn4eCdj0zxYXXdcq//33KdzNPFtw0k5n8fm3SclYCOEEAGkqqpfNWzKTonS\nuQdsbKprCqxWq3hkVXaIrbvlvIUQTU+dBWxSUlLo2rWrR1t8fDwnTpwot+1HH31Ebm4uDz30UJUD\nNjExxhqli1d13fRwo56LWUWg0XhdiqupCPSa8k2JjI1vMj7eydg0ba5lvas5JQrsNyLGDu3My+//\nxJFTWRw/m0O3Dk1r+XMhhKgvVpvqCsb4qmHja0qU1eo2JUqjKZNhIwWHhRDVV2cBm8LCQkJDPZe9\nDgkJobi42KMtNTWVFStW8I9//AOz2Vzl18nIKKh2hk111k3XO6LrGVmFZGbmV/2FG4GarinflMnY\n+Cbj411NxqapB4ebEmsNp0Q59evWkg6xYZy9lM/mb07y59v6BaB3QgghnNk1AEFBnss7eWTY4Dkl\nSqPYz+1Wm4rF5l50WEPX9i347eBOtDDoaddSbswIIaqvzgI2oaGh5YIzxcXFGAwG1+82m43HHnuM\npKQkWrduzdmzZ6v8OqqqYrVWv582m1qlddND9PYhLCw2N/m16Ks6Ns2JjI1vMj7eydg0bZYAZNiA\n/S7v2KFxJG86xI+/ZnDqQh5xbaQughBC1JSzfg1UkGHjXsPGsZmzlo1Go7gCNu41bHRaBUVRmHRd\nt1ruuRCiOaizZb27dOlCSkqKR1tKSgrdupWezC5cuMAPP/zAwoULGThwIOPHjwdgxIgR7N+/v666\nWiWhjqW9C0tqECUSQgjRJDkzbGpSw8ZpYI9WtIm23+T4z+6TNT6eEEIIsLhn2JQtOuyxrLcjw8ax\nuaIorvqVzmXBoeYZlUII4a7OAjYJCQmYTCbefvttzGYz//rXv0hPT2f48OGubdq1a8ePP/7I/v37\n2b9/Px999BEAO3bsYODAgXXV1SoJDbZn2MgqUUIIIcpyZdgE4AJeo1G4OSEOgO9+vkTK+dwaH1MI\nIZo7k4+AjfuUKGdmjc1jSpR9+xKTW8CmhhmVQgjhrs7OKHq9nldffZWPP/6YQYMGsX79elatWoXB\nYGDBggUsWLCgrroSUAYJ2AghhPCiNEU+MB+3Q/q0pm2MPcvmX9t/dd3xFUIIUT1mvzNs7D+dARtF\nU5phU2IuDdgE6nwvhBBQhzVsAHr27Mm7775brn3RokUVbt+hQweOHTtW292qEcmwEUII4Y2rCGV1\nquFXQKvRMHFEV17+wL5i1MGUTPp2iQnIsYUQojnyFbBRKlglyjklSqMo6BzZkyaPgI1MiRJCBI6E\ngGvIGbAplICNEEKIMqyOgI02gBfw/bu3dC3r/c7WXzy+bAghhKgas1v9mfJFh0sfOzMabTinRCml\nU6LMpedhqWEjhAgkCdjUkEyJEkII4Y1zSlQgAzaKojDlhu4oClzILOTTfacDdmwhhGhu3FeJ0vnK\nsHFMiVIdDxSl9NzuXsNGpkQJIQJJzig15MywMZltrtR3IYQQAtyLDnt+3JZYTVwoSKv2cTu3acH1\nV3YAYPPuk6RlF1W/k0II0Yw5sxR1Wo1HzRrwrGFTruiwxj3Dxr3osGTYCCECRwI2NWQIKS0DVGyS\npb2FEEKU8rasd/KPb7B470v8nPVrtY99yzXxtDDqMVts/OO/P0sBYiGEqAZnwKZs/RonZ8zGNSXK\ncarVKFJ0WAhR++SMUkPODBuQOjZCCCE8eVvW+0zeWQDOF1ys9rENIUHcfn03AH78NYPvfk6v9rGE\nEKK5qixg48yyccTfPaZEOYsOe2TYSA0bIUQAScCmhtwDNkXFErARQghRqqJlva02K0WWYgBMVlON\njj+4d2t6xUUBsP6/x8gvMtfoeEII0dw4a9iULTjs5Kxjo1Y0Jcqxj0kybIQQtUTOKDVkCNa6Hkvh\nYSGEEO5cy3q7XcDnmwtdj822mgVYFEXhrpt6oNdpyMk3sf7zYzU6nhBCNDdmcyUZNo5mZ6BGdZ8S\nVUGGjSzrLYQIJAnY1FCQTus6MUvARgghhDtrBVOiCswFrsdmW80/N9pEG7jtOvvUqH1H0vjm0IUa\nH1MIIZoLV4ZNJVOinIEaZ+DGY5Uoj2W95euVECJw5IwSAM5pUVLDRgghhDuLo+iB+6oh+e4BG2tg\npjBdN6A9fTrbp0a99ekxzqUXVLKHEEII8KfosKOGjXNKlGOqq0ZR0DlXiTJZXG0aqWEjhAggCdgE\ngDNgIxk2QojmLD09nYSEBL788ksAcnJymDNnDldddRXXXnstGzdudG2rqirLli1jyJAhXH311SxZ\nsgSrtemttOfMsNG53XEtcJsSZbLVrIaNk0ZRmD6uDxFhekrMVv7+758olLpqQghRKVfAxkvtGWf8\npdyUKI1SLsNGlvQWQgSaBGwCQAI2QggB8+fPJzs72/X7k08+icFgYPfu3axcuZKXXnqJ77//HoAN\nGzawfft2PvroI7Zs2cJ3333HunXr6qvrtcZaSYaNyRq4z40Io55ZEy5Hoyiczyhk1aaDrtcXQghR\nMX8zbFTH6bR0SlRpDRtn0WGpXyOECDQJ2ASAwRWwaXp3h4UQwh/vvPMOoaGhtG3bFoCCggK2bt3K\nAw88QHBwMFdccQVjx47lww8/BGDTpk1MnTqVVq1aERsby8yZM/n3v/9dn2+hVpQu6+2eYeNewyaw\nqzpd1jGSO0ddBsChlEze+vSYa2UTIYSoqf3793Pbbbdx1VVXccMNN/Duu+8CjTujstIaNhpvU6JK\nz+0mR9BH6tcIIQJNV/kmojJSw0YI0ZylpKTw+uuv889//pPf//73AJw6dQqdTkfHjh1d28XHx/P5\n558DcOLECbp16+bxXEpKCqqquu5mVkZRFOri2th5sV6dugTOKVH6II0ry6bA4rlKVKBT6EcO7EBa\ndhGf7j3N1z+exxCi444buvs9rhWpyRg0FTIGMgbQvMcgJyeH2bNn8+STT3LzzTdz5MgRpk2bRqdO\nnXj33XddGZXHjh1jxowZdO/enf79+3tkVCqKwsyZM1m3bh0zZsyo77cEgMVSfjU/d0rZKVGOdo2i\nlDt/S4aNECLQJGATAKGOpb1lSpQQormxWCw8+uijzJ8/n8jISFd7YWEhISEhHtuGhIRQXFwMQFFR\nkcfzoaGh2Gw2TCYTwcHBfr12TIyxRkGIqoqMNFZpe5tNdV3gR0YaiI4OA8CslJRupLW52gNp1sT+\nmKwq2/af4bN9ZwgJ0XPP+D41Hq+qjkFTJGMgYwDNcwxSU1MZMWIE48aNA6BPnz4MHjyY7777jq1b\nt/LZZ5+Vy6js37+/R0YlwMyZM1mxYkXDCdjYfAdsyq0SZSudElV2H8mwEUIEmt8Bm+Li4nIX38JO\natgIIZqrv//97/Tq1YsRI0Z4tIeGhlJSUuLRVlxcjMFgAOzBG/fni4qK0Ol0fgdrADIyCuoswyYy\n0kh2doHrQt0fzroIAEWFJWRm5gOQmZ/jai8sKXa1B9qdN3Yjr6CEb4+ksemrX8nMLuTuMT29finx\npbpj0JTIGMgYQN2PQW0EdKurV69evPjii67fc3Jy2L9/Pz169GiQGZX+ZkNZHX+PQTpNhRmPGrc+\narWKK8PGvm2ZDBtd+aybhqg5Z4r5Q8bHOxkb72prbPwO2IwbN46VK1fSq1evgHagKQjR2zNsis0N\nZz6uEELUhS1btnDp0iW2bNkCQH5+PnPnzmX69OmYzWZSU1Np164dYJ865bxo79q1KykpKfTr18/1\nXJcuXar02qqqUpdlEGw21TXFyR8lptLOaVBc++aZSmvYlFhMVTpm1SjcO643eq2GXQcv8PWP50nL\nKmL2LZcTbtBX64hVHYOmSMZAxgBkDPLy8khMTHRl2bz11lsezzekjMrKsqE0jkiQ0aivMECmc9S2\nCTXYn9c6gt5Gg94V7HEK1usaVJCtMs0xU6wqZHy8k7HxLtBj43fAxmw212nqeWPiTH9srnebhBDN\n16effurx+/XXX8+TTz7Jddddx9GjR1m2bBlLlizhl19+YfPmzaxZswaA8ePH89prrzFkyBB0Oh2r\nV69mwoQJ9fEWao37hbz7HdfaLDpcllajYdrNvYgIC2bLnlMcO5PNU+v2MWNcH3rFRdXqawshmqYz\nZ86QmJhIx44d+b//+z9+/fXXBplR6W82VGGR/TxsNVsrzHh0Fm7Pz7dnRJpM9oz64hJzaUEbB0Wl\n1rImA0my5XyT8fFOxsa7mo6Nt2Cv3wGbsWPHMm3aNG6++WY6duxYbnrU5MmTq9yppsKZ9lQ2yi6E\nEM3Z4sWLeeqppxgxYgQGg4FHHnnElVEzZcoU0tPTmThxImazmXHjxjFt2rR67nFgWa2lU6K0bumx\n+R4Bm9qfSqtRFCZe25X2LY28+dlRsvNNvPjOAa67sj23juiKIUTK2Qkh/HPo0CGmT5/O+PHjeeyx\nx9BoNMTFxTXojMrKsqEsjnO1RqNUuJ3zhrXFaj+O83pfcXvOydsxGqrmnilWGRkf72RsvAv02Ph9\nlbZlyxZCQ0PZtm1buecURWnWARvnhbhEGYUQzZ37Z0RkZCQrVqyocDutVktSUhJJSUl11bU655lh\nY781bLZZKLGaXO0mt8e1LeHyNsS1CWf1R4c4k5bPlwfO8e3RNEYP6cT1AzoQHKSts74IIRqf9PR0\npk+fzrRp07j33ntd7WFhYYwcObLRZlQ6AzbeVnhyxtudRYedPzWKUq5WhawSJYQINL8DNhUFaoSd\nsxiZBGyEEEI4WdwybJwX8e7ToaD2p0SV1a6lkSenDuTTvaf5aNdJ8ovMbPzyVz7/9gy/HdSJoZe3\nqXZ9GyFE45Cdnc2xY8ewWCyu6T5Ow4cP97rfv/71LzIzM1m1ahWrVq1ytf/xj39s1BmVFsed8MpW\niXJe5ztX/1MUxSN70tcxhBCiuqqUB52RkcHGjRs5efIkjzzyCHv37qV79+507969tvrXKGhlSpQQ\nQogyLG7psM7PiQJzocc2ZpsFm2pDo9TdRb5Oq2Hs0M4M69uW/+w+ydc/pJKTb+K9bcf51/ZfubJ7\nS4Ze3pZenaMk60aIJuaDDz5g4cKFmEzls/sUReHIkSNe901MTCQxMdHr8401o9Jq9b2st3Pak+oo\nWOMM3GiU8st4N4YVooQQjYvfAZvDhw/zxz/+kW7dunHw4EHmzJnDrl27eOKJJ0hOTiYhIaE2+9mg\naWRKlBBCiDLcg/jOLwL5poJy21lsFvTaus9qiQoP5o839eC3gzvx8e6T7D1yEZPZxv5jl9h/7BI6\nrYbLOkbQJz6a7h0i6G8MqfygQogGbeXKlUyaNIk///nPhIU1ntWMapPFca7WeVmK19lcNsNGo1HK\nTYHSVbUyshBCVMLvgM2zzz7L1KlTuf/++7nyyisBWLp0KVFRUbz00ku8//77tdbJhk4ybIQQQpTl\nOSXKEbAxlw/YmGzmegnYOLWKDGXamF7cPrI73x5NY+dP5zl+NgeL1cbhk1kcPpkFgKJAm2gDbaIN\ntIwIJTYyhJaRocRGhNDCqMcYElSunoMQomHJysri7rvvlmCNG4vFfq7Wesuwcd6YLVPDRlGUcvtI\nho0QItD8DtgcOnSIJUuWlGufPHky69evD2inGhtXho0qARshhGjoNCknAFAjI1GjoittU2KiwLHU\nomu7iAjU6BifbbqLeYQX5ZIX2gKtRkFzMgXO/0JYXjH54fZslVYXc+HXX1BadSzd92QKqCpqiwjU\nmAC0nToJNlulbcYLZ7m2hY0Rv40jO7QFh1MyObPvICnnsjhv1pFriOB8RiG2EymcV20U6g3kGiLs\n7yPnIlrVhhreAjU2lrBQHW1z0wjWKRAejjUmluAgDTFZF9ArYA0LxxIdg06jEH4pFS0qalgYtphY\ntFoF48VUtNhQwsIwR7dEURRCz58FmxWbYzuAkNSzKKoVmyEMW8tYUECfehatzYrVGIbaMhZFgaDU\ns2isVmxGA9aWrQDQnTuDYrG32Vq2dmuzYDMascW28mgjPAyrEk9ObjHKmTMoZjM2oxFry1YoioIu\n9axHG1BhW1DqWRSLGZvBgDXW+br2NtW9LfUcVNRmNoFbmzb1HIrZZN+uVZm20FCsrdrUvO18Koqp\nBI3BgFbfldwCE5w7h2IqqXA7NSQEa+u2VWu7cB6lpLhmbcEhWNtUse3ieZRi/9uCDCEQfbn939GF\n81BUBMHB2Nq199lmcxynvg0dOpTdu3czadKk+u5Kg2Gx+Vt0uEyGjdSwEULUAb8DNhEREaSmphIX\nF+fRfujQIaKjowPescZEMmyEEKLxiBncH4CChx6j8LH5nm1zH6Xw8b8AEJ0wAMVmo+ihR+Gl5+1t\nQ69CsVopSHqYwicW2NuGDUSxWCh88CEK5j9lbxt+NTFmM7dcfStv/eYudFqF6N8M4rclJRSP78+m\nP15PgaWQ5Q+/h978DwrnPEjBU4vt+44YglJUROHsByhYaL9REn3tUJTCAgpn3U/B00sBiLpuGJqC\nfAoT76Ng0TP2tuuHo8nPo3DmbAoWP1falpdL4b2zKFhifx9RN1yDJiebwhmJFCx9wd524zVosrMp\nuude1GdfIuHyNoy9dTCarCxyp97DLw8u5eAvl7htylQM+Tl8cfVYVl4zA5uqsuwfjxJZlMOWfr9l\n1chELgJPJc8kqjCHT664ib/fMAuAt5LvJqowm0/7juKVG2cD8MbqacQUZPFp3xv5+41zHG1/IqYg\nk88vv4GXR90HwOtr7qFlfgaf9xnJyzfdD8C6NfcQm5/Bf/uMZKWj7bVXZ9Aq7xJb+1zPipseAGDt\n2hm0zr3EF72v4/9++6Cj7V5a56axrde1/HX0nwFY89pM2uZc5MteI1g+2l5vY/VribTLucCXPUew\nfIyjbd0s2mWfZ0eP3/DSzQ8BkLxuNu2zUz3aVr0+hw5Z5/iqx3BevPlhAP7+xn10zDzLzsuG8vzY\nRwF45Y376JR5lp3dh/L8OHvb3958gLiM0+zqnsBz4x4D4OU3H6Bzxml2dxvCs+MfB2DlWw/SOf0U\n33QdzDMTnnC0/ZnO6SfZ03UQSyfMA+D/3k6i66UUz7b1c+madoJ9XQay+Hf2/+//un4u3dJO8G38\nQBbdYm9bvuEhul/8lW/jr2L2LU8CsGzDI1x28Rf2dx7A07+3/1t46R+P0OPCL/wv7koW3mr/t/Di\nPx6l54Wf+S6uP0/duhCAF955jF7nj3Egrh8Lbn0agOfffZzeqUf5vtMVPDlxkaPtCXqnHuGHjn35\ny232fx/PvfcEfc55tj373nwuP3eIHztczvxJ9n8zz/xzPn3PHuJg+z48Mdn+b2bpP//CFWcPcrB9\nb56YbP83s2Tjk/Q78xOH2vfi8cnPArD4Xwvof/pHDrfrxWO329sW/esprjz9A0fa9uDrtzZx44D2\nhN+fiH7Hl5ivuprsT74AIPzB2ei//ALzVQPJ/sS+YEf4n+eQ8+4HNAR9+vRh6dKlbNu2jfj4eIKC\ngjyenzt3bj31rP5UueiwWw2b8lOiJMNGCBFYfgds7rjjDhYsWMDDD9svOI4dO8ZXX33Fyy+/7Hel\n98OHD7NgwQKOHz9OXFwcTz/9NP379/fYRlVVVq5cycaNGykoKODyyy9nwYIFDbqwsdSwEUII4UvZ\nwpTh+jAKLIVetm549DotV/VsTXwrIyF6eyHioZe3oc8jI8jKK8H4ug6KoFdcNLeP7E5+kZngtfbt\nWkeFcmX3lhSbrK7pAoaQIFpHG7Baba4vQ0FaDcF6rasAqBANVW5++YK9jcXevXu54oorKCgo4ODB\ngx7POYvrNjfOc4636UyuosMVTYkqV3RYMmyEEIGlqGXX8/Nhw4YNrF27lvPnzwPQsmVLpk+fzt13\n313pviUlJdx4440kJiZy2223sWnTJpYtW8bWrVsxGo2u7TZu3Mjrr7/O66+/TmxsLC+//DLbt2/n\n3//+t199vHQpz9+340GrVYiODiMzMx+rtWqBl31HLpK86RChwTpeSbqmWq/fkNVkbJo6GRvfZHy8\nq8nYxMaG11KvGpfqnu+rOiVKExNFVNdOZGbmox7/1b6dH1Oijp3O4m/bzpBvaMFrj12P5mQKW1I+\n58vcQ7Tq0JMTOSdpdTGXGX3upH2HXvU+JcrVFt4CtWVLjzZNZAuiLou3j8GJlPLbnT4FVmvN2sLC\nUWNjXW2qxYLFYMQaE4vq3M5in+pkbdkSVQXt6VOoVis2gxFLy1h729nTYLZgNRqxxMSiqiq6s2dQ\nLRbXdgBB586guO3r3mYNLd9GeBjGLp3IySlEc/asfeqUwYglxv4+7FOdSl9DraANFYLOn0UxW7AZ\nDFicU6e8tpmxhZa2BV04V3GbyYy1TJvGbMYWEorZMXWqZm2paMwmMBowdO1Mbm4RmnPn0JhNFW5n\nCw7B7Jgm5XfbxfNoTCU1a9MHY3ZMk/K7Le0CmpJiv9t0xhB6XnMlWVkFqOdSa31KlJzv7apzvvf3\nc3bWsh2UmK3MueVyrurRqtzzz63/Hz+fzeHmhDhuHdGVx1d/Q1pWEX+48TIijHr+/mFp4Ou6Ae25\na1SPKve1rsn1mW8yPt7J2HhX07Hxdr73O8MmNTWVO+64gz/84Q8UFhZitVoJDw/HarXy008/0bdv\nX5/779mzB41Gw5QpUwCYOHEib775Jjt27GDMmDGu7SZOnMjNN9+MwWAgPz+fvLw8oqKi/O1mvSib\nKimEEKLhssV3qVKb4nbXtSr75psvkRea41o1xNY5nvSiaPLVELoE2W9UpLVuQX7HNqhRMaX7do4v\nf7yatMV1rnGbxxhUtF2nuFpp0zr+ANDVPs4eEzh6dCu3L70qyMiNquALVExP/9pa2ttcF2J6Ddbo\nCo4XW8G+FbW1qkFbaz/b2gS6zf5+Sy9GdVhjKxiDNjVpq+BCtQG2abWKK+OioiCMv211aefOnQwZ\nMgSdTsfOnTu9bqcoCsOGDavDnjUMFmslRYfLZNh4LOstq0QJIWqZ3wGbkSNHsmvXLqKjozEYDK72\nU6dOceedd/LDDz/43D8lJYWuXbt6tMXHx3PixAmPNkVRMBgMfPDBB8ybN4+wsDDWrVvnbzdRFIXq\nnCud05qqs8JFkM5xMW5Tm2R1+JqMTVMnY+ObjI93MjZNn2upWLfPBbPNDECoLgQFBRUVk9VcJEnO\nwwAAIABJREFUL/0TQjQP06dPZ9euXcTExDB9+nSv2ymKwpEjR+qwZ/VPVVVXDUqvRYfLLC7iPiWq\nbN0bb8cQQojq8hmweeedd3jllVcA+wlt3Lhx5ea3FhQU+FVfprCwkNDQUI+2kJAQiouLK9x+7Nix\njB07lrfffpvp06fz+eefExkZWenrxMQYazQHNzLSWPlGZURE5ANgVVWio5vuMonVGZvmQsbGNxkf\n7xrC2GRlZbFmzRoOHjyI2Vw+ePDuu+/WQ68aP9ddW7egnDM4o9fqCdIGYbKaMNkkYCOEqD1Hjx6t\n8LEoLTgM3rNjnF8ryq0SpSm/SpTUsBFCBJrPgM3EiRMJDQ3FZrMxb948Zs6cSXh4aZqoMxtmyJAh\nlb5QaGhoueBMcXGxR7aOO71eD8A999zDhg0b2LdvH6NGjar0dTIyCqqdYRMZaSQ7u6DKU5sKC0oA\ne4ZNRkZekyvaVpOxaepkbHyT8fGuJmMT6MDw448/zk8//cT48eMJC2u6Qee6ZjJbAdAHaUvbrPZi\npUEaHXqNPWBjlgwbIUQdU1W1wgC98/q7ubC4FTmvfJUo++/OgI2iUH5Zb8maFUIEmM+ATVBQEL/7\n3e8A6NChAwMGDECn83sWlYcuXbqwfv16j7aUlBTGjh3r0bZy5UqsVitJSfalM1VVxWQyeQSKfFFV\nFau1Wl0E7EGXmhRQMlts5SrGNxU1HZumTMbGNxkf7xrC2OzZs4e33nqLfv361Ws/mpq8QvuXofDQ\n0qorzmwavVZPkCbIo00IIWrb3r17WbRoESdPnsRmKw1WqKraLKdEWd1umHgra+BtSpRGUcpl1DTF\n0ghCiPrlM/qyfPlyZs2aRWhoKDt37vRZqGzu3Lk+XyghIQGTycTbb7/N7bffzqZNm0hPT2f48OEe\n2/Xr149HHnmE0aNH06VLF1avXk1YWBgDBgyowtuqW+7RdZtNRbIhhRCNSUxMDMHBwfXdjSYnt9Ce\nTRNuLL1j7axho9cEodcGebQJIURtW7BgAd26deOxxx4jJCSkvrtT79wzbIK8FR12/HRNiXIVHVbK\n1azxlqUjhBDV5TNgc+DAAcxmM6GhoRw4cMDrdv5MAdLr9bz66qssXLiQ5cuXExcXx6pVqzAYDCxY\nsACARYsWMWLECObOncucOXPIy8vjyiuvZO3atQ36y4R70VCrTfVcwUIIIRq4++67j8WLFzN//nzi\n4uIICvI8izW3FPlAcWbYtDC4Zdi41bDROzJsZEqUEKKupKWlkZycTHx8+RXlmiP3gE3lGTb2352B\nG0VDuax6CdgIIQLNZ8Dm7bffrvBxdfXs2bPC4pWLFi3y+P3222/n9ttvr/Hr1RVNmQwbIYRoTJYv\nX052dja33nprhc83txT5QMktcGTYGEoDXu41bIK0MiVKCFG3brzxRnbs2CEBGwf3Kcnegi2ly3o7\niw7b2zVKBUWHpYaNECLAfAZsfE2BcqcoCsOGDQtIhxojbZkMGyGEaEyWL19e311okvIcU6JaVDQl\nyq2GjWTYCCHqyty5cxk/fjybN2+mY8eOaMpkiCxbtqyeelY/zH4VHbb/dN6Uda0SpSjlsnKkho0Q\nItB8BmymT5/u10GaY5EydxpFMmyEEI3XoEGD6rsLTVKus+iwoYKiw1LDRghRD+bPn4+iKHTo0EFq\n2OCZYeMt2FKaYWP/XbWprvayS4HLlCghRKD5DNgcOXKkyS1RXRskw0YI0djcfvvtrFmzhhYtWjB5\n8mSf5/qKprIK32yqWpph45gSpaqqK5smyH2VKMmwEULUkf3797N+/Xr69u1b311pEDyW9fayymvZ\nVaJKp0SVD/JIwEYIEWg+AzZDhw7l6quvZtiwYQwdOpSOHTvWVb8aFalhI4RobIYPH+4qLvyb3/ym\nnnvT9BQUmV13Y51Toiw2Cyr2Rr1GVzolSjJshBB1JC4uDpPJVN/daDA8AjZeM2zsP1XXst7OosPl\na9jopIaNECLAfAZsPvvsM/bs2cOuXbtYu3YtNpuNYcOGkZCQQEJCApGRkXXVzwbNI8NGlYCNEKLh\nu++++1yPx44dS+fOneuvM02QczoUlE6Jci8urNfq0Wv15dqFEKI2zZo1i8cff5y77rqLTp06odN5\nfhUYPnx4PfWsflhslRcddpY+cG7qvqx32VWipIaNECLQfAZsWrRowahRoxg1ahQAZ8+eZffu3Xz6\n6acsWrSItm3bMnToUNef5sr9ZC0ZNkKIxmb06NH07t2bcePGMWbMGFq1alXlY2zZsoWXX36ZCxcu\n0K5dO5KSkujduzc333yzx3Ymk4kOHTrw2WefoaoqV111letuJcBVV13F2rVra/ye6lteQekdbOcq\nUe6ZNEGaIII09o9gKToshKgrSUlJADzzzDPlnmuONSmtjgwbjaJ4ZMy7c8+wUVVnniRoNOWzcrQy\nJUoIEWA+AzZldejQgUmTJjFp0iRUVeXQoUPs3r2bNWvWNOuAjUZq2AghGrGtW7fyySefsHnzZl58\n8UUGDBjAuHHjuOmmm4iIiKh0/5SUFObNm8e6desYMGAAu3fv5t577+Wrr77iwIEDru0uXbrELbfc\nwvz58wE4deoUAN99912Tq5eW66hfE6zXEhykBUqX9AZHho1GlvUWQtSto0ePBuQ4P/74I7Nnz3at\nKPvTTz8xadIkj0LGM2fOJDExEVVVWb58ORs3bsRqtTJhwgSeeOIJtFptQPpSExZH0WFv06HALcPG\npuKeSK9UsEqUTIkSQgRalQI2mZmZGAwGQkJCOHz4MDt27ODyyy/n3nvvra3+NQpaqWEjhGjE2rdv\nz/Tp05k+fTpnzpzhs88+4/3332fJkiUMGzaMVatW+dw/Pj6eXbt2YTQasVgspKenYzQa0ev1Hts9\n9dRTjB49mmuuuQaAw4cP06NHj2oHaxRFwUuNyIByBuW93X2tSH6RPQjTwqB3XdBbsbieDwkKIjjI\nWdvGjFarcC7/AjklufSOuSxQXQ+Y6oxBUyNjIGMAjXMMJkyYwNChQxk2bBiDBg0qd26uClVVef/9\n93nuuec8Ai5HjhzhmmuuYfXq1eX22bBhA9u3b+ejjz5CURRmzpzJunXrmDFjRrX7ESjOGja+MmNK\niw6XFh4GR1ZOmc8vKToshAg0vwM2W7duZe7cuSQnJ9O+fXvuvPNO2rVrx9q1a5k7dy533XVXbfaz\nQZMMGyFEUxEVFUVsbCytW7fm559/Ji0tza/9jEYjZ86c4aabbsJms7Fw4ULCwsJcz3/zzTd89913\nvPjii662I0eOkJ+fz4QJE0hLS+Pqq69m/vz5tG7d2q/XjIkx1mlmTmSk0e9tzY6PguiIEKKj7eOQ\nbiv9yG3dMoqIXHu7VbESERnKIzuSyTMV8NJNf6FTZPvAdTyAqjIGTZWMgYwBNK4xeOGFF9i9ezdv\nvPEGc+fOpWfPnq6alH379q3SeTQ5OZlPPvmExMREXn31VVf74cOH6dmzZ4X7bNq0ialTp7qm286c\nOZMVK1Y0qICNrwyb0mW9VY8pvBrFsbS3VnFl6kgNGyFEoPkdsFmxYgX3338/Q4cOZdmyZbRr146P\nP/6Ybdu2sXTp0mYdsJEMGyFEY5afn8/WrVv59NNP2b17N+3atWPs2LEkJSURHx/v93Hatm3LDz/8\nwP79+5k9ezZxcXEkJCQAsGbNGv70pz9hNJZ+ydHr9fTv358HH3yQ4OBgli5dyv33388///lPv14v\nI6OgzjJsIiONZGcX+H2OT0svAMCg15KZmQ9AelaO6/mCHBOWEvsXhSJTCSnnU8kz2fc5dO44YbbK\np6LVpeqMQVMjYyBjAHU/Bs6Ab0306NGDHj16MG3aNEwmEwcOHGD37t0sWrSIs2fPMmjQIBISEhg6\ndChxcXE+j3XrrbeSmJjIvn37PNqPHDmCXq/n+uuvx2azMXr0aJKSktDr9Zw4cYJu3bq5to2Pjycl\nJQVVVf0OFlUno9KfbCjnX6FOq/EabHFe56sqHv3V6ez7aDUaLFYrAPogbaMI2jTGTLG6JOPjnYyN\nd7U1Nn4HbE6ePMnYsWMB+PLLLxk5ciRg/xBIT08PaKcaG8mwEUI0ZgkJCURFRTF69GjmzJlD3759\nq3Uc52ojCQkJjBo1ii+++IKEhATOnz/Pt99+y7Jlyzy2v//++z1+f+yxxxgyZAhpaWl+FT5WVRXH\nNXKdsNlUrFb/zvE5jqLD4YYg1z7FFnubVtGCqkGHY1lvq5n0wmzXvumFWX6/Tl2ryhg0VTIGMgbQ\neMdAr9czePBgBg8eTFJSEjk5Oa4VYdetW8d///tfn/t7OzdHRUUxePBgJk+eTEZGBg8++CArV67k\n4YcfpqioyKO2TWhoKDabDZPJRHBwsF/9rklGpa9sqOAQ+3k4WK/1GhwLDbVvowvSEhFpKD1uhIHo\n6DB0Og0lZvuHUUy0MSBBtrrSmDLF6oOMj3cyNt4Femz8Dti0bt2aw4cPk5WVxfHjx3n66acB2L59\nOx06dAhopxobybARQjRma9asYciQIdW+GN6xYwevv/46b7zxhqvNbDYTHh4O2IP8gwYNIjo6utzr\nDhs2jD59+gD2FaQAvy/gGzJn0WHnClEAJsdqUEGOYsNBWkfAxmYmq6Q0YJNZnFVX3RRCNEM2mw1V\nVdFqtRQWFlJUVMSdd97JokWLqn3M5ORk12ODwcDMmTNZvnw5Dz/8MCEhIZSUlLieLyoqQqfTVelc\nX52MSn+yoXJziwBQwJUNWZbJZK8/VlJiJsNtm7y8IjIz83G/mV6QX0ymruFnHki2nG8yPt7J2HhX\n07HxFuz1O2Dzpz/9iQcffBCA/v37c9VVV/G3v/2N5ORkXnjhhSp3qCnxyLBR5X9cIUTD995773n8\nfvr0aa/bTp482eexevfuzcGDB/nwww8ZP348X3/9NTt27HBNbfrhhx/o379/uf1OnDjB119/zcqV\nK9HpdCxdupSRI0f6tTJVQ+dc1ruFe8DGsRqU3hGocV8lKqvYPWBT+lgIIQJp//79PPDAAyxbtoyu\nXbsyceJETCYTRUVFvPDCC4wZM6bKx8zJySE5OZk5c+a4apeVlJS4AjJdu3YlJSWFfv36AfaVBbt0\n6VKl16hJRqWvbCiTxVF0WKPxnjGllh7HYindRlXBalU9Cw072hqLxpopVldkfLyTsfEu0GPjd8Bm\nypQp9O/fn9TUVIYPHw7A8OHDueGGG7wWGWsuNIqCgv18LpFGIURjUNFKHhVRFKXSgE1sbCzJyck8\n88wzLFq0iM6dO/PKK6/QtWtXAM6dO1dhwOYvf/kLS5cuZfTo0ZjNZq699loWL15c9TfTAOUW2oMz\n4cYgV5vZsay3M1ATorNPEbCpNs4XXHRtJxk2Qoja8vzzzzNmzBj69+/P66+/TnBwMF9++SWbN2/m\nb3/7W7UCNuHh4fz3v/9FVVUeeughUlNTSU5OZtKkSQCMHz+e1157jSFDhqDT6Vi9ejUTJkwI9Fur\nFoul8qLDHqtEuV3nO1eIcs+0l1WihBCBVqVlvXv37k1xcTEff/wxN910E0ajsdLiZM2FRqNgtalS\nw0YI0Shs27YtoMcbOHAgH3zwQYXPrV+/vsL2sLAwnn322YD2oyHILzJTVGJPoY8KK035L82wsWfd\ntDbEup47nHnM9TizOLtKxTiFEMJfx44dY8WKFYSGhrJt2zZuuOEGV12bhQsXVuuYGo2G5ORklixZ\nwpAhQwgJCWHy5MlMnToVsN/0TU9PZ+LEiZjNZsaNG8e0adMC+K6qz7m6k69Ai/NUrKqq57LemooC\nNnLeFkIElt8Bm8zMTGbNmsWhQ4ew2WwMGjSIZcuW8euvv7Ju3To6duxYm/1s8LSOgI1k2AghGqOM\njAw2btzIyZMneeSRR9i7dy/du3ene/fu9d21RudsWmmNgw6tSucjm8vUsAnXhxEWZCTfXECeqXQf\ns81MvrmAcH3jKVwphGgcIiMjOXfuHFarlUOHDpGUlATAjz/+6Fexd6fBgwezd+9e1+/dunXzqGPm\nTqvVkpSU5HqthsRi8yPDxhGxsdlU3CsfOAM5Wrdgj1YybIQQAeb3WWXp0qXExMSwd+9e15zU559/\nnk6dOrF06dJa62Bj4UqXlICNEKKROXz4MDfddBPbt29n8+bNFBYWsmvXLiZOnMg333xT391rdM44\nAjbRLYIxhpROiSpbwwagrbF1hceQaVFCiNpw2223MXv2bCZPnkxcXBwJCQm8/fbbPPbYY66MmObE\nWWfCV6DFme3obUqUzi3DRitLHQshAszvDJvdu3fz5ptvYjSWLlMVERHB448/zh133FErnWtMnCdo\nmRIlhGhsnn32WaZOncr999/PlVdeCdiD9FFRUbz00ku8//779dzDxsUZsOkY65khY3LUsAnyCNi0\n4ZfsE+WOkVmcTVyL5p25KoQIvPvvv59evXpx9uxZxo4di0ajoVOnTqxcuZJrr722vrtX5yxWe4ZN\nkI+AjaayKVFaCdgIIWqP3xk2VqsVmyNt0F1eXh5arTagnWqMJMNGCNFYHTp0iPHjx5drnzx5Mr/+\n+ms99KhxcwVsWnsGbMzODBtN6cpR7cI8M2x0iv3zVDJshBC1YdKkSXTq1Im7776bli1bAjBixIhm\nGayB0ho2Wr+KDquobgEb15Qox3rjOq0itceEEAHnd8Dmhhtu4MUXXyQzM9N1Mjp+/DiLFy9m5MiR\ntdbBxkIjGTZCiEYqIiKC1NTUcu2HDh0iOjq6HnrUeFltNs6lFwDQsVW4x3OmMjVswJ5h466TI6sm\nS5b2FkLUgrNnz6LTVWnNkSbNmWHju+iw25Qot8v8sqtESf0aIURt8PuMPW/ePObPn8+wYcNQVZVx\n48ZRUlLCddddx7x582qzj42C1i36LoQQjckdd9zBggULePjhhwH7KiJfffUVL7/8coNZyaOxuJBR\n6PoC0LFVmSlRldSwMQYZaGOI5UTOScmwEULUittvv505c+YwefJkOnToQEhIiMfzw4cPr6ee1Q9X\nwMbHVCbXlKgyi4u4athoy9eyEUKIQPE7YBMWFsaKFSs4c+YMv/76KxaLha5duxIfH1+b/Ws0nCdt\nybARQjQ29957L0ajkeeee46ioiLuu+8+WrZsSWJiYrMsQlkTzulQ+iANrSJDPZ5z1rBxD9gYgwxE\n6MPJMeURHRxJdEgUIFOihBC14+9//zsAzz33XLnnFEXhyJEjdd2leuVP0WGNW4ZNhVOiHPtKho0Q\nojb4DNikpKRU2B4XF1dum+YeuNFKDRshRCNis9nQaEovLv/whz/whz/8gcLCQqxWK+Hh9uk8ly5d\nIjY2tr662eg4AzYdYsNcU2WdnFOi3GvYgH1aVI4pj8iQ0oDNpaJMVFWVeghCiIA6evRofXehQSmd\nEuX9XOs8D9uLDpe2u4oOazwzbYQQIpB8BmxGjx6Noige0WR3zueaY0S+LKlhI4RoTB588EH++te/\nlqtlYDAYXI+3bNnC008/zd69e+u6e43WsTP22jPxbVqUe85ZdNi9hg3A1W2u5ETOSa6M7Utroz04\nVmwtJteUT0RweLnjCCFETZhMJj777DNOnjzJXXfdxbFjx+jataurCHFzYnFct/uqYeOMvdvUiqdE\nuQI2GsmwEUIEns+AzRdffFFX/Wj0JMNGCNGY7Nu3jzlz5vDyyy+j13tmfOTk5LBw4UI++eQThg4d\nWk89bHwKis2knM8FoHfnqHLPV1TDBmBI24EMajMAjaKhyFLkak8rTJOAjRAioM6cOcPUqVOxWq2k\np6fzu9/9jg0bNrB3715ef/11evfuXd9drFNWf4oOa9ymRFF+SpTONSVKMmyEEIHnMxTcvn37cn8K\nCgr44YcfOHDgADk5Oa725q40w6b80udCCNHQvPXWWxw+fJiZM2dSVFQaJNi+fTs333wzO3fuZPHi\nxaxbt64ee9m4HD2Vhara77r2jCsfsDFXUMPGSaPYP45DdaGE6+3Fii8WXqrF3gohmqOlS5cyfPhw\nvvzyS1ewfvny5Vx77bU8++yz9dy7uudc1tvXdCaN+5Qot8v8slOitJJhI4SoBX6fWXJzc0lMTGT8\n+PE8/fTTLFy4kN///vdMmTKFvLw8v45x+PBhJk6cSP/+/ZkwYQLff/99hdv985//ZNSoUQwYMIBb\nb72V/fv3+9vNeiMZNkKIxqRHjx784x//4MyZM9xzzz1cvHiRefPmkZiYSN++fdm8eTO33XZbfXez\nUTmUkglAl/YtCA0un8DqrYZNWa0N9mlRErARQgTa//73P+6++26PGmY6nY5Zs2Zx8ODBeuxZ/TA7\nMmx8Fx22/7TZVI/VYF1TorRSw0YIUXv8DtgsXbqUtLQ0Pv74Y/bu3cv+/fv5z3/+Q3FxMS+88EKl\n+5eUlJCYmMjvf/97vv32W+666y5mzZpFQUGBx3Z79uxh+fLlrFixgv3793PnnXeSmJhIVlbDXjFD\natgIIRqbjh07smHDBnJzc7n++uvZvn07L730EqtWraJ169aVH0B4OHTSHrDp0zm6wuddNWwqyLBx\n5wzYpEnARggRYHq9ntzc3HLtZ8+exWg01kOP6pe1ikWHfa0S5WtalRBCVJffZ5Zt27bx1FNP0bVr\nV1db9+7dWbBgAZ9//nml++/ZsweNRsOUKVMICgpi4sSJtGzZkh07dnhsd+HCBe655x569eqFRqPh\nlltuQavVcvz48Sq8rbqndS35JwEbIUTj0bp1azZs2ECfPn2IjY2VmjXVdDGrkEvZxUDFARtVVUtr\n2Gh8B2xaSYaNEKKWjB8/nsWLF7uyaXJyctixYwcLFixg7Nix9dy7uueaEuVjOpPGrYaNz6LDkmEj\nhKgFPosOe2yo0xEcHFyuPSQkBLPZXOn+KSkpHsEesC8FfuLECY+23/3udx6//+9//6OgoKDcvt4o\nikJ1ppA6T8Zll2H1lzO6rqpNr+hYTcemKZOx8U3Gx7v6HpudO3d6/H7PPfewePFi7rrrLh599FG0\nWq3rueHDh9d19xqd/x2zB1eMITri25UvFGxVrdhU+53cimrYuHNm2GQUZ3GxIA2dJoiY0PI1cYQQ\noqoeeughli9fzpQpUzCZTEycOBGdTscdd9zB3Llz67t7dc6/Zb3tP21elvWODg8BIMrxUwghAsnv\ngM3QoUN54YUXWL58OZGRkQBkZmby4osv+nVHtrCwkNDQUI+2kJAQiouLve5z/PhxHnjgAR544AGi\noytOMS8rJsboSl2sjsjI6qWDBjvqFQTpdURHh1X79Ruy6o5NcyBj45uMj3f1NTbTp0+vsD09PZ2Z\nM2e6flcUhSNHjtRVtxqtfYcvAjCwZ6sKC08669cABPlZw8am2li09yW0ipb7+k/nsij/blwIIYQ3\nOp2ORx99lAcffJDTp09jtVrp1KkTBoOBjIwMYmJi6ruLdcrq17LezilRVDgl6voB7WkZEUKPTpG1\n11EhRLPld8Dm8ccfZ+rUqYwYMYIOHToA9vmuXbt25Zlnnql0/9DQ0HLBmeLiYgwGQ4Xb79y5k6Sk\nJKZNm8a9997rbzfJyCiodoZNZKSR7OyCahUOtjki9IWFJjIz86vegQaspmPTlMnY+Cbj411NxiYQ\nQeGjR4/W+BjC7nxGAafT7Of9Qb0qrv3jrF8DlWfYxIR43qCwqlbW/vQ2Dw+c45ouJYQQ1dGrVy92\n7dpFdHQ03bt3d7WfOXOG8ePHc+DAgXrsXd2z+LGstzNg463osD5Iy8CerWqxl0KI5szvgE1sbCwf\nffQRX331FSdOnCA4OJguXbowdOhQvzJaunTpwvr16z3aUlJSKpwv+/7777N06VIWLVpU5fm0qqpi\ntVZpFw82m4rVWvUvls6TtsVqq9b+jUF1x6Y5kLHxTcbHOxmbxm+vI7smIkxPj44V32EttJQunR6i\nLT+92J1WoyUsyEi+2V6U36gzUGApZOMvHzGn3z0B6rUQorl4//332bhxI2C/Tp4xYwZBQZ6B40uX\nLtGqVfMLOlhcq0RVPiXKfVlvBWqU0S+EEP7yO2AD9jTK6667zqOegbN+jV7vO8U7ISEBk8nE22+/\nze23386mTZtIT08vVxvhm2++4emnn2bdunUMHDiwKt2rVxpZ1lsIIZodm03lm0MXALi6ZyuvNYmy\nirMBUFCICG5R6XHHdbmJL8/uYlL3CWSVZPP2kX/yc+Zxii0lhOh8B3yEEMLd6NGjOX/+PADff/89\ngwYNKrcilNFoZNSoUfXRvXrlKjrsK8PGreiwc0qUBGuEEHXF74DNvn37ePrppzl58iQ2Z3gZ+4nL\nnxoHer2eV199lYULF7J8+XLi4uJYtWoVBoOBBQsWALBo0SJeffVVzGYzM2bM8Nh/xYoVXHPNNVV5\nb3VKlvUWQojm538/X3KtDvWbK9p53S67JAeAcH0YOk3lH73D2w9hePshAOSbC1BQsKhWjmUdp19s\nnwD0XAjRXBgMBu677z4A2rdvz80331zpjdbmwr+iw6XLejunRFWn/IIQQlSH3wGbJ598km7duvHY\nY48RElK9Kug9e/bk3XffLde+aNEi1+N169ZV69j1TSsZNkKIZmzLli28/PLLXLhwgXbt2pGUlMQN\nN9zATz/9xKRJkzw+N2bOnEliYiKqqrJ8+XI2btyI1WplwoQJPPHEEx4rVDVkqqqyZc8pAC7vEk3H\nVt5rCzkzbCKDI6r8OmFBRuIjOnEi5xSHMo5IwEYIUSXvvfcet9xyC3q9HpPJxL///W+v206ePLkO\ne1a/bDYVZ0karc8aNo7t3aZEaSTDRghRR/wO2KSlpZGcnEx8fHxt9qfRcp64JcNGCNHcpKSkMG/e\nPNatW8eAAQPYvXs39957L1999RVHjhzhmmuuYfXq1eX227BhA9u3b+ejjz5CURRmzpzJunXrymVY\nNlSHT2Zx6kIeAKMHx/nc1plhExVSvVVE+sT0dARsjrkyW4UQwh+rV69m1KhR6PX6Cs91XHvaAAAg\nAElEQVTFToqi+B2w+fHHH5k9ezY7d+4EICcnh3nz5rFnzx7Cw8OZM2cOt912G0CDDc47s2sAgnwE\nbBRX0WGZEiWEqHt+B2xuvPFGduzYIQEbL7QyJUoI0UzFx8eza9cujEYjFouF9PR0jEYjer2ew4cP\n07Nnzwr327RpE1OnTnUVupw5cyYrVqxocAGbnPwSsousRIaWfrkoKrHw1mf2lbbi27agZyXLuWY5\nAjbVybAB6BPTi/+c+IzskhxO550lrkXHah1HCNH8rF27luzsbLKzs3nttddqdCxVVXn//fd57rnn\nPAIuTz75JAaDgd27d3Ps2DFmzJhB9+7d6d+/f4MNzlvcCv77KjqskSlRQoh65HfAZu7cuYwfP57N\nmzfTsWNHNGXOVMuWLQt45xoTKToshGjOjEYjZ86c4aabbsJms7Fw4ULCwsI4cuQIer2e66+/HpvN\nxujRo0lKSkKv13PixAm6devmOkZ8fDwpKSl+Z5AoilInF80vvvM9Z9Ly+eNvezDyqg7YVJV3vviF\nS9nFaDUK08b0RKfz3RFnhk10aITPLwbexEW0IzokisziLN44/A6PXn0fYXpj5TsGiPMzzltR5eZA\nxkDGABrnGIwZMwZFUXyeW/2tSZmcnMwnn3xCYmIir776KgAFBQVs3bqVzz77jODgYK644grGjh3L\nhx9+SP/+/RtscN7iVpNT5+PDRHGfEuUM2EiGjRCijvgdsJk/fz6KotChQ4dq17BpyiTDRgjR3LVt\n25YffviB/fv3M3v2bOLi4oiKimLw4MFMnjyZjIwMHnzwQVauXMnDDz9MUVGRx+dJaGgoNpsNk8lE\ncHDlKyHFxBjrJC09vn0EZ9LyWf/ZMWyKwtGTWew/Yl/K+w+/7cmVvdtWeoxskz1g0zGmDdHR3mvd\n+HLfkKks/epl0grTWbJ3OZe37snEPmNoF966WserjsjIugsSNVQyBjIG0LjG4IsvvgjYsW699VYS\nExPZt2+fq+3UqVPodDo6dizN/IuPj+fzzz8HqHFwHqoXoK8suOac3gSg12u8BtOdAXlVLZ0KpdEo\n1Qq+NxSNMfBYl2R8vJOx8a62xsbvgM3+/ftZv349ffv2DWgHmorSJf8kYCOEaJ50OvtHSkJCAqNG\njeKLL74gOTnZ9bzBYGDmzJksX76chx9+mJCQEEpKSlzPFxUVodPp/ArWAGRkFNRJhs2dN3bn9IU8\nTp7PZf0nR13tw/q24bp+bcnMzPe5f5GlmCKzfSWpIEtwpdt70y6oPX/sPYk3Dr5LdnEuO0/t48fz\nh5k7cBZtjK2qdUx/aTQKkZFGsrMLmm0mqYyBjAHU/RhUN8Drrn379gHoiZ0zS8ZdYWFhuZu5ISEh\nFBfbz3s1Dc5DzQL03oJrJrX0eC2jw7yOdcSlQtfj0FD76lpajSYgfzf1rTEFHuuDjI93MjbeBXps\n/A7YxMXFYTKZAvriTYlk2AghmqsdO3bw+uuv88Ybb7jazGYzqqry/PPPM2fOHMLC7Be2JSUlrgv0\nrl27kpKSQr9+/QB78eIuXbr4/bqqqmK1Bu59eBMcpOWp6UNY+e53nL2Uj1ajYfTgTgy/oi2qClar\n7/N+RmG263FEUESl2/sysNWVtBvUlkMZR/n05DZyTfks35/Mn6+cSetaDtqAfdpvTfrfFMgYyBiA\njIG70NBQj+A7QHFxMQaDAaDGwXmoXoC+suBaekaB63F+fhGZXmog5+cXux7n5BY5HqnVDr43BBJ8\n9U3GxzsZG+9qOjbegsB+B2xmzZrF448/zl133UWnTp1cd1Kdhg8fXuVONSVSw0YI0Vz17t2bgwcP\n8uGHHzJ+/Hi+/vprduzYwXvvvedavvuhhx4iNTWV5ORkJk2aBMD48eN57bXXGDJkCDqdjtWrVzNh\nwoR6fjcVaxkZyp8n9avwC1p6UQYnc04zoHU/NEr5bxTOJb0VFCKCW9S4L+3C2tAurA3dIuP52/dr\nyTXlseLAau7rP4N2YW1qfHwhhKiKuLg4zGYzqamptGvXDrAH4J3ToGoanIeaBei9BddM5tIaNhoU\n7wE4t2azxb6PovjYvhGRwKNvMj7eydh4F+ix8Ttgk5SUBMAzzzxT7jl/ipQ1dZJhI4RormJjY0lO\nTuaZZ55h0aJFdO7cmVdeeYVu3bqRnJzMkiVLGDJkCCEhIUyePJmpU6cCMGXKFNLT05k4cSJms5lx\n48Yxbdq0en43VWOxWVh5YA0ZxVnkmPIY2emacts4Cw6H68PQafz+2K1UfEQcc/rfw9++X0uOKY9n\n9v2VPjE96N/qCvrG9KrTosRCiOYrLCyMkSNHsmzZMpYsWcIvv/zC5s2bWbNmDdBwg/Puy3prfS7r\nXfrY+SVMyncIIeqK31eOR48erXyjZsxZLV4ybIQQzdHAgQP54IMPyrV369bNY6qUO61WS1JSkuuG\nQGP0zflvySjOAuCL019xTYehBJUJyjgzbKq7pLcvXSI6M7vfPbx2cD25pjwOZhzlYMZRtIqWvi17\nk9B2IL1jelSY+SOEEIGyePFinnrqKUaMGIHBYOCRRx5xZdQ01OC8+x1wna9lvd2iM84bs3VR8F4I\nIaAKARvhm2TYCCFE82K2Wfj05DbX7zmmXPZfOEBCu6s9tnNm2ETVQsAGoFtkPIuGPsH3aT+x/+IB\njmUdx2yz8P2ln/j+0k+EB4XROaIjncI70Cm8A3EtOhKub/zFMoUQ9Wfw4MHs3bvX9XtkZCQrVqyo\ncNuGGpw3u2XY6Hxm2JQGZ5xZObJCjhCirkjAJkCkho0QQjQv3144QHZJDgoKnVt0IiX3FFtP72BI\n24EeF/hZjoBNZEhkrfUlSKPj6jZXcnWbKym2lPBd2o98c/5bTuScJM+cz0/pR/gpvXTqcoS+BW2N\nrWkb1pq2BvvPVoZYjDqD3DkWQjQLVvcpUT4CMBpFMmyEEPVHAjYBIhk2QgjRvPyYfgiAvi17M7LT\nNfz1u1VcKEwjuySHKLfgTFYtZ9iUFaILZmi7qxna7mouFKRxNOsXTuee5XTeWS4UpKGikmPKJceU\ny9GsXzz2DdWFEBsaQ0vHH+fj1mEtiVQNddJ/IYSoCxbHlCitRvEZgPGoYWNzZNhIvEYIUUckYBMg\nrgwbVQI2QgjR1FlsFn7OOg5An5gedApvj4KCisqFgjSPgE12cd0GbNy1Mbaijdty38WWEs7mp5Ka\nf57zBRddf/LN9uVtiyzFnM47x+m8c+WOpVU0tNCH0yK4BZH6FrQIbkGEvgURweEYggwYdCGE6uw/\nDUGhBGuDpXaOEKLBck5v8jUdCjwzbJxBHpkSJYSoKxKwCRCNZNgIIUSzkZJzihKrCYBe0T3Qa/VE\nh0SRUZzJ+cKL9Iq5DLAHQIqtxUDtTonyV4gumG6R8XSLjPdozzPlc6konUuFGaQXZXCpKJP0Ivvj\nPHM+AFbVRlZJDlklOZzy47UUFIK1eoI0QQRpgwjSBKF3/tQEEaTVodMEoVU0aBQNGhw/NZrSNkWD\nVtGiQXH8rkVRFBTH8e3/2T9/nXfIFWeLopQ+57atq0VxPVPmWM4t8Li1rtFAWE4I+QXF2EpnUgRM\nXX79U6r5ahoNGHNCKKilMaipupimotfpGB5+Va2/jqh9FpszYOP7/5uKVomq7r8hIYSoKgnYBIjW\nVcOmAV7BCCGECKjDmT8D0NrQipjQKADaGlvZAzb5F13bOQsOQ/1k2PgrXB9GuD6MLhGdyz1XbCkm\n05SJWVfCuYw0sopy7FOqSvLIMeWSW5JHoaUIs83ssZ+KSrG1hGJrCZjLHVaIRutYzi9MuWxifXdD\n1JAz+OJrSW8ou0qUs+hw7fVLCCHcScAmQKTosBBCNB9HHAGb3tGXudraGttwMOMoFwpLAzbOJb0B\nIoJb1F0HAyhEF0LH4PZER4fRJTTfYylcd2armUJLMUWWIgotRRSaCymxmjDbzPY/VjMmm8XtsRmL\nzYJNtbn+WFUbquNnaZvV8Vi1/8QGqoqKPSgEoDqmI6vOFtX1jL1F9fac577ux4Ky71NBo1Ecn/PV\n+6yvrSsEtbaOXH4I0CiKffp3NV+y1vpaR3QaHVe06VXf3RABYDJbAdDrfEdflAqKDmuk6LAQoo5I\nwCZAtIpMiRJCiOagwFzIGUeNF+fUJ8BVK+Z8QRqqqqIoiivDJlwfhk7TtD9yg7RBRGiDiAgOr++u\n1AqtViE6OozMTO9Bq6ZOxsBzDETjVmK2Z8sE67U+t9NUNCVKAjZCiDoiCX0BIhk2QgjRPJxxK8jb\nJSLO9bitsTUARZYick15QGmGTVRw/devEUIIUarYZAEgOKiSgI1MiRJC1CM53QSILOsthBDNw9n8\nVABahkQTqgt1tbc2lK7GdL7APi0qu46X9BZCCOEfkzPDppKAjVLRKlGSYSOEqCMSsAkQWdZbCCGa\nh7N59oBNh/B2Hu0humCiQ+wFiJ0BmyxHwKYhrBD1/+3deXhU1f0/8PfsW0IWEjCRECYhBkiFBBJM\nCgiCglIgrYRKQdvHjVXwhxW1qBhBik8tIoKoUNGytFRav6BCKyIaKIjKXkBAyYjBEDFkn2T2+/tj\nMkOGZJJJMpn1/XoeHoabe+eeHPBz4nvOOZeIiK4xNu5h0+YMm6ZLomxcEkVEvsXAxks4w4aIKDw4\nZtj0irix2deu7WPjGthwhg0RUWBxBjZt7mHTZEmUtXFJFPMaIvIRBjZewj1siIhCn8lqxo/1PwEA\nekUmNPt6UmOIo6u+CACoatzDJpqBDRFRQDGaHDNsPH9KlMXxlCgmNkTkIwxsvIQzbIiIQt9lfRls\ngv0T1l4Ric2+3jdaCwAo1ZehvKECBqsRABDDJVFERAHFMcNG3p4lUY0zbLgkioh8hYGNlziSdkHg\nPjZERKHKsX+NRqZucdZMSlQyxCL70Hr4x+PO45xhQ0QUWDzdw0bk8pQobjpMRL7FwMZLJE0KN5dF\nERGFJsf+NUkRN7b4CatSqnQui/rqx2PO49GKbr5pIBERecQR2Cjbs4eNM7DpunYRETXFwMZLmq5l\nZWBDRBSaHJsJJ0bc4PacvjH2ZVFljefGKKIhFUu7vnFEROQxY+Njvdu3JIpPiSIi3/JpYHPmzBkU\nFBQgMzMT+fn5OH78eKvnv/POO5g/f76PWtc5EvG1ruQ+NkREoamycRNhx+O7W5IWneLy5/HaO7q0\nTURE1H5GkwWAB0uiXGbYND4lilNsiMhHfBbYGI1GzJo1C3fffTe++uor3HfffZg9ezb0en2zc+vr\n6/GnP/0JL774oq+a12kuM2y4hw0RUcixCTbnY7pjW9lEODVKCxHsY0J2z0zkJWT7pH1EROQ5xwyb\ntgKbpkuiLFYuiSIi3/JZYHPo0CGIxWJMmzYNMpkMBQUFiIuLQ1FRUbNzH3nkEVy8eBH33HOPr5rX\naZIWNiQjIqLQUWuqg1Ww73kQo3Af2KhlKtyT/ksMT7wFU9Pv5tR5IqIAZPJw0+Emk+idM2xY14nI\nV3y2qF6n0yE1NdXlmFarRXFxcbNzly9fjp49e2L16tWorKxs131EIpFLYfWUY4ZMR6c4SqXXrhMB\nkEhCp5B3tm9CGfumdewf90Ktb3bt2oXVq1ejrKwMiYmJWLBgAW6//XaUlZVhyZIlOHLkCKRSKe68\n8048+eSTkMvlEAQBQ4YMgdBkVuKQIUPwl7/8xY/fiXsVjcuhgLYf0z3ixryubg4REXWQxWpzfsCq\naGPTYVFLmw6HyNhNRIHPZ4FNfX09VCqVyzGlUgmDwdDs3J49e3b4Pt27azqVekdHazp0XZ3J5nwd\n2U2N2BhVK2cHp472TThg37SO/eNeKPSNTqfDokWLsGHDBgwePBgHDx7EjBkzsG/fPixcuBBpaWnY\nt28fampqMHfuXLz22mtYsGABLl68CAA4evRoUHxa6di/RiaWIkIW/H9vRETe9tZbb2HlypWQyWTO\nY+vXr0daWhoWLVqEQ4cOITIyEnPnzsWUKVP81k6Dyep83Z4lUVYuiSIiH/NZYKNSqZqFMwaDAWq1\n2qv3uXpV3+EZNtHRGlRV6Tv0lKe62mvfW0VlHSSCtZWzg0tn+yaUsW9ax/5xrzN9Exsb0UWt6hit\nVosDBw5Ao9HAYrGgvLwcGo0GcrkcKpUKs2fPhkKhQHx8PCZOnIiPP/4YgH0j+vT09KAIawCg0mDf\nvyZGER00bSYi8qUzZ85gwYIFePDBB12Oz58/H2q1GgcPHsS5c+fw8MMPIy0tDZmZmX5pp2M5FAAo\nZK3/j0PTcm+xckkUEfmWzwKblJQUbN682eWYTqfDhAkTvHofQRBg7URWYrMJzvS8Xfdt8tpstnXo\nPQJdR/smHLBvWsf+cS9U+kaj0aCkpATjxo2DzWZDYWEhIiIisG7dOpfzPv30U/Tr1w8A8PXXX6Ou\nrg75+fm4cuUKcnJy8PTTT3s8y7KjS2DbyzH1vdJon2ETo4oOqWWvngi1JXwdwT5gHwDsg7Z8/fXX\nmDx5sssxvV6PPXv24KOPPoJCocDAgQMxYcIEbN++3W+BjdHs+Qwbkci+jbyAJkuiGNgQkY/4LLDJ\ny8uDyWTCpk2bMHXqVOzYsQPl5eUYPny4r5rQpSQtrG8lIgonCQkJOHHiBA4fPow5c+YgOTkZeXn2\nvVwEQcCyZctQXFyMl156CQAgl8uRmZmJRx99FAqFAsuWLcO8efPw7rvvenS/zi6Bba86ax0AIKFb\nfMDNcvKVUFjC11nsA/YBwD5oSUNDA3Q6HTZu3IiFCxeiW7duePDBBzFgwABIpVIkJSU5z9Vqtdi9\ne7fH792RgL61cM1svbaVgVolbTOEF4lE9g+FG3/Gl0hEQR3cM3hsHfvHPfaNe13VNz4LbORyOdav\nX4/CwkK8/PLLSE5Oxuuvvw61Wo3FixcDAJYsWeKr5nidy2O9GdgQURiSSu1DSl5eHsaOHYtPPvkE\neXl5MBgMeOKJJ3Du3Dls2rQJ3bt3BwDMmzfP5fonn3wSubm5uHLlCnr06NHm/Tq6BLa9HMvXymp/\nAgCoRRpUVNR1/Y0DCJc3sg8A9gHg+z4IpnC4vLwcQ4YMwW9+8xu8+uqrOHnyJGbNmoX7778fSqXS\n5Vx3+1i605mAvqVw7YfKa/e+oWeUB0+KEsFmvRbYKJWyoPq7cYfBY+vYP+6xb9zzdt/4LLABgH79\n+mHr1q3NjrsLaq7/YT6Q8bHeRBSuioqK8Pbbb+Odd95xHjObzYiMjERVVRUeeughqNVq/OMf/0B0\n9LWnK61btw7Dhg1DRkYGAMBkMgEAFAqFR/ft7BLY9qpssO9hE62ICollbB0RKkv4OoN9wD4A2Act\nSUpKctn+IDs7G/n5+Th8+DCMRqPLue3dx7IjAX1r4Vr5VXvoLgJQV1MPfRthkOPLjvcxmyxBHdwz\nfG0d+8c99o17ne0bdyGwTwObUOYyw0bgP14iCh8DBgzAqVOnsH37dkyaNAn79+9HUVER3n33Xcyb\nNw9xcXFYvXq1y1NDAKC4uBj79+/Hq6++CqlUimXLlmHMmDGIiory03finsVqQY2pFoB902EiInJ1\n+vRpHDhwADNmzHAeMxqNSEhIgNlsRmlpKRITEwHY97Hs27evx+/dmYC+pXCtwWh/M7lcApsNcN2N\nsrnr96wRQRQSgR2Dx9axf9xj37jn7b7xwWTy8MAZNkQUruLj4/HGG29g48aNyM7OxqpVq/Daa6+h\nuroaX375JQ4ePIihQ4ciKysLWVlZmD59OgDgmWeeQa9evXDXXXdh1KhRkMlkWL58uZ+/m5ZVNFRB\naPyBPlbJwIaI6HpqtRpr1qzBf/7zH9hsNnz++efYuXMnpk+fjjFjxmDFihVoaGjAyZMn8eGHH2Li\nxIl+a6ux8bHebS2Fcrh+Ao6I/wdFRD7CGTZewj1sup4gCLAJ9sTSarO/FgT7cQEAmrx2HEeT14L9\nFPtx4dprQWj5eOttsf8ukYhQWW9BTU2Dx0mq0MqnOJ2ZnNXWta3dt40PljpMLBGhW60JNTUNsDGF\ndyGWiPAztWdLf4JBdnY23nvvvWbHz5075/aaiIiIgA1orldeX+F8Hc0ZNkREzWi1WrzyyitYuXIl\nnnrqKfTs2RPLly9HRkYGli5diueeew4jR46EWq3GwoULMWjQIL+11fGUKKWHgc31M2z4lCgi8hUG\nNl7CGTaAxWpDjd6EugYz6hrM0Bss0DeYoTeYYTBZYTRbYTLbYLI0/m62wmS2wmixv7ZYbfYpZI2/\nbDYBlsbfHceJQklsNwVemvNz2B8YSoGsonH/GpVUCaU0dII2IiJvGj16NEaPHt3seHR0NFatWuWH\nFrXMEdjIPQ1sxAxsiMg/GNh4SbjMsKmtN6G0XI/Scj1+rGxARY0BFbVGVNQYUK03dWqGCFG4iY9W\nQywWQbC1fS75V51JDwDQSD3fJJOIiAKTc0mU3LO1TVwSRUT+wsDGS8Qi+2fkAkJnho3RbMV3l2tQ\nfLkG31/R4+x3FajWmzy+XiGXIEIphUYlg1IuhUImgVwmhlwqgUImhlwmgUxq/10uFUMmFUMsFkHS\n+EssFkEqdj3mOC5pPC4S2QdREUTOwVQkajxu/wPE7o4DQJNrHY+LFIvsX/fksxOJRISYGA0qK/Ut\n/r239R4dfUSl63t4cI43zuhAU136h0uiXEgkIiTeEGXvm65ak0ZeozfVAwDUMpWfW0JERJ3lmGHj\n6R42XBJFRP7CwMaLxGKRcylPsLpSWY+TF67iZPFVnPu+CmZLyx/9R0XIkRCrRvduSsR0UyK2mwKx\nkUrERioQqZZBo5JBKgn9jx8kEhGiIhSwmswMJFogkYigVspgUEjZP9eRSEReCezIN5yBDWfYEBEF\nPVM7A5tmM2w4fhORjzCw8SJJY2ATbDNsqvUmfHHmR3x+qgwXf6xt9vUeMSoM0HZHrzg1knpEIDFO\nA41S1sI7ERGFpjqzPbBRcYYNEVHQMzgCG3lH97DxepOIiFrEwMaLHMXcFgQbuQiCgDMXK7HnqxL8\nr7jCpc0apRQZ2lgMTO2ODG13++yZ2AhUVNRxlgQRhaVrM2wY2BARBbt2P9YbXBJFRP7BwMaLHE+K\nstoCdwdRs8WGQ2fK8PFXJbj0k955XCGXIPumeOT97Aak946GRBz6y5mIiDzFwIaIKHS0d0nU9T8W\nizjFhoh8hIGNFzkCm0Dcw8ZiteHA/y7jg4PfoaLG6DyuTYjE7UOSMDg93uNBi4go3NRx02EiopBh\nNNs/XPV8DxsuiSIi/2Bg40Vi5wybwAlsbIKAQ6fLsOO/OvxUZQBg3zhtyE3xGJvTG6k3duPGaURE\nbeAMGyKi0GEwtXMPGz4lioj8hIGNFwXaDBvd5Rps3n0euss1AOwPbs7p3wP5w7VI6K7xb+OIiIKI\nY9NhtYxPiSIiCnZ8ShQRBQsGNl4kDpDAprbehH8VFWP/iVI4WpKVFodfjUhBrx4Rfm0bEVGwsdqs\nMFrsS0k5w4aIKPgZGwMbucyzPRubPSWKa6KIyEcY2HiRuHFHMn8tibLZBHx2/Af8375i6A0WAEBC\ndzWm3XETMvrE+qVNRETBTm9pcL5mYENEFPyM7d10+LoZNZxgQ0S+wsDGi/y5JOqbS1XYsvs8vr9S\nB8C+Jjd/mBa3Z/eCVMInPhERdVR943IogJsOExEFO0EQnI/1Vnq4h831AQ33sCEiX2Fg40WO4m0V\nfBfYVNUZse3TC/j8dJnzWF5GT0y5rS+iIxQ+awcRUaiqN3OGDRFRqDBbbM4tAzo6w4ZLoojIVxjY\neJEvZ9hYrDZ8cuQSdvxX59zpvld8BO4dexNuSoru8vsTEYWL+sYlUSKIoJQq/dwaIiLqDMdyKACQ\nd/Cx3pxgQ0S+wsDGi3z1WO+TF67iH3u/weWrjU8tUUjxq1tTMCorERIxlz8REXmTY4aNSqqEWMQa\nS0QUzJoGNh7PsLmu9HNJFBH5CgMbL+rqGTY/lOvxj73f4FRxBQD7Y7pHDErA3SNT0U0t75J7EhGF\nu3qL45HeXA5FRBTs9A0W52vP97BxDWg8DXqIiDqLgY0XddUMm4oaAz44+B32n7gMW+P+OH17ReE3\nY9KgTejm1XsREZErxwwb7l9DRBT8viurAQBolFJER3q23+P1M2qSb4j0eruIiFrCwMaLHDNsLFab\nV96vtt6EXYcu4pMjPzjfs3s3Jabcloqcfj2apf1EROR9zsCGM2yIiILehVJ7YKNN7Obx0qamewyr\nFVL0iOF4QES+wcDGi+Kjlfj6InDhh5pOvU/JlTrsPXoJn58ug8lsD2oiVDKMz03G6ME3erxBGhER\ndZ5j02G1VO3nlhARUWcVNwY2qYlRHl/T9ENSbUIk97AhIp9hYONFg/rGYd+Jy7jwQzWq9SZEaTzf\nV8ZgsuDo+Z+w78RlnC+pch5XyCUYl5OEcUN7Q6XgXxcRka/pzdzDhogoFNQbzCgt1wMAUhM931ag\n6QwbbTuuIyLqLD7uwosG9ImFXCqGAODkt+Vtnt9gtODkhXKs/+AM/t/q/+IvH37tDGviopT49W19\n8ec5P8cvR6QwrCGigLZr1y7cddddyMrKwi9+8Qvs2bMHAFBdXY25c+diyJAhGDVqFLZt2+a8RhAE\nrFixArm5ucjJycELL7wAq9Xq7hZ+45hho5Fxhg0RUUedOXMGBQUFyMzMRH5+Po4fP+7zNugu1zpf\ntyd4sViv7U+ZkuD5zBwios5iCuBFCpkEA/rE4vi35Tj2TTlGDEp0fk0QBNQ2mPHtpWqcL6nC+ZIq\nXPyxFkKT/YklYhF+po3FyKwbMTClu3MTYyKiQKbT6bBo0SJs2LABgwcPxsGDBzFjxgzs27cPhYWF\nUKvVOHjwIM6dO4eHH34YaWlpyMzMxJYtW/DZZ5/h/fffh0gkwsyZM7FhwwY8/PDD/v6WXHDTYSKi\nzjEajZg1axZmzZqFKVOmYMeOHZg9ezb27NkDjUbjs3ZcKK0GACR0V0OjlHl8XbBiTFwAABC9SURB\nVMmVOudrbQI3HCYi3/FpYHPmzBksXrwY3377LZKTk/H8888jMzOz2XkffvghVq5ciatXr+KWW27B\nsmXLEBcX58umdlhWWhyOf1uO099V4NV/nsTVGgPqGsyorTe73YxYmxCJvIwbMHRATz6em4iCjlar\nxYEDB6DRaGCxWFBeXg6NRgO5XI49e/bgo48+gkKhwMCBAzFhwgRs374dmZmZ2LFjB373u9+hR48e\nAICZM2di1apVgRfYWLjpMBFRZxw6dAhisRjTpk0DABQUFOCvf/0rioqKMH78+C6559ffVaD2XDn0\neqPzKavHvrHPgE9p51NW643XHgUeFeHZk6WIiLzBZ4GNp8n62bNn8dxzz2HDhg1IT0/H0qVL8Yc/\n/AHr16/3VVM7ZVDfOIgAmC02HG9hWZRYJELyDRG4KSkaNyVFI61XNCJUnif8RESBSKPRoKSkBOPG\njYPNZkNhYSG+//57SKVSJCUlOc/TarXYvXs3AKC4uBh9+/Z1+ZpOp4MgCB49BU8kEkHsg4W9JqsJ\nABAhV0MiCc+Zj44Zn+E885N9wD4A2AcdpdPpkJqa6nJMq9WiuLjYo+vbW+9LrtThxS3H3H49tVdU\nh+t5KIwD/HfcOvaPe+wb97qqb3wW2HiarH/wwQcYM2YMBg0aBAB4/PHHkZeXh/Ly8qCYZdNNI8ek\n4VocO/8TboyPQGKcGpFqOSJUMkSqZegVH8H9aIgoJCUkJODEiRM4fPgw5syZgwcffBBKpdLlHKVS\nCYPBAABoaGhw+bpKpYLNZoPJZIJC0fYnmN27azwKdjrrF+lj8PVP5/Hz1MFQyZRtXxDCoqN9t3Qh\nULEP2AcA+6C96uvroVK5zlJsOh60pb31XqVRYlBanHOD4aZ6xKhx57AURLZjVnvhw7n420dn8eCk\nnyE2NsLj6wId/x23jv3jHvvGPW/3jc+SA0+T9eLiYmRlZTn/HBMTg6ioKOh0Oo8Cm45+4urNROzu\nkSm4e2RKp98nUDBJdY990zr2j3uh2DdSqX1IycvLw9ixY3Hq1CkYjUaXcwwGA9Rq++a9SqXS5esN\nDQ2QSqUehTUAcPWq3iczbG6/cSQKMsajqkqPBltd2xeEILFYhOhoDaqq9LDZhLYvCEHsA/YB4Ps+\nCJVwQKVSNQtnmo4HbelIvX98aqbbvyuzwYQKg8nj99L20ODp+4YAACoqgn8c4H/LrWP/uMe+ca+z\nfeOu3vsssPE0Wb/+E1fAXuQbGho8uk9nP3FlWuge+8Y99k3r2D/uhULfFBUV4e2338Y777zjPGY2\nm9G7d2/s27cPpaWlSEy0b8Ku0+mcy6BSU1Oh0+mcMyp1Oh1SUjwPuwVBgC8fKmWzCbBaw/uHE/YB\n+wBgHwDsg/ZKSUnB5s2bXY7pdDpMmDDBo+s7U+/5d+Ue+6Z17B/32DfuebtvfBbYeJqsuwtxujKB\nB5gWtoZ94x77pnXsH/c60zeB9onrgAEDcOrUKWzfvh2TJk3C/v37UVRUhHfffReXL1/GihUr8MIL\nL+Cbb77Bhx9+iHXr1gEAJk2ahLfeegu5ubmQSqV48803kZ+f7+fvhoiIvC0vLw8mkwmbNm3C1KlT\nsWPHDpSXl2P48OH+bhoRUUDzWWDjabLu+MTVoaKiAtXV1c2WU7nT2U9cmRa6x75xj33TOvaPe6HQ\nN/Hx8XjjjTfwxz/+EUuWLEGfPn3w2muvITU1FUuXLsVzzz2HkSNHQq1WY+HChc4ZNdOmTUN5eTkK\nCgpgNpsxceJE3H///X7+boiIyNvkcjnWr1+PwsJCvPzyy0hOTsbrr7/u8QeyREThymeBjafJ+oQJ\nE3Dvvfdi8uTJuPnmm/Hyyy/j1ltvRUxMjK+aSkRE7ZSdnY333nuv2fHo6GisWrWqxWskEgkWLFiA\nBQsWdHXziIjIz/r164etW7f6uxlEREHFB9s12jmS9Z07d2Lo0KHYvHmzM1lfvHgxFi9eDADo378/\nli5diqeffhp5eXm4cuUKli9f7qtmEhERERERERH5nU+fL+0uWV+yZInLn8ePH+/yqG8iIiIiIiIi\nonDisxk2RERERERERETkGZEgCMG92yURERERERERUYjhDBsiIiIiIiIiogDDwIaIiIiIiIiIKMAw\nsCEiIiIiIiIiCjAMbIiIiIiIiIiIAgwDGyIiIiIiIiKiAMPAhoiIiIiIiIgowDCwISIiIiIiIiIK\nMAxsiIiIiIiIiIgCDAMbIiIiIiIiIqIAE/aBzZkzZ1BQUIDMzEzk5+fj+PHj/m5SQDl8+DCmTJmC\nIUOG4Pbbb8fWrVv93aSAU15ejry8PHz66af+bkrAKCsrw8yZMzF48GDceuut2Lhxo7+bFFCOHj2K\nu+++G4MHD8a4cePwwQcf+LtJ5AbHCI4DDuFe61nXWbuDCWu3e6zpbQv3eu8OxwH3unR8EMKYwWAQ\nRowYIWzZskUwmUzCtm3bhNzcXKGurs7fTQsIVVVVQk5OjvD+++8LVqtVOHXqlJCTkyMcOHDA300L\nKDNmzBD69esn7N27199NCQg2m0341a9+Jbz44ouCyWQSzp8/L+Tk5AhHjhzxd9MCgsViEXJzc4V/\n//vfgiAIwldffSUMGDBAKCkp8XPL6HocIzgONBXOtZ51nbU7mLB2u8ea7plwrvfucBxwr6vHh7Ce\nYXPo0CGIxWJMmzYNMpkMBQUFiIuLQ1FRkb+bFhBKS0sxcuRITJw4EWKxGBkZGbjllltw9OhRfzct\nYPz973+HSqVCQkKCv5sSME6cOIErV67g8ccfh0wmQ1paGrZu3QqtVuvvpgWEmpoaVFRUwGq1QhAE\niEQiyGQySCQSfzeNrsMxguOAQ7jXetZ11u5gwtrtHmt628K93rvDccC9rh4fwjqw0el0SE1NdTmm\n1WpRXFzspxYFlv79++Oll15y/rm6uhqHDx9Gv379/NiqwKHT6fD222+jsLDQ300JKKdPn0ZaWhpe\neuklDBs2DOPGjcOJEycQExPj76YFhJiYGEybNg2PPfYYMjIyMH36dDz77LP8wSAAcYzgOACw1gOs\n6wBrdzBh7XaPNb11rPfucRxwr6vHB6lX3iVI1dfXQ6VSuRxTKpUwGAx+alHgqq2txaxZs5CRkYHR\no0f7uzl+Z7FY8MQTT+Dpp59GdHS0v5sTUKqrq/HFF18gNzcXn376KU6dOoWHHnoISUlJyM7O9nfz\n/M5ms0GpVGLVqlUYPXo0Dh48iN///vfIyMjgD0wBhmOEq3AcB1jr7VjXWbuDCWu3Z8KxpreG9b51\nHAfc6+rxIaxn2KhUqmbF22AwQK1W+6lFgamkpARTp05FVFQU1qxZA7E4rP/ZAADWrl2L/v37Y+TI\nkf5uSsCRy+WIiorCzJkzIZfLnZtvffLJJ/5uWkDYvXs3Tp48iTvvvBNyuRyjRo3CqFGjsH37dn83\nja7DMeKacB0HWOvtWNdZu4MJa3fbwrWmt4b1vnUcB9zr6vEhrP/rTElJgU6nczmm0+nQt29fP7Uo\n8Jw+fRq//vWvMXz4cKxduxZKpdLfTQoIu3btws6dO5GdnY3s7GyUlpbisccew7p16/zdNL/TarWw\nWq2wWq3OY441nQRcvnwZJpPJ5ZhUKuU+CAGIY4RdOI8DrPV2rOus3cGEtbt14VzTW8N63zqOA+51\n+fjgla2Lg5TRaBSGDx8ubNy40WUXeb1e7++mBYSffvpJyM3NFd58801/NyXg3XbbbdxJvlFDQ4Mw\nYsQI4ZVXXhHMZrNw5MgRITMzUzh27Ji/mxYQzp49K2RkZAj//Oc/BZvNJnzxxRdCVlaWcPLkSX83\nja7DMYLjwPXCtdazrrN2BxPWbvdY0z0XrvXeHY4D7nX1+CAShPCOxc6ePYvCwkKcO3cOycnJKCws\nRGZmpr+bFRDeeOMNrFy5stkU0t/+9rdYsGCBn1oVmEaPHo1nn30Wt912m7+bEhAuXryIJUuW4H//\n+x8iIiIwd+5cTJ482d/NChh79+7FqlWrUFJSgsTERDz66KO44447/N0sakG4jxEcB1yFc61nXWft\nDibhXrvdYU33XDjXe3c4DrjXleND2Ac2RERERERERESBJqz3sCEiIiIiIiIiCkQMbIiIiIiIiIiI\nAgwDGyIiIiIiIiKiAMPAhoiIiIiIiIgowDCwISIiIiIiIiIKMAxsiIiIiIiIiIgCDAMbCmpPPfUU\n0tPT3f5avXo1vvjiC6Snp8NoNHZ5ey5duuS89/z58zt8/YULF9o89/HHH3fey5PziYiCFWs9az0R\nhQfWe9Z7ciUSBEHwdyOIOqq2thYGgwEAcOzYMcybNw979+6FXC4HAKjVashkMlRXVyM+Pr7L23Pp\n0iWMGTMGW7ZswU033YRu3bq163qr1YqKigrExsZCIpG0em5tbS10Oh2mTJmCXbt2ITU1tTNNJyIK\nWKz1rPVEFB5Y71nvyZXU3w0g6ozIyEhERkYCAKKiogAAcXFxUCgULuf5oqA3FRMT0+6CDgASicTj\ntkZGRiI2Nrbd9yAiCjas9az1RBQeWO9Z78kVl0RRyLt+2mR6ejp27dqFiRMnYuDAgXjggQdw+fJl\nLFy4EJmZmRg7diwOHTrkvP7HH3/E/PnzkZWVhREjRqCwsBB6vd6jezumQX722We44447MGjQICxY\nsAClpaWYMWMGBg0ahPz8fJw9e9blfMc0yNGjR2PTpk249957cfPNN2PcuHEoKirycg8REQU/1noi\novDAek/hhIENhaUVK1Zg8eLF2Lx5M06fPo1Jkyahf//++Ne//oW+ffvi+eefBwAIgoBHHnkEMpkM\n27Ztw5o1a3D27FksWrSoXfd79dVX8corr2Dt2rX4+OOPMXnyZIwfPx7btm2DUqnEn//851avnTZt\nGnbu3In09HQsWrQIZrO5U98/EVE4YK0nIgoPrPcUqrgkisLSfffdh5ycHABAbm4uSkpK8MADDwAA\n7rnnHsyaNQtWqxVffvkldDod/va3v0EmkwEAli9fjjvvvBNlZWW44YYbPLrf7NmzkZGRAQDo168f\nEhMT8ctf/hIAkJ+fj/Xr17u9duLEiRg/fjwAYM6cOcjPz0dZWRmSkpI69s0TEYUJ1noiovDAek+h\nioENhaXevXs7X6tUKpcCqVQqYbPZYLFYcOHCBdTV1WHo0KHN3kOn03lc1K+/X69evVzuZzKZ3F7b\np08f5+uIiAgAYApPROQB1noiovDAek+hioENhSWp1PWfvljc8upAi8WC3r17t5iSt2ezM0/v1xJH\n+t8UH+5GRNQ21noiovDAek+hinvYELUiNTUVZWVliIyMRHJyMpKTk2GxWPDiiy+irq7O380jIiIv\nYK0nIgoPrPcUbBjYELVi2LBhSE1NxWOPPYbTp0/j1KlTWLhwISorK9GjRw9/N4+IiLyAtZ6IKDyw\n3lOwYWBD1AqxWIy1a9ciIiIC9957Lx544AH06dMHa9as8XfTiIjIS1jriYjCA+s9BRuRwAVzRF5z\n6dIljBkzBrt27UJqamrI3Y+IiFjriYjCBes9+Rtn2BB1gcrKStTU1HTpPWpra1FRUdGl9yAiIvdY\n64mIwgPrPfkLAxuiLjB9+nQ888wzXXqP559/HlOmTOnSexARkXus9URE4YH1nvyFS6KIiIiIiIiI\niAIMZ9gQEREREREREQUYBjZERERERERERAGGgQ0RERERERERUYBhYENEREREREREFGAY2BARERER\nERERBZj/D73wfNvGHj6iAAAAAElFTkSuQmCC\n",
"text/plain": [
""
]
},
"metadata": {},
"output_type": "display_data"
}
],
"source": [
"from ipywidgets import interact\n",
"\n",
"IC = [C0,T0,Tcf]\n",
"\n",
"def sim(Tsetpoint,kp,ki,kd):\n",
" global Tsp, qc\n",
" Tsp = Tsetpoint\n",
" \n",
" # control parameters\n",
" beta = 0\n",
" gamma = 0\n",
"\n",
" # create python list to log results\n",
" log = []\n",
"\n",
" # start simulation\n",
" c,T,Tc = IC\n",
" qc = 150\n",
"\n",
" eP_ = beta*Tsp - T\n",
" eD_ = gamma*Tsp - T\n",
" eD__ = eD_\n",
"\n",
" for t in np.linspace(ti,tf,int((tf-ti)/dt)+1):\n",
" # PID control calculations\n",
" eP = beta*Tsp - T\n",
" eI = Tsp - T\n",
" eD = gamma*Tsp - T\n",
" qc -= kp*(eP - eP_) + ki*dt*eI + kd*(eD - 2*eD_ + eD__)/dt\n",
" qc = sat(qc)\n",
"\n",
" # log data and update state\n",
" log.append([t,c,T,Tc,qc])\n",
" c,T,Tc = odeint(deriv,[c,T,Tc],[t,t+dt])[-1] # start at t, find state at t + dt\n",
" \n",
" # save data for PID calculations\n",
" eD__ = eD_\n",
" eD_ = eD\n",
" eP_ = eP\n",
"\n",
" qplot(log)\n",
" \n",
"interact(sim,Tsetpoint = (360,420),kp = (0,80), ki=(0,160), kd=(0,10));"
]
},
{
"cell_type": "markdown",
"metadata": {},
"source": [
"## Simulation 5. Using the PIDsim Controller Class\n",
"\n",
"From this point on, this notebook describes the development of a PID control class that could be used in more complex simulations. Regard everything after this point as 'try at your own risk'!\n",
"\n",
"Examples of PID Codes in Python:\n",
"\n",
"* [DISCRETE PID CONTROLLER (PYTHON RECIPE)](http://code.activestate.com/recipes/577231-discrete-pid-controller/)\n",
"* [ivPID](https://github.com/ivmech/ivPID)\n",
"* [python-pid_controller](https://gist.github.com/chaosmail/8372717)\n",
"* [PID Controller](https://github.com/chrisspen/pid_controller)\n",
"* [pypid 0.4](https://pypi.python.org/pypi/pypid/)\n",
"* [PIDController](http://cgkit.sourceforge.net/doc2/pidcontroller.html)\n",
"* [PID.py](http://examples.oreilly.com/9780596809577/CH09/PID.py)\n",
"* [APMonitor.com](http://apmonitor.com/do/index.php/Main/NonlinearControl)\n",
"\n",
"Below we demonstrate the use of `PIDsim`, a python module that can be used to implement multiple PID controllers in a single simulation. "
]
},
{
"cell_type": "code",
"execution_count": 8,
"metadata": {},
"outputs": [
{
"data": {
"image/png": "iVBORw0KGgoAAAANSUhEUgAAAtgAAAHvCAYAAABuRKiNAAAABHNCSVQICAgIfAhkiAAAAAlwSFlz\nAAALEgAACxIB0t1+/AAAIABJREFUeJzs3Xl4VFW29/HvqarMCZkI80wYZR4EBERRQGVyalq9otKo\nqG3bgEO3zVVbRW3f2+gFlAYRnJrr0E6gIiiKqAgqMggoCiQgYyAJmSup8f2jkiKBEEKoOpWE3+d5\n+iGpOnXOyrL6ZGXX2nsbXq/Xi4iIiIiIBIQl1AGIiIiIiNQnKrBFRERERAJIBbaIiIiISACpwBYR\nERERCSAV2CIiIiIiAaQCW0REREQkgFRgi8g5aeLEiXTq1Mn/v86dO9OnTx+uu+46vvzyy1CHF3AP\nPPAA/fr1w+FwVPr8+vXr6dSpExs2bKjxNZ599lkuvPDCah//zTff0KlTJ/bu3XvKY+677z6uv/76\nGsckIhIKKrBF5Jx1wQUX8Oabb/Lmm2/y+uuvM3v2bBo0aMAdd9zB9u3bQx1eQI0fP578/Hy++eab\nSp//6KOPaNGiBX379q3xNa6//nrmz59f49eLiNQXKrBF5JyVkJBAr1696NWrF71792bo0KHMmTOH\n2NhY3nzzzVCHF1CDBg2iUaNGfPzxxyc953Q6+eSTTxg3bhyGYdT4Gk2aNKFr165nE6aISL2gAltE\npJzIyEjatGnDwYMHAfjrX//Kn//8Z/70pz/Rs2dPHn74YQD27t3L3XffzYABA+jXrx/Tpk0jIyOj\nwrnWrl3L73//e3r27Mnw4cN54YUXKjy/evVqrrzySrp3784ll1zCkiVLKjy/adMmrrvuOnr37s3A\ngQP5y1/+wrFjxyq8/qqrrqJHjx4MGTKEmTNnUlxcXOnPZbFYGDNmDJ999tlJbSJff/01OTk5jB8/\n3v/YqlWruO666+jVqxc9e/bkuuuuY9OmTf7nr7/+eh5//HFuvPFGevTowcKFC09qEcnJyeGRRx7h\nwgsv5LzzzmPo0KE8/fTTuFyuCtffsGEDY8aMoUePHtx4443s2LGj8v84gMPh4Omnn2bIkCH06NGD\niRMn8uuvv57yeBGRUFCBLSJSjsvl4sCBAzRv3tz/2KpVq4iNjWXevHlcffXVHDx4kAkTJnDkyBGe\neOIJHn30UbZv386NN95IQUEB4CuOb7vtNpo1a8bcuXO55ZZbmDNnDq+++ioAX375JXfddRddu3Zl\n3rx5XHXVVTzxxBP+Ijs/P5877riD5s2bM2/ePB566CHWrl3Lo48+CvgK/HvuuYfzzz+fhQsX8uc/\n/5l33nmHuXPnnvJnO1WbyIcffkivXr1o06YN4Ct47777bvr06cOCBQt46qmnyMvL47777sPj8fhf\n98Ybb9C3b1/mzp3L8OHDT7retGnT+Pbbb3nwwQd58cUXGT9+PIsXL2bp0qUVjps5cybXXHMNs2fP\nxul0cvPNN5OdnV3pz3D//ffz9ttvc9dddzF79mxsNhsTJ0486Y8bEZFQsoU6ABGRUPF6vf7RVI/H\nw+HDh5k/fz5ZWVlce+21FY595JFHiIyMBODJJ58EYPHixcTGxgLQo0cPLrvsMt5++21uueUWXnzx\nRTp16sSzzz4LwIUXXkhGRgabNm3ipptuYs6cOVxwwQX+cw0dOhSXy8XcuXOZMGECu3fvJicnhxtv\nvJHevXsDEBsbS1paGgDbtm3D4XAwefJkGjVqxIABAwgPDz9pdLi8zp0707FjRz7++GMuuugiAOx2\nO59//jn33Xef/7i0tDSuvPJKHnjgAf9jhmEwdepUMjIyaNq0KQCNGzdm2rRplV6rqKgIr9fLzJkz\n6devH+BrU1m9ejU//PAD11xzjf/Yu+++m0mTJgHQs2dPLr74Yt59911uvfXWCufcsWMHK1asYPbs\n2Vx22WUADBkyhJEjR/Lyyy/zl7/85ZQ/u4iImVRgi8g56+OPPz6pJzkpKYnHHnuM7t27+x9r0qSJ\nv7gG+OGHHxgyZIi/uAZo2bIl3bt3Z+PGjdxyyy1s2rSJCRMmVDj3/fffD/iKz23btvHggw9WKIiH\nDBnCv/71L3bu3ElqaioJCQnceeedjBkzhuHDhzN48GCGDRsG+Ar68PBwJkyY4H9+7NixWCxVfzA5\nbtw4FixYgMPhIDw8nNWrV+N0Orniiiv8x0yYMIEJEyZQUFDA7t27SU9P59NPPwV8/dplyka8KxMd\nHc3LL7+M1+vlt99+Iz09nR07dpCdnV3hHAAjRozwf52UlESPHj3YuHHjSef8/vvv/Xkqy5thGAwc\nOJBvv/22yp9bRMRMKrBF5Jw1ZMgQpk6dCvh6lBs0aECLFi1OmuiXnJxc4fu8vLyTHgNfcVjWIpKb\nm0tSUlKl183Ly8Pr9fLkk0/6R7DLO3r0KF27duW1117jueee4+233+a1114jOTmZe++9l2uuuYaW\nLVvy0ksvMX/+fF5++WUWLlxI8+bNeeSRR/xFeGXGjRvHM888wzfffMNFF13Ehx9+yLBhw0hMTPQf\nU1BQwIwZM/jkk0+wWq106NCBZs2aAb5R//I/b1VWrlzJU089xaFDh0hJSaFXr15ERkZWOEdl50lM\nTKzQa14mJycHoNKVTlJSUqqMRUTETCqwReSc1aBBgwoj1WfyuqysrJMez8rK8heisbGxJxWJhw4d\nYt++ff6VNqZPn84FF1xw0nlat24NQMeOHZkzZw4Oh4N169axaNEi/vu//5vBgwfTpEkT+vXrx4sv\nvkhhYSFff/018+fPZ9q0aaxfv57w8PBKY2/cuDEDBgxgxYoV9O3bl6+++opZs2ZVOOaxxx5jy5Yt\nvPbaa/Ts2ZOwsDA+//xzVq1aVe0c7d69m3vvvZeJEycyefJkGjZsCMDVV1990rH5+fkVPg3Izs6u\n9A+YuLg4oqKieO211056LiwsrNqxiYgEmyY5ioicoT59+vD111/7R6sB9u3bx7Zt2+jVqxcAvXr1\nYs2aNRVe9+qrrzJjxgxiY2Pp2LEjBw4coHv37v7/ZWdnM3fuXEpKSli3bh2DBg0iOzub8PBwhg0b\nxr333ovH4+HIkSO8//77XHLJJTidTmJiYhg1ahS33XYbhYWFFeKqzLhx41i9ejWrV68mKirK349d\nZvPmzVx66aX069fPX7iWTYwsP8mxKtu3b8fpdDJlyhR/cX3kyBF27tx50gj22rVr/V9nZGSwZcsW\nf992eX369MFutwNUyNt7773HypUrqxWXiIgZNIItInKGbrnlFt577z3+8Ic/cPvtt+NwOJg9ezaN\nGzf2T9677bbbuOmmm7j//vsZP348u3bt4t///jczZswAfBP7pk2bRkxMDBdeeCH79+/nn//8J+ed\ndx4pKSlERkZiGAb33HMPt912GwAvvPACLVq0oEuXLiQmJpKZmcn06dO57rrrsNvt/Otf/6JPnz6n\nbd0YOXIkjz76KM899xyXX375SaPd3bp1Y/ny5fTq1Yvk5GQ+++wz/+omZQXu6XTt2hWLxcI//vEP\nrrzySv8EUqfTSVFRUYVjZ8+ejcViISEhgblz55KSksJVV1110jl79OjB0KFD+dOf/sTdd99N8+bN\nWbFiBW+88YZ/MqmISG2gEWwRkTPUvHlzlixZQlxcHPfddx+PPvoo3bp14/XXX/e3OvTr14/58+ez\ne/du7rzzTpYsWcJf//pXrrvuOgBGjRrl74W+7bbbmDNnDqNHj2bOnDmArx1i4cKF2Gw27r33XqZO\nnUpMTAyLFi0iLCyMli1b8q9//YuMjAzuvvtuHnzwQTp06OB/fVViY2O59NJL2bt3b4W1r8s8+OCD\n9OzZk0ceeYSpU6eyc+dOXnnlFcLDw9m8eXO1cpSamsoTTzzBhg0buP3223n++ecZNWoUt99+O1u3\nbq0wEv7www+zYMECpk6dSnJyMq+88kqFlpHy5syZw6WXXsqzzz7LlClT2Lx5M7NmzaowSVNEJNQM\n74mf1YmIiIiISI1pBFtEREREJIBUYIuIiIiIBJAKbBERERGRAFKBLSIiIiISQCqwRUREREQCqNau\ng330aH5IrmsYBsnJMWRlFZ60GYIElnJtLuXbPMq1eZRrcynf5lGuzVPdXKekxFX7nBrBPoHF4ku0\nRZkJOuXaXMq3eZRr8yjX5lK+zaNcmycYudZ/NhERERGRAFKBLSIiIiISQCqwRUREREQCSAW2iIiI\niEgAqcA2kdPl5qsfD3LkWFGoQxERERGRIKm1y/TVR6t+2M9/Vu8GYPFfh4c4GhEREREJBo1gm2jd\ntgz/126PJ4SRiIiIiEiwqMA2UZPkaP/Xh7PtIYxERERERIJFBbaJkuIi/F/vP1IQwkhEREREJFhU\nYJvI4zm+/eb+oyqwRUREROojFdgmcrmP913v0wi2iIiISI0NGdKPSy4ZzIgRQxkx4kJGjhzG9Ol3\nk5a2C7vdzqhRw/jii89Oet2BA/sZNmwABw8eCFpsKrBN5CxXYGsEW0REROTsLFz4Cp9++hWffvol\ny5d/Rrt2qdx3358JDw9nxIjLWLHio5Ne89FHy+jb93yaNWsetLi0TJ+J3O7jLSLZeSUUFjuJiQwL\nYUQiIiIiVXO5PWTnFZtyraQGkdisNRv/tdlsXHHFWN5449/k5+czbtxV3H77LRw7dozExEQAPB4P\nK1Z8xNSp9wcy7JNjCerZpYLyI9jgm+jYqVViiKIRERERqZrL7eFvL6wnM9ecArthfCRP3j6wRkV2\nXl4eb7/9Bu3atSchIYGEhARSUzvy6acrmDDhegC+/XYdXq+XwYOHBjr0CqodfWZmJoMGDWL16tUV\nHvd4PEycOJGnn37a/5jX62XWrFkMHDiQ/v37M3PmTNxud+CirqNcrooFtvqwRURERGrujjsmc9ll\nF3HZZRfxX/91LVlZmcyc+f/8z48ff3WFNpGPPlrGmDHjsVqtQY2r2iPYM2bMICcn56THFy9ezIYN\nG+jWrZv/sSVLlvDFF1+wbNkyDMNgypQpLF68mNtuuy0wUddRrhNGsPcezg9RJCIiIiKnZ7NaePL2\ngbW2RWT+/EW0a5d6yucvvXQUc+c+S1raLpKTG7J+/Vr+/Od7AxFqlapVYL/++utERUXRtGnTCo/v\n2LGDd999lxEjRlR4fOnSpdx88800atQIgClTpjB79uxzvsB2lvZgGwZ4vbBldxZujwerRXNNRURE\npHayWS00Sow+/YG1UFRUFCNGjGLFio9ISWlM//4DSElpFPTrnrbATk9P56WXXuKtt97i6quv9j/u\ncDj4y1/+wuOPP85//vOfCq9JS0sjNfX4XxNt27YlPT0dr9eLYRjVCswwDEJRd1osRoV/A6lse/RB\n3ZrwzdbDFNid7Nyfy3ltkwJ+rbogmLmWkynf5lGuzaNcm0v5No9yXT0Wi4HVWnWOrrrqah588AGS\nkpKYPHnKSccHI9dVFtgul4sHHniAGTNmkJCQUOG5WbNmMWTIEPr27XtSgW2324mMjPR/HxUVhcfj\nweFwEBERQXUkJ8dUuxgPhoSEmICf04vv50ltmUjGMTu79+eyJS2boX1bBfxadUkwci2npnybR7k2\nj3JtLuXbPMp11eLjo0lKiq3ymEGD+pOcnEROTg5XXDHilPVlIHNdZYE9b948unTpwrBhwyo8vm7d\nOtavX39SYV0mMjKSkpIS//d2ux2bzVbt4hogK6swZCPYCQkx5OQUVth5MRCKS1wAOB0u+nRoyO79\nuXzz40F+f3G7c7JNJJi5lpMp3+ZRrs2jXJtL+TaPcn1669b9AEB29ukXjVi48BUAjh0rPOm56ub6\ndIV8eVUW2MuXL+fo0aMsX74cgIKCAqZPn47T6SQsLIwLLrgAgOLiYgzDIC0tjQULFtC+fXvS09Pp\n2bMn4GszadeuXbWDAt9KJKFceMTj8VZYtzoQylYRsRgGfTum8J/Vu8kvcrI9/RjntTk320QgOLmW\nU1O+zaNcm0e5NpfybR7l2jyBzHWVBfaKFSsqfD98+HAeeughLr744gqP//WvfyUxMZG//OUvAIwb\nN45FixYxcOBAbDYbCxYsYPz48QEJuC5zlfZg26wGjRKjad04jr0Z+azfdvicLrBFRERE6pOgbDRz\nww03kJmZybXXXovT6WTs2LFMmjQpGJeqU8pGsMuWnxncvQl7M/L5/pcj3DCiI1ER2vdHREREpK47\no4ru888/r/Txf/zjHxW+t1qtTJs2jWnTptU8snqobJm+MJuvwB54XhPeWr0Lh9PDdz9nMKxX81CG\nJyIiIiIBcO7NrAuhso1mykawY6PC6N0hBYCvfzwUsrhEREREJHBUYJvoeIF9fHmYoT19m/fsPpjH\nwcyTZ7aKiIiISN2iAtskbo8Hb+nE1PJbgHZtnUR8bDgAm3dlhiI0EREREQkgFdgmcbmOL/tSvsC2\nWAy6t00GYFtalulxiYiIiEhgqcA2ibO0PQSOT3Is062db4m+nftzsZduRiMiIiIidZPWhTOJq1yB\nbT1hr/uubZIwDHB7vPy89xh9OqaYHZ6IiIhInfPbb3uZN282mzdvwu1206xZc373u98zZsyVLF/+\nAf/4x+PldhI3iI6OZvjwEfzxj3/GZgteGawC2ySuKkawY6PCaN8snl0HctmWlqUCW0REROQ0PB4P\n9957D6NHj+XRR58kLCycLVs28be/3U9sbBwAHTp0YtGi1/yvOXr0CFOn3kVERAR33HF30GJTgW0S\nl7vyHuwy3dolsetALlvTsvF6vRiGcdIxIiIiImZzeVwcK8415VqJkfHYLNUrT3Nzczh06AAjRlxG\nREQkAL179+Wuu+7B6XRW+pqUlEZccMFQ0tJ2BSzmyqjANknZLo5QeYHdvV0y73+VTlZeMQczC2me\nEmtmeCIiIiIncXlcPLb+f8gqPmbK9ZIjE3l44P3VKrITE5Po3bsv06b9kVGjrqB377506XIeY8de\nCcDy5R9UON7j8bBnTxpr1nzOVVf9Lijxl1GBbZLykxzLr4NdpnWTOOJjw8ktcLDx16MqsEVERERO\n45//nMPSpe+wZs1q/v3vlwG48MKLmT79AQB27fqVyy67CACv10tiYhLDh49gwoTrgxqXCmyTuE/T\nImIxDHp3SOGLTQfY+GsmYwe3NTM8ERERkZPYLDYeHnh/rWwRAYiIiGDChBuYMOEGSkpK+PHHzfzr\nX3N56qnHuPDCi0lN7VihB9ssKrBNUtUyfWX6dGzIF5sOsDcjn8xcOw3jo8wKT0RERKRSNouNlOjk\nUIdxks8++4T585/nrbfexzAMIiIi6N9/AA6Hg2eeeZoLL7w4ZLFpHWyTVLVMX5nOrRKJivD9zbNp\np3Z1FBERETmVfv3Ox24vZPbsWRw75lskYv/+fbz99hsMHjw0pLGpwDZJ2SRHm9U45QohNquFnu19\nfyFu+vWoabGJiIiI1DXx8QnMm/cimZlHmTjx91x66RCmTr2LLl3O409/mh7S2NQiYhKXx9eDXVn/\ndXl9Oqaw/qcMftmXQ26hg/iYcDPCExEREalzWrVqw8yZT1f63BVXjOWKK8aaHJGPRrBNcnwEu+qU\nd2+fTESYFa8XNuw4YkZoIiIiIhJAKrBNUjbJ8VQTHMtEhFnp1aEhAN/9nBH0uEREREQksFRgm6Rs\nkmNla2Cf6PwujQDYuT+X7LzioMYlIiIiIoGlAtsk1W0RAejWNtm/msj3ahMRERERqVNUYJukupMc\nwddG0qej2kRERERE6iIV2CY5kxFsgAFdGgOQfiifIzn2oMUlIiIiIoGlAtsk/kmO1ejBBujcOpHY\nqDAAvtcotoiIiEidUe0COzMzk0GDBrF69WoADh8+zF133cWAAQMYPHgwjz/+OA6HAwCv18usWbMY\nOHAg/fv3Z+bMmbjd7uD8BHVE2SRHazVHsG1WC/06pQDw3c/qwxaR+sfr9fLp9/vYqI21RKSeqXaB\nPWPGDHJycvzf33///TRp0oQvv/yS999/n61bt/L8888DsGTJEr744guWLVvG8uXL2bhxI4sXLw58\n9HWIy+3rwT7dMn3lnV/aJrLvSAGHsgqDEpeISKikHcrj9c928ty7W7V7rYjUK9Wq9l5//XWioqJo\n2rQpAA6Hg6ioKO68804iIiJISUlh7NixbNq0CYClS5dy880306hRI1JSUpgyZQrvvfde8H6KOuD4\nMn3VL7A7tkzw7+SoUWwRqW+y80r8X7/40U9kZBeFMBoRkcA57Vbp6enpvPTSS7z11ltcffXVAISH\nh/PCCy9UOG716tV07twZgLS0NFJTU/3PtW3blvT0dLxeL4ZRvR5kwzCwhKBD3GIxKvwbKG7P8RFs\nazX7sK1Wg/O7NuLT7/fz3c8ZXHVh22rnry4IVq6lcsq3eZTr6rGXuMp97WbOOz/y3zf3A2BfRj4d\nWyVgPc0vAuXaXMq3eZRr8wQj11UW2C6XiwceeIAZM2aQkJBQ6TFer5cnnniCtLQ0/ud//gcAu91O\nZGSk/5ioqCg8Hg8Oh4OIiIhqBZacHBPSYjIhISag57OU/pKIiQ4nKSm22q8bObAtn36/n0NZReSV\neGjbLD6gcdUGgc61VE35No9yXTVvueLZajE4lFXE/7y+maxcO/lFTtq3iOeeCb1p1/z09z3l2lzK\nt3mUa/MEMtdVFtjz5s2jS5cuDBs2rNLni4uLeeCBB/jll1947bXXSE5OBiAyMpKSkuMf/dntdmw2\nW7WLa4CsrMKQjWAnJMSQk1OIp3TUORAK7b4JoG6Xm+zsgmq/LiUujOQGkWTlFfPJunR+d3Hq6V9U\nRwQr11I55ds8ynX1HM32zS3p3DqBoT2asfCDn9hzKM///O79uUz/3zU8OLEPHVpUPsijXJtL+TaP\ncm2e6ub6TAZIqyywly9fztGjR1m+fDkABQUFTJ8+nTvvvJMJEyZw6623Eh0dzZtvvllhhLt9+/ak\np6fTs2dPwNdm0q5du2oHBb6R8VAuPOLxeHG7A/eGdpatg22xnPF5+3dpxIpvf2P99gyuGtquXrWJ\nQOBzLVVTvs2jXFetoMg38BAdEcag85qQW+Dg3S/T6JWazPldGvPm5zvJyivhpY928Mik/lXOYVGu\nzaV8m0e5Nk8gc11lgb1ixYoK3w8fPpyHHnqIiy66iJtuuomGDRsyd+5cwsLCKhw3btw4Fi1axMCB\nA7HZbCxYsIDx48cHJOC6yj/J0XbmxfGALo1Z8e1vZOYWk34on3bNGgQ6PBER0xUW+3qwYyJ9v4ou\nG9CKkf1b+vsgE+MiePK1HziQWcjK735j9KA2oQpVROSMnHaSY2U2bdrEd999R0REBOeff77/8a5d\nu7JkyRJuuOEGMjMzufbaa3E6nYwdO5ZJkyYFLOi66Ex3ciyvVeNYGidGkXHMzrrth1Vgi0i9UGh3\nAhATdXyQpvwko/bN4xnWqxlfbD7I0q/30Cw5ht4dU0yPU0TkTJ1Rgf3555/7v/7ll19OeZzVamXa\ntGlMmzat5pHVM87SjxxqUmAbhsEF3Zrw3lfprN9+mAkXp57RetoiIrVR0Qkj2JW55qL2bE3LJiuv\nmOfe3crvh6cyon/LetcqJyL1i6o0k7j962DX7JfC4O5NMfB9pLp5V2YAIxMRCY3C4tIR7MiwUx4T\nExnG3yb2pXXjOLzAG5/v4t+f/Irb4zEpShGRM6cC2yTO0gI7rAYj2ABJDSLp2iYRgK9/PBSwuERE\nQqWgdAQ7uooRbPD1Yv/1v/rQK7UhAKs3HWDRhz/j9Wril4jUTiqwTXJ8kmPNUz64h28nzW3pWRzL\nLznN0SIitZfL7aHE4VsqqnwP9qlEhFu5++ruXNqvBQDrf8rg258yghqjiEhNqcA2iesserDL9OmQ\nQnSEDa8XvtpyMFChiYiYrqz/GiC2ihaR8iwWg+sv6UC3tkkA/PuTX8nOKw5KfCIiZ0MFtklcZ9mD\nDRAeZmVwd98o9potB9WDKCJ1Vln/NZy+RaQ8wzCYdEUXoiNsFJW4WPyRWkVEpPZRgW2S4wX22aX8\not7NADiWX8LmnVlnHZeISCgUlhvBrmqSY2US4yK4cWRHALamZbNi3Z4ARiYicvZUYJvE6fKNsNR0\nkmOZpskxdGntm+z4xab9Zx2XiEgolK2BbRgQGWE949cP6NqYfp18a2Iv+mA7h7OLAhqfiMjZUIFt\nAq/X6x/Btp5lgQ1wce/mAGzfc4xDWYVnfT4REbMdXwM7DEsN1rQ2DIOJozoRHxNOicPNc+9spcTp\nDnSYIiI1ogLbBG7P8f7AsLPowS7Tu2NDkhtEAPDp9/vO+nwiImYr8K+BXaMNhQGIiw5nyvjzsBiw\n70gBr67YoX5sEakVVGCboGz0Gs5umb4yVouFS/q2BGDttsPkFTnO+pwiImYq8q+BfWb91yc6r20S\nN17eBYB12zP4SvsEiEgtoALbBGVL9MHZT3Isc2HPZkSEW3G6PHyx6UBAzikiYpayHuyYqJqPYJe5\n5uIO/k1o/m/Vr2qdE5GQU4FtAqfr+Aj22U5yLBMdaePCHr4VRT77Yb96D0WkTqnONunVZbEY3Dq2\nC/Gx4TicHhYs217hk0MREbOpwDZB+Ru9NQA92GVG9G+B1WKQX+TkS208IyJ1SKF/kuPZj2CDrx/7\n1jFdAfgto4Bla/cE5LwiIjWhAtsE5QvsQI1gAzSMj2JQtyYArPj2twoj5SIitVkgR7DLnNcmyb+V\n+vJ1e0k/lBewc4uInAkV2Cao0IMdgEmO5Y0e2BrD8G08s3arJveISN1QFOAR7DLXDGtP48QoPF4v\nL374E06X2udExHwqsE1QYRWRAI5gAzROimZAl8YAfLhuj0axRaROOD7JMXAj2AARYVYmj+mKYcCh\nrCLe+yo9oOcXEakOFdgmKF/02gLYg11m7OA2GAZk55VoRRERqfW8Xq+/Bzs6wCPYAKnN47lsQCsA\nVn77Gzv35wT8GiIiVVGBbQJ3EEewwbd9+uDuTQHfKHaxwxXwa4iIBEqJ0+3fgCuQPdjlXTmkHc0b\nxuAFXvzwJ+wlui+KiHlUYJvAWdqDbQBWS+BHsAHGDW6DzepbUeST77S7o4jUXmX91xCcEWyAMJuF\nW8d0xWp0nP6XAAAgAElEQVQxOJpTzJJPfw3KdUREKqMC2wSO0jWqw8OsGEZwCuyG8VFc3Lt09vy3\nezmWXxKU64iInK2icqPJ0RHBKbABWjeJ45ph7QH4Ztth1m8/HLRriYiUpwLbBA5XWYEd3HSPHdyG\nmEgbDqeHd9fsDuq1RERqyowR7DIjz29J1zaJALz2yS8czbEH9XoiInAGBXZmZiaDBg1i9erVAOTm\n5vLHP/6Rvn37ctFFF/Gf//zHf6zX62XWrFkMHDiQ/v37M3PmTNzuc3eppBKnrwc73GYN6nVio8K4\ncmg7ANZuO6w1YEWkVirrh7YYBhFhwb0vWgyDyaO7EhsVhr3EzcIPfsLt0WpLIhJc1S6wZ8yYQU7O\n8ZnYDz30ENHR0XzzzTfMmTOHf/7zn2zevBmAJUuW8MUXX7Bs2TKWL1/Oxo0bWbx4ceCjryPKWkQi\nwoP7iwRgWK9mNE2OBuDVlb/g8XhP8woREXOVtYhERQSvba68xLgIJl3RGYBdB3L5QLs8ikiQVavA\nfv3114mKiqJpU99KFYWFhaxatYp77rmHiIgIevTowZgxY3j//fcBWLp0KTfffDONGjUiJSWFKVOm\n8N577wXvp6jl/D3YAd5kpjI2q4UbR3YCYO/hfFZr2T4RqWXs/gI7uO0h5fXukMLFvZsD8ME3e7R0\nn4gE1Wnvbunp6bz00ku89dZbXH311QDs3bsXm81Gy5Yt/ce1bduWTz75BIC0tDRSU1MrPJeeno7X\n6632aIVhGFhC0CFuKV3lwxLA1T6cpcv0RYRbsQZhHewTdWuXxAXdmvDNtsO8++Vu+nVOIalBZNCv\ne6aCkWs5NeXbPMp11YodvkGHmMiws74nnkmurx/RgV/25XAws5CFH/zEY7eeH7RlAusrvbfNo1yb\nJxi5rrLAdrlcPPDAA8yYMYOEhAT/40VFRURGVizYIiMjKS4uBsBut1d4PioqCo/Hg8PhICIiolqB\nJSfHmPLR4akkJMQE7FyG1dcaEhsdTlJSbMDOW5U7ru3Jlt1ZFNqdLFm1i4cnDwhpPqsSyFzL6Snf\n5lGuK+ctvRc1iI0I2D2xurn+6839mf6/X5KZW8wbn+3mvhv71tp7Y22m97Z5lGvzBDLXVRbY8+bN\no0uXLgwbNqzC41FRUZSUVFwGrri4mOhoX+9vZGRkheftdjs2m63axTVAVlZhyEawExJiyMkpDFj/\ncl6+7w8PAy/Z2QUBOWd13HBpBxZ+8BMbfs7ggzW7GNKjqWnXro5g5FpOTfk2j3JdtezSlTzCrJz1\nPfFMcx0faeX3w9uz5NOdfLn5AJ1axte6e2Ntpve2eZRr81Q312cyIFBlgb18+XKOHj3K8uXLASgo\nKGD69OnceuutOJ1ODh48SLNmzQBfK0lZW0j79u1JT0+nZ8+e/ufatWtX7aDAtxJJKBce8Xi8uN2B\neUOXlH4cGma1Buyc1TGwa2O++ymDLbuz+Pcnv9KxRQLJ8bWvVSSQuZbTU77No1xXrrDYCUBUuC1g\n+TmTXA/v04Ifd2ezNS2LV1f8QuvGcTRrqFHCM6H3tnmUa/MEMtdVjhGvWLGCH374gQ0bNrBhwwaa\nNWvGM888wx//+EcuueQSZs2ahd1u58cff+TDDz9k7NixAIwbN45FixZx+PBhMjMzWbBgAePHjw9I\nwHVRiYmriJRnGAY3XdaZmEgb9hIXCz/Yrr+CRSTkikIwybE8wzD4w+guxMeEU+J089y7W7WVuogE\nVI2bMB5//HFcLhfDhg3jnnvu4f777/ePWN9www0MHz6ca6+9ltGjR9OnTx8mTZoUsKDrGjNXETlR\nYlwEt1zeBYBf9+fywTd7TI9BRKQ8e+lGM8HeZKYq8THh3HllN6wWg8PZRSz+6Ge8Xg1AiEhgnNHd\n7fPPP/d/nZCQwOzZsys9zmq1Mm3aNKZNm3Z20dUTJa7SjWaCvKHCqfTtlMJFvZrxxeaDLPs6nTZN\n4uiZ2jAksYiIhHoEu0zHlglMuDiV1z/byQ+/HuXjb3/jioGtQxqTiNQP2irdBI7SHuyIIG+VXpXf\nX9KBVo1j8QIvfLCdQ1mFIYtFRM5tZQV2dIgLbIBL+7VgYNfGALyzZjfb92SHOCIRqQ9UYJsg1CPY\nABFhVv50dQ/ion3bBc99ZytFxeo5FBHzhWKjmVMxDIObL+tMi5QYvF6Y9942Dhw1b7UnEamfVGCb\nwL9VeggLbIDk+EjuKtdzuPCD7XjUcygiJnK5PTicvkGHUPZglxcRbuXua8oGIFw8+58tHMsvOf0L\nRUROQQW2CUI5yfFEnVolcv2lHQDYsjuLNz7bqYk9ImKa8qt11IYR7DKNEqL487U9CbdZyM4r4dm3\ntmhlERGpsdBXfOeAktLRmlCPYJe5uHdzhvXyrV++asN+ln6dHuKIRORcUVSuaK0NPdjltWvWgDvG\nd8MwYP/RAp5/bysutyfUYYlIHaQCO8g8Hq//Bh3KHuzyDMPgxpEd6dcpBYBla/ew4tvfQhyViJwL\nKoxg15IWkfJ6dWjIxJGdAPhpzzHmvbcNp0tFtoicGRXYQeZwHd+OMjyEq4icyGqxcPu48+jWLgmA\nt1bvYvWmAyGOSkTqu/KTq2vbCHaZi3o3Z9zgNgBs3pXJc+9uxekK4dbCIlLn1M67Wz1S1h4CtadF\npIzNauGPV3Xn2be28Ou+HF5b+QsOp5tR57cKdWh1hsfrxe324HT5PqlwuT24PV4MfJ8UGMYJ/4L/\n6zCrBZvNwGqpPX94iQRb2Qh2uM2CzVp73/tXDm2HxTB4/+t0tqZlMeftH7n7mh617j5eW3m9Xtye\nsvuiF6fLg9vjwcB3P4Ry98bSrzHAYhhYLQZhNgtWi+F7XKQOUoF9ArfHTY49l9ySQtwBGLDIKrJD\nmG82uoMicmvhxPRJ49qyYOk29mQU8OZX2zlScIyxF7QN+o3NagWL3R2wXJ8Nr8dLgd1BbpGT3MIS\n8god5BY4yC9yUFTixl7i8v3P4aa4xInD6ful4Q7ABFED/MW2zWrBZrFgtVkIs5Z+bzWwWi2+Y6wW\nrFaDMIuBtbRAsVl9RXqY1YLVZmAzDGw2i+81Ft/xNqtBeJiVhIQoiu0ODAxsFt81LRYDi1H6L75f\ndBbL8T8KfM+Xfm2E6BdeaZ792faClxMeq3Cot/zLKPfQ8dd5T3xNxbOd+J/WWz4G7wlHe73+r72A\n1WKQ4yokN6cIt6dm75G4yCiSYmJr9NpA8nq95DkCt2xdVlEuhJUQGR1Gbkn+WZ8vmPeRYf0b4rLY\n+XDdXrYfOMyst4uYdEUXoiPDAnuh2szrpdjpJrfAQW5hCQV2JyVuL0eyCimwOykucVNU4sTucGMv\ndlHidON2e3G6PSf9f/NMGfg+bQ2zgbX03mizWrDZLNgsvvua755Yev+zGv7nrKX3xkpfU3bvLL0/\nhpUOdNisltLCvuy+67sPWkrvfZbSPwgspYW/774ZvHtjtd7bldwbff+cfH888T7n9VbxutPcH08+\nprJ7dOUxVozpzN8lsRGRJMfGnfHrzGZ4a+kSEkePnv2N90wVu0qY+e0/OVaSa/q1RUTK83oMRqSM\n56qeF4Q0jrmbFrLj2M6QxiAiUsbrMbgoaQwT+gwN2DmtVoOkpFiyswtwu09dFqekVL+wr72fz4WA\nx+uh2F0Lh5hF5JxjWLxsy9wR0hg8Xo+KaxGpVQyLt07UamoRKSc6LIonh87AFV5Mbm4RngBMHE8/\nmMdLH/t+ST58cz9stWAt7Kp4vV4+23iALzcfBCAm0sY1F7UntXl8wK9lsUB8fHTAcl1U7GLv4Tz2\nHM5nz6E8DmfbT/kRZWykjaT4SJLjI0luEEHD+CgSYiOIiwknJsKGxVL/+v4MA2LjIsnOLsTh9Pj7\nI90er++jQq8Xj7fc1x4vHko/RvSUPocXj+f4sZVeB+PEB6r61h9bZUf5ezUrvNDgxK+OP2X4vz/p\nnOUeqHDek85R9o9RyTmOP1dZ7GUfEVssBnGxkRQUFOMp7cmv5Ic8pSU/foQ9ah8Oiqr9mmC7rtPV\ntItvfdbnWb5+D+u3HyG1eQNuuqzzWZ8v0PeRquzNyOeNVTspLHZhGDCiXwsu6N4USy3vE3a5POzP\nLGTPoTzSD+Wx72gBLlfl//8NsxkkN4gkOT6K5PgIkhtEkhgXSYPoMBpEhxEebjUt32Zyezy43V5f\n65/Hg8vlweP14im7/+FbFcyLF68H/3OUu1dSel+sMi+VvFVO9e6xWg2ioyMoKirB4281q969sfx3\n5e8//scq3PxOeF01748nn6fic5Wc2n+PrPzc1fv/UXxkNO1SmlTr2FBSgX2CKFskSQkNyfZU/TFB\ndWVZw/Da4zAMaBXfrE5M2Lh5WDO6NMrg5Y93UHDMzavvHWTMBeGMG9ImoBPyrFaDpITYGufa4/GS\ndiiPzTsz2ZqWxf4jBeUKahvg+yinUWIUbZrE0bpxHK2bxNGqcRyxUedQD2Wpso/AEo3AvLfl1Kr7\nceOpvLM5ATv7cGIPQnQ10zAqieaxTc/6PJaSY3jtdhJsKQE539neR85E89imdExuyZx3fmT/0UJW\nfpnLvt8sTB7dlcS4iKBe+0wdzi7y3xt3Hcg9YalBX29/bFSY797YJM5/j0yOj6zy95SZ+T7Xne19\nREJLBXaQlW0JHB5mrRPFdZnzuzSmdeM4/vX+Nn47UsAH3+zhx7QsbrmsM62bhG5yQbHDxfb0Y2ze\ndZQfd2eRX+Ss8LzFMGjdJI5OrRLo3CqB1Obx59aEJKkXrJ5IABy1qMAOlLJl+mrTLo5nomFCFH+b\n2Jd/f/Ir32w7zE97jvHfL67ndxelcmGvZiEbzXZ7POzan8vmXZls3pVFRvbJn340Soyic6sEOrVK\npEOLeJIbVF1Mi0jN1c07XB1SUrpNel1c2qlxUjQzburLG5/51sjeezifx175nmG9mnPlkLY0iAk3\nJY7svGK27Mpk065MduzNOWlntUaJUfRKbUjXNkl0aBFfZ39xi5Sx+QvsIrxeb70qgsqW6YuuhZvM\nVFdkuI1bx3SlW9sklnz6K4XFLl5d+QtrthzkuuGpdGqVaEocRcUutqVnsXlXJlt3Z1FYXHFr99io\nMHq0T+a8Nkl0apVAUoNIU+ISERXYQecoLbDDa3nv9amE2axMHNWJfp1SeGXlLxw5ZueLTQdYt+0w\nF/Vuxsj+rQL+0WhZ68ePuzPZsiuLfUcqLhFmGJDaPJ5eHRrSK7UhTZKi61UBImL1RAHgNTzYXXai\nw6JDHFHglG2VXh/+EB54XhO6tEni/z79le93HGHv4Xye/r9NdGmdyOhBrenSOjGg9yav18vBrCK2\n7s7ix92Z7Nyfe9IykM0axtAzNZleqQ1p3yy+Xs4nEakL6v4drpYr22imLo5gl9elTRKPTz6fVT/s\n58Nv9mAvcbPyu318+v1+enVoyAXdmtCtbVKNt4PPyi1m5/4ctqZlszUtiwJ7xdaPyHAr3dol0ys1\nme7tkomLNmf0XCQUylpEAPIcBfWqwPaPYNeDAhsgPiacO6/sxsV7j/HG5zv5LaOAn/ce4+e9x2iS\nFM3g7k3o37kRjRJr9t8wr9DB7gO5bEvP5sfdWWTlFVd43mox6NgygZ6pDemVmlzj64hIYNWPO1wt\n5h/BrkXbpNdUmM3K5QNaM7RHM1Zt2MdnP+ynsNjFxl+PsvHXo4SHWejQIoEOLeJpkRJL48Qo4mMj\niAy3YrUYuNxe7CUuMnOLOZJTVPpvMT+lZZGdf/KSO40SouiRmkzP9g3p1CqhVu/6JhJIFQvsfJrE\nNAphNIFV9sdzTB1uEalM59aJPHxLf7bszOTDdXtJP5TH4ewi3lmTxjtr0vz9z60ax9E0OYbEuAii\nI22EWS14vF5KHG6y80rIzLVzNLeYQ5mF7D6Yy9Gc4pOuFRcdRvd2yfRon0y3tkmaZyJSC9WvO1wt\nVOKquz3YpxIbFcaVQ9tx+cDWbNhxhK9+PMTOfTk4nB62p2ezPT27Rue1WS2kNm9Aj/YN6ZmarNYP\nOWdZsOF12TBsLvIc5m+6FSxFxS7sJb57Yn3sB7YYBr07ptC7Ywrph/L4cstBfvjlKAV2J0eO2Tly\nzA4cqtF5WzeJo0d7X1HduklcrV8aUORcpwI7yMqvIlLfRIRZGdy9KYO7NyW30MHW3Vn8ui+HtEN5\nZGRXvUV0RJiVRolRtGrSgJYp0bRvFk+rxnGE1dFedZFA8zrD612BnZ1/fDQ2qUHtWtYu0No2bUDb\npg24cWRHdu3P5ee9x9h9MI/9RwvILXCc8nWR4VYaxkfRKDGKtk3jSG0eT5smDYgIr3+/Q0TqMxXY\nQVZSxyc5Vld8TDhDejRlSA/furYut4ec/BJyixyUONy4PV7CrBYiwq0kNfBtWmCzWbTGp8gpeJ0R\nEFVEvqPg9AfXEdml/cOGAQmx9bvALmO1WOjUKrHCyiIlTje5BSXYS9w4XR4sFoPwMAsJsRHERNr0\nyZ1IPaACO8gcdXiZvrNhs1pomBBFw4SoUIciUid5nb4CNK+kHo1g5/nmWiTERpzTcyp8n+BpMqJI\nfVatO9zy5cu5/PLL6d27N6NHj2bVqlUAZGRkcMcdd9C/f3+GDBnCrFmz8JTuEer1epk1axYDBw6k\nf//+zJw5E7fbHbyfpJaqzy0iIhJETt9KOfWpRaRsBYz63h4iInLaAjs9PZ2//e1vPPHEE2zatIkZ\nM2YwdepUsrOzmTlzJq1atWLdunW8/fbbLF++nGXLlgGwZMkSvvjiC5YtW8by5cvZuHEjixcvDvoP\nVNs4XPVnFRERMU/ZCHZ+PSqwy1pEkuvhBEcRkfJOW/W1bduWtWvX0qdPH1wuF5mZmcTExBAeHs6e\nPXtwu93+UWuLxUJEhO+XwtKlS7n55ptp1KgRKSkpTJkyhffeey+4P00tVJd3chSR0PG3iNSjAjur\ntEWkPq4gIiJSXrV6sGNiYti3bx+jRo3C4/Hw97//ndjYWCZPnsxDDz3E66+/jtvt5qqrruLyyy8H\nIC0tjdTUVP852rZtS3p6erW3/TUMA0sIBn3Ldr0K1O5XztIWkchwK1arJq6UF+hcS9WUb/Ocba4N\nw8Bb2iKS7yzEsHixGObfEA3v8fgtFuOs72HHSlcRaRgfGbD7od7X5lK+zaNcmycYua72JMemTZuy\nZcsWNmzYwF133UXr1q0BmDJlCpMnT+bAgQPccccdvPHGG1x33XXY7XYiI4+PUkRFReHxeHA4HP5R\n7qokJ8eEdCZ1QkJMQM7jKl2qLiE+mqSk2ICcs74JVK6lepRv89Q01z06prDtyzQAPF4PYTEQH2n+\n/cPj9fi/bhAXdVb3MLfHy7HSDaVaN08I+P1Q72tzKd/mUa7NE8hcV7vAttl8hw4aNIiRI0fy0Ucf\n8cEHH/D9998THh5Oamoqt99+u7/AjoyMpKTk+O58drsdm81WreIaICurMGQj2AkJMeTkFOKpYh3n\n6ioq9m0L7HK6yM6uP8ttBUKgcy1VU77Nc7a5HtQlhf+sOT5A8VtGBs3jzL8hli+w8/LtZIfX/B52\nLL8EV+lynBEWAnY/1PvaXMq3eZRr81Q312cyMHDaAnvNmjW89NJLvPzyy/7HnE4nubm5OJ1OnE4n\n4eG+jzKtVitWq6/XuH379qSnp9OzZ0/AN1myXbt21Q7M6/USykVHPB5vQNZmLlumL8xq0VrPpxCo\nXEv1KN/mqWmuoyPCGNSlNd+Vfp9ZmEuT6CaBDa4aPN7jsZ/t++boMbv/64TYiIC/B/W+NpfybR7l\n2jyBzPVph0S6du3Ktm3beP/99/F4PKxZs4Y1a9YwdepUmjRpwtNPP43D4WD//v0sXryY0aNHAzBu\n3DgWLVrE4cOHyczMZMGCBYwfPz4gQdclWkVERGri8v5t8DrDAFi785cQR3P2skvbQ8LDLMREagsG\nEanfTlv1paSkMH/+fF599VX69evH7Nmzef755+ncuTMvvPAC+/fvZ8iQIUycOJErrriCm266CYAb\nbriB4cOHc+211zJ69Gj69OnDpEmTgv4D1SZuj8f/kajWwRaRM9EoMZokWgLwY/735NoLQxzR2cnK\nLV0DOy5SOxWKSL1XrWGEfv368e677570eGpq6inXtrZarUybNo1p06adXYR1WNkmM6Bl+kTkzN3U\naxz/u3U2RlgJi777iOnDJoQ6pBo7vga2NpkRkfpPfQtBVNZ/DRBuU6pF5Mx0bNyMFkY3AHY5NrE3\n62iII6q547s4ag1sEan/VPUFUYHd6f86Wj2HIlIDt/YfB64wDKubed+/7t/Yq645mFUEQKPEqBBH\nIiISfCqwg6hsxMYwfLPmRUTOVKMG8ZyfcBEABeH7eXfLN6ENqAbsJS4ysn0FdusmcSGORkQk+FRg\nB1HZtsAJsRHYrEq1iNTMxH6XEFnSGIDVR1ayPyc7xBGdmd8yjm/33rqxCmwRqf9U9QXR8Uk96jkU\nkZqzWCzc1vv3eN1WCCvhf9e/jMsTwo0CztDew74CO7lBBHHR4SGORkQk+FRgB9HxST1qDxGRs9O5\nSQsGJVwKgD38MPO+XhriiKpvb+kIdiuNXovIOUIFdhBll677mhyvEWwROXsT+19Ckqs9ADuc6/lg\n67chjqh69mb4tkVvo/5rETlHqMAOorIebLWIiEig3D/0JmwlCRgGfHx4KZv2pYU6pCqVONwcyvJt\nkqMJjiJyrlCBHSRuj4djpVsDa91XEQmUBlFRTO0/GZwRGFYXL/70MjszDoY6rFPad6QAr29DW1o3\naRDaYERETKICO0hyCxx4Sn+raARbRAKpbcPG/FeH/yqd9FjM7E0L+S07M9RhVWrP4TwAEmLDiY/R\nBEcROTeowA6SsgmOoK2BRSTwLmjXmfEtfofXY+ANL+T/ffd8rRzJ3rk/F4A2Gr0WkXOICuwgKSuw\nI8OtREVoF0cRCbxRXfowstGV/iL7fzfPZ/P+PaEOy8/t8bA93bdm93ltk0IcjYiIeVRgB0l2uQmO\nhmGEOBoRqa+u7DGI8c1/728XWfjTQtal7Qh1WACkHcyjqMQFQPf2ySGORkTEPCqwg+T4GtjqvxaR\n4BrVpQ83tJsIrjCwOXlt9yu8s3ltqMNia1oWAI2TommUEBXiaEREzKMCO0i0BraImGlI+67c2qVs\ndRE3n2cv5Z+r3wzpjo8/7vYV2N3bqT1ERM4tKrCDJMu/TbomOIqIOXq3bMcD/e4hrMRX0KZ7f+Bv\nnzxHVkGB6bHkFpTwW+kGMz3aqT1ERM4tKrCDwOv1kpmrFhERMV/r5BSeumQ6ya4OABSGH+CRr2ex\ndvfPpsaxeZdv2cBwm4VOrRJMvbaISKipwA6CIzl2ih2+j2VbpMSGOBoROddEhYfz90sn0zNymH+F\nkSXpLzP7y3dwuc1pGVm79TDgm9wYZrOack0RkdpCBXYQ7DmUD0CYzUKzhtEhjkZEzkUWi4XbLxjN\nxHaTsDhiMCxefnV9ywOfPMuuI4eCeu2DmYXsOuBb/3poj2ZBvZaISG2kAjsIynYua9UoFqtFKRaR\n0BnUrjMzh93nbxkpiTjCM1vmsGj9CjweT1Cu+fVWXwGfGBdBN61/LSLnIFV/QVA2gq2dy0SkNoiP\niuGxkbdxUeJYcIVhWN1sLPqc+1c+y68B3v3R5fbwTWmBPbh7EywW7QMgIueeahXYy5cv5/LLL6d3\n796MHj2aVatWAeBwOHj88ccZMGAAAwYMYMaMGTgcDsA30W/WrFkMHDiQ/v37M3PmTNwm9f6Fksfr\nZW+Gr8Bu3SQuxNGIiBz3u95DmXH+vTRwtgKgOCKD//1xLi9881HAlvP79qcM8oqcAAzp3jQg5xQR\nqWtOW2Cnp6fzt7/9jSeeeIJNmzYxY8YMpk6dSnZ2Ns888ww7d+5k5cqVrFy5kl27drF48WIAlixZ\nwhdffMGyZctYvnw5Gzdu9D9Xn2VkF/knOLZpqgJbRGqXZglJPDXq7tLR7HAMq5stxWt4YOUz7Di8\n/6zO7XJ7WPp1OgC9OzSkUaLmoIjIuem0BXbbtm1Zu3Ytffr0weVykZmZSUxMDGFhYbz55ps8/PDD\nJCQkkJCQwJw5cxg7diwAS5cu5eabb6ZRo0akpKQwZcoU3nvvvaD/QKG257Bv9Do8zELTZP1yEZHa\n6Xe9hzJjwL3EO9sAUBJxlDnbnmP+2g9rPJr99dZDZOYWYwBXDW0XuGBFROoYW3UOiomJYd++fYwa\nNQqPx8Pf//53MjIycLvdbNmyhbvuugu73c6YMWOYPn06AGlpaaSmpvrP0bZtW9LT0/F6vRjG6Xvy\nDMMgFPMDy/oFa9o36G8PaRxHeJiWpqrK2eZazozybZ66kuuWSUk8fcUfeXvj16w6vAIjrIStJV/y\nl0928Mf+N9I+pbH/WIvFwGo99c9TVOzig7V7ABhwXmNam/QJXl3JdX2hfJtHuTZPMHJdrQIboGnT\npmzZsoUNGzZw1113MXnyZJxOJ6tXr+btt9+msLCQKVOmEBcX5y+4IyOPb7ISFRWFx+PB4XAQEXH6\n3Q2Tk2OqVYgHS0JCTI1eV7ZzWee2ySQlaQ3s6qhprqVmlG/z1JVc337pZYw5NoDHVrxItiWN4vAj\nzNo4hyGNRviPaRAXdcp7mtfr5YXXNnAsvwSb1cItY7uZfv+rK7muL5Rv8yjX5glkrqtdYNtsvkMH\nDRrEyJEj2bx5Mx6Ph6lTp9KgQQMaNGjApEmTeO2117jrrruIjIykpKTE/3q73Y7NZqtWcQ2QlVUY\nshHshIQYcnIK8Xi8Z/TaAruTX347BkCbRjFkZ5u/PXFdcja5ljOnfJunLuY6kjCeHHUnb/7wJauP\nrACbk6+zPvY/n5dvJzu88nvaZxv2s3aLbzWS6y5JJcqKafe/upjrukz5No9ybZ7q5vpMBg5OW2Cv\nWbOGl156iZdfftn/mNPppE2bNqxdu9a/aghQYZWQ9u3bk56eTs+ePQHfZMl27arfk+f1egnloiMe\nj0kAmCQAACAASURBVBe3+8ze0Ft3Z+H1gtVi0LlV4hm//lxVk1xLzSnf5qmLub6211AG5pzH3PX/\npiD8+KTH/EIH7oSKP4vX62Xld/t4a/UuAHqlNuTi3s1D8jPXxVzXZcq3eZRr8wQy16cdI+7atSvb\ntm3j/fffx+PxsGbNGtasWcP111/PpZdeyjPPPENeXh4ZGRm88sorXHbZZQCMGzeORYsWcfjwYTIz\nM1mwYAHjx48PSNC11dbdWQB0aBFPVES1PxwQEalVWiQk8dTIuxkQOxKvy4bXY+AoiKpwTF6hgxc+\n+MlfXLdt2oA/jO4S0tY+EZHa4rRVYEpKCvPnz+fJJ5/kscceo02bNjz//PO0b9+ep556iqeffpor\nrrgCp9PJlVdeyR/+8AcAbrjhBjIzM7n22mtxOp2MHTuWSZMmBf0HChWP18vW9GwAurdPDnE0IiJn\nx2KxMLH/JXz9jAcXLpbtziAlJonmDWP56seDfPjNHgqLXYBvSb7bx51HhCZ2i4gAYHi93lr5ucPR\no/khua7VapCUFEt2dsEZfUyw93A+j778PQCPTT6fFima4Hg6Nc211IzybZ76lOuvthzk1ZW/4K6k\nLzEy3Mo1w9pzcZ/mWEI0cl2fcl0XKN/mUa7NU91cp6RUf3Uk9TEEyJbdmQAkxkXQvKFm/IpI/TC0\nZzOaNozhhWXbycwtBnzzTAZ3b8r4IW1JjKvexHURkXOJCuwA8Hq9rNueAUCvDg3Vgygi9Upq83j+\nMWUQmbl2Mo7ZaZYcQ3J85OlfKCJyjlKBHQA79+eSkV0EwJDuTUMcjYhI4FksBo0So7X9uYhINYRg\npen656vS9V9bpMTSpok5u5eJiIiISO2kAvssFRW7+H7HEQAu7NlU7SEiIiIi5zgV2Gfp662HcLg8\n2KwWBp7XJNThiIiIiEiIqcA+C/YSFx+t2wPAwPMaExsVFtJ4RERERCT0VGCfhU837CO/yInNajB+\ncNtQhyMiIiIitYAK7BPYS1zYS1ynPS630MGKb38D4OLeLbRklYiIiIgAKrArKLA7mT53LXf/z+fk\nFTpOeZzX6+Wl5T9T7HATGW5l9AWtTYxSRERERGozFdjl2KwGHq+XI8fsLP7oZ061i/zqTQf4cXcW\nANdf2oEG0eFmhikiIiIitZgK7HIiw23814iOAGzamcma0vWty/t57zHe+GwXAP06pWhjGRERERGp\nQAX2CYb2bMqA0uX2Xlv5C5/9sN//3K79ucx5+0dcbg8N4yO56bLOWvdaRERERCrQVuknMAyDP03o\nxcHnv2bfkQKWfPor3/+cQUJcxP9n787DoyrP//G/z+wzSchGwg6SIAHZwg6CQsFdAbWgltoPBUVQ\nq9aFX9Vebh9oq1dFpaIiFK24VL9YF4pWEYroBxREBAQEhUQWIUL2bfY5vz8m58w5Z5bMTCbJTHi/\nrsuSmTlz5uRh+szNPfdzP/jq4GmIIpCVbsJ9vxrOtnxEREREFIQZ7BAy08344/+MxOCCHADA9ydq\nsOM7f3Cdl2XBvTcMR36WtZ2vkoiIiIiSETPYYVjNBvx+5jB888MZ7Pr+DE5X2zFmYBdMLu4Bo4H/\nLiEiIiKi0BhgR6DTCRhZlI+RRfntfSlERERElCKYiiUiIiIiSiAG2ERERERECcQAm4iIiIgogRhg\nExERERElEANsIiIiIqIEYoBNRERERJRAgiiKYntfBBERERFRR8EMNhERERFRAjHAJiIiIiJKIAbY\nREREREQJxACbiIiIiCiBGGATERERESUQA2wiIiIiogRigE1ERERElEAMsImIiIiIEuisDLAPHDiA\nmTNnori4GDNmzMDu3btDHrd+/XpMnToVxcXFWLBgAcrLy9v4SjuGnTt3YtasWRg5ciQuuugivPnm\nmyGPW7BgAYYOHYrhw4fL/1FsVq9ejcGDB6vGcOfOnUHH8b3dcuvWrVON8/DhwzFgwAA89NBDQcfy\nvR2/vXv3YuLEifLtmpoa3H777Rg5ciQmT56MtWvXhn1utHM9BWjHu6ysDLfddhvGjh2LCRMmYPHi\nxXC5XCGfG+38Q37asf72228xcOBA1fitWLEi5HP53o6NcqxPnjwZNHcPGjQIl156acjnxv2+Fs8y\nDodDvOCCC8TXX39ddLlc4tq1a8Vx48aJ9fX1quO+++47ccSIEeLu3btFu90uPvjgg+LNN9/cTled\nuqqrq8XRo0eL69atE71er7hv3z5x9OjR4tatW4OOnThxorh37952uMqO45577hH//ve/RzyG7+3W\nsXXrVnHChAniqVOngh7jezt2Pp9PXLt2rThy5EhxzJgx8v133HGHeN9994kOh0Pcs2ePOGbMGPGb\nb74Jen60cz35hRvvG2+8UXzsscdEh8Mhnj59Wpw1a5b41FNPhTxHNPMPhR/rt956S7zllluafT7f\n29ELN9ZKp0+fFidMmCBu2bIl5OPxvq/Pugz2l19+CZ1Oh9mzZ8NoNGLmzJno3LkztmzZojru3//+\nN6ZOnYphw4bBYrHgvvvuw+eff85MX4xOnjyJSZMmYdq0adDpdBg0aBDGjh2LXbt2qY6rqKhAZWUl\n+vfv305X2jF89913GDhwYMRj+N5OvIaGBtx///149NFH0bVrV9VjfG/HZ8WKFVizZg0WLlwo39fQ\n0ICNGzfizjvvhNlsxtChQ3HVVVfhvffeC3p+tHM9+YUab5fLBavViltvvRVmsxl5eXmYNm0avvnm\nm5DniGb+odBjDfiz0gMGDGj2+XxvRy/cWCs98sgjuPzyy3HhhReGfDze9/VZF2CXlpaisLBQdV/f\nvn1RUlKiuq+kpAT9+vWTb2dnZyMzMxOlpaVtcp0dxcCBA/HXv/5Vvl1TU4OdO3cGTSIHDhxAWloa\nFixYgHHjxuGGG24IO4lTaHa7HaWlpVizZg0mTJiAyy+/HG+//XbQcXxvJ97f//539O/fHxdddFHQ\nY3xvx+eXv/wl3n//fQwZMkS+7+jRozAYDOjVq5d8X6j5G4h+rie/UONtMpmwcuVK5OXlyfdt3rw5\nZBAY7fxDocca8Adyu3btwpQpUzB58mQ88cQTIctx+N6OXrixlnzxxRfYtWsXfv/734d8vCXv67Mu\nwG5sbITValXdZ7FY4HA4VPfZ7XZYLBbVfVarFXa7vdWvsaOqq6vDwoULMWjQIEyZMkX1mNPpRHFx\nMf74xz/is88+w/Tp0zF//nycOXOmna429ZSXl2PkyJH41a9+hc2bN2Px4sV4/PHHg7IafG8nVkND\nA1577TX87ne/C/k439vxyc/PhyAIqvsaGxuD3ruh5m/p2GjmevILNd5KoihiyZIlKCkpwYIFC4Ie\nj3b+ofBjnZ2djSlTpmD9+vV49dVXsX37dvztb38LOo7v7eg1975euXIl5s2bh7S0tJCPt+R9fdYF\n2FarNehN6HA4YLPZVPeFC7q1x1F0jh8/jhtuuAGZmZlYvnw5dDr1W++iiy7CypUrce6558JkMmH2\n7Nno1q0btm/f3k5XnHp69eqF1157DZMmTYLJZMKoUaMwY8YMbNq0SXUc39uJtXHjRnTv3h3FxcUh\nH+d7O3GsViucTqfqvlDzt3RsNHM9Nc/hcOCuu+7C559/jldffRW5ublBx0Q7/1B4K1aswNy5c2Gz\n2dCrVy8sWLAAn3zySdBxfG8nxqlTp/DVV19h5syZYY9pyfv6rAuwCwoKgr4KLy0tVX1lDgCFhYWq\n4yorK1FTUxP0tQw1b//+/bjuuuswceJEPP/880EZKAD46KOP8OGHH6ruczqdMJvNbXWZKW///v1Y\nuXKl6j6n0wmTyaS6j+/txNq8eTMuv/zysI/zvZ04ffr0gdvtxsmTJ+X7Qs3fQPRzPUVWXV2NG2+8\nEdXV1XjrrbdU5TlK0c4/FFpNTQ2eeOIJ1NfXy/eFmyf43k6MzZs3Y8yYMcjJyQl7TEve12ddgD1+\n/Hi4XC68+uqrcLvdePvtt1FeXq5qlQMAV111FTZs2ICdO3fC6XTiqaeewoUXXojs7Ox2uvLUVF5e\njptvvhlz587FAw88EJS5ljQ2NuJPf/oTDh8+DLfbjb///e9wOByYMGFCG19x6rLZbFi+fDk++ugj\n+Hw+fPHFF/jggw9wzTXXqI7jezux9uzZEzZ7DfC9nUjp6emYOnUqli5dCrvdjr1792L9+vWYNm1a\n0LHRzvUUniiKuOOOO9C5c2esXr0aWVlZYY+Ndv6h0DIyMvDJJ59g+fLlcLvdOHr0KFasWIFrr702\n6Fi+txOjubkbaOH7Oua+Ix3Ad999J15//fVicXGxOGPGDLnF00MPPSQ+9NBD8nEffPCBeMkll4jD\nhw8X58+fL5aXl7fXJaesF154Qezfv79YXFys+u+pp54KGu8VK1aIkyZNEocNGyb+6le/Eg8ePNiO\nV56aNm3aJF511VXisGHDxEsuuUT8z3/+I4oi39utxePxiEVFReLhw4dV9/O9nThffvmlqr1WVVWV\neOedd4qjR48WJ02aJK5du1Z+7P333xevuOIK+Xa4uZ7CU473119/Lfbv318cMmSIav6ePXu2KIrB\n4x1u/qHQtO/tH374QZwzZ444YsQI8fzzzxeXLVsm+nw+URT53m4p7ViLoij++te/Ft94442gYxP1\nvhZEURQT9I8BIiIiIqKz3llXIkJERERE1JoYYBMRERERJRADbCIiIiKiBGKATURERESUQAywiYiI\niIgSiAE2EREREVECGdr7AoiIKLL7778f7777btjHx4wZgx07dmDv3r3cIZKIKAmwDzYRUZKrq6uD\nw+EAAHzzzTe444478N///lfertdqtcJutyMvL689L5OIiJowg01ElOQyMjKQkZEBAMjMzAQAdO7c\nWZWtTk9Pb5drIyKiYKzBJiJKcdu3b0dRURGcTicAoKioCB9++CGmTZuGoUOHYt68eTh16hQWLVqE\n4uJiXHLJJfjyyy/l5//888+48847MXz4cFxwwQV49NFH0dDQ0F6/DhFRymOATUTUAS1duhQPP/ww\nXnvtNezfvx/Tp0/HwIED8a9//Qv9+vXDY489BgAQRRG/+93vYDQasXbtWixfvhwHDx7Egw8+2M6/\nARFR6mKJCBFRB/Sb3/wGo0ePBgCMGzcOx48fx7x58wAA119/PRYuXAiv14sdO3agtLQUb7zxBoxG\nIwDgL3/5Cy677DKUlZWha9eu7fY7EBGlKgbYREQdUO/eveWfrVYrevXqJd+2WCzw+XzweDw4cuQI\n6uvrMWbMmKBzlJaWMsAmIooDA2wiog7IYFBP7zpd6IpAj8eD3r17Y9WqVUGPsSsJEVF8WINNRHQW\nKywsRFlZGTIyMtCnTx/06dMHHo8Hjz/+OOrr69v78oiIUhIDbCKis9iECRNQWFiIe+65B/v378e+\nffuwaNEiVFVVIT8/v70vj4goJTHAJiI6i+l0Ojz//PNIT0/HjTfeiHnz5uGcc87B8uXL2/vSiIhS\nFndyJCIiIiJKIGawiYiIiIgSiAE2EREREVECMcAmIiIiIkogBthERERERAnEAJuIiIiIKIEYYBMR\nERERJRADbCIiIiKiBGKATURERESUQAywiYiIiIgSiAE2EREREVECMcAmIiIiIkogBthERERERAnE\nAJuIiIiIKIEYYBMRERERJRADbCIiIiKiBGKATURERESUQAywiYiIiIgSiAE2EREREVECMcAmIkpx\noii29yUQEZECA2wiSmlTpkxBUVERnn322ZCPr1+/HkVFRbjuuusS+rrPPvssJkyYkNBz3n///TFd\np8vlwmOPPYZt27a16HVPnDiBoqIifPbZZ0GPNTQ0oLi4GI8++mjY5//hD3/AxRdf3KJruPDCC/H0\n009Hffx9992HX/3qV2Ef93g8KCoqwtq1a1t0XURE8WCATUQpTxAEbNy4MeRjGzZsaJXXnDVrFlau\nXNkq547W6dOn8cYbb8Dr9bbaa6SlpWHq1Kn45JNP4PP5gh53Op3YuHEjZsyY0aLXWbFiRcSAmYgo\nlTDAJqKUV1xcjIMHD+LEiROq+x0OBz7//HP0798/4a/ZtWtXDBo0KOHnTUYzZsxAeXk5duzYEfTY\np59+ivr6ekyfPr1Fr3Heeeeha9euLToHEVGyYIBNRClvxIgRyM3NDcpif/7558jOzg4KhGtra/HY\nY4/hwgsvxODBgzFx4kT8+c9/htvtBgBs374dRUVF2LVrF2bOnIkhQ4bg0ksvVZ1fWyIilSPMmzcP\nQ4cOxeWXX47//Oc/8uPvvPMOioqK4HQ65fuOHDmCoqIibN++PeTvVVZWhkWLFuH888/HoEGDMGXK\nFLzwwgsA/GUdU6dOBQDMnz8f999/PwB/PfbKlSsxZcoUDBkyBNdee23Q+Q8fPow5c+aguLgYV155\nJfbt2xdxfCdMmIDOnTvjo48+Cnrsgw8+wPDhw9G7d28A/oz2k08+ialTp2Lw4MEYN24cHnjgAdTX\n1wMAjh49iqKiIrz66qu44IILMGbMGBw/fjyoRGTnzp347W9/ixEjRmDIkCGYMWMGNm/eHPT6f/vb\n3zB27FiMHTsWixcvhsPhCPt7/PDDD7jppptQXFyMcePGYcmSJaq/DyKiRGGATUQpT6fTYcqUKdi0\naZPq/g0bNuDSSy8NOv6ee+7Btm3b8MADD2DVqlW49tpr8corr+Ddd99VHXfffffh6quvxosvvoju\n3bvjnnvuQXV1ddjrePzxx9GlSxcsX74cQ4cOxd13340vvvgirt/J5/Ph5ptvxo8//ojFixdj1apV\nOP/88/HMM89g69atyM/Px/LlywEADzzwAG677TYAwNNPP41nn30W119/PZ577jn07dsXN998M/bv\n3w8AqKurw5w5c+B0OvH000/j+uuvxx//+MeI16LX63HllVcGlYnU19fj008/xdVXXy3ft3jxYrz3\n3nu44447sHr1asybNw/r1q3DSy+9pDrnypUr8fDDD+OPf/wjevXqpXrs2LFjmDt3Lrp27YrnnnsO\nzzzzDKxWK+699145UAeAPXv2YOPGjfjTn/6Eu+66C++8807YWvGysjLceOONcDgceOqpp3Dvvffi\n3//+Nx544IFm/iaIiGJnaO8LICJKhIsvvhi33norqqqqkJ2dDbfbjc2bN2PVqlV466235OMcDge8\nXi8ee+wxjBs3DgAwfvx4fPrpp/j6669Viwznz58v1wV369YNl112GbZv3x4yaAf8ZQ5/+ctfAPgX\n7R05cgQvvfQSxo8fH/PvU1ZWhuzsbDz22GMoKCiQr/Pjjz/G119/jQkTJmDgwIEAgIKCAvTu3RvV\n1dV4+eWXcccdd+CWW26Rr+PXv/41nnvuOTz//PN49913UV9fj+effx45OTkA/AsCn3jiiYjXM2PG\nDLzyyivYsWOHPG4bN26Ez+fD5ZdfLh9XXV2NBx98EFdccQUAYOzYsdi5cye+/vpr1flmzZoVdmHk\n4cOHMW7cOPzlL3+BIAgAgLy8PMyaNQsHDx7EqFGjAPj/YbVq1Sp06dIFgD97v2TJEtx7773Izs5W\nnfPll1+GwWDAqlWrYLPZAPjLfG6++WYsXLiwVcqIiOjsxQw2EXUI48ePh9VqlcsIvvjiC1itVhQX\nF6uOs1gsePnllzF27FgcO3YMW7ZswcqVK1FRUSGXiEiGDRsm/yzVB9vt9rDXIAWVkilTpmDXrl1x\n/T7du3fHq6++ij59+qCkpASbNm3C8uXL4fF4gq5TsmfPHrhcLlxwwQXweDzyfxMnTpTLRHbt2oXB\ngwfLwTWAqDqADBo0CIWFhaoykfXr1+MXv/gFMjMz5fuWL1+OK664AidPnsTWrVvx8ssvo6SkJOia\n+/btG/a1pkyZglWrVsHhcGD//v1Yv349/vnPfwKA6jxDhw6Vg2sAmDx5Mnw+H/bs2RN0zq+++gqj\nRo2CyWSSx2Xs2LEwGo1hS3SIiOLFDDYRdQgmkwmTJk3Cxo0bce2112LDhg24+OKL5QyoklRWcPLk\nSXTu3BnFxcUwm81B/aTNZrP8s07nz0eE6qQhycvLU93OyclBfX19xOdE8tZbb+Hpp59GVVUVunXr\nhpEjR8JgMITtey2VryhLNpTsdjtqa2uDsru5ublRXc+MGTOwZs0aPPzww6iursYXX3yBZcuWqY75\n6quv8PDDD6OkpARZWVkYMmQIrFZr0DVHek2Px4PFixfjX//6F3w+HwoKClBUVARA3fNbew7p96qr\nqws6Z3V1Nfbv3x+yjvzMmTPN/OZERLFhgE1EHcbFF1+M+++/Hw0NDfjvf/+Lp556KuiYH3/8Eb//\n/e8xe/ZszJ8/Xw6KZ82a1eLXr6mpUd2uqKhAVlYWdDqdHOgrW+o1NjaGPdeOHTvwyCOP4N5778V1\n110nZ4nPP//8sM/p1KkTAGD16tWqrLLEZDIhMzMTZWVlEa87nGnTpuHpp5/GV199hZKSEqSnp2PS\npEny49XV1bj11lsxadIkrF69Gt27dwcA3HHHHSgvL4/qNQDgueeewwcffIDly5dj/PjxMJvNOHTo\nENavX686rra2VnW7oqICAFTZeUlGRgYuv/xy3HTTTUGPaf9hRETUUiwRIaIO48ILL4Qoinj++efh\n8/kwevTooGMOHDgAt9uNBQsWyIFVeXk5vv/++7gzzZItW7aobm/atEmuF5bqfpXBrbYuWWn37t0w\nm82YP3++HCwfPHgQFRUV8nXq9XrVc4YOHQqDwYCamhoMGTJE/m/r1q345z//Cb1ej9GjR+Pbb79V\nXcfnn38e1e/XvXt3jB49Gps2bcLGjRtxxRVXwGg0yo8fOXIEdXV1mDdvnhxcNzY24ptvvolpt8nd\nu3dj1KhRmDx5svwtgrSZjvLvaO/evapFj5988gmMRiOGDh0adM7hw4ejpKQEgwYNksclLy8PS5cu\nxbFjx6K+NiKiaDCDTUQdRlpaGs4//3y88soruPrqq4MCUAAYMGAA9Ho9Hn/8cfzyl7/E6dOnsWLF\nCjidzoj11dH45JNP8MQTT+D888/HunXr8P3338tdLcaMGQOj0YglS5Zg/vz5KCkpwZo1a8Kea8iQ\nIXA4HHjiiSfwi1/8Aj/++COWL18OQRDk68zIyADgD5B79uyJgoIC3HDDDXj00UdRXl6OoqIi7Nix\nAy+88ALuuusuAMA111yD1atX45ZbbsGdd96J8vLysLtghjJ9+nQ899xzqKiowJ133ql6rLCwEDab\nDcuWLcPcuXNRU1OD1atXo7y8PGRWOZzBgwdjzZo1WLt2LXr37o3t27dj1apVANQ18F6vF7feeisW\nLFiAI0eOYNmyZfjNb36D7OxseDwe1TnnzZuH999/H7fffjtuuOEGuFwuPPvss6iurpbLT4iIEoUZ\nbCLqUC6++GK43e6wC/cKCgrw5z//Gbt378Ytt9yCZ599FlOnTsXChQvx7bfftmhXxFtvvRX79u3D\n7bffjiNHjmDVqlVyNjU7OxvPPPMMTp06hfnz52PdunVB9ctK48ePx6JFi/Dhhx9i/vz5ePnllzFn\nzhxcc8018iK+9PR0zJs3D2+99ZbcBeTBBx/EnDlz8Morr2D+/Pn44IMP8Ic//AELFy4EAFitVrzy\nyivo2rUr7rvvPqxevTriNuhal112GSorK9G9e3fVIlAAyMrKwjPPPIOTJ09iwYIFeOKJJzBo0CA8\n+OCDKCkpCVkbHcrChQtx2WWX4cknn8Ttt9+OrVu3Yvny5ejRowd2794tHzdp0iQMHjwYd911F1as\nWIHf/va3WLRoUchz9u7dG6+99hocDgfuuusuuT3gmjVrQpbTEBG1hCDG8r0dERGFVFRUhEcffZTb\nfRMRETPYRERERESJxACbiIiIiCiBWCJCRERERJRAzGATERERESUQA2wiIiIiogRK2j7YZ85E184p\n0QRBQG5uGioqGmLaGIFix7FuWxzvtsOxbjsc67bF8W47HOu2E+1Y5+VlRH1OZrA1dDr/QOs4Mq2O\nY922ON5th2PddjjWbYvj3XY41m2nNcaaf21ERERERAnEAJuIiIiIKIGiCrA//PBDXH755Rg+fDiu\nvPJKbNy4EQBQU1OD22+/HSNHjsTkyZOxdu1a+TmiKGLp0qUYN24cRo8ejSVLlrRoC2IiIiIiolTQ\n7CLH0tJSPPjgg3jppZcwYsQIbNu2Dbfccgs+++wzPProo7DZbNi2bRsOHTqE+fPn49xzz0VxcTFe\nf/11fPrpp1i3bh0EQcCCBQvw0ksvYf78+W3xexERERERtYtmA+y+ffti69atSEtLg8fjQXl5OdLS\n0mAymbBx40Z8/PHHMJvNGDp0KK666iq89957KC4uxvvvv485c+YgPz8fALBgwQIsW7YsJQJsr0/E\nF/vL0Ds/A11zbHGd46cz9dh9uDzofr1Oh1ED8tA504ryGjt2HjwDr8+HLtk2jCzKgyAIAACX24ut\n355Co9PTot8lHiajHhk2I/r3zEJOJ0ubvz4RdRx2pwdf7i8LOZf17pKBIQW5QfdX1jrw3dEqjB6Q\nD5NR36rX1+Bw46vvTqPB4W72WJNRj042E/r1yERuJudGIgovqjZ9aWlpOH78OC699FL4fD48+uij\nOHbsGAwGA3r16iUf17dvX2zYsAEAUFJSgn79+qkeKy0thSiKchAZSXutnNXpBHx98GeseG8/+nTJ\nwP/ePCau8zz7zrc4XWUP+dj+Hyvx/80ejlc/PoRvSyrl+x/+7SgU9sgEAHzyxXH8a0tJXK+dKAKA\n8/rm4LKxvTGkICeqv7dY6HSC6k9qXRzvtsOxDti06wTeCTOXCQD+evv5yMuyqu5/Y+P32PV9Odwe\nH6aO6hnx/PGOtc8nYu3mw9j49Qm43L6YngsAA/tk45LRvTC8f+eEz43JjO/ttsOxbjutMdZR98Hu\n1q0b9uzZg507d+K2227DTTfdBItF/S94i8UCh8MBALDb7arHrVYrfD4fXC4XzGZzs6+Xm5vWs1e5\nBgAAIABJREFUbpNWxYHTAIA6uws5OelxnaOy1j8O3XLTYLP6h7m6zomKGgfq7W7k5KSjzq7O6Dh9\nkF/vmx8qAAC5mRZkZTQ/XokiioDD6UFFrQNOlxf7Syuxv7QSRX2yMfuSARiuyLInSlZWWkLPR5Fx\nvNsOxxryPJduNaJLrv8bQVEESn6qgQjAJQpB86z0nEa3L+o5ONaxfm/LEXz45TEAgNmkR8/8yK8j\niv5sfGXT3Pjd0Sp8d7QK/XpmYvalAzBqYJezKtDme7vtcKzbTiLHOuoA22DwHzp+/Hhccskl2Ldv\nH5xOp+oYh8MBm80/gVosFtXjdrsdBoMhquAaACoqGtotg+3x+puMe7w+VFbWx3wOr88nn+M3l/bH\noL45AICPth/DPzf+ALfHi8rKerg96kWflVWNqKysR0WNAyUnawAAN105UH5+W3J7fPjmhzPYsOM4\nfjhRg0NHq/DIqi9Q2KMTrrmgAIMTkNHW6QRkZaWhuroBPh+b6Lc2jnfb4VgHVNf5kw0j+nfGTVed\nB8C/CP6mxzfD6xPx8+k69MhWJ2tcbv/cWFvvaHYOjmesz1Tb8ep/DgAARg3Iw9wrBiLdaozquR6v\nD3sOV2DjzuM48GMVDp+owf+u3o6C7p1w7YWJmRuTGd/bbYdj3XaiHetYkq7NBthbtmzByy+/jH/8\n4x/yfW63G71798Znn32GkydPonv37gD8CyKlspDCwkKUlpZi2LBh8mMFBQVRX5goimivpiMer//r\nQp8P8Hpjf1PbnYEL1+uEwDma/vA2nVf7l2h3euD1itj1/RkAgNWsR78emXFdQ0vpBAEj++djxLl5\nOHC0Cu9/XorDP9XgyE+1ePLN3Sjs0QkzJvbFoHNa/mHi84nt8juerTjebYdjDdgd/my0yahXjYXN\nYkBdoxv1dnfQGEm3XW5f1OMX7Vj7fCJWr/8OLrcPnWxG/M+lA2A1GaJ+HQECivt1RnG/zjh0rArv\nfl6K749Xo+Skf27s1yMTV1/QFwP7ZHfoQJvv7bbDsW47iRzrZnPE5513Hvbt24f33nsPPp8PW7Zs\nwZYtW3D99ddj6tSpWLp0Kex2O/bu3Yv169dj2rRpAIDp06dj9erVKCsrQ3l5OV588UXMmDEjIRfd\n2gIBdnyD7PYE6vlMhsACHam2R2w6r/b8zqasze4f/IsjB/fNhUHfvq3KBUHAoHNy8MCNI3Dv9cUo\n7NEJAHDkp1o89dYe/OW1XdhfWsltXIkoJIfLP69ZTOp8js3svx1q8aM0n2i/5UuEdz4rwXdHqwAA\nsy/uH3XmOpSi3tn4w+zhuO+GYvRrWj9z+KcaPPnmbjzxxjc4dKwqIddMRKmn2Qx2Xl4eVqxYgT//\n+c/43//9X5xzzjl47rnnUFhYiMWLF+ORRx7BpEmTYLPZsGjRIjljPXv2bJSXl2PmzJlwu92YNm0a\n5s6d2+q/UCJ4mgJkX5xBo/T1JgCYjIEAWaqdl86rPb/T5YXd6ZEn/+JzO8f1+q1BEAQM6puD887J\nxoEfq/De/5XgyE+1OPxTDZa+5c/azJjYF+ed07GzNkQUG4fLH0BbzepuIDaL/+NHynAreX1SgB37\n4sNIvjxQhg+/PAoAmFTcHaMH5Lf4nIIg4LxzcjCwTzb2l1bi3c9LUXqqFt8fr8YTb3yDwh6d8Ivh\nPTB6QD6MhtbtiEJEySOqGuxRo0bhnXfeCbo/KysLy5YtC/kcvV6Pu+++G3fffXfLrrAduL0tDLDD\nZLCFpghbDrA1nx0utw8/nKiG1ydCEBCyfVV7Uwba+3+sxPufl+LIyUCg3TXHhvGDu2L4uZ3Ro3P7\nLVQlouQglczFksH2iYkPsH8sq8XLHx4EAPTvmYlfX9w/ofOTIAgYXJCLQX1zsPdIBd77v1IcLavD\nkZ9qceSnWvxz4w+YMKQbRg3IR0G3TuwMQdTBRb3I8WwiLVDUBsDRcim+1jSqMthNAbYvTAbb7UVd\no78Xa4bV2KKvLlubIAgY3DcXg87JUQXaZZWNePezErz7WQmyM8wo7JGJPl3SkZdlRX62FXlZVtjM\nBgbeRGcJKYNtMamzt9ZIAbaUwfYmJsCuqXfi2X99C7fHh9xOZtx2zZBWK78TBAHD+nXG0MJc7C+t\nxOZvfsLuw+VocHiw4avj2PDVcaRbjejXIxPndMtAl2wbuuRYkZ9lk7P6RJT6+P/mEKQa7HjripU9\nVc3KGmwpwG46bagAO1y9YrJSBtolp2qxbV8Zvj50BrUNLlTVObHz4GnsPHha9RyzUY/MdBOyM8zI\nz0mDzaRDToYFPfPS0KdrBmyW5P2HBRFFTxRFeU6zajPYEUpEpLnRk6AM9tufHkFVnRMmgw53/HIo\nOqWZEnLeSKSM9uCCXFTWOvDZnpP48sDPOF1lR73djd2Hy4M2I7Oa9chKNyMzzYSsDDOy0s3I7WRB\n985p6N0lHWmcG4lSRmpEcW1MrsGOc5Fj2Ax204/SeaXFjnqdAK9PbAqw/R82ZlNq1eoJgoDC7pko\n7O7/6vVoWR0OHatGyckanKpoxJlqu1w643R7cbrKjtNVdhw6Vq0+D/y7uw0tzMWYgfnokRdfH3Ii\nan8er0+up9ZmsG1mf7AYKoOdyBpsj9eHXU0Lx6++oAC9u2S0+JyxyulkwdUXFGDGxL4oq2zEd0f9\n7f1OnGnA6epGOSljd3phdzbiVEVj0DkEAD3y0jG4IAejivLRt1sGvwkkSmIMsEOQM9hA1DtPKrmb\nJkuDXpCz1kAggy3Kixz991vNBtTb3XC5fYoMdmoF2Eo6QUDfbp3Qt1sn+T5RFFFd78KZajuq652o\nqXehttGFRpcXP1c04OdKOypqHRABHP25Dkd/rsO/t/2Igu6dcNGonhg9IB/69miMTkRxs7sCyQap\nJES+3ZTBbgyRwRYTGGD/cKIG9qYgflRRXovP1xKCIKBbbhq65aZhygj/DpXS3Hi6qhHV9S5U1zvl\nObKqzokzNXZU1johAjhxph4nztTjo+3H0C3XhikjemLi0G4wt/J28kQUOwbYIXgUdX8+UYQ+xgDb\n2ZTBNmlWjOuCFjkGMjv1djecbi+cKVYiEi1BEJCdYUa2YldKvd6/g1tlZT28XhGNDjd+OFGD/T9W\n4utDZ1BV50TJyVqsXHcA/976I2ZOKkTxuWfXtsREqcyhyE4HZ7ClGmx30PO8CazB3tNUhtEjLw2d\nNVuyJ4NQc6NWo8ONIydr8d3RKuw6dAanq+04VdGI1z/5Huu3/YhpE87BhcO6t3tbVyIK6FhRXIJ4\nFE3GfT4g1jlLymAry0MA5SLHpnOLUoDt/2tQ1mCnWolIItgsRgzr1xnD+nXGDVPPxYEfK7Fx5wns\nPVKBUxWNePadb9GvZyaum9wP/XpmtvflElEzHIoMdrgAW7kxlyRRXUREUZT3FSjulzxtT2Nlsxgx\npCAXQwpyMWtyIUpO1WLzrp+w/cDPqGlw4bUN3+PjHcdwzQUFGHve2bVlO1GyYoAdgjaDHSup1tis\nyWALYfpgS/1hXYoa7FQuEUkEXdPiycF9c1F6qhZvf3pErlv882tfY/ygrrjuF4XITA+f9SGi9qUK\nsCOUiGhL8aQkREsD7FMVjThdbQeQ2gG2knK9y/SJffH+5yX4cv/POFPtwMp/H8B/v/kJN17cv11q\nzYkogN8nhaCc1ONZ6CgtcgyfwVa3AZRqE51uLxxNm9RYWFMn69utE+67oRh3XzcMPfPSAABf7C/D\ng6u+xIavjsMbbz9FImpVUu2zIAAmg3o+lDLYPlGUd7GVJGqR454j/ux1J5sRfbt3aubo1JOfZcX8\naYPw2LwxGFro3zfh8IkaPPaPr/DahkNocASX3xBR22CAHYIygx1Pqz6pRERbgy1tNCOdUhTVq+ud\nLkWbPjMDbCVBEDCkIBePzB2NX1/cH1azAXanF29u+gGPvvwVtyQmSkLKtqPasgVlz2ftQkd5q/QW\n1mBLu+IO6pujWnDe0fTMT8fvZw3D72cNRX6WFaII/HfXT3jgxS/x2Z6TcW+aRkTxY4AdgrpEJPbn\nBxY5hslgBy1ylDLYvg67yDFR9Dodpo7sib/cMg4Th3QDAPx0pgFPvPENVq7bj6o6ZztfIRFJwm2T\nDgQy2EAg0y1RZrDj3Y/A4/Xhh+M1AIABvbPjOkeqGVrYGYtvHoNrLyyAyaBDvd2Nf/znIP605muU\nnqpt78sjOqswwA5BublBPP/ylzPYRm0XkcDPPp8I6czSh4/H65M/aNh2KbJOaSbMu3IgHvzNSPTu\n4u+V/eWBn/Hgqi/x0fZjqn8kEVH7CLdNOqBu26fsha2dc5WLzmNxtKxOLj0Z0OfsCLABwGjQ46rz\nz8Gf5o+T2xKWnqrFkld24h//OYi6Rlc7XyHR2YEBdgiqEpEW1GCHy2ADUNUNK3c4q23wT35n+yLH\naPXrkYmH54zGby7pjzSLAU6XF/9v82E88tIOfHmgjPXZRO1IzmCHmM8sJr288FtZIqJd9xJvHfbB\nprKx3E5mdM60xHWOVJabacFt1wzBvdcXo1uuDSKAz/acxAMvfon3/68U9XbWZxO1JgbYIaja9MWR\nPJG6iIRb5Kh9DeXqeum5DLCjp9MJ+MWInvjTLeNw4TB/2cipikasXHcAD678Epu/+QluT3ArMCJq\nXZE2zhIEQdELO0KAHee3UQebdokd0Dv7rG5bN6hvDh6bNwbX/aIfzCY9Gp0evP9/pVj0wjb8v/8e\nZlkdUSthoW8IqhrseDLYYRY5ShvNaF8jdHaHfzWx6mQz4beXD8Sk4h7499YfsftwOc5UO/Dqx4fw\nr0+PYPTAfIwf1BXn9sw86z5wfT4RHp8P9XY3auqdcLl9cHt98HhFeDw+eHw+f9mSYgGuKDb9icCu\nphClf3T6HxcEf6Ak/amD5rbiZ0EI/CNT+ln7mOp80N5WnzvoPvl4yH+/ynNS2wu0HQ09n9ksBjQ4\nPKoabG9QBju6fxx7fT58vvcUPvjiKPQ6ARU1DgBA0VlSfx2JQa/DZWN7Y9ygLvh4xzF8+s1JOF1e\nfLTjGD7+6hgG9c3B+YO7YsS5eUGljR2dKIr+edDra/ov+GevT5r7RPk50vzoAwDlXCnGNzcq5zDp\nMYNBh0aPiNpaO0SfGNfcCCjnQfV90BxHicUoLoSW98GOpkQkcF7tFsIAM9gt0bdbJ9w5cyhOnK7H\nf7YfxfYDp9Ho9GDL7pPYsvskOmdaMKywMwaek40BvbNgsxjb+5LDEkURDpcXdqcHjQ4PGpv+tDul\nn93yfco/7U1/utxeuL0+sImA/4MEqiBfgE7w3xfXB0wzYyoICBp3sbknxfE6baFrrg33/3pEzP/w\nb64rkjT3KUtEtIsaoy0RefKfu3Hgx+BuQgN6Z0X1/LNBVroZ1085F1eOPwcbdx7Hpq9PoMHhwb6S\nSuwrqYTFpMegvjk475wcDDonG/nZtva+5LBEUYTb4wsx/7nl+S9oXlT87HR74fH4gv5BdzYKNTdq\nA/JmRTGMUc1/URxiNRtwy7TzMPCcnOYPbkcMsENorUWOgiLeVgbxoYLps3Enx0TrmZ+O+dMG4ZeT\nCvHlgZ/xxb4y/FTegPIaBzbtOoFNu05AEIBe+enolZeOHnnp6JmXhrxsK7LTzQnJ5PhEEU6XVzHR\nu0MEyeqgWPqQ8B/jTboWW9JkLEBqOxlX2NjmmhLv8MrjmQpXnRyO/VyPIz/VYlDf2D7QIi1yBBCy\nRCQ4g918gO1ye+XgelhhLnQ6Ad/8UI6iXllJuT16e0u3GnH1BQW4Ylwf7D5cjm37yrCvpBIOlxdf\nHzqDrw+dAQDkdDKjd34GeuanoWdeOrpk29A5y4KsrJb/f0cURbg8Ps385w4KigOPB8+d8S6AbU2C\n/D/B/8BOVqk2N7o8Lhw/Xc8AOxWpt0pvpQy2sgY7xIcPM9iJk9PJgivG9cHlY3vj+Ol6fHXwNA4e\nrULpqTr4RBHHfq7HsZ/rg55nMxuQmW6CzWyA2aSH2aiHsenvtKlaAmj6OtDp8cLp8v/ncAf+dLm8\nCZ+qdIIAm8Xg/89sgNUc+DnwpxE2swEmox5GgwC9XgezUYec7DQ0NjghCAKMegEGva7pPwE6nRCy\nzEL+2hHhsxnar0dFUYRPjPF20/PljZjEwPiqzg3pOcqfA38qnxvu61sxxH3xCJfc0ekEpKdbUF/v\nCDGHxJ4tb89vcJ9751uICA58o9HczrTSt0eqRY6alwkVYFfVOZGZZpLL7mrqA50xZk4uRI+8dNTb\n3UFzMKmZjHqMGdgFYwZ2QU29E19/fwb7Sytx8FgV7E4vKmudqKx1YvfhctXzDHodstJNsJoNsJr0\nsJgNMBp0Qe9sEf6SSafLA6fb1zQ3euSWtIlOHghA0HwYuG1U3W826uW5T54HDeo5UfpZr1OXVeia\nomjlXBlufpTmF588z0Q/N+p0AjIzbaiqboDH41MfB2XZnlS6otkpOsI82TRFhngs+L6oxz+qear5\ng5o7j81sQP9eyf/NFAPsENwt7IMtLVQMatOnWuQYeA2jwf9/YOUHGGuwE08QBPTukiFvIdzo8OD7\n49UoOVWLn87U46czDThTbZcD4samLEmi6XVChODYCGtQsKwNmnVxlTTo9QJyctJRWVmv+gdeIkgf\nMO0aCSaR1hzrtqZrmpviSTZIJSKhyuCAQAZbWYPdXBeRvUfK8czavZgwuCtuuuo8AEB1vUN+PCPN\nBMCfpaXoZaabMWVET0wZ0RNenw+lp+pQcrIWJ07X4/iZepwsb5D/LjxeH8prHM2cMXaCANVcGGqe\nDJ4fjYGg2aRPug2F5DUmcfzD2j+P2GCAL+XnkbMRo7gQWtymr6n3qlGbwVYtcgycVxD8fa+VwRwz\n2K3PZjGg+NzOKD63s3yf0+1FVZ0TVXVOVNc7UVPvgsPlr9dzurz+DxipPKLpr1OA/+/PbNLD0pTp\nNpv0sJr8GWTth4LJEF+ATNQe5AA7jmyjFDiHm8/kGuwYuogcOu7vDnL4ZGDjFCmDLQhAehKvqUgV\nep0O/Xpkol+PTPk+n09EbaMLNQ0uuEUBJ07VoMHhhsPlhcPphdsbejGqyRCYG01GPSxN86PZ6L8v\nzWKUM83+1o2cG6ljYIAdQktrsAMZbG2JiOI1FP2ZdTpBbp8EQP5qitqe2ahH1xwbuuYk7+IeorYk\n70Dbkgx2hC4igLpExCtGzmBLwXSjI9DHubqp1VyGzaRKZFDi6HQCstLNyM20+L+d6ZHBrCpRBIzi\nNERRVJVqxBVgR9GmTzkx6QRBVU7CXRyJKFlIO9DGMxdG6oMNhF7kqP3WMDjA9gfTDXaPXHMv3dfJ\nxuw1ESUHBtga2oU88WwEKPVtDc5gh67B1ukEmBXHsv6aiJJFvBlsqZcwEGmRY3ANdnNdRGqadrv1\nNbWwBIDq+kAGm4goGTDA1vBo6v1izdr4fKJcX63NYAtharB1gjprzfprIkoW0jdvsXYRkYJfQL1b\nrZKcwVZ1EYlcg12t6BgiPU8KsDPTGGATUXJggK2h7asZa9bGpdh1LGiRo6I00KvMYGtKRBhgE1Gy\nkALsWJMN9igWbUv3K7PdkbqIeLz+3UglDU112DV1zGATUXJhgK2hzWBrdxVrjlR/DQTXUofbyVEQ\nBGawiSgpxVsiospghyl7U65Lkc4faav02gaX6rGGpgy2tPCxUxprsIkoOTDA1tCuik5kBluIsgbb\nzBpsIkoScoAd4xpHaZMZALCG2SpdrwvMe1JgHVQioshgK8tDgEAnEakPNjPYRJQsGGBrBNVgx/h8\n5YdB0CJH1mATUYqRdrGLNdkgbZMu9YkPRTknyrtwRuiDLXULkTQ4PPCJoiKDzQCbiJIDA2yNoBKR\nWDPYihKRoDZ9YfpgCzpNmz4G2ESUJIQ4A2wpg22OsHmIIoEdyGBHqMGuCSoRcaPR4ZGf24kZbCJK\nEgywNYIWOcZag60oEYmUwdb2wWYGm4iSkT7ORY7NbZMOqNel+MIE2B5ViYg6g93o8KjqstkHm4iS\nBQNsjaASkRhrRCJnsMPUYAuCKhhnH2wiShZSXiDmALuZbdIBzSJHUf2nJHIGWx1gZ7BEhIiSBANs\njaCNZuLMYBv0QtCWveE3mtHUYHMnRyJKEi3tIhIpYaBXfqvXlM0I6iKiqsEOXuRY2+hqeh09d8El\noqTBAFvDG5TBjq8G22gInuhV9YYsESGiFKCLd5GjK94MdqQuIppFjna3nMFmBxEiSiYMsDUSVYNt\nMgQPrapNn0/Tps/ERY5ElHxaupNjxAA7ihrsUCUi2RlmAOoSEfbAJqJkwgBbo6VbpUsZbO0CRyDy\nIkf1To6swSai5BDYyTG250WzyFFdIhI5wPaJohxMd++cBkBa5Ojvhc1t0okomTDA1vBoJncxxkWO\n0oeBdoEjoK3BVu7kyD7YRJScpHkr1l1t7VEsclR+qyeG22imKelRb3fLQXj3XH+A3aCowWaJCBEl\nEwbYGi3OYEslIiEy2EDgw0oqEREEbpVORMlLSjLHXyKSmAy2coFjj7xABpubzBBRMmKArdHirdIj\nLHIEAgsdpcWUUsCtrLu2RPhKlYioLcW7k6O00Uy4bdIB7SJH//m1gbxLCrAbAgscu+bYAAAigGM/\n1wEAcjtZYro+IqLWxEhOo20y2KJcIiJ9wHTLtWFoYS4Meh3yMvlBQUTJIe6dHJ3NZ7BDLnLUzLme\npjnV6QosIM9QbCgjBeB9u2XEdH1ERK2JAbZGcIAd2/PlRY5hMtiCZkW+9AGjEwT8ftaw2F6MiKiV\n6aU+2DHv5Nh8DXYsJSLSn0aDDmlWdccQg16HnvnpMV0fEVFrYomIRtBGMzFG2O5oa7ClEhH+DRBR\nEou3i4g9ihpsZYmItIgyaCfHprlS+tNg0MGmKaMr6NEJBj0nUyJKHpyRNFrcB1vOYIcLsP1/SrXe\nyq9IiYiSTWAnx+hbKnl9PjnjbIlYg618TuQMtkfKYOt1MOh1qnUr/XpmRX1tRERtgQG2hrZERIx1\nkWOENn1AIGMjvY7AAJuIklhgJ8fonyN1EAEAa6w12Jo51+MV4RNFVYkIAKRZAuc9t1d29BdHRNQG\nGGBrBG2VHnMNtv+DxRimRETQlogwviaiJBbPTo5SD2yg+T7YUowdrosI4M9eSyUiRr0UYAfqsM/t\nxQw2ESWXqALsnTt3YtasWRg5ciQuuugivPnmmwCAmpoa3H777Rg5ciQmT56MtWvXys8RRRFLly7F\nuHHjMHr0aCxZsgRerzfcSyQNbYlIrJsrNJvBbvowkTa0ERhhE1ESi2ejGWUGu7m+/npNAB/qddze\nQMmJQZPBNhm5wJGIkk+zXURqampw22234aGHHsKVV16J7777DnPnzkXv3r3x5ptvwmazYdu2bTh0\n6BDmz5+Pc889F8XFxXj99dfx6aefYt26dRAEAQsWLMBLL72E+fPnt8XvFbdWb9MnfZho+mATESUj\nqU46lrlQVSLSTF9/nU4AvKJcgiIF2nqdIP/sDpHBtjVlsM/pmgE9FzgSUZJpdlY6efIkJk2ahGnT\npkGn02HQoEEYO3Ysdu3ahY0bN+LOO++E2WzG0KFDcdVVV+G9994DALz//vuYM2cO8vPzkZeXhwUL\nFuDdd99t9V+opYIWOcbaRaSZNn2BLiJc5EhEyU+ao2IpEXEoSkTMzWSwdZo2gNKca1Lsbuv2+ODx\n+O+XarAH9vHXXY8Z2CXq6yIiaivNZrAHDhyIv/71r/Ltmpoa7Ny5E0VFRTAYDOjVq5f8WN++fbFh\nwwYAQElJCfr166d6rLS0FKIoRrWwTxCEdmlhp/0QEQHo9dEHwVKJiMWkD/m8QD2jT74dy/k7Emks\ndCyTaRMc77bTkcZaan8nitHPhdI3eRaTXg6Iw5FKRESI0OsFSDOwxaSXa7l9ogiPL7DIUa8XcMmY\nXrhgWDe5J3ZHGOtU0JHe28mOY912WmOsY9popq6uDgsXLpSz2GvWrFE9brFY4HA4AAB2ux0WS2BH\nQqvVCp/PB5fLBbPZ3Oxr5eamtUuHDb3mw8BsMSInJ/r6PulrzOwsW8jnSR82oijIt2M5f0eUlZXW\n3pdwVuF4t52OMNbWpgBWH8NcpTNUAABsFkOzz5HKO2w2M3Jy0mFqKimxWQyoqvNvj261maGTFjfa\nTCHP2RHGOpVwvNsOx7rtJHKsow6wjx8/joULF6JXr1545plncOTIETidTtUxDocDNpsNgD/YVj5u\nt9thMBiiCq4BoKKioV0y2I12t/p2gwuVlfVRP9/VVHvodrpDPk9awON0e+TbsZy/I9HpBGRlpaG6\nuiHmUhyKHce77XSksXY3zVVOpyfquaqiqgGAv1SuuedIaZTaWjsqK+vR2OgCANXGMRWVDWhoul/0\n+VTn7EhjnQo43m2HY912oh3rWBKiUQXY+/fvx80334zp06fjD3/4A3Q6Hfr06QO3242TJ0+ie/fu\nAIDS0lK5LKSwsBClpaUYNmyY/FhBQUHUFyaKItqj6Yi0mYF82+eTN4Vpjs8nyhlsvV4I+TwhRA12\ntOfvqHw+8awfg7bE8W47HWGsBQRqsKP9XRodgW3Sm3tOYG8A//mlheZmRQ22w+WVN/HS63Qhz9kR\nxjqVcLzbDse67SRyrJvNEZeXl+Pmm2/G3Llz8cADD0DXlFZOT0/H1KlTsXTpUtjtduzduxfr16/H\ntGnTAADTp0/H6tWrUVZWhvLycrz44ouYMWNGQi66NQVvNBP9c92K4Ly5RY5ebjRDRCkgsJNjDH2w\nXYEa7FjPL3UTMRl0cnbb7fEFbTRDRJTMms1gv/3226isrMQLL7yAF154Qb7/f/7nf7B48WI88sgj\nmDRpEmw2GxYtWiRnrGfPno3y8nLMnDkTbrcb06ZNw9y5c1vvN0mQlmyV7vQEUu5h2/RpM9j8rCCi\nJBbYyTH2Nn2WCLs4SrR9sKXX0ekEGI06uNw+uD1eOfnBAJuIUkGzs9/ChQuxcOHCsI9lVrRRAAAg\nAElEQVQvW7Ys5P16vR5333037r777vivrh14NfsBx/KhIrXoAwBj2K3S1a/DNn1ElMzkOSumPtj+\nEhGrufkMtrTZltymTwyUz5kMerjcPrgUGWwDe14TUQrgTKXRko1mXIoMtjlMliU4g80Am4iSl7yT\nY0x9sGPPYEvJDOVGM9I3gS63lyUiRJRSOFNpBJeIRP9clzKDbQydudFujc4MNhElM50mwxwNKYNt\niSKDHVSD3fQ6gk6Q17K43L5AiQgz2ESUAjhTaQRlsGMpEVEtcoycwQ7cjuHiiIjaWDw7OdpjyGBr\nt2L3hcpge5jBJqLUwplKQ/shkvhFjprbjLCJKIkFSjiif46cwY6ii0jQIke5BjvQqs/p9sktUBlg\nE1Eq4EylEdymL/ZFjnqdAH2Y9iDagJpt+ogomUlzVGwlIv5kgzWaDHZQm77A+hSTUSoRUWSwWSJC\nRCmAM5WGtsF4TDXYTRnscNlrIESJCDPYRJTEtIsQo2GPIYOtrfGWA2xBkEvtXG6vnME2MINNRCmA\nM5VGUAY7li4iTRnscJvMAMEZayawiSiZxbrI0ecT5bkwmkWO2hIRZRcRZYmIhxlsIkohnKk0pC4i\n+jhWzksZ7Eg1gkE12IywiSiJSXNWtBlsqTwEiK5ERJDbAPpvS1OuoFjk6HB55MCbNdhElAo4U2lI\nGWxpYo/la1E5gx2mRR8QXBLCAJuIklmsGWxpgSMQ3yJHOYMtBNr0NToC5zToOWcSUfJjgK0h1WBL\nE3tcNdgRM9iswSai1BFrmz67MoNtjqZNX5idHHUCzE0BeoPDLR8fbpdcIqJkwgBbw6NpBRVPH+xI\nAXbwRjOxXiERUduRAuBoOyo5nLFlsCN2EWmaSxsUGWyWiBBRKuBMpeDziZA+QuQSkXgWOUYqEWEf\nbCJKIYEMc3TH19v92WadIMASRQY7XImITgi06WuwBzLYLBEholTAAFtB2UHEKJeIJHiRI2uwiSiF\nxFoiIgXY6VZDVPObEKFERO6D7VHOzfzYIqLkx5lKwaPogS19NRnLRjNSBtscMYPNjWaIKHXE2lGp\nrrEpwLaZYju/pkRErygRUWINNhGlAgbYCh7FXsBS5iSeRY6R2/RpFznGcIFERG1M0NRIN0fOYFua\nLw8BItRgC6GTFUaWiBBRCmB4p6DcxbFFixwjtunT3GYGm4iSWKw7OcoBdpQZbGlODNrJUVEiosQS\nESJKBZypFNQ12PEscmSbPiLqWLQBcHMCNdjGqI7XLnJUtekzBs+lBu7kSEQpgDOVgjLANsUTYEeR\nwQ5u08cAm4iSlzRHiSIgRjEf1je6AAAZtugCbG2JiGqjGc1catALXLdCRCmBAbaCV7XIsakG2xfu\n6GBxZbD5YUFESUz5LVs0CYe6GDPYOu0iR+VW6Zq5lOUhRJQqOFspKBc5GuPpg+1Rb1ITSlAXEf4N\nEFESU85Z0dRhx1oiIgXYXrkG2z+P6kPUYBtZHkJEKSK6Zd5niax0MwQB6JRmQrrF/+EQW5u+pgw2\nFzkSUQehymA3842eTxRjD7ClEhS5i0jgfm0XEQMz2ESUIhhgK2Slm7Fk/lh0ze+Ej7eVAoitTV80\nW6VzkSMRpZJYSkTsTg+kQ9KjrMGOtMjRpFnkyAw2EaUKBtgaPfPSkZNtk7c0j2ZRjySaRY7cyZGI\nUolyjmpuN8f6xsCW5hmx1mA3ndqn3Cpds6kMa7CJKFVwtgpDF+PuZT5RjCqDrV0Bz41miCiZ6WPI\nYEsLHAEg3RplH+xwXUR0AnQ6QdWWjy36iChVcLYKQzvpN0cKroFmMtiahLUAZrCJKHkpcwLKNSk1\n9c6gb/ikDLZeJ8Bqjm5Lc22JiHROaQG4shc2M9hElCo4W4UhB9hRZrClBY5ArFulM8AmouSlzGBL\nQfCO737G3cu34p+bflAdW2f398BOsxqj7lctaL4tDGSw/fOoMmHBAJuIUgVnqzDk3cui7IMdbQY7\neKOZmC+NiKjNhFrkePBYNQDgu6NVqmOlDiLR1l8DwVuxy4scm15WOZ+yRISIUgVnqzCEGDPYTkUG\nO3IXEc1tRthElMRC9cGurnMCACpqHKoyEalEJNoWfcrzywG2L9BFBADMBpaIEFHq4WwVRqyLHFUZ\n7EgBNruIEFEKUWew/X9W1/sDbIfLC7vTIz8u98COskWf8vw+eaOZpvub5kaWiBBRKuJsFYZ284Pm\nuNzRLnLU7OTIAJuIklioDHZVU4ANAOU1DvnnWDeZAdSLHEVRlANt6X5lL2z2wSaiVMHZKgxtb9bm\nuDxc5EhEHY96J0cRXp8PtQ0u+b6K2kCAXRdHgC2d3ucTVd8YSq+r7IXNnRyJKFVwtgpDOelHQ9pk\nRq/p2xp0Xi5yJKIUol3kWNvghrJyrkKZwW6MfZGjskREuahcSkaYTYoSEWawiShFcLYKI9YabKlN\nX3M1glzkSESpRDlFeX2iXH8tqawN3G5JDbbXJ6oSGoEMNhc5ElHq4VbpYcTaB9sdxTbpQKg2fQyw\niSh5aXdyVJaHAEB5U4mIzyeiQS4RiW4XR0C93iVkiYiRGWwiSj2crcIItI6K7ngpgx2pg4jyvPJt\nZrCJKIkpF2KLvkCLPolUIlLT4IIUHmemRR9gKxc5ehUZ7JCLHJnBJqIUwdkqDCnw1W4FHI4rygx2\ncBeROC6OiKiNqHdy9KGqXp3BrmzKYJ+uapTvy8+2Rn1+5YJyVQZbqsHmIkciSkGcrcIQYl3kGG0N\ntuZhlogQUTLT9sGWarCtZn+FYU2DC26PFz9X2QEAnWxG+bGozq/YaCZkDTZLRIgoBXG2CkOuC0R0\nWWwpg21miQgRdSDaPthSgF3YvZN8f2WtEz83ZbDzs20xnV8fbpFj08uaWSJCRCmIs1UYysA3mioR\naaMZY3MlIlzkSEQpRDlF+UQR1XX+EpECRYBdXuvA6Up/BjuW8hAgsPBbFKPIYDPAJqIUwdkqDJ3m\nQ6U50kYzXORIRB2JIAiqMg4pg52XZUWnpnZ8lTUOuUSkS4wBtmqRYzNdRCLtMUBElEw4W4WhbKcX\nTR12o8MDALA1U3uo3Rqd8TURJTsp2HW6vXKv66wMM3IzLQD826Wfro6vRESZdPB6FV1EBPbBJqLU\nxdkqDFXdYRQZ7EZnU4BtibzBAhc5ElGqkeatKkWLvqx0M7rlpgEAvv7+jFwm1yUntgy28ls8j9cX\ndD8XORJRKuJsFYZq5XwUvbAbHf6sjs0SWwZbe5uIKNlIiYCK2sC26NnpJgwtzAUAnCxvkO/Pz4pv\nkSMAuEME2GYj2/QRUeqJabbau3cvJk6cKN+uqanB7bffjpEjR2Ly5MlYu3at/Jgoili6dCnGjRuH\n0aNHY8mSJfB6vYm78lYWawa7QSoRaSbADq7BjuPiiIjakBQESxlsk0EHq9mAIQW5MOgDc1qGzdjs\nHKilnBM9Hl/Q/aqNZpjBJqIUEdVsJYoi3n77bcybNw9ut1u+/6GHHoLNZsO2bdvwt7/9DU8++SR2\n794NAHj99dfx6aefYt26dfjwww+xa9cuvPTSS63zW7SCWBc5SjXYac0F2CwRIaIUI33TJs1zVrMB\ngiDAajZgYJ8c+bhYO4gA6m8LQ2WwLaZABttsityliYgoWUQVYK9YsQJr1qzBwoUL5fsaGhqwceNG\n3HnnnTCbzRg6dCiuuuoqvPfeewCA999/H3PmzEF+fj7y8vKwYMECvPvuu63zW7QCVZu+ZhY5iqIY\nWOTYXA22tkSEqxyJKMlJGWyHK3hDreH9O8s/x1oeAqiTDh5P8E6OuZ0s+MXwHpg4tFvMHUqIiNpL\nVN/l/fKXv8TChQuxY8cO+b6jR4/CYDCgV69e8n19+/bFhg0bAAAlJSXo16+f6rHS0lKIohhV3bEg\nCO1SPiEF1nrlV5ECoNeHv2a70ytnuTNsxojHamsIjXoh4vEdmTTWbFXYNjjebaejjbWyiwjgL9uQ\n5q2R/fOw5qNDAIBuubaY5zNlsO4TAxnswGsI+O0VA5q9to4y1smO4912ONZtpzXGOqoAOz8/P+i+\nxsZGWCwW1X0WiwUOh38RjN1uVz1utVrh8/ngcrlgNpubfc3c3LR2XQCYlRnIxHTKtCEnQuup05WN\n8s/du3RCTk56+PNWO1W3MzNtEY8/G2RlpbX3JZxVON5tp6OMtZQYkEo4rBajPG/l5KRj3OCu+OrA\nzxhf3CPm+azOFQiqzRaT/HPnzukwGqIvCekoY50qON5th2PddhI51rGtRlGwWq1wOtXBosPhgM3m\nD0QtFovqcbvdDoPBEFVwDQAVFQ3tlsHOykpDfb1dvq+qqgEGMXwrkZ/K6uSfPU43Kivrwx6rPK//\ntiPi8R2ZNNbV1Q1R9RqnluF4t50ON9ZNv4K9qRROAFTz1oLp52HOpUWwWQwxz2d1tYE5sbpG8XN1\nA/RRfAh0uLFOchzvtsOxbjvRjnUsCYS4A+w+ffrA7Xbj5MmT6N69OwCgtLRULgspLCxEaWkphg0b\nJj9WUFAQ9flFUUSyNB1xe3yqDRC06hsDCz8tJn3EY4PWS4qIePzZwOcTz/oxaEsc77bTUcZa+tZU\nrsHW64J+L7Mx8twXDZc7MOmLPqh2dmxORxnrVMHxbjsc67aTyLGOO0ecnp6OqVOnYunSpbDb7di7\ndy/Wr1+PadOmAQCmT5+O1atXo6ysDOXl5XjxxRcxY8aMhFx0W1C16WvmX45Siz6jQdfsV5rcKp2I\nUo00T0kbwSRyR0W9Yk50N7XpEwTuEUBEqS3uDDYALF68GI888ggmTZoEm82GRYsWyRnr2bNno7y8\nHDNnzoTb7ca0adMwd+7chFx0W1D3wY58rLzJTDPbpGvPG+o2EVGy0SYCEhlgh2rTp2figYhSXEwB\n9tixY7F9+3b5dlZWFpYtWxbyWL1ej7vvvht33313y66wnSjj3uba9EW7yQwQ/EHF+JqIkp02EdBa\nAban6atZJh6IKNVxW6wwVFulN1MH2Oj0Z7DTmumBDag3sNG+DhFRMtLOU6ZEBtjKnRybMticF4ko\n1THADkM56Te3zqYlGWxmaogo2QVlsPWJ21FRVSLSVIPNeZGIUh0D7DBiymBHuU06wEWORJR6tDXR\nRmMCFznqmMEmoo6HAXYYsXURaVrkGEWJiHZrdH6OEFGy085TRj1LRIiIImGAHYZyf4PEZrC1t/lB\nQkTJrc26iHjYRYSIOgYG2GHEksFulGuwo1nkyBIRIkotrbrIUXEqt9xFJGGnJyJqFwyww1DXYEc+\nVuqDHVUGO6hNHz9JiCi5tWqbPmWJiIclIkTUMTDADkO90Uykrc/FQBeRKDaa0QbU/BwhomSnDXgN\nCQywBUGQ51u5BpuJByJKcQyww4h2oxmX2wdv0+NRtenTfG5oFz0SESUbbcBrMiSuTR8QKBPhIkci\n6igYYIchCIIcZEfKYEsdRIAoN5phH2wiSjGtuchReX43A2wi6iAYYEcgBb8+X/hjpAWOQLQZbAbY\nRJRaWnORIxCYBz0efzJDz3mRiFIcA+wIpA+VhGawg7qIxHlxRERtJKgPdoIDbKktH0tEiKijYHgX\nQSCDHT7AljLYep0AUxS7m2kDamawiSjZaftSJ3KRI6AoEWEXESLqIBhgRxBNDXajM7DJTDQt94K6\niPCDhIiSXKsvchRYg01EHQsD7AjkDHaEALuyzgkAyLCZojsnFzkSUYrRdjtqrUWObNNHRB0FA+wI\npElfjLDI8WhZHQCgV5f06M6p+eDg5wgRJTttiYhR30qLHL3cKp2IOgYG2BHooigROVpWCwDo0yUj\nhvP6TywI3MmRiJJf0E6OUaw3iYVersHmVulE1DEwwI5AaKaLSF2jCxW1/hKRc7rGEGA3jTq/BiWi\n/7+9e4+NsvrzOP7pQC/Ti5TrqvsDhCKXRaGltoAtlgWC4lKIWkQRYiBq8QIaAwliUAgaMf4g0TTQ\nIP4j1UCKURCQeGORsIIgN7ltVBqWBBArtj96mV7P/lE6zHSmw9A+M+WZvl8JoXOeM2ceTk7OfPn2\nnPPYgc852FZnsH1OEeGrCYC9MYsF0BwAt5bAbl4eIkn92pDBZiMPADvw2eRocQbbd2+Kpc0DQNjd\n+MkonVhrx/QdPHNZFdV1qqhuOgP733rEyxkbfFc2Z8bJYAOwA8+EsiMqSl0szjD7Ph+AuRGAvRFg\nB9D8HeK5ROTiX5Va98UJSVKis+nBMjezPETyzGBbcJMAEGKeAa/VJ4hIvpsau1i8BAUAwo1ZLAB/\nGez/PnLB/XNzBvtmNjg2tevdPgDcyjznqlAE2C2TDfE38RtBALgVEWAHcP1R6U2va+sa9D8nLvrU\n63+zGWxH8ykiBNgAbn1dQpzBbrkkJMFJgA3A3giwA2iZwT545rIqXfVyREVpYvo/JDV98fQP8gzs\nlu2yzhCAHYQ8g90i2ZAYF235ZwBAOJEmCCDKfYpIU4C991jT8pCRg3pq5oRBSoqPVp9kp+Jv8svA\n4d7kaOHNAkCIeD7JMSYMa7ATnATYAOyNADuAlpsc/+9yhSTpvqF91LWLQ9OyBrSp3eZkDUtEANhB\nuJeIsAYbgN2xRCQAzyUidfUNctU2SJK6J8Za0i6bHAHYgfcSkS4hbV8igw3A/giwA/Dc5Hi1qs5d\nnhTfvsmfY/oA2Emoj+nz2eQYRwYbgL0R4gXQnFRpbDT6V1WtuzwpIaZ97fKgGQA24hn/Wv2Y9Kb2\nW2xyJIMNwOYIsANwLxExRv+qbMpgR6n9O9zd52CzyxGADXiuwbb6Mekt25ekBE4RAWBzBNgBeAbY\nV69lsBPjo9sdGDvIYAOwEc9TREKSwfZov4sjKiRBPACEE7NYAM2Tvmm8vgY7Kb59y0Ok64E1p4gA\nsIPQP8nxevsJzmjmRgC2R4AdQPOc75nBvq2dGxyb2mWTIwD78D6mL7SniLDBEUAkYCYLIMpxfYlI\npZUZ7GuBNUtEANhB6DPY13/miD4AkYAcagDNXyreS0SszGATYAO49YX6mD7PDHkCD5kBEAEIsAPw\nt8nxNgsy2BzTB8BOHCF+VLrDI4VNBhtAJCDADsDh8AywQ5DBJr4GYAOec1XXUATYHu1zRB+ASECA\nHYB7k6PHg2asOUXk2t9E2ABsIPQZbM9TRFgiAsD+CLADaJ70XbUNqq1rlGRRBtvBMX0A7CPkmxy9\nThEhgw3A/giwA2ie9Msratxlt7XzMeme7ZLBBmAHoT6mz2uTI8f0AYgABNgBNGeYyytr3WXWHNPH\nJkcA9hEV4lNE5LkGm02OACIAAXYAzRvby64F2I6oKMVbkF1hkyMAOwn1EpGa2gb3zywRARAJCLAD\naP5SaZ78k+KjLck6NzfBGmwAduC1RKSL9V8b1TUeATabHAFEgJAG2KdOnVJeXp5SU1M1ffp0HT16\nNJQfZ7mWAbAVGxwl1mADsBfPxEJMtPVfG67aevfPZLABRIKQBdg1NTWaP3++Hn30UR08eFBz5szR\n888/r8rKylB9pOVaBsBWrL+WPB80Y0lzABBSjpBnsK8H2PE8yRFABAhZgL1//345HA7NmjVL0dHR\nysvLU69evbRnz55QfaTlWgbA1mWwr/1NhA3ABjwetKjoaOtPEan2WIPNvAggEoQsVVBSUqKUlBSv\nsgEDBujs2bNBvT8qKsprUg8X9wkfjijFtPgiSU6MVZcu7Z/8m9vt2tVhSXt25dnXCD36O3wira89\n50JnbBfL563uSbE6d+mqJN1025HW17c6+jt86OvwCUVfhyzArqqqktPp9CqLi4uTy+UK6v09eyZ0\n6CbA5OQEPTh2gP73fJmuVtUp0Rmth7MHqkePxHa3/XDWQP1Z5tJ/WdSe3SUnJ3T0LXQq9Hf4REpf\nJycn6P4RdyjRGaOB/Xpa3v6Cx9P0z09+1n+m/6PNc2Kk9LVd0N/hQ1+Hj5V9HbIA2+l0+gTTLpdL\n8fHxQb3/r78qOyyDnZycoLKySvVI6KrlczO8rl+5UtHuz/j3HnFaOmeUZe3ZlWdfNzaajr6diEd/\nh08k9nV+7n9ICs2c1VXSkqfS2tR+JPb1rYz+Dh/6OnyC7eubSQCELMAeOHCgioqKvMpKSko0derU\noN5vjFFDw43rhUpjo1FDAwM6HOjr8KK/w4e+Dh/6Orzo7/Chr8PHyr4OWY547Nixqq2t1caNG1VX\nV6ctW7aotLRU2dnZofpIAAAAoMOFLMCOiYnRhx9+qB07digzM1NFRUVat25d0EtEAAAAADsK6YGj\nQ4cO1aZNm0L5EQAAAMAthUelAwAAABYiwAYAAAAsFGWMYWsqAAAAYBEy2AAAAICFCLABAAAACxFg\nAwAAABYiwAYAAAAsRIANAAAAWIgAGwAAALAQATYAAABgIQJsAAAAwEKdMsA+deqU8vLylJqaqunT\np+vo0aN+623fvl0TJ05Uamqq8vPzVVpaGuY7jQyHDh3SjBkzlJ6erkmTJmnTpk1+6+Xn52vEiBFK\nS0tz/8HN+eijj3TPPfd49eGhQ4d86jG222/btm1e/ZyWlqahQ4dq2bJlPnUZ2213/PhxZWdnu1+X\nl5frxRdfVHp6usaPH6/i4uJW3xvsXI/rWvb3pUuX9MILL2j06NHKysrSypUrVVtb6/e9wc4/aNKy\nr3/55RcNGzbMq/8KCwv9vpexfXM8+/rChQs+c/fw4cP14IMP+n1vm8e16WRcLpcZN26c+eSTT0xt\nba0pLi42Y8aMMRUVFV71Tp8+bUaNGmWOHj1qqqurzdKlS80zzzzTQXdtX2VlZSYjI8Ns27bNNDQ0\nmBMnTpiMjAyzb98+n7rZ2dnm+PHjHXCXkePVV181GzZsCFiHsR0a+/btM1lZWebixYs+1xjbN6+x\nsdEUFxeb9PR0k5mZ6S5fsGCBWbRokXG5XObYsWMmMzPTHDlyxOf9wc71aNJaf8+ePdusWLHCuFwu\nc/nyZTNjxgyzZs0av20EM/+g9b7evHmzee655274fsZ28Frra0+XL182WVlZZs+ePX6vt3Vcd7oM\n9v79++VwODRr1ixFR0crLy9PvXr10p49e7zqffnll5o4caJGjhypuLg4LVq0SHv37iXTd5MuXLig\nnJwc5ebmyuFwaPjw4Ro9erQOHz7sVe+vv/7SlStXNHjw4A6608hw+vRpDRs2LGAdxrb1KisrtWTJ\nEi1fvly333671zXGdtsUFhbq448/1vz5891llZWV+vbbb7Vw4ULFxsZqxIgRmjp1qr744guf9wc7\n16OJv/6ura2V0+nU888/r9jYWPXu3Vu5ubk6cuSI3zaCmX/gv6+lpqz00KFDb/h+xnbwWutrT2++\n+aamTJmiBx54wO/1to7rThdgl5SUKCUlxatswIABOnv2rFfZ2bNnNWjQIPfr7t27q1u3biopKQnL\nfUaKYcOG6b333nO/Li8v16FDh3wmkVOnTikhIUH5+fkaM2aMnnjiiVYncfhXXV2tkpISffzxx8rK\nytKUKVO0ZcsWn3qMbett2LBBgwcP1qRJk3yuMbbb5rHHHtPWrVt17733usvOnTunrl27qm/fvu4y\nf/O3FPxcjyb++jsmJkbr169X79693WW7d+/2GwQGO//Af19LTYHc4cOHNWHCBI0fP17vvvuu3+U4\njO3gtdbXzX788UcdPnxYr7zyit/r7RnXnS7ArqqqktPp9CqLi4uTy+XyKquurlZcXJxXmdPpVHV1\ndcjvMVJdvXpV8+fP1/DhwzVhwgSvazU1NUpNTdXrr7+uH374QdOmTdOzzz6rP//8s4Pu1n5KS0uV\nnp6uJ598Urt379bKlSu1atUqn6wGY9talZWVKioq0ksvveT3OmO7bfr06aOoqCivsqqqKp+x62/+\nbq4bzFyPJv7625MxRm+99ZbOnj2r/Px8n+vBzj9ova+7d++uCRMmaPv27dq4caMOHDigDz74wKce\nYzt4NxrX69ev17x585SQkOD3envGdacLsJ1Op88gdLlcio+P9yprLehuWQ/BOX/+vJ544gl169ZN\nBQUFcji8h96kSZO0fv163X333YqJidGsWbN0xx136MCBAx10x/bTt29fFRUVKScnRzExMbrvvvs0\nffp0fffdd171GNvW+vbbb3XnnXcqNTXV73XGtnWcTqdqamq8yvzN3811g5nrcWMul0svv/yy9u7d\nq40bN6pnz54+dYKdf9C6wsJCzZ07V/Hx8erbt6/y8/P1zTff+NRjbFvj4sWLOnjwoPLy8lqt055x\n3ekC7IEDB/r8KrykpMTrV+aSlJKS4lXvypUrKi8v9/m1DG7s5MmTevzxx5Wdna21a9f6ZKAkadeu\nXdq5c6dXWU1NjWJjY8N1m7Z38uRJrV+/3quspqZGMTExXmWMbWvt3r1bU6ZMafU6Y9s6/fv3V11d\nnS5cuOAu8zd/S8HP9QisrKxMs2fPVllZmTZv3uy1PMdTsPMP/CsvL9e7776riooKd1lr8wRj2xq7\nd+9WZmamevTo0Wqd9ozrThdgjx07VrW1tdq4caPq6uq0ZcsWlZaWeh2VI0lTp07V119/rUOHDqmm\npkZr1qzRAw88oO7du3fQndtTaWmpnnnmGc2dO1evvfaaT+a6WVVVld5++2399ttvqqur04YNG+Ry\nuZSVlRXmO7av+Ph4FRQUaNeuXWpsbNSPP/6oHTt26JFHHvGqx9i21rFjx1rNXkuMbSslJiZq4sSJ\nWr16taqrq3X8+HFt375dubm5PnWDnevROmOMFixYoF69eumjjz5ScnJyq3WDnX/gX1JSkr755hsV\nFBSorq5O586dU2FhoR599FGfuoxta9xo7pbaOa5v+tyRCHD69Gkzc+ZMk5qaaqZPn+4+4mnZsmVm\n2bJl7no7duwwkydPNmlpaebZZ581paWlHXXLtrVu3TozePBgk5qa6vVnzZo1Pv1dWFhocnJyzMiR\nI82TTz5pzpw504F3bk/fffedmTp1qhk5cqSZPHmy+eqrr4wxjO1Qqa+vN0OGDAl/NoQAAAMsSURB\nVDG//fabVzlj2zr79+/3Ol7r77//NgsXLjQZGRkmJyfHFBcXu69t3brVPPzww+7Xrc31aJ1nf//8\n889m8ODB5t577/Wav2fNmmWM8e3v1uYf+NdybP/666/m6aefNqNGjTL333+/ef/9901jY6MxhrHd\nXi372hhjnnrqKfPpp5/61LVqXEcZY4xF/xkAAAAAOr1Ot0QEAAAACCUCbAAAAMBCBNgAAACAhQiw\nAQAAAAsRYAMAAAAWIsAGAAAALNS1o28AABDYkiVL9Pnnn7d6PTMzUz/99JOOHz/OEyIB4BbAOdgA\ncIu7evWqXC6XJOnIkSNasGCBvv/+e/fjep1Op6qrq9W7d++OvE0AwDVksAHgFpeUlKSkpCRJUrdu\n3SRJvXr18spWJyYmdsi9AQB8sQYbAGzuwIEDGjJkiGpqaiRJQ4YM0c6dO5Wbm6sRI0Zo3rx5unjx\nohYvXqzU1FRNnjxZ+/fvd7//jz/+0MKFC5WWlqZx48Zp+fLlqqys7Kh/DgDYHgE2AESg1atX6403\n3lBRUZFOnjypadOmadiwYfrss880aNAgrVixQpJkjNFLL72k6OhoFRcXq6CgQGfOnNHSpUs7+F8A\nAPbFEhEAiEBz5sxRRkaGJGnMmDE6f/685s2bJ0maOXOm5s+fr4aGBv30008qKSnRp59+qujoaEnS\nO++8o4ceekiXLl3S7bff3mH/BgCwKwJsAIhA/fr1c//sdDrVt29f9+u4uDg1Njaqvr5ev//+uyoq\nKpSZmenTRklJCQE2ALQBATYARKCuXb2nd4fD/4rA+vp69evXTx9++KHPNU4lAYC2YQ02AHRiKSkp\nunTpkpKSktS/f3/1799f9fX1WrVqlSoqKjr69gDAlgiwAaATy8rKUkpKil599VWdPHlSJ06c0OLF\ni/X333+rT58+HX17AGBLBNgA0Ik5HA6tXbtWiYmJmj17tubNm6e77rpLBQUFHX1rAGBbPMkRAAAA\nsBAZbAAAAMBCBNgAAACAhQiwAQAAAAsRYAMAAAAWIsAGAAAALESADQAAAFiIABsAAACwEAE2AAAA\nYCECbAAAAMBC/w/QFRrTSh5vGwAAAABJRU5ErkJggg==\n",
"text/plain": [
""
]
},
"metadata": {},
"output_type": "display_data"
}
],
"source": [
"from PIDsim import PID\n",
"\n",
"# reactor temperature setpoint\n",
"Tsp = 390\n",
"\n",
"# set initial conditions and cooling flow\n",
"IC = [C0,T0,Tcf]\n",
"\n",
"# do simulation at fixed time steps dt\n",
"tstart = 0\n",
"tstop = 8\n",
"tstep = 0.05\n",
"\n",
"# configure controller. Creates a PID object.\n",
"reactorPID = PID(Kp=8,Ki=30,Kd=5,MVrange=(0,300),DirectAction=True)\n",
"\n",
"c,T,Tc = IC # reactor initial conditions\n",
"qc = 150 # initial condition of the MV\n",
"for t in np.arange(tstart,tstop,tstep): # simulate from tstart to tstop\n",
" qc = reactorPID.update(t,Tsp,T,qc) # update manipulated variable\n",
" c,T,Tc = odeint(deriv,[c,T,Tc],[t,t+dt])[-1] # start at t, find state at t + dt\n",
"\n",
"reactorPID.manual() # switch to manual model \n",
"T -= 10 # change process variable by -10 deg\n",
"for t in np.arange(t,t+1,tstep): # simulate for 1 minute\n",
" qc = reactorPID.update(t,Tsp,T,qc) # continue to update, SP tracks PV\n",
" c,T,Tc = odeint(deriv,[c,T,Tc],[t,t+dt])[-1] # start at t, find state at t + dt\n",
"\n",
"reactorPID.auto() # switch back to auto mode\n",
"for t in np.arange(t,t+tstop,tstep): # integrate another tstop minutes\n",
" qc = reactorPID.update(t,Tsp,T,qc) # update MV\n",
" c,T,Tc = odeint(deriv,[c,T,Tc],[t,t+dt])[-1] # start at t, find state at t + dt\n",
" \n",
"# plot controller log\n",
"reactorPID.plot() # plot controller log"
]
},
{
"cell_type": "markdown",
"metadata": {},
"source": [
"### Contents of PIDsim.py"
]
},
{
"cell_type": "code",
"execution_count": 9,
"metadata": {
"collapsed": true
},
"outputs": [],
"source": [
"# %load PIDsim.py\n",
"import matplotlib.pyplot as plt\n",
"import numpy as np\n",
"\n",
"class PID:\n",
" \"\"\" An implementation of a PID control class for use in process control simulations.\n",
" \"\"\"\n",
" def __init__(self, name=None, SP=None, Kp=0.2, Ki=0, Kd=0, beta=1, gamma=0, MVrange=(0,100), DirectAction=False):\n",
" self.name = name\n",
" self.SP = SP\n",
" self.Kp = Kp\n",
" self.Ki = Ki\n",
" self.Kd = Kd\n",
" self.beta = beta\n",
" self.gamma = gamma\n",
" self.MVrange = MVrange\n",
" self.DirectAction = DirectAction\n",
" self._mode = 'inAuto'\n",
" self._log = []\n",
" self._errorP0 = 0\n",
" self._errorD0 = 0\n",
" self._errorD1 = 0\n",
" self._lastT = 0\n",
" self._currT = 0\n",
" \n",
" def auto(self):\n",
" \"\"\"Change to automatic control mode. In automatic control mode the .update()\n",
" method computes new values for the manipulated variable using a velocity algorithm.\n",
" \"\"\"\n",
" self._mode = 'inAuto'\n",
" \n",
" def manual(self):\n",
" \"\"\"Change to manual control mode. In manual mode the setpoint tracks the process \n",
" variable to provide bumpless transfer on return to automatic model.\n",
" \"\"\"\n",
" self._mode = 'inManual'\n",
" \n",
" def _logger(self,t,SP,PV,MV):\n",
" \"\"\"The PID simulator logs values of time (t), setpoint (SP), process variable (PV),\n",
" and manipulated variable (MV) that can be plotted with the .plot() method.\n",
" \"\"\"\n",
" self._log.append([t,SP,PV,MV])\n",
" \n",
" def plot(self):\n",
" \"\"\"Create historical plot of SP,PV, and MV using the controller's internal log file.\n",
" \"\"\"\n",
" dlog = np.asarray(self._log).T\n",
" t,SP,PV,MV = dlog\n",
" plt.subplot(2,1,1)\n",
" plt.plot(t,PV,t,SP)\n",
" plt.title('Process Variable')\n",
" plt.xlabel('Time')\n",
" plt.legend(['PV','SP'])\n",
" plt.subplot(2,1,2)\n",
" plt.plot(t,MV)\n",
" plt.title('Manipulated Variable')\n",
" plt.xlabel('Time')\n",
" plt.tight_layout()\n",
" \n",
" @property\n",
" def beta(self):\n",
" \"\"\"beta is the setpoint weighting for proportional control where the proportional error\n",
" is given by error_proportional = beta*SP - PV. The default value is one.\n",
" \"\"\"\n",
" return self._beta\n",
" \n",
" @beta.setter\n",
" def beta(self,beta):\n",
" self._beta = max(0.0,min(1.0,beta))\n",
" \n",
" @property\n",
" def DirectAction(self):\n",
" \"\"\"DirectAction is a logical variable setting the direction of the control. A True\n",
" value means the controller output MV should increase for PV > SP. If False the controller\n",
" is reverse acting, and ouput MV will increase for SP > PV. IFf the steady state\n",
" process gain is positive then a control will be reverse acting. \n",
" \n",
" The default value is False.\n",
" \"\"\"\n",
" return self._DirectAction\n",
" \n",
" @DirectAction.setter\n",
" def DirectAction(self,DirectAction):\n",
" if DirectAction:\n",
" self._DirectAction = True\n",
" self._action = +1.0\n",
" else:\n",
" self._DirectAction = False\n",
" self._action = -1.0\n",
" \n",
" @property\n",
" def gamma(self):\n",
" \"\"\"gamma is the setpoint weighting for derivative control where the derivative error\n",
" is given by gamma*SP - PV. The default value is zero. \n",
" \"\"\"\n",
" return self._gamma\n",
" \n",
" @gamma.setter\n",
" def gamma(self,gamma):\n",
" self._gamma = max(0.0,min(1.0,gamma))\n",
" \n",
" @property\n",
" def Kp(self):\n",
" \"\"\"Kp is the proportional control gain.\n",
" \"\"\"\n",
" return self._Kp\n",
" \n",
" @Kp.setter\n",
" def Kp(self,Kp):\n",
" self._Kp = Kp\n",
" \n",
" @property\n",
" def Ki(self):\n",
" \"\"\"Ki is the integral control gain.\n",
" \"\"\"\n",
" return self._Ki\n",
" \n",
" @Ki.setter\n",
" def Ki(self,Ki):\n",
" self._Ki = Ki\n",
" \n",
" @property\n",
" def Kd(self):\n",
" \"\"\"Kd is the derivative control gain.\n",
" \"\"\"\n",
" return self._Kd\n",
" \n",
" @Kd.setter\n",
" def Kd(self,Kd):\n",
" self._Kd = Kd\n",
" \n",
" @property\n",
" def MV(self):\n",
" \"\"\"MV is the manipulated (or PID outpout) variable. It is automatically\n",
" restricted to the limits given in MVrange.\n",
" \"\"\"\n",
" return self._MV\n",
" \n",
" @MV.setter\n",
" def MV(self,MV):\n",
" self._MV = max(self._MVmin,min(self._MVmax,MV))\n",
" \n",
" @property\n",
" def MVrange(self):\n",
" \"\"\"range is a tuple specifying the minimum and maximum controller output.\n",
" Default value is (0,100).\n",
" \"\"\"\n",
" return (self._MVmin,self._MVmax)\n",
" \n",
" @MVrange.setter\n",
" def MVrange(self,MVrange):\n",
" self._MVmin = MVrange[0]\n",
" self._MVmax = MVrange[1]\n",
"\n",
" @property\n",
" def SP(self):\n",
" \"\"\"SP is the setpoint for the measured process variable.\n",
" \"\"\"\n",
" return self._SP\n",
" \n",
" @SP.setter\n",
" def SP(self,SP):\n",
" self._SP = SP\n",
" \n",
" @property\n",
" def PV(self):\n",
" \"\"\"PV is the measured process (or control) variable.\n",
" \"\"\"\n",
" return self._PV\n",
" \n",
" @PV.setter\n",
" def PV(self,PV):\n",
" self._PV = PV\n",
"\n",
" def update(self,t,SP,PV,MV):\n",
" self.SP = SP\n",
" self.PV = PV\n",
" self.MV = MV \n",
" if t > self._lastT:\n",
" dt = t - self._lastT\n",
" self._lastT = t\n",
" if self._mode=='inManual':\n",
" self.SP = PV\n",
" self._errorP1 = self._errorP0\n",
" self._errorP0 = self.beta*self.SP - self.PV\n",
" self._errorI0 = self.SP - self.PV \n",
" self._errorD2 = self._errorD1\n",
" self._errorD1 = self._errorD0\n",
" self._errorD0 = self.gamma*self.SP - self.PV\n",
" if self._mode=='inAuto':\n",
" self._deltaMV = self.Kp*(self._errorP0 - self._errorP1) \\\n",
" + self.Ki*dt*self._errorI0 \\\n",
" + self.Kd*(self._errorD0 - 2*self._errorD1 + self._errorD2)/dt\n",
" self.MV -= self._action*self._deltaMV\n",
" self._logger(t,self.SP,self.PV,self.MV)\n",
" return self.MV \n"
]
},
{
"cell_type": "code",
"execution_count": null,
"metadata": {
"collapsed": true
},
"outputs": [],
"source": []
},
{
"cell_type": "markdown",
"metadata": {},
"source": [
"\n",
"< [PID Control - Laboratory](http://nbviewer.jupyter.org/github/jckantor/CBE30338/blob/master/notebooks/04.10-PID-Control.ipynb) | [Contents](toc.ipynb) | [Interactive PID Control Tuning with Ziegler-Nichols](http://nbviewer.jupyter.org/github/jckantor/CBE30338/blob/master/notebooks/04.12-Interactive-PID-Control-Tuning-with-Ziegler--Nichols.ipynb) >
"
]
},
{
"cell_type": "markdown",
"metadata": {},
"source": [
"# Implementing PID Control in Nonlinear Simulations\n",
"\n",
"A task common to many control projects is to simulate the PID control of a nonlinear process. This notebook demonstrates the simulation of PID for an exothermic stirred tank reactor where the objective is to control the reactor temperature through manipulation of cooling water through the reactor cooling jacket.\n",
"\n",
"\n",
"\n",
"(Diagram By [Daniele Pugliesi](http://commons.wikimedia.org/wiki/User:Daniele_Pugliesi) - Own work, [CC BY-SA 3.0](http://creativecommons.org/licenses/by-sa/3.0), [Link](https://commons.wikimedia.org/w/index.php?curid=6915706))"
]
},
{
"cell_type": "markdown",
"metadata": {},
"source": [
"## Model\n",
"\n",
"The model consists of nonlinear mole and energy balances on the contents of the well-mixed reactor.\n",
"\n",
"\\begin{align*}\n",
"V\\frac{dc}{dt} & = q(c_f - c )-Vk(T)c \\\\\n",
"\\rho C_p V\\frac{dT}{dt} & = wC_p(T_f - T) + (-\\Delta H_R)Vk(T)c + UA(T_c-T)\n",
"\\end{align*}\n",
"\n",
"where $c$ is the reactant concentration, $T$ is the reactor temperature, and $T_c$ is the cooling jacket temperature. The model is adapted from example 2.5 from Seborg, Edgar, Mellichamp and Doyle (SEMD), parameters defined and given in the table below.\n",
"\n",
"The temperature in the cooling jacket is manipulated by the cooling jacket flow, $q_c$, and governed by the energy balance\n",
"\n",
"\\begin{align*}\n",
"\\rho C_p V_c\\frac{dT_c}{dt} & = \\rho C_p q_c(T_{cf}-T_c) + UA(T - T_c)\n",
"\\end{align*}\n",
"\n",
"Normalizing the equations to isolate the time rates of change of $c$, $T$, and $T_c$ give\n",
"\n",
"\\begin{align*}\n",
"\\frac{dc}{dt} & = \\frac{q}{V}(c_f - c)- k(T)c\\\\\n",
"\\frac{dT}{dt} & = \\frac{q}{V}(T_i - T) + \\frac{-\\Delta H_R}{\\rho C_p}k(T)c + \\frac{UA}{\\rho C_pV}(T_c - T)\\\\\n",
"\\frac{dT_c}{dt} & = \\frac{q_c}{V_c}(T_{cf}-T_c) + \\frac{UA}{\\rho C_pV_c}(T - T_c)\n",
"\\end{align*}\n",
"\n",
"These are the equations that will be integrated below.\n",
"\n",
"| Quantity | Symbol | Value | Units | Comments |\n",
"| :------- | :----: | :---: | :---- | |\n",
"| Activation Energy | $E_a$ | 72,750 | J/gmol | |\n",
"| Arrehnius pre-exponential | $k_0$ | 7.2 x 1010 | 1/min | |\n",
"| Gas Constant | $R$ | 8.314 | J/gmol/K | |\n",
"| Reactor Volume | $V$ | 100 | liters | |\n",
"| Density | $\\rho$ | 1000 | g/liter | |\n",
"| Heat Capacity | $C_p$ | 0.239 | J/g/K | |\n",
"| Enthalpy of Reaction | $\\Delta H_r$ | -50,000 | J/gmol | |\n",
"| Heat Transfer Coefficient | $UA$ | 50,000 | J/min/K | |\n",
"| Feed flowrate | $q$ | 100 | liters/min | |\n",
"| Feed concentration | $c_{A,f}$ | 1.0 | gmol/liter | |\n",
"| Feed temperature | $T_f$ | 350 | K | |\n",
"| Initial concentration | $c_{A,0}$ | 0.5 | gmol/liter | |\n",
"| Initial temperature | $T_0$ | 350 | K | |\n",
"| Coolant feed temperature | $T_{cf}$ | 300 | K | |\n",
"| Nominal coolant flowrate | $q_c$ | 50 | L/min | primary manipulated variable |\n",
"| Cooling jacket volume | $V_c$ | 20 | liters | |"
]
},
{
"cell_type": "code",
"execution_count": 1,
"metadata": {
"collapsed": true
},
"outputs": [],
"source": [
"%matplotlib inline\n",
"import matplotlib.pyplot as plt\n",
"import numpy as np\n",
"from scipy.integrate import odeint\n",
"import seaborn as sns\n",
"sns.set_context('talk')\n",
"\n",
"Ea = 72750 # activation energy J/gmol\n",
"R = 8.314 # gas constant J/gmol/K\n",
"k0 = 7.2e10 # Arrhenius rate constant 1/min\n",
"V = 100.0 # Volume [L]\n",
"rho = 1000.0 # Density [g/L]\n",
"Cp = 0.239 # Heat capacity [J/g/K]\n",
"dHr = -5.0e4 # Enthalpy of reaction [J/mol]\n",
"UA = 5.0e4 # Heat transfer [J/min/K]\n",
"q = 100.0 # Flowrate [L/min]\n",
"Cf = 1.0 # Inlet feed concentration [mol/L]\n",
"Tf = 300.0 # Inlet feed temperature [K]\n",
"C0 = 0.5 # Initial concentration [mol/L]\n",
"T0 = 350.0; # Initial temperature [K]\n",
"Tcf = 300.0 # Coolant feed temperature [K]\n",
"qc = 50.0 # Nominal coolant flowrate [L/min]\n",
"Vc = 20.0 # Cooling jacket volume\n",
"\n",
"# Arrhenius rate expression\n",
"def k(T):\n",
" return k0*np.exp(-Ea/R/T)\n",
"\n",
"def deriv(X,t):\n",
" C,T,Tc = X\n",
" dC = (q/V)*(Cf - C) - k(T)*C\n",
" dT = (q/V)*(Tf - T) + (-dHr/rho/Cp)*k(T)*C + (UA/V/rho/Cp)*(Tc - T)\n",
" dTc = (qc/Vc)*(Tcf - Tc) + (UA/Vc/rho/Cp)*(T - Tc)\n",
" return [dC,dT,dTc]"
]
},
{
"cell_type": "markdown",
"metadata": {},
"source": [
"## Simulation 1. Same Initial Condition, different values of $q_c$\n",
"\n",
"The given reaction is highly exothermic. If operated without cooling, the reactor will reach an operating temperature of 500K which leads to significant pressurization, a potentially hazardous condition, and possible product degradation. \n",
"\n",
"The purpose of this first simulation is to determine the cooling water flowrate necessary to maintain the reactor temperature at an acceptable value. This simulation shows the effect of the cooling water flowrate, $q_c$, on the steady state concentration and temperature of the reactor. We use the scipy.integrate function `odeint` to create a solution of the differential equations for entire time period."
]
},
{
"cell_type": "code",
"execution_count": 2,
"metadata": {
"collapsed": true
},
"outputs": [],
"source": [
"# visualization\n",
"def plotReactor(t,X):\n",
" plt.subplot(1,2,1)\n",
" plt.plot(t,X[:,0])\n",
" plt.xlabel('Time [min]')\n",
" plt.ylabel('gmol/liter')\n",
" plt.title('Reactor Concentration')\n",
" plt.ylim(0,1)\n",
"\n",
" plt.subplot(1,2,2)\n",
" plt.plot(t,X[:,1])\n",
" plt.xlabel('Time [min]')\n",
" plt.ylabel('Kelvin');\n",
" plt.title('Reactor Temperature')\n",
" plt.ylim(300,520)"
]
},
{
"cell_type": "markdown",
"metadata": {},
"source": [
"Once the visualization code has been established, the actual simulation is straightforward."
]
},
{
"cell_type": "code",
"execution_count": 3,
"metadata": {},
"outputs": [
{
"data": {
"text/plain": [
""
]
},
"execution_count": 3,
"metadata": {},
"output_type": "execute_result"
},
{
"data": {
"image/png": "iVBORw0KGgoAAAANSUhEUgAAA7gAAAEhCAYAAABVzwz5AAAABHNCSVQICAgIfAhkiAAAAAlwSFlz\nAAALEgAACxIB0t1+/AAAIABJREFUeJzs3Xl8FOX9wPHPzOy9uU+uEM4ESMIlpwJVQFQKAqIVLbWK\nBbFqFUVB26JVi7QieP4qIKiIJxXFiop4gMollygg9xHO3Mkmm73n+f2xyZIlAYKGQOB5v7qdmWee\nmfnOGDL7zTzzPIoQQiBJkiRJkiRJkiRJDZx6rgOQJEmSJEmSJEmSpLogE1xJkiRJkiRJkiTpgiAT\nXEmSJEmSJEmSJOmCIBNcSZIkSZIkSZIk6YIgE1xJkiRJkiRJkiTpgiATXEk6i2Qn5Weurq6ZvPaS\nJElSbcj7Rd2T11Q6lwznOgBJqgt/+MMf+P7778PKjEYjcXFx9O3bl4ceeojo6Oh6i8fr9fLUU08x\ncOBALrvssrNyjF27djF37lxWr15NcXExTZs2ZciQIdx2221YrdazcsyzqaSkhEcffZS//OUvtGrV\n6lft6+2336akpITx48cDwZ+PhIQEZs6cWRehSpIkSb/AxXKvXrt2Lbfccssp64wYMYJp06bV2THP\nJ2vXruX999/n3//+97kORbpIyQRXumBceuml3HvvvaFlt9vNjz/+yEsvvURJSQkvvvhivcWSm5vL\nW2+9xRVXXHFW9v/VV18xYcIEunXrxqRJk4iNjWXLli3Mnj2b1atXM3fuXEwm01k59tmyfft2Pv30\nU+65555fva85c+YwePDg0PKjjz6K0Wj81fuVJEmSfp2L4V6dkZHBu+++G1p+6623WLVqVdi5xcXF\n1ekxzycLFy7k8OHD5zoM6SImE1zpghETE0Pnzp3Dynr16oXT6WTWrFk4nU7sdvs5iq7u5OXlMXny\nZAYPHsxTTz0VKu/duzedO3dm9OjRvPvuu/zhD384h1GeX9q0aXOuQ5AkSZK4OO7VERERYef4xRdf\nYDKZqp23JElnh3wHV7rgRUREAOHvg3z99dcMHz6crKwsBgwYwJtvvhm2jcPh4B//+Af9+vUjMzOT\nPn36MHXqVHw+X6hOYWEhDz74ID179qR79+785S9/IScnh0OHDjFgwAAAxo4dy+TJk4FgU6gXX3yR\nQYMG0bFjR0aMGMHXX38d2t/atWtJT0/nnXfeoXfv3vTp04eysrJq5/PBBx/gcrl48MEHq63r3r07\nd999N40aNQqVHThwgLvvvpuePXvSrVs3JkyYQE5OTmj95MmTmThxIrNnz6Zv37507NiRO+64I6wO\nwPz587nqqqvo1KkTw4YNC4tdCMHs2bPp378/WVlZXHfddaxdu7bauW3cuJHrr7+erKwsrrrqKr74\n4ovQ+srmXIMHD+aFF17g0KFDpKenM3/+fPr160ePHj3Yv38/Xq+XGTNmMHDgQDIzM+nVqxeTJ08O\nXav+/ftz+PBh5syZQ//+/YFgs7gJEyaE4snPz2fy5Mn06dOHzp07M3bsWPbs2RNa/8ILL3DTTTex\naNEiBg4cSFZWFr///e/ZvXt3tWsuSZIk/XoX2r26toQQzJo1K3T/HDlyJOvWrQutX7VqFenp6axf\nv57hw4fTsWNHRo4cye7du/nkk08YOHAgXbt2ZcKECZSXl4dts2rVKgYPHkynTp0YPXo0O3bsCDv2\nhg0bGDVqFFlZWfTr148XX3wRXddD6/v168ezzz7L8OHD6dSpE0uWLAGCT2iHDRtGx44d6dKlC7ff\nfnvoHjpx4kT+97//sXHjRtLT0zl27BgzZ86kX79+Ycf++uuvQ+sBbrrpJp544glGjx5Nx44dmTNn\nDhB8Hev222+nc+fO9OrViyeffBKPx/OLr7d0kRCSdAEYPXq0uPfee4XP5wt9HA6HWL58uejdu7cY\nO3ZsqO6KFStEu3btxMMPPyy++eYb8cILL4j27duLBQsWhOrcfvvtYtCgQeKTTz4Rq1atEs8884xI\nS0sT7777rhBCCJ/PJ4YOHSr69+8vFi9eLL766isxZMgQMWLECOHxeMTnn38u0tLSxKuvvioOHDgg\nhBDinnvuEV26dBGvv/66WLFihZg4caJIT08XX331lRBCiDVr1oi0tDRx1VVXieXLl4tPPvmkxnO9\n9dZbxY033lir63L48GHRo0cPccMNN4hly5aJjz/+WFx55ZVi4MCBorS0VAghxKRJk0TXrl3FjTfe\nKL788kvx0Ucfie7du4t77703tJ85c+aIDh06iJkzZ4qVK1eKRx99VGRkZIiffvpJCCHEM888IzIz\nM8XLL78sVqxYIe6//36RmZkptmzZEnZuV1xxhXjjjTfEypUrxa233iqysrJEUVGRKC0tFQsWLBBp\naWliyZIl4ujRo+LgwYMiLS1NXHbZZWLp0qXigw8+EEIIMWXKFNG7d2+xaNEisWbNGjFnzhyRkZEh\nZsyYIYQQYuvWreKyyy4TDz74oNi6dWvo5+O+++4TQghRWloqBgwYIAYNGiQ+/vhjsWzZMjFy5EjR\no0cPceTIESGEEM8//7zo2rWrGDx4sPjkk0/EsmXLxBVXXCGuv/76Wl13SZIkqbqL6V5d1dNPPy2u\nuOKKGtf961//EllZWWLWrFlixYoVYsKECSIzM1P8/PPPQgghVq5cGbp/Llq0SHz55ZeiT58+on//\n/mLIkCFi2bJlYsGCBSI9PV3MmjUrbJuePXuK2bNniy+//FJcd911omfPnqK4uFgIEbxXZmZmirvu\nukssX75cvPrqq6Jjx45i+vTpodj69u0rMjIyxGuvvSaWLl0qcnJyxOLFi0X79u3FrFmzxJo1a8Si\nRYtEnz59xO9//3shhBAHDhwQY8aMEUOHDhWbNm0SHo9HzJgxQ/Tt2zfsvL/66iuRlpYmjh49KoQQ\nYtSoUaJDhw5ixowZYvny5WL37t3i6NGjokePHuLmm28WX375pXjvvfdEjx49xIQJE057zaWLm0xw\npQvC6NGjRVpaWrVPly5dxOTJk0O/0IUQYuTIkWLMmDFh28+cOVP07NlTeL1e4XK5xK233ipWr14d\nVmfo0KHioYceEkIIsWzZMpGWlha6AQkRvFn0799fZGdnh5KzFStWCCGE+Pnnn0VaWppYvHhx2D5v\nueUWce211wohjt80K2/MJzN48OBa/3L/5z//KXr06BFKZoUQIjs7W3To0EG8+uqrQohggpuZmSlK\nSkpCdZ555hnRuXNnIYQQgUBA9OjRQ/zzn/8M2/fo0aPFq6++KoqKikRmZmboxlrp5ptvFnfeeWfY\nub311luh9Xv37hVpaWnis88+C6uze/duIYQIXcPKxLXSPffcIz766KOwsvHjx4durkIIccUVV4in\nn346LNbKBPe1114TGRkZ4uDBg6H1DodDdO/eXTz55JNCiGCCWzUWIYR45513RFpamigsLBSSJEnS\nmbuY7tVVnSzBLSgoEBkZGeKVV14Jlem6Lm688UZxzz33CCGOJ6tV758zZ84UaWlpYtOmTaGy0aNH\ni7vvvjtsm5deeim0Pi8vT2RlZYl58+YJIYS46667xNChQ0UgEAjVefvtt0VGRkboPte3b9+we6sQ\nQrz00kth91chgn8Eb9euXWj5gQceEKNGjQot1zbBPfEaTZ06VVx66aXC6XSGyr755huRlpYmduzY\nISTpZGQTZemC0adPH/773/+ycOFCpkyZgsVi4brrrmPq1KmhXhnLy8vZsmUL/fr1w+/3hz59+vSh\nqKiIXbt2YbFYePXVV+nZsyfZ2dmsWLGC2bNnU1BQEGr2tGnTJho1akS7du1Cx+/QoQNffvklKSkp\n1WLbsGEDiqJw9dVXh5UPHjyY7du3hzVvatmy5SnPU1VVAoFAra7Jhg0b6NOnT6jpF0BKSgpZWVls\n3Lgx7JhRUVGh5eTkZFwuFwD79u2juLi4Wiccb7zxBrfeeiubN2/G6/XSt2/fate0ajNlgE6dOoXm\nK5tRVx7nZE68Hs8//zxDhw7l6NGjrFy5ktdee409e/aENUk7lQ0bNpCZmUmzZs1CZZGRkfTt25cN\nGzaEyiIiImjduvUZxytJkiSd3MVyr66NH374AZ/PF3b/DAQCp71/JiQkhM6lUmxsLKWlpWHb/Pa3\nvw3bplOnTqF7/7p167jsssvQdT3s+vp8PjZt2nTS8/zzn//MxIkTKS4uZv369SxcuJAVK1ag63qt\nv5ucTIsWLcKW161bR7du3TCZTKEYe/bsidForHZ9JKkq2cmUdMGIiooiKysLgI4dO2K325k0aRKJ\niYnccccdQPB9HSEEU6dOZerUqdX2kZeXBwQ7hPjnP//JkSNHSEhIoHPnzpjN5tC7QSUlJWfUA2JJ\nSQmRkZHVejaOj48HwOl0hspOt98mTZqE3lmpSX5+PjExMRgMBhwOR+gYVcXFxYXdqC0WS9h6VVVD\n51pcXAwEb541qVw/fPjwGtdXTQjNZnPYMYCw931qcuL1WL9+PY8++ii7d+8mJiaGzMxMLBZLrcfc\nO9U12bJlS2j5xGuiKEqt4pUkSZJO7mK5V9dG5f1z6NChNa73er2h+RM73tI07bSjJSQmJoYtx8bG\nUlxcjBCC4uJi5s2bx7x586ptV3l9ofp55uTk8PDDD7Ny5UqsVivp6ek1vj/9S5x4rOLiYrZu3cpn\nn312yhgl6UQywZUuWMOHD2fx4sW8+OKLXH311aSmpoZ+Cd9///1ceuml1bZJTU1l//793Hfffdx8\n882MHTs2dIO44YYbQvUiIyMpKiqqtv2KFStCN+6qoqOjKS0txev1ht2Q8vPzQ+trq3fv3kyfPp3i\n4mJiYmKqrb///vtxOp28//77REVFUVBQUK1OQUEBTZo0qdXxIiMjAaqd77Zt29A0LfTkd+7cuTWe\nR10OV1RaWsqdd95Jnz59mD17Nk2bNgXgvvvu48iRI7XaR1RUFLm5udXKCwoK6nX8RUmSJOnCvVfX\nRuX987XXXgtraVXJYPh1X9NLSkqw2Wyh5cLCQhISElAUhcjISEaMGMG1115bbbvKe2tN7r//fkpL\nS/nwww9JT09HVVXeeOMNvvvuu5NuoyhKtT8OV3aIdSqRkZFcc8013H777dXWnZi8S1JVsomydEF7\n+OGH8fv9PPPMM0Cw2WlaWhqHDx8mKysr9CksLOSFF17A4/Gwbds2fD4fd9xxR+gXaH5+Pjt37gz9\ngu7UqRNHjx4N65Fw586djBs3jr1796JpWlgcXbt2RQhR7a+Qn376Ke3bt6/2tPBUrr32WiwWC9On\nT6+2bvXq1axbt45rrrkmdNzvvvsu7GntwYMH2bJlS62HK6hsvvzNN9+Elf/tb39j/vz5dOzYEYPB\nQElJSdg1XblyJW+//Xa1a3Eytam3d+9eHA4Ht912W+gG7Ha72bhxY9jNs/LpcE26du3Kli1bOHTo\nUKisrKyM7777Tg7hIEmSdA5ciPfq2ujYsSOaplW7f37zzTcsXLjwlPey2lixYkVoPi8vj82bN9O9\ne3cAunTpQnZ2dthxAZ599lkKCwtPus/NmzczYsQI2rdvH4pv5cqVwPEWTideV5vNRnFxMW63O1RW\n9ZWgk+nSpQt79+4lIyMjFGNiYiLPPPMM2dnZtbkE0kVKPsGVLmhpaWkMHz6cRYsWsWnTJrp06cLd\nd9/NhAkTsNvt9OvXj0OHDjF9+nQyMjJITEykXbt2aJrGtGnTGDlyJLm5ubz88st4PJ5Qc9v+/fuT\nlpbGPffcw3333YfZbObZZ5+la9eudO3aNfSXyW+//ZZmzZrRvn17Bg4cyGOPPUZxcTEtW7bk448/\nZu3atWc8qH1cXByPPfYYDz30EDk5OYwcOZKoqCg2bNjAvHnz6NGjB3/84x8BuPXWW/nggw8YM2YM\n48aNw+v18txzz5GcnMzIkSNrdTyj0cif/vQnnn/+eex2O127dmXp0qXs2rWLqVOnEh8fz6hRo3js\nscfIz88nPT2d77//nv/85z/ce++9tT6vyifFX375ZVhT5qpatmyJzWbjueee4/bbb6e0tJR58+aR\nm5sb2h6CfxXfvHkzP/zwQ7WkdeTIkcyfP5/bb7+de++9F5PJxOzZs9F1PXTdJEmSpPpzId6rayMp\nKYkbbriBRx99lNzcXNLS0lizZg0vv/wyDzzwwK/e//Tp0xFCkJSUxEsvvURiYiIjRowAYPz48dxy\nyy088sgjDB48mKKiImbOnElERES1d2GrysjI4J133iElJQWz2cyHH37I8uXLgeArSSaTicjISLKz\ns1m9ejVdu3alb9++PPPMM0yZMoXrrruODRs2sHTp0tPGP2bMGBYvXsxdd93FqFGj8Hq9vPDCCxQX\nF5Oenv6rr4904ZJPcKUL3n333YfFYuHf//43AFdddRUzZsxg1apVjB07lueff57f/va3PP/88wC0\natWKqVOn8sMPPzBu3DheeOEFBgwYwPjx4/npp58IBAKYTCbmzZtHZmYmf//733nkkUdo164dL730\nEqqqEhERwZgxY3j33Xf517/+BcAzzzzD7373O2bPns1dd93Fnj17+M9//sPAgQPP+JyGDBnC66+/\njqZpPPnkk9x5550sXbqU8ePHM2vWLIxGIxBsZvTmm28SGRnJxIkT+cc//kFmZiZvv/12jc2hTuaO\nO+7ggQceYNGiRYwfP56tW7cyZ86cUMcdjzzyCH/84x95/fXXGTt2LEuWLGHSpEmMHz++1sdo27Zt\n6L9DTe8EQTBxfe6558jNzWX8+PFMmzaN9PR0/v73v7N///7Q+0x/+tOf2LFjB2PHjsXv94ftIzIy\nkjfeeIP09HT+9re/MWnSJGJjY3n33XdP2SxLkiRJOnsuxHt1bfz9739n9OjRvPrqq/zpT3/i008/\n5ZFHHmHs2LG/et+TJ0/mtddeY+LEiSQlJfHGG2+EmixfcsklvPLKK+zZs4c777yTp556iu7duzN3\n7txTNo2eNm0aycnJPPjgg0yaNAmn08krr7wCBDvNAhg1ahRms5lx48axY8cO2rdvz6OPPsq6desY\nN24cmzZtYtq0aaeNv3nz5ixYsAC32829997LX//6V1JSUpg/f758pUg6JUX82jfCJUmSJEmSJEk6\nL6xatYrbbruNzz//nNTU1HMdjiTVO/kEV5IkSZIkSZIkSbognJME98cff6RPnz4nXf/xxx8zYMAA\nOnfuzB133BHqvU6SJEmSJEmSJEmSTqZemygLIXj//feZNm0amqbVOEjz9u3b+f3vf8+8efNIT0/n\niSeeIDc3lzlz5tRXmJIkSZIkSZIkSVIDVK9PcF9++WXmz59/yo5n/ve//zFgwAA6deqExWJh4sSJ\nfPvtt/IpriRJkiRJkiRJknRK9Zrgjhw5ksWLF9c4uHalvXv30qZNm9BybGws0dHR7Nu3rz5ClCRJ\nkiRJkiRJkhqoeh0HNykp6bR1XC5XtYG0rVZraEyz0xFCoCjKL4pPkiRJkqS6l5dXWif7URSF+Hg7\nBQVOGtIgEDLu+iXjrl8y7vrXUGOvy7gTEyNPuq5eE9zasFgsuN3usDKXyxUat+t0CgqcqHXwXFpV\nFWJi7BQXO9H1hvODI+OuXzLu+iXjrn8NNfa6jDsurvZjRktnl6oGvyCpKgQC5zqa2pNx1y8Zd/2S\ncde/hhp7fcV93iW4rVu3DmuOXFhYSElJCa1bt67V9kKIOr1gui4IBBrOl7pKMu76JeOuXzLu+tdQ\nY2+ocUuSJEmS9MucdwnukCFDGD16NCNHjiQrK4sZM2bQr18/YmNjz3VokiRdJIQQIAKIig9CRyBA\n6MF5oQfX6QJd96PrOkIPoAd0dD2A0INTXegIPbhN0VEDTqcrVKdyP7quVxxLgBAV+w5O0YPbi4rj\nVl1fdTk4FUDFPAKlYoqA4P9VLHN8GSFA4fi0olwJ1QlSFI43JQptE14nWLGmRFJQZYOKelQ5Tk31\nK+ucoGL/1VeJsMLgKSnYzVm0vPSaGo8iSZIkSdKF6bxIcKdMmQLA448/Tvv27XniiSf461//Sl5e\nHt26deOpp546xxFKklQfhNARuq/i40eIimnFvEIA4VYpLnLg83nR/T78fi96wBf6CN0XTDJFAKEH\ngskjwYSSKlMFHRQ9OEVHUY5/1BoTtV/HfZLyU75RoQBanYdy0SjO2w7IBFeSJEmSLibnJMHt2bNn\n2Bi4jz/+eNj6wYMHM3jw4PoOS5KkX0AIHT3gDn78rtC80L3oAS8Bvxu/143f70H3ewj4vQj9+Ad8\nIHwoih9V+XXvFygVn1DSWK/9xNdM1xWEqPoBXSggFPSKsuDD1OA6RMXz0yoPTIMPYpXjD16Bqg9j\ng3WV4/NV8vOw5YpjhTYPrat6zKrPR5XQ/5/47LSyTKn2PPVknfwpYWtr3rZyRXi5UuWZsoJS9eCh\n64UCilBCB1AUjXbdLz9JLJIkSZIkXajOiye4kiSde0IIRMBNwF9OwO9E95ej+50E/OXo/nICfhd+\nrwu/r7wigfWA8KDgO+NjVSaiofymlh2f67qCrqsEAioBXUUPTbWKqRJMKAOh1sShh7ZCB3RRsRyc\nBpdFxVQPm1f04LIqgkmVoigoqCiqgqpoKKqKqigomoaqqiiKhqppqJqKqhpQDQYMBgOq0YBm1IiI\nsuHx68H6BgOqQUHVNBSDAUVTUTQNVA1F0yrmq5aduF4Nr6sF46ncDkWps97kNU0hLi6CwsKyBvUu\na9W4JUmSJEm6eMgEV5IucLruI+ArDX68pQR8DvRAGcWH3Licxfi8TnRfObruqmiuWzunSp/8fi34\nCQSngcDx5UBAI+BX0X2g+0D4BcInwCvAp4MvgOrzo/gDKH4/qi8QLPMH0AJ+VOFH0wPBqfCj6gEM\nBLCaDGgWM5rVgmqxolqtqJaKeYsF1WxGMZlQTWYUswnVZDq+bKpcNgenZnNovVIH3bI31CRRkiRJ\nkiSpoZEJriQ1YELoBHyl+D1F+L0l+L1FBLwloYTW7y1F6Cd7+zPciQlrIKDg9Rrx+kx4vQZ8PiM+\nvwGfz4DPb0D3a4iAhvABHh1R7gWnG9XlweD3YtQ9GAMuDLoHY8CLUfdh1b3BpFQEqjdsNRhQIyLQ\n7BFodjtaVASqPTY4b7ej2iMwRtqJSY7H6QdMllASq5jMdZKISpIkSZIkSQ2bTHAl6TynB7z4PYX4\nPAX4PQXBRNYTTGT93hI4g6euPp+G22PG4zHh9piCCazXiMcbnEexoiomVN2AUQ9g9Lkwuh0YSgvR\nSnKwekqJ9rswBdyopzuuoqBFRKLFRGGIikeLisIQFYUWHY0hKhotKir4iYwKJrAm02nj1zSFmLgI\ndPkkVJIkSZIkSaqBTHAl6TwghCDgc+Bz5+P3FOBzF+D35ONzFxDwOWq1D6/XQLnLgstlwe0x43YH\nk1hPRUKrGiOx2u1ERJqxW1WirX6EtwCDKw9D4RGUYwfRiwqO9250CorBgCE2HkNsXMUnFmNcxXxc\nHIaYGLTIKPlUVZIkSZKkUxJCEKgYszygCwK6HhzDXBf4dVExnrlesS74CZWJ4HaV9XURnA926Bic\n18WJy8Ex0hUFzBYjTqcnuA8RXCdOso+q64LlwQH4KvcpKsqD51TRt0nlfEUPkWFlFXX1igIdKjqS\nFBXrq9YXoU4mK7dTVJVAIFARJxVDBoZvD8HYKq+zqLIPUaU8FPPx/yrH11eJi9A8oW2q1j1eVuWc\nq/y31lSFW4d0oF9Wo1/2w1JLMsGVpHqmB7z43Hn4XDl43bn4XDn4XDnogVM3Jfb7VZzlVlwuSyiR\nPT41YzRZiIqxBj+xFpJirETaNcyuQrS8I/gO78F75Ajeo0cJlJWG7fuEjndBUTDExWFMTMKYmIgp\nMSk0b4iLR4uMrLNOjCRJkiRJqj+6LvD4Anj9Oj5fAI9fx+sLBD9+Ha9Pxx8IfnwBHb9fxx8QJ5SJ\n0HxAF6iqitPlxeevrF9RLyDw+3UCul4lWRVVklW9Nn9Xly4QAV2w93CJTHAlqSHTdR++8mN4yg/j\nLT+Ct/wofk/BKbcpd5lxOq04nTbKnDbKnFac5VbcbjOqphITZyM23kZ8io22CXaiY4NJrcHnwn1g\nP56D+/DsyMadfYDynBzKT3Hn0KKiMDVqjKlJU0yNG2NKTg4msvEJKAb560GSJEmSzhV/QMftDeD2\n+iumFfOeQLVyl9ePx1s1Sa0y9el4/AF8Ph2vP4D/AnvFR1UUNE1BVRVURUFVQFWDowlUzquKgqIE\n64aWVQWTQUPX9WBdlYptTtzHqfYZXFaqHJuKYwVnldCoEWrFgwG1YmVovVI5Op5SUValnGCBqhC2\nraYq2O1mysu9oWHzKvcf2r7KfKic48erHHav8nmFgsLhg3tY+MaL5Bw5QEJyU343+m5atG4X2r6y\nx5aNa5fz8aLXKXUU0bZ9J35/2wSiYmKprHH8GUiV8wBMJo0eWU0pdZTX7Q/BCeQ3WEmqI0II/J4C\nPM6DeJ1H8JQfxufK5WTvyHp9BhwOO6VlEThK7ZSW2ilz2ggENBQFYuJtJCRF0LZ1BDHxwaQ2KsaC\nqqoIvx/PoUO49/6I67s9HNm7B19e7klj0yKjMDdvjrlJU0yNm2Bp1oTkDm0p9SnyXVZJkiRJOgt0\nXVDu8eN0+yh3V536KXf7TpgGPx5/AKfLh9sbwOevfR8bdcmgqRgNCgZNDc5rKgaDikFTgvMVy8F5\nBaNRI8JmIuAPoKkqBkOVehV1DJqKph5PRA1qcFmtKNMqlkNlqoKmHS/TqpRVJrSVddXj2dQZacgj\nHJyN2D0eD9P++iS33DKGoUOH89lnS3j5P0/w3nuLsdlsoXq7d+/iv2++yIwZL9GmTRtmznyaj955\ngenTn69V3EbD2X99TSa4kvQLCaHjKc8jt/QYBbk7cZceQPfX/Bcpj9dMUVEExSWROEqDCa3HYwIU\nDEaV+KQImrWJICEpgoTkCOIS7BiMWmj7gMuFa9dOCr7cjnvvHtz79yF8NY8/a0xOxpzSHHNKcyzN\nUzGnNEeLjg5rUqxpCsbICJBjhEqSJElSrfj8AUrLfRUfL6XlPhwV09ITpy4fLo//rMRhMqpYTAYs\nJq3ic3wCLl/YAAAgAElEQVTeZNQwGzRMRhWTsWJq0DAZTliuMjUbNYwV640G9YwTxoacKErHbdy4\nHkVRGDHiegCGDBnGe++9xerVKxkw4MpQvc8//5Q+fX5DRkYmAHfeeQ9DhlxJYWEBcXHx5yT2E8kE\nV5JqqfIJrcuxB3fpXjzObETAU62erhtxlEaRn2+juCSSYkckHo85tD423kbLtCiSm0aR3CSK2AQ7\nqhp+M9HdLpw7dlG+fTuuHdtxH9gPevW/5GqRUVhat8baqjWWVq2xtGiBarHW+blLkiRJ0oXK6wtQ\n7PRSXOqhuMxDcZm3YuqpKAsuu72BX30sq1nDZjZitxiwWQzYLcbQNMJmJCHWRsAfwGysnrxWzp/4\nnUFqePwBnUJH7YZxrImqKXh0heKScvTT/FEhLsqCQTv9U9Ps7P20aNEqrKx581Sys/dXq5eR0TG0\nHB0dQ1RUFNnZB2SCK0kNge534y7bF0xqHXsI+Eqq1xEWiktiOHrUTkFRNKWldirfNtA0heSm0TRO\niaZR02iSm0Rithir7UPoOp6D2Th/+hHnlp9w791TPaFVFMzNUrC2TcPSug3WVq0xJCTIzp4kSToj\nc+fOZebMmRiNx38XzZkzh7Zt2/LII4+wZs0aIiMjueuuu7jhhhuA4B/4ZsyYwcKFCwkEAgwbNoyH\nH34YTdNOdhhJOi8IIXCU+8gvcVFQ4qbA4Sa/xB2aLy714HT/sietEVYjkTYjkVYjkXYTkTZTcN5m\nJNJmwm4NT2CtZg3tFKMLyCehFwd/QOeR2WvIL/nlCe6ZSIi2MHVcr9MmuS6XC4vFElZmNltwu90n\n1HPXqt65JBNcSTqB31NMecl2XCU78JRlc0L/wgjMlDoTOXzYTk5uJE6nlcqEVlUVGjeLoknzGJqm\nxpDcJCqsqXFVgXIn5du24vzxR5xbfyJQUj15NqekYE1vhy29Hda26WgREXV9upIkXWS2bdvGhAkT\nuP3228PK//KXv2Cz2Vi1ahU7duxg7NixtG3bls6dO/Pmm2+yfPlyPvroIxRF4Y477mDevHmMHTv2\nHJ2FJB3n8+vkFrvILSwnp8hFbnE5JU4fR/OdFDjcZ/Quq8moEhthJibCTEykmZgIEzERZqIjTETb\nTKFENsJqOGWyKkkNjcViweMJb5no8bixWq0n1DPj8bhrqGfjfCETXOmiJ4TA587FVbyd8pId+FzH\nTqih4BdJ5OXHsHePleKSSI73BwdxCXaatYwlpWUsjVNiMJ4koQXwOxyUbdpI2cb1lG//GQLhzZ0M\nsbHYMrOwZ2ZhS28vE1pJkurczz//zMiRI8PKnE4nX3zxBUuXLsVsNtOxY0eGDBnChx9+SOfOnVm8\neDF//OMfSUpKAuCOO+7gueeekwmuVG90ISgscXOkwMmxQhc5ReWhhLagxE1tnneajCrxURbioy0k\nVEzjIi3BJDYymNRaTJpsGSWdVQZNZeq4Xr+6iXJMtJ3iEmedNVFOTW3J++8vDCvLzj7AlVdeXa1e\ndvaB0HJxcTEOh4MWLVrU/gTOMpngShctn6eQ8sKfcBZtqTZ0j6KacfubcehwDLt3mvH7j/9T0TSF\nJqmxpLaKo0XbeFq1STplcyJfYSFlGzdQtnE9rl07CRvwTdOwtm6DPasT9qwsTE2byRurJElnjcvl\nYt++fcyfP58HH3yQqKgobr/9djp06IDBYCAlJSVUt2XLlnz++ecA7N27lzZt2oSt27dvH0KIWv3O\nqhyC49eqfPewob2DKOOuPSEERaUeDuc7OZzn5HBeGYfynBzJd572HVhNVUiMsdIo3kaz5EgiLYaw\nZDbSZjyv77Hy56R+ncu4NU2jcYL9F2+vqgoxMXashmBv3XWhR48e+HxeFi16lxEjRvLpp59QVFRI\n796XomnHr9FVV13Nn/88lmuvHUa7dh2YPftFeve+lLi42FrFXXV6tsgEV7qoBHxllBdtxVn0E97y\nI2HrVEMEXr05Bw5Es3O7hq4f/zZmMKqkto6ndbtEmreKw2gK/tOp+g8+7Djl5ZRtXI9j9SpcO7aH\nrVNMJuxZHYno2g17Vkc02/nTpEOSpAtbfn4+l1xyCTfddBPPP/88P/74I+PHj+e2226r9k6VxXL8\nnaoT382yWq3ouo7X68VsNnM68fH2Ok0sYmJ++RfDc0nGHS6gCw7nlrLncAm7DxWz51AJ+486cLpq\nHiUAQFUgKc5Gk4QImiTYaZxop2liBE0SIkiKtaLV4knV+U7+nNSvhho31H3sc+e+wmOPPcbLL79E\namoqL7/8Mk2bJjJlyhQAHn/8cXr1uoQnn3ySadOeJC8vj27dujF9+tPExdW+1eHZvuYywZUueELo\nuB27KcvfiMuxi6rv1KoGO7rWhoMH49m2JYDfd3ydyazRok0CrdITSWkZe9J3aUPH8ftxbt2CY/Uq\nnJs3hQ3jo1qt2Dt2JuKSbtgzMlFr8YVQkiSprqWkpLBgwYLQcrdu3Rg2bBjr16+v9u6V2+0OjX14\n4rtZLpcLg8FQq+QWoKDAWWdPcGNi7BQXO+vsqUV9kHEHmxgfyXey94iDA8dK2X+slOycUry+k78f\nmxhjpWminWYVSWyzRDuN4m2YDDXdjwUlJeV1Hnd9knHXr4YaN5y92JOSmvF///dKWFlhYRn33fdQ\naB6gV69+9OrVr1q906nLuE+VUMsEV7pg+b0llBVswlmwiYCvNFSuqCZMEW3JK2jC5rUCR7EXCPag\nqCjQvFU8aZnJtGgTf9qkFsCdk0Pu4k8o/vYbAg7H8eMYDNg7diKq96XYMjuiGqv3nixJklSftm7d\nysqVKxk3blyozOPx0LhxY3w+H0eOHKFJkyYA7Nu3L9QsuXXr1uzbt49OnTqF1rVq1ar6AU5CCHFi\nlwO/iq6LBtnL7MUUt8vjZ+8RR/Dp7OES9hxxnHRcWLNRo3lyBKnJkaQkR9AsMYLG8TYsppq/ptY2\nlovpep8PZNz1r6HGfrbjlgmudEERQuAu3Utp3lrcjt1h68z25viVdmzfYWP3z0XoVcawTWwUQVpm\nI9q0T8JmN53+OH4/ZT9uxvHNcpxbt4S9V2ttm0Zkr0uJ7NYdzd5wm71IknThsdlsvPjiizRv3pxB\ngwaxdu1alixZwoIFCygtLeWZZ57hySefZNeuXXz88cfMnj0bgGuvvZa5c+fSq1cvDAYDs2bNYtiw\nYef4bKTzicPpZXt2ETuyi9l1qITDeWU1dvxkNWukJkfSPDmSFo0iSW0USXKsrcG9vylJ0vlLJrjS\nBUHofpxFP1GauxafOzdUrmpWbLGdyC9qzprvSinIdQLBd8oMRpW0jGQyujQhITmyVscJlJVRvPwr\nipd/RaC4OFSuRUURdVlfovv+BlNFL6OSJEnnm5YtW/Lss88yc+ZMJk+eTHJyMk899RQZGRk88cQT\nPProo/zmN7/BZrPx4IMPhp7Y3nzzzeTn53P99dfj8/kYOnQot9122zk+G+lcKnf72JFdzM8Hitie\nXcShPGeN9RrH22jdJJo2zaJp3TSaxvE21PO4oydJkho+meBKDVrAX05Z3jpK89ej+4/fXE22plhi\nLmHfvmh++uYozrLjQ//EJ9rJ6NqEth2SMZlr90/Ae+woRcs+x7F6JcLrDZXb2ncgZeg1KG07oCun\nb84sSZJ0rvXv35/+/ftXK4+JieG5556rcRtN05gwYQITJkw42+FJ5yldF+w76uDHPQX8tLeAAzml\nYYMCQLAX41ZNokhLiaFN02BCG2GVr+dIklS/ZIIrNUgBXxmO3NWU5a9H6JWdOSnYYtpjsHdl6486\nPy85is97/Clry7QEOnVvRqNm0bXuzbN85w6Kln6Kc/MPoTLFbCG6b19iLh+AtWlj4uIigi/WN8B3\nICRJkiTpZMpcPn7aeIhVmw/z454Cyk7o3VhRoEWjKNqlxtA+NZa2TWMwm+QfeyVJOrdkgis1KH5f\nKaU5qyjL34AQFR1DqSYi4rui2Trz44YStv1wMPTiusGgkt6xER27NSMmrnbD8QghcO3YTsH/FocN\n8WOIjSNmwECi+/0GzSbfrZUkSZIuPIUONxt25LFhRy67DpdUe0qbGGOhY6sEMlrGkZYSg80iv0pK\nknR+kb+VpAYh4C/HcexbSvPXgwh2xaloZiITe6BZu7B5XR7bfvg5lNiaLQY6dmtGRtcmWG2n7zQK\ngolt+batFH78Ea5dO0Pl5pTmxF49mMhLuqEY5D8ZSZIk6cKSW1TOhh15rN+Rx76jjrB1Bk0hvXks\nWS3jyGodT6M4W52OaSxJklTX5Ld16bymB7yU5q3FkbMSoQfffVU1K5FJPTFFduGH73P5af3msMS2\nU48Usi5pWuv3awHKd2wnf9F/ce853vOyuUVL4ocOw96xk7yZS5IkSReU4jIPa7bmsGbbMbJzwsev\ntJoNdG6TQLd2ifTpmoK73NMghyKRJOniJBNc6bwkhE5ZwSZKjq5A9wdvvIpqIiqpN7a4HmzbnM+G\nVZvxuIPNlH9pYus5dJD8Rf/F+ePmUJmlVWvirx2GLSNLJraSJEnSBcPjDbBxZx6rth5j2/7CsObH\nEVYjXdomcEl6Eh1axGLQVDRNwWYx4i73nHynkiRJ5xmZ4ErnHXfpfooOfXZ8uB9FJTKhO5HJl7F/\ndzlrPtxMaUnFUD8GlU49UujcM+WMEltfQQEFixfhWL0qNIatObUFCdddj61DhkxsJUmSpAuCEIJd\nh0r4ZvMRNuzIw+MLhNZZzRqXpCfRs0My7ZrHoKnqOYxUkqRzbfPmH3jxxZlkZ+8nOjqGm2++heHD\nR+JwOHjqqcfZuHEddnsEY8aMZciQ4TXuY+fO7Tz99FT27dtLs2bNmTjxYTIzs+r1PGSCK503vO5i\ncvd8iLNoa6jMFptJTOMrKCkx8vG7uzh6qAQI9tyYntWIHn1bYo801/oYusdD4acfU/TZpwh/8Omv\nMTGRhBHXE9GtO4q8uUuSJEkXAKfbx6qfjrFi8xGO5B8fRk9TFTJbxtE7sxGd2yRgMspejyVJAofD\nweTJ9zNhwkMMHDiIXbt2ct99f6Zp02YsXvw+NpuVjz76nD17djFx4r20aNG6WuLq8XiYNOl+brll\nDEOHDuezz5YwefL9vPfeYmy22nX2Whdkgiudc0L3U5y7hgNHv0WvGPLHZG1MbMo1oDVi7bf7+WnD\noVBTquat4uh1eSvikyJqfwwhKNuwjrz33sFfWAiAFhlJ3NBhxPS7XHYeJUmSJF0Q9hwpYfnGw3y/\nPRefXw+VpyRF0LdjY3p0SCaqlp0vSpJ0dvh1P0Xukl+8vaaB1+SipNxJIHDqurGWaAzq6b/n5uQc\n5dJLL2PQoKsBSE9vR9eul7Bly498++0K3nrrfcxmMx06ZHLllVfx2WdLqiW4GzeuR1EURoy4HoAh\nQ4bx3ntvsXr1SgYMuPKXnewvIL/VS+eUx3mIwuz/4XPnAaAarMQ0HoAtrhO7f85n9VffU+4Mdi4V\nGW2hz5VtaNEm4cyOcfgwuW8vwLX952CBphE78ErihgxDs1rr9HwkSZIkqb7pumDjzjyWrstmz+Hj\nvSAbDSo92idxeeemtGoSJV+/kaTzgF/38/iapylwF9XL8eItsUzp9eBpk9y2bdP5+9+fCC07HA42\nb/6B1q3bomkGmjZtFlrXvHkqK1Z8XW0f2dn7adGiVVhZ8+apZGfv/3UncYZkgiudE3rAS8nRrynN\nWxsqS0zpjTW+L6UOWPLeFg7tD/7D1zSFLr1T6dIzBcMZNKXSPR4KPvqQomVLQQ/+FduWkUnSqJsx\nNW5StyckSZIkSfXM5fHz3Y9HWbb+IPkVfVMANI63cXmXplya2Qi7xXgOI5QkqSEqKytj0qQJpKe3\np2vXbixc+E7YerPZgtvtrrady+XCYrHUqu7ZJBNcqd65HHsoPPgxAW+waYbBkkBii2tp3Kwd33yx\nk1Vf78HnDba3SG0dR58r2xIVc2ZPWst/3kbO/Nfw5QU7qjIkJJB0403YO3eVf8GWJEmSGrTSci+f\nrzvIVxsP4/L4Q+UZLeO4qnsKGS3j5L1Oks5TBtXAlF4P/uomytExdkqK666JcqUjRw7z0EMTaNq0\nKY8//hT79+/H6w3vSd3jcWO1Vn+n1mKx4PHUVLd+W0zKBFeqN7ruo/jwMsry1wcLFJWo5D5EJ/eh\nrNTH6/9ZRfbe4PuxFquRvoPa0rpd4hndpANOJ3kL38Hx3bfBAlUl7urBxA25FtUk3zmSJEmSGi6H\n08vS77P5auPhUG/IBk2hV0YjBnVLodkZ9E0hSdK5Y1ANJNrif/H2mqYQFxGByWut0zGqd+zYzgMP\n3MNVV13DXXfdh6qqpKSk4Pf7OXbsGI0aNQIgO/sALVu2rLZ9ampL3n9/YVhZdvYBrrzy6jqLsTZk\ngivVC2/5EfL3f4DfUwCAydaEuObXYrQksnXTEVZ/tQd/RWcYbTok0WdgG6xn2AlG2aYN5LzxOgFH\n8P0jc2oLkv94G5bmqXV7MpIkSZJUj0rKPHy6Npvlmw7jrbhXmk0a/bs2ZVC3FKIjaj+agCRJUk0K\nCwt44IF7GDXq94wefWuo3Gaz06dPP2bNepFJk/7Gvn17WLZsKU8//Vy1fVxySXd8Pi///e87DB9+\nPZ99toTCwkJ69Ohdj2dSzwnutm3bmDJlCrt37yY1NZV//OMfdO7cuVq9hQsX8vLLL1NcXEzbtm35\n29/+RmZmZn2GKtURIXQcOasoOboc0AGF6Eb9iGrUF1e5ny/+u4UDe4JJb0SUmX6D2pJ6hp1IBVwu\n8t55C8fK4FNbxWQiftgIYgcOQtHk8AeSJElSw+Ty+Pl07QE+X3cQry+Y2FrNGgMuSWFQ9xQirPL9\nWkmS6sbHHy+muLiI11+fy+uvzw2VX3/9KCZN+htPP/0UI0YMxmq18uc//4WMjGBu9vnnnzJ//qss\nWPAeJpOJ6dOfZ/r0p5g16/9o1qwZ06bNuHCbKHs8HsaPH8/48eO54YYbWLx4MXfeeSdffPEFdrs9\nVG/79u1Mnz6dd955h9TUVF555RXuvfdevvzyy/oKVaojfq+DggOL8JRlA2AwxRLfYgRmezMO7Cng\n6yXbcZUHhwVq3S6RETd3xe3xnlFTi/KdOzg2bw7+/HwArG3TSL7tT5iSkur+hCRJkiSpHvj8Oss3\nHeZ/q/ZT5greJ21mA1d2T2Fgt2ay4yhJkurcLbeM4ZZbxpx0/RNPTKuxfNCgaxg06JrQcps2bXn5\n5Xl1Ht+ZqLcEd82aNaiqys033wzA9ddfz+uvv86KFSsYPHhwqN6BAwfQdZ1AIIAQAlVVq/XGJZ3/\n3I695B9YhO4vB8Ae34XYplchhIHvlu3ipw2HATAYVfoMbEtGl8bY7CbcHm+t9q/7fBQs/oCipZ+C\nEKBpJIwYSeygq1FU9aydlyRJkiSdLUII1v6cw6IVe0O9Ihs0lSu7NWNw71SZ2ErSRUKI4MMeQfWH\nPkIIUBT8eoCAHsBfMVLIKR8PiZOvPfVjpZrXnmobcYpjGamflpX1luDu27eP1q1bh5W1bNmSvXv3\nhpX16dOHFi1a8Nvf/hZN07Db7cyfP7++wpR+JSF0HMe+peTYCgAU1UR882uxxXagtMTN5x9uIvdo\nKQBJjSMZeG17omNtZ9SRlDcnh6Oz/g9P9gEATE2b0fhP4zCnNK/7E5IkSZKkepCdU8qby3ay61Cw\nZ1VFgcsyGzO8b0viouQf+hsyIQS60AmIQPCjB+f9enBZFwECQkcoOvm6iaKSMnz+wPH6QidQUbdy\nXzo6uhAIEZwGl/Xj6yvKRGUdKssr6lRsH1w+yT4Qwe0REJonVBZKZBSBZlDx+QIV2xOsj0CI0FxF\n/Zr3IaruHx0E6AgIlVWNpcr2lalWaBKeXIUt11BHUUCvjEGcULFK3eOrRLUa4cvV61SLqYYEsKZE\n9kKkoHBj1lAub9T3rB6n3hLc8vLyau2vLZbq4yJ5PB7atGnDlClTSEtLY86cOdx9990sWbKkVk9y\nFUWhLh7gqaoSNm0ozmXcAZ+T/H0f4HLsAcBkTSap9Q0YLfEc2F3A5x9tw+MKDmfQtXdzev6mJZqm\nnlHcjnXfc3TeXHS3GxSFuEFXkThyJKrx3PSQLH9O6peMu/411NgbatzSxafM5eODb/eyfNPh0Bfs\njq3juf7y1jRLlL0i1xUhBH7dj0f34g0EP56qU92HJ+DFr/vw6X78ur/K1IdfD1RM/Sesr7l+ZRIb\nTGD1c336knReEAhyy/LP+nHqLcG1Wq3Vklm3243NFj6G0osvvkijRo3IysoC4K677uK9995j1apV\n9O/f/7THiY+31+nYbzEx9tNXOg/Vd9zljsPs3vIaPncxAPFNe9C83XBQDKxYuoNvv9gFBIf/GXZT\nZ9IzGtW4n5PFrXu97Hv1dY598hkAxugo0u6/j5jOnc7C2Zw5+XNSv2Tc9a+hxt5Q45YufLoQfPfj\nUf67fE/oPdukWCs3D2xLx9Zn1tnihcyn+3H73bj8Llx+Ny6/u2LZjStwfDlU5nfj1b0E8FPudYcl\nsQ3xKZmqqGiKFvyoKioqqqKgKCqqoqKioCoqSmVZxXKoTkX9YJ0T1yuoVJRX1qlSX1UUFBRQFFQU\nILiNAhXT4PdtTVWxWk143P7QH2nUiu0qax2vX/M+KudDUxSC/6ss54TyYEQolXuA43OE9hm2XHms\nymurqUREmCkr8yB0EapVm21RTliuYe5k+6geX/i+wuopNW+raSpRkRZKS93ouqi2j5qdfO0vyZ1O\njCl8fzWXmzQDnVLTKS4uP+PjnYl6S3BbtWrFggULwsr27dvHkCFDwsqOHDkS9qRXURQ0TUOrZW+4\nBQXOOnuCGxNjp7jYWfGD0zCci7idRT+Tt+8DhO5DUQzEp/6WyITOHDtWxtIPtnJwXxEAiY0iuGZk\nJlExVgoLy2odtzc3h8P/9xLuA8Emybb0djS5Yzx6bGy1/dQ3+XNSv2Tc9a+hxl6XccfFyadoUt06\nVljOa59uZ+fB4B+FTUaVoZe2YFD35hgNF24/Et6AF4e3DKfPSZmvHKfPidNXTtkJU2eVqU/3n/W4\nFBRMmhGTZsKkGjGoRgyqhlE1YlQNGFQDxiplwWVDlanxhGVDRUKqoYUlqFWWK+bVinUGVcNkMJAQ\nF4WjxAW6Gkpcz3eaphAXF0FhYVmdjsl6tjXUuKFK7MaGFbumKaj10FdOvSW4vXv3xuv18sYbbzBq\n1CgWL15Mfn4+ffr0Cat3+eWXM3PmTAYPHkx6ejpvvPEGgUCASy65pFbHEUIQCNRd3LouGtQPTqX6\niFsIgSPn24ohgEAzRpLQ6kbMtibk55Tx6ftbKClyAdChc2MuG9gGg0E7ZVwnxl32wyaOzZ2N7nIF\nmyT/dgjxQ4ejaKfeT32TPyf1S8Zd/xpq7A01bunC5A/ofLL6AIu/248/EGy22i09kVED2jbY92yF\nELj8Loo8JTg8pTi8pZR4HeHz3lIcnlLcAU+dHNOoGrEYzFgNFqyaFavBgsVgqZgGy2MiIwh4wKgE\nE1ezZgqbmtTj80bVcF4kkpqmYDNZcWsBAg3wabMknS/qLcE1mUzMmTOHxx57jBkzZpCamsp//vMf\nbDYbU6ZMAeDxxx/nxhtvxOFwcM899+BwOGjfvj2vvPIKERHyL+jnE133UXhgMeXF2wAw2ZqQ2OpG\nNGMk2XsLWLZ4G15PAFVV6HdVGu07NT6j/Qtdp3DJ/yhY/AEAWmQkjf50B/YMOR6yJEmS1PDsOVTM\nzLc2ciAn2NFiTISJP1yVTpe2iec4slML6AEKXEXklxdR6C6i0F1MkbuIQk9xaN4TqN0ICFUpKNgM\nVuwmG3aDnYiKqd1kI8Jgx260YTfasBmtweQ1lMiaMain/vrakJ/MSZL069VbggvQrl073nnnnWrl\njz/+eGheURTGjRvHuHHj6jM06QwEfE7y9r6Nt/wIALbYLOKbDwVFY/O6g6z+ag9CgMVm5OoRGTRO\niTmj/etuF0fnzsG5aSMA5hYtafLnezDGxdX5uUiSJEnS2aTrgk/WHOCDb/YSqGgu/5vOTbjh8jbY\nLPX6NeykAnqAQncxua588lz55JXnk+cqIM+VT4GrkMAZdJIUYbQTZYokyhRJtDkqOG+uWDZFEmmK\nJMJkx2awoioXbnNsSZLOnfPjN6vUYPg8heTtfhO/N/hebUyTAUQmXYquC775bAfbfzwGQHyinWuu\nzyIy+syaXHmPHePgC8/hPRJMnqMuvYykP/zxnPWSLEmSJEm/VF6xi1c+3hYa+icxxspt17SjXWrs\nOYnHr/vJLc/nqPMYR5w5HHXmcNR5jHxXYa16+rUarMRZYoizxBBrjg1OLTHEWWKJNUcTaYo47dNV\nSZLOf4WFBdxyyygefngKl13WF4fDwVNPPc7Gjeuw2yMYM2YsQ4YMr3HbnTu38/TTU9m3by/NmjVn\n4sSHyczMqtf45W8hqdY8zsPk7X0b3V8OikZ86nDssRl4PX4+//B4Z1It2yYwYGg7jKYz+/Eq2riJ\nff+ege4qB1Ul8Xc3ETNg4HnxXowkSZIk1ZYQglVbjvHmsp24vcGOQQb1TGVkvxYYa9lp5q9V6i0j\nu/QwB0sPcaTsGEecx8gpzztlIqugEGOOJsmWQKItgWRbAq2Tm2EO2Ik2RmM1NMz3hCVJOjPTpj2B\nw1ESWv73v5/EZrPy0Uefs2fPLiZOvJcWLVpXS1w9Hg+TJt3PLbeMYejQ4Xz22RImT76f995bXG3k\nnLNJJrhSrbhKdpK/778I4UfRLCS2/B2WyBaUl3lYsvAn8nOCvRl37plCr8tbnXFSWvjVl+S8uQB0\nHTUigibj78LWrv3ZOBVJkiRJOmtcHj+vf7ad73/OBSDCamTMb9tzZe+WZ+2d0DKfk2zHIbJLD5Nd\neohsxyGKPMUnrW9UDTSyJ9PE3ojG9mSSbIkk2RJIsMRh1IyhevJdVkm6+Hz44X+xWKwkJSUDUF5e\nzuk+gsMAACAASURBVLffruCtt97HbDbToUMmV155FZ99tqRagrtx43oURWHEiOsBGDJkGO+99xar\nV69kwIAr6+0cZIIrnVZZwQ8UZv8PEGjGKBJb34zJmkRRgZMl7/1EaUlwfOO+V7Yl85KmZ7Rvoevk\nvfcOxV98DoC5WTOa3H0vxoTzu9MNSZIkSTrRodwyXvpwCzmFwTEeO7aO57Zr2hF3hq/rnIoQgjxX\nPnuK97O3ZD97SvaTU55XY10FhSRbAs0imtDY3ogmEck0tjciwRon33+VpHNI+P34Cgt/8faaBi5P\nGd4S52lHjzHGxaEYapfyZWcf4J133mT27NcYM2Y0AIcOZaNpBpo2bRaq17x5KitWfF3D9vtp0aJV\nWFnz5v/P3pnHV1He+//9zMzZsyckEBJCIOwg4oKC+wK4gGJdcGtte12rtpfW1tva9tda23qvXuvS\nKthqW1tr3W5LKyiCqIiCKLKHnbAkJJB9OfuZmd8fc3JyTk4ICSSB4PN+vc5rZp555pnvDOGc+cz3\n+3y/Rezbt6dL5+8ppMCVdEpz9Wrqy98BwObMY8Dwm9DsaVSWN/L2GxsJBiKomsKls8YwbFT3RKkR\nCFD5h/l4160FIOO0SeTefhfYZQiURCKRSPoXKzZU8td3txGKGKiK4PoLhzPtzMJjnmZjmiaV3oNs\nq9/Jzobd7GrYQ3O44xrwee4BFKYOZkhqAUNSCyhMzccpw4olkhMKMxKh7Mf/RaSmpk/Op+XkUPzI\no0cUuZFIhF/84qd85zsPkJaWHmv3+wM4HI6Evg6Hk0AgkDSG3+/H6XR2qW9vIgWu5LA0Vn1EY6X1\ndsbuKSB32M0ompM9O2p4d0EpesTA4dS44roJDCxIP8JoiUQa6ql4+kmC+/YCkHHRxYy9/27qG/0y\nDEoikUgk/YZgWOfld7ezYmMlAJmpDu6ZPZ6Swd37XYynMdjEtvqdbK3bwda6HTSGmpL6OFQ7xWlF\nDM8YyrD0oRSlFco5shKJ5Kj5859fYMSIkUyZck5Cu9PpJBRKrGEdDAZwuZLn1DqdToLBjvq6et7g\nTpACV5KEaZo0HniPpkOfAOBIKWbAsDkoqp0dpQd5799bME1ITXdy5Q2nkJndvUnjwf37qHj6SSL1\ndSAEA264kewZMxB9lHhDIpFIJJKeoKrOx7P/2Eh5tReA8cOyuGPmWFLd3cv8b5gG+5rL2VBdysaa\nUg54q5L6pNg8jMgcTkl6McMzhpLvGYiqyN9NiaS/ITSN4kcePeYQ5fR0D409GKL83nvvUltbw7Jl\nSwDwer387Gc/4pZbbiMSiVBVVcXAgQMBK5S5uLg4aYyiomLefPP1hLZ9+/YybdplXbyynkEKXEkC\npmlSX/42LTWfA+BKH0nO0OsQikbpugN8+M52ADJz3MyaMxFPqqOz4ZLwlm7mwO+ewQwGEHY7g+68\nh5RTJ8lMyRKJRCLpV3y29RB/XLSFQEhHCJh93jCunFKE0sXfs7AeZlv9TjbUlLKpppTGUHPCfk3R\nKEkvZnTWCEZnjWBwyiA5b1YiOUkQmoY9N/eoj1dVgSsrBb+j5xLA/e1vbyZsX3fdLObO/QHnnHMe\nO3ZsY/783/Lggz+mrGwXS5Ys5rHHnkoa4/TTzyQcDvHGG39n9uzreOedhdTV1TF58pQesbGrSIEr\niWGaJnX7/o23bh0A7szxZBddjRAq6z7dx8r3dwMwYGAKV95wCq5uvqFuWr2Kqhd+D7qOmp7O4Pvn\n4hw6tKcvQyKRSCSSXiOiG7z2/k6Wfl4OQJrHzl1XjWNMF2rb6obOppptfH5wPRtqNhHUQwn7s51Z\nnDJgLOOzxzAsfSj2uIzGEolEcrx48MEf89hjv+aaa67A5XLxrW99m3HjxgPw7rtv89JLf+Svf30N\nu93O448/zeOP/5r585+loKCARx99QoYoS44PpmlSt/+tmLj1ZE8iq/BKQLB6eRlrPrHmyg4qSOeK\n6ydgd3Szxu3SJVT//WUAbHkDKZj7PZkpWSKRSCTHnWZfCI/L1iXPa11TgOf+uYldB6w5sSMLM7j7\n6nFkpBw+mskwDXbW7WHjrk2s3P8F3rAvYX9RWiGn5IzjlJyxDPLkyYgmiURyQvDGG/+OraelpfOL\nXzzaYb/p0y9n+vTLY9slJSOYN+/FXrevM6TAlbSJ21orm3FK9ulkFl4BwMfv7WTj5xUAFA7LYsY1\n47DZuj7nxzRNav/xJnWL3gLAWTyM/G//J1pqWg9fhUQikUgk3ePjjZW8sHALp5bkcP+1EzoVlxt2\n1fCHt7bQ4g8DcNlZQ7j2gmGoSsdhw3WBelYe+IyVlZ8n1aQtTivijLxTOTV3PBmOo09GJZFIJJJk\npMD9kmOaJvX7F8bErSf7tJi4/ejdHWxeewCAYaNyuPSqsahq1+f/mLrOwb/8iaYVHwHgHjee/Hvu\nQ3HKLI8SiUQiOf58sb0aMNkcXs6jH6/iO2fdituWmDgxEIrw6rKdfLjO+j10OTT+48oxnDYyOQpJ\nN3Q21pTy8YHVbKnbjknb3Lii9MFMGnAKpw2YSLYrq1evSyKRSL7MSIH7JcZKKLWIltovgPiwZFj+\n7g5Ko+J25Pg8LrpiFMph3lJ3hBEMUjn/Wbwb1gOQevYUBn79P7pcaFoikUgkkt6m2R9GOL1oefuo\nCJi8/cZvueKM2bhGjEQ3DD4tPcibH+6mvtkqezEsP407Z40lNzNRBB/yVfPJgc9YVfU5zaG2GrUe\nzc3kQadxbsFZTBhSQl1dzyWEkUgkEknHSLXxJaUtW/IaADxZp5JVOBNIFLejJgzkwstHoShdnxOk\nt7RQ8cyTBHbtBCBz+mXkXHcDohsCWSKRSCSS3iYU1hEOPwAj9waZsLKUvcu28NkF/8GGOpNanxWO\nrCqCq84t5oqzh8RCkkN6mHXVG/nkwGp2NOxOGHdUZglT8yczccB4bIqGqsp5tR1hmiboOqZhgKFj\nRuLWdQNT12Pr1lKPthlt66aBaZhgGGCamKaJIkwMt53mZj+GboBhWvsMw+pvWtsY0XXDjI5jjdH6\nad2Ob29bN8DEarMuBlo99mbbtmlGG2JtcduYUVusdQHUOmwEgyHrmkysKIC4Pm1BAVF7zLZ1iDtH\n/LGt7bHzd/iP0Z1m4gxBCKi0aYRDEet6usrh+nbYfDj7uvnCyEy0u0JTiUR0634fxRhd695B/+7a\n3Q4hoLzV9n70zkyxaYgbroXikb16Hilwv4SYpknDgSWxUkCerIlkDZkFwPLF2yldZxWrHz1hIBd0\nU9yG62qp+M3/Eqq0BHLO9XPImnH5EY6SSCQSiaTvCYUNhBoBIKfBWga0FFr2BykSCi3A2BE5XH9R\nCQOz3Jimyf7mClZWfsbqqrX4I/7YWGn2VM4edAZTB01mgDv7eFxOlzFNEzMSwQyFMEIhzGAQIxTE\nDIasZchaEg4R0AQtDS0Y4bB1TCSMGY60rUci0e3W/ZHEvtH96DqmHrEEYkzUGsf7Vkgkkj6mavES\n8u6WAlfSwzQdXEHzoVUAuDMnHFbcXnjFqG5lcwweqKDiN/9LpL4OVJWBX/8maVPO6fkLkEgkEomk\nBwhFdLDrADhDlthqcOaBUFGBa8d5GTlkEU21hXxSm876ms3U+GtjxwsE47JHMzV/MuOzR6MqXU/C\neDSYpokZDKJ7W9C9Xgy/v+0TsJZ6B22x9ZiYDZ1c4lIIUBTrmUVRQAgURcEUAoRACCXaR1jRZPHt\n0W0R3Y9QENFlrF9rn9Zjo1586xlJgIjaQNwSYa3Gt8c/UwmBaHesUAR2u41QKBI3VrRPXF9B65it\n7Yl9RJwNHfU5/KPdYXYc7oDYpQmcThuBQPjw3sTDjHF4UzrY01N2R1GUNrsNw+ywf7ezmh/hXnWh\nsUsk2d5PUG0aQy6/FN+Rux4TUuB+yWiu/ozGyvcBcKWPIrvoakAcs7j179pJxVO/wfB5EXY7+d+6\nD8/4U3rjEiQSiUTSA9TU1DBr1ix+9atfcdFFF7Fx40ZuuOEGnHGJAO+66y7uvvtuTNPkiSee4PXX\nX0fXda6++mp++MMfoqq9K+h6m1DYAJclJjzCAQQwRNs1Zbl2gxHC2bKLzU0+mtVTSXFfiN1czzmD\nSjh70OnHlAXZNE0Mn49IQwORhnr0xkb0lmb0lhZLxLYkfgxvC2YkcqyXfUSEpiEcDhS7A83lxFRV\nUDUUm83ap2kIzYawaQhVs5atbZqG6LCfCqqKUNTouhK3rlriUVERqmK1ta4r0f2xdQWhapagVdU2\ncRqHqgqyslL63ZxnaXff0l/thv5ru6oKnFkp+Opajtz5GJAC90uEt24j9eVvA+BIGUrO0GvpCXHb\nsmEdlfOexQyFUFJSGPztubiGDe+NS5BIJBJJD/HQQw/R0NBWvmbLli2cf/75zJ8/P6nvyy+/zAcf\nfMC//vUvhBDcddddvPjii9xxxx19aXKPoxsmQlgPh6pp/e6ZWW0Zjm1am5i8wJPBO+bpAKTYp3HZ\n0GIAqhv8PP3GBgZkuLj3K+Njc3SNQCAmXI2mRvwhH00HqtgaqKSCRiZt8yPqGjDD4WO7CFVFcblQ\nXS4UpwvFlfhRXW4UpxPF5ULYHSgOO8JuR7E7otsOFHu0zeFA2O2xnBn99SFaIpF8uZEC90uCr3Eb\ntXv/CYDdnc+AYXNAqIni9hQroVR3xG3TJx9T9acXwDDQsrIpmPs97IPye+UaJBKJRNIzvPLKK7hc\nLgYNGhRrKy0tZfTo0R32X7BgAbfddhu5ubmA5dl96qmn+r3AtcISrVBdNRqxa8vIgahzQVHbwniz\nFBtY0cw0hiKUfrGOgoCX5TtaqKixUVHj5e3HX2Rc0070hgaMQCDpfGEV3vhKDmGbgrfRx7kHE8Wt\nmpKKmpKCmpqK4vFY656UaHt0OyUVxZOC6nGjuNyWt7S7YZQSiURyEiMF7peAQPMeasreAExszgEM\nGH4zQrEfs7itW/w2Na+/CoA9fzCD5z6ALTOzNy5BIpFIJD1EWVkZf/zjH3nttdf4yle+EmvfsmUL\ndrudiy++GMMwuPzyy5k7dy52u53du3dTUlIS61tcXExZWRmmaXbpd0MIQU8k0m9Netid5IedYQlc\nyzOpRCcPmtF5tDlZduyiTYAaJF7Ae3sPMmPhK1TmTIYM68XAR/5MRlRVJc2sU9xuHNlZeHM9hG31\nAKwZ5+Er59yGOysbLSMTLT39hCul19P3u6+Qdvct0u6+p7dsP3ToIP/9379i3bq1eDwebr31a9xw\nw000NTXxy1/+nDVrPiMlJYVvfvNOrrpqdodjbNu2lf/+719RVraLwsIh/OAHP2L8+Am9and7Tqxv\nUkmPE/QdoHr338HUUe0ZDCi5FUV1sWLJjqMWt6ZpUvPGa9QvtsKdncNLGHz/f6KmpPTadUgkEonk\n2IlEIvzgBz/goYceIiMjI2FfZmYmZ511FnPmzKG2tpbvfOc7PP300zzwwAP4/f6EubkulwvDMAiF\nQjgcjiOeNzvb06NexowMT4+Mo0dLygBo0RBl1WYDwJXmISXTTTjUSHlFLu78YMKxlQXDODhqHCKc\nFmurdmTim30bk0qysWdlYs/Kwp6ViRq9R5sPbYf3fxPrXz7CxUXDTu2Ra+lNeup+9zXS7r5F2t33\n9KTtpmly++3f56yzzuL55+exZ88ebrnlFiZPPp0//elPZGSksXLlSrZt28Ydd9zBqaeO59RTE7+/\ngsEgDz74Xe6++26uv/56FixYwIMPfpelS5fi8bTZ2tv3XArck5iwv5rqnS9jGiEULYXckltRtRQ+\nfm8nm75IrHPbZXEbiXDwpT/S9MnHAHhOmcigu76F0oUHHIlEIpEcX5599lnGjBnDBRdckLRv3rx5\nsXW3281dd93FE088wQMPPIDT6SQYbBN4fr8fTdO6JG4Bamu9PebBzcjw0NDgPebMoaZpYhgmSjRE\nWUTnmIaiS8M00Y1oHVxnEfsONEGedaxbD+JTHayadh0Z5X7YVBUb9+36NE4fewq6EPgBvzeM4o+Q\nkeHhUH1dgg3/t/kdJqSPRxEnZp34nrzffYm0u2+RdncfXTdoaQoeueNhEALS0tw0NfmOWAc3Jc2B\nqh75O2bTpo0cPHiQb37zbpqbg2RnD2L+/Bex2x0sXbqUV1/9P7zeMAUFw7j00hm8+urrDBlSkjDG\nypWWPrjssqtobg5y8cWX8cILL7Jw4WIuvXR6j97zrKzDO9akwD1JCQfrObTrrxi6H0V1kltyK5o9\nk5XLdrHx8woARo7L65a4NYJBKuc/i3fDegDSpp5D3te+ccKFVEkkEomkYxYtWkR1dTWLFi0CoKWl\nhe9+13rb3tDQwL333ktKNBonGAzGBOzw4cMpKytj4sSJgBXmPGzYsC6f1zRNdL3nrsMwzGNOeqS3\nlslpDVGOPmy1hiIrisA0LKPHn1ZIpDwDglbd22n25fwrcin14Qjl5VbIsceu4g3p7Kxo5KP1lZwz\nYRDtCUUS59we9FWzpmojp+We2FUHeuJ+Hw+k3X2LtLtr6LrBK8+vprkxeZ5+b5Ca7uSmOycfUeRu\n2bKFoUOH8cwzT7FkyTu43R5uu+2bDB9egqpqDBw4OHafCguH8OGH7yfdt7KyMoqKhiW0DxlSxJ49\nexLaevueS2VyEhIONlG1/S/o4WaEYmPA8JuxOQew6oPdrP+sHICSsblcdOXoLsfA6y0tVDzzJIFd\nOwHIvOwKcq69Xia2kEgkkn7EO++8k7B98cUX85Of/IQLLriA6dOnY5om3/ve9zhw4ADz5s3jhhtu\nAOCqq67ihRde4Oyzz0bTNObPn8/VV199PC6hx2j1HoiYwI22xwSugmlaWZSFYmPQ0CzYZr0gHkAd\nZ6obWG1MJGSz+rtDOjagAXhp4Ra2fLCb7EwXqelO0jNc5A5Mo9JrZa1O0VIoTM1nS/12FuxcxISc\nsdgU+UgmkUiOH01Njaxdu4bTTz+TN998i61bS/ne977NY489mRSt43A4CXSQSK/9dJbO+vYm8tv0\nJEOP+Nmx5iUiwXoQKgOGzcHuHszq5WWs+3Q/AMNHD+CSmV0Xt+Gaaiqe+g2hSiusOef6OWTNuLzX\nrkEikUgkfYuiKMybN49HHnmEs88+G6fTyZw5c7jtttsAuPnmm6mpqeG6664jHA4za9YsvvGNbxxn\nq48NvTU8LipwRXTbjIYLKypgWh5coajocXGA6QMmM6l6FYdEFvWG9TCnZNkoqtdpMg3CQKk3SIk3\nRFV5U+y4ugH7oBiC3gjhtbkwfjs1gTp+9+9XGScm4XTacLo0nC4bDlfbutNlw+G0YXeo8sWyRNLP\nUVWFm+6cfEwhyqoqSE9309joO6IntKshyna7ndTUNL76Veu7fcKEiVx44cW88MJ8QqFEW4PBAC6X\nO2mM9tNZ2vq6jnj+nkQK3JMIQw9RvetvBL1VgCBn6LU4U4fx2UdlfLFyHwDFI3O4ZNYYlC5OhgqU\n7abi6SfRm5tAVRl42zdJm3pOL16FRCKRSPqKZcuWxdZLSkr405/+1GE/VVWZO3cuc+fO7SPLeh+j\nncBtC1GOJptS24SkECp61MOrANmF03GnD2fG/nfZbI4mCDSnODljSpDhNS7+79NGGgBRmE5JipPm\npgABX5i6aNkhYSo4/WlkHRpCXd4+dno2Ym5KwxlI7dRmIYgJXbtDw2ZXsds17A4Vm0PDblex2+PW\no300TUGztS1VTcFmU1A1tV9mkJVI+juqqpCeefSiz6pR7QGl50J9hwwpQtd1dF1HVa1s8rquM2LE\nKNavX0tVVRUDBw4EYN++vRQXFyeNUVRUzJtvvp7Qtm/fXqZNu6xHbOwqUuCeJJhGhJqy1wh6rRDk\nnKFX4c4Yzecf7+Hzj/cCMHRENtOuHtultzgALevWUvn8c5ihEIrLxaB77sMzdlyvXYNEIpFIJH1F\nzINLYpmgmMDV2mrgCqERifZXo4LQlTacIWPuZMDqj2jGRAhYE8ijIOUAY/ObKT2Qymf7G5g4cwyX\nTcwnKyuFV78I8OaOUjLSPcy68RTqmoby19qX8CotVI/bzNn1Mwj7DQL+MAF/hFAwkmCzaRLdlziX\n91hQVJEkgDVNQdUUNE3B6bShGwaKIlAUBUUVqKqCqratd7iM9lUUES0TJRBKu21BclsHfdq3C0Wg\nCAHCEv2tXm3p3ZZIjp4zzzwLp9PJH//4e77+9dvZsmUzy5d/wG9+8zuqqg4wf/5vefDBH1NWtosl\nSxbz2GNPJY1x+ulnEg6HeOONvzN79nW8885C6urqmDx5Sp9eixS4JwGmaVCz5/8INO8GoGDUVdhS\nT+WzFXv47KM9ABQNz2b67HFdFrf1y5ZS/crLYJpomVkM/s5cHAWFvXUJEolEIpH0KTEPbnTybasH\n14zOwdXUtr7xIcpqnIgSiopQU4Am8l0mzUA5+dhHZpLrK+dQg5MXF5YSbtnBrEvOIhINeXbY7RQM\nzaKALL5ZdxPPrPs9jWodvol7uHHUNXE2GgQDEQL+CMGosA0EIoRDEUJBnXBIJxSKEA7qhIIRQiE9\ntq+1PRJpE+od3gfdJKTrhII9mAXsOGL98wiEAoKORHCiIBaCaB/Rdqx1GETbRExMi2h73Hp0O3pk\nwnayXW07k7S4sI7XbCqRiE5i7+SV5HMnnajD87R/CZBsR8fjd4YAbHaNcChCV3yJvfcaonsjCxFn\n9+EM7wVje2JIIQQ2u0o4pGO2Gt8DA18387ssXvZX/v7KKzgcTi457ybKtwnOGHsti5b+mZlXzsBu\nd3D+2deyfyvs37qJjVs+4ePVb3H3bb8C4JrLvs0br/6ZZ3/3WzIzcpl92b18uGgXAKqmcO4lI0hJ\ntx+7sZ0gBW4/xzRN6vb9G3/jVgAyBl1AXtF5LF1YyqcflgEwZFgWM67pmrg1DYPq11+lYcliAByF\nQxj8nbloGZm9dxESiUQikfQx7T24IlYeKNmDi9A6FLgAkehxp+Tlkl+cxYK9BwnhglOG4VpzAL9X\n8NIHYRpq/og/1yoTpBhhTCOCUDRGZ41getFFvLv3fT6qWMnglEGcN/hsq5+i4HLbcbmP/mHQMEz0\niEEkohMJxy8NImE9ttQjRtJ+0zSx2zS83iCRiIGhG+i6aS2N6FI3MHQzcWlYSz1iYpompmFiRJdH\nKmlyrFjjm9Hp0/0vo69EcnzRmDLh6zChraVsRw0Ap42aw2mjkttTtJHMmPrd2Da4uOD0u2P9wi3E\n7bOiYKbNHttbFwBIgduvMU2Thop38dZZZXtSB5xFRv4FrHhvB58ss96UFAzNZMZXxqFqRxa3utdL\n5e/n4du0EQD3+FPIv/seFGffTgyXSCQSiaS3OeIc3LifTSHaPLhaksA1ov0VJuWkMTTVxRtlBylr\n9pN6Wj6RtdWEW8Is2DSSYRM+AxcYwRrKN/wPjtShOFOKmZ43nvLmCkrrtvPa9n+S5cxgXPboHrlO\nRREodhWbXT1y53ZY8/xSqKtr6bF5fqbZKnqtf4PWesSt62bcumEQFcVmQt/WPtZ4AJZwtj4mioCU\nVBfNTX503Yjta+vXJrRj69GliQkd9CdufLDqJFsDJF9f8jV33BBrbv1TFOBy2fH5Q9Z1t9vftpl4\n7uR+8femIzs6P779+EdCKAKn00YgEMY8TnVwj+bFiVDA6bARCIYxOwx06Plr6akRFSFwOGwEg2Hr\nb7EXbntv/EtqqmDKBSVH7nis5+n1M0h6jaaq5TRXfwqAJ+tU0vOn8dlHe1gdDUsuGJrJ5deOR9OO\n/KMWqjxAxW+fInzwIADpF11C7o03I9Tu/yBKJBKJRHKi0ypYRSyLsvWEa3lwTVQ1bg6uEufBVToW\nuLboi+RMh43bRw1mXW0zi/bXwGm51K+vJtwYYl9DKjZXLcIUmGaEQNNOAk1W+b3pqBzSHNREgvx+\n40t8a8LXGNlDIvdEwgoFFqBAbz1h9IYw7wuk3X1Lf7Ub+q/t8Xb3JlLg9lOaDq2isepDAFwZY8gs\nvJLPPtoTy5ZcNDyL6deM65K4bdmwjqrfz8fw+0FVyb3lq2Scf2Fvmi+RSCQSyXGlzYMbzWwc8+Ba\nqGrbQ6MQKhHDSux0uBDlxKzLgkk5aYzO8PD+gTpWqgq1W2rRo/N9yxtzaBl0Ibm2PQS9+9DDzTjQ\nud6j8nKzoMmI8Nz6F7k5K5fi9KHY3YOwuQZidw9C1ZJLc0gkEomkjS4L3EAgkFS4V3J8aKldR0PF\nuwA4U4eRPWQ2qz7Yw/rVVp3bUePyuHjm6A4yByRiGgZ1i96idsE/wDRRU1PJ/9b9uEaM7PVrkEgk\nEonkeJJUBzdhDq6JEidwEUpciHLiOK0eXK2D8nsuTeWKIQM4Lz+Tj4ZksWjtOkwgHIL//WcTp585\nnlumzsKDl0DLPjzefdxi282faw/gM03+VneIa0PNDLGVxsZUtBRszhxszgFxywEomltmEZZIJBK6\nIXBnzZrF008/zZgxY3rTHskR8DVsoW7fvwGwewrIHno9nyzbw8Y1FQAMHz2A624744iFnyNNTVT9\nYT6+0s0AOIYUkX/vt7FlZ/f+RUgkEolEcpxpPwe3LUTZalZEogf3SCHKWnvlG0eGw8Ztpwylojad\ndYcAoWIaJp9/WsGmXbWceXYB5w8ZwbDCU8geIvjPxr38dsMfaQr7eN0b4NqMHIbit+yLtBBsaSHY\nsifhHEJ1otkz0RwZaPaMuPVMNHsGQpFTjiQSyZeDLgvccDh8zG8GS0tL+elPf8rOnTspKiri5z//\nOaeeempSv88//5xf/vKX7Nmzh4KCAn70ox8xZUrf1k86EfE1bqNmz5uAic2VR07xjXy4uIxtG6sA\nKBmby/SrxxwxW7Jv21Yqn5+H3tgAQNq555F7060oDkdvX4JEIpFIJCcESR7c1hDlaHNCCgqhENWx\nySHK0QM68uC2R4uGQ08qyaXcn0ZFeROBmgAr3t7F+pJaCodnMDk3g4lZg/nu6ffz9LrnqQvUkRMC\nqgAAIABJREFU83pDLdeXXMnkzCGEAzWEA9VEAtWEAzXo4WYATD1A2F9J2F/Z4bkVzYNqS0W1paJF\nl4kfD4rqlkJYIpH0e7oscGfOnMk3vvENrrzySgoLC5PClefMmdPp8cFgkLvvvpu7776b66+/ngUL\nFnDPPfewdOlSPB5PrN/Bgwe55557eOSRR5g+fToLFy7k/vvvZ8WKFV/qEGl/43Zqyl4H00BzZJNV\ndCNLFuxiz85aAEaNz+PCK0ajdPIDa+q6FZL8r3+CaSLsdvJuvY20qef01WVIJBKJRHJCkDAH1zQR\nZqLAFUpckinRcR1cIFZnVutCKb7WOripDgcP33I6H26o5NX3dhAM6TRtq2d7lZeqUT7eSbVTnOri\n0qG38uG+VznoO8SrO/7NocJz+UrJTBTRdi5DDxAO1BAJ1hEJ1hMJNUSX9THxC2BEvBgRL2F/Vac2\nCsWBqrlRNDeqzU2TJ42wbkdR3SiqE6E6URR7dN2BojpRVAdCscsQaYlEckLQZYG7aNEiXC4Xy5Yt\nS9onhDiiwF21ahWKonDzzTcDcN111/HnP/+ZDz/8kCuuuCLWb8GCBUydOpUZM2YAlrAuLi7uVLid\n7Pgbd1AdE7dZZBTexNtv7qayvBGAiZMLmHLR8E5/WEJVlVS9+HsCu3cDYM8fzKC778WRn98n1yCR\nSCQSyYlEqwdXCJO4hMkYrZ7ahDJBCpHWObhxIcqmacbG6SxEue2cenQMDSEEF07MZ1JJDi8v2c7n\nWw8RbgxRu/ogrnwPO4als7tZRaiXk+b4gKbgXt7fv4KD3mpuG3cjKTbLOaCoThyeAhyegqTzmUbE\nEryhBvRwc+InFF1GWtodEyQSCkKoHgB/4xEvq+0+qQ4UpVXw2hCKHUWxRdfbPodtExooKkK0fuK3\nNYRQ47a/vM+FEomkc7oscDsStt2hrKyM4cOHJ7QVFxezOyq4Wtm8eTN5eXnce++9fP755wwdOpSH\nHnoIu71rRc6FEPSEFlaiP2CKcnzfRvoad1FT9hqYOpojk/SCm1j4+m5qDlk/SFMvHsaks4fExG17\nu03DoP69pRx643XMUAiAjIsuIm/OTSdUSPKJcr+7i7S7b5F29z391fb+arek74ifg6vE1e6MzcFV\nTCulsrBCdluFbLwHV487rishyhEjEh2jrW+6x863Zo9n/c4a/r5sJwfrfPgPeAkd8uMuTMFVmIqw\nXYrdWEkoXEpp3TZ+tvIJvjJiDpPzRiQI7vYIRYsmoso5bB/TNNDDLRgRH4buQ4/4MSJe9IgPU/ej\nqSH83ib0sBdDD2DoQUwj2PFYehBdD6KHj3gregARFbxanCBWQQiEUKjSNHQdq9gpVlv7dUF0GW2P\nrcf2RdsQsXO2JvAUsXXRaXub86FdOyQdrygCw+fE6wtFX7Qk/9smOzMOsy0Ot5+464kN2rUxO9gl\nsOxWdRe+Zn/sBdHhx+7Ypt4i6VrjUFRBk+nE3xzAOJZSO312OW0nUhRBk+nC3+xv+y7rATaXbuN3\n816kvLySrKwMvnrL9Vxy0Xk0N7fw+G+eZe36TXg8br52y/VcPuOSDsfYsbOMJ5+Zz9695QwePJDv\n3HcnY8dYCWxVzYaZ0fvJbLtVJqi2tpbXX3+dPXv28P3vf59PP/2UESNGMGLEiCMe6/P5cLlcCW1O\np5NAIJDQ1tjYyPLly3nmmWd48sknee2117jzzjtZvHgx6enpRzxPdranR0NkMjI8R+7USzTVbufQ\nrr9jmjp2VxbZQ27l9b9so7HejxAw8/qJTDprSIfHZmR48JVXsOu5+TRtshJJ2bOyKLn/W2SeNqkv\nL6NbHM/7fSxIu/sWaXff019t7692S3qf1pBjhIkS93zY+oAuogK31VPYUYhyONL2NN+dEGVNSX78\nmliSw7jiLJatKWfBx3vwByM0lzURrPAyuCQTW+5UfEo2/uAn+CPNvLzlBf5RdhajsyZTku5mWKqb\nfLcjKQnWkRBCQbOngT0tad/ham2apolphNoEb3RpGMG2bSOM2e7TeVsI6M6DuolpRkCPdHhUn2js\nXqD2eBtwlBw83gYcJZ0H7J/Y9LTthmHyk/+3mG/ccApnTRrP1p21/PLxZxjoLuXlf27GblN59pGL\n2VfRxP889wJpyiZGFGcljBEK6zz08HtcPX0ED31rGitWl/Pjn/6cJ392KU6H9b0XajqDtPwre9j6\nRLoscEtLS/na175GSUkJmzZt4t577+Xjjz/mhz/8IfPmzTtiEiiXy5UkZgOBAG53Yj03u93O+eef\nz7nnngvALbfcwgsvvMAXX3zBRRdddEQ7a2u9PebBzcjw0NDg7dE3I13FW7+VQ7vfsDy39gxCtln8\ned5GwiEdVVOYMXssRSOykgolK4og1aWx6+VXqVm4EDNivS1OO3sKA2/9KqbH0+vFlY+G432/jxZp\nd98i7e57+qvtPWl3VlZKD1klOZGIn4Or6sntsSzKrR7cDgRuvAc3vg7u4WgNUVZFx4mcNFVh+uQh\nTBk/kLdX7WPZF+WEQjplpTW4d2mcMrYEbVAh6+sWEjEa8QVWsf7gHrY3XoCqpGFTBPluB4UeJwUp\nTgo9TjLsWo/PjRVCROff9mwkmGkamKYOho5p6phmJG47Em2L3zaspaGDqWOaBmAgMHC5bfi8AQxD\nB9OM7TNNA0wDExNa100DSzC37ktsxzRpFd9mbN2M20d0vMP0a32ZgtnueCzb4sZThMAwWs+ddIfa\n37AO95vtthNXOz7mcGOaHb4+MBMWYFqe2rjrlPQ/vP4wTS0hdMP6OxUKaJqCogg+31DF//7kYuw2\nlZKhmUw9o4CPVpcnCdzS7TUIIZh2XjEAF04Zwtvv72Ld5oOcfdpgQOD05Pb6tXRZ4P7617/mtttu\n4/7772fSJMsD+Mtf/pLMzEwef/xx3nzzzU6PHzZsGH/9618T2srKypg5c2ZCW3FxMfv27Utos/6j\nd+0/i2maVkhKD2EYZqfldnoDb90GavcuAEw0RxZ1vktZ8VYZpgkuj43Lr51AXn5ah3a1bNzIrr/9\nlUCV9V5Hy8wi9+ZbSJl0OkCfX0t3OR73uyeQdvct0u6+p7/a3l/tlvQ+8XNwE0KUo7pCiSaZavXg\ndjQHt7VEEICtCx5cPebB7TxTcarbzg0XlzBjciFvf7qP99dW4AtGWLW2EmWdYNLoKwgP2sCOls3o\nehVe75s4HJMxbWPZ2xJgb0sg5lLzaCqD3HbyXA4GuuzkuR3kOu3Yu2BvXyNaQ4QV2zGNczjP84nO\nl8Hurj7P9wydn+t43m/T0NHD3Zjg3g5FgYx0Dw2N3uSw8HaotrSuZUc34ZrZHn77pzd57i/rMAyD\nBx98CHf+SDRtOWdc8qtY17E7X2f58vcpnPhQwhCfbHuF4SNEQvuwkc20iBIKJ96Oqgqyc9J73dnW\nZYG7efNmHnnkkaT2OXPmJAnXjpgyZQqhUIi//OUv3HjjjSxYsICampqYp7aVq6++mjlz5vDBBx9w\n/vnn8/LLLxMMBjnrrLO6amq/prn6c+rLFwGgOQawu2IqG9dYYjV7gIfLr5tAanpyNulgRTnVr7+K\nb9NGq0FRyLxkGtlXX4PyJc4+LZFIJBJJR8QeaIVJa8JkkzbhqyitArj9HNy2MSJxIcpd8eBGjuDB\nbU96ioMbLxnBZWcNYdkX5Xyw9gAt/jBrttTDlkLyitIJDlxL0PQTCH5CqrKHkuyLaY5kUukLEjFN\nvBGdnU1+djb5Y+MKIMthI8dpI9tpJ9thI9tpI9thI8NhS8oULZH0FH2babvzc4nofG3r03cC1zR0\nKrc+hx5qOKZxKrrYT7VnkD/m3iOKXMM0cLrc/OIXj3LuuRfw2Wef8vOfP8Sjjz6Bw+FION7pchEI\nBpPGDAQCOJ2uxL5OF8FgCKGoiD7Ki9FlgZuens6BAwcoKipKaN+8eTNZWVmHOaoNu93O73//e372\ns5/xxBNPUFRUxHPPPYfb7eanP/0pAA8//DBjx47lueee4/HHH2fu3LkUFxczb968hFJCJyOmadJU\ntZzGqg8BUB2DWP3FeCrLrSyGRcOzufSqMdgdif9kkcYGahf8g8aPlsdCQ1JHjSTn5luxDe54fq5E\nIpFIJF92dKPVQ9vmwTXjHoiFiAZndhKiHIlPMtUND67azVqzGSkOvnL+cK6cMpSVm6tY8tl+Kmt9\nHNybBhVTcAzdgpJVSbX/ADXlLzM1/0xuGn8ZLbpGhTfIQX+QKn+IKl+QgG5gArXBMLXBMDT6Es6l\nCMiw20i3a2Q6NAamu7HrJmk2lfRou0tVZEkgieQk48MPl1Fauol77/0OAFOnnsvUqefx4ovPEwol\nJpYLBgO4XO6kMZxOJ8FgR31dSX17ky4L3Jtuuomf/vSnPPDAAwBs27YtlgzqG9/4RpfGGD16NH//\n+9+T2h9++OGE7XPPPTfJs3syYxo6dfvfwlu33mpQB/PesuF4W6w/kElThjD5vOKEbKCRxkbqF79N\nwwfLYtmRbTkDyL3+eopmXEx9vbdfhbdIJBKJ5MjU19fz/PPPs2nTJsLh5DQ6Hf3GSjpGj5uDG/Pg\nxmU3bhW4nSWZig9R7lKSqWgWZa2LHtz2OGwqF546mAsm5rN9fwMrNlTy2bZDBHdOREkbjK1oC4rL\ny8cHVrO6ch0XF57P9KHn49SsJJ2madIUjlDlC3HIH6I2GLKEbiBMY8hK1mSYUBcMUxcMU9YM1DQn\n2aEK8GgaKTYVj6bGlh6bSkp06VJVnJqCU1VxqQo2RUhRLJEAQlHJH3MvkWMIUVYVQUaGm4YGX0Iu\ngI7QbOldClE+eLCKUFRTxM6jqowcOZoNG9ZRVVXFwIEDAdi3by/FxcVJYxQVFfPmm68ntO3bt5dp\n0y474vl7ki4L3DvvvBOPx8Ojjz6K3+/nvvvuIycnh7vvvpuvf/3rvWjiyY0RCVBd9hrBlj0A+MND\n+eDdAgwT7A6VS2aOYeiItvT+kcYG6t55m8YP348JW8XtJuvKWWRcfCk2pyy0LpFIJCcr//Vf/8XG\njRu56qqrSEmRya+OhYQQ5ah4NeKEZ2vIYmuIciTaXT3MHFytO0mmuunBbY8QglFDMhk1JJObp43k\ns62H+GRjJTs2ZaHm7UUbvJOwGmLxvqUs2fMRY11ncuXI8ynMSY96YW2MapdhPGwYlrANWOK2MRSh\nKRzBa5jU+II0hyK0Xq1uQlPY2t9VFEFM7DpVJSp+FexK9KMKbHHrdsUSxbFltK8mBKoi0IRAUwSq\nEKiir0NfJZJjQygqNseRI2APh6oKHO4UbIEWlB5yaJ155tnMn/87Fi78F1dcMYt1675g+fIPePrp\n56iqOsD8+b/lwQd/TFnZLpYsWcxjjz2VNMbpp59JOBzijTf+zuzZ1/HOOwupq6tj8uTOkxH3NF0W\nuAcOHOCmm27illtuwefzoes6qamp6LrOxo0bmTBhQm/aeVISDtRSXfYqkUANABUHh7NuXT4gyMlN\nYfo140jPtFz6oapK6pe8S9MnKzCjb+0Vl4uMS6eTeel01JM8hFsikUgksGrVKl566SUmTpx4vE3p\n9+ixekAGaoce3Fi9oGj/aJKpBA/u0YUoa6JbVRo7xeXQOH9iPudPzKehJciabaNZvX0Me8y1qLn7\nMJQgm4Ir2PjFauwNwxibOokxBXmMLMwgN8MVE4Y2RSHP5SDP1ZYZOT4JTyhi0BKO0BiK0BLW8UZ0\nWsJ6dD2CN6LjjW3rSel9DBN8ER1fpAczgcbRXvjaNRVhmlZ7dJ8qQEGgCFBEdIloW09oa93uqM3a\nbv1TEETncxItnYtVf7Vt3doRq3orovshbgyBpgpSwxFaWgKYBknjdVKONnm7neAXh+lHnA1tfTs+\nNqnEbhRVFbSoCo3eQFuZrSPa14EhfYyqCIJ2lQZ/EKN3/ix7DVUVBFsCNPpDRxWx2dHtT8sfwvd/\n8kv+9qff8+RTj5OTm8f93/8x2UUl/Me3H2TeU//D7Gsux+l089U77iV32Eiq/SE+fG8xb77yEk//\n4WUAHvrl/zLvqf9h3vxnGZQ/mP/6+X/TgkqLP4RdU8jsg0RjXf6GveSSS/j444/JyspKKO2zd+9e\nbr31VtavX98rBp6s+Bq2Ubv3n5hGENMUbN4ykr378wCYcMZgzr5gGKqm4NtSSv2SxXg3tN1fxe0m\nc9oMMi65FNUtha1EIpF8WcjOzsbh6NnSLF9W2soEtSWZMmgTqYponaPbSZmgboco94wH93BkpDi4\n5PQCLjm9gCbvGXy6s4zlVcup03YibCHCA7ayTt/Bmk2DiSwbQqqaxYiCdEoGpzMkL5WivBTczo4z\nGKtCxLy/R8I0TUKGiT+iE9AN/LpBUNfxR4zYdiC6L2QYhA2TkGEQ0k3ChkHIMAnF7evK43DENIno\nJrHZf6Gue5clEkmU1EKG3P8wrVl8VgIrN+0FQLvmDk675g4ANgIbo+3kjab4P3/Fb1q3sZF3x0Pk\nRbcWGbAotg8ubchl2sCMXr2MTgXuK6+8wu9+9zvA+rKaNWtW0hshr9fLiBEjes/CkwzTNGis/ICm\ngysACEfsfP7FaOrqM/Ck2LnoytEMHuSiaeVHNCx7j1D5/tixWlYWGRdfSvr5F6K6kyd2SyQSieTk\n5r777uMXv/gFDz30EEVFRdhsiWLDbrcfJ8v6H5EEgRtNMhXnwaWdB7e1TFB8iHJY714W5ViSqaOc\ng9sd0jx2pk0cxbSJo6jx1fGv7ctYV/cFuhpBG7gPbeA+Ak2ZrKsuZM32PDAtmwZkOKNiN5XCvBTG\nDAe7MDhSRtp4hBA4VIGjB0oRmaZJxDQJ6ZYI1k2TiGG16a3Ldm2GAIfTTqM3QDhitLUDhmlan9h6\nuyXt2tpvxx1r0prfs23djG7F1uP2G2Zbpdr4/Wbc8QjrmtvGkkhOLvqiQlmnAve6667D5XJhGAY/\n+tGPuOuuu0hNTY3tF0Lgdrs5++yze93Qk4FIqJHavf8k2GK9xWhoSGXNurEEgg6Gjx7A5DEOgh8t\nYNenn2IGA7HjHEOLyZw+g9TTzkBoPRfWJJFIJJL+xRNPPEFDQwPXXntth/u3bNnSxxb1X6ywPjM6\nB9dqi5+Dq7Sbg9tWJig5RFlVrFDWI9HqwT1SHdyeJsedxTdPvQ5v+Ao+qljJ8vKVNIaaUNPqUdPq\nYaiNSG0eeu0gqhuyqG4IsGZbdex4VRHkZbkZlOVmYLabvEw3OelOctKdZKY5UJXee2IVQmATApsC\nHrp23062erJmVFS3NSQsaJPNybSPBm3fJ8E/njRux9vx45qYqKogI8NDQ4M3IWz/yDYc338bRWmz\nuz/9nQAocffc6Kbtx/NK7ZrC0IEZx7cOrs1mY/bs2QAUFBRw2mmnoUmBdVR46zdRt28hpmEFz+zd\nN4jSrcNxeZycXxQgZcNrHHqrzX2PqpIy6XQyL5mGs6REJk+QSCQSCU888cTxNuGkQTcMiIrYmAc3\nQai1hignZlHWOghR7kp4smmaferB7QiPzc1lQy9h2pALKa3bxscHPmVTzVZMNYyWW46WW45DuEkJ\nFhGozqGu0o1hCHTD5ECNlwM13qQxFSHITLWTne6yBG+qg4wUB+keO2keO+kpdtI9dpx2+fx4tAgh\nEqV90iPh8X1GVFVBmsNGxKahK/1HKKqqIMtlh6Ocx3o8abVd9DPbuxLp0hN0+m3zxBNPcM899+By\nuVixYgUrVqw4bN/vfve7PW7cyYAe9lJfsRhf/SYAQmGNjZtHcPDgAIY56ijcshR1nS82Z8SWN5D0\n8y8gbeo5aKlpx89wiUQikZxwTJ48+XibcNKgG2YsDDlWJkhtC/luzaKcVAdXSfbgdimDshk3X7eP\nPbjtURWVCTljmZAzlvpAA58dXMuag+spbzlA0PQRtG+BwZA+xMHIrJFk6AXYfHnU1UFlrZfqhgAt\nfivhpWGa1DYFqW0Ksn3/4c/psKmkp9hJddvwOG14nJq1dNlwOzVSnDY8LqvN5dBw2lWcdhWHXe1V\nD7FEIjn56FTgrl27lnA4jMvlYu3atYftJ72LyZimibduHfXlSzANK9y4uiaD9ZtG4W5u4syqf5Ea\nqgNAaBopp59J+vkX4Bo5St5PiUQikcS48cYbef7550lLS2POnDmd/kbIOrhdR9fNmAdXjXpwDTX+\nsSjRgxuJhSi39WgtE6R2owauNcaJ483MdGYwvegiphddRJX3EGsOrWftoQ1Ueg8S0INsqN6IlVIG\nBubkUjJiGJdnDKPQPYRIwE5NY4DapgA1jdanoTlIozdIY0uIUKRN1AfDOofq/Ryq93fbRk1VEgSv\n067itKk47Bp2m4JNVbBpClp06bCppKc5CYciKIqI7Y99VAVFEaiKYoWXK9Esy9GPoghUVYmFnqtq\nW3tXQtElEsnxpdNv2L/85S8drks6J+SronrvIvRAOQDhiMq27cUc2uVmVM0Kcnz7EYBz2HDSppxD\n6pmTUWU9Q4lEIpF0wLnnnhtLJnXeeecdZ2tOHiwPbmuIstWW4MFtnfXYSRblSCxEuSvzb+ME7gnq\nkRzoyeXK4mlcWTyNWn8dpfXb2Na4g00HtxI2IlT5DlHlO8SKilUA5LiyKUotYEhOAacWF1CYWoJL\ncwLWi/5ASKfRG6KxJRhdhmjxh/EGwngDEbz+6DIQxusP4wtEOpwfGNENWvxGzGt8PBGCqBBWUJS4\nckDR8kFCHH47qS12bGt/gd2mWqHvAhTa+ibaIBLsgfhSPqJtO64EUXzfWFeSnVTxmyJ+rITxEs8j\nBDgcNkKhcGy+bWvZpD7jKE4mhMBh1wiGIpjdKF3Tt684Oj6bUGiz3eiwS/fO0kcXZdMUZl80gix3\n777k63T0zkKS4xFCcM455/SIQf2ZSKiJ6j1LCLVsjv2hVFblsGNTPoOqtnJ243bMDA/NZ0yiefxQ\nglkpOFQ7zqYtZATTyXUPINOZjiJOzB8+iUQikfQ99913X2x95syZDB069PgZcxJhzcGNemBbMyqr\n8Vmp25cJslo7qoPbnRJBcGJ5cA9HtiuLC1Om8pWJ06mqrmNH3R52NuxmR8Nu9jTtJ2JEqPHXUuOv\nZc2htlKGue4c8j0DGejJY5A7l4GePIoHD8CuHrm8kGGa+IMR/MEIwZBOIKwTCOkEo59AKEIg3Lpu\nfcIR3cqUrJux9bBupU4KBCNxbQbhiBl7KXG0mKb17x7R+1nhVInkBCEQNvjW7HG9eo5Ov2Fvv/32\nLg0ihPhSZ26MhFuo3PkBum8dimIgBHi9TraXFuDZtp/xvrfYPURj1ZlpHBhgA1EBDRXQkDyWS3Mx\nKnM4Y7NHcVruxNibUIlEIpFILr/8csaOHcusWbO44ooryM3NPd4m9Vt0w4zNs1VbPbiaJcIURRAf\nomxGS9FA+zm4XU8yFe/BPd5zcLuLXbUzOmsEo7OsspBhPcyepn2UNe1jX3MF+5rKqQ1Y064O+Wo4\n5KuB6k2x4wWCbGcmOa5ssl1Z5DizyHJlku3MIseVRYrNE/NgWvNzjyyGO6OzLMqGaaLrhlU2yLDK\nCxmGia5b/8bWenS/GW03zFh/a92IrbeOaZpt5X3ab5vREkNJ2yRuC8DptOH1hdANM9qemNo4djVx\nWYyTshTH7TvcsWb7zMtJfdr2m+06tTMJgeXBDQTDlifUpFcz9XbD2dopyV7Q3rG6N0aNeZ+D4R67\nH/H01r+fpgq+cvHIXho97jyd7dyyZYucD9oJoUAj+za8jarsRFUNFAVCIY2yHYNgw0Hszo/YPM7G\nroJMdM26j6n2FFyaE5tiQxUKQT2EL+KnOWSly/ZH/Kyr3sS66k28sf1fTB50OlcMvZR0h0w4JZFI\nJF92li5dyttvv81bb73FY489xmmnncasWbOYMWMG6enpx9u8fkV8iLIaFUFGnMA1oxmPESpG3NNe\nhyHKypGflfQED27/Erjtsak2RmQOZ0Tm8FhbS9jL/qYK9jdXUOk7SJX3IFXeQ4SMMCYmNYE6agJ1\nUN/BeIpGmj2NNHsq6Y5U0uxpsWWaPYVUewpuzY3H5sKpOY8p0k0RAkVTOTYJ3TucbOWNTnT6q93Q\nf22Pt7s36VTgTp06lTPPPJNzzjmHqVOnUlhY2KvG9Beqdm+icucHZGbXY7dZf1ThiErl7kyattRS\nMeALtk7OpMUYhelLxdiZihl0Y4acDBqUwVlj8jj3lEG4HG23P6iHOOSrpqxxL1vqdlBau5WQEWZF\nxSpWV67hymHTubjwPBm+LJFIJF9iBg8ezO23387tt9/O/v37Wbx4MW+++SaPPPII55xzDs8999zx\nNrHfYCWZag1Rttpa5+CqmkLrxDYhFCJxLpKOQpS7lmSqTeD2Nw9uV0ixeRiTPZIx2W3eGcM0qA80\nUuWzxG5toJ5afx21gTpq/XWEDGtObdiIWG1RL3BnCARuzYXb5sJtc+PR3LhtLhyqA4dqx6E6cNkc\nZNWloQdNbKKt3ak5sCkamqJhU2yxdflsJZEkUlq6iR/+8AEWLHgHgKamJn7964f54ovP8HhS+OY3\n72DmTKuUrGmazJ//O956659EIjqXXXYl998/F1VN/p5ramriJz95kJUrVyaN05N0KnAXL17MqlWr\n+Pjjj/nDH/6AYRicc845TJkyhSlTppCRkdHjBp2oHKrcz8YV/yI320t6ZoDsAVZ7KKRRuSuF/Qca\n2JzaQvWQEYjIVESd0wq30U3shoEvaH2J7z7QxO4DTSxYUcbs84q5+LQCFEXgUO0Upg6mMHUw5xdM\npSXk5ZPK1SzZ+wG+iJ9/7FzIltrt/Mf4W3HbXMfxTkgkEonkRCAzM5MBAwaQl5fH9u3bOXTo0PE2\nqV8RXwc35sFVrMciVVMSPLiROBeu1kGIsk3rbhblk0/gdoQiFLJdmWS7MhmXPTphn2matIS91Pjr\naAg20hhqoinYbC1DzbH1lpC3LdQWK7TWG/HhjfjAX9tjdtqioleLCWAtJoBVoaIIBUWbyTXUAAAg\nAElEQVQoqEJFja5bH6uMkRpdt/oo7Y5REEKJJXWykkslL1VFkFLnxOcLgSFAgECxnidjiaKsdSvC\nUiRvR6+powjMjtI+tfWLS1zVvn984ql2YwgEiipICztpbg5gGGaHYxFn/+HsStzV+xGkqgI1hpum\nJh/dnZp9vONbFUVQY7hoavJH73k8R2+daZosX7KMv/3hjyiqSlnjPgCe/tX/YLc7ePqvL7C/bC+P\n/79f4BjgoWT0KJb8exEfrnifXzzzOCB44ue/4tk//ZaZ112TNP5vf/04uVnZLFy4hO3bt/PAA99h\n6NDhjB8/4aht7ohOBW5aWhrTp09n+vTpAJSXl/PJJ5/wzjvv8PDDDzNo0CCmTp0a+5xMvP/eYuoO\n7MYtAuRkRsgc4GdESdsfkLfFSeUelXXNGrV5RQyeNJxxKTqqCl4M6kw7dbS9AMgyTMapDpqrvKze\nchBfMMLflu7gs62HuPeaCaR57AnnT7F7mF50Eefmn8UbO/7Np1Vr2Fq/g9988Rz3nXoH6Y7UPrsX\nEolEIjkxaGlpYenSpbzzzjt88skn5OfnM3PmTObOnUtxcfHxNq9fEV8HtzXJVMyDq8Z5cFGIJNSw\nbXt4DIWtdnuXBG5ciPJJ6MHtLkIIUqPhx51hmAa+iB9f2Ic37McX8eEN+/CF/Xgjvlh7SA8S1EME\n9SBBI8T/Z+/Ow6Mqz8aPf88ye/aFBEhIQlBZXRAVXHCpiCJC9UVttUpbFVFb++K+VIuKfa2KFrUW\ntVqrUrH+aosCRVELbrigouxrIMQQsm+zn+X3x5nMTMgKJAPR53NdrcyZk3OeM+HizH3u+7mfsBHC\nFw4Q1EOtHi50dA7rZ0M9eYmC0OfsXbmT+nWVpJ/Sn8qPSnn0y6fQgxrrVn3G0BvH8sTaZwFwDE/j\nqYVPkXfBUWx9azUZJwzgLyV/B8A43sW/F/+TTUXftTq2HtRY98kqfvnor3A4HAwfPpIJEyaybNmS\nxAa4+8rLy+OSSy7hkksuwTRN1q9fzyeffMKzzz77vQpw//Xa8xxbXE7hsNZPRHRDomavm5IKG2Vy\nGjmBBvKbGznyu/VgptKclYMrS6c4dxf9suvwmU42mcWsZRR+JL4xQ4wekcH/nT6YN1ZsZ9X6vWwt\na+CBv33BrZeNpl9a28ys2+bmyuGXUpxWyMLN/6LcW8Gfv32B/z1uJk7VkaiPRBAEQTgMjBs3jvT0\ndM477zxuuOEGRo3q2S8FPyS6biLJrUuUDVkFM5LBjQRFkqy2yuDa4spZw/uRwY2fg6v+QDK4PUGW\nZJJsHpJsnm7/zL7zE3VDtwJfPUTYCBM2NDRDi/tvbFvL9rARRjN0NCOMbhropo5hGhimgW4aGEY7\n2+L2s15H/mwYmBhWkylaGkOZGJhgmtHXJiayLKHputWoKtrUycCI2weTuNdGpBFUbP/4rkNm9L9t\n52m2tzROe/sJvUFGlrr/d/pgGKaXlqZ5XckY3Z9+4wvw7ox1wg3W+pFkCUdGLE5xZrlp2GBVDQWq\nvTizY9fiyHITrPZhmmarjH3LcYoLB0e3DRpUwMqV/z3QS+tQpwFuSUlJpz/s8XiYMGECEyZM6NFB\nHXKSgpXeNwkGVRpqHVTWqTQ6cphw9nmMSbMaefiDGqs2lPOvtR+RrX7N6WvWYO4u4KusE0nOCHDs\n0ZsZ7V7PcHMrn7insMVn46vqJkwTrpo8nGOGZPH8ko3UNAaZu/Br7vzZ8aQltR+0njLgJFyqixfW\nLWB303e8tPE1rhl5hWgCJgiC8APy7LPPMnbsWPFvfw/Q9LhlgiIlyqasgm4FSKYZC3DDHZQoh8NW\n0NqdADekx9ZwVeTDf5mg7xNFVnDLbtw296EeSqcOx8ZBptm6PLy99xRFIj0jibra5lbLMLXXkZl9\n3o0G4K2C7cRcu6JIpKcnUVe3f5/3wY5ON0ye3riH+lBilppKsytcPyy3VQf4rnyT8jX3//MeHh1/\nH+u+/ZZ7XZt4dPx90feXNS9hyY43eXT8fUycfQa3jv0Vg4uHALCnvJwr/3Qp/zf2buyOWFzTcpxp\nIyZFm0w5HE4CgUAPXWlMp//CnnfeeUiS1OHixy3vfd+WCbrwkp+zdetGdMlk6JgRHf6ldzlUzjpu\nEMcWX8T//SOH10/9gHPX7OaEskq+Ms/lo1XHcdKYDaSl1nNm8A3S03/KZ3UGX9c0keNyMH5YDslu\nO4//4xuq6gPM//c6br3suA4XgB/d72jqj2jgn1vf4puqdXyy53NOGXBSb34UgiAIwiH22muvtXpd\nWlra4b6XXnppbw/neyOsGbBvBleJBLjxGVxJbd1kKj7A3Y8MbkuAKyGJDK7QZ8Q/TGszhzfyUpEl\nVFmxSu/NvtOwS1EkXDYnflVDlxL3QEEzzHbnQ/cWCQmn6upWt/cWdsWOJFnLl6Z4UgmFQrjUWAbX\nCOt43Em4VBdOpxM0Kfa+ZqIoCqme1r2aWo4TLxgM4HL1/IOnTgPc9957r8dP2FcMHTq8222sM1Kc\nzPrxWGb/PcCyEz9k2ooajit/hy/yJ/PZ6uGcMf5bHLZmjg8swZ8xjW9rvbxTVk1xiothBelcPXkY\n8xetZ0tZA0s/LeWCkws7PNeZeaeypW47a6s38MbWJRydNaLL+SuCIAhC3/XMM890az9JkkSAux+s\nADeSgTWsL35GJPBUFRkz0uFXkm2tm0zFfeEPay0BbtcBaygyv9Om2EQGXhB+wFRZYtaoQhpC4a53\n7oCiSKSmemho8HaZfU612/YruN1Xfn4+mqZRUVFBbm4uAKWlu6J9HwoKCikt3cmIESOj7xUWtu0J\n0XKc8vJynM6UNsfpSZ0GuAMHDmyzbcuWLWzbtg3DMBg8eDDDhw/v8UH1RQOyPFwwZgRvbqrk7ZPX\n8bMlNRxR9QWb+43js8+PZPwpazDC9ZyVtoly52CqA2He3FXFtcPyOHFYDpt21bFiTTmLP9nJuBE5\nZKW23ylZkiQuG/o/3LdqBwE9wOKSd/jpURcl+GoFQRCERHn//fcP9RC+l8K6gRQpUVbN1gGu1UW5\n/Tm4rZpMad1vMtWSwbXLh+MKrIIgJJIqS2Q67V3v2AFFkcjwOFCD4V4vZ3e7PZx66nieeeYpbr/9\nt5SUbGf58rd55JF5AEyceB6vvvoyxx9/Aqqq8vLLLzJx4qR2j3Paaaczd+5cbr75DrZta32cntTt\nOoLGxkZmzpzJlClTuO+++5g9ezYXXXQRl112GU1NTT0+sL5owpg8HI1F1CvJfHOUm4GNW0gO1dLU\nnMTeGmtCdbD6U84faHVA3u0NsDaSIZ52RjHJbhthzeCfK3d0ep4UezLnFp4FwCfln1Pj73rdOEEQ\nBOH7oaamhvnz53PHHXdQU1PD0qVL2bp166EeVp8TX6Ks7pPBVZR9S5QjpcwSyO1mcLsT4EYyuCLA\nFQShj7n99t+iaRoXXjiJu+++jeuvvzGasb3wwos59dTTueaa6fzsZ5cwatQxXHrp5QBUVFQwYcJp\nVFRUAHDHHdZxpkxpe5ye1O0A98EHH6SyspIlS5bw2WefsXr1at566y0CgQAPP/xwjw+sL3LaVc4a\nnY+2t5DVw9zoiklR9dcArFnTDyQ7pqmRE1jDUalWvfnKPbWYponbaePC06wg+PONe6ms83V6rjPy\nTiHJ5sEwDVaUfdy7FyYIgiAcFjZs2MDEiRNZsWIFixcvxufz8fHHHzNt2jRWrVq1X8eqrq5m3Lhx\n/Pe/VgfLhoYGbrjhBo4//njOOOMMXn/99ei+pmkyd+5cxo4dywknnMCcOXPQ9cQ0SOktrQNca1tH\nGdyWJlOqJLc9BmBT9iODq4gAVxCEw9vo0WNYsiQ2VTUlJZUHHniI//znfd54YwmTJ0+NvqcoCjNm\nXM+iRctYuvQ9/vd/b0FRrH9Lc3NzWb78w2hpc2pqKvPmzeOdd/7b5jg9qdsB7vvvv8/vfvc7iouL\no9uOOOII7r33Xt55551eGVxfdMrI/ujVAwioNrYMcpLl243H8KJpKvXN1mfXXPM1p+daE68r/CG2\nNVrB7Cmj+pOWZMc04e0vdnd6HptiY3yetTTTJ+WfE9B6vgOZIAiCcHj5v//7P6ZPn87ChQux2axA\n6cEHH+SKK67g0Ucf3a9j3X333dTXx5aCuOeee3C73XzyySc88cQTPProo6xZswaABQsWsGLFCt58\n802WLl3KV199xQsvvNBzF3YIhDU92kVZjcTqRiSAVRQpNgdXipUo7zuPLaxF5vDauv46FdREBlcQ\nBCERuh3gqqqKw9F2CRun00k4fOCTpL9vcjLcFPRLR6/PZn2xEwnIrdsEwPp1VlBraD76GaXkeazP\n86tqq8Tbpsr86Pg8AD5dv5dQuPOn4+MHjkORFAJ6kG+q1vfSFQmCIAiHi/Xr1zNlypQ22y+99FK2\nb9/e7eO8+uqruFwu+vfvD4DX6+Xdd9/lxhtvxOFwcPTRRzN58mT+/e9/A7Bo0SKmT59Ov379yM7O\n5tprr+Vf//pXz1zUIRLWDKSWJlOROWyxDG5sP0mOdVFWpX0DXJHBFQRBONx0O8A9+eSTefjhh1s9\n7a2treWRRx7h5JNP7pXB9VWjBmei1+ZSnm3D65TJabLm1NbX2zAVq3GXr249o7OsDmIb6psJREq9\nTh7ZH0my1tj9emt1p+dJticxPPNIAL7Y+3VvXY4gCIJwmEhNTaW8vLzN9vXr15ORkdGtY5SUlPDX\nv/6V2bNnR7ft2rULVVXJz8+PbisqKmLHDuv+tWPHDoYMGdLqvZKSkg6XEewLwnpcibJmXYeOFeDa\n4nq/SLJKqGU5IKV1gBuMPIi227ruohwUc3AFQRASotsrjd9xxx1Mnz6d008/nbw8K8tYVlZGcXEx\nv//973ttgH3RyKIMFn+WiYnEjjw7o7Z5SaOJepKprc8lM/k7/I3bGTVwCktKIWyYbK73cUxmMunJ\nDkYUZbBuRy2fb9zLScNzOj3XCTnHsbZ6I5tqt9Ic9pKqiCWDBEEQvq9++tOfcu+993LLLbcAsHnz\nZj744AOefPJJfvGLX3T585qmcdttt3H33XeTlhZbo9Dn81lrGcZxOp0EAtb0F7/f3+p9l8uFYRiE\nQqF2q7v2JUkSHSzxvl/kSImwfBBLXrQIa0asRDmSidUjz/2dcZekqDY0rADYocgocUFuIGQFuG6n\n2mp7e+NuaTJlV2yd7ns46cnPO5HEuBNLjDvx+urYEzXubge42dnZvPnmm3zwwQfs2LEDh8PB4MGD\nOfnkk8V6bvsYPCAFFTumL4XdOQFGbQuQVldCffrR7NjuIfNYMI0gSvA7CpJc7Gjys6XByzGZVnfl\n44/MZt2OWjbsrCOsGZ12ZxyROQxFUtBNnU21WzlpwHGJukxBEAQhwWbMmIHH4+Ghhx7C7/fzq1/9\niqysLGbOnMn06dO7/Pmnn36aYcOGcfrpp7fa7nK5CAaDrbYFAgHcbqshotPpbPW+3+/vcOpSezIz\nPT36XSEtzXNQP28YJppuYmspUdbiS5RNkpNViDSeSk9PQ262glOPw0ZGhvUg2TTNaAY3OzMpur0j\nIc0qUU5yubvc93BzsJ/3oSLGnVhi3InXV8fe2+PudoAL1jzcM888k1NPPTW6rWX+rd1+4Gs5fd+o\nikx+v2TKmtL5rl8dAJneMnamH03lXlDs2eihKvyN2zky9fhIgOvDME1kSeLo4ixgM8Gwzpbd9Ywo\n6rjszKk6KE4tZEv9djbUbBYBriAIwveMYRjIcenPyy+/nMsvvxyfz4eu6yQnWw9Hq6qqyM7O7vRY\nS5cupaqqiqVLlwLQ3NzMTTfdxNVXX004HKa8vJwBAwYAVilzS1lycXExJSUlHHPMMdH3Bg8e3O1r\nqKnx9lgGNy3NQ329F8M48PLolsAUxfqvHMnghqzGyehGrHFjY5NGg9cK7mXDpDayvF8gpNFSoa2F\nwtHtHY3bp/kjx1A63fdw0lOfd6KJcSeWGHfi9dWx9+S4O3tQ2O0A9/PPP+e+++5j586dGIYR3W6a\nJpIksXHjxoMa5PdNUf9kSnem4stVqE9WSGmqRpZMDFMipOegUEXIu5sj805jWRl4NZ0Kf4gBbgfp\nyQ4G9UuitLKZtTtqOg1wAYZnHsWW+u1srN3Sp+dDCYIgCG395je/4fHHH0dVW9+yW7KrYAWu9913\nH5999lmnx1q2bFmr12eddRb33HMPZ555Jps2bWLu3LnMmTOHrVu3snjxYp599lkApkyZwvPPP8/Y\nsWNRVZVnnnmGqVO7v7yDaZr05KpChmGi6wd+v/P6I5FsJIOrRALeaNyrGBD5s4mdoO4FwCZJ0fNG\nj4HVZKqr8fjDVtDslJ0HNfZD4WA/70NFjDuxxLgTr6+OvbfH3e0A95577mHIkCHcfvvtbebpCG0V\n5CZjbLCaSO3NUElrCpIi+6nX3dQ3pJKZBEFfOXkOGbcq49MMdjf7GeC2yr2GFqRTWtnMtu8aujzX\nUenWE/bGUBPV/loySe69CxMEQRAS6vPPP+eGG27gySefbFMt1dDQwOzZs/nPf/5z0A0fH3jgAX73\nu99x+umn43a7ufXWW6MZ28suu4zq6mqmTZtGOBzmggsu6Nac38NVIJKqlRQNyTCRI02ktMgXLlXR\nowGuJNsIRt63x3VLbpl/C+B0dP11Khrgqt0r6xYEQRAOTLcD3MrKSubPn09RUVFvjud7o3+GBzPg\nxjRkqtNUjtoVJMVfSb29kPLvXGQeBZg64cBe8j1ONjf42N0c4KR+1s8PGZjKO1/sZldFE6Gw3mmH\nxoFJ/bHJNsJGmJKGUo7KK0jMRQqCIAi97qWXXuLqq6/m2muv5emnn8blcgGwYsUKfvvb3xIMBnng\ngQe4+OKL9/vY77//fvTPaWlpzJs3r939FEVh1qxZzJo168Au4jATCEaCU1lHjQS1JhBZ1hZV1SEE\nkmxHkiRCkco1uxwf4MYyuE57112UfWGrRNmpiiSBIAhCb+r2jJgJEyawcuXK3hzL90puphuQMX1J\nVKVbzxGS6ssAqNijI6tWaVnIv5f8JOvLSqk3NudnSF4qALphsrOiqdNzKbLCoGSrs3VJw64evQ5B\nEATh0DrqqKP4+9//zu7du7nqqqvYu3cvd911FzNnzmTUqFEsXrz4gILbH7JYBlePNpjSpdgzfyUy\nN1eSrYx5KJrBjeugHIxlcF3dCHCjGVxFBLiCIBzeNmxYx9Sp50ZfV1bu5c47b2bSpB8xZcpEHn/8\nYUIhq/meaZrMn/8Ukyefzbnnnskf//goegdzUhobG7nhhhs4++zxXHTR+Sxe/O9eGX+3M7g33XQT\nU6ZMYfHixeTn57dqeAEwd+7cHh9cX5bkspHkshEMeKhOsxpNuYPWGsKhoI5sy8bQdhH2V5KXMhSA\nmkCYkG5gV2TSkqy5uHVNQUr3NnFkflqH5wIoTM1ne0MJOxt39+6FCYIgCAmXn5/PggULuOqqqzjr\nrLNITU3l0UcfZfLkyYd6aH2SPxTL4DpCkQA3bn1aRdbQAVmxAtxAJMB1xpcoRybsKrKEqnSdL/Bp\nokRZEITDm2maLFnyJk899TiKEntwd//99zB4cDH/+tdSmpubuPPOW3jxxb8wY8b1vPHGP1i16iNe\nfHEhkgS33TaLhQtf4fLL23b2f+ihOSQne1iyZDlbtmzhllt+Q2FhMSNHjurR6+h2Bvfuu+9GkiTy\n8vJwOp3Y7fZW/+uODRs2MG3aNI499limTp3KmjVrOt1/1apVDB06FK/X291hHlZyM9yYQTdel4ym\nynhCsfm0mmEFrOHAXvo5rc/PBKoCoeg++f2s7mBlVV1f/0BPfwD2eCtFoylBEITvoZycHBYsWMCI\nESPIzs4+6Dm3P2QtGVxZ1XCEWwLc2DN/WY5keCMZXH+ky7Ir7gtfyzGcdqXLJZAM0yCoWZ2YXaJE\nWRB+8DTdoLLOd8D/21vnY0+1l73d2FfTja4HFPHSSy/w+usLufLKX0a3hcNhXC4X06dfhcPhIDMz\ni3POOZd1674FYNmypVx88U/JysoiMzOLK674Of/5z+I2x/b5fHzwwQpuvPFGHA4Hw4ePZMKEiSxb\ntuTgP9B9dDuDu3r1al555RVGjTqwCDsYDDJz5kxmzpzJxRdfzKJFi7juuut499138XjaroXU0NDA\nXXfd1aeDtew0FyV7XSBJNCYpZNSHcdkM/GEZvz8ZlwxhfyXZNgWHIhPUDfb6Qwz0WDe/gdkevt1e\nQ1lV18sJ9E/KASCgBajx1yEjlm0SBEH4Pvjoo49avb7qqqt44IEHuOKKK7jttttaPWWPX8ZP6Fi0\nQZSs4QhZX/50OXbflIgsC6RYU4j8kXI7l9q2yVR35t8GtNgawqJEWRB+2DTd4K5nP6W6IdD1zj0g\nK9XJ72eM7ValyfnnT+XKK3/J119/Gd1ms9l45JHW/Rk+/vhDhgw5AoDS0p0UFsaWjRs0qIDS0l3R\nlXZalJWVoigq+fn50aXSBg0qYOXK/x7U9bWn2wFuQUFBtNb6QHz66afIssxll10GwLRp0/jb3/7G\nypUrmTRpUpv9Z8+ezaRJk/jLX/5ywOc81DJSHJil1lzbuiSJjHpIkgL4cdPQ4MSVDoYeACNIjtNO\nqTdApT8ug5vdksFtjq6R25Fcdw4SEiYmuxvKKXAU9uq1CYIgCIlx9dVXt7u9urqaa6+9NvpaLNnX\nfYGgDpIBSlyJsjPuYbtpffFUVBe6YRKKrNcYn8H1BawMrqsbHZQDWuyLrEuUKAuCcJjKysrq9H3T\nNJk371F27drJvfc+AEAgEGi1wo7D4cQwDEKhEA5H7N87vz/Q6nXLvoFAzwf63Q5wr7vuOu644w6u\nuOIKBg0a1GY9vq6eGpeUlFBcXNxqW1FRETt27Giz75tvvkljYyM333xznw5w05IcGAErwG3wWDdF\nV7gJcFPfoJKbbu2nherp54oEuHElygOyrJttKGzQ0BwiPbnjm6JdsZHlyqDKX0NZQwUF/Qp75ZoE\nQRCExNq0adOhHsL3TiCkgRIGiJUou6wl9hRVBjOWwfXHNUuJz+A2+6yfT3Z3XTHl0/zRP4suyoLw\nw6YqMr+fMZbaxgMP7GRFIi3VQ32DF6OL9WQzUpzdyt52JRgM8MAD97J9+zaeeupZ0tMzACtIDQaD\nrfZTFKVNMOt0OgmFgm2O6XK56WndDnBblgb4/e9/3+a97jw19vl80aUNWjidbaP28vJy5s2bx9//\n/nfC4XB3h9dqLPLB/w6RZanVfw9EVqoTwg5MU6Ih2QpwHb46cOXQUCchFSmYpo6h1ZPpskqMG0Jh\nlEiXxpyM2C+8ptFPVlrnN8V+7myq/DXs9VYd1LgPhZ74vA8FMe7EEuNOvL469r46biExAiEdSY0E\nuJESZc1hVU05XSpGJCCVVSc+LTZ/zaXGMrhNkYqrJFesOVVHmkKxqUZJtrbTsgRB+GFRFZl+6Qce\n2CmKREaGB4dsoncR4PaExsYGbr7517hcbp555q+kpKRG3ysoKKS0dCcjRowEoLR0F4WFbZeVzc/P\nR9M0ysvLcTpTovv2xhK03Q5wD/YJssvlahPMBgIB3O7YL9cwDG6//XZmzZpFTk4OZWVl+32ezExP\nl80e9kda2oHfiArywoAEYTtNbuva7c014ILmphB2VwZBXxV2xU9eRjLsrqE+pJORYd1kM7CeDDf5\nQvjCZnR7Rwam9WN9zSaqvLUHNe5DSYw7scS4E6uvjhv67tj76riF3uUPatEA1x7J4Gp2N5jgdNkw\n9EiAq7gIxGdwlbYZ3O4EuM0hq1mkS3Wiyt3+6iUIgnDImabJXXfdSkZGJg8++EibKt6JE8/j1Vdf\n5vjjT0BVVV5++UUmTmw7/dTt9nDaaaczd+5cbr75DrZt287y5W+3md/bE7r9r+zQoUM7DBxtNhs5\nOTlMmjSJG2+8sVXDixaDBw/mlVdeabWtpKSk1RIHFRUVfPPNN2zcuJHZs2djRBZWP/3005k/fz5j\nxozpcpw1Nd4ey+CmpXmor/diGAf2ZESOjN8MO/C5rEE5go0ABPxhkJOBKhrq9mJLHw6AX9P5rrIh\n+pQ4K9VBky9ESVk9tcUZnZ7PI1sBcJW35qDGfSj0xOd9KIhxJ5YYd+L11bH35Li7ergo9D1NvjAo\n1hzaljm4YdUF4UiAG83guqIdlCXAER/g+rsf4DaFrQA32S7+LgmC0LesW/cta9Z8hd3u4Lzzzoxu\nP/LIofzpT89x4YUXU1tbyzXXTCccDnPOOedx6aWXA1Zsd8UVF/Pyy6+Tm5vLHXf8lj/+8WGmTJmE\ny+Xi+utvjGZ+e1K3A9z777+fefPm8etf/5pjjz0WgLVr1/LEE09wySWXUFxczNNPPw3EypnjjRs3\njlAoxMsvv8xPfvITFi1aRHV1dau5uwMGDODbb7+Nvi4rK+NHP/oRK1eubLfTcntM06SDtYUPiGEc\neOrfZbc+XjPswOu0bopOLbbkj25a833CwXpSbbGHAjX+MP3d1v5ZqS5K9jRRWefrchwZDisArvLW\noOsGRve7gh82DubzPpTEuBNLjDvx+urY++q4hd7V7A9HM7hJYevhfVhxRgJcFT0ug+vVYh2U45s9\nRgNcd/dLlEV5siAIfcHo0WNYsuQ9AEaNOoaPPlrd4b6KojBjxvXMmHF9m/dyc3NZvvzD6OvU1FTm\nzZtHbW1zr96bux3gPv/88zz44IOcccYZ0W1Dhw4lNzeXBx98kGXLlpGTk8NNN93UboBrt9t57rnn\nmD17No899hgFBQX8+c9/xu12c++99wJWEP19YlNlHHYFI+TAl2wFrHY91mhCN6w5tXq4mWSbiiyB\nYUJ9MEx/tzUxu2XebVU3WolnOq2uVUE9RFPYi0cRN1JBEARB2FeTPww2K0D1RHqehGXrvut0S2Ba\nQa2sumjyW5neZFvrr0wtAW5yt0qUrQBXZHAFQRB6X7cD3MrKSvLz89ts79+/P8rItDwAACAASURB\nVHv27AGsKL2hoaHDYwwdOpSFCxe22d5RYJuXl8fmzZu7O8TDUpLTRkPYjqFIBJ0qjoCGqoCmQzjs\nQAUMrRlZkki1q9QFNeoji8cDpCdZN9z6pmAHZ4jJiAS4ALX+OjxJIsAVBEEQhH01+0JImVaTKLff\nCmbDkhWoul2xBpeKLZmmsPV+fIAb1nS8kWWCkj1dd1FuFAGuIAhCwnR7turo0aN55JFHWgWwjY2N\nzJ07l9GjRwOwfPlyCgsLe3yQfVmS24apWTc/vyvSSVmxaoeDQWu7HvZimibJkUnbzeFYjXVaJMBt\n8IYwzc5T+R6bG1myfqWNoaYevApBEARB+H7QDQNvQEOyWQ+OXZEA169b92i3Oy7AVZNoCrVkcGNT\nieqaY0v6ZXSyhF+Lhkj/jRR78kGOXhAEQehKtzO4DzzwADNmzGD8+PEMGjQIwzAoKyujqKiIp556\nig8++IDHHnuMJ554ojfH2+ckuWzQZD0V9tsl0gCHrONFJhCwYU0tNjB0P57IzbNZi2VwU5OsIDis\nGfiDGm5nx6VQsiSTbE+iIdhIQ1AEuIIgCIKwL2+k5FiyBVF0EzWkYQL+SLMplzMEIZAUB7Jipync\ntkS5Lm79ypYH0Z2pC1jJgQxnWk9dhiAIgtCBbge4AwYMYNGiRaxatYotW7agqipHHHEE48aNA8Dt\ndrNixQoyMjrv9PtDk+yyYdZZH7PPZt08HYQBG16vQmakilgPN5PUEuDGZXBT426c9c2hTgNcgFR7\nMg3BRppEBlcQBEEQ2mjwWtlXyR7EHbAqqsKyI9qY0W4PoYes8mQgrkQ5LoMbmTbkcqi4HJ1/lQob\nGg0hK4ObLgJcQRCEXrdfi7EpisKpp57aqvNxCxHYts/jsoFuBaUBu9V90W4EADfN3liFuKF5SVKt\nRY+98SXKcXN7GpqDDMjqfF5tiiMZmqBBBLiCIAiC0EZdk5V9lWxBPA2RKUNq7N5qUwPoWOXJhmlG\n+2Kk2m1xx7AC3O6VJ8emdokAVxAEoff1wIqxQmecdgVTs26KwUiAq2rWjTEYAEm23jO0+BLlWIBr\ntyk47db2Rl9sXlBHWub3NIoSZUEQBEFoo7YxCJhItiCpXut+G3KnAiBJgGFlW1V7Go0hDT3S/yIz\nroKqZWWDjBRn1+cL1Ef/nOFM7YlLEARBEDohAtxe5rQroFuJ8oDd+riVsLVUUCigISvWzdHQA3El\nylqrY7QsIu8LdB3gtnRobFlzTxAEQRCEmNqmIJI9ABKkNFsBbjA1FwBPsgM9VAeA6sigNhi772Y4\nYgHu3lofADkZri7PV+WrBiDVkYxT7TogFgRBEA6OCHB7mdOuts3ghqwbYzDYOsB1K1aAGzJMdCPW\nMdkTeWrcHGgd+LbHY3MD4Av7eugKBEEQBOH7o64xgOSw7pEpkQxuwG1Ns0pNd6IFawFQHenRANej\nKjiU2FemikiAm5vh7vJ8Fb5KAAak5PbQFQiCIPSuDRvWMXXquW22G4bBr399LU899cfoNtM0mT//\nKSZPPptzzz2TP/7xUXRdb/OzYK3Ac8MNN3D22eO56KLzWbz4370yfhHg9jKHTQFDAVMiGMngygEr\nuxraJ8B1xt08gy3dLgC308oAe/1dZ3DdqvU02av5e+YCBEEQBOF7pLohgOS0AtQMn/Xg2ada03sy\ns2RM03qYbHNkUBMJcDPjsrfBkB6dg5vTjQB3r68KgIHJOT10BYIgCL3DNE0WL17ETTf9Ck1rG3cs\nXPgK33zzdattb7zxD1at+ogXX1zIggWvs3btNyxc+Eq7x3/ooTm43W6WLFnOnDl/4Omnn2TdurU9\nfh0iwO1l1vxZCVNXCdqsG6kSF+BK8QGuGvt1+OPm4XoiJcrebpQou21WgOsLiwBXEARBEPZVUeeL\nZnBTm62HyV7DahaVlhZb31Z1pFMdsF5nxM2/3VsXq5DKTe9GgOsVGVxBEPqGl156gddfX8iVV/6y\nzXvbtm1l6dK3GD/+jFbbly1bysUX/5SsrCwyM7O44oqf85//LG7z8z6fjw8+WMGNN96Iw+Fg+PCR\nTJgwkWXLlvT4dexXF2Vh/7U0iDJ1JZrBVXXrhmmaIEnWTXXfDG5Aj2Vwk6IZ3K5LlN2REuWwESZs\naNhk8SsWBEEQBAB/UKOhOYQ914eqmbibghhIeEPWA2iPxwtBkFUPsuJkj8+6X+e6Yt2Sv6vyAmC3\nyaSndN5FOaiHqAlYc3oHiAyuIAgRmqFF18c+EIoCIbufBp+XDqqBo9KdqajdjAfOP38qV175S77+\n+stW20OhEHPm/I7bbrubt95qXVZcWrqTwsLB0deDBhVQWroL0zSRJCm6vaysFEVRyc/Pp7a2Obrv\nypX/7dbY9oeIfnqZIxLgoquEI5+2asSeEBtYywB1FuDuVwZXjTW88Gt+bJGuyoIgCILwQ9eSfZWc\nXjIaNCTAa08l0igZp62eUBDsrlwCuh6dg9vfHVuyb2eFtUrBoJxk5Lgvb+3Z3fQdJtbBB6fnY4ji\nKkH4wdMMjfs/fST68Ku3ZTrTuXfsrd0KcrOystrd/swzT3HiiWM5+uhj2wS4gUAApzPWQM/hcGIY\nBqFQCIcj9hDQ7w+0et2ybyAQ2J/L6RZRotzLnPbIXyZDIaxGmkzpcQGuEQtwVVnGJlv7+LW4ANfZ\n0kW5OxncWIArypQFQRAEIaai1geSgeTyklVv3VObkwcAYHcoYFgdj+3uXPb6Yvfq/u7Yl7JdFdYy\nQoU5XT9ALm3cDVjr36a5xBJBgiD0PV9++QVffrmaa665rt33HQ4nwWAw+joYDKAoSptg1ul0EgoF\nW20LBgO4XF1P9dhfIoPby1oyuKahEI7MwY3P4OpGZB1c3Xp64VRkwoZOMK7eoKXJlC/YjS7KcRlc\nn2g0JQiCIAhRZZVeJFczkmRGA1xvej4AWf3chP3WfFmbK5fdXuu+nGxTSLJZ92HdMNi11yqtK+zf\ndYC7q6nM2jclr2cvRBCEPkuVVe4de+tBlyinpnloqO/ZEuX2vPvuO3z3XRkXXDABsDK2sixTWrqT\nhx/+IwUFhZSW7mTEiJEAlJbuorCwqM1x8vPz0TSN8vJynM6U6L5FRW33PVgiwO1lzmiJskLYFWky\nZcYCVcNomaNrlUE5FZmmsI4/rkTZEVkfNxjq4m8w4FAdSJKEaZpiqSBBEARBiLOrohHZbWVg+9da\n99kmRwYEYcDAcLSDst3dn5Jq6yFxYVLswfHOiiaCYeteXDyw84ysaZpsrdsOQFFqQc9eiCAIfZoq\nq2S7Mw/45xVFIiMpCXvIha6bXf/AQbj99ru5/fa7o68ffHA2qalp/OpX/wvAxInn8eqrL3P88Seg\nqiovv/wiEydOanMct9vDaaedzty5c7n55jvYtm07y5e/zSOPzOvxMYsS5V7mtMVlcCMlyhLQMt02\nGuBGsrrOyFq4gfYC3LCOaXb+l1iWZFyRheSDerDTfQVBEAThh8I0TXZWNCF7GlB0k+yaELqkUBey\nyugyM63AV7GlINvS2NlkBbhFKbEAd+NOa85cZoqDfmkuOrPHu5eGkDVfd1jmET1+PYIgCIeDCy+8\nmFNPPZ1rrpnOz352CaNGHcOll14OQEVFBRMmnEZFRQUAd9zxWzRNY8qUSdx9921cf/2N0cxvTxIZ\n3F5ma1n6x1BAktBVGUUzUBXQDdB0BTtgRAJch2IFwaH4ADeSBdYNE003samdN7VwKHZ8YT9Bveum\nVIIgCILwQ1DdEMAb0HAk15FTE0YxTGpd/TAiz41djirCGjiSCqgMhKOVVEXJcQHuLivAHVaQ0ao7\naHs21W4BIMnmYWBS/164IkEQhN4xevQYlix5r9337r57dqvXiqIwY8b1zJhxfZt9c3NzWb78w+jr\n1NRU5s2bR21tc69mnkUGt5dJkoSqyKBbQaoWyca2ZHB1LfIHU8c0DWyy9TpkxH7p0TJniJZGdcah\n2iPHCHWxpyAIgiD8MGwurQc1hOxuZmCV9QC4IbMYgIxsJ5rfagjlTCpgU721FFCSqtDPad1TfYEw\nW8vqARhemN7l+dZWbwRgaMYRyJL4uiUIgpAo4l/cBLCpMmakFFmPZHRVyQpgw1rsV2Aa4WgX5bDR\ntkQZuj8PFyCkiwBXEARBEMDKvsrJtQAUlFv3x9okq/nT4OJAbKpQSjHr66xGUsPTk6KZ2q+3VqPp\nJqoicXRx+0tptKgPNrC1fgcAx2WP6vmLEQRBEDokAtwEsCkSmNZHrUdKkBXZCnA1LRa8mkYIezsZ\n3PgAN9CNDK5TiWRwRYArCIIgCJimyabSOpS0KpwBgwHVYfxqEvWa1bMiJ7sKALt7II2Gm3Kf1cNi\nRLoneozPNu4FYGRRZnR1g458ufcbTEycipMRmUN745IEQRCEDogANwFsqgxG6wC3JYOrhWO/AsMI\nR0uUW2Vw7QeWwQ2KAFcQBEEQKKvyUtcUQEmrpKg8iGRCZcpgAFxuCUkvAcCdNozV1dbSHR5VYXCy\ntT5jZZ2P9Tus7O/YETmdnsswDT78bhUAx/YbiU2x9co1CYIgCO0TAW4CqIoczeC2JGxlrAA2HBfg\nmnoIeztNpvZ3Dq5dFRlcQRAEQWixelMlcnIdki3MsJIAJlCRORyAkaP8mHoAkLCnjuDLaqub8vFZ\nKSiRaUPvflmGCaQm2Rl9ZHan51pfs4kqfw0AZ+Sd0luXJAiCIHRABLgJYM3BbQlwIyXKWIFqKK7R\nsWlq0RLlcFyJsqrI0ZtsdzK4LSXKoouyIAiC8ENnmiarN1eiZJWR0qyTvzdMnas/zaYTMMnJ2gWA\nK/VI1jZBU1hHAk7ITgGg0Rviw2/3AHDmcQOth9YdMEyDxTveAeCItMHkJw/s1WsTBEEQ2hIBbgK0\nyuBGPnElksHVw7FlBkxDizaZCsWVKENsHm4grHV5vmiTKdFFWRAEQfiB217eyJ76BpTMCo7e4gOg\nLMtq/HTEkX5MzZpb6846gf+WW2XIozKSyIx0T37z4xKCIR2XQ+HM4zoPWFfvXUNZczkAkwdP7JXr\nEQRBEDonAtwEsObgRpYJinzikmllYrW4hGx8Bje+yRTE5uF2bw6uKFEWBEEQBID3vypDzS7DFdY4\neluABkcWVY4BgMmQwTsBcCQN4ovmVOpDGhJw1oBMAHZVNLFyjRWwThpbQLLb3uF5mkLN/HPrWwCM\nyhrGkLSi3rwsQRAEoQMiwE0AVYkvUba2yZEMra6bIFkbTUNHjWRwtX0D3EgGNxhundltj1MEuIIg\nCIJAXVOQ1Zv3oOaWMHqjD1Uz2ZZ9AgDDhlUjm9UA6Jln8H55HQAnZKfSz2UnrOn8ZfEGdMOkX7qL\ns8fkd3gewzRYsOl1msNeHIqdi4+Y2vsXJwiCILRLBLgJYFNjJcrhyPJAsmmVGuu6iSRbyw2YphYL\ncM3WAW7LnB9N7zrAdShiHVxBEARBWPzJTsjeSVrQx+hNPspTjqDemYPL5ado0DYAbGnH8M89Mppp\nkqQqTMzLxDRNXvzPJr6r9iJJcPX5w1st2dfmPDveYW31RgD+Z8gFZLoyEnF5giAIvWLDhnVMnXpu\n9HU4HObxxx/m/PN/xKRJP+Khhx4gHLZ6/Zimyfz5TzF58tmce+6Z/PGPj6Lr7VecNjY2csMNN3D2\n2eO56KLzWbz4370yfhHgJoBNkcGMBK6RAFcyrF+8rhlIUiTANTTUyILy+j4ZXJsaCXC1bgS4kQxu\nUMzBFQRBEH6g9tb6+GDjdmy52zj7syaCSjLbsk5ElnVOHrsFiTAoblZox7PHF0QCLh6cg1OReeOD\nHaxab83NnXpKEUPyUjs8z9s73+ftXe8DMK7/CZw84MREXJ4gCEKPM02TxYsXcdNNv0LTYs1q589/\nipKSHbz66hssXPhG5M8vA/DGG/9g1aqPePHFhSxY8Dpr137DwoWvtHv8hx6ag9vtZsmS5cyZ8wee\nfvpJ1q1b2+PX0flK5UKPUOPn4Ea6KMuGlcE1dKPDDK5pmkiRgLclwA13I4Nrj6y5F9a7bkglCIIg\nCN83pmny4rINKAVrOWFzE7lVJqvzzkJXFE4cvRGnvQHDlPnC+WPW1vsBOGtABsXJbl57fxvvfLEb\ngBOH9eOCUwrbPYdu6Pxr+xL+u/sjAIamH8FPjrowet8WBEHoiKlphGtrD/jnFQX8wWZCDV46SJZG\n2TIykNTuhXwvvfQC77//Llde+UsWLPgbAJqm8eab/+LZZ18kJcV62Pfggw+jaVacsWzZUi6++Kdk\nZWUBcMUVP+cvf5nP5ZdPb3Vsn8/HBx+s4O2338bhcDB8+EgmTJjIsmVLGDly1P5cfpdEgJsAiixF\n5+Dq0QA3ksHVYxlc4jK4ALppRl/bIj8X7kYGV40EzJohAlxBEAShtaVLl/Lkk09SUVHBgAEDmDVr\nFmeffTZr167lkksuwel0Rve99tprmTlzJqZp8thjj/H666+j6zpTp07lzjvvRFE6Lts9lN75Yjfb\nta8Y1vAdJ6wNsmbABALuFI4/egNZmbWETJWVtqlsb7CqpU7KTuWEtCQe/8ca1u+05uKOOSqbqycP\nbzdgrfRV8fLG19nRsBOAkZlDuXrkFdH7ryAIQkdMTaPkt3egVVcn5HxqVhZFcx7qVpB7/vlTufLK\nX/L1119Gt+3eXYqu62zYsI4777yZQCDAhAnncu21NwBQWrqTwsLB0f0HDSqgtHRXq0QdQFlZKYqi\nkp+fT21tc3TflSv/21OXGiX+JU4ARZaic3D1li7KehjkjufggpXFbfkF2VTrS0R3Mrg2JRLgmiLA\nFQRBEGJKSkq46667eOGFFxg9ejSffPIJM2bM4IMPPmDjxo2MHz+eZ555ps3PLViwgBUrVvDmm28i\nSRLXXnstL7zwAtdcc80huIrObdhZyz/XrmCoup4zPwuzZuA5hNNSGHfcGlJTvOwxs1gpnUl9wLpX\nnpidQnqDxm8XfU6z3yrJO+O4gVw+4QgUufVMLr/m573SD1leuiL6EPms/NP4cfEkFPnwDPYFQRC6\nqyULG6+xsRFNC/Pxxx/y3HMv4fN5ue22WXg8Hn7+86sJBAKtHow6HE4MwyAUCuFwOKLb/f5Aq9ct\n+wYCgR6/DhHgJoAiS9CSwY0EsC3LBFkZ3JYuyhpK3JMOzTAhcr9UIxnc7szBtcm2yM+LAFcQBEGI\nKSoq4uOPP8bj8aBpGtXV1Xg8Hux2Oxs2bGDo0KHt/tyiRYuYPn06/fr1A6zM7rx58w67AHdzaR1P\nrniTEwKrGbkpia8GTiCnsJFhR32JX3GwQj+RTeZgQEIyTYYZNr56bxdlVVY2wWFT+Nk5R3LKqP6t\njlvtr+GT8i/44LtP8GvWl7FUezKXHHUhx2aPTPRlCoLQh0mqStGchw66RDk11UNDD5cot8dut2EY\nBtdccx3JyckkJyfzk59czv/7f6/x859fjcPhJBgMRvcPBgMoitImmHU6nYRCwVbbgsEALpf7gMfW\nERHgJoD1BNgKUCNxrlWirIChtT8HF1ovFbQ/c3BjJcp6m/IAQRAE4YfN4/Gwe/duJk6ciGEYzJ49\nm6SkJDZu3Ijdbuess87CMAzOO+88Zs2ahd1uZ8eOHQwZMiR6jKKiIkpKSrp9j5EkCfkg21q+s+wt\nMs1N1DUonHjB1aR4UqLvGabJ26u3899vX2fyjr2E9aOpGJ3OiUM24fc4+dAYwza9AM2U0bxhlOog\n/gof73tjzRiPPyqby885kswUKxOx11vFuppNrKlcx9a6HdH9bLLK6Xknc37xBFxqLGvRGTlyb5fl\nvnU/FuNOLDHuxDqk41ZsqP1zDvjHZVnCleYh6PJi7NOYticokcSaokgUFBQgyzK6Ho5uN00DMFEU\nicLCQsrKdnH00dY82rKyUgoLi6L7tigoGISmaZSXl+N2W3N5d+8uZfDgtvseLBHgJoCiSGBEAlyp\ndZMpXTfb7aIM1hzcFtFlgrqTwY2UKJuYGKaBIomyKUEQBCGmf//+fPPNN6xevZrrr7+egoIC0tPT\nOemkk7j00kupqanhN7/5DU888QS33HILfr+/VQmay+VqtwStI5mZnoN+2Nq8dxdHjPCRkQ2bVzzL\n9vosTjzzbLaWlbFp9Spyqps4IykL+bj+ePrrVKhOFutnUFPvItwUJtzQQKgmgB5sne44bmg2k8cP\nICk9xKbaNWzfs4ttNSVUemta7ZfiSOLMopM5/6gfkeZM4UCkpXkO+PoPJTHuxBLjTqy+Om7ovbEn\nJ7uQJImMjCQyMpI4++yzef75+Tz22GP4/X7+3/9byAUXXEBGRhIXXXQhr722gLPPPgNVVVmw4G9c\ndNGFZGQktTpmRkYSP/rRj5g7dy5z5sxh69atvPvu2zz77LNt9j1YIsBNAOvJUOsMrqS3BLgGtJQo\nmwZq3CPucHsZ3G6VKMd+rWFDE/OCBEEQhFbUSLnauHHjOOecc3jvvfeYP39+9H232821117LY489\nxi233ILT2boEze/3o6pqt4JbgJoa70FncM+YcjkbVzzPgHwvWTleMrJ9VH7xGt4qGR2VtYUFNNhS\n0IJ29M0mRkjHCO8EDJANJEWDtBCqGsLh1snIkFCcfkpCdTy6pv1l9Tw2N8MyjmRM7jGMyhqGIisY\nPqj1Ne/X2GVZIi3NQ31972RbeosYd2KJcSdWXx039P7Ym5r8mKYZbQZ1222/5cknH+e8884jFAoz\nadJkfvzjS6itbebcc6dQVraH//mf/yEUCnPuuecxderF1NY2U1Gxh8suu5i///11cnP7c8std/LY\nY39g/PjxuFwurr/+RvLzi6Pn2R+dBcUJDXA3bNjAvffey7Zt2ygoKOC+++7j2GOPbbPfP/7xD/7y\nl79QXV1NUVERd955J2PGjEnkUHuUIkuYkSZTRnQObizAbZmDi6l3ncHdjxJlaGk01b0vIIIgCML3\n28qVK/nrX//Kiy++GN0WDocxTZM//OEP3HDDDSQlWV8agsFgNIAtLi6mpKSEY445BrCaVQ0ePLjN\n8TtimmaX88S6kpqSxrHnXMd/3/gLRxQ3Y7dr5AxsJmcgHK3L1NXoNO5pwlcRQvM2otvqCDqDeN0y\nXoeC1y3T7JLxumTCNplqE/C3Pke6I42ClHwKUvIYkjaYwpR8ZCkSmZtW1dXBMAzzoI9xKIhxJ5YY\nd2L11XFD7439mGOOZ8mS96LHdjrd3Hrr3dx6a+v9rPdlrr76Oq6++ro272Vn57J8+YfR18nJKcyb\nN4/a2ubosXtj/AkLcIPBIDNnzmTmzJlcfPHFLFq0iOuuu453330XjyeWXv/000957LHH+Otf/8pR\nRx3FokWLmDlzJsuXLyc9PT1Rw+1RVhflfebgtmRwNQMpcvO0MrhdzMHdjxJl6xii0ZQgCIJgGT58\nOOvWrePf//43U6ZM4cMPP2TlypW89tpr0eWAbr75ZsrLy5k/fz6XXHIJAFOmTOH5559n7NixqKrK\nM888w9SpUxM+fo/Hw5W/uZvtW3fyxbsLyUr3kZbuR1EMsvo1kNUPOAa8PhdV1QOo25uEWh5ioK+O\n5EA1KcEaHLofzaYQTnZhpiShpqXhysgmKTMXT3p/bBk52LKzke32hF+fIAiCcPASFuB++umnyLLM\nZZddBsC0adP429/+xsqVK5k0aVJ0v4qKCq666iqGDRsGwIUXXshDDz3Etm3bOOGEExI13B4VH+Dq\nkfi1pUTZNIH4ADe+i3JcBtem7H+TKbAaTQmCIAgCQHZ2NvPnz+f3v/89999/P4WFhfzpT39iyJAh\nzJ8/nzlz5jB27FicTieXXnop06dPB+Cyyy6jurqaadOmEQ6HueCCC/jFL35xyK4jPTOLH02z1mAM\nBZuo3vUN3trNqMpeFFXD4w7gGbSHwkGgGxK1talUVQ9le1UGRn2YDH8F6f49pO+uwL6zAp1NNAAN\ncedQ0tKwZ/fD1i8He04O9rx8HHl5qOkZonmjIAjCYSxhAW5JSQnFxcWtthUVFbFjx45W23784x+3\nev3ll1/i9Xrb/GxHeqJTI/RsZzWbKsdlcCMlynGZVQlrwJJkYFNj5zOlWBczuy2yzJBhdtppTJal\nVnNwTUnv8c5kvUF04EssMe7E6qvjhr479r467kQYM2YMb7zxRpvtQ4YMaVW6HE9RFGbNmsWsWbN6\neXT7z+5IZsCRpwKnYpoGId93eOu24q3bihHeiyKbZGfVk51Vz/ChO6irT+a78hw2VwwmHLaRqgTI\nMarIaizBWb0LKVJLrdfX46+vx791S6vzyW43jkiw6ywqxlk8BFt2tgh6BUEQDhMJC3B9Ph8ul6vV\nNqez88V9t23bxo033siNN95IRkZGt87TE50a4/VEd7KkJCdE5+Ba21oyuAB2hx0vYLPJZGYmo0gS\numni9jiiE6iTk63ulSZSl53GaiML1QN4ku1kpPVsZ7Le1Fc72YlxJ5YYd+L11bH31XELB0aSZBye\nfByefDLyzkIPewk07cDfuI1A43YM3Ud6WhPpaU0MH7qdir1Z7CwdwJb6PLYk55M00EHhoCQKMw2S\ngzWEqyoJVVYS2lNOeO9eME0Mnw//ls34t2wG3gNASU7BWVyMa8gRuIePwJGXj9QTT9sFQRCE/Zaw\nANflcrUJZgOBAG53+4v7fvTRR8yaNYtf/OIXzJgxo9vn6YlOjdCz3cmCwXDbDK6mxb1vPS0OBYPU\n1jajSKCbUN/op9Zm/YqCAStoDYW0TjuNybKEzRX7tdbUNZFs7H9nskTrq53sxLgTS4w78frq2Hty\n3D29fIGQOIrNgydjFJ6MUZimQaBpB97ab/HXb0KWNQb0r2JA/yqampPZvmMA5RXZrFsXZB2Q2S+Z\no0YewRGTcnB77BjBIKE95QTLygiW7SZYuovAzhLMUAi9qRHvmq/xrvnaOm9yCu7hI/CMHIln5NEo\naQe2rJAgCIKw/xIW4A4ePJhXXnml1baSkhImT57cZt9//vOfPPjgg9x/fznSigAAIABJREFU//3t\nvt+ZnujUGK8nupNJLf9vRpfDRdJjWVajJbtr6Oi6iSxJgElYj527pcxO07sejzOuRDmohftUZ7i+\n2slOjDuxxLgTr6+Ova+OW+h5kiTjShmCK2UIhh7AV7eepqovCAcqSU5q4tijNzNyZBklO/PZti2L\nmkovn7y/nU9X7KB4WDajjs8jp7AIZ2FR9JimphEsK8O/YxuB7dvwbd6EXl+P3tRI02eraPpsFcgy\n7qOGEjz9FJShI5GSUg/hpyAIgvD9l7AAd9y4cYRCIV5++WV+8pOfsGjRIqqrqzn11FNb7bdq1Sru\nu+8+XnjhhT69NFA8pWUOmCnHSpTNuCjcbHnfaiClREqs45cJajmGZuxfkyldNJkSBEEQhFZkxUlS\n1vF4MkcTbN5JU9Vn+Bu2oMpejhi8iSOGJFFVdyRrvk4mFDTZur6Sresr6dc/mVFj8hgyLBtZlpFU\nFWdhIc7CQjjrbEzTJFT+Hb716/CuX4d/8yZMTcO3cQM7Nm4AScJ15FGknnoaSaPHIHdzHWFBEASh\n+xIW4Nrtdp577jlmz57NY489RkFBAX/+859xu93ce++9ANx///0899xzhMNhrrnmmlY/P2/ePMaP\nH5+o4faoWJMTKVqiLJuxQNUk1kUZiHZSNtpZB7c7mQhFVqJ/1k0R4AqCIAhCeyRJwplchDO5iHCg\nioaKD/HVrQejmezUrzh3QhpNodGs+dJGXY2fyj1NvPfWRr74sITR4wo4cmQOiiK3Op5jYB6OgXmk\nn3MuRiCAd923NH+5Gu/abzECAfybN+HfvAl5wcskn3gSKaechnNwsWhSJQiC0EMSFuACDB06lIUL\nF7bZfv/990f//MILLyRySAmhtkwKNuVYiXJ8gGu2vG8Foy27a+0FuN3I4EqShCzJGKYhAlxBEARB\n6AabM5uswosI546PBLrr0MP1uKX3OWN8HmHlJNZ+bbBrew2N9QFW/Gczqz/eyXEnDWLYMf1R1LYN\nQGSnk+QxJ5J20kmkemzsXvkJ9R99FA12Gz5YScMHK3EMKiB9wjkkn3ASkprQr2aCIAhtbNiwjjvv\nvIVFi5YBUF1dxcMP/55vv12Dw2HnvPMuYMaM65FlGdM0eeaZP7F48b/RNJ1zzz2fX/96FoqitDlu\nY2Mj99xzO6tWrcLjSeKXv7yGyZN/3Ga/gyVa/CVAdJkeU0JvaTLVKoNrbTP3LVE22ilR7uZcMkVS\nIscQAa4gCIIgdJfNmUVW4YXkDp2BM9laojDkK8Ns+icnjtnCtOlDGTIsG4DmxiAfLt/Kq899zpZ1\nFZhmx/doxeEgZcwJDPz1/zL4kcfImnYJ9v4DAAiW7qLi+efYcfvN1Ly1CK2xsfcvVBAEYR+mabJ4\n8SJuuulXaFqsX9Djjz9MXl4eixcv57nnXuK995bz9ttLAXjjjX+watVHvPjiQhYseJ21a79h4cJX\n2j3+Qw/Nwe12s2TJcubM+QNPP/0k69at7fHrEAFuAshSSwArRefgyrTN4O4b4MY3/lSVlgC36wwu\nxMqURQZXEARBEPaf3ZVDvyGXk118GTZnPwB89esJVL7ISeOauPTq4zlyZA6SBE0NAd5bvInX/7qa\n0h01nQa6AGpqGhnnTqLg/gfJu+V2PMceB5KE3tBAzaJ/UXL7zVS+uoBwbW0iLlUQBAGAl156gddf\nX8iVV/6y1fbdu0vRdT36b5v8/9u78/io6nvx/69zzmyZ7CFAWAKEgOwSVqGAKCIoikiVulsvt6LF\n1l9dqtZWi+1t9Xettlqvt9bWtu6Pi7auqKACKoqKChQQFQgkkATIvs5yzvl8/5jJJEMCTJTMJPH9\nfDzGOft5zxjmM+/5bLqG2x2awvT111exZMklZGdn06tXNldccRWvvfZKm2s3NjbyzjvruP7663G7\n3YwePZYzz5zP66+/esJfh7SDiYPWNbh2ezW4sQwyZTQnwaFRQVv69R7lnlq4SbOKLSEWQgghRFtJ\nacPwpA6lvuJTqkveRlk+qg+sxunZwszTFjBh2iA+XL+HvV9VUHGogVf/79/0H5TBjDOGkd332FNM\naZqGd+QovCNHETh8iOq336L2vXewm5qofmsNNevXkjZjJllnnYOzd+84vWIhRGezLJv6Wv/XPt8w\nNLA1amqajjs+T0qaO2qsgGM555xFXHnlUj777JOo7ZdeeiX//d+/4YUXnseyLM4++1zmzJkLQFHR\nXoYMGRo5dtCgwRQV7UMpFTW2wP79RRiGg9zc3MiUp4MGDWb9+rUxxdYRkuDGQXPzYmVrkT64ejsJ\nriJU29pegutoldBato2ut23X3ppDC/2vNaWJshBCCPGNaJpOavZkvOmjqC55k4bKLQR9Bzn41d9I\nzipg3qK5HCobxMZ1uynbX0tJUTXP/X0Towr6M3XWEFJSjz9asqt3H/pcdAm9Fi6ieu1bVK15A7u+\nnpr166h59x3Spk0na+EiXL37xOEVCyE6i2XZPPPnj6ir8cXlfqnpHi5ZNjWmJDc7O7vd7Uoprrji\nP7jkkisoLS3h1ltv4IUXnuf88y/A5/Ph8Xgix7rdHmzbJhAI4G41UnxTky9qvflYn+/Evw/SRDkO\njMggU1qraYKOVYMbWm2vBhdi64fbfE9poiyEEEKcGIYzmV6DF9Fn+FWRZssNlZsp/fxhUpP2sujS\nAs767ljSMjwoBTs+K+HpRz5iy8f7sWLtYuT10uuchQy953dkL7kIIy0NbJva9zew9xc/4+BTj2NW\nV3fmyxRCiIjy8nJ+97u7ueyy7+PxeMjLG8rll1/FSy/9CwglqX5/S2203+/DMIw2yazH4yEQiK61\n9vt9JCV5T3jMUoMbB5HKV6W3NFFu3QfXPqIPrt5cg9tyjeY+uBA9+NTRRAaZkgRXCCGEOKE8KYPI\nGXk1dYc/oqZ0HbbZSGXRizRUbmHgoAVc/IOpbN20n0/e30fAb/Lu6q/YuaWU6XPyGTA4M6Z76B4P\nWfPPJuP0M6h57x0qX30Zq6aGmrVvU7vhPTLmzCXrrAUYKcduBi2E6FoMQ+eSZVO/cRPl9HQvNTWN\nJ7SJcnsqKsoxTRPTNHG5XOH7G5FRkgcPHkJR0V7GjBkLQFHRPoYMyWtzndzcXEzTpKSkBI8nLXJs\nXl7bY78pSXDjINJfVmlYzTW4gKaF+9SqcFW6OlYT5dY1uMf/FTgyyJQ0URZCCCFOOE0zSOszHW/G\naKr2v0ZTzZf46/dSuvMR0vrOoGDqTE4a25cP1xfyxb/LOHywnpee2cKQ4b2YccYw0jKSYrqP7nKR\nOWcu6TNmUf32m1S+tgq7sYGq11dRs34tmfPPJnPuPPRWTQSFEF2bYeikZ8b2GdD++RpZWcmgq+Mm\nuN9UXt5Qevfuw0MP/Z6f/OSnVFSU88wzT7Jw4SIA5s8/m2eeeYJJk6bgcDh44om/M3/+gjbX8XqT\nmTVrNvfddx833XQbu3btZs2aN7j33gdOeMzSRDkOopoot+ps3Zz4Hm0U5ahpglrV4MaU4EoNrhBC\nCNHpHK50eg+9mOy872E4U0FZ1Ja9Q+nOP2GoEuacM5IlV01iwKAMAPZ+VcGzj37Eh+v3EAyYMd9H\nd7vJOvsc8u75b7LOXYjmdmM3NVHxwj8p/NktVL25BjsYPP6FhBCiA1wuF/fe+wClpSUsWnQWP/rR\nMubOnceSJZcAsHjxEmbOnM3VV3+fyy//HuPGjeeiiy4DoKysjDPPnEVZWRkAt932C0zT5LzzFvDz\nn9/C8uXXR2p+TySpwY0DLfIzQksfXABdB8tq3Qc3lIzqxxhFGTrYRNmWUZSFEEKIzubNGIknNY+a\n0nXUHf4I01/JoV1P4M0cR/ag+Sz98Uw+eHc377+1m8aGAJ9+UMQX28qYdlo+w0f3iRpt9FgMbzLZ\n519AxulzqXztFWrWrcWqq+Xws09RteZ1shd9l9Rp09F0qcMQQnw9EydO5tVX34qs5+UN5fe//592\njzUMg2XLlrNs2fI2+3Jycliz5t3Ienp6Og888ACVlfWdWvMsn35xYLSqqbVbjYbcnMjadvP+ow8y\n1XoU5VgGmXJE5sGN/ddhIYQQQnx9uuEmc+B8ckb8AJe3PwCNVf9m/7aHqCj5iBFj+3LJsqlMmJaL\nrms01AV46+XPeeGpzRwuq+vQvRzp6fS5+DKG/OYe0mbMAk3DrKig7LFH2bfiDuo/+/S48/EKIURP\nJAluHDQnsqEmyq22N7dcPmIUZcfxanA71ERZanCFEEKIeHJ5+9H3pKVkDjwLTXdhWz727XiO0i/+\nBnYl007L56IfTGFwfi8AyvbX8NzfP2H961/Q1Bjo0L2cvbLJ+Y//ZPBd/0XKhEkABEoOUPI/D1J8\n93/RuPPzE/76hBCiK5MENw5aDzIV1US5uaY2UoN7ZBPllmM7PIpy8zRBMsiUEEIIEXeappPaeyr9\nRi3HmzkaAH99MWU7/0x1yVukZThZsGQcC5aMIz0rNNjMjs2lPP3IR2zdFPu0Qs3c/QfQ/7ofk3v7\nHSSNHAWAb89u9v/u/2f/73+Hb+/eE/r6hBCiq5I+uHFgtEpw0bTQk2pJcJWtNQ+jjFKq1TRB32AU\n5XANrimDTAkhhBAJ43Cl0Td/CYZVzN7tz2MGaqg9uIGGqu1k5S5gcP4wBg7J5N+bDrBpw14CfpMN\nb+5ix+YSZs4dzsAhsU0r1CxpaD65N99Kw47tlD+/Ev++vTRu30bR9m2kTJ5C9vnfxZXTr5NerRBC\nJJ7U4MZB6ybKACqcrDYnuNHjQNmRUZRtdbRRlGOpwZVRlIUQQoiuIr33KAaMWU5qn+8AGlagmsO7\nn6a88DmwGyg4JZdLl01lxLgcAKrKG3n52S28/s9t1FQ1dvh+yaPHMOgXv6TfD6/DmRO6Zv2mj9l7\n5885+PjfCFZWnsiXJ4QQXYbU4MZB6ybKAErXwGqd4Lb8zqBUS4Jrtp4mqNUgUx3pg2tLE2UhhBCi\nS9ANF5kD5pKcNY7K4lcJNOynsXoHTbW7yeg/h5TsScw5ZyRjJvTnvTVfcai0jsIvy9m3q4KR4/sx\n+TuDSU51x3w/TdNInTSFlIKJ1L7/HhUvvYhZVUnNO+upfX8DGXPmkjn/LBzpGZ34qoUQIr6kBjcO\nWpLT6BpcTQslsFE1uMqKjKLcugZX07TIdcyOTBMkg0wJIYQQXYorqS99h/8HmbnnoBkelO2nav9r\nlH3xF5pqd9O3fxrfvXIip58zkuRUF7at2PFZCU8/8iEb1+3B7+vYfLeaYZA+azZDfnsPvb93MXpK\nCso0qVr9OoW3/ZRDTz9BsKKik16tEELEl9TgxkHz3HaqdQ0ubQeZCh3TUoN7ZEtkh6Fj2VZMNbjN\n0wSZtkwTJIQQQnQ1mqaRmj0Jb/oIqg6sprFqG8GmMg7vfgpP6lAy+p/ByHH9GDayN9s+PcCnHxTh\n95l8trGI7Z+VMGFaLmMnDsDljv2rnO50kTnvLNJmzaZq9etUv7kau6mJ6rffonr9OtKmf4ess8/B\n1TenE1+5EEJ0Lklw48A4ooly81y4OuEaXKvV3EFRCW50hhupwY2lD67U4AohhBBdnuFMIXvId/Fl\nT6L6wJsEGg/gq9tD2Rd78GaOJb3faRScMohR4/ux+cNitm7aT8Bv8uH6QjZ/WMy4yQM5efIA3B5n\n7PdMSiJ70WIyz5xPzbq3qVr9BlZ9HbXvvUvthvdInTyFjDPnkzJ8WCe+ciGE6ByS4MZBu31waW6i\nrEU1UVbKOmqC2zxVkGXH0Ae3eZogGWRKCCGE6PI8KYPpe9JSmmq+oLrkLUx/BY1V22is2h5KdHNm\ncsrsoYydNIBP3t/H51tK8ftMNr23ly0fFTN2Yn9OnpKLN9kV8z0Nr5esBeeSccaZ1Ly7nqo3XsOs\nqqLu44+o+/gjkvLzsRYvQh8xBsI/nAsheq4tWzbz0EO/p6hoL+npGVx66ZWcf/4F1NbWcvfdv+LT\nTz8mOTmFpUuv5txzzwdAKcUjj/wPr7zyAqZpcdZZ5/DjH9+AYbT9zKitreWOO27lgw8+aHOdE0kS\n3DhoGUVZDz+F1g3CCe6RNbjtTBMEYBih82OrwQ39r5V5cIUQQojuQdM0vBkjSUo/iYaKz6gpewcr\nWEdj1b9prPo33ozRpOXM4tR5JzFx+mC2fFTMjs0lBAMWn20sZuumA5w0pi/jJg+gV++UmO+ru91k\nzp1H+uzTqf1gA9VrVhMoLaFp926+/N39ODIzyTj9DNJmnYojNa0T3wEhRKLU1tZy2203csMNtzB3\n7jy++upLfvKT5QwYMJAXX3werzeJl15aze7dX3Hzzf8fQ4bkM3bsOP75z//jgw/e4+9/fxZNg1tu\nuYFnn32Syy77fpt73HPPf5Gamsyrr67hyy+/jLrOiSQJbhwcOchUcxNlLdxE2Yzqg2uia6FmRkdr\nohzTKMpSgyuEEEJ0S5qmk5I9ieSs8dRXbKb24AasYA2N1TtorN6BJzWf1D6n8J05+UycPoitm/az\n7ZMDBPwWn28p5fMtpQwYnMG4yQMZnN+rpSXZcehOJxmnnkb6rNk07thO9VtraNi6BbOqivJ/Pkf5\ni/8iZcJE0mfNxjtqNJouY5UK0VMcPFjKd74zg3nzzgJgxIiRTJw4iW3btvLuu+t5+unncbvdjB49\nljPPnM/rr7/K2LHjeP31VSxZcgnZ2dkAXHHFVfzlL39qk+A2NjbyzjvreOONN9q9zokkCW4cHLWJ\ncqQPbsuxyrZwaKHmRUfmsY7mGtwOjaIsCa4QQgjRHWm6g9Tek0npNYGGqq3Ulr2HGajCV7cbX91u\nHO5epPaeypQZ4ymYOoidW0v59ycHqKvxcWBfNQf2VZOW4WHkyf0YMS6HlBinGNI0jeQxY0k7eRye\nxhr2Pv8i1RveQ/n91G/6mPpNH+Po1Yv0maeS9p2ZOHv16uR3QoieRdkWZrDma59v6xr+Rh9BXyPW\ncfIChzMdTT9+F4Phw0dwxx2/jqzX1tayZctm8vOHYxgOBgwYGNk3aNBg1q9fC0BR0V6GDBkata+o\naB9KqchAuwD79xdhGA5yc3OprKxvc50TSRLcOIj8wBkZZCq8PZzgWlbreXBj6IMbQxPl5lGUpYmy\nEEII0b1pukFKrwkkZ42nqXondYc/xN9QjOmvoGr/a1SXvE1y1lhGjilg7KSpFO2uYOumA5QUVVNb\n7eOjdwr5+N1CcodmMXJcP4YM7xXp9nQ83oEDyLniSnp990LqPv6ImnfX49uzB7OigooX/0XFi//C\nM2w4aVNPIWXSFBzp6Z38bgjRvSnbouTz/8EKVH+j6+yP8TjDlUH/UdfFlOQ2q6+v59Zbb2DEiFFM\nnDiZlSufjdrvdnvw+XwA+Hw+PB5P1D7btgkEArjdLT+qNTX5otaPvM6JJAluHOhHTBNka83TBDUn\nuK374FqR49s2UW7ugxtDE2WpwRVCCCF6FE3T8WaOxps5Gn9jCXWHPqSxejvK9lNf/gn15Z/g9PSh\nV68Czl0yjuoqxY7NJXy5/RABv0nR7kqKdlfiSXIwdERv8kf2of+gdPQYmhrrniTSZ80mfdZs/PuL\nqXnvHWo/eB+7oQHfrq/w7fqKQ888hXfkaFKnTiV5/AQcadJfV4jupqTkALfccgMDBgzgV7+6m717\n9xII+KOO8ft9JCV5gVCS6vf7o/YZhtEmmfV4PMe8zokkCW4caJqGptFmmiAtPIWP1bqJckyjKMs0\nQUIIIcS3mdvbH/eQxVjBM2mo3EJ9xWZMfwVB3yGqD6ym+sAaPKlDmDBxDFNPHU/RnkZ2bi1j/94q\nfE0mOzaXsmNzKUleZzjZ7U3OwPSYanbdA3Ppc/FlZF/wPRq3b6Puow+p3/wpKhCg8fPtNH6+HbS/\n4xmaT8r4ApLHT8DVv39Uc0Uhvq003aD/qOu+URNlQ9fIyPBSXX3imigDfPHFTm666cfMn3821133\nE3RdJzc3F9M0KSsrIycnNEd2UdE+8vLyABg8eAhFRXsZM2ZsZN+QIXltrt18nZKSEjyetDbXOZEk\nwY0TQ9faaaIcSj7tIxPc8H77qKMoxzLIVOgP2ZQmykIIIUSPZThTSOs7g9Q+3yHQUEx9xeZwrW4Q\nX10hvrpCQCM9NY/TzhiByUgKd/nZvfMw5QfraWoMsv2zErZ/VoLLbZCbl8Xg/F4Mys86bp9d3ekk\npWACKQUTsP1+GrZuofajjTRu+zcqGMS3exe+3bso/+dzOLN74x03Du/I0XhHjMRIiX2UZyF6Gk03\ncLqzvvb5hqHh9qbg9NWjx9B1MRaVlRXcdNOPufjiy7j88qsi273eZGbOPJVHHnmIW2/9BYWFu1mz\n5g3uvfcBAObPP5tnnnmCSZOm4HA4eOKJvzN//oI21/d6k5k1azb33XcfN910G7t2RV/nRJIEN050\nXcMOj6KstOYa3HATZZvQ/HLKOqIGl6gO2h3pgytNlIUQQohvD03TcKcMwp0yiMyBZ+Gr3UVD9XZ8\nNV+hlImvbg++uj0A5GRkkzdvGBaD2LfPxe7PK6g43EDAb7F752F27zwMQN/+qQwf1ZesPsn06ZeG\n03X0WiDd7SZ1ylRSp0zF9vtp/HwH9Vs+o2HrFqyaGoLlh6lZ+zY1a98GTcOdOwjvqFF4R47Gkz8M\nw3vimykKIWL3yisvUl1dxT/+8Vf+8Y+/RrZfeOHF3HrrL7j33rtZvHgBSUlJLF9+faTGdvHiJVRW\nVnL11d8nGAwyb97ZXHTRZQCUlZVxxRVLeOKJleTk5HDbbb/gD3/4b847r+11TiRJcONE1zTs8Dy4\nzWNKaZEaXBtNM1DKAtvCMFqa8FgKHOHVSB9cuwPTBEkNrhBCCPGtohuuSF9d2wrQVPsVTdWf01S7\nG2X7MX3l1PnKgY308ToYeOpANMcAyivTKNzj4MDeWixLcbCkjoMldaFr6hp9+qXSf1AG/XLT6dMv\nDU+Ss/37u92Rml1l2/j37aV+6xYad2zHV7gHbBt/0T78RfuoeuN10DRc/frjGZpPUn4+nqHDcPXr\nJ9MQCRFHV165lCuvXHrU/b/+9T3tbjcMg2XLlrNs2fI2+3Jycliz5t3Ienp6Og888ACVlfUxVdh9\nXZLgxomha5hHNlEO94+1bRVKcInugwvNzZSlBlcIIYQQHacbLpIzx5CcOQalLPwN+/HVfEVT7S6C\nvkMoZeKv3wvsJUWHccMNJp7cn0Awi4pKLyX73RQXWdg2lB2opexALXwQunZahoc+/dPo0y+Vvv3S\n6NUnpU0tr6brePKG4skbCosWY/uaaPzyS5o+30Hjzh34i4tBKQIlBwiUHKD2vXdCcScl4c4dFH7k\n4s4dhKt/f3SnK75voBCi25EEN070qD640YNM2VYowYW2CW7rgaY60gc3Mk2QJLhCCCGEADTNwJMy\nGE/KYDIGzMUM1OKv34uvfh/++n2Y/kpQFsHGYjSKyU6F7FEwfowHi17U1ady+JCTslIH9Q1eaqt9\n1Fb72LXjUOQeaRkeevVOIat3cuSRkZUUGalZ9ySRcvJ4Uk4eD4BVX49v7x6adu8O9dkt3IPd1ITd\n1ETTl1/Q9OUXLS9A13Hl9MM9YADOnH64cnJw9e2HK6cvuicpru+lEKLrkgQ3ThyGHpkmqHlWoJYa\nXBv01gluy3mtE1xHODE2O1KDG0NzZiGEEEJ8+zhcaTiyTiY562QArGAdvvoi/A3FBBpLCDaVoWwT\nZfvQOUC6F9KHwLAhofMtlUJjUwpVVR6qq500NnpobPKwd1cThV+VR+6j6xqp6R7SM5NIz0wiLfyc\nnplEarqX5LEnkzw2FIOybQJlpfj27MG/vwh/cTH+4iLsxkaw7UhN75GM9AxcOTk4e/fB1Tsbc9AA\n/J4UjMwsHJlZaEbsc4AKIbo3SXDjxOnQobkPbnj+Wy1cu9q6BhdlhUZcDouuwW2eJqgj8+Ca3zz4\nHsxWNqZtYSkTS9nY4YdlNy9b0dtV9HY0RbLPRXVtA6bVajuhAcJsFCiFQqGanyPLtLOt5RkFdngd\nVNRy5Nrha7SmUO0uNy8qFLqm4fE4afIFUOqIY6NOUUduilprubVqZ2/ra7a3v73rtL1ba5qm4XY7\n8PvNNq+7q2gvKk0Dt9uJ3x+ki4YdpfXfja5puNwOAn6zzcjuXZlTN1gw+nQy6ZXoUIToNgxnaqQ5\nM4CuK5Jc9Rwu3Y2v/gDBpkMEfIdQVmguS0OrJ9VbT6oXBg1ouY5SGv5AEvX1bhoa3fj8bvw+F021\nbqoOufH5XQSDDpq7YHlTXKSkuklOdYee09ykZJ1EyuBxpKW48SQ50OprCOwPJbuB0lICZaUEDh5E\n+X0AWDXVNNVU0/TFTgBa0mtA03BkZuLslY2RnoEjPR1HejpGWjqOjPBzejpGapr0+xWiB5AEN05c\nDh2s8NQ9WigB0sKJqmW3aqJsH9lEueUajnAT5Vj64DqN0MAPQdvEVja61j0+sC3bojHoJ2AH8FsB\nAlboOWgF8dst64Hmhx2MrAftIKZthh8WQdvEVC3rLftC25vfGyFEz1QdrOO68f+R6DCE6LY0Tceb\n2p/UYBrezAIg9KOqFawj6DscejQdwvRXYPqrsMz68HkKj7sRj7uR7KP8xmRZOj6/C7/fRSDoJBhw\n4A+4aKhwUFXqJBB0EgiEnoNBBwonniQ3Sd4BJGXkkdTficfrxK1bGL569MZatNpKtNpK9Opy9KrD\nUFcdmpJRKczKSszKyuO9YIyUFPTkZIzkFIzwc2g99NDD2/WkJHRPErrHE3q43WgO+VotRFcQ13+J\nO3bs4M4772TXrl0MHjyYu+66i4KCgjbHvfLKK/z+97+noqKCU045hd/85jdkZ2fHM9QTzukwUL5w\nghuuHdEjNbh2aJog2umD22ryZiPSRPn4SVmSwxNZ9lt+khyd2zfFthX+oEljMECdr5E6fwP1gSYa\ngk00BJpoMhtpDPrwWaGH3/Lht/0ElZ+A8mMSwCSATfercVYq3L9Eqls6AAAW00lEQVS6+YEWrsZr\n6XcdOQ6i90PLMa2OjVwHoo5V7R1L6/Pa2XbcF3C8Y9vZ3+5vLEe5zteISR03JiGOQ+k0+obA+EQH\n0vWsWrWKP/7xj5SVldG/f39uuOEG5s6dS01NDbfffjsbN24kNTWV6667jiVLlgChpOb+++9n5cqV\nWJbFokWL+NnPfoYhzT6/dTRNCzVtdqWRlJYftc+2g5j+KsxAVejZX4UVqMEM1mEFa7HNhsixhmGT\n7PWR7PXFfG/L0jFNg6DpwDSN0CPgwG8amLoDM9XA8rqweudi24OxLB2lDDRNR7c1lAWaaaOCFviD\n0BRA8/vQTRPNNDGUhWGbGA0mRl0QXVWgq0NoykJXdvhhoSsLLbIcetaw0R2OSNKrNSe+zcmvy4Xu\ndKI5XWhOJ5rTie5qXg7tMzwuyEqjwWehDCeawwDDQHM40Awj1MzaCC87DLTwMoYRmVJSCBHHBNfv\n93Pttddy7bXXsmTJEl588UV++MMf8uabb5KcnBw5bufOnfzyl7/kscceY8SIEfz617/mZz/7GY8+\n+mi8Qu0ULocOduiLQEAPJ6hWEAglh7oeGhVQWb6oBNds1STQ5Qyd7w8ef+Co1gltk+kjyZGEUgp/\n0KLRb9IYMGnwmzT6LZoCJo0BiyZfkPqgn8ZAI43BJnxmKBEN2D4CdgBT+TGVH0sLYBNA6UGUHgQj\niGaYoWe985owKiv0HirbCL2XVvOyA2w9vBxqCq7Czx1bb52gaqH9CpTSW21rJ5ntSDIZqyMvqYX+\n0+6dTvTtY75ejAee2MM65e0WPZCuoY/vk+goupzCwkJuv/12HnvsMSZOnMj777/PsmXLeOedd1ix\nYgVer5f333+fL774gquvvprhw4dTUFDAU089xbp163jppZfQNI1rrrmGxx57jKuvvjrRL0l0Ibru\nxJXUB1dS+//2lLKwgvVYwbrQI1CLZTZgm01YViO2GXpY4ecjf001DBvDsHG7g50Sv22Hynzb1rFt\nD0pp2HZomkfb1lC2jh3eplRoGaWhVOiH2VAvolAXI2WHww8/lAqCCqBUQ2hbAPARdQ6tlrVwFycA\nTamW7i0q9B9NNS8qtNDR4a8KLd9LWpLeVuvhAzWlobSWbxZa+HtG8z5arYMWvqwWtS1yvBaanrJ1\npYzWcttW50RtbL4p7R+qEV3ea0csarHd46hbwj/WOA3MoBXdpeu4Z34dJ+g64ctomobDoWOadjtd\ntk7MvTrjNxNNd1Awdz54U0/8xVuJW4K7ceNGdF3n0ksvBeDCCy/kH//4B+vXr2fBggWR415++WXO\nOOMMxo8P/ex+8803M336dMrLy7t1La7HZYAVersbPeGmxlWVkDqE+lo/9bUBnAZUlm6nbncAkoYD\n8PbmTWTbdQQ1jf2HDUDni+Jqfv/yOvwWBE2NoKlhWhA0wbTAMsHCB8NC9779n/+H7fOCHgDDRHME\nwRFEM4Kh5ciz2ZKgGuHHMcSS2ikFWA6U5Qy9fsuJsp0QfmgqtB56dqEpJ+BAwwHKgaY5AAdoDgxN\nBz38Aa2HP2x1LbTuAK2573L4Q7jlM77Vstbqg1zToj4oWj6rW5Yj14m86OgP2LafpdoR60ccd+QH\ndtTnfE/J3LpPP82O0nrwa+tpdGxGeEqB0YkOpUvJy8tjw4YNJCcnY5om5eXlJCcn43K5ePPNN3nj\njTdwu92cfPLJnHvuubzwwgsUFBTw4osv8v3vf58+fUKJyzXXXMMDDzwgCa7oEE0zcLjScbjSj3us\nUgpl+ULJru1HWX7s8EPZfmzLF1q2AqFl249tBbCtAMoyARPLDKCUCQRj+vwOdb+1CTVMkFkohOgM\nm9aWMPGc6zv1HnFLcAsLC8nPj27KkpeXx549e6K27dmzhwkTJkTWMzMzSU9Pp7CwsFsnuL3SPdhF\noVrVOm8owfXUlEH4B4x9+5wMGwoOo4bM1E2kmv2oI4Xtzj5A6AuFP7UJKEcp+Pf24zVTNvAEXWjO\nAEbO7uPlqkentFAyqkJJqKYcaDgxcKLjwMCJoTkwNAdO3cChO3BqDtyGjls3cBkaTkNhaDaGptAI\nPXQUmma3LKPQsNAxI8e07DvyuOjl5nVQaJpqbszb7nNzAaehInmYFvl59Yj9kd9EiTq3+WfWyHWj\nrtOhhrhHrB/tTBV16LGu3/qKWtSG6HtptLwCjnNce3pKKg49OR0/mp70f+/olK1RsrsvtO0F862X\nnJxMcXEx8+fPx7ZtVqxYQVFREQ6Hg9zc3MhxeXl5rF69GgiVzcOGDYvaV1hYiFKqB/04J7oSTdPQ\nHEnoX6OLlWFoZGWlUFlZHxm3RCkrPCJ0EGUHse1gKPlVFsq2UMoKLSu71bKFbVnYtolthR+2hW2Z\nKNvCVhbKtkMPZYdmxVDh9cggk3a4VrZ5W0vVroqq5g0t61p4dg3VeiDJqOpbWlfnalr0eujNa7Xc\nfFyr5ejK0NbntJx3zH/VzTuP/NLRvK51bsl63E+c4x7w9eOTj7tvxrY1kpP7dfp94pbgNjY2kpQU\n/SHl8Xjw+aL7XjQ1NeHxeKK2JSUl0dTUFNN9NE3jRAyAp4drA3X9xPwlnzKmL+9uLcGq7s2egQep\nTGsko/YwGU1lVCflUFTcjwH9D5LkCYSO17ew1p6GhYGmbJyYJGXYBHs7qT8cDDUHcYBhgMMAw6Fw\nGAqnoXDooUegbhg1GTtRmsKwHTiUA4dt4FAGLmXgsnXcSsdt6bhtDbel4zI1nJaO09JxWTqGrYWa\n8Lb6fFVKQ9kq3EzXDje7CUbyMKW05s/pyHkKre3nMrQ6p9XHe1S/1dCzFrlGS71xpEmG0lqOjbpH\nq3vSsu/rJ6BttXutrznSbOy1g9++lKw7kDKva9GUjZlcfMI+w3uafv36sWXLFjZt2sTy5cv5z//8\nzzZlb+sy+siyOSkpCdu2CQQCuN3u496vq5bN8SJxx1f7cYdbhOFp75QuQdc1MjKSqa5uwLa7T1kv\nccdfd429ddydKW4JblJSUptk1ufz4fV6o7YdLek98rijyc5O+WaBHiEjI/n4B8XgO1kp/LMgFzgv\ntOGK0NOsqKOWRJYmARe1d6HzTkg4QgghvsUc4dFep0+fzrx589i2bRt+vz/qmNZltMfjidrf1NSE\nw+GIKbmFrls2x5vEHV8Sd3xJ3PHXXWPv7LjjNnfM0KFDKSwsjNpWWFgY1eQJID8/P+q4yspKampq\n2jRvFkIIIUTHrF+/nquuuipqWzAYZNCgQQSDQUpKSiLbW5fRR5bNhYWFDB06NC4xCyGEEB0RtwR3\n+vTpBAIBnnjiCYLBIM899xzl5eXMnDkz6rhzzz2X1atXs2nTJvx+P/fffz+nnnoqmZmZ8QpVCCGE\n6JFGjx7Ntm3beOGFF7Btm/Xr17N+/XouuugizjjjDO677z6amprYunUrr7zyCgsXLgTgvPPO469/\n/StlZWWUl5fzyCOPsGjRogS/GiGEEKItTbUdW7rT7Ny5kxUrVvDFF18wePBgVqxYQUFBAXfeeScA\nv/rVr4DQHH0PPPAAhw8fZvLkydx999306nWUmcKFEEIIEbNNmzbx29/+lr179zJkyBBuueUWpk2b\nRnV1Nb/85S/54IMP8Hq9/OhHP+LCCy8EwLIsHnzwQZ5//nmCwSALFy6UeXCFEEJ0SXFNcIUQQggh\nhBBCiM4StybKQgghhBBCCCFEZ5IEVwghhBBCCCFEjyAJrhBCCCGEEEKIHkESXCGEEEIIIYQQPYIk\nuO3YsWMHF154IQUFBSxatIjNmzcnOqQO2bp1a5vpl7qyTZs2sWTJEiZNmsTcuXN59tlnEx1STFat\nWsXZZ5/NhAkTOOecc3jzzTcTHVKHlJeXM336dNauXZvoUGLy17/+lbFjxzJhwoTIY9OmTYkO67jK\nysq45pprmDhxIqeeeiqPP/54okOKyUsvvRT1Xk+YMIGRI0dyxx13JDq04/r000/57ne/y8SJE5k/\nfz4vv/xyokMSJ4CUzfElZXNiSNkcH1I2x19cy2Ylovh8PjVr1iz11FNPqUAgoFauXKmmTZum6uvr\nEx3acdm2rVauXKkmTZqkpk6dmuhwYlJdXa2mTJmiXnrpJWVZltq2bZuaMmWK2rBhQ6JDO6Y9e/ao\n8ePHq08++UQppdSGDRvUmDFjVEVFRYIji92yZcvUyJEj1dtvv53oUGJy4403qr/85S+JDqNDbNtW\nixcvVvfcc48KBALqyy+/VFOmTIn83XQnGzZsUDNmzFClpaWJDuWYTNNU06ZNU6+99ppSSqmPP/5Y\njR49WhUXFyc4MvFNSNkcX1I2J46UzZ1Pyub4i3fZLDW4R9i4cSO6rnPppZfidDq58MILyc7OZv36\n9YkO7bj+9Kc/8fjjj3PttdcmOpSYlZSUMHv2bBYuXIiu64wZM4ZTTjmFTz/9NNGhHVNeXh4bNmxg\n4sSJmKZJeXk5ycnJuFyuRIcWk2eeeYakpCT69euX6FBi9vnnnzNq1KhEh9EhW7Zs4dChQ9x88804\nnU6GDx/Os88+S15eXqJD65CGhgZuu+02VqxYQU5OTqLDOaba2loqKyuxLAulFJqm4XQ6Zb7Wbk7K\n5viSsjkxpGyODymb4y/eZbMkuEcoLCwkPz8/alteXh579uxJUESxu+CCC3jxxRcZN25cokOJ2ahR\no7j33nsj6zU1NWzatImRI0cmMKrYJCcnU1xczMknn8wtt9zCDTfcQEpKSqLDOq7CwkL+9re/sWLF\nikSHErOmpiYKCwt5/PHHmTFjBmeffTbPPfdcosM6ru3btzN8+HDuvfdeZsyYwfz589myZQuZmZmJ\nDq1D/vKXv3DSSScxd+7cRIdyXJmZmVx66aXceOONjBkzhssuu4w77rijW31hFG1J2RxfUjbHn5TN\n8SNlc/zFu2x2dMpVu7HGxkaSkpKitnk8Hnw+X4Iiil2fPn0SHcI3UldXx7XXXsuYMWOYM2dOosOJ\nSb9+/diyZQubNm1i+fLlDB48mOnTpyc6rKMyTZNbbrmFn//852RkZCQ6nJiVl5czadIkLrnkEh58\n8EG2bt3KtddeS+/evZk9e3aiwzuqmpoaPvzwQ6ZNm8batWvZtm0bP/jBD8jNzWXy5MmJDi8mDQ0N\nPPnkkzz66KOJDiUmtm3j8Xh44IEHmDNnDu+//z433XQTY8aM6RZfzkX7pGxOHCmbO5+UzfElZXP8\nxbtslhrcIyQlJbUpMH0+H16vN0ERfTsUFxdz8cUXk56ezkMPPYSud48/TYfDgdPpZPr06cybN4+3\n3nor0SEd08MPP8yoUaO6dMHTntzcXJ588klmz56Ny+Vi8uTJLFq0qMu/3y6Xi/T0dK655hpcLldk\nYIWuHndrb775Jv3796egoCDRocRk9erVbN26lbPOOguXy8Vpp53GaaedxgsvvJDo0MQ3IGVzYkjZ\nHB9SNseXlM3xF++yuXt8UsXR0KFDKSwsjNpWWFjIsGHDEhRRz7d9+3a+973vMXPmTB5++GE8Hk+i\nQzqu9evXc9VVV0VtCwaDpKamJiagGK1atYpXX32VyZMnM3nyZEpKSrjxxhv585//nOjQjmn79u1t\nYvT7/V2+X1VeXh6WZWFZVmRbc/+T7mLt2rWcffbZiQ4jZqWlpQQCgahtDodD+uB2c1I2x5+UzfEj\nZXN8Sdkcf3Evmztl6KpuzO/3q5kzZ6rHH388aqTGhoaGRIcWs40bN3abkRoPHz6spk2bph555JFE\nh9Ihhw4dUpMmTVL/+te/lGVZat26dWrixIlq165diQ6tQ04//fRuMVLjnj171Lhx49Rrr72mLMtS\n77//viooKFDbtm1LdGjH1NTUpGbNmqX+8Ic/qGAwqD755BNVUFCgPvvss0SHFrPTTjtNffDBB4kO\nI2Y7d+5UY8aMUc8995yybVt9+OGHasKECWrr1q2JDk18A1I2x5eUzYklZXPnkrI5/uJdNkuC247P\nP/9cXXTRRaqgoEAtWrSoW/3BK9W9CtH//d//VSeddJIqKCiIetx///2JDu24Pv74Y7V48WI1YcIE\ntXjx4m71QdOsuxSiSin11ltvqXPPPVeNHz9ezZs3LzLUfFe3d+9etXTpUjVlyhR1+umnq+eeey7R\nIcXMNE01YsSIbvfl8K233lLnnXeemjBhgjrnnHPU6tWrEx2SOAGkbI4fKZsTS8rmzidlc/zFs2zW\nlOpG9fFCCCGEEEIIIcRRSB9cIYQQQgghhBA9giS4QgghhBBCCCF6BElwhRBCCCGEEEL0CJLgCiGE\nEEIIIYToESTBFUIIIYQQQgjRI0iCK4QQQgghhBCiR5AEV4gEuO222xgxYsRRH3/84x/58MMPGTFi\nBH6/v9Pj2b9/f+Te119//dc+f/fu3cc99uabb47cK5bjhRBCiHiQslnKZtEzyDy4QiRAXV0dPp8P\ngM8++4wf//jHvP3227hcLgC8Xi9Op5Oamhp69+7d6fHs37+fM844g6eeeoqTTjqJtLS0Dp1vWRaV\nlZVkZWVhGMYxj62rq6OwsJAlS5awatUq8vPzv0noQgghxAkhZbOUzaJncCQ6ACG+jVJTU0lNTQUg\nPT0dgOzsbNxud9Rx8ShAW8vMzOxwAQpgGEbMsaamppKVldXhewghhBCdScpmKZtFzyBNlIXooo5s\nBjVixAhWrVrFwoULOfnkk1m6dCmlpaX89Kc/paCggHnz5rFx48bI+QcPHuT6669nwoQJzJo1ixUr\nVtDQ0BDTvZubNa1bt44zzzyT8ePHc8MNN1BSUsKyZcsYP348ixYtYufOnVHHNzdrmjNnDk888QSX\nX34548aNY/78+axfv/4Ev0NCCCFEfEnZLETXJwmuEN3Ifffdx5133smTTz7J9u3bOe+88xg1ahTP\nP/88w4YN46677gJAKcWPfvQjnE4nK1eu5KGHHmLnzp3cfvvtHbrfgw8+yB/+8Acefvhh1qxZwwUX\nXMCCBQtYuXIlHo+H3/3ud8c899JLL+XVV19lxIgR3H777QSDwW/0+oUQQoiuRspmIboWaaIsRDdy\nxRVXMGXKFACmTZtGcXExS5cuBeCiiy7i2muvxbIsPvroIwoLC3n66adxOp0A3H333Zx11lmUlZWR\nk5MT0/1++MMfMmbMGABGjhxJ//79Of/88wFYtGgRjz766FHPXbhwIQsWLABg+fLlLFq0iLKyMnJz\nc7/eixdCCCG6ICmbhehaJMEVohsZNGhQZDkpKSmqQPJ4PNi2jWma7N69m/r6eqZOndrmGoWFhTEX\nokfeb+DAgVH3CwQCRz13yJAhkeWUlBQA+ZVYCCFEjyNlsxBdiyS4QnQjDkf0P1ldb7+XgWmaDBo0\nqN1fcTsyOEas92tP86/Trcmg7UIIIXoaKZuF6FqkD64QPVB+fj5lZWWkpqYyePBgBg8ejGma3HPP\nPdTX1yc6PCGEEOJbR8pmIeJDElwheqAZM2aQn5/PjTfeyPbt29m2bRs//elPqaqqok+fPokOTwgh\nhPjWkbJZiPiQBFeIHkjXdR5++GFSUlK4/PLLWbp0KUOGDOGhhx5KdGhCCCHEt5KUzULEh6ak4b0Q\n33r79+/njDPOYNWqVeTn5/e4+wkhhBDdjZTNQnw9UoMrhIioqqqitra2U+9RV1dHZWVlp95DCCGE\n6CmkbBaiYyTBFUJEXHbZZfziF7/o1HvcddddLFmypFPvIYQQQvQUUjYL0THSRFkIIYQQQgghRI8g\nNbhCCCGEEEIIIXoESXCFEEIIIYQQQvQIkuAKIYQQQgghhOgRJMEVQgghhBBCCNEjSIIrhBBCCCGE\nEKJHkARXCCGEEEIIIUSP8P8AOk99z1wDtEQAAAAASUVORK5CYII=\n",
"text/plain": [
""
]
},
"metadata": {},
"output_type": "display_data"
}
],
"source": [
"IC = [C0,T0,Tcf] # initial condition\n",
"t = np.linspace(0,8.0,2000) # simulation time grid\n",
"\n",
"qList = np.linspace(0,200,11)\n",
"\n",
"plt.figure(figsize=(16,4)) # setup figure \n",
"for qc in qList: # for each flowrate q_c\n",
" X = odeint(deriv,IC,t) # perform simulation\n",
" plotReactor(t,X) # plot the results\n",
" \n",
"plt.legend(qList)"
]
},
{
"cell_type": "markdown",
"metadata": {},
"source": [
"The results clearly show a strongly nonlinear behavior for cooling water flowrates in the range from 140 to 160 liters per minute. Here we expand on that range to better understand what is going on."
]
},
{
"cell_type": "code",
"execution_count": 4,
"metadata": {},
"outputs": [
{
"data": {
"text/plain": [
""
]
},
"execution_count": 4,
"metadata": {},
"output_type": "execute_result"
},
{
"data": {
"image/png": "iVBORw0KGgoAAAANSUhEUgAAA7gAAAEhCAYAAABVzwz5AAAABHNCSVQICAgIfAhkiAAAAAlwSFlz\nAAALEgAACxIB0t1+/AAAIABJREFUeJzs3Xd8FNXawPHf7mx2N70SgkgJJQGS0KS3eymK0hFRVEQB\nQRBRUBDkKnjVi+iVpvgqICBgvd6LoqIigoICgiAiRUIHqenZlO1z3j8WVtYEjRpCe76fz5jdM2fm\nPDOSnX0yZ84xKKUUQgghhBBCCCHEZc54sQMQQgghhBBCCCHKgyS4QgghhBBCCCGuCJLgCiGEEEII\nIYS4IkiCK4QQQgghhBDiiiAJrhBCCCGEEEKIK4IkuEJcQDJI+R9XXudMzr0QQoiykOtF+ZNzKi4m\n08UOQIjycNddd7F58+aAsqCgIGJiYmjfvj2PPvookZGRFRaPy+Xi2WefpUuXLrRt2/aCtLFv3z4W\nLFjAxo0bycvLo2rVqvTo0YPBgwcTHBx8Qdq8kPLz85kyZQoPPvggtWrV+kv7evvtt8nPz2fEiBGA\n799HXFwcM2fOLI9QhRBC/AlXy7V606ZNDBo06Dfr9O3bl2nTppVbm5eSTZs28b///Y/nn3/+Yoci\nrlKS4IorRps2bXjooYf87x0OBz/++CMvv/wy+fn5zJkzp8JiycjI4K233qJjx44XZP9r1qxh7Nix\nNGvWjAkTJhAdHc3OnTuZN28eGzduZMGCBZjN5gvS9oWyZ88ePv30U0aPHv2X9zV//ny6devmfz9l\nyhSCgoL+8n6FEEL8NVfDtTolJYV3333X//6tt95iw4YNAccWExNTrm1eSt577z2OHz9+scMQVzFJ\ncMUVIyoqisaNGweUtWrViqKiIubOnUtRURGhoaEXKbryk5mZycSJE+nWrRvPPvusv7x169Y0btyY\ngQMH8u6773LXXXddxCgvLXXq1LnYIQghhODquFaHhYUFHOMXX3yB2WwucdxCiAtDnsEVV7ywsDAg\n8HmQL7/8kj59+pCWlkbnzp158803A7ax2Wz885//pEOHDqSmptKuXTumTp2K2+3218nJyWH8+PG0\nbNmS5s2b8+CDD3L69GmOHTtG586dARg2bBgTJ04EfF2h5syZww033EDDhg3p27cvX375pX9/mzZt\nIjk5mXfeeYfWrVvTrl07CgsLSxzP+++/j91uZ/z48SXWNW/enAceeICEhAR/2ZEjR3jggQdo2bIl\nzZo1Y+zYsZw+fdq/fuLEiYwbN4558+bRvn17GjZsyH333RdQB2DJkiV07dqVRo0a0bt374DYlVLM\nmzePTp06kZaWxs0338ymTZtKHNv333/PLbfcQlpaGl27duWLL77wrz/bnatbt2689NJLHDt2jOTk\nZJYsWUKHDh1o0aIFhw8fxuVyMWPGDLp06UJqaiqtWrVi4sSJ/nPVqVMnjh8/zvz58+nUqRPg6xY3\nduxYfzxZWVlMnDiRdu3a0bhxY4YNG8aBAwf861966SVuv/12li1bRpcuXUhLS+POO+9k//79Jc65\nEEKIv+5Ku1aXlVKKuXPn+q+f/fr147vvvvOv37BhA8nJyWzZsoU+ffrQsGFD+vXrx/79+/nkk0/o\n0qULTZs2ZezYsRQXFwdss2HDBrp160ajRo0YOHAg6enpAW1v3bqVAQMGkJaWRocOHZgzZw66rvvX\nd+jQgVmzZtGnTx8aNWrEihUrAN8d2t69e9OwYUOaNGnC0KFD/dfQcePG8dFHH/H999+TnJzMqVOn\nmDlzJh06dAho+8svv/SvB7j99tt5+umnGThwIA0bNmT+/PmA73GsoUOH0rhxY1q1asUzzzyD0+n8\n0+dbXCWUEFeAgQMHqoceeki53W7/YrPZ1FdffaVat26thg0b5q+7du1aVa9ePfXYY4+pdevWqZde\neknVr19fvfHGG/46Q4cOVTfccIP65JNP1IYNG9T06dNVUlKSevfdd5VSSrndbtWzZ0/VqVMntXz5\ncrVmzRrVo0cP1bdvX+V0OtXnn3+ukpKS1KJFi9SRI0eUUkqNHj1aNWnSRC1evFitXbtWjRs3TiUn\nJ6s1a9YopZT69ttvVVJSkuratav66quv1CeffFLqsd5zzz3qtttuK9N5OX78uGrRooXq37+/WrVq\nlfr444/V9ddfr7p06aIKCgqUUkpNmDBBNW3aVN12221q9erV6sMPP1TNmzdXDz30kH8/8+fPVw0a\nNFAzZ85U69evV1OmTFEpKSlqx44dSimlpk+frlJTU9Wrr76q1q5dqx5++GGVmpqqdu7cGXBsHTt2\nVEuXLlXr169X99xzj0pLS1O5ubmqoKBAvfHGGyopKUmtWLFCnTx5Uv38888qKSlJtW3bVq1cuVK9\n//77SimlJk+erFq3bq2WLVumvv32WzV//nyVkpKiZsyYoZRSateuXapt27Zq/PjxateuXf5/H2PG\njFFKKVVQUKA6d+6sbrjhBvXxxx+rVatWqX79+qkWLVqoEydOKKWUevHFF1XTpk1Vt27d1CeffKJW\nrVqlOnbsqG655ZYynXchhBAlXU3X6nP9+9//Vh07dix13XPPPafS0tLU3Llz1dq1a9XYsWNVamqq\n+umnn5RSSq1fv95//Vy2bJlavXq1ateunerUqZPq0aOHWrVqlXrjjTdUcnKymjt3bsA2LVu2VPPm\nzVOrV69WN998s2rZsqXKy8tTSvmulampqWrUqFHqq6++UosWLVINGzZUL7zwgj+29u3bq5SUFPX6\n66+rlStXqtOnT6vly5er+vXrq7lz56pvv/1WLVu2TLVr107deeedSimljhw5ooYMGaJ69uyptm3b\nppxOp5oxY4Zq3759wHGvWbNGJSUlqZMnTyqllBowYIBq0KCBmjFjhvrqq6/U/v371cmTJ1WLFi3U\nHXfcoVavXq3+85//qBYtWqixY8f+7jkXVzdJcMUVYeDAgSopKanE0qRJEzVx4kT/B7pSSvXr108N\nGTIkYPuZM2eqli1bKpfLpex2u7rnnnvUxo0bA+r07NlTPfroo0oppVatWqWSkpL8FyClfBeLTp06\nqaNHj/qTs7Vr1yqllPrpp59UUlKSWr58ecA+Bw0apHr16qWU+uWiefbCfD7dunUr84f7v/71L9Wi\nRQt/MquUUkePHlUNGjRQixYtUkr5EtzU1FSVn5/vrzN9+nTVuHFjpZRSXq9XtWjRQv3rX/8K2PfA\ngQPVokWLVG5urkpNTfVfWM+644471MiRIwOO7a233vKvP3jwoEpKSlKfffZZQJ39+/crpZT/HJ5N\nXM8aPXq0+vDDDwPKRowY4b+4KqVUx44d1b///e+AWM8muK+//rpKSUlRP//8s3+9zWZTzZs3V888\n84xSypfgnhuLUkq98847KikpSeXk5CghhBB/3NV0rT7X+RLc7OxslZKSol577TV/ma7r6rbbblOj\nR49WSv2SrJ57/Zw5c6ZKSkpS27Zt85cNHDhQPfDAAwHbvPzyy/71mZmZKi0tTS1cuFAppdSoUaNU\nz549ldfr9dd5++23VUpKiv861759+4Brq1JKvfzyywHXV6V8fwSvV6+e//0jjzyiBgwY4H9f1gT3\n1+do6tSpqk2bNqqoqMhftm7dOpWUlKTS09OVEOcjXZTFFaNdu3b897//5b333mPy5MlYrVZuvvlm\npk6d6h+Vsbi4mJ07d9KhQwc8Ho9/adeuHbm5uezbtw+r1cqiRYto2bIlR48eZe3atcybN4/s7Gx/\nt6dt27aRkJBAvXr1/O03aNCA1atXU61atRKxbd26FYPBwI033hhQ3q1bN/bs2RPQvSkxMfE3j9No\nNOL1est0TrZu3Uq7du38Xb8AqlWrRlpaGt9//31AmxEREf73lStXxm63A3Do0CHy8vJKDMKxdOlS\n7rnnHrZv347L5aJ9+/Ylzum53ZQBGjVq5H99thv12XbO59fn48UXX6Rnz56cPHmS9evX8/rrr3Pg\nwIGALmm/ZevWraSmpnLttdf6y8LDw2nfvj1bt271l4WFhVG7du0/HK8QQojzu1qu1WXxww8/4Ha7\nA66fXq/3d6+fcXFx/mM5Kzo6moKCgoBtunfvHrBNo0aN/Nf+7777jrZt26LresD5dbvdbNu27bzH\nef/99zNu3Djy8vLYsmUL7733HmvXrkXX9TJ/NzmfmjVrBrz/7rvvaNasGWaz2R9jy5YtCQoKKnF+\nhDiXDDIlrhgRERGkpaUB0LBhQ0JDQ5kwYQKVKlXivvvuA3zP6yilmDp1KlOnTi2xj8zMTMA3IMS/\n/vUvTpw4QVxcHI0bN8ZisfifDcrPz/9DIyDm5+cTHh5eYmTj2NhYAIqKivxlv7ffa665xv/MSmmy\nsrKIiorCZDJhs9n8bZwrJiYm4EJttVoD1huNRv+x5uXlAb6LZ2nOru/Tp0+p689NCC0WS0AbQMDz\nPqX59fnYsmULU6ZMYf/+/URFRZGamorVai3znHu/dU527tzpf//rc2IwGMoUrxBCiPO7Wq7VZXH2\n+tmzZ89S17tcLv/rXw+8pWna786WUKlSpYD30dHR5OXloZQiLy+PhQsXsnDhwhLbnT2/UPI4T58+\nzWOPPcb69esJDg4mOTm51Oen/4xft5WXl8euXbv47LPPfjNGIX5NElxxxerTpw/Lly9nzpw53Hjj\njdSoUcP/Ifzwww/Tpk2bEtvUqFGDw4cPM2bMGO644w6GDRvmv0D079/fXy88PJzc3NwS269du9Z/\n4T5XZGQkBQUFuFyugAtSVlaWf31ZtW7dmhdeeIG8vDyioqJKrH/44YcpKirif//7HxEREWRnZ5eo\nk52dzTXXXFOm9sLDwwFKHO/u3bvRNM1/53fBggWlHkd5TldUUFDAyJEjadeuHfPmzaNq1aoAjBkz\nhhMnTpRpHxEREWRkZJQoz87OrtD5F4UQQly51+qyOHv9fP311wN6Wp1lMv21r+n5+fmEhIT43+fk\n5BAXF4fBYCA8PJy+ffvSq1evEtudvbaW5uGHH6agoIAPPviA5ORkjEYjS5cu5ZtvvjnvNgaDocQf\nh88OiPVbwsPDuemmmxg6dGiJdb9O3oU4l3RRFle0xx57DI/Hw/Tp0wFft9OkpCSOHz9OWlqaf8nJ\nyeGll17C6XSye/du3G439913n/8DNCsri7179/o/oBs1asTJkycDRiTcu3cvw4cP5+DBg2iaFhBH\n06ZNUUqV+Cvkp59+Sv369UvcLfwtvXr1wmq18sILL5RYt3HjRr777jtuuukmf7vffPNNwN3an3/+\nmZ07d5Z5uoKz3ZfXrVsXUP7444+zZMkSGjZsiMlkIj8/P+Ccrl+/nrfffrvEuTifstQ7ePAgNpuN\nwYMH+y/ADoeD77//PuDiefbucGmaNm3Kzp07OXbsmL+ssLCQb775RqZwEEKIi+BKvFaXRcOGDdE0\nrcT1c926dbz33nu/eS0ri7Vr1/pfZ2Zmsn37dpo3bw5AkyZNOHr0aEC7ALNmzSInJ+e8+9y+fTt9\n+/alfv36/vjWr18P/NLD6dfnNSQkhLy8PBwOh7/s3EeCzqdJkyYcPHiQlJQUf4yVKlVi+vTpHD16\ntCynQFyl5A6uuKIlJSXRp08fli1bxrZt22jSpAkPPPAAY8eOJTQ0lA4dOnDs2DFeeOEFUlJSqFSp\nEvXq1UPTNKZNm0a/fv3IyMjg1Vdfxel0+rvbdurUiaSkJEaPHs2YMWOwWCzMmjWLpk2b0rRpU/9f\nJr/++muuvfZa6tevT5cuXXjyySfJy8sjMTGRjz/+mE2bNv3hSe1jYmJ48sknefTRRzl9+jT9+vUj\nIiKCrVu3snDhQlq0aMHdd98NwD333MP777/PkCFDGD58OC6Xi9mzZ1O5cmX69etXpvaCgoK49957\nefHFFwkNDaVp06asXLmSffv2MXXqVGJjYxkwYABPPvkkWVlZJCcns3nzZl555RUeeuihMh/X2TvF\nq1evDujKfK7ExERCQkKYPXs2Q4cOpaCggIULF5KRkeHfHnx/Fd++fTs//PBDiaS1X79+LFmyhKFD\nh/LQQw9hNpuZN28euq77z5sQQoiKcyVeq8siPj6e/v37M2XKFDIyMkhKSuLbb7/l1Vdf5ZFHHvnL\n+3/hhRdQShEfH8/LL79MpUqV6Nu3LwAjRoxg0KBBTJo0iW7dupGbm8vMmTMJCwsr8SzsuVJSUnjn\nnXeoVq0aFouFDz74gK+++grwPZJkNpsJDw/n6NGjbNy4kaZNm9K+fXumT5/O5MmTufnmm9m6dSsr\nV6783fiHDBnC8uXLGTVqFAMGDMDlcvHSSy+Rl5dHcnLyXz4/4sold3DFFW/MmDFYrVaef/55ALp2\n7cqMGTPYsGEDw4YN48UXX6R79+68+OKLANSqVYupU6fyww8/MHz4cF566SU6d+7MiBEj2LFjB16v\nF7PZzMKFC0lNTeWJJ55g0qRJ1KtXj5dffhmj0UhYWBhDhgzh3Xff5bnnngNg+vTp3HrrrcybN49R\no0Zx4MABXnnlFbp06fKHj6lHjx4sXrwYTdN45plnGDlyJCtXrmTEiBHMnTuXoKAgwNfN6M033yQ8\nPJxx48bxz3/+k9TUVN5+++1Su0Odz3333ccjjzzCsmXLGDFiBLt27WL+/Pn+gTsmTZrE3XffzeLF\nixk2bBgrVqxgwoQJjBgxosxt1K1b1///obRngsCXuM6ePZuMjAxGjBjBtGnTSE5O5oknnuDw4cP+\n55nuvfde0tPTGTZsGB6PJ2Af4eHhLF26lOTkZB5//HEmTJhAdHQ077777m92yxJCCHHhXInX6rJ4\n4oknGDhwIIsWLeLee+/l008/ZdKkSQwbNuwv73vixIm8/vrrjBs3jvj4eJYuXervsnzdddfx2muv\nceDAAUaOHMmzzz5L8+bNWbBgwW92jZ42bRqVK1dm/PjxTJgwgaKiIl577TXAN2gWwIABA7BYLAwf\nPpz09HTq16/PlClT+O677xg+fDjbtm1j2rRpvxt/9erVeeONN3A4HDz00EP84x//oFq1aixZskQe\nKRK/yaD+6hPhQgghhBBCiEvChg0bGDx4MJ9//jk1atS42OEIUeHkDq4QQgghhBBCiCvCRUlwf/zx\nR9q1a3fe9R9//DGdO3emcePG3Hffff7R64QQQgghhBBCiPOp0C7KSin+97//MW3aNDRNK3WS5j17\n9nDnnXeycOFCkpOTefrpp8nIyGD+/PkVFaYQQgghhBBCiMtQhd7BffXVV1myZMlvDjzz0Ucf0blz\nZxo1aoTVamXcuHF8/fXXchdXCCGEEEIIIcRvqtAEt1+/fixfvrzUybXPOnjwIHXq1PG/j46OJjIy\nkkOHDlVEiEIIIYQQQgghLlMVOg9ufHz879ax2+0lJtIODg72z2n2e5RSGAyGPxWfEEIIIcpfZmZB\nuezHYDAQGxtKdnYRl9MkEBJ3xZK4K5bEXfEu19jLM+5KlcLPu65CE9yysFqtOByOgDK73e6ft+v3\nZGcXYSyH+9JGo4GoqFDy8orQ9cvnH47EXbEk7oolcVe8yzX28ow7Jqbsc0aLC8to9H1BMhrB673Y\n0ZSdxF2xJO6KJXFXvMs19oqK+5JLcGvXrh3QHTknJ4f8/Hxq165dpu2VUuV6wnRd4fVePl/qzpK4\nK5bEXbEk7op3ucZ+ucYthBBCiD/nkktwe/TowcCBA+nXrx9paWnMmDGDDh06EB0dfbFDE0KIq5JS\nCqV0lO5F170opaN7vb5F96LrHnS3G93jRvf6Xns9HrxeN94zZV6vG+X1nKl/ZvGc2Zfu27e/nbNl\nKDj788w6UL56Z8rORHhmOfNaKcBAYsMOVK3T6KKcMyGEEEJcHJdEgjt58mQAnnrqKerXr8/TTz/N\nP/7xDzIzM2nWrBnPPvvsRY5QCCEqjlIKpbtRyoPudeFxO/G4XXjcLrweJx6nE6/LjtflwOt24nW7\n0L1uvF4PuteNrrtRuhfwAvovi0HHgA4GhdGoMBgUBoOOwagwGBVGg8Jg5Ez52QWMxr92B9TAmYuN\ndmapICf3fi4JrhBCCHGVuSgJbsuWLQPmwH3qqacC1nfr1o1u3bpVdFhCCPGHnU1Gdd2J8jrxehw4\n7cW4nA48Lgcetx2vq4hDyo2zuAjd60QpFyg3GDwYDB4MBi8Go47xzKJpZU8oNQNo5gt4gOVIKVDK\n4P8JZ19z5rXBdwMWfnntX3d2mzNlv1r36/e6DpmFsRfhKIUQQghxMV0Sd3CFEOJSoHtd6J4ivJ4i\nPK4inPYinPZC3K5iPK5idFcRutcOyoEBF0bNjaZ5fvcO59lx7yyW8onTqxvQvRperxGvbkTpBnTd\ngPKC0g0oHd973Zfo/fqnruNPAnUddOV7VlVXoCsDulJ41ZlyDCjAiwF1djEYz/lpxHfb14gyaGDQ\nUEYNg8GE0WQCUxCaUQOTCZMpCJPZhGYKQjOZCAoyo5mDMAWZMGkmgjQTmqahGQyYjCZMmkaQ0YhJ\n09CMRkyaEaPRgGYwYDT6Fs1owGg48/PMe4MBTCYjMTFh5OQUls9JF0IIIcRlQRJcIcQVTfe68LoL\n8LpteFwFOIpsOOw2PM5CvJ4ilKcIA3Y0oxOjpp93P6az/ynjp6auG/B4NN/i1fC6jXg9RnSPAd0N\nuhu8HtC9Ct2r8Oq+5049usKjFG5d4VbgUgqPMuDUDTgVOHUjLoMRt0HDjYbXGAQmKwaTBWNQMJrZ\ngiXIjEUzYw0yExxkISTITLD5zGIxYQ4yYjZpmE1GgkxGzEGa76fJSJBJO7Pel1CemyjKYE1CCCGE\nuNRJgiuEuGzpuhuPMxevKx+3Kx97YS4uez4elw3lLcRAEZrRXeq2RsBoAIJK37dS4HabfIsnCI/L\niMdlQHcZ8DpBdyl0l8Lj1vF4Pbi9XlzKgwsPduXFYQSnZsBhMuIwmnAaLbjQcBmsODGjey3gDUJ5\nTFg1K6FBwYSYgwkLNRNiDSLEYiLUaiLaaiLEGkSo1USI1USIxfc62Goi2GwiyFQO86IJIYQQQlwh\nJMEVQlyylNJ9d16dubidebiKszmZnk9xYRbo+RgNjlK30+CXfsHncLs1nC4zLlcQLmcQHocBrwN0\nh44q1vE4PXjcHpxuF07cuM12nBYXxRYvxUFmio1WCrUQ7MZg3FhRmgVUEEqZUboZ5TGDNwiTUSMq\n2EJkmJnwYDNVwszEx4ZiMkBYcBDhIUFEhJgJDzETFhwkSaoQQgghRDmRBFcIcdHpXhceZzZuRxYu\neyb2wgzc9izQ8zAYSnYbNoJvaN4zlAKn04zdYcHpNOO0m/AWg7dIh0IPqtCF2+XEbXTiNtuwW50U\nhXgoCtUoslgpCA3FFhqK8lhRrgiU24pyWVBuC8ppBbvmbzAsOIhKERaiwyxEhf/yMyrMQnS4hagw\nX9JqMPwSoKYZpJuvEEIIIUQFkARXCFFhlPLidmThtp/GVXwae+FJ3I5MDKqoRF2D/z8+TmcQxXYr\nxXYrjiIznkKFbvOCzYXRVgSGYtyWbIpD7BSEKfLDNGxRGvZrw3Br4Xgc4dgLYtCdwShnMMoVjCqw\nggqctybEYiIuykpcbDBxkdYzi+91bKSVYIt8bAohhBBCXKrkm5oQ4oLQvS5cxSdw2U/hKj6Fo/Ak\nXne2bx7Wc5yTw+LxaBQWBVNYFEJRYTBuG+i5bkz5xViLbRiMp3AEF5Ef5iU3UiMvzkReooYrLJgQ\nLRqDqyquIiv5OWZcBVZUZijoJT/mTJqB+OgQEqqHkBBzzhIbQljweR7KFUIIIYS4CuzevZPHHhvH\n8uWfAbBnz26GD78HyznTQdx112AGDRqCzWZj+vRn2bzZNwVsmzZtGTt2AmFhYSX2a7PZeOKJCWzc\nuJHQ0DCGDBlGjx59yj1+SXCFEH+ZUjpuRyauouM4i47jKDyGx5V1ZoKZX5xNZj0eIwUFYdgKQiks\nDMadpzDkOLDk5xPqykFpR/CGO8iN0siJ0chNNJEboYHFQkJoMmZPJN6iMIpyrOQe1HA5TOQGpMo+\nmtFAQqUQqlUKo2qlUK6tFEaV2BDiIoMxGkvWF0IIIYS4WimlWLHiQ+bMmYmm/dLDbe/edFq1asPz\nz88qsc2sWf8GDCxbtgKlFI8/PoFFi+YxevTDJepOm/YM4eGhrFixir179zJu3EPUrFmb1NS0cj0O\nSXCFEH+YrrtxFR3DUXgEZ+ERnEUnQAWOVnw2fSy2W7DZwigoDKUo34Ke5cSUW0iYM48Q91GwFpId\nbSQr1kRmbRNZ0SYcFguR5jiqhV9LjKES4YWhhGWYOZzuJb3YU2pMIRYTNauEU71yuD+hrRIbKgM4\nCSGEEEKUwZIlC1mz5gsGDRrCm28u9pfv3ZtOnTpJpW4zadIUdF3HbDaTmZmB3V5MZGRUiXrFxcWs\nW/cVK1euxGKx0KBBKtdf35XPPlshCa4QouLpXhfOoqM4C4/gKDiCq/gEUHLwJ7fbRF5+uG/JC8Wb\n4SHYlkuEI4tYZzphliJOx5o4USOIU7FBZEWb8GpRhAaFUCO8GinhVbF4YrFlhXD0mJsfj+fjdHkB\nBTj97ZhNRqpXDiexSgSJVXw/46ODAwZ2EkIIIYS4FHm8Ojm20meCKAujZsCpG8jLL0b/ncErYyKs\nmLSy/bG/e/feDBo0hG3btgaU79uXjtlspn//Xni9Xjp1up7hw+/HbDZjMvnSyalT/8mnn35MYmIt\n+vTpV2Lfx44dRdNMVKtWjZycQgCqV6/B2rVflim2P0ISXCFECUop3PbTOAoOYLcdwFl4lNIS2sLC\nYLJzI8nNi6Qw24w5O48oewZRjsMkePI4HadxPMHEntggTsWacVitAMQHx1E7KpHrI2sSbazCiRPw\n08FcVh7Jxe4sAAoC2okMNVO3WhT1qkfRPPUawi0GUJLMCvFnLFiwgJkzZxIU9Mvz5vPnz6du3bpM\nmjSJb7/9lvDwcEaNGkX//v0B32fCjBkzeO+99/B6vfTu3ZvHHnssoAubEEKI3+fx6kya9y1Z+X8+\nwf0j4iKtTB3eqkxJblxcXKnlUVFRNGlyHb179yMnJ5snnpjIggVzGTlytL/OI49M5MEHH2HatKeZ\nNGk8c+bnSuAJAAAgAElEQVTMC9iH3e4IeIYXwGKx4nCU/3mQBFcIAYDudWC37cdh8yW1uqewRB1b\nQQg5uVFk50RSlBlEaH42UfbTVLPvwqIKyIgzc6S6xqbKQZyOjcGr+ZLQqmFVaB1Vm9pRidQIr86J\nUx5+2JfF8rU5ZOTtLdFOXKSV5OpRJFXzLfFRvruzMt2OEH/d7t27GTt2LEOHDg0of/DBBwkJCWHD\nhg2kp6czbNgw6tatS+PGjXnzzTf56quv+PDDDzEYDNx3330sXLiQYcOGXaSjEEIIUVGee26m/3XV\nqtcyaNBg5s59OSDBtVgsWCwW7r//QW69tTc2Wz4REZH+9VarFZfLGbBfp9NBcHBIuccrCa4QVzGP\nq4DC3D3Y8/bgKDwMKvAurcNpJjMrmsysaHIywwizZRNbfIKaxT8S4rZhqxTGvmqwoYqJU7Gx6GcS\n2khzOM1ikqgXU5d6MXUx6VZ+PJjNpo1ZvHbwB+xOb0A7wRaNetWjSU2MISUxhvjo8v+wE0L4/PTT\nT/TrF9h9rKioiC+++ML/bFTDhg3p0aMHH3zwAY0bN2b58uXcfffdxMfHA3Dfffcxe/ZsSXCFEOIP\nMmlGpg5v9Ze7KEdFhpKXX1SuXZRLY7PZWLJkIUOGDCMkJBQAl8uF2ey7Gzt27ChuuWUAbdu2B8Dj\ncaNpGlZrcMB+qlWrhsfj4cSJE1itEQAcPXqExMTEPx3b+UiCK8RVxuPMoyB/Nxn79lKUfzRgna4b\nyMmN9Ce1eraLuOLjJBR/Tz17BiokiOPXhPB1JThaJQ671feBaTQYqR1Zk7S4BtSPSaJKaGWKnR62\npmcy/6v97Dmah1cP/ACuXjmMxnXiSEmMIbFKxF/68BVClI3dbufQoUMsWbKE8ePHExERwdChQ2nQ\noAEmk+/ZqLMSExP5/PPPATh48CB16tQJWHfo0CGUUmV69t1gMGAsh1/xs6OfX26joEvcFUvirlgS\n9x+naRpV4kL/9PZGo4GoqFCCTaDr5d+jTTtzw0LTDERGhvP1119iMCjuv380J0+eYsmShfTufTOa\nZqBevXosWbKAtLQ0NE1jzpxZ3HhjN4KDA7sjh4eH0b7935g+fTrjxz/G/v0HWLVqJTNmzPa3V14k\nwRXiKuD1FFOcu5vi3B04i34OWOfxGMnMiuFURhwZGdGEFuZQqegoqUWbCXEX4IkO50hSKF/ER3Aq\nNghlNABBWDQzTWLr0TCuASmx9QgNCsHh8nU9/u9PO9hxMDsgqdWMBupVj6Jx3Uo0rhNHbKS1gs+C\nECIrK4vrrruO22+/nRdffJEff/yRESNGMHjwYKzWwN9Jq/WXZ6PsdnvA+uDgYHRdx+VylXimqjSx\nsaHlOghcVNSf/2J4MUncFUvirlgSd8W7ULGHh/seDYuJ8c1lO2/ePJ555hluuqkLVquV2267jZEj\nh2EwGHj00XE8//zz3HXXbRiNRm644QbGjRtHSEgIJ06coHv37qxYsYJrrrmG5557lilTptCrVzdC\nQkKYMOFR2rdvXe7xS4IrxBVK193Y89Ipyt2Bw3aAcweJcrlMnM6I5VRGHNlZEUQVniK+cB+1i3/G\n7HXgrRzH4bpRbIoLIjNKA4MOmAk2BdOkUipN4htSN7o2QUYTuq7YfTiHb3Yc5If9Wbjcv7Rj0ow0\nqhNLs+R40mrFEmKVjxwhLqZq1arxxhtv+N83a9aM3r17s2XLFpzOwGejHA4HISG+xwWsVmvAervd\njslkKlNyC5CdXVRud3CjokLJyyu6IHctLhSJu2JJ3BVL4q54Fzr2unVT+PTT1f7RjmNiEpgxY05A\nndzcIv/r++8fw/33j/G/dzh0HI5CrNYIVq/+GoCcnEKMxiBmz54dEPfZNv6os8l3aeTbphBXGJf9\nNIVZ31OUuwPl/eX5Dq/XyKmMWE6cjCczK5rI4gwqF6RTr/AIQboTQ0w0J5tXZ118ISfDPPim5TFh\n1sw0ikvhusqNqB+ThMno+9jIyrPzzY6jfLPjJDm2X774Gg0GGiRG07J+ZZomVSLYIh8zQlwqdu3a\nxfr16xk+fLi/zOl0UqVKFdxuNydOnOCaa64B4NChQ/5uybVr1+bQoUM0atTIv65WrVplblcphdf7\n+/XKStfVZTnQnMRdsSTuiiVxV7zLNfYLHbd88xTiCqB7XRTn7qQw+/szc9T6KAWZWdGcOBnPqYw4\ngh0FJOTvo27hIayeYoxhoRRcV5tvqrj5MTQPDHkAGDBQPzaJVgnNSIurj1kzA76h7Tf/dJp120/w\n0+Fczv1oqnVNBG1TE7iuXjwRIeaKPHwhRBmFhIQwZ84cqlevzg033MCmTZtYsWIFb7zxBgUFBUyf\nPp1nnnmGffv28fHHHzNvnm+ah169erFgwQJatWqFyWRi7ty59O7d+yIfjRBCCFGSJLhCXMY8zlwK\nMjdTmL0Npbv85cXFFn4+nsDPxxPwOk1Uzt9PU9tmwp3ZGAwGgurV5WhqHB+bf6ZYZZ3ZykBccCyt\nqzSnZUJToq1R/v3Zilys/eE4X247Tl7hL+2EBQfRJjWB9g2rULXS+buKCCEuDYmJicyaNYuZM2cy\nceJEKleuzLPPPktKSgpPP/00U6ZM4W9/+xshISGMHz/ef8f2jjvuICsri1tuuQW3203Pnj0ZPHjw\nRT4aIYQQoiRJcIW4zCilcBYeoSBzE/b8dH+5rhs4lRHLz8eqkJUdRZQ3j1pZ31G58DCa8qBFRuFq\n24YN17r53nMEyAMFmkGjaXxD2l7TgjpRtQIGgjl8ysbqLcfY9NNpPOd0JUlNjKFDo2toXDdORj8W\n4jLTqVMnOnXqVKI8KiqK2bNnl7qNpmmMHTuWsWPHXujwhBBCiL9EElwhLhNK6RTn7sKWsQG3/bS/\n3OE0c+RoFY4eq4LHHURC0WFaZH1NuCsHAGtSMtnX1ebTkJ85bt8PHt920cGRtLumJW2qtCTCHH5O\nO4pdh3L45Nsj7Dma5y+3mjXapVWh03XXkhAj89QKIYQQQohLjyS4QlzilO6hKOdHbKfX43Hl+svz\n88M4dKQqJ09VwmzQuTZ7J1Vzf8KsO0HTCG3VmsONqrDSvYtsxxaw+7arFVmTTtXb0qlea2x5dv9D\n/rqu2Lo3k082HuHI6QJ/OwkxIXS+7lrapCbIgFFCCCGEEOKSJt9WhbhE6bqboqzvsWVsxOu2+ctP\nZcRy8NC15OZFEK65qHd6A5VtBzGiYwwNJfzvXdiZFMrnOd9RWHDAv13DuBRuqPF3EiNroGkGTEYN\n8A0ctXHnKT7ZdJTTOcX++rWrRtC9VU0a1onFWI7zVwohhBBCCHGhSIIrxCVG6V4Ks78n/9TX6B7f\n3GBKwYlTlThwsBoFhWFEm+w0PPklcUVHMQBaRASR19/AjqQQVp74hoLTvu2MBiPNKzfh+hp/p0po\n5YB2vLpi485TLFt3kIxcu788tVYM3VvVIKlaVMDzuEIIIYQQQlzqJMEV4hLhe8Z2J3knv8LryjtT\nZuDY8XgOHKpGUXEIlSwO6pxcRXTRcQyAKTqGyK43srtOMAuPryPvSD7gGziq7TUtub7G34ixRv+q\nHcWWPZksX3+Yo6d+6YrcrF483VvVoEZCOEIIIYQQQlyOJMEV4iJTSmHP30v+yTW4HZlnyuD4iXj2\nHqiB3R5MjNVN8ukviC44BoAWGUVMj14cTIri9SOfk33QN6CU0WCkVUIzbqzZmdjg6BJt7TmSy3++\n3M/hcxLbxnXi6NM+keqVJbEVQgghhLja7d69k8ceG8fy5Z8BsGfPboYPvweLxeKvc9ddgxk0aAg2\nm43p059l8+ZNALRp05axYycQFnb+6SPz8/MYPHgQzz03nVq16pR7/JLgCnERuYpPkXt8Jc7CI/6y\nU6djSd9Xk8KiUKKCFck53xCTsx8DYAwJIeam7uQ1S2bh0ZUcTPdtZ8BAs8pN6JbYhfiQuBLtnM4t\n5j9r9rNtX5a/rHHdSvRqW4OaCREX/DiFEEIIIcSlTSnFihUfMmfOTDRN85fv3ZtOq1ZteP75WSW2\nmTXr34CBZctWoJTi8ccnsGjRPEaPfrjUNrZs2cKkSf/g5MnjF+owJMEV4mLwuovIP/klhdnf+8ty\ncqP4Kb0mefkRhIUYaeTcTuz+rRgAg9lMVOfrMXRsw/KTa9myY55/uwaxyfSt3Z1rwhJKtFPscPPh\n+sOs3noMr+4bLblGQji3d65D26bVyckp9I+iLIQQQgghLjyP7iHXkf+nt9c0cJnt5BcX4fX+dt1o\nayQmY9lSviVLFrJmzRcMGjSEN99c7C/fuzedOnWSSt1m0qQp6LqO2WwmMzMDu72YyMioUutu376N\nxx+fyP33P8hTT00uU0x/hiS4QlQgpXspyPqO/FNrUV4nAHZHCDt3J5KRGUNQkEYD83Hid3yBpnyf\nWOGt2xDVuw9rCraz6seXceu+iWyrhFbm5jo9aBCbXKIdXVes3X6C99cdpNDuBiAqzEy/v9WmdWoC\nQSZjBR2xEEIIIYQ4y6N7eOrbf5PtyP39yuUg1hrN5Fbjy5Tkdu/em0GDhrBt29aA8n370jGbzfTv\n3wuv10unTtczfPj9mM1mTCbffqdO/SeffvoxiYm16NOnX6n7T0yszerVqyku9kiCK8SVwFF4lNyf\nV/ifs/XqJvbuq8bhI1XRlZHESCfX7vwIs9M3ArKlZiLxt9/J4SiduXsXkWnPBiAsKJQetbrSpkpz\nNKNWop0jpwpYsjKdQyd9UwuZTUZubFmdm1rWwGIuWV8IIYQQQoi4uJKPuQFERUXRpMl19O7dj5yc\nbJ54YiILFsxl5MjR/jqPPDKRBx98hGnTnmbSpPHMmTOvxH4iIiKwWq0UFxdesGMASXCFuOB0j528\nE6v93ZGVguMnq/BTeg1cLjPxsUHU+fkrgvfvA0CLjCTu5v7oTVN468AKvj/yI+B7zvbv17ale63r\nCTYFl2jH7vTw/rqDrP7+GOpMr+MW9eO5tWMdYiKsFXOwQgghhBDivExGE5Nbjf/LXZQjo0LJzyvf\nLsrn89xzM/2vq1a9lkGDBjN37ssBCa7FYsFisXD//Q9y6629sdnyiYiI/Evt/lmS4ApxgSilKM7d\nRe7xleieIgCK7OH8sL02efkRWCwaTSJOEr3pEwwABgNRnbsQ06sP32T/wEebZ+A40425ZkR1BiT3\npVp41VLb2ZKeyVtf7CW/0AVA5ehgBnZNJqVmTEUdrhBCCCGEKAOT0USlkNg/vb2mGYgJC8PsCr7g\nY6nYbDaWLFnIkCHDCAkJBcDlcmE2+0ZUHjt2FLfcMoC2bdsD4PG40TQNq7XkzZiKIgmuEBeAx2Uj\n5+ePcdj2A6Arjb37qnPwcFWUMlIrwUj1Hz9As/m6HVuqVafy3YOxxYfx4k+LOZB/GIAQUzC9a99E\nm2taYDSUfG42v8jFGyvT2brX1+3ZpBnp3roG3VpVJ8gk3ZGFEEIIIcSfFxYWxrp1X6KUYuTI0Zw6\ndZIlSxbSq1dfAJKS6rF48QIaNEhF0zTmzJlF167dMJvNFy3mCk1wd+/ezeTJk9m/fz81atTgn//8\nJ40bNy5R77333uPVV18lLy+PunXr8vjjj5OamlqRoQrxpyilKMr5kdzjn/kHkcrNj2Pb9kTs9mAi\noyykuPcQ/M1awDc6cmzvvkR27sLaExv5cPNn/kGkmlduQr+6PQk3l5xHTCnFpp9O8+bneyly+OrX\nrxHNoK7JVI4JqaCjFUIIIYQQVzKj0chzz81k1qwX6N69MxaLld69b6Z//9sBGDr0Pv7v/2YzaNBt\nGI1G/va3jowc+SAAp06d4q67+rN06XskJJSc7eNCqbAE1+l0MmLECEaMGEH//v1Zvnw5I0eO5Isv\nviA0NNRfb8+ePbzwwgu888471KhRg9dee42HHnqI1atXV1SoQvwpXnchOUc/xm7bC4CuzPy4I5Hj\nJ+MxGAykJJpJ+PYdKPA9cxGSkkrlu+4mJ1gx84d5HLL55rSNNIdze71+pMU1KLWd/EInSz/fy/dn\n7tpazRoDOtelfcMqGAyGCjhSIYQQQghxpWratBkrVvySeyUm1mL27P8rta7ZbGbMmPGMGTO+xLqE\nhARWrfq61O2++WZL+QRbigpLcL/99luMRiN33HEHALfccguLFy9m7dq1dOvWzV/vyJEj6LqO1+tF\nKYXRaMRqlQFyxKWtMGcn2Uc+QffaAcjNr8TWbbVwOi1ERltppPYStGoNAAaLhUq33k5E+w6sO7GR\nD3au8N+1bZlwHbfU7UlIUOl3YTf/dJqlK9P9d21TE2O4+8Z6xEbK74gQQgghhBAVluAeOnSI2rVr\nB5QlJiZy8ODBgLJ27dpRs2ZNunfvjqZphIaGsmTJkooKU4g/RPc6ObTjI3JOnh0hOYgdu2vx87F4\nwECD2iFcs/ldVK7vWdvgpGQqDx6KMzKEV3e8zq7sPQBEmiO4o14/UuPql9qO3enhrVV7Wb/zlG8/\nFo3bOsldWyGEEEIIIc5VYQlucXExwcGBo2lZrVYcDkdAmdPppE6dOkyePJmkpCTmz5/PAw88wIoV\nK8p0J9dgMGAsORbPH2Y0GgJ+Xi4k7orjLDpBxsH/4nH6JuouLK7Epu8ScTisRERauS4qA+Pni1FK\nYQgKIv6W/kR3uZ5dOXtZvPlVCly+OcCaVm7InfX7EXqeu7YHjufz6vJdZOT67g43qBnNvT0bEPsX\npv65HM83SNwXw+Ua++UatxBCCCH+mgpLcIODg0sksw6Hg5CQwC/1c+bMISEhgbS0NABGjRrFf/7z\nHzZs2ECnTp1+t53Y2NByvaMVFRX6+5UuQRL3haOUzukj6zix71NQOmBk/+HapKcnAAbqN4ilVvqn\nOLbuBiCkZg2SHxmLqWoCb2xfxmf7vgLAYrIwtOlt/K1mq1L/zXp1xX/X7OWtlenoukIzGrjrpvr0\n/XudcvvSfjmc79JI3BXvco39co1bCCGEEH9OhSW4tWrV4o033ggoO3ToED169AgoO3HiRMCdXoPB\ngKZpaFrZpjzJzi4qtzu4UVGh5OUVoesXdn6p8iRxX1gedyFZhz7Abjvge+8N59vNdci3hRNk1miR\nZCL481dwFBcDEN2pM/EDBnDYlcf8z6ZyvNDXxbhmRDWGpN1BfEgcublFJdrJsTmYu3wXe47mAZAQ\nE8KIPikkVokgL69k/T/qcjnfvyZxV7zLNfbyjDsmpuRI5kIIIYS4NFVYgtu6dWtcLhdLly5lwIAB\nLF++nKysLNq1axdQ7+9//zszZ86kW7duJCcns3TpUrxeL9ddd12Z2lFK4fWWX9y6ri74BMoXgsRd\n/hwFh8k6/D90jy/BzMqtypatNfB6TVRKCKOJSsf7wSoUYAwJJWHwEMKaXMeW09t5c897OL0uDBjo\nWqMj3RKvRzNqpR7rrkM5zP1wF4V2NwAdGlXh9s5JWMyl1/8rLuXz/Vsk7op3ucZ+ucYthBBCiD+n\nwhJcs9nM/PnzefLJJ5kxYwY1atTglVdeISQkhMmTJwPw1FNPcdttt2Gz2Rg9ejQ2m4369evz2muv\nERYmf0EXF4dSioKMjeSdWA0oIIhde+pw+EglAFq0qELshndxHfYNmBaclEzCvcMhKpL/7P2Atcc2\nABBuDmNwgztIjqlTaju6rvhow2E+/OYQCt9AUoNvqk+zevEVcJRCCCGEEEJc/ioswQWoV68e77zz\nTonyp556yv/aYDAwfPhwhg8fXpGhCVEq3esk++iH2PN+AsCjR/LNhroUFYVgtmi0axRC0Ecv4Srw\nDRgV06Mnsb36kuPMZ8HWVzhS8DMAdaISGZJyJ5GWiFLbsRW7mP/RbnYdygGgenwYI/umUjm69IGn\nhBBCCCGEECVVaIIrxOXEZc8g69B7eJy+KX7yCqry7aaaeL0asfGhtIw8hfPthXiUwhgSQsK9wwlr\n2JidWT+xePc7FHt8ox7fUKMjPRJvQDOW/hz5/mP5vLJ8J7kFTsDXJfmOLkmYg8r23LkQQgghhBDl\nZffunTz22DiWL/8MgD17djN8+D1YLBZ/nbvuGsygQUOw2WxMn/4smzdvAqBNm7aMHTuh1N63GRmn\nefzxR/nuuy2YTCY6duzMqFFjMJvN5Rq/JLhClKIodyc5Rz9C6W58oyTXJT3dN7dtUv046h7+AseG\nHwAITUwkYcT9GGJi+fTQF3x86HMAQkzBDGpwG2lxDUptQynFF1uO8Z8v9+PVFWaTkbu6JtM2rUoF\nHaUQQgghhBA+SilWrPiQOXNmBgzwu3dvOq1ateH552eV2GbWrH8DBpYtW4FSiscfn8CiRfMYPfrh\nEnWffPIJGjSox4cffkp+vo3HHhvH66+/xvDh95frcUiCK8Q5lFLkn1yD7fR633tC2bwliazscDTN\nQKtmcYStfB1HViYAke3aU//BkZzOL2DRzjfYnrkTgOrhVbk39S5ig2NKbcft8bL4s3Q27PSNqlw5\nJoRRfVK5Nl6eNRdCCCGEEBVvyZKFrFnzBYMGDeHNNxf7y/fuTadOnaRSt5k0aQq6rmM2m8nMzMBu\nLyYyMqpEPbfbTXCwlZEjR6JpFmJj47jhhhtZt+6rcj8OSXCFOEP3usg+8j72/HQAXN7KrF2XiMtl\nJjTcwt9STLjeewmP04HBZKLSHQOJ+fvfyXTl8/zm/+NE0WkAWiQ05fbkfpi1oFLbyS1wMmfZDg6d\ntAHQNKkSQ7vXJ9giv45CCCGEEFc65fHgzsn509trGtidhbjyi3539pigmBgMprJ9x+zevTeDBg1h\n27atAeX79qVjNpvp378XXq+XTp2uZ/jw+zGbzZjO7Hvq1H/y6acfk5hYiz59+pWMIyiI6dNfJCYm\njJwc39g169d/TZ06dcsU2x8h36iFADyufDIPvoPb7ktS8wpqsmHjtShlJL5KOK2iTlH05n9BKbTI\nKK4Z9SDBtWrxU85eFux4kyK3HaPBSN863el4bTsMBkOp7Rw4ns+c93eQX+gCoE+7RHq0rYnxPPWF\nEEIIIcSVQ3k8HHp8Ip6srAppzxQXR+Iz08qU5MbFxZVaHhUVRZMm19G7dz9ycrJ54omJLFgwl5Ej\nR/vrPPLIRB588BGmTXuaSZPGM2fOvPO2o5Ri1qx/c+TIYSZPfvqPH9TvkARXXPWcRT+TefA/Z+a3\nNXDkeD127owDDNStX4l6p7+h6KONAFhqJnLNqAcxRUXxxdG1fLD/ExSK0KAQhqTcSb2Y8/8V6psf\nT7Jk5R48XoXFrDGsRwOaJlWqmIMUQgghhBDiT3juuZn+11WrXsugQYOZO/flgATXYrFgsVi4//4H\nufXW3ths+URERJbYl8Ph4B//mMD+/fuYM2ce0dGlP873V0iCK65qRTk/kn30I1BeMJjZvrMBx475\npvJp3jKBuPXvUnz4EADhLVpS+Z6heDR4fffbbDntG2SqemRVhqXeRYyl9F9Qr67z7pr9fLHlGACV\noqyM7teQayvJ87ZCCCGEEFcTg8lE4jPT/nIX5cjIUPLLuYtyaWw2G0uWLGTIkGGEhIQC4HK5MJt9\nIyqPHTuKW24ZQNu27QHweNxomobVGlxiX/n5+Qwf/hBms4W5cxeVmgCXB0lwxVWp5GBSEXyzMRlb\nfjCmICMdWsZi+uBVXHl5AMT27UdMtx7kOfOZu30xPxccB6Bp5YaMaTeEYpsbr1eVaKfQ7uaVD3by\n05FcABrUjGZE71TCgkt/PlcIIYQQQlzZDCYT5vj4P729phkIjgnDbiks9ftneQoLC2Pdui9RSjFy\n5GhOnTrJkiUL6dWrLwBJSfVYvHgBDRqkomkac+bMomvXbiWm/lFK8dhj44mLi+Opp57FYLhwaagk\nuOKq8+vBpJyeeNauq43bHUR4hIUO9cD55iy8bjcGi4WEocMJb3odh/KPMm/HYmyuAgwY6FGrK91q\ndcJqslCMu0Q7xzILeel/P5KZ5wDg+mbVuLVTbTSjsUKPVwghhBBCiD/DaDTy3HMzmTXrBbp374zF\nYqV375vp3/92AIYOvY//+7/ZDBp0G0ajkb/9rSMjRz4IwKlTp7jrrv4sXfoemZmn2bZtKxaLhRtu\n6Ojff1JSPV5+eX65xiwJrriqeFx5ZB54F7fDN5hUbkFNNp4ZTCrh2ghaWA9T9NaHAJhiY6n6wBgs\n1aqx+dT3vLnnv3h0DxbNzOCUO0iLa3DewaS2pmfy2se7cbq9mDQDg7rWo11Dmd9WCCGEEEJc2po2\nbcaKFav97xMTazF79v+VWtdsNjNmzHjGjPl/9u47TKrybPz495zpM1tme2cLu0sHQZCOwY5SDKJY\nURRFLIkmmhjNmzdF3yS/JMbEboIFuwYNFhALKtI7LizsAtv7zu5O7zPn98dslaVIX3k+18Xl7MyZ\nc+4ZZ3fOfZ77uZ8HD3osNTWVzz77pvP2+vVbO7son8yRZ5HgCmeN7zaTqqgZwO7dSYDEgCFJFFZ+\niWvHFgAMBYWkLboHOTqKZQdW8GnllwAk6OO5c/gtpEel9nqMsKLw4doKlq2JzNuNjdJyz+xh9E8/\nOXMMBEEQBEEQBEHoIhJc4azw3WZSO4oGUVsbiyTBeWNTifvqddw11QDETJpM8g3z8EshXi5aQpGl\nGIB8cy63D51HlNbU6zE8viCLP97DttJmAPLSY7j7x8OIi9admhcpCIIgCIIgCGc5keAKP2iKomCr\n+wJ70zoAwkSzdt0A7HYjWp2KKaPNyO89TcDhAEki6ZprMV90Ca3eNp779mXqXA0ATEg7j7kDrkQt\n9/4r09Tm4Yl3d1Lb7AJg4tBU5l02AI1adWpeqCAIgiAIgiAIIsEVfrgO10wqxqxncp4f72tPEAqF\nkA0G0hbehWnoMPZby/lX0RKcARcSElcVzOBHmRMPOd92Z2kzf1yyCZcniCTB3Kn5XDwm65DbC4Ig\nCMLZKui3Iss6ZPXBS4gci2qnlzcP1DM8PprLshJPyD4FQejbRIIr/CB9t5lUqy2HDRsjzaTS+8Uy\nirzGMBkAACAASURBVBI873wCgCY5hYx7f4o2LZ11dZt5q+Q9QkoIg1rPbUNuZFBCYa/HUBSFlZuq\neeuL/YTDCia9mjuvHMqQnBO/YLUgCIIgnA5ub4A2h48Yo5YaRx3ekI98cy5KMEjLxx9iyC/ANGQo\nAB63n68/KcUcb2Ts+bkHXej1e5poKHkBlTqKtEGLkFXHP4VnVV0LVn+Q1Q1tjE+JJVYrluEThLOd\nSHCFHxyfs4rm8nc7m0mVVw+guDjSTGrQsCTySj7BU1wEgHHQENIWLkIyGVm670NWVUc6vSUbErlz\n+C2kmHpfoywQDLHkkxLW7oqUMGckmbh39jCS44yn4iUKgiAIwkkXDiv89PGvaGr1cO81A3i56hn8\nIT/3nLOAtKJaWj9cBkDeX/+O2hzHvuImykstAOQWJpKSHtNjf177flDChAJ2XK3fEp005rhjDCld\nnVi3Wxz8KF1cZBaEs51YkFP4QXG27KBx/5L25FbL9m+HU1ycjCRJjB+XQvb6V/G2J7fmCy4i476f\n4dereHbnS53J7YC4fB4Yfc8hk9s2h48/vb69M7kdPyyN/7l5tEhuBUHoUywWC+PHj+fLLyNd4ouK\nihg0aBAjR47s/Pfcc88BkYqVv/3tb4wbN44xY8bw6KOPEgqFTmf4winQ0OqmocVNWFF4d8N2/CE/\nACvKv8BXVdW5nfXrrwBwOXyd9xXvqDtof4oS7LzttGxDUY5/mRB/t6VGtrXYT8g+BUHo28QIrvCD\noChhrLWf42jeAEBYieabdQNwOo3o9GqmnGOE954k4HaDSkXy9TdhPv9H1LsaeaHoFZrckSvO52dO\n4Kr8Gajk3ptDHai18dT7RdickS/5KyfnMn/mMKxW10ldz0sQBOFEe+SRR7BarZ0/79mzhylTpvD8\n888ftO3rr7/OV199xQcffIAkSSxcuJAXX3yR22+//VSGLJxiLm+g83aD1YkuJXL7gK0ctyuz8zHb\n11+ScMUM/L6uBHb/niYmXpiPVtd1qqmEuy6KBLyN+N116EwZxxVjoFtCa/EGqHZ5yY0VF5wF4Wwm\nRnCFPi8c8tJ84M3O5NbjT+HzL4fidBqJSzByUYGH8BtPEXa7kaOiyPzZg5jP/xE7mor4y5YnaXJb\nUEkqrh3wY64pvLLX5FZRFL7cXsuf39iGzelHp1Fx94+H8eMpeciyaCYlCELf8uabb2IwGEhLS+u8\nr7i4mIEDB/a6/bJly7j55ptJTk4mKSmJhQsX8v7775+qcIXTJNj9wq0U7vFYi7W+83bIbsexdQvh\nbtsHA2EO7G3uuUOl56i/07L1uGMMf2fEdkOj7bj3KQhC3yZGcIU+LeBtobnsbYK+yAhsc2sOm7dE\nmkn1yzUz3LUdz/tfA6DNyCTjnp+iSkzggwOfsLJyFQAx2mgWDL2J/uacXo/h84dYsnIv63dHGlYl\nmfXcO3s4mclRJ/8FCoIgnGDl5eW89NJLvPPOO8yePbvz/j179qDVarngggsIh8NMmzaN+++/H61W\nS1lZGfn5+Z3b5ubmUl5ejqIoR9UxXpIk5BNwSb3jgmJfu7DYV+MOh7uSR0numeA6Ha10n2Fr+/Jz\nwkOv6rHNrq21DBmZ1vUZ+U6S7LbuJrHfpchq/bHH2J7gJhu0NHn8fNvqYJo/iXj63vvdVz8nIu5T\n72THvnv3Lh566Od8+OFKAPbsKWbBgpvR6boaw82bdyu33HIrdrudv/zlj2zcGBlomjhxEj//+S+I\nioo+aL/79+/jH//4K8XFezCZTFx55Wzmz19wwlceEQmu0Gd57AdoqVhKOOQFZPaVDaB0XxIAw4cn\nkLHlP3iqKgEwjTiHtNsX4lUpvLDzJYpbI0sH5cZks2DYjZh1sb0eo6HVzdPvF3Wub3tOfiK3TR+E\nSS+6NAqC0PcEg0F+8Ytf8Mgjj2A2m3s8FhcXx9ixY5k7dy4tLS389Kc/5Z///CcPPPAAHo8Hvb4r\nCTEYDITDYfx+f48TnkNJSDCd0BMYs9l0wvZ1KvW1uPUNzq4fpK5kVyXJ0D4HO6qgAOe+fXj274f+\nHgASkky0NLuwNDlpaXRTODhS2+xsjFzlMJmzcdtrUcIBgq5dpOb+6NiDbD/BPz87iZVljdj9QTa0\nOMhLNfe597uDiPvU6qtxw4mPXVEUli5dyp/+9CdUKhXx8ZHBnNraikNOYfm///stOp2G1au/RlEU\nfvKTn/D66y/zq1/9qsd24XCYX/7yZ9xyyy28+uqrNDQ0cM011zBy5HAuvPDCE/o6RIIr9DmKomBv\nXIut/ktAQUHH5q0DabbEolJJjB9uwrD8OfwuF0gSCTNmET99JtWuOhZvew2LtxWASeljmVM4C43c\n+6/Blr1NvLh8D15/CEmC2VPymDYuG1msbysIQh/1zDPPMGjQIM4///yDHutoKAVgNBpZuHAhjz/+\nOA888AB6vR6fr6uBkMfjQa1WH1VyC9DS4jphI7hmswmr1dVjdPFM11fjttkjCatWI5OeYqQRkIJ6\nzkkfgCocaU6mGzIEv92Ov7ERZ00dEEVqZiw6g4a6KitffrKXhBQjkiTh9XgBUDASlTACR/NW6stW\noTYNPeZRXH8wMioc8gWYkGLmk2oLq6ssXJybjMYf7FPvd1/9nIi4v79QKIzT7jvyhocgSRATY8Ru\nd3OkvmpRMTpUqqP7A/zyy4v54ovPmDfvVl577WVaWyMXubZv30lOTv/On7v7xS8eIRxW8HrDNDU1\nYbc70OlMvW77xhvvkpqagNXqorKylmAwhCxre932SDqS796IBFfoU8IhLy2Vy/DYIiOw/kAsa9YX\n4vEYiIrWMS6pBeU/LxNWFGSjkdQFCzENG87Xtet4f99HBJUQaknFNQOuZGL62F6P4QuEeGfVfr7c\nXgtAjFHDwplDGCTWtxUEoY9bvnw5zc3NLF++HACn08nPfvYz7rzzTqxWK3fffTdRUZGTBp/P15nA\n9u/fn/LyckaMGAFEypzz8vKO+riKonAimy6Hw0qfbOzX1+IOtCePapXMsP5xNDZGBm7j/QORlEiC\nW+Nqot/Ui2h+63V8rVYwRSFJMGp8P+qqrDTW2tlX3Ez/gUmEO5tMyUQnT8Zp2Uk45KGtYR3mtKnH\nFGNHibKExLikWNY2tOEIhFi6t5ZrcpL71Pvdoa99TjqIuI9OKBTmzRc24bB5T8nxomP1XHfHeUeV\n5E6bNpMbb5zP9u2R+fEd70tJSQlarZbZs2cQCoW44IKLueOOu9BqtUiSGpUK/vCH37JixUfk5uYx\na9bsXt9TnU6PJEnMnj2TurpaLrlkGoMHDz/h779oMiX0GX5PIw0l/+5Mbi2tGXzx1VA8HgMZWdFM\nDG5G+fQ/oCjosrLo9z+/RR5UyL93vcq7pcsIKiES9HH87Ny7DpncVjc5+cMrWzqT2/zMWP53/nki\nuRUE4Qfhk08+YevWrWzZsoUtW7aQnp7O448/zu23385nn33GU089RSAQoLKykueee65zju7MmTNZ\nvHgxDQ0NWCwWnn/+eWbNmnWaX41wsgVDHQmuRFyMNnKnIrN2gxejFPl5l7UUw6QJqBMTUaSueYGZ\nOXFk5sQBsP7LAwSDIZT2JlOSrEKtjSGqfR1cR+N6gr62Y4qxYx1cGdCqZC7OSABga4OVAzb3Me1T\nEM5WiYmJvU4nMZvNTJw4mSVL3ubJJ59n27YtLF7cs1z55z9/iBUrvqRfvxwefvjBwx7njTfe5e23\n/0tJyR5eeulfJ/Q1gBjBFfoIV+u3tFZ9hKIEUZAp2ZfPgbIUQGJ4oZGUta8StEWWu4geP4GUG2+m\nytfIi5tfpKW9JPmcpKHcMPBqjBrDQfsPKwqfb6nhP1/tJxhSkCSYMSGHGRNzUJ2IujpBEIQzmCzL\nPPfcczz66KOMGzcOvV7P3LlzufnmmwG4/vrrsVgszJkzh0AgwIwZM5g/f/5pjlo42Tq6KKtlmWDH\nGrZhmRa7D1VAC7hxhb18WvsNU+fMRflgf+R5liYkqYCJF+bzzoubcdi8bFtXRW5W+wiuFFmtIDZl\nMq7WbwkHXbRWLyep//Xfe652x8CPqv15oxJj2Nhso9bl4z9ljfxkSD+0R1meKQingkolc90d5x1X\nibJKJREba8Rmcx9x9PP7lCgfyp///PfO2xkZmcybN5/nn3+aRYvu7bxfp9Oh0+m4666fcM01s7Db\nbcTE9N7jRqfTkZGRyfXXz+Odd97gttsWHld83yUSXOGMFg75aK1ejrutCIBgyMDGzQOw2mLQ6lSc\nl9CKbsUrhBUFSasl+fobMY2fwPLKVaysXEVYCaOWVMwumMGUjPG9fnFarB5e/mQvxRWRq8cJMXpu\nnzGYwizzQdsKgiD8kKxatarzdn5+Pi+//HKv26lUKu6//37uv//+UxSZcCboGMFVqSRC7eXFMUY9\nzYDPE8QAhGX4rOorRoy6G4wNAHh2FRFyDCc+KZqh52ZQtKWWbesrSU+MnNBL7QmurNYTl3kZLRVL\n8ToO4G4rwhQ//HvF2FGi3NEfQ5Yk5uSl8NSuKlp9AT6rbeGKfknH+1YIwgmlUsnExh084HL0z5eI\njzeBfPLLq+12O0uWvMitt96O0RhpauX3+9FqI1NY7r//bubMuZaJEycDEAwGUKlU6PU9X19bWxsL\nF97C+++/R0cKGggEeu22fLzEJS3hjOVz1dKw94XO5LbNlsCXq0dgtcWQnGxgomc9utXvd5YkZ//P\nb3GPHMBftz3NiorPCSthko2J/Hz03ZyfOeGg5DasKHyxtYb/WbypM7k9b1Ayv7t1jEhuBUEQhLNe\nKNQ1BzfYXl6cEG0gOc6ATOQxky6KsBLmtb3vokpsTyR9HhpffxWAsVPyiI0zoCjQ0uQAuhJcAKN5\nMPqYyBJUrdXLv3epckeC232AKt2k57L+qQCsa7SyX5QqC8Ixi4qKYvXqL1m8+AWCwSA1NdUsWfIi\nV1wxA4DCwoG88spi2trasNvtPPXUE1x66eVotdoe+4mLiyMuLo6///3v7VNhKnjjjSVMn37ip7uI\nBFc44yhKGFvDGhpLXyLob0NRZPaW5rFuw2ACAS1DUoMM3bwYuawYAPOFF5P+0COsDuzjT5v/QbUj\nMn/2/MyJ/GrMffSLzjzoGPUtLv78+jZe/6wUXyBEtFHDnbOGsHDmEIxiCSBBEARB6CxRVqnkzhFc\ntaxm/rSByEp7guvPR0KiztVAWyDSCVUmjHPLJmzffI1Gq2Lq5QMACAUDkR1LXaefkiSR0G8GstqI\nEvZjqVjaOVf3SBRFOahEucMV/VNJM+pQgLfLGrD7g8f0HgjC2U6WZf7857+zf/8+rrjiQu66awFT\np17E1VdfB8Btty1k8OAhzJs3l5tuuobU1DTuv/8XADQ0NHDxxZNpaIhUdzz22J9pbGzkiisu5sEH\nf8rcudczbdr0Ex6zKFEWzih+TzPN5cvwuyNJqtdnYvO2Quz2aEwmNcNd2zCu2QaAymwmZd4ttOQk\n8OLO56h21gEQpzNz46CrGRhfcND+Pb4gH62r4NPN1YTaW8KPG5zCdRcVEG3UHrS9IAiCIJytOr4n\n1bLUOQdXLasY0C+OYpUEISirhGHnjOVb5wZcfg96otGlpEBbEU2vv4quXzZp2TmMGt8PKbgDgDaL\nh7iMruOoNNEk9JtJc9lb+N11WGs/Jy7z0iPGF+52+7tL+GlUMjcWpPGPokpcwRBvlzVw64CMgxJh\nQRAONmrUaD7++IvOn3Nz8/jHP57pdVutVst99z3Iffcd3FgqNTWVzz77pvPnlJRUnnvuOVpbnSe1\ntFokuMIZQVHC1Jetou7Ap9B+5ba6NpXde/oTCqnIMfvJLnoXtc8FQMyEiURfNZuPG79h9ZY3UWhP\nVtNGM6dgBga14Tv7V9hQ3Mg7X+7H5vQDEBet48ZLChlZIObmCIIgCMJ3BbuVKHeM4KrkSHmxRo58\n84aR2fFNPDkTs5GUSPIonTsEdfN2gi0t1D37FNm//i1jJudQsiHy3Ir9VmLTnCQkd61jaYgtJDpp\nLI7mjTiaN6IxpBCVcM5h4wt3WwC0t8Q10aBldk4Kb5U1UO7wsLyqmRnZycf+hgiC0CeIBFc47Xyu\nWtpqluN310d+9uvZWZRPsyUeo15mkGMT5v27AFDFmkm+aR7fJgX4sOhpHP5IOVSKMZnrBvyYgrj+\nB+2/uKKVpV+XUV5vByLLHVx6Xj+mj89Bp1UdtL0gCIIgCD2XCeqYg6uW2k8d2xc21hu0+AJhmnYO\nJFuJXEBea9nG7Qvm0/a3JwhaLNQ98yQZ9z9AjFlLyAfBICz/TxGzbxqFKVrXeTxzxkX4PY34nBW0\nVn+MRpeALirrkPGFuiW4h5pzNzwhmiqXl3WNVtY32UjQa5mQIvpsCMIPmUhwhdMmFHRjrVuFq2Vb\n532V1WnsLcklGFKTrWkhp/gT1OEASBLmCy6iefJQ/ln7BXUlkVp+tazmsuwLuSj7fDRyz4/zgVob\n760uY09lV8OK4f0TuO6iAlLijKfmRQqCIAhCH9XbHNyOEVylPcGdPimP4i1+2trC5Kr0ALjCLhZb\nv2DBjTfR8vJLeEpLaHzp38iTIQQgqXDafSx/t4hZN5yDVhf5/pYkFYm5c2gsWUzQ30Zz+dukFMxH\no0/oNb5wtwrHw5UeT8tKpMXrp8Tm5uOqZhJ0GgaYTcfz1giCcAY76iZTXq/3ZMYhnEUUJYSjeTP1\nxU91JrdOl5ENm4ezq7gAkxzm3NpPyN/zIepwAH3/fPjpAt4c4OSZ0tepc0WS23OTR/CbsQ8wLffC\nzuRWURR2l7fyt7e289irWzuT29y0GH5+7Tncd/UIkdwKgiAIwlHo7KIsSwQ7mkxJKhRFgXDksZTE\naBZMHwxA+yYokkKVo5a3ovZjnhnpkOrYtJGgPbJefeGQNAAsTU4+XVbceRwAldpIUt61SCod4aCb\npv2vEfTbe4+v+wiufOgEVyVJXNs/jVSDFgV440A9lQ7P9307BEHoI446wZ0xYwZ79uw5mbEIP3CK\nouBq20X9nmdpq1lBOOQlGFRRvDeP1WtH4bDFMKB1M+fueROzpwF1XBzha2aw9JJ4/tG0jFLrAQDy\nYrN54Nx7uHXoDSQY4oFIGdX63Q387qXN/O3tHexuX/YnI8nEvbOH8et55zIkJ/60vXZBEARB6Gt6\njOC2N5lSyarO8mQASaVi9MBkrr0gn44UMyaQA0Bxawn/zbETPXESACFvZLmehJQYJl0cWRqouqyV\nz76T5GoMSSTlXYckqQkFbDQdeI1Q8OClfnrOwT38a9GpZOYVpBOrURMIK7y8r45alxi8EYQfoqMu\nUQ4EAgetI/p9FRcX85vf/Ib9+/eTnZ3N7373O8455+AGAlu2bOGxxx6joqKCzMxMHn74YcaPH39c\nxxZOH0VR8Nr3Ya3/ioCnof0+qK1PZm9pLgGflkx7CTmW7WjDPmSjEefkkXyW6aLCsxFskf1kRWcw\nLecihicO7vws1re4WL2zjrVFDTg9gc5j5qXHcNl5/RhVmHTYq7qCIAiCIPSu+xzcUPcR3G4JLqpI\nyfIl5/Wj7KtylLBCU3U0qYkjaNDsZKdlN7pzR3CRayRBuQUAd9Euhk0djccVYOu6SspLLXzx4R4u\nmjkIWY6Mveij+pGYezXNZW8T9Fpo2v8ayfk3olJ3VWGFjrJEuYNZp+G2gRm8sKcGZzDES6W1LBiQ\nSapRd8TnCoLQdxx1gjt9+nTmz5/PFVdcQVZWFnq9vsfjc+fOPezzfT4fd955J3feeSdXX301y5Yt\nY9GiRXz++eeYTF3zIBobG1m0aBGPPvool1xyCR9//DH33nsva9asOeiYwplNUcK423Zjb1xLwNvU\neX9jUzwl+3JwOE2kOQ6Q27IdQ9CFbNBjGTmE5dlOWqR90F49lB2TxeU5FzEkYSCSJGF3+dla2syG\n3Q3sq7F17lcCRuQnctnYfhRkxh73BRlBEARBOJsFO5YJUsn4lY45uGqUUNeaspKqq1mjJIFC5F/5\n9lQyRwRo0RWzybKT8KRhXOiLfGc7Nm9B40lk9GWXEwqF2bGxmgN7m5EkiQumD0SliiS5htgCErKv\npKXyfQKeBpr2LYkkuZpI9+XuI7jfXSboUBL1Wm4dkMG/9tbgDob5d0kN8wszyDCJc0xB+KE46gR3\n+fLlGAwGVq1addBjkiQdMcHdsGEDsixz/fXXAzBnzhxeeeUVvv76ay6//PLO7ZYtW8aECRO49NLI\n+mfTp08nNze384qecOYLB704W3fiaN5AyN+VgLa0xlKyLwdrWzQpjjKGtBVhCtgImwyUjO7Hqkw3\nfm0zABISwxMHc37mRArj+mNz+fl6Rx2b9zaxt6qNbt9pxEXrmDQsjcnD00g0G74bjiAIgiAIxyDU\nbQTXc4gRXEnV1QMj3D6kOjw/kdX7LdTszCJ1WAiboYQtrUVMMUejQYGwguW9/xByOhl71dWEQwrf\nbqlh/54m/L4gl/x4CBpNJHE2xQ8FiCS53iYa971Ccv5NqLUxPebgHqlEubtUo475AzJ4qaS2Pcmt\nZV5BOrnR4hxCEH4IjjrB7S2x/T7Ky8vp37/nEi65ubmUlZX1uG/37t2kpKRw9913s2XLFnJycnjk\nkUfQarVHdRxJkjgRuXBHWWtfK289XXErioLfXYe9eQuull0oStfV3YamBMrKM7G1msiwlzLYuht9\n0IUrMYqv88wU5WgIqb2AjEljZEL6GCalj6PVIvNtUStvHNhMdZOzx/G0GpkR+YlMGpbGsP7xqE7T\nBRDxOTm1RNynXl+Nva/GLQhnks45uLJM8DBzcCFyHtDhwnMziU4y8fH6ShqKckgZpMIeXQyEAQlt\nVjbe/cW0ffoJIaeD8Tfdgkots31DFVVlrXz01k4uv3oYOr0GiCS5kqzCUrGUoK+FxtIXSep/HSGl\na7mfox3B7ZBp0nP7wExeLKnFGQzxcmkt1/VPY6DoriwIFBfv4le/eoBlyz4BYO/eYu644xZ0uq5y\n/ptums+8ebdit9v529/+yKZNGwGYMGEi99//S6KionrdN0A4HObee+9kwIBB3HPPfSc8/u+1TFBL\nSwvvvvsuFRUVPPjgg2zcuJGCggIKCgqO+Fy3243B0PPKmF6vP6g7s81mY/Xq1Tz55JM88cQTvPPO\nO9xxxx2sXLmS2NjYIx4nIcF0QktTzX30D92pitvvtdHWsIOWum14nHWd94dCMnUNSZSVZxJqC5Jh\nL2WYfT8qyc++LB07C8zUJ2pAklBJMqNShpKtH0ywNZG9262sXLYLnz/U41hajYoxg1OYPCKDcwcm\no9edOatcic/JqSXiPvX6aux9NW5BOBOEDjkHt6shVMcc3HC3CbEqlcxV5/dHr1Wx9OsyGvf0w5wr\noTJXAPB5XpCp7tEENm7Bvm4tQZuNMQsXoTdoWP/lARpq7bz/6namzRlKbPvKB0bzIJJyr8FS/h9C\nATuNpS/hT5vTdcxjOPdLNepYOCiTxSW1WP1BXt1Xx+VZiUxIMYtpTsJZSVEUPv74A5566u+ouk0/\nKC0tYdy4Cfy///fEQc954om/ABLvvfcxiqLw61//kpdeeoF77/3ZIY/zxhuvsXPndgYMGHQyXsbR\nJ7jFxcXMmzeP/Px8du3axd13383atWv51a9+xXPPPXfEJlAGg+GgZNbr9WI09lyyRavVMmXKFCZN\ninTcu+GGG1i8eDHbtm1j6tSpR4yzpcV1wkZwzWYTVquLcPeF1s5wpyLuUNCNq20PDsu3+FxVdP8K\ncDoNVNakUVebiLmtngL7N8R5GqhL0vDNuXr29YvGq5ORkUlRZ2LwZeCsS2LdJj9rFStg7XGs9EQT\nw/snMCwvnsJ+ZrTqyC+b2+XF7TopL+97EZ+TU0vEfer11dhPZNzx8Ye+Ci0IP2Qdc3BVKplgjzm4\nB4/gdv89k9vrha8Yn4M5SsfLK/ZiK89ELqwEoCXg4JlCLwv045G+Xo979y6qHvsDg+/9KforBvL1\nihLaWtwsfWUbl1w5hMycOAAMsYUkF9xMc9lbhIMuWmo+Ay6KHPMY89EEvZaFgzJZsq+eerePj6st\nNHsDzOiXhEpUgAhnmSVLXmTVqs+ZN+9WXn/9lc77S0tLyM8v7PU5Dz/8v4TDYbRaLc3NTXg8bmJj\nzb1uC7B3714+/vgDpkz50YkOv9NRJ7h//OMfufnmm7n33nsZOXIkAI899hhxcXH89a9/ZenSpYd9\nfl5eHq+99lqP+8rLy5k+fXqP+3Jzc6mqqupxXzgc7lH6cjiKonSvnDlu4bBCKNR3Tuo6nMi4FUUh\n6LXgtpXibN1L0FuHJEX2LQGBgIqGxkRq6pJRat2kOCsY51xNqznMnkIdJdkJOKJUyIoGlSsZf00C\nIWsSrpCm/Qj+zmOlJ5rIz4ilIDOWQdlxxMf0bPpwpv6/EJ+TU0vEfer11dj7atyCcCbotYuyrIIe\nTaYiowo91rJVdY00TByWRmKsnmff39n1HDS4Q16eyiznhukTiV+xgUBjA1WP/Z7M2+9k+tzhrHx/\nNz5vkI/e3smEC/IZNjoDSZLQmTJILbyNprI3CHdbylZSgkDXiNP3EavVcMfATN4pa2CP1cWmZhvN\nXj9z81KJ0Z451WLCD4cSDhEM2I684SGEZQmf20vA6yZ0hIu4ak0sknx0vxtXXDGLefNuZfv2rT3u\n37evBK1Wy9VXzyQUCnHBBRdzxx13odVqUasjvyP/93+/Y8WKj8jNzePKK6/qdf9+v59f/vKXPPTQ\nr1m27P2jiulYHPVv7e7du3n00UcPun/u3LkHJa69GT9+PH6/n1dffZVrr72WZcuWYbFYOkdqO8ya\nNYu5c+fy1VdfMWXKFF5//XV8Ph9jx4492lCFEyAUcOB1VuFoKcfr2IeMo/MxSYqUIDc1x1NXl4i/\n2k+Co5ZC9w6akkLsHaBjRUY0Tr2asNNM2JpAuCqBsDOW7ksvxxg19EuJJjs1msIsM2OGpRPwN/Fu\n9wAAIABJREFU+sXJqCAIgiCcAboSXJlQxwjuQcsERU4le4zgfmfkc0C/OB668Rz8VasBcOwbjLbf\nAfwqG6/G7GPSlUMZs/IAYaeTun/+nbhLLuOqG6ez4r/FtFncrP1iP7VVbUy9fCB6gwa1zkxqwa3U\n7f+MjtMTy75XSM6bg0Z3bGve61QyN+Sn8Um1hTWNVsodHp7cXcXc/qnkxxiPvANBOEpKOETdnqcJ\n+a1H3vgwao5yO5XWTPqgu48qyU1MTOz1frPZzMiR5zJr1lW0trbwP//zEIsXP8+iRfd2bvPznz/E\nT37yc/70pz/w8MMP8tRTLxy0n2effYpJkyYxYsQ5Z0aCGxsbS11dHdnZ2T3u3717N/HxR/5jotVq\n+de//sVvf/tbHn/8cbKzs3n22WcxGo385je/AeD3v/89gwcP5tlnn+Wvf/0r999/P7m5uTz33HM9\nlhISTqyOEVqPswpHSxl+VzUquaupU0dK6vNpaGqOp6UxGqXSSZyjFrOuiPoUia0FGmrjYvH74iJJ\nbbWZsCMOwmqQwGjSEp2hwxSrw2TWY4jRIetkQgo0KApNXjcbd5QTDoUACRnQyjJ6lYxOFfmvQS0T\no1UTo1Fj1mqI0apQi+7agiAIgnBSdDSZUqtkgt1GcJVgLyXKoUMnuABJsVpq22+HfAZs28cQVbiH\nUEwta3T1VExL4qoNMVBdR9unn6ArLWH6LbezdnMbZSXNVOxr4d2XtnDRjEGkZZmR1XqiU6eCoz4S\nq6eBhr0vEJd5KTFJI4/p9cqSxOX9ksg06XmvohFXMMRLJbX8KC2eqenxqEXJsnCW+vOf/955OyMj\nk3nz5vP880/3SHB1Oh06nY677voJ11wzC7vdRkxMV/+krVs3s3XrZt57bylOp5+T6agT3Ouuu47f\n/OY3PPDAAwCUlJR0NoOaP3/+Ue1j4MCBvPXWWwfd//vf/77Hz5MmTTpoZFc4cZRwEL+7DkdrOc6W\nAyihRlSqQOfjHZVF4bCEzRZNa0s0nuowqvo2FM0B3GYnNfkaaqLj8PgHRRLaZjPUR6E2alEb1agT\nNaj7qVEbNaiMaqT2LwV3+z8CfggcFNr3IgMJeg0pBh0pBi3Z0Qb6mfRoVSLpFQThh6mtrY0XXniB\nXbt2EQgc/Ee0t+9YQThWHaWPKlkiFOoYwT3UHNyuEmW5t+9hpes5l4/P4+XPW3DuHYoqyYwuZy81\nGhdPTlCYW5ZP8ub9+CrKqf/j7xgz+2oyLxnE2lUHcNp9LHtjB8PHZDFmcg4dR5QBlcZIOOimtepD\nvPZSYqLmAseWkA5PiCbdpOON/fU0ePx8Wd/KXpuLObkppBl1R96BIByGJKtIH3T3cZUoq2QJs9mI\n1XpiS5R7Y7fbWbLkRW699XaMxsiAo9/vR6uN/C7cf//dzJlzLRMnTgYgGAygUqnQ63s2F/7880+p\nra1hwoQJKIqC1+tFlmWqqip6bV51PI46wb3jjjswmUz86U9/wuPxcM8995CYmMidd97JLbfcckKD\nEk6sUNCDx15JW30pPkcFao0NWY78MsjQOWUlEFDTZo3B2mrE1xjG32rHZ2iiNaaS+ngzzUkJhLzD\nkEJmZH8cao8Wg1GDOl6NyqhB1spIkoREGC1BdFIIvexHJ/vQqSR0Kg0atQ6NWo9GpUUjy6hkCVmS\nkCTQ6TW4PX5CYQVFAV84jC8UxhuK/NcdDGHzB/G2l0yFgWZvgGZvgF1tkdegkiKt/wfHRTEsLgqz\nTnPQ+yEIgtBXPfTQQxQVFTFz5szDLsEgCCdC9y7KQV9k3q36EMsEHWkEV1G6EuAxg9LIyshl8UfF\nVDVl4XHGYizcRVhn580CO8Pis7hgTQuKy43lzdeIzi9g5qwb+GptE20WNzs3VVOxz0LW1BwgcrKf\nNvBOWqs+wmMvxW0toXjd3zBnXITBPPyYOiIn6rUsGpzFJ9UW1jfZqHf7eKa4iqnp8UxJjRMVZMJx\nkWTVMZfTA6hUEjpjFBqvE/kkT+2Liopi9eovURSFRYvupaGhniVLXmTmzB8DUFg4kFdeWczgwUNR\nqVQ89dQTXHrp5Qct8frLXz7Cww//mvj4KFpbnfz+9/9LbKz59C4TVFdXx3XXXccNN9yA2+0mFAoR\nHR1NKBSiqKiIYcOGnfDghO9PURS8LguNB7bTUlcMigWDsat7tbbbhUe3W0dbWwwuixqPxY/DZ8dm\naqTBqKM5NolQbD9U8khU+igMRolMU5BYox+TyoeRNgySFyNeDHgxSl70+NDhR02QHt8lChBs/+eL\n3CWrTehMGWiNmRhi8tFHpZKQEE1rq/OIc3B9oTB2f5AWn59Gj58Gt586t5dmb4CQApVOL5VOLyuq\nLeREG5iQbGZwnOl7r5EnCIJwptmwYQNLlixhxIgRpzsU4SzQMQdXddg5uJEEN9RjmaDeEtxuz5Fk\nspKj+PXNo/loXQUfr6/E9e041OkH0KSXUZTgY/+lRq7cHUNySQPe/fvw/eNRpl48jbLcYezcUoet\nzUP9mgoYFt8+ghtFYt5cXK07aKtZSTDgwlKxDJ1pG3FZV6A1JH/v16+RZWZkJzMkLoqlFY20+YJ8\nXtvKdouDGdlJFMaK6XPCD58sy/z5z3/niSf+yhVXXIhOp2fWrNlcffV1ANx220KeeeYfzJs3F1mW\nOf/8qSxa9BMAGhoauOmmq3n11XdJTU09ZTEfdYJ74YUXsnbtWuLj43ss7VNZWcmNN97Izp07D/Ns\n4WRRlDCOlipq923G56rFZHKj00eusnZfdlhRwO6IwtZiwG1RsNu8tEpemkx2mqJTkbJSidGYiI8K\nkWvwco7kQx/0oQlUovEFUQISgWY1waCaUEgmHJYJKxKOsAG7Evk8yHIYWVKQ5DBqVRitNkCsWSIm\nFgwGPzIOwsFIN4hw0IXHVorHVoqtfhVqbSyetBFooocjaw5/RUunkkkyaEkyaBnYrQu5IxCkwuGh\n1OZmd5sTbyhMhcNDhcNDvE7DJRkJDIuPEmvbCYLQZyUkJKDTiRJJ4dQ45BzcYylRDnd7jqTq3O+V\nk/MYWZDEkpV7Ka8tINSWjC5vNx6jnTfPDVOQnsAlWzyoHW5sKz4k2fwNl108h021OlxS5JgBX4ht\n6ysZPiaTqISRGGPzcDZ+jrVpNz5XNQ17nycqaQyxKZNRab5/UpoXY+QnQ7JZWWNhY5ONFl+Al0vr\nGGQ2cXlWIgl67ZF3Igh9yKhRo/n44y86f87NzeMf/3im1221Wi333fcg99334EGPpaam8tln3/T6\nvEce+e0JibU3h01w33zzTZ5++mkgMjI4Y8aMg5IDl8tFQUHBSQtQ6Mnv91C1ayPN9Xsw6JzExPpQ\nq8OY9GDqtqJOMChjtUbjalFjbwvR6A5gMUjYE4zoUs0kpsskhLyku9zo7E7CbishT4CQNwBKCEUO\nEgoHkcJ+FCWMFAZdMEyMX0EdgrCkJihrIv9UOrxqIz61CbcmCpchkQAHlwabonUMGJpA4SANasmC\nz1WDz1lBKOAg6LfRWLkaWI0+Oo/YtPPRmbK+13sTrVEzLD6aYfHRzMpOZp/NxfomG/vtblp9Ad4q\na2Bdk57ZOSkkG8SXkSAIfc8999zDH/7wBx555BGys7PRaHr+rf1uSZggHI8eywT1GMFtL8eSJKT2\nUt3DdVGGniO4HQluh+zUaB6ZN5o139bzn68O4Nw1HlVSNdqsfexLg8rLjEzao2XoHjshq5XQu/9m\nTL8c5HGzaAWkkMLGteUU76jn3AnZDBqRSv9zbqGmbBuWqhWE/FaczZtwtewgJnk80cnjkVXf73dF\np5KZmZ3M6MQYPqhqpsrpZY/VRYnNxejEGKamxxOrFdOiBOFMcNgEd86cORgMBsLhMA8//DALFy4k\nOjq683FJkjAajYwbN+6kB3q2ctla2LH+U8KeRsyxfqJjvWhlyEjvuZ3Xq8HeasTRChYHNCgyTnM0\nMVojiSYnsWEran+I6CYLAaMNv9FEnS6GcGwScnwGMipQJIJB8AeD+IIBXH4vDq+XoORF0viQtD4k\nvRuN1k5UwI/Z4cLsCJFgDZFmCZDYHERSIhXJPpURhzEZZ84IWjTJ2OwBXA4f29bXsWOjROHQFEZP\nnEZCto6Apx6PvQR3604CPjteRxleRxmG2AHEZV6KWnvoxaIPRS1LDIqLYlBcFPVuHytrLJTa3FQ5\nvTxdXMUVWUmMSYoRo7mCIPQpjz/+OFarlauu6n2NwT179pziiIQfso6yY1mGcPscWrWshpAb6Bq9\nhe/MwT1CifJ3E1yIdDCeMiKdUYVJLFtTzlfbZTytqWgy90NyNauGy2zNNXPBTj/9qpwEqirQmVbD\nlMvRqCJLGDpsXr5aUcL2DVX86NIBZOTmkzZoEY6m9dgb16GE/dgavsZh2UJM8jiiEs9FVukPiuVw\n0k16Fg7MZHuLg5U1FhyBEJua7WyzOBibHMvk1Dixdq4gnGaH/Q3UaDRceeWVAGRmZjJq1KjOxXyF\nk6Ohupxdmz/DqHJiNgeIivGRlXLwdk6HHnurBqtVotGrwqbTEaXWYgz60KUlUpCeQUpyJunmOOKj\nDWjUx9YMQVEUXN4gDS1uKhsdVDY4KC5vodFjxxLdhmxuQc62IOu8aANhshoCDKwNklfjJ8lRQVJR\nBbmSBGOm0txvNKWlbXjdAfZ+28D+PU2MnpjD8DGZxGdk0H/IFdSUb6WtdjUBbyMeWwleRzlxmZdh\nih9xzMlomlHHLYUZlFhdvFfRiCMQ4r+VTTR5/VyelSjm5gqC0Gc8/vjjpzsE4SzSMYIryd3m10pd\nywRJ3SoIepQo99aAqccc3EN3dI0yaLjh4kIuGZPFB2vKWbdbS7AhB3XGfmwJdbw/SU1Go5pJu7yE\n2zvDal1WJvk2UJk+niqLgq3Nw7K3dhAdq2fYuRkMHD6BqMTR2Bu+wWHZQjjowlr3BbaGNUQnjSY6\naSwqzdE3bZMkiVGJMQyLj2JDo42vG1pxB8OsbbSyocnKiIRoJqXEkSo6LgvCaXHYbPXxxx9n0aJF\nGAwG1qxZw5o1aw657c9+9rMTHtzZwNpiYd2q94hRO4mPjyS0hbk9t1EUcNgM2FpVNDtVNIV0qLRG\nUuLiyZ86gqlpaejarxaqVFJnd7IjNWs6GpIkEWXQkJ8ZS35mbHs8CvUtbjbvbWLNt/W0VHiQo9oI\nJtVSllnPgawwmoCB82rVjC7xQUsbbFpFyu6NDL5+HtVyNlvXV+J1B9jwVRkV+y1cNnso8fFRmOIG\no4seiKv1W6y1nxIOeWit+gC/u564zEuRpGPvWjjAbOInQ7L5T3kDJTY36xqtuAIhrs5LEUmuIAh9\nwnnnnXe6QxDOIsHOEdyu84nIOriRXh+Squs0MnTELsqHH8H9riSzgdumD+aycdl8tK6CzXtMBOvy\nUGfspya5gbcv0JDsiySlcjiEtmovBVV7yYjLpDp3CjV2LQ6bl3WrDrB5TQUDh6UyZNQk0gePxd64\nHlfLdpSwD3vjWuxN6zHGDiIq8Vx0UdlHfUFdI8tMTotjTHIMaxusrGu04gmF2WZxsM3ioDDWyHlJ\nsQyINaESa+gKwilz2AR3+/btBAIBDAYD27dvP+R2oszz6Pn9fr5c8S4afzMJcQFi47wMze+ZiIZC\nEg6rAWubimaXCitGcvLyGX3pecRGGQ+x51NHkiTSE03MmpTLjIk5bC9tZtmaCmrK4wnUFGDoV04g\noZK1OWE2ZGm4rm0MSd/sIuxy0fyvZ0m75DKuve3HbFxdwZ6d9TTU2Hln8RZuvHMcWr0qklQnjMAQ\n05+WymV4HQdwWjYTCthIzLn6uNbyMmlU3FiQzgeVTWxutrOz1YFRLTO9X5L4HAuCcEa69tpreeGF\nF4iJiWHu3LmH/Vsl1sEVTpSwohBW2s9P5K7R2cgIbvsazOqDS5R766AMB3dRPloZiSYWzhzCVVPy\n+HRLNd/sjMVX40CdUoE1U4cBsJsk9mXpyK/xYWyrYUDbG2To4mjImUCNkkTAH6Joay1FW2tJyYhh\n4LAR5BZMwGfbisOyGSXkw23djdu6G7UukajEUZjihqDSRB8xPgC9SsWFGQlMTo1ji8XO2oY22vxB\nSm1uSm1uojUqRiXGMDoxRjSkEoRT4LAJ7quvvtrrbeH7qaouY8c3y0mO8pGQ7GNQv2CPxyMjtHra\nWtU0OdX4DAlMmTSV4enph9jjmUOWJM4dkMzIwiRW76jj3a/24zkwCLk+ncRhJThUrbyWWMmUG8cx\nYXU9npK9tH36CYHWVs6/fSFZuXF8ubwEt8vPkmfWM/O6ESQkR67IqjRRJPW/jrbaT3E2b8JjK6Wl\n8r8k5Pz4uEZyVZLEldnJSEhsaraxvslGkkHLuOTvP9dXEAThZJs0aVJnM6nJkyef5miEs0X3Edke\nJcpyV4myrD64RLnXDsoAHevgSvKxrUtrNnD9RYXMnJjL2qJ6Vu9MxN4gQS74ZYnlk2OJcYYYUeJm\ncLmXKF8b+SUf00/WUZc4lFrzILxhNY21dhpr7az9XCY7P4W8whtJSq7Dbd1OwNNA0GfBWvsp1trP\n0EVlY4obitE8CFltOGKMWpXMhBQzY5NjKW5zsrHJRpnDgyMQ4uv6Nr6ubyPTpIs0xIyLwqwTTakE\n4WQ4bIJ7uJLk7iRJYuLEiSckoB+K1V+txNOwl6S4AHEJHs4Z1HOU1uPW0mbR0mxX4dEncPm0Kxka\ndfTzP840siTxo5EZDM2N58n3iqhugqbNo8g7r5L68D5Wu3bjuXQ4l6WkYl/9Fc4tm2jUaMibfxsx\nZgMfvf0tXk+AD9/eyeybRhFjjnyRSJJMfOZlyLIGe+PayNXV+njM6VOPK15JkpiZnYQ9EGSv1cXy\nKgvZUQbSxHwZQRDOMPfcc0/n7enTp5OTk3P6ghHOGh3zbwEkqeu2WlKhhNov1HcbwQ0d5Qju0ZQn\nH06UQcOl5/XjkjFZvL2njm9dbhRXNL7S82hNqmH1yAbWnRNFXo2PwWVesut95DRtJbtpG63GNOrN\nA2k2ZhIMwoG9zRzY24xKLZOVO47+BRBrOoDfWYIS9uNzVuBzVtBasxydKRtDbAGG2EI0usMvZ6iS\npM5VHVq8frY029lqseMMhqhx+ahx+VhRbSHLpGeQ2URhrLH9/ENUkgnCiXDYBHfBggVHtRNJkkTn\nRuCb1V/gri0iNSlAdrwHKbbrMUUBW5uBZoualmAUky6cwYCJp27B41Ml0Wzg4RvP5en/FrGrrJWy\nDXkMm2xiv28Hmy3fYh5/PhO0l2D9/FPs69eiSU0l6YoZXHnDObz/2nY8rgArlu7iqptHoe72xRmb\ndgGhoBtXy3bsjd+gi8rCEJN/XLHKksTVuSk8ubsKqz/I0vJG7hqcJebjCoJwxpo2bRqDBw9mxowZ\nXH755SQnJ5/ukIQfqFC3ZX8kqfsIrhol0Msc3GAkCVYdYgS3s0T5OBPcrpgkYqN14HJTmBnL8IzJ\nbN7bxLc7GwjENLI3vp7S8y1Ee4IUVnrJr/aR1lJHgruOgKylMSqXpqhsrIY0QsEwFftaqdgHEEdi\n8o/IL/CQEFcHwQpQQvic5fic5VhrP0WtS0Af0x99VDa6qGxU6kNPH0vQa7k0K5GLMhI44HBT1Opk\nd5sTbyhMtctLtcvLp7UtRGtUFJpNjPLGkyzLmFQn5n0ShLPRYRPcPXv2iHmJR7Bx01pa928hLTFA\nVoIbeWDXY8GgTGuzgcY2Nar4HC6bNuv0BXoK6bQq7p09jH/+51t2V7RRvDaVcy4YQbF9J59Vf03u\nBTeR7HTg2LCelv++hyGvP4lDh3DVTefy5r830trsYuPX5Uy8sCuBlSSJ+MxpBNwN+D31tFZ9SNqg\nu5BVxzfialCrmJObwr9Laqlz+9jcbGdscuyRnygIgnAafP7556xYsYKPPvqIv/zlL4waNYoZM2Zw\n6aWXEhsr/nYJJ073EVyl2xxctawi1D6C272Lcih0+BJlJXxiRnC7C7XPEdaoZMYWpjB2cAoe3yCK\nylrYW21j854aWvW1bEppZmuhhWifj/41vkiy21xCpr0Ev6zDYsqiKSqbNkMaYVmNpcmLpUkCMtBo\nUsjr7yEtpQ2jvh4JH0FfC87mFpzNmyLH1yeji8pGZ8pAa0xHrUs46PxZJUsUxpoojDUxKzuZA3Y3\nu9uclNpc2AMhHIEQW5vtbG22AxCv05AdpScn2kB2lIFEvUZcgBeEo3TYBHfChAmMGTOGiRMnMmHC\nBLKysk5VXGe0HTu2UrN7LekJflKSPKR1Kz8OBmVaGg3UWbUUnHsho8cMPo2Rnj4atYpFVw7lD0u2\n0tjqpnxTP3LPs1Jur+T1kqU8PPcefDU1+GuqaXzlJUyPPkb+wGRGju/HtnVVfLu5hv4DkkjN7Dph\nk2Q1CTk/pn7v84QCDmz1XxGXeelxx5oXY2R4fBTftjr5tMbCOQnR6A41h0gQBOE0ysjIYMGCBSxY\nsIDq6mpWrlzJ0qVLefTRR5k4cSLPPvvs6Q5R+IHonuAi9WwyFezsoty9RLl9BPdQyxKeoBLl7jpG\nmVXdEj+DTs34oalcMSUfi6WQ/bU2ig60sLuihUpHNVuim9g+zoJOayOzyU+/Bj/Z9eWcU7+fkKTC\npk+mxZhOqzEDpy6eQEBNyd5oSvZGA1nEme2kp1lJSrJj1FuRJIWAt4mAtwmnZXPkNcpatMY0tMZ0\ntIZUNPpE1PpEZDlyQUAtSwwwmxhgNqEoCg0eP6U2F/tsbqpcXoJhhVZfgFZfgO0tDgA0skSaQUe6\nSUe6UUeaUUeSXotWnK8IJ0Fx8S5+9asHWLbsEwD27i3mjjtuQafrGli66ab5zJt3K3a7nb/97Y9s\n2rQRgAkTJnL//b8kqpepl3v2FLNgwc297udEOmyCu3LlSjZs2MDatWv597//TTgcZuLEiYwfP57x\n48djNp89TXmKdu+gcsdqUuMCJCR7iB/c9cc+FJJobTZS16Ihe8T5jJk+4jRGeuYw6jXc/eOh/O6l\nzbTaAwxqHUe9rhFXwM1/qz7jutvuoPIP/0vA0ozlww9IvGM+Y6fkUrGvhdZmF2u/2M/seaN6XAXV\n6BOJSZmIvWE1jubNRCedh1oXd9yxTstKZHebC08ozKYmG5PTjn+fgiAIJ1NcXBxJSUmkpKRQWlpK\nU1PT6Q5J+AEJdmsypdDVAVkja/B2JLjqrtPII3dRbn+OfNhTz++lYwRXdYiRTVmW6J8eS//02P/P\n3nnHR13ff/z5Hfe9leQyScIOQ9G6iloBLSoWFQfYKmitYtVWQH8OtI7iLmqdOGvFFlSsq7ZVrOLC\ngVuLIg62hJ2QnUty67t+f3zvLgm5SCAXYuLn+fDM977fz33ufcfdfb+vz3txys+HENNHUloWZM3m\nOlZtraI0uok1xVXIw2vJkmvoWxOhuLKOvlWVDN3yOYbsod7ThzpPAfWePjS486mtC1BbF4CVoCgm\nOdlB8nLqyM2tJ5DVhKKY8fzdjUQbN7ayR9VyUD35uDz5uNy5KFo2qjuHIk+AYl8u4/rnkRnw8dXm\nKtbXh9nQGGZjY4SoaaFbNpuaImxqirSaM1tTKfBo5Hs0Crwu8j0aOZpKQHOhitZEgl3Etm1eeeUl\nHnroXpQWC1hr1qxm1Kgx3HnnfW0ec999dwES//nPK9i2zXXXXc1jjz3KxRe3bSG7Zs0qxo4dy5//\nfE9a2pm2x/f+ymRlZXHsscdy7LHHArBlyxY++ugjXnvtNf70pz9RXFzMmDFjkrfexDtvvU7NtvX4\n5Qi5AZPcgjAHtBC1liVRU+WlrMpF4YhRHDLhMA7uRnt/qPQvyODkMYN58YNSPvqinkmnjOP1bYv4\n3/ZljO0/hpxjj6f2tUXUvP460VMnoSgahx8zjP8+u5yKsga+W1XJsH1a55hlFR5OY9UXWEYj9ds/\nIG/gyZ22M6C5ODg/k88qg3ywvZbRhQHUVI3qBQKBoBtpbGxk8eLFvPbaa3z00Uf07duXk046iZkz\nZ1JSUrLzCQSCDqIbLYpMtQhRdslqcx9cdRdycJMhyntO4O6I5lLYe2AOew/M4WRKsOxDqKwLs7G8\ngdLyetbHtvG+XEasXy2au46iaDUFwRoK6ioYULucnHKLsJpHgzuXRncODe5cavQcqqoTi+I2Gf4Q\ngUAD2VmNBAINZGY0oarOe2PEajFitUSCa9vYJquZuDw5+DPzCVgeRqp+Ds3PRCryUWdlsV1XKQtb\nbAtF2RaKEol7zOtiBnUxg7XBUKv5JCDTpRDQXGS7VXI0F1maSoaqkOFSyHCpZLoUPMruVbUW9E4W\nLJjP228vZurU83jqqSeS+9esWc2wYXulfMysWTdiWRaaplFZWUE4HCIQSO0EXbNmNSNGjEh5LJ3s\n0q9M//79mTJlClOmTMG2bb799ls++ugjHn300V4lcF94bh4HDd3G4B0qH1sW1FX7KK9yERh4IKOP\nO6p7DOxhnDB6EB98XUZVfYQN32bTt38R25rKWVT6JjNO/A317y/Bampi679fIPu00+k/OIeBQ3LZ\ntL6GZZ9sYuiI1j1qZdlFVuHh1G19nabq5QSKjkTVsjpt59iiXP5XGaRBN1lZ18T+uR3rfycQCAR7\nitGjR5OTk8OECRO46KKL2H///bvbJEEvpaXAteMhyhKS0ybITCFwzZ0VmUq/B9dICNzdXI+WJYnC\nHB+FOT5+tk8h4FzAN4RilFWHKKtuYmNNJavqt/NBtIqwWUuOUUueXk1OeAs5DSbFDRbesBfTDhB2\nZRGqz6KpKotKV19iqg+w8XiiZGaEyPCHyIj/9fkieNyxpC2W0UC0sYFo46aUtuYBubbMvrYbW/IQ\ncgWol3MISlnU2T7qbC81potG03kzbCComwR1k81N7b8HiiQlRa9XlfEoLf4qMh5Vxqs4QtijyGiK\njCZLuGQZTXH+KqL68y5jWDb1MX23H68oEkZTlPpIbKee0F3x5p944iSmTj2PZcs+b7WSAw2kAAAg\nAElEQVR/7drVaJrG5MkTMU2TcePGc8EFF6JpGmr8d+C2227m1VdfpqRkCKeccmrK+desWc2WLZtY\nuPDkNvOkk+/9lSktLf3eB/v9fsaPH8/48ePTalS3Iyk4a1820ahKfbWbqnqVjAH7ccSxx3S3dT0O\nVZE55ecl/P3llXy5toYzD/g5LzQ9z8qaNWw1qsk59niqX/g35W+8SebxJ4LXz09HDWTT+hqqtjey\ndWMd/Qe3DhnOyB9JsHwJlhmhqeZLAkVjO21nrsfFsCwfa4MhPq8KCoErEAh+cDz66KOMGjVKeFwE\nXY5uNIcl25Kz7YqL02QV5VQCt50cXNuKP6aLc3DTQaZPI9OnsdeAbKBfcr9l29Q3xqgORqisa2Jr\nfTUrGqupDdcSjdbgCtXgjWwmMxYmMxolN2rjjnhRa71g+TAkH5Wqn82uIqKKD11z4/ab+LwRfN4I\nXm8EjyeKxx3DrcVwu2O4XM3/DpJkIUlhIEymXUumuaG14RLoikIjfhptHw34abD9NOKnwfYRwksY\nDzotioPZNvW6QX3833R3kG0LxTZRWv21kLCRsZFs56+MjWzbyICMtcN9nLGAhI2EhBTfn7wvOYss\nsgSSbSNJEnJyP4CNZEtJuS3LzoHEHufK3nk8koQs2Xg8LqIRPdmhSWoh1pu37OS2FLdwx+Nyi0dJ\nzZutRiW2LRveIZMQe6ZStg+L4+QQbbIH4u/DjmzasoW169ZhGCZffPmlM1SSGTR4KGeedR4NDUHm\nz/srVdVVTDplcvJxvzj2RI4adyxPP/U4l1x6IZdedlWbuRXVxbhx4zj22JOorKzi+uuvYd68ucyY\ncXEaX/FOBO6ECROQJAnbTr0ykDjW29oE/XLKb1m7diWmZDPikJ90aYz4j4VR+xbx4vulVNVH2PJd\nBoXZBWwPVfL2pvc5++hJ1LzyX+xYjPqPPiRwzLEUDwjQpziTirIGVny5rY3AlWUX/twDaaj8lMaq\nL8gqPAJJ6nxI8cH5WawNhlhbH6I+phPQRBN2gUDQvTz33HOt7m/alNrDA3D66ad3tTmCHwl6yyrK\nSYHrnBNTenCNjuXg0o0hyp1FliRyMt3kZLoZ1i8A9G0zxrZtwlGD+qYY9Y1RKhuD1ISD1EcbaArV\nYTTWQrgWObQFNRLCjOpEGi0MU6XRVJFtF5LlxsYHdjYoCqpHRvFJKF6QNeemahaaS8flMuK3+LZq\nElAayJGD7b4O3VYI4yGMh5DtSW5HbY0YLqJoRNGI2S22cdFen15LkrEkmd33R3YjUbqu/fAPRD6E\nkHnBalvw6fuoM71EgX/pfgByz7+GGuC/AIFcMo/5FR++8hyxE85p/UAFXCdOZd0tl/J0rYXL3/p5\ns8+5gqraNXi9Xvr168/Uqecyd+5f9qzAfeutt9L6ZD2JESP2JTc3g5qaxu42pVcgyxLjRvbnn++s\n45Nvt3Pqr0bxYul/+bLya6bsdQpZh42i/v33qH3nHbLGjUeSJPY5sJiKsgZK11YRjei4Pa3FZkbe\nSBoqP8XUg0QbN+LJ7Hz+2T45ftyyTNSyWFnXxKg+P55CagKB4IfJ3LlzOzROkiQhcAVpIxGirCoy\nZjx/Vk2EFxsp+uDutE1Q1xWZUn9AEQ2SJOHzuPB5XBTn+RlBbscfK0NGhpey7fU0hKM0xiI0RSOE\nwiEiTQ1Ew41EwyFMPYIRiWA2SVgxFUk3sA0XGDYYNpJhg22jyKAoIMtOmyJFjW8rNori5FbLcgiv\nHMIv20gyyArI8W3nvo0s2yDbWLKKIcuYkoopy5iSgikrzt/4zZAUTJxtS5KwkFv8dfy2FjImMiYK\nlt36vo2EFffZ7rjt3Jd3uN/sURV0DXqokc1vvsjA405F9XgBsAwdOd4m7Ku/3ka/sceT95ORgLMA\nJskKitudcp6isYcl98ViMTStcy0/U/G9vzL9+vVrs2/NmjWsW7cOy7IYMmQI++7742yDI9h1jjig\nmBfeX09Mt5DqBqLKKrpl8HnFcg476ijq33+PWHkZ0Y0b8QwezNARffhg8TpMw+K7VZXse1DrlVKX\ntwCXtxA9vJ1Q3aq0CFyXLLNXwMfXtY2srBUCVyAQdD9vv/12d5sg+BGSELiaS0a3HN9cQuBaunNf\ncu1CiHIX5ODq8RBlVy8pCilLEh63SiDDTYZXA3pGqpSiSEmnUCLq0TJNbNvC1HUs28IyLExsTN3E\nsm0sw8TGwrZtTNME244vpNhYtg2miQXYtoVtW1iWhW1Z8c+R7USX2haS5Wxb4DwPjuPUspzPo41N\n/D+wnHGJ/bIEmttFJKIT/yhht7hB877v2075N/E/u/Xo9py6tp1KpO84c/z14HxWNE0lGjPaibSV\nkmN3im23sNlhg17JWtvi6NhWbNniwa8+wTIbGDvhROpra3nmjec54mejGBXbitk3nw2vP8eEYj+y\nLPPCC89wwE9HMs6qhFiLp5EtHvr6E9yD8jAMna1by1iwYD4TJ/6yY3buAh3+lQkGg1x11VW8++67\nBAIBTNOksbGRkSNHMnfuXDIze8aXUNB9ZHhdHDAkj8/XVLJ8dT0H7LcvX1R8xZcVX3Pkwb/H3acP\n0YoKGr9YimfwYNwelYFDcildU8WGtVVtBC6ALzCC+vB2wvWrsPsfn5a8tBHZfr6ubWR9Q5ioaYme\nuAKB4AdFdXU1zz//PBs2bODKK6/k008/Zfjw4QwfPry7TRP0IpICV1XQ497X5hzcuMBt4XmxEgK3\nnWI2XVFFuVngCg/eDw1ZUQAFRf3hpnqlEuY9ha62/QtvBi+6nmH84UcBMLz/YO67727uvfVPuN0e\nJk36FeeddwGSJHHkoWN4+OH7mffQA8iyzJFHHs2MGZfg9XopLy/n7LMn8+STz1NUVMQ+gwbz4INz\nOP74Y5LzTJ7867Tb3+Er91tvvZWKigpeeeUVPv30U5YuXcp///tfIpEId955Z9oNE/RODo23/Fm5\nsY69s5wy4WvqviNiRMgbMwqAhs+XJscPHpYHwJaNdei6yY54s505TL0BPbw9LTbune3kG5i2zabG\ncFrmFAgEgnSwYsUKjjvuON59911efvllQqEQH374Iaeddhoff/zxLs1VVVXF6NGjeeeddwCor6/n\noosu4uCDD+aoo47i+eefT461bZt77rmHUaNGceihh3LLLbc4XhdBryWRg+tyyRg7CtyY45aRW1Q+\nTebgtuPBxU5/kamEwBX9XgWC9DJy5CG88kpzqmpJyRDuv/9hXn99CS+99Drnnz8t6VTSNI3LLruS\n//73DRYufI3LL78ar9cJZS4qKuLNN9+nqKgoOc/jjz/O4sXvtZknnXRY4L799tvceOONDB06NLlv\n+PDh3HDDDbzxxhtpN0zQOzlwaD6qImHZNnawD7IkY9kW31SvJu+wnwGgby9Hr6oEYOBQR+CahsW2\njXVt5nN5+iCrjiCNNG5Ii40+VaGP1zlpb2iI7GS0QCAQ7Dn+/Oc/c8455/Dss8/iiuc/3XrrrZx9\n9tncfffduzTXtddeS11d8+/q9ddfj8/n46OPPuKBBx7g7rvv5st4Bc2nnnqKd999l5deeolFixbx\nxRdfMH/+/PS9MMEPjmYPbssQZeczZ0WjAEgtBK4Rr7qsqqkFbFfk4OrxMFThwRUIBC3psMBVVRW3\nu20SsMfjQdd7ZN00QTfg1pR45UFYu6mJYdlDAFhVvZaMvYYjxT9jodWrAPD5NfILnQps2za3FbiS\nJOHJGARANE0CF2BwhrPytEF4cAUCwQ+Ib7/9lokTJ7bZf/rpp/Pdd991eJ5nnnkGr9dLcXExAE1N\nTSxevJhLLrkEt9vNAQccwEknncSLL74IwMKFCznnnHPo06cPBQUFTJs2jRdeeCE9L0rwg8SIC1yX\nqmAki0w54jURotzSg6vr8aJUrp3k4HZJiLJIJRIIBM10+BdhzJgx3Hnnna1We2tqarjrrrsYM2ZM\nlxgn6J3sO9ipKLhiQw3DAoMBWFdXiqyq+IY7TdbDq1Ylxxf3dwRx+Zb6lPO5M505Io0b221ptasM\nzvQAsLkxgmH1rLwMgUDQewkEAmzbtq3N/m+//Zbc3I5Vay0tLeWxxx7jpptuSu7buHEjqqoyYMCA\n5L6SkhLWr18PwPr16xk2bFirY6WlpWn7zRX88GhZZCpmOiHJLiXuwY2l8ODG04hUV3se3Hjebho9\nuIYIURYIBCno8K/MNddcwznnnMORRx5J//79AdiyZQtDhw7ltttu6zIDBb2PfQfn8p/31lMTjFLg\ncj5L20OV1EWC+EaMoOmbr5MeXIDiAQG+/nwrFeUNGIbZJvzJ7R8IgG1GMaLVuDz5nbZxoN/x4Bq2\nTWUkRrEv/SXMBQKBYFf59a9/zQ033MAf/vAHAFavXs17773Hgw8+yLnnnrvTxxuGwVVXXcW1115L\ndnZzlfhQKITH42k11uPxEIk4aRrhcLjVca/Xi2VZxGKxlNFdOyJJEulwsslxISP3MEHTE+2OxUOO\nPZpK1HQErVf1oChSMgdX9biTfW8THl+3W0nZCzchcBVVa7dX7q6i2/HnVOVWc/bE9xuE3Xuanmo3\n9Fzb95TdHRa4BQUFvPTSS7z33nusX78et9vNkCFDGDNmTJckBwt6LwMLM1AVGcO0MBqyUCQF0zZZ\nVbmO/vEqoEZNNUYwiJqVRVE8pNkybSrLG5Me3QQuTz6SpGLbBrFQeVoEbrZbTfbDLQtFhcAVCAQ/\nCC644AL8fj+333474XCY//u//yM/P5/p06dzzjnn7PTxDz/8MPvssw9HHnlkq/1er5doPK8yQSQS\nwefzAY7YbXk8HA63m7qUirw8f1qvFbLjxQB7Gj3JbltyViS8bhVTdsRuwOcnNzcjGaKcmZtFbq6T\nRmTFBW4g25fc15JtkhOinJGZlfL4rmJYdrK1S14g9XP2pPe7JcLuPUtPtRt6ru1dbfcuxYmoqsrR\nRx/NEUcckdyXyL/VWoSpCATfh6rIDCrM4LttQTaVhemXVcymhi2sr93E0AGHgySBbRPdtAF1vwPw\nZ7rxZ2o0NcSo3t5W4EqSjMtbSCy0lVi4DD/7ddpGWZIo8mlsbIxQHoru/AECgUDQRViWhdzC/fmb\n3/yG3/zmN4RCIUzTTLbpq6yspKCg4HvnWrRoEZWVlSxatAiAxsZGLr/8cn73u9+h6zrbtm2jb1+n\nJVtpaWkyLHno0KGUlpZy4IEHJo8NGTKkw6+huropbR7c7Gw/dXVNWD0ofaQn2l0XL7Lodas0hkPO\nTlOmurohWWQqFLOoqWkEIBp1BGw0ZiT3tcSIOTUtwhE75fFdJWI0V/EON0WpkZvn7InvNwi79zQ9\n1W7ouban0+7vWyjrsMD97LPPuPnmm9mwYUOyeTI4rQMkSWLlypWdMlLw46KkOIvvtgUpLQsysG9f\nNjVsYWPdFpT+XlyFhejl5UQ2bsS/3wEA5BVk0NRQQ3Vl6pOi5uvrCNxQWdpsLPK52dgYoSwsBK5A\nIOg+Lr30Uu69915UtfUpO+FdBUe43nzzzXz66affO9drr73W6v64ceO4/vrrOfroo1m1ahX33HMP\nt9xyC2vXruXll1/m0UcfBWDixInMmzePUaNGoaoqc+fOZdKkSR1+DbZtk86uQpZl97i+ldCz7A5H\nHMHqdatUGo7Y1WQ3RlSHeO61rbiSryfRyk9R5JSv0bKcsGYkLS3vQcRovhZVbNp5zp7zfrdE2L1n\n6al2Q8+1vavt7rDAvf766xk2bBhXX311mzwdgWBXGVTkeBy2VDYxOsOp4rmhbgsAnoGD0cvLiW7a\nmByfW+Bn0/oaqiuaUs7n8jheCyNanTYbC+OtgqrCokq4QCDoPj777DMuuugiHnzwwTbRUvX19dx0\n0028+uqrnS74OHv2bG688UaOPPJIfD4fV155ZdJje+aZZ1JVVcVpp52GruucfPLJHcr5FfRcIrFm\ngZvIwXUrGnakuX2e7HHqVViWhRW/WE1VRdm27RZFplxpsS9mNgtctyKqKAsEgmY6LHArKip45JFH\nKCkp6Up7BD8S+uY7sffhqEFAcZo/14braYg1osXD42Ll5cnxeX2cMITqysZk1EBLXJ54v1y9AcuM\nIiudz5nNczsn4XrdQLcs0YZAIBB0CwsWLOB3v/sd06ZN4+GHH8brdUTFu+++y3XXXUc0GmX27NlM\nnjx5l+d+++23k9vZ2dncf//9KccpisLMmTOZOXPm7r0IQY8jEnM8sl6PSjTseF89ihsz3Nw+T45/\nFg29WWy6UlRRthPeW0CW01PTIiIErkAgaIcO/yKMHz+eJUuWdKUtgh8RRbnNoXV2uDmGvrypAq3I\nEbx6xXbseDh8Tp4z3tAtmhpj7Ijqzktup8uLm+tuXmWuiQovrkAg6B723ntvnn76aTZv3sz555/P\n9u3bmTVrFtOnT2f//ffn5Zdf3i1xKxB8H0mB61aJGAkPrhsrFEqOkX2OwI3F828BNHdb34nVQuBK\nSnpqtkSFwBUIuowVK75h0qTjk/dXrVrB2LE/Y/z4nydvCxbMByAYDHLjjX9kwoRxTJgwjtmzr6ex\nMXVKoa7rzJ49m+OPH8cJJxzD7bfPTtZzSicd9uBefvnlTJw4kZdffpkBAwa0KngBcM8996TdOEHv\nxetWyctyUx2MUlNr4nf5aNJDVIar6VfohCzbuo5RU40rv4CsbG/yscG6MBmZrVeAFVcmkuzCtnT0\nSA2ar2+nbcx2u5AlsGxH4BZ6RSVlgUDQPQwYMICnnnqK888/n3HjxhEIBLj77rs56aSTuts0QS8l\nqjcL3GSIsurGqmsWuIrXWXyORloIXE/bS0vbbK5lIctpErjxBXBZAlV08xAI0oJt27zyyks89NC9\nKEpzNMaaNasZNWoMd955X5vH3HffXYDEf/7zCrZtc911V/PYY49y8cWXtxn7178+yPr163juuRew\nLJsrr7yMZ555kqlTz0vr6+iwwL322muRJIn+/fvvdg7uihUruOGGG1i3bh2DBg3i5ptv5qCDDmp3\n/Mcff8y5557L559/jt/fM8tgC9qnOM9PdTBKWXWIPvn5lNZvojJUjavfAckxsfJyXPkFuD0qHq9K\nJGwQrA3Td0B2q7kkSUJ156GHy9PmwVUkiWzNRU1UpzoiPLgCgaB7KSws5KmnnuL3v/890Wi00zm3\nAsH3kcjBdbtkdMvZdkKUgwBILhdSvPBZtIUH153Cg2u38uCmZ7E44cF1y7JoVynocRimRU0wsvOB\n7SArElFLoq4+lMx/b4/cLA9qB6McFiyYz9tvL2bq1PN46qknkvvXrFnNsGF7pXzMrFk3YlkWmqZR\nWVlBOBwiEMhuM84wDBYufIHnn3+eQCCAadrceuudGIaRYtbO0WGBu3TpUv7xj3+w//7779YTRaNR\npk+fzvTp05k8eTILFy5kxowZLF68OKV4ra+vZ9asWdh2z6sMJugY+XGvbHUwQv6AvLjArULWNNTc\nPIyaavSK7YDzmcvK9hIJNxCsS/2DoGoBR+DqwbTZmOt2BG5tNP1fPoFAIOgIH3zwQav7559/PrNn\nz+bss8/mqquuarXK3rKNn0Cwu9i2nQxRVlzN5a/dioYVbxmUyL8FiMU9uLIspSwyZZktc3DTFaJs\nx20S4cmCnoVhWsx69BOq6ndf4O4K+QEPt10wqkMi98QTJzF16nksW/Z5q/1r165G0zQmT56IaZqM\nGzeeCy64EE3TkhX+b7vtZl599WVKSoZwyimntpl78+ZNGIbJ8uXLefjh6UQiEcaPP55p0y5Kzwtt\nQYcF7qBBg4jF2uY+dpRPPvkEWZY588wzATjttNN44oknWLJkCSeccEKb8TfddBMnnHACf//733f7\nOQU/bPKynFXc6voIe/mcHNrKsON9deXFBW5tbXJ8VraXirIGgnXhtpMBiisLADOWPoEb0JyvSFAX\nAlcgEHQPv/vd71Lur6qqYtq0acn7omWfIF3EdCvRCQhJbeGdVdxY8SJTsre5lkbCg6u51ZTeVMtK\nhCjLILUtQrU7RON9p4TAFQjSR35+fsr92dnZ/PSnBzNp0qnU1FRz/fXXMG/eXGbMuDg55oorruGS\nS67g9ttnM2vWlTz00KOt5ggGgxiGzjvvvMP8+U/S0NDIVVfNxO/389vfpj7P7S4dFrgzZszgmmuu\n4eyzz2bgwIFt+vHtbNW4tLSUoUOHttpXUlLC+vXr24x96aWXCAaDXHHFFULg9mLyAk6oe1UwQp4n\n19kO1wCg5uQAYNTWJMdnZTvjg+2seKlaXOCm0YOb5XI+5w0xIXAFAkH3sGrVqu42QfAjo6llWo7a\nvJ2h+dGTAretB9edIv8WwIr30ZVVT9rCiUPxPrg+NT2CWSDYU6iKzG0XjOp0iHJ2wE9dfVNaQ5Tb\n44477k1u9+vXn6lTz2Xu3L+0Erhutxu3282FF17ClCmTCAbrycoKJI9rmgvLsrjsssvIzMzE58vg\njDN+w7/+9Vz3CdxEa4DbbrutzbGOrBqHQqFka4MEHo+HSKT1P+62bdu4//77efrpp3erqpYkSaSj\nm4ssS63+9hR6kt19cpzPQzRm4lecvrhNeggLA1euI3jN2loUxXktmVmOwA01xpL7WuJyO18iUw+m\nPL47BNzNHtxUc/ak97slwu49S0+1G3qu7T3VboHgh0BDqMX1l9JcIMqv+qiJV1FW2vHgpsIy42HN\nqi/l8d0hZDgeXJ8qPLiCnoeqyPTJ2f3vg6JI5Ob6ccs25k4EbmcJBoMsWDCf8877PT6fk1Yai8XQ\nNCcSc+bMizjttDM4/PCfA2AYOoqi4PG01n39+w9EluVWEcGmaXZJOmqHBW5nV5C9Xm8bMRuJRPD5\nmv9xLcvi6quvZubMmRQWFrJly5Zdfp68PH9aiw1kZ/fM4lY9we5hLfLGvK7mFR68BoH+RdQAVn0d\nublOG6HCYsdDG2qMkZPT9t/ZRSGVpWCZUQJZKoq6e8XQWtJPN2BDBQ26SXaOH7mdz1ZPeL9TIeze\ns/RUu6Hn2t5T7RYIupPGcLPANSXnYtSrelFkBTMucBMtgqAjHty4KFa6QuAKD65A0JVkZGTw3nvv\nYNs2M2ZcTHl5GQsWzGfixF8CsNdeI3jiiXnsu+9+KIrCQw/dx3HHnYCmtc63z8zMZOzYo5gzZw7X\nXz+bpqYQ//zn0xx77IS029xhgTtixIh2haPL5aKwsJATTjiBSy65pFXBiwRDhgzhH//4R6t9paWl\nrVoclJeXs3z5clauXMlNN92EFS8Bf+SRR/LII49wyCGH7NTO6uqmtHlws7P91NU1YVk9p9BVT7Lb\ntm0UWcK0bOqqmvvZbdheRqHbEbXRqmqqqxuQJAkLZ4xpWpRtq8PjdbWaT48236/cXo7mTZ1HsCvI\n8VVp07bZUlFPhqv1V6Ynvd8tEXbvWXqq3dBzbU+n3YlFNoHgx0JD2BG1fo9Kk97kbLsccWo2OGlA\nSlZWcnyiTVB7HlzTiIc1p9GD2yQErkCwR5BlmTvuuJf77rubE088Brfbw6RJv2Ly5F8DcP7503j4\n4fuZOvV0ZFnmyCOPZsaMSwBH25199mSefPJ5ioqKuO66m3j00Yc488zTiMV0Jkw4kTPOOCvtNndY\n4P7pT3/i/vvv5+KLL0629vn666954IEHmDJlCkOHDuXhhx8GmsOZWzJ69GhisRhPPvkkZ5xxBgsX\nLqSqqqpV7m7fvn356quvkve3bNnCMcccw5IlSzrcJsi2bUxz5+M6imV1veu/K+gpdmf5NWobooRC\n4FHdRIwoNeF6igNODq5t6Oj1DSiZmXi8zStBwfoILq31x1eSmy9C9WgQRcvrtH3eFqslwaiBV059\nIu0p7/eOCLv3LD3Vbui5tvdUuwWC7iQRopzhc9EQa3S2Xc51mBF0BK6a2SxwQyFHEPv8rReeEyQ8\nuLLqTXl8dxA5uAJB1zFy5CG88spbyfslJUO4//6HU47VNI3LLruSyy67ss2xoqIi3nzz/eR9v9/P\n7NmzmTnz6i49N3dY4M6bN49bb72Vo446KrlvxIgRFBUVceutt/Laa69RWFjI5ZdfnlLgaprG3/72\nN2666SbmzJnDoEGD+Otf/4rP5+OGG24AHBEt+HGR5XMEbrApRq43m20N26mPBlGzBybHGPV1KJmZ\n+DKaBW6oMUZeQeu5JFlFkjVsK4ZphEgHLU+ciZOpQCAQCAS9mca4wM30ajREnWKPGQkPblzgKi0F\nbmNC4KZuAZTIwVW6IAfXLwSuQCDYgQ4L3IqKCgYMGNBmf3FxMWVlZYCj0uvr69udY8SIETz77LNt\n9rcnbPv378/q1as7aqKgB5IZX+0NNsUIFGayrWE7jXoTSkazN9ZsdFaPFUXG7VGJRgzCodQFyGTV\nhxmLJVeLO4tLllAlCcO2kydTgUAgEAh6M4kc3Ayvi2DUOQf7XX4nSi5FiHK4KZ6n257ATYQopykH\n17JtwqLIlEAgaIcO/yqMHDmSu+66q5WADQaD3HPPPYwcORKAN998k8GDB6fdSEHvJcvnnAyDoRgZ\nmhP+1KQ3IWsaktupzpYQuNBcwCIaTi1wE6vDlpm6V+6uIklS8uQpBK5AIBAIfgzUNTqVk7MzNWrC\ndQAE3FlYkQh2vMOFGhe4tm0nF53b8+AaegMAiis9Rd9ChkkipmrH2hgCgUDQ4V+F2bNnc8EFFzB2\n7FgGDhyIZVls2bKFkpISHnroId577z3mzJnDAw880JX2CnoZWfGTYX1TjKHxwlJNejyUyZ+BEY1i\nNjYkx3u8LoJ1ESLtCFxZcfJ70hWiDE6YclA3hcAVCAQCwY+CmgZH4OZmelgfqgUg2x3ADDY7ORIh\nypGwnizk1jKVKIFlxrBNp4uG4spqc3x3qG/Rmz6gCYErEAha0+Ffhb59+7Jw4UI+/vhj1qxZg6qq\nDB8+nNGjRwPg8/l49913yY33LxUIOkLSg9sUIzPuwW2MV2xUMjIwaqpbeXATlXA+nEQAACAASURB\nVJMTFRt3JFGhMV0hytCchysErkAgEAh+DNQGHUGanalRU+2I2mx3AKOqJjlGzXGKQSbybyG1B9fU\ng8ltRUuPwA3GBa4siRxcgUDQll1a9lIUhSOOOKJV5eMEQtgKdge/1/kINoUNMnf04GZmAjuEKMfH\nt+fBVbpU4IoiUwKBQCDo3eiGRTAecuz1mZiVzuJujjuAXr0ecM7PcjyNKFjniGFZlvBluNvM10rg\nujLTYmO97gjcLJfabn96gUDw40Vk5gu6FZ/b8ciGogZZcYHb0oMLtA5R9jjjI+16cJ0Q5URBi3Tg\njefghoUHVyAQCAS9nNp4/i2A7IkktwPuAHp1NQBqXnOf+WC9c77NyHIjy23FphFzzuGy4kWWU7cR\n2lUSHtwskX8rEAhSIASuoFvxuR3vqG5YeOP5s016CNu2UeK9j83GpuT4ZIhyOx5cSXZWjy0rmvL4\n7uCO98KNWcKDKxAIBILeTUVtcwSULjsRVJrsIkvLwKiuAsCV19xnviHuwc3KTt3j1og5Yc2Klp02\nG2uizjVAtlsIXIFA0BbxyyDoVnye5tVc2XZydyzbImbpyB7nZGlHm1eQdxaiLCtxgWvGUh7fHTTF\nEbjRLmxILRAIBALBD4Hyakfg5mW5qYk6HttCfwGSJKFXVgLgaunBTQpcT8r5jIgjcF2evJTHd4eq\niHMNkO9JXbVZIBB0jhUrvuGPf/wDCxe+BsCqVSu44ILf4nY3pyGcffa5TJ16Xryrzp/57LNPARgz\n5nBmzryajBYtPwHKy8s5++zJSJKEbTvX1LFYjOLifjz77H/Sar8QuIJuxetuLg5hm83bESOK7HFO\nllakOdxYi1dL1GOpw4Vl2TnZ2en04CYErvDgCgQCgaCXs73GOecW5vooD20CoI+vANu2iW7bCoDW\nt29yfCJEuT0Prh4XyS53egSubdtURZxF7HxPekKeBQKBg23bvPLKSzz00L0oSvN1+Zo1qxk1agx3\n3nlfm8fcd99dgMR//vMKtm1z3XVX89hjj3LxxZe3GldUVMTbb39Abm4GNTWNVFRUct55Z3HZZX9I\n++sQAlfQrbT04JpG8xcpakbQvPF82kizB9elxUOa9dQCV1ISAlfHti0kqfNR+MkQZVMIXIFAIBD0\nbsprnLSgolwf20KOx7bQV4DZ0IDV5BzTih2Ba5oW9bXtC1zbtjHiAldNk8Bt0E1i8bZEwoMrEKSX\nBQvm8/bbi5k69TyeeuqJ5P41a1YzbNheKR8za9aNWJaFpmlUVlYQDocIBHaeknD33X9m3LjxjBo1\nJm32JxACV9CttPTgmrFmMRoxo3gSHtxwW4Fr6BaWZbcpaJEIUQawrRiSkjpkaldIenCFwBUIBAJB\nL6esxglR7pPjZVmTI3D7+PKJxb23AFpxMQB1NSGsePpOXh9/m7lMvQHbcsKJ0xWinPDeAuS7hQdX\n0DMxLIPaSP3OB7aDokBMC1MfasLcSQ3UHE8AVe6Y5DvxxElMnXoey5Z93mr/2rWr0TSNyZMnYpom\n48aN54ILLkTTNFTVmfu2227m1VdfpqRkCKeccur3Ps/SpZ/x9dfLuf762R2ya1cRAlfQrSiyjNul\nENVNTL2FB7edEGWXq3mMoZtoOxSYSBSZAicPV06LwHVEdNSysG0bSbQkEAgEAkEvJBiKURN0UnwC\nOTqRBmd7QGZfYt9+A4ASyEbxOWK2usLx6KoumUBOWw+uHi6Pb0lp8+CWhRybslwqHtEDV9ADMSyD\nP31yF9WR2j3yfHmeHG4YdWWHRG5+fn7K/dnZ2fz0pwczadKp1NRUc/311zBv3lxmzLg4OeaKK67h\nkkuu4PbbZzNr1pU89NCj7T7PggWPc/rpZ+Hz+Xb9BXUAUUVZ0O34PM4XLhyxkeMhxREz2lxkyjCw\nDaclQMKDC6nzcGWlOVzJNtOTh6vFQ5QtGwxbFJoSCAQCQe9kY7nT0kcCTE8dAJriosjfh8iGUgA8\nAwcmx1dXOFWW8woyUi7+RkPbAHB5ClqdnzvD1rjA7edv23NXIBB0DXfccS9nnHEWXq+Xfv36M3Xq\nubz33jutxrjdbjIyMrjwwkv48ssvCAZTe6jLyspYtuxzTj75lC6zV3hwBd2OJy5aIzETj+ImZITj\nRaYCyTFWJIKSkdFa4KbIw5VbenDTVGgqEaIMTpiySxbrQgKBQCDofWwoCwJQlOejLOyI08HZA1Bk\nhcj69QB4hgxNjq8ocwRxXmEGqYiFygDQfMVps3FrkxC4gp6NKqvcMOrKTocoB7L91NelN0Q5FcFg\nkAUL5nPeeb/HF4/eiMViaJrzHZw58yJOO+0MDj/85wAYho6iKHg8qQvPvfPOO4wceTDZ2elrHbYj\nQuAKuh0tHnYcjRl41LjANZtDlMEJU24jcFN4cKWWObhpahXUUuAmClsIBAKBQNDbWLfVEbiDi7Io\nrV8KwNDcQZihJmLljlhNCFzTtNi+zRlf3D/QZi7btomF4lWX0yRwI4aZzMHt5+t8CpJA0F2oskqB\nb/fD9hVFIjcjAy3mxeziNpYZGRm899472LbNjBkXU15exoIF85k48ZcA7LXXCJ54Yh777rsfiqLw\n0EP3cdxxJ6BpqaM2li9fzn77HdClNgtXlKDbcbtae3ABomYUqcXKT6KScssc3JQCV5KRJGfdxrLS\nI3A1ubUHVyAQCASC3oZuWKze7OQEDh3gY0NwMwD7Fe5NaNVqZ5Ak4SkpAaCyvAHTcM6JqQSuHt6O\nZTgFq9wZg9JiY2ljGBsnhHpghhC4AsGeQJZl7rjjXtatW8uJJx7DhRf+jqOP/gWTJ/8agPPPn8a+\n+/6EqVNP5+yzp1BUVMzMmVcBTu/b8eN/Tnl5eXK+rVu3kpeXOtc3XQgPrqDbaRa4Bm6vI3AjRhTF\n28KDG6+krKgykgS23X4vXEnRnLzdNAnclh5cXfTCFQgEAkEvZP22emK6c45z59Ri19hISPykYC9K\nn58HON7bRIGpLRscMZyR5SYz0FZsRhqcnF1Z9ePy9EmPjUGn6GRfnxuvKDAlEHQZI0cewiuvvJW8\nX1IyhPvvfzjlWE3TuOyyK7nssivbHCsqKuLNN99vte8f//gHNTWNXep5Fh5cQbejueJteHb04Got\n8mljTs6NJEk774Ub9+AmWhN0FrVF4QxdhCgLBAKBoBfyTWkN4PS/3RzZAMDgwAC8Lg9NX38NgH+/\n/ZPjN6ytAmDQsNRhlpGG7wDwZA5JS/cB27ZZXe9UbR6S1TWVVwUCQe9ACFxBt+NO5uCaaPEqizFL\nR5JlpHhvLVtvFqtJgdueB1d2+uLZlpEW+2TJCYcCMITAFQgEAkEvw7Zt/reyAoD9h+bwZYUjaPfP\n34em0g3oVY6Y9f3EEbiNDVEqy50KyoOHtQ01NI0wkYYNAHizhrY5vjtURGJURZxrgX2z2/bcFQgE\nggRC4Aq6nZY5uK54lTfddE5ikisuVmPN4caqa2cCNy6K7fQIXEmSUGVH4goPrkAgEAh6GxvKG6io\nc8J/iwaGadAd8Xpw4YFUvrsEADU/H8/gwQCsW7EdcBac+w1sWwk1XLcSsJAkFW9g77TY+E2NY1Om\nS2GAyL8VCATfgxC4gm6nZQ6uS3EErW61FriW3kLgxnNizXYKPjUXmUpPiDKAKy5wDVvk4AoEAoGg\nd/H+V06F5D7ZXkqj3wIwMLMffdw5VL7n5M9ljRqNJMvYts3Kr5yCMcP26YOitr2UbKpZDoAnMBxZ\n6Xw7H8u2+aLaqdj8k5wM5DSEPAsEgt6LELiCbqdlDq5LTghcx/sqxUuM27FmsZo4mbYrcBO9vtIU\nogygSs5zihBlgUAgEPQmmiI6H33jCNyfHZDFskonPPnwvocR/OxT9No6kCSyRh8OwLZNddRVO9WR\n9zmwbfufWKiMaJNTgTkj98C02Li2PkRt1DmnH1rQtmKzQCAQtEQIXEG344oL1phhoiUEbjxEWXbF\nBW6LHFwl4cE1duLBTVOIMiBClAUCgUDQK1m8dAsx3UJzyURz1mHZFj7VyyGFP6X61UUAZPz0p2iF\nRQB8/tFGAAqKMulTnNlmvmDFxwCo7lw8WcPTYuP75U7F5kEZHop9nfcICwSC3o0QuIJuxxUv9a8b\nFqoSz8H9nhDlpAe3PYEbF8np9OA2hygLgSsQCASC3kF9U4zXPtsEwKiDsvik/BMAjhpwBLHPvyC6\nZQsAecefADje260b6wA4eMygNtWRY6EyQrXfAJDZZ3Raqid/FwyxvsHJDx5blNPp+QQCQe9HCFxB\nt+NS4t5Rw2r24O4YopxC4Bo7CVG20hqiHBe4woMrEAgEgl7C8++sIxoz8bplQnlfYdgmGS4/Rxcc\nStW//glAzqEH4xs+HNO0eO+NNQDkF2YweHjr9kC2bVO79Q3A8d5m5B3UaftMy2bRZqeCc3+/mxGi\nerJAIOgAQuAKup1E0SjdaJmDmwhRTlRRbg5RVnfmwU30wbXTV2SqOURZFJkSCAQCQc9n2dpKPvrG\nKRZ18M90vq1ZAcCkoRMI/vNfGLW1oCiUnPtbAL74aCO1VU7u7c+PHd7GO9tY9T+ijU74cnbfXyBJ\nSqdt/GB7LWWhKAATBhSkxSMsEAh6P0LgCrqdRA6ubli4lNRtglqFKO8sBzfNfXChRYiy8OAKBAKB\noIdTXhNi3ssrARg82OYbw2kFtE/uXvykNErwow8AKPjlqXj79WXT+hqWfuiI15/8tC9F/VoXeoqG\ntlG3dTEAvuyf4Mse0WkbNzSEeXNrNQCHFmRRkunt9JwCgaBjrFjxDZMmHZ+8v2rVCsaO/Rnjx/88\neVuwYD4AwWCQG2/8IxMmjGPChHHMnn09jY2NKeetrKxk+vTpHHvsUUyadByPPPIQVhc4j4TAFXQ7\nzR5ca5eqKBvtCtz4qnE6i0zFqyjrIgdXIBAIBD2Y2oYo9z2/nFDUwJ+lE+n/KVEzRpaWyWR7P7Yv\neBwA74h9yJswgW2b63j1305ebV4fP2PGDW01nxGto+q7Z7FtA8WVRc6ACZ22sToS45nvyrBsyPe4\nmDAgv9NzCgSCnWPbNi+/vJDLL/8/DKP52nvNmtWMGjWGN998P3mbOvU8AO677y5A4j//eYV///tl\namvreOyxR1POP2fOnQwcOJBFixbzt78t4K233uT11xel/XWoaZ9RINhF1BYeXG2HEGVJjYcbm81i\nVdlpH1xH4Npp7Fkbf0osIXAFAoFA0EOpqAtzz7PLqKyLoHib8O23nPpYEJes8nvpZwQfnQemiVbc\nl77TL6J8WwOLnv8aPWbi9bs47pf7obqaQ4/1SDUV657ENBqRZBcFQ85AUX2dsrE6EuPvq7bSoJto\nssRvhhXjUTof7iwQ/JCwDQO9pma3H68oEI42EqtvwjS/f6wrNzd5Pb0zFiyYz9tvL2bq1PN46qkn\nkvvXrFnNsGF7pXzMrFk3YlkWmqZRWVlBOBwiEMhOOXbTpo30798XO349LcsSbrenQ7btCkLgCrod\nV0KwWjZyXJyatvNtTQpco/nbu7MqyiQF7k6+8btAoqm8KQSuQCDo4SxatIgHH3yQ8vJy+vbty8yZ\nM/nFL37B119/zZQpU/B4mi82pk2bxvTp07Ftmzlz5vD8889jmiaTJk3ij3/8I4oQHj2GL9dV8ff/\nriAUNXDlVeAd9g2NRgwNlWmVwzHeeBZsGzU/n76XXs7a9Q289/paTNPC7VE5+fQDCeQ0hwmH69dS\nvfEFLDMSF7eno/mKOmXjd8EQT68rI2xauGSJc/bqR6FXtAUS9C5sw6D0umswqqr2yPOp+fmU3HJ7\nh0TuiSdOYurU81i27PNW+9euXY2maUyePBHTNBk3bjwXXHAhmqahxue97babefXVlykpGcIpp5ya\ncv6zzjqH22+/hWeeeQbTNJkw4STGjftF51/kDogQZUG3k8jBBbDtuJC04gI3fvHU0oObLDK1Ew8u\nVvoErhIXuKLGlEAg6MmUlpYya9Ysbr31VpYtW8a1117LZZddRk1NDStXrmTs2LEsW7YseZs+fToA\nTz31FO+++y4vvfQSixYt4osvvmD+/Pnd/GoEHSEYivH3l1fwwL++ImQ24R3+NerQL9DtGAMbVGZ8\nrCK/vgRsG/eAgeT931W8/V457yxajWlaZOd6OXXqSPL6ZABgmRFqNr9K5fpnsMwIsuKhYOiZeDKH\n7LaNumXx2uYq5q/eSti08Coy5wzvK/JuBYI9TH5+fspibtnZ2Rx++M9ZsOA5HnxwLl98sZR58+a2\nGnPFFdfw6qvvMHDgYGbNujLl/LZtM23aNBYvXsKTT/6T5cuX8eKL/0776xAeXEG3k8jBBZDseH6t\nbWLbNlK86BRGs8CV422F2q+i3AUhypLogysQCHo+JSUlfPjhh/j9fgzDoKqqCr/fj6ZprFixghEj\nUhcHWrhwIeeccw59+vQBHM/u/fffz+9///s9ab5gF2gM67zxv00sXrqFiBVG7b8BrWgTtmyQHTQ4\nap3MoNVlED+veQ4fx9aBo3nj2VXJGheDhuVx2lkHE9V1dD1GU9UX1G//EMtwCsi4vMUUlExGdacO\nR9wZlm3zVU0Di7fWUBN1UpP6eDTOHl5MnkdLw7sgEPzwkFSVkltu73SIciDgpz7NIcrtcccd9ya3\n+/Xrz9Sp5zJ37l+YMePi5H63243b7ebCCy9hypRJBIP1ZGU1F6SrqqrizjtvY+nSpTQ2xigpGcJZ\nZ/2WF1/8d7se391FCFxBt6MqzStFCQ8ugGVbLXJwW4QoxwWxZbYjNuMFodIZopwwUeTgCgSCno7f\n72fz5s0cd9xxWJbFTTfdREZGBitXrkTTNMaNG4dlWUyYMIGZM2eiaRrr169n2LBhyTlKSkooLS11\nFiI70LpFkiTkTsaMvfHaf8mzV1Fbr/Czk39Hlj+rcxPuQeR4Jf7E367CtCxWbazj/eXbWLqqAtNX\nhdJ/K57cclTLpGRLlJ9s0Bm0NYxkgw00FY+geu+xlJYZmNu3AuDxuhh99BD2G9kXRWmgdsOHNFQu\nwzSaAGchObt4LIGiw5sLO+4CIcNkWWWQj7fXURmJ19wAjuybwy/65+Hq5IdlT73f6UbYvWfpVrsV\nF2px4W4/XJYlvNl+ot4mrC7o8KHEL3wVRSIYDPL44/M4//wL8PudXtSGEcPtdqMoEpdeeiGTJ5/B\nEUeMBcCyDBRFwe/3JecBqKurwjAMdF1Pvucul4qqKq3GpQMhcAXdTqsQZav5A27aprNEhZOvkCDx\npWjvC50MURY5uAKBQJCS4uJili9fztKlS7nwwgsZNGgQOTk5HHbYYZx++ulUV1dz6aWX8sADD/CH\nP/yBcDjcKjfX6/ViWRaxmHORszPy8vyd7mHauH0jw38SIrcAVi+ZS4U+mLPPPbdTc+5psrP9aZ3P\ntm3KqptYtaGGpau288V3m4i4KpADVSgHVJIbjTCgPMbAVTEGl8XQdBtd1qjyDaAuZwhVWSWEYsAW\npxWf5lY49PBBjDzURzi4lvLVrxIKbml+Qkkmr/hgioccg9uXt0u2NsQMvq6oZ9n2Or6tDKK3OIcf\n2CfAKXsV0z+rcwWqdiTd7/eeQti9Z+mpdkPX2Z6Z6UWSJHJzM8jO9vHBB0vweFxcccUVbNu2jSef\nfJwpU6aQm5vBgQcewD/+8ThHHDEKRVF45JEHmTRpEkVFua3mPPjgAykqKuKOO+7guuuuo6Kiguee\ne4rJkyeTm5uRVvuFwBV0O64WIcrYzduGZab04HZU4KbXg5sQuGmbUiAQCLqNRFGQ0aNHc+yxx/LW\nW2/xyCOPJI/7fD6mTZvGnDlz+MMf/oDH4yEajSaPh8NhVFXtkLgFqK5u6rQH96iJv2Hlu/PpO6CR\n/D4h8lnB4mduoKZWJRh1YbkzGT7iQIYN3yvpZfihIMsS2dl+6up2z9ti2zYNIZ3ymhDbqprYXF3L\n5trtbG7YTkwO4nHVkmPXMsQXpqDWoGCzQX6tgRZz06hl0+TOYV1ODg3uPBrdzRedsmGRmRGmsMhk\n8BCLzIwgevhD1n8Za/X8iiuDjLwDySw4GJc7h6YINEVS97kEJ6e2MqxTFoqyoSHMhoYwFeHWc6qS\nxEH5mYwuzKZ/hgcMi5qa9ufcFTr7fncXwu49S0+1G7re9oaGMLZtJ7+Td9wxhzlz7uKwww7D7fZw\nyim/4uSTT6WmppGzzjqPurogJ5xwIooic+SR47jookuoqWmkvLyMM8+czNNPP09RUTF3330/Dz10\nL4cffjg+n4+TTz6Fk0761W59979PFO9RgbtixQpuuOEG1q1bx6BBg7j55ps56KCD2oz75z//yd//\n/neqqqooKSnhj3/8I4cccsieNFWwB1FaClyredu0zeYiUylycK12ikx1RRVlRXhwBQJBL2DJkiU8\n9thjPP7448l9uq5j2zZ33HEHF110ERkZzkVDNBpNCtihQ4dSWlrKgQceCDjFqoYM6XhRIdu2d5on\ntjMCWdkcdOx03vr339hrSAi3RycnL0xO0pFYA8ZGyr6R0WMKuq5gxGQMQ8I0wTQlDAMMS8K0JHRT\nwrTAsCVMW0ZHwpJULFkBRUNyudG8fvweD363G7/Xj8+fgd/nx+X1oKgqsqwgyxIyErIkI0kSiiQh\nSTIJh7URP1dFTJvK6iDhmIFu6uimSdQwCEd1GmMRGiNhwuFGopEmYuEmouEmjEgTZqwJyQjjNmN4\nTQOvYeLRJfbWXewXc6HFXEi2hi73Jap6iah+Kjw+yoa4UV0WqmridsfwuGNkuevxuCvweGJkZERx\na2FaOtZjTc3bipaNLzCcwgEHYEh9seIRVoZhEbNsQoZJg25QFzWojxnUxQzqYjoV4Rg1UZ1UZ0tV\nkhiW5WOfHD/75WTgVeOdE7po9diy7C6buysRdu9Zeqrd0HW2H3jgwbzyylvJuQcOLOG++x7e4bkB\nbBTFxaWXXsmll7YuLGWaNgUFRbz55vvJ+4MHlzB//nxqahqTc9t2+n8D9pjAjUajTJ8+nenTpzN5\n8mQWLlzIjBkzWLx4cauV1k8++YQ5c+bw2GOPsffee7Nw4UKmT5/Om2++SU5Ozp4yV7AHaRV3b7cO\nUU7twY3n4O40RDn9RaZEDq5AIOjJ7LvvvnzzzTe8+OKLTJw4kffff58lS5bw3HPPJdsBJULQHnnk\nEaZMmQLAxIkTmTdvHqNGjUJVVebOncukSZP2uP1+v5/fXnYdX321kqVv/YeCLJ1AjoHXF016iBXF\nQvFaeLx6p5/PNGVMQ8YwZcwGGbNWotaUMA0J0wDDdMSzYYJhObeYKWHYEjEbYrZEVJKdGwq66sJQ\nFWzFhSSrqLKCioxLlnEh4ZIlXJKN3+0i05uNIgWQZZAkkCWQJAtZspHjf5EsDMlCli1UycajGOQp\nISCMjRS/gYWEjYyFQhM+GvFTiYRtS1i2hIkLW83CdgWw1SwsxY8puTGiNpTKNES20KRbhA2TsGl2\nOJpJkyX6+z0MyvQyOMPDwAwvbkU08BAIBF3LHhO4n3zyCbIsc+aZZwJw2mmn8cQTT7BkyRJOOOGE\n5Ljy8nLOP/989tlnHwB++ctfcvvtt7Nu3ToOPfTQPWWuYA+itkzu3yFEOdljMUUOrtmewJW7og+u\n81d4cAUCQU+moKCARx55hNtuu40//elPDB48mL/85S8MGzaMRx55hFtuuYVRo0bh8Xg4/fTTOeec\ncwA488wzqaqq4rTTTkPXdU4++WTO7cb81/79B1B81iXJ+5VVlfzvw7eJNdUi2zqaZOFSbTTFQlFA\nVW0UBRTVRlFsFNVCUWxU1URRLWQ59W+7olgoikU66/kahoxpKhiGjB5TMWIyekxGj0kYUTB0CyNm\noesm0ZhJKGYQky2iqk3IJRHS3IRVLyHVR0z2gu0GXEi4kWw3ku1BkhUkRUJSZSRVQlZlJJeMrMhI\nrvh9VXbGJFy4sfit7Z3/b+/Ow6Os7sb/v+9l1uwLWSAJhLAjEhYVBMQdxQV9lNraalta17b+Hq1t\n1adatb8+tYtb9bG1Lr2K+tTvV21d0bohOyqKILuQARJIAtm3We/7fP+YLBNIJEjIJPHzuq5cM3PP\nvXwy1yRnPnPO+Zwv5dQ1UpwmKU4HKU6TIW4HWR4X2R4nKU6zvYaFEEL0lT5LcH0+H0VFRZ22FRYW\nUlJS0mnbJZdc0unxJ598QnNz82HHdqc3KjWCVITrS05HRwVGLXZpZs1GdzqA6Dq4bT29pqO1SrKt\nuqy6phsdCW5vVWVrmydsq849zgPx9QaJu68N1Lhh4MY+UOPuC9OnT+ef//znYdtHjRrVaehyLMMw\nuPnmm7n55puPc3RfzZDMIcxfcMVXPj4cDNBQd5D62loaG6pprq8nFGjGCgfADqMpCw0r2lOqtybJ\nbT9m7K2NYUaHBXeX15mmjWnauFxAQrDrnQ4RDDoIBFz4A07CfoNIM0QaQlj1LYSbgthaC2FXM35P\nC00J0OB20aC7qdMSCUY8qBYXKuRBBT2ooBcV9ES/UNbAdBqYLgOn28TlMnC5Hbg9Jl6vg6QkJ1mZ\nibhNDY9h4DENvIaO1zRwmzrJDhO3oR9zATEhhOhNfZbgtrS04PF0XrDb7XYTCAS6PWbnzp3cdNNN\n3HTTTaSnp3e7X6zeqNQYa6BWVhtIcUdi5tLGvkcSkpw4kxOoBDSl2ieTJyc3ANEhyl1NMDfsBA4A\nKJu0tN55PyTVtU5M0rUurzmQXu9YEnffGqhxw8CNfaDGLfqWw+UmIzufjOz8XjmfUgplRwiHAgSb\nG7ECTZiEqa+uJRz0Ew61EIn4sW0/SgXRtBCaHsEwIhiOCA6H1alX2eUK43KFSUnp6lrg93toakmn\nsdGLv9YkfMBGr2shKVCHrpUS8DZRm6xTm2JSnWNwINUkpKKJrvInEW5JJNSSREN1IliOw67hdhpk\nprjJTPGQk+5l2JAE8oYkkpZhSnIrhOh3+izB9Xg8hyWzgUAAr7frkvAr+mi4zgAAIABJREFUV67k\n5ptv5vvf/z7XXnttj6/TG5UaYeBWVhuIcauYYb8tzR1zpmrqGkkORocmW6Fwe4W1lpboN96W1XXF\nRX9T27AqRU1NI5p27G+IYGv1x1DE6nTNgfh6g8Td1wZq3DBwY+/NuHt7+QIx+GmahmY4cHkcuDxJ\nGEb0y9HYwipfJpogB7HCjYQC9QRb6gi21BEK1BEJ1oNdj2k0o2kKTQOvN4DXGyArsxYKo+eIRHQa\nGhOpq8+hqSYJVWEzdHc94wMHSAlU0OKt5UCGg8p0k/JMB1XDTZSuYUS8aIFUQvXJhOtTsJuTCYSg\n7GAzZQebO8WpaZCV6iE/O4miockUDU1heE4iDvPo18YVQoje0mcJ7siRI3n22Wc7bfP5fFx44YWH\n7fvSSy/xm9/8hnvvvbfL579Mb1RqjDVQK6sNtLgNXcOyFbbV8U1wKGKhtI4qyu2/T1vBJ6vr39GO\nnccbiaDrh38b/VVZ3byuA+31biNx962BGjcM3NgHatzi6y2aILvRDTcO9xASUg/fRymbSKiOSKCa\nUKAaf2MloZZKVOQgmmZhmjbpaQ2kpzUwckT0mIbGBGpqc9l1cAJNlSYpTQcZd7CcWev3YetB9mU7\nKM1uoSy7geqhBq5hGoZmkG5mkWwPxWzJprEqgf1VLfiDFkpBZa2fylo/67YdAKLteUF2EuMKUplQ\nmM6YvBRJeIUQfarPEtyZM2cSCoV45pln+OY3v8krr7xCVVUVs2fP7rTfmjVruOeee3j66adlaaCv\nkbYEN7YuVLSKcmuCa3UUmYqdA2vb6rA5du1VlKMHAsee4JqyTJAQQoh+RNN0HK50HK50PCmjScmO\nblfKJhKsJtRSQbBlP/76PURClWgokpOaSU5qZkTBfixLp6o6lQMHi9h14CRcDQ1kNpcx7fNS5n5S\nQ3Oiyc6hDkryXOzL2s9BvRxc4C5wceLkIkYmjCbZyqe6RrG7vIFd+xuobQxi2QpfeQO+8gbe/HAv\nDlNnTH4qkwrTmTpmCJmpni//xYQQ4hj1WYLrdDp54oknuPvuu3nggQcYPnw4f/7zn/F6vdx1110A\n3HvvvTzxxBOEw2GuueaaTsc//PDDnHbaaX0VruhjpqETitioTlWUI2hG61u0i2WCAGzbRtc7fzMc\nm+Aq24Je+OJYb09wj/1cQgghxPGiaToO95Boz2/6JMgD2woSbC4l2LQXf0MJYf9+DMMmO6uG7Kwa\nTpgANbUp7CvPZX3lJMzmFnKafIzx7aJ4Rx2Wy2RvfgKf5WuUZis2Vm1hY9UWdE1nTGoRxSdP4qoh\nkwkHTEr2N/BFWT1b9tSw72Az4YjNZl8Nm301PP/+TobnJDF97BCmjc0iJ73raWpCCHEs+izBBRg3\nbhzPP//8Ydvvvffe9vtPP/10X4Yk+om2XlnLUmhoKBSWsjt6cCMRlFKtVbJjenAtdfi7OGbOraJ3\n1sI1pAdXCCHEAKUbLjzJo/AkjyJ16JlY4Sb8DTvxN3xBoGEX2CEy0uvJSK/nhPE7OViVxt6yfNZW\nTSbRX01O4y7ySnZRuDOEleRl36gM1uQGqEi12Vb7Bdtqv+D/7niZiRljmZF7EgvHjMPUR1PbGGTL\n7ho2767h813VNAci7KloZE9FIy8tK6EwN4lZk3I5ZUI2Ce7em04khDg2W7Zs4vbbb+WVV94CYNu2\nLVx77fdwuVzt+1x11fe5+upFNDQ0cP/9v+Wjjz4E4NRTZ3Hzzb8gMfHw+hX79pXx85//kfXr15Oa\nmsZ3v/sDzj//6Kaj9kSfJrhCdMdoTVptBYamE1EWlh2B2N5ZpUDT0A8ZonyozkOUeyfBlXVwhRBC\nDBaGI5HEjGISM4pRdgR/wxc0127CX78DXbfae3b9fiel+3IoLStmV2A62Y0+htVvJ399KQXrwcrL\nYeeEdJal1+HXI3xetZXPq7aS6Ejg5JypzB56CrMm5TJrUi4Ry2bb3lo+2X6Q9TsO0tASxlfeiK+8\nkeff28nUMZnMOXEoE0akSWVmIeJEKcUbb7zKo48+iGF0fJ7esWM7M2acyu9//9Bhxzz00B8AjX/+\n8w2UUvzyl7/gb3/7Kz/5yS2d9rMsi1/84qdMnnwir732FuXlldxyy49JTU1l5szZh533WEiCK/oF\no3XYsWUpdE0HZWErG83o+EZXWRE03dm5B7er6qixPbiqdyqOtfXgSn4rhBBiMNF0E2/qeLyp47Gt\nIC1122iu+Yxg0x48nhBjRu1ldNFeKg9kULI7j3V1RSRZDQyr3kTOvhLGllUwPiGBluIxfFio8Zld\nRlO4mfdLV/B+6QompI/l9PxZjE8fwwmFGZxQmMFV545l695aVn1ezifbDxKO2Hy09QAfbT1AboaX\nM6fmceoJOXhc8jFViL60ePHTvP/+u1x99SKee+7v7dt37NjOqFFjujzmjjt+hW3bOJ1ODh48gN/f\nQkrK4ZXxSkv34vOV8OKLLxAI2BQUDOfSSy/n9ddfkQRXDE5ma69sxLIxdAPscLTIlO7u2MmywXHI\nHFzr8B7aTssC9VoPrgxRFkIIMbjphovEjMkkZkwmHKiiueZTmms2YoVbyMmuJie7mtq6JEp257Hd\nPBVf9skMq/qcvPptuFetZ+5qjXOKT2RncQ5LKaEuWM+Wmu1sqdlOlieTuXmzmDn0JFyGk4kj0pk4\nIp2WcyJ8vK2SFRvLKdnfQHl1C8+9s4N/Lt/FrEm5nD09nywpTCUGGcuyaWoIfuXjDUMDW6O+3n/E\nlQISk10YRs+WzLzgggVcffUi1q//pNP2L77YjtPpZOHCi7EsizPPPIdrr70Rp9OJaUbTyf/+73t4\n883XKSwcySWXXHbYuW3bwjAMnE5n+9KxmqZTVlbao9iOhiS4ol9on4Nrt/bgApaywTikYBQccYgy\nxPbg9tYc3NbzAbZS7QmvEEIIMRg53Jlk5M+j6ISLKS35iPqKtYT9FaSlNjKteCstLW5KduexWytm\nT+YUhjWXkHdgPazfQMH6DVw3dhy1M2fxnnsfuxp2c8BfxQtfvMKS3e9wRt5s5uaditfhxes2mVs8\njLnFw/CVN/DeJ2V8tLUSf9Di3XVlvPdJGSePz2b+jOHkZ8ma1GLgsyybf/z1IxrrA31yvaQUN9+6\n9uQeJbmZmZldbk9NTWXKlGksWHAZNTXV3HnnbTz11OPccMNP2vf56U9v46abfsp99/2aO+74GY8+\n+tdO5ygoGEFu7lDuv/9+rrrqB5SXV/Dqq/86tl+uG5Lgin6hfYiyrTBa59BaykaL+WNUrZWUY4co\nd/Wt1fHswYXWYcqS3wohhPga0A0HSRmT8aRMItjko6FyDYHGXXi9AU6YsJNRRXvZVZLP3rIRlCYU\nMSxURsG+tbB9G57t27gsPx/77LNYkVrLugMbaA638Lrvbd7Z+wGnDTuVM/LnkOJKAqAwN5kfXjiB\nb5wximWf7eP99fuobwrx4ZZKPtxSyeSiDC6YOYJReSlxflWE+Hr53e8ebL8/bFgeV1/9fR5//H86\nJbgulwuXy8WNN97EN76xgIaGepKTO/5WTdPkd7+7n0ceeYAFC+YzYkQh8+adz+rVK3o9XklwRb/Q\nVmTKsuz2HlxbWWgxPbi0DkeOTXBVl3NwY3p9e6mKcmyCaymFIRmuEEKIrxFN03AnjcSdNJKQ/wCN\nB9bQXLMRtyvExPG7GFVUyq6SPPaW5bKv8HKGqUry96yE0lL42z84bVge5553Hqsy6lm5/0OCVoh3\n9n7A0rKVnJp7MvNGnEGqK/phODnByUWzCjnvlOGs2lTOW2v3cqDOz4Zd1WzYVc3Y/FQunl3I+OFp\ncX5VhDh6hqHzrWtPPuYhyikpXurrW3p1iHJXGhoaWLz4aRYtugavNwGAUCiE0xmtqHzzzT/i8su/\nyaxZcwCIRMIYhoHb3XlqgW3b+P1+nnzyyfah1X/+8yOMHj32K8fWHUlwRb9gtv7hRSwVnYMLWLbd\nqYpyew+u0Xkd3EMdjx7cmFHR2Mg8XCGEEF9fTk8WGcMXkJJzGvWVK2mu3oDLGWLCuBJGjSxlly+P\nPaVD2TfiMoZpVeT7lsG+MkJP/Y1pw/I4bf75fJjZxAf71uCP+Fm+bzWryz9iztAZnDP8jPYeXYep\nc3rxMOacmMvH2w6wZM0eyg42s720jj/8Yz3jClJZMLuQsQWS6IqBxTB0UtK++txyw9BIT08AXR0x\nwT1WiYmJLF++FKUUN9zwEyoqylm8+GkuvvhSAMaMGcff//4UEyacgGEYPProQ8ybNx+n09npPLqu\nc+edt3PNNT/knHMuYP36T3nttZd54IFHez1mSXBFv9AxB9fGMNt6cO1OPbjtc3DjUEVZj+mx7XLa\nrxBCCPE1Y7rSyCi4iJTsOTRUrqSp5jOczjDjx/ooGlnGrpLWRHf4ZQzTYxLdJ57khGF5zJx/Pp/k\nB3m3dDnNkRaWlq1k5f4POW3YTM4ZfjpJzuicW0PXmTEhh1PGZ7NhVzWvrfLhK29k2946tv3vesYP\nT+PSOSNl6LIQx4Gu6/zudw/y0EN/5IILzsLlcrNgwX+wcOG3APjBD67jscce5uqrr0DXdebOPYMb\nbrgJgIqKCq66aiHPPPMCOTk5/PrXv+WBB37H73//e7KycvjFL37JuHHjez1mSXBFv9A+RNlW6O1z\ncK1Oc3DpYg6u3WdzcGPCkErKQgghRDvTlUp6wYUk58yhoXIVTdXrcTq6SHTbenRLoolu9RNPMCYv\nn2kXzOfjzGbeLV2BP+LnvdLlrNi/ltPzZnFWwWkkOqLDIjVNo3hUJpOLMtiwq5qXV5Swt7KJrXtq\n2brnE04oTGfBnELG5B++RIkQouemTp3OG2+81/64sHAkDz/8WJf7Op1O/vM/f8Z//ufPDnsuJyeH\nd97pmGM7fvwEXnrpJWpqmo5rz7MkuKJfiF0H19A6enAxOt6iqm0ObhyqKMfOwZUeXCGEEOJwpjOF\n9Pz5JGfPak10P+1IdAv3sbMkjz2luewrvIw8rYr8kg+grJTqxx9nfH4BJ10wn7Vp9bxftoqAFeDt\nPUtZXraa0/Nnc1b+HLwOL9A50V3/RRUvr/BRdrCJTb4aNvlqmFyUwXcvmkhGgiO+L4gQIi4kwRX9\nQqd1cNuXCbLQ9NgqyhHgkHVwu5yDqxEtc6yOSxVlW3pwhRBCiG51SnQrVtJUsx5n6xzdjh7dXMoK\nLyefg+T5lkHpXoJ/+QsnFAznlAsvYE1yDUv3rSJgBXlr93t8ULqKM/Jnc2b+HLyO6NxFTdOYOmYI\nxaMz+XT7QV5Z6WNfVTMbdlVzy0PLKR6VyUWzRlCYmxznV0QI0ZckwRX9Quw6uJ2XCYqpomwfXkW5\n2+ENmg7K6rUqyp2KTEmCK4QQQhyR6UwhveACknNaE93qz9qLURWNLGNnSR57S3MpLbycfCrJ9y2H\nvXsIPvZnThxRyMz5F7EyqYpl+1YTsAK8uftdPihbyRl5szkjJtHVNY3p47KYOnYI67Yd4JWVPsqr\nW/hsZxWf7ayieFQmF88ewYgcSXSF+DqQBFf0C7Hr4LYvE2RbYBxeRRmiw5RtS3UzRDk6D1cp67j0\n4B7nYnVCCCHEoGI6W+foZs+mvnJFe9XlieNKGFVYxs6SfPaW5VJWuJB8VUHe7uWw20fwsT8zdeRI\nZs9fwPKESpbvW4s/EmDJ7ndZWraSM/LncGb+bDxmR6J78vhsTpmQzea99Tz31lZJdIX4GpIEV/QL\n7T24lt25B7fTEOWYBFdvTXC/rAeX4zUHVzJcIYQQ4miZrtTWqsuzW5cX+gxX6zq6bUOX95blUjry\nG+Tb5eT5VkBJCYFHH2N60SjmzL+EFZ4Klu9vTXR977C0dCVn5c/h9PxZHYmurjF3ah4TC1JYs7mC\n11btPizRXTC7kOE5SXF+RYQQx4MkuKJfMPWOdXD12Dm4sUOUD0lwoes5uACaZkRXqz0e6+BKgiuE\nEEJ8ZR3LC82mvmIFzTUbcLuC7YnuzpI8SstyKS36BvnWfvJ3r4BdOwk8spOTRo9hzvmXsty9nxX7\n1+KP+Hnd9zbvl67gzPzTmJt3KklGtBiVrmvMmJDDyeOy+WhbZZeJrvToCjH4SIIr+oVO6+DqHT24\nnYYoxySz0UJTFqrbksZtPbi9tA6uVFEWQgghepXpSiNj+MUk58ymoWIFzTUbcbuCnDB+F6NGlrFz\nVz6lZTmUFl1BgbWPYbtXwRc78H+xg5NGjWb2WReyIuUgK/d/SEvEz+u+f/PO3qXMyZvBZSeeh0a0\ninKnRHdrJa+u2k1FTUeiO354GufPKGDiiPTWQpVCiIFMElzRL5ht6+BaCjNmmSBN00DXwbYPm4ML\n3S0TFLMWrqyDK4QQQvRrDlc6GcMXRNfRrVhOc83n0UR3wk5GFZXi2z2UvWU57Bn5DXKtSvLKPoSd\nXxDY+QXTc3KZdeZcVuf4WXFgHUErxLt7lrO0dBUn50zhrPy55CZkA62J7sQcTh7fOdGNrqNbS35W\nIuefUsBJ47Paa4MIIQYeSXBFv2AYrUOUbYW3bQ6uHU1oNV2P9t52OUT5CHNwe62KckwPbq+cUQgh\nhBCxoonuJSRnz6G+YgUttdFEd/xYH6NH7aW0LJvde4axv+BiMrR68vZ9REbFPkL/+zzFycmcevpc\nPitysrTmE5rDLazZv441+9dxQsZ4ziqYw+jUIjRN60h0J2Tz2RdVvPnhHnbta6D0QBN/fW0LLy0r\n4dyT8pl9Yi4el3xUFmKgkb9a0S8YemyRqY4e3OiTJkQiKLuLBLebIlO93oOLFJkSQggh+oLDnUHm\niEsI58yh8eCHNFd/hmlEKBy+nxEF+6k8kIFvzzA2qLNJ0oMMrfyMnIZdWK++xiink+KTT6Z82lD+\n1biBmkAtm6q3sql6KzkJ2cwdNpOTc6biNt3orevoTh0zhC/K6nhz7V4+21lFdUOAf7z3Bf9cUcKp\nE3M4Y+ow8oYkxvtlEaLPbNmyidtvv5VXXnkLgG3btnDttd/D5XK173PVVd/n6qsXdTruySf/wpo1\nq3jqqWe6PG9DQwN33vkL1qxZQ0JCIosWXcOFF17S6/FLgiv6BbN1yHHEVujtVZRbe3ANHcWhywS1\nLSvUTQLb61WUO+7LEGUhhBDi+HO4M0jPn09K7uk0VX1C08GPsSJN5GRXk5NdTVOTh71luZQY09iZ\ndTI5DbsYVrsVtXIliSvhmsJCaqdM5u20A+z2l1PRXMn/2fEyr+x6k5NzpjE3byY5rcOXR+elMvry\nVPZXNfPWR3tZu7mSYMhi6fp9LF2/jzH5qZw5dRhTxwzBNGT4shiclFK88carPProgxgxdXB27NjO\njBmn8vvfP9TtsZs2fc5zz/2dkSNHdbvPfff9/yQlJfDGG++wY8cObr31/2PEiCJOOGFSr/4ekuCK\nfqF9HVxLtd9v68Ftr6RsxRaZOsIcXFqP6aUEV9M0dKLDk6XIlBBCCNF3DNNLSs4ckrNOpaVuMw0H\n1hL2V5CY6GfCuBLGjvZRUTmEvWU5fJQ0ihSrnqFVm8navRuPz8elCQkw9QTW5+usxEfACrJ832qW\n71vNyJQRzMidxtSsE/GYHoZmJrBo/ngWnl7Eyo3lLF2/j6r6ADtK69hRWkdygpMZE7KZPSmXvCzp\n1RWDy+LFT/P+++9y9dWLeO65v7dv37FjO6NGjen2uJaWFu67714uvfRyNmz4rNt9li//gH//+9+4\nXC4mTDiBc86Zx1tvvSEJrhicOlVRjlkmCIDWqsqHroML3Q9R7ujB7Z0qyhCtpGwrJUOUhRBCiDjQ\ndIOE9BPxpk0i1LKfpupPaandhEGYYUMPMGzoAZpb3Owvz2Jv8hS2N88ks3EPuY27SF/xEZNRTBua\nS+X4PN7PrGO/0URJ/W5K6nfzwo5XKR5yAjNypzMmrYgkr5PzZwxn3skFbCyp5v1Py9hUUkNDc4i3\nPy7l7Y9LGZ6dxKxJOZwyIZskrzPeL48YQJRtEQnXf+XjbV0j2BIgHGjBOkLPi+lIQdONL92nzQUX\nLODqqxexfv0nnbZ/8cV2nE4nCxdejGVZnHnmOVx77Y04ndH3/SOPPMC8efPJyMjsNsEtK9uLYZjk\n5+dTU9MEQEHBcJYtW9qj2I6GJLiiX2ibgxuxFEYXQ5SBrufgdltFuXVMcS/14ELrMGUlQ5SFEEKI\neNI0DVfCMFwJw0gbdg7NNZtoqv6EsL+SBG+A0UV7GV20l/r6RPaVZ7GtYg7KD1kNu8iu8ZH+3qcs\n1DTsUcPZOdzDipQamlxhPq5cz8eV60lxJlOcNYmpWScyMmU4xaMyKR6VyYE6P6s/L2fV5xVUNwTY\nU9nInspG/s/7O5kwIp3p44YwZfQQEj2OeL9Eoh9TtsX+rf+DFao7pvOU9XA/w5nK0PE/6lGSm5mZ\n2eX21NRUpkyZxoIFl1FTU82dd97GU089zg03/ISVK5exe3cJt956O//+95Juz+33BzrN4QVwudwE\nAoEe/iY9Jwmu6BfMmDm1ensP7qFDlLtaJqjrBFZrTZJ7q4oytK2Fq2SIshBCCNFP6IabpCHTScyc\nRthfQXPN57TUbcIKN5GSEv2ZMK6E2rokKg5ksq3yDMKNGkOa9jCkbA+jvtjDGF0jODyHrcMMPh7S\nQj0NLCtbxbKyVaQ4kyjOmsSUISdSlDqCS+aM5OLZhWzfW8fqz8v5ePsBQmGbz0uq+bykmsX6dsYP\nT2P6uCymjM6Unl0xKPzudw+23x82LI+rr/4+jz/+P1xxxZU8/PD9PPjg/3Sas9sVt9tNKBTstC0Y\nDODxeHs9XklwRb9gxKyD29aD2568tg9Rjp2D2zpP9whDlHu3B7c1qZYeXCGEEKJf0TQNpzcXpzeX\njIJzcFBJ+e6PaK7dgrJDpKU2kpbayPgxPpqaPFQcyGTPwRlsrnGS0bSfjIP7mLB3P8V2EP/QdHZl\nG2zKDFOZ0cCystUsK1tNgullfMYYJmaMY0LuWH4wfAJXnjOGz76oYt32A3xeUkPEstnkq2GTr4a/\nvwUjhyZz4sgMTizKJD87sf2zhPj60nSDoeN/dExDlA1dIzXVS11d7w5R7kpDQwOLFz/NokXX4PUm\nABAKhXA6XXz88YfU1tbwwx9eBUA4HCYSiXDeeafz1lsfdDpPfn4+kUiE/fv343YnA7B37x4KCwu/\ncmzdkQRX9AtGew+uiunBbRui3JbgRtr3bxuirLr9o+7dKsoArZ3GkuAKIYQQ/Zim6SSnjyai5ZKW\nN59AYwkt9dvx12/HjrSQmOhnVGIpo0aWEo4YVNekUlVdxO6q6eg1zaS3lDNsyz4mBOqxvQ725brZ\nmqUoy7ZYF/mMdZWfoaExIjmf8eljGDOsiOvHjycS0diwq4p12w7yeUk14YjNrn0N7NrXwL9W+EhO\ncDJpZDoThqcztiCV9GR3vF8qESeabuBwpX/l4w1Dw+VNxBFoQu+us6eXJCYmsnz5UpRS3HDDT6io\nKGfx4qe5+OJLmTdvPvPmzW/fd8mS13jppf/b5TJBXm8Cc+bM5f777+enP72NnTt38c47/+YPf3i4\n12OWBFf0Cx1zcG0MvW0ObtsQ5dbe2JjhyB1DlPtmHVyI6cHttTMKIYQQ4njSdBNPyhg8KWNQ6gJC\nzWWtye4OIsFqHKZFTlY1OVnVALT4XdTWplBVN5md1QnoVc2k1h7gpP2VnB04SCjZZE+mRukQk31Z\nPpbU72HJ7ncxdZPC5AJGp47k3DOK+O75o9hZ1sTnu6rZWFJNTUOQhuYQqz6vYNXnFQBkpXoYW5DK\n2IJUxhWkMSTNE8+XSogu6brO7373IA899EcuuOAsXC43Cxb8BwsXfuuIx1ZUVHDVVQt55pkXyMnJ\n4bbbfslDD/2eiy+ej8fj4cYbb2LixBN6PWZJcEW/YLZXUe7owW1bJggj+jbtqopyt8MyenkdXACj\nNcGVIlNCCCHEwKNpOq7EAlyJBaQNO4dIsI5AYwmBxhL8jSUoK4DXE8TriVZkBgiHDWrrkqmtG8e+\n+ulYB8N462uZdKCKUz+tRjMbKM/QqUw3qczYxrvpO1ni1NE1nWGJuYwoLOCyE/PwWEPZX6axyVfD\nrv0NhCM2B+r8HKjzs2JjOQCpiS7GF6aTl+llRHYSI3KT8bjko7roe1OnTueNN95rf1xYOJKHH37s\niMfNn38R8+df1P44JyeHd95Z0f44JSWFhx9+mJqaJqzj2PMsfzWiX2gfotxVFeXW+badE9y2Objd\nFZk6Hj240VspMiWEEEIMfKYrlUTXVBIzp6KUTailnGDTboJNpQSa9qLsAA6HRdaQWrKG1LYf5w84\nqW9Ip6ahgMZ6N3Z1iCH7GigsqSUxVEvIWU91OhxMa6Q0pYTPUk0avTpu003+uGGcMTUHZySFploP\n5ft0SspaCEds6pqCrPm8vP06GpCT4SU/K5H8rESGDUkkf0gi6cmujtUihBCHkQRX9AvtRaZi1sG1\nD62ifFRDlI9DFWWkyJQQQggxGGma3r70ENmglCISqCLYXEqweS+BpjIioRo0wOMO4XF3DGsGsCyd\npmYvTU2ZNDV50Ro0MmtC5O5rxhuox7QbCXhqqEs5SG2KSV2SQW2SQWOGQU5BJilmBkY4iXCzl8py\nqK4yUWEn5dUtlFe38NHWA+3X8rhM8oYkMCwzgex0L9lpXrLTPQxJ9bSvSiHE15kkuKJfiF0HV2/r\nwW1b97a9yFTP18HtqKJsdf38V4lxgBWZspXCshRhK0I4YkdvLYtwOIytIpTXOairaSAUDBIOR7DD\nYcKRCFY4jB2xsCKR6K0VvVWWBXYElB0d+q1sUAqFQlMKZduggYZq7znXUIBqv227r6JPgta6VdNQ\n0YNR6ChNi341oWnYmoZCb7+PptN+9phvsJXWdtK2+wB69L4WjQBi99E6H6dFo6M1StqWUu64c5j2\n57S2Jw//Rr39GQ2U6ryPat1+2Pm6OL6D1uXd7o7vDVqnOHr4/o+7DKVkAAAag0lEQVRz74JmKyZl\nZjBz2ilxjUMIMTBpmobDMwSHZwiJmVMBsK0gIX8l4ZZygi3lBJr2Y4Wr0VAYhk1KchMpyU2dzqMU\nBAJOWvzDwO8mtdlBYiPkHQyj7QpgNjWhtAaCnoM0e0M0JNqYCTopeQaBBBe2Jw0VSSLY5KSp3sQO\nuQkEPXxR7ueLsjpiGwJNg4xkN9lpHrLSvWQku0lPdpGe5CYj2U1qkhNDlwRYDH6S4Ip+IfYbx7YE\nN9I2RNmMLpiuQqGOfdoS3G7G77cNUVZ27yW4evsc3KM/1rJtAiELfyBCUzBIY8BPU9BPSyhAwN9E\nKNBMOOAnEmjGCvixgwEIRlDhCIQttLBCtxSaBZqto6FhGqAbOg5TwzA0NFPD1EEzotX1dIPWHxW9\nNUEzbAwDdNOmVldohkLXFQ5doTSFoRtYmo7tMLBcBhHNwNIMIppJmOhPBJOwchHBwMLAQm+9jbmv\nDCIY2Oit+3XsY6NjIw2sOP6amyuYGe8ghBCDhm64cCcW4E4sIKl1m7IjhIPVhANVhP0H8DcdIOw/\niLLq0DQbTQOPJ4THEyKDw5eFUQqCoSQCgQwCQRehgEmkBexGBZURaAlihRoIWSECRpigO0TAHcKf\nEsHvchAwPTTgoVlLIGy7qQ27qG10srXGhQo7UWEXRByAhqZF5/m2Jb3JCU6SE5ykJDhJ9jpbHztI\nSXDiML/6sjJCxJskuKJfMIyObyAdWjShDVnRhFZ3RRdJt2MWh+7owe1mDq4ePUbZ4V6Jz1aqveOq\nqsHPhpaD1LW0UBdsImAHqa6vIdzSgO2vRws2YwT8mKEIZkhhWuCIaJi2gWEZ6LYDTRkYDhPdaaK5\nNZxuHbcLDBcYToUjUcdM1TFNA9ME04xgmjamGcFhWhhG17+3UhDGJICLIE4CqvU25nEAZ8z96PYw\nJjbdNGZtna/9lKY6eoY7+l9VawdxNPDYW03F3G/fHnOcarsfvT3seodtObRvOHbfL3vhYo47bLfu\nzxn7/Jf1kX75tY9RT08dx05cXdkMa/7qawwKIURPaLqJ05ON05MNaRNJbd2ulEUkWEckVEskWEug\nuZpgSw2RUB3Y9eha9POJpoHbFcLtCgFN3V4HwLJchEIJhMIOQiEHkaCOFdRQYYUK2dihIFbET8Sy\niVgWIWURMiIEjAgBFCEDAppGsFmjPODCV+UiRPQnbLtQthMiDpRl4jacJDg9JDjdJLidrT8mXrej\n9dYkwe3A6zZJ9DrwWxBoCeIwdFwOo/1zmhDx0KcJ7pYtW7jrrrvYuXMnw4cP55577qG4uPiw/V5/\n/XUefPBBqqurOeWUU/jNb35DZmZmX4Yq+pgZM2TGoUWT04AVTWh1V3SdODsQm+C2FZnqpge3NcG1\n7dBhz0Usm2Z/mCZ/mMaWEPUtAWpaGqgLNNEQaCLQVE/EXweBJvRAM0YohCNkUzPpDEjPofTzz1Bb\ntqHbDlBODN1BrssEj4nmcqO73ZjJNg5HBKcjjNMRweEIRx87wzgdQRyOyGFxHcpSOgGiCWkLnk4J\nacCKSVyVs32/IM7uE9WvSEfh0MCpgVMHp67h0HUcuobZet80NExNx9F6a+odjw1dx6HpmHrbj4au\ntf4QrU4dfRztJTda7xuHbgMcpk5mRiJ1tc3YlhowRTYMQyM9PfG4Vw08HgZq7LFxi86WLFnCI488\nQkVFBUOHDuXmm2/m7LPPpr6+njvuuIO1a9eSlJTEj370IxYuXAhE5yM+8MADvPDCC1iWxYIFC7j9\n9tsxDOnlEaIrmmbgcGfgcGcAkDSk4zmlFLYVwAo3YIUbCQfrCTTXEQ7Uo6xmQsF6NLsZXQ92mu1h\nGHZ7b/DRMbFtsCyj9UfHsnRsC6xIGGVFsCPN2JaGbSlsK/r5yrIUylZYQYUdUFgqOootrKDGhgMK\nLKURsaPbQ0ojgk7YBkvTsTBRhokyHGCaGA43DtPEdDjRTRdOhwPDdGA6HDhMBw6nE6dp4jRMnKaB\n0zBxORy4TBOnw8Bh6hi6jt76+cPQNXRdwzB0jNbHhh4d1aZr2oD5jCB6X58luMFgkOuvv57rr7+e\nhQsX8sorr3DDDTfw7rvvkpCQ0L7ftm3b+NWvfsXTTz/N2LFj+fWvf83tt9/OE0880VehijiI7cE1\nYnpwbWWjuVwAqJgeXKc7+qEq4A9TVeenKRCmvsVPdXMjdf5m0qxyitxQUVXOc2v/ghZswgz4MUNh\nnBFwWjoOy8CwHNEeVduJUzkYYjrB40Bzm2judHR3Oo5kG6czTMhj4AcSh3qYkA0ORwtORz2G8eUf\n+m2lEcRBEBeNJEYTUjuakMb2sAaUmyAugrgI4CDSS4mqy9DxGjpe08BrGnhMnQSHQUaSBy1s4dZ1\n3IaO09Bx6jpOQ8Oldzw2+9G3sIahYeo6uqbFzLMVQvSUz+fjjjvu4Omnn2bq1KmsXr2aa6+9luXL\nl3P33Xfj9XpZvXo127dv55prrmH06NEUFxfz3HPP8cEHH/Dqq6+iaRrXXXcdTz/9NNdcc028fyUh\nBhxN0zBMD4bpAU82HiB5yOFfKCplY1sB7EgzVqSFcLCJYEsToUAj4VAzVrgZO9yCbQXQVAhND6Pr\nEUzz8OlZug66buFw9N7UraOlVPQzEUrDVhrKbhuNp6GUBhaoiIZSoFT0ltb7ARv8rfdjzxd7i2od\nu6RiBhqpLvbrdEzbmC3VcXxX56abmhwah2870uvQfqetjkcXx2udr334BTuO66oKSE/DiX09j+pj\nVdtLcdTfe2uMKJ7LyLGTj/bAo9JnCe7atWvRdZ0rr7wSgMsvv5y///3vLFu2jPnz57fv99prr3HW\nWWcxeXL0F7/11luZOXMmVVVV0os7iCV5He339+4LtN//++rlDK06SCFQtX0rLzz9IEawBbMlExjL\ngfJa3nn0tzgsE8N2oFlOdOXCURSGKZBshiisTUJzp2AkgcNp4XSEcThbe1edYRyOME5nM05HGF3v\n/i91n5VDpRpCs9NLwHBRr5KiPah2W4LqbE1O3QRwE8RJUDkJ9tKfmalprUmqjqc1WfWaOl7D6GJ7\nNJH1GkZ7Aa9YA7VXTghxbAoLC1m1ahUJCQlEIhGqqqpISEjA6XTy7rvv8u9//xuXy8WJJ57IhRde\nyMsvv0xxcTGvvPIK3/3ud8nKygLguuuu4+GHH5YEV4jjSNN0DNOLYXpxAO5ESMo48nFKKZQdxLZa\nhyyHAoQCLYRCfiKhIFYoiBXyY4X82FYI2wpFp3SpCIoIEEHTLDTdbp1HrNB0G11XaFq0dseXfV7q\n/vcBQ4tmrTL24+vr4BfvDJ4E1+fzUVRU1GlbYWEhJSUlnbaVlJQwZcqU9sdpaWmkpKTg8/kkwR3E\nUhNdOEydcMTm9Q8O4pkW3b4uuISxiQEKAU8wxPzVGwA46A2wcehYwKTOPPOwd3LErqGATRimYtqc\nXV8pJqUAzYVmeNAND2mWC4JwkAz+ac37yr+rDq2JqN6aiHZOVGOfa09cDQOnlP4XQvSChIQESktL\nmTdvHrZtc/fdd7N3715M0yQ/P799v8LCQt5++20g2jaPGjWq03M+nw+lBs5UASG+LjRNQzPc6IYb\n05mCywsJRz7sqCilQFkoFUHDIinJSXVVPeFQGCsSIRIKRBPpcBgViWBbYWwrgm1FV2mwrDC2HcG2\nLWzbQtkWtrLAjq7UoGJWbFAq2putWldpULHdsq0rM2golAKtU1GLmPtaa40NreO5Q/91RR+39tNq\nsds6znXEf3exvbHdL3rQxca22hptJzikW/cI1+3pv2Gti2sek6P8969sjRYr69ivewR9luC2tLTg\n8Xg6bXO73QQCgU7b/H4/bre70zaPx4Pf7+/RdTRNozcqoLdNjh9ok+QHbtw63zp3LIuXbAXLiVU5\nHCN7DwC+oS4OpDnIqo0WZIgYGl6tisRwNU2O6FeZGjYO3cbpALdLx2OkE/Cn4PZEi8woNDTNhaZ7\n0E0vpjMBh9OL4fCiG223HgzTi976baluuturMQMkBkJs2lxKY7hjeI/H0El0mbh1HU/bMGBHTO9q\na5Ka0Najahq4DT3uHwgH7vtE4u5rAzX2gRp3X8nNzWXDhg2sW7eOG2+8kR/84AeHtb2xbfShbbPH\n48G2bUKhEK7WaSRfRtpmibsvSdx9QSP6lb0DXdfwJCSQGHZ1v3xjP6TrGqmpCdTVNQ+ouGHgxh4b\n9/HUZwmux+M5LJkNBAJ4vd5O27pLeg/drzuZmYnHFughUlN7+zuvvjEQ41541hgWnjWm9dGCzk9e\nffj+5x7xjBcde1Ax0oEHhqb36jnjbSC+T0DijoeBGvtAjft4M81o8z9z5kzOPfdcNm3aRDAY7LRP\nbBvtdrs7Pe/3+zFNs0fJLUjb3Ebi7lsSd9+SuPveQI39eMfdZ2MeR44cic/n67TN5/N1GvIEUFRU\n1Gm/mpoa6uvrDxveLIQQQoijs2zZMr73ve912hYOhykoKCAcDrN///727bFt9KFts8/nY+TIkX0S\nsxBCCHE0+izBnTlzJqFQiGeeeYZwOMyLL75IVVUVs2fP7rTfhRdeyNtvv826desIBoM88MADnHba\naaSlpfVVqEIIIcSgNGHCBDZt2sTLL7+MbdssW7aMZcuWccUVV3DWWWdx//334/f72bhxI6+//joX\nXRQdCXPxxRfz1FNPUVFRQVVVFY8//jgLFiw4wtWEEEKIvqcpdfQFnr+qbdu2cffdd7N9+3aGDx/O\n3XffTXFxMXfddRcA9957LxBdo+/hhx/m4MGDTJ8+nd/+9rdkZPSgbJwQQgghvtS6dev47//+b3bv\n3s2IESP4+c9/zowZM6irq+NXv/oVa9aswev18uMf/5jLL78cAMuy+NOf/sRLL71EOBzmoosuknVw\nhRBC9Et9muAKIYQQQgghhBDHi6w7IoQQQgghhBBiUJAEVwghhBBCCCHEoCAJrhBCCCGEEEKIQUES\nXCGEEEIIIYQQg4IkuF3YsmULl19+OcXFxSxYsIDPPvss3iEdlY0bNx62/FJ/tm7dOhYuXMi0adM4\n++yzef755+MdUo8sWbKE888/nylTpnDBBRfw7rvvxjuko1JVVcXMmTNZunRpvEPpkaeeeooTTjiB\nKVOmtP+sW7cu3mEdUUVFBddddx1Tp07ltNNOY/HixfEOqUdeffXVTq/1lClTGDduHHfeeWe8Qzui\nTz/9lP/4j/9g6tSpzJs3j9deey3eIYleIG1z35K2OT6kbe4b0jb3vT5tm5XoJBAIqDlz5qjnnntO\nhUIh9cILL6gZM2aopqameId2RLZtqxdeeEFNmzZNnXzyyfEOp0fq6urUSSedpF599VVlWZbatGmT\nOumkk9SqVaviHdqXKikpUZMnT1affPKJUkqpVatWqYkTJ6rq6uo4R9Zz1157rRo3bpx6//334x1K\nj9xyyy3qySefjHcYR8W2bXXppZeq++67T4VCIbVjxw510kkntb9vBpJVq1apWbNmqfLy8niH8qUi\nkYiaMWOGevPNN5VSSn388cdqwoQJqrS0NM6RiWMhbXPfkrY5fqRtPv6kbe57fd02Sw/uIdauXYuu\n61x55ZU4HA4uv/xyMjMzWbZsWbxDO6K//OUvLF68mOuvvz7eofTY/v37mTt3LhdddBG6rjNx4kRO\nOeUUPv3003iH9qUKCwtZtWoVU6dOJRKJUFVVRUJCAk6nM96h9cg//vEPPB4Pubm58Q6lx7Zu3cr4\n8ePjHcZR2bBhAwcOHODWW2/F4XAwevRonn/+eQoLC+Md2lFpbm7mtttu4+677yYnJyfe4XyphoYG\nampqsCwLpRSapuFwOGS91gFO2ua+JW1zfEjb3Dekbe57fd02S4J7CJ/PR1FRUadthYWFlJSUxCmi\nnrvssst45ZVXmDRpUrxD6bHx48fzhz/8of1xfX0969atY9y4cXGMqmcSEhIoLS3lxBNP5Oc//zk3\n33wziYmJ8Q7riHw+H3/729+4++674x1Kj/n9fnw+H4sXL2bWrFmcf/75vPjii/EO64g2b97M6NGj\n+cMf/sCsWbOYN28eGzZsIC0tLd6hHZUnn3ySMWPGcPbZZ8c7lCNKS0vjyiuv5JZbbmHixIl8+9vf\n5s477xxQHxjF4aRt7lvSNvc9aZv7jrTNfa+v22bzuJx1AGtpacHj8XTa5na7CQQCcYqo57KysuId\nwjFpbGzk+uuvZ+LEiZx55pnxDqdHcnNz2bBhA+vWrePGG29k+PDhzJw5M95hdSsSifDzn/+c//qv\n/yI1NTXe4fRYVVUV06ZN41vf+hZ/+tOf2LhxI9dffz1Dhgxh7ty58Q6vW/X19Xz44YfMmDGDpUuX\nsmnTJn74wx+Sn5/P9OnT4x1ejzQ3N/Pss8/yxBNPxDuUHrFtG7fbzcMPP8yZZ57J6tWr+elPf8rE\niRMHxIdz0TVpm+NH2ubjT9rmviVtc9/r67ZZenAP4fF4DmswA4EAXq83ThF9PZSWlvLNb36TlJQU\nHn30UXR9YLw1TdPE4XAwc+ZMzj33XN577714h/SlHnvsMcaPH9+vG56u5Ofn8+yzzzJ37lycTifT\np09nwYIF/f71djqdpKSkcN111+F0OtsLK/T3uGO9++67DB06lOLi4niH0iNvv/02Gzdu5LzzzsPp\ndHL66adz+umn8/LLL8c7NHEMpG2OD2mb+4a0zX1L2ua+19dt88D4T9WHRo4cic/n67TN5/MxatSo\nOEU0+G3evJlvfOMbzJ49m8ceewy32x3vkI5o2bJlfO973+u0LRwOk5SUFJ+AemjJkiW88cYbTJ8+\nnenTp7N//35uueUW/vrXv8Y7tC+1efPmw2IMBoP9fl5VYWEhlmVhWVb7trb5JwPF0qVLOf/88+Md\nRo+Vl5cTCoU6bTNNU+bgDnDSNvc9aZv7jrTNfUva5r7X523zcSldNYAFg0E1e/ZstXjx4k6VGpub\nm+MdWo+tXbt2wFRqPHjwoJoxY4Z6/PHH4x3KUTlw4ICaNm2a+te//qUsy1IffPCBmjp1qtq5c2e8\nQzsqZ5xxxoCo1FhSUqImTZqk3nzzTWVZllq9erUqLi5WmzZtindoX8rv96s5c+aohx56SIXDYfXJ\nJ5+o4uJitX79+niH1mOnn366WrNmTbzD6LFt27apiRMnqhdffFHZtq0+/PBDNWXKFLVx48Z4hyaO\ngbTNfUva5viStvn4kra57/V12ywJbhe2bt2qrrjiClVcXKwWLFgwoN7wSg2sRvTPf/6zGjNmjCou\nLu7088ADD8Q7tCP6+OOP1aWXXqqmTJmiLr300gH1j6bNQGlElVLqvffeUxdeeKGaPHmyOvfcc9tL\nzfd3u3fvVosWLVInnXSSOuOMM9SLL74Y75B6LBKJqLFjxw64D4fvvfeeuvjii9WUKVPUBRdcoN5+\n++14hyR6gbTNfUfa5viStvn4k7a57/Vl26wpNYD644UQQgghhBBCiG7IHFwhhBBCCCGEEIOCJLhC\nCCGEEEIIIQYFSXCFEEIIIYQQQgwKkuAKIYQQQgghhBgUJMEVQgghhBBCCDEoSIIrhBBCCCGEEGJQ\nkARXiDi47bbbGDt2bLc/jzzyCB9++CFjx44lGAwe93jKysrar33TTTd95eN37dp1xH1vvfXW9mv1\nZH8hhBCiL0jbLG2zGBxkHVwh4qCxsZFAIADA+vXr+clPfsL777+P0+kEwOv14nA4qK+vZ8iQIcc9\nnrKyMs466yyee+45xowZQ3Jy8lEdb1kWNTU1pKenYxjGl+7b2NiIz+dj4cKFLFmyhKKiomMJXQgh\nhOgV0jZL2ywGBzPeAQjxdZSUlERSUhIAKSkpAGRmZuJyuTrt1xcNaKy0tLSjbkABDMPocaxJSUmk\np6cf9TWEEEKI40naZmmbxeAgQ5SF6KcOHQY1duxYlixZwkUXXcSJJ57IokWLKC8v52c/+xnFxcWc\ne+65rF27tv34yspKbrrpJqZMmcKcOXO4++67aW5u7tG124Y1ffDBB5xzzjlMnjyZm2++mf3793Pt\ntdcyefJkFixYwLZt2zrt3zas6cwzz+SZZ57hO9/5DpMmTWLevHksW7asl18hIYQQom9J2yxE/ycJ\nrhADyP33389dd93Fs88+y+bNm7n44osZP348L730EqNGjeKee+4BQCnFj3/8YxwOBy+88AKPPvoo\n27Zt44477jiq6/3pT3/ioYce4rHHHuOdd97hsssuY/78+bzwwgu43W7++Mc/fumxV155JW+88QZj\nx47ljjvuIBwOH9PvL4QQQvQ30jYL0b/IEGUhBpCrrrqKk046CYAZM2ZQWlrKokWLALjiiiu4/vrr\nsSyLjz76CJ/Px//+7//icDgA+O1vf8t5551HRUUFOTk5PbreDTfcwMSJEwEYN24cQ4cO5ZJLLgFg\nwYIFPPHEE90ee9FFFzF//nwAbrzxRhYsWEBFRQX5+flf7ZcXQggh+iFpm4XoXyTBFWIAKSgoaL/v\n8Xg6NUhutxvbtolEIuzatYumpiZOPvnkw87h8/l63Igeer28vLxO1wuFQt0eO2LEiPb7iYmJAPIt\nsRBCiEFH2mYh+hdJcIUYQEyz85+srnc9yyASiVBQUNDlt7hHUxyjp9frStu307GkaLsQQojBRtpm\nIfoXmYMrxCBUVFRERUUFSUlJDB8+nOHDhxOJRLjvvvtoamqKd3hCCCHE1460zUL0DUlwhRiEZs2a\nRVFREbfccgubN29m06ZN/OxnP6O2tpasrKx4hyeEEEJ87UjbLETfkARXiEFI13Uee+wxEhMT+c53\nvsOiRYsYMWIEjz76aLxDE0IIIb6WpG0Wom9oSgbeC/G1V1ZWxllnncWSJUsoKioadNcTQgghBhpp\nm4X4aqQHVwjRrra2loaGhuN6jcbGRmpqao7rNYQQQojBQtpmIY6OJLhCiHbf/va3+eUvf3lcr3HP\nPfewcOHC43oNIYQQYrCQtlmIoyNDlIUQQgghhBBCDArSgyuEEEIIIYQQYlCQBFcIIYQQQgghxKAg\nCa4QQgghhBBCiEFBElwhhBBCCCGEEIOCJLhCCCGEEEIIIQYFSXCFEEIIIYQQQgwK/w8Q2qPqU0au\nyAAAAABJRU5ErkJggg==\n",
"text/plain": [
""
]
},
"metadata": {},
"output_type": "display_data"
}
],
"source": [
"IC = [C0,T0,Tcf] # initial condition\n",
"t = np.linspace(0,8.0,2000) # simulation time grid\n",
"\n",
"qList = np.linspace(153,154,11)\n",
"\n",
"plt.figure(figsize=(16,4)) # setup figure \n",
"for qc in qList: # for each flowrate q_c\n",
" X = odeint(deriv,IC,t) # perform simulation\n",
" plotReactor(t,X) # plot the results\n",
" \n",
"plt.legend(qList)"
]
},
{
"cell_type": "markdown",
"metadata": {},
"source": [
"There's a clear bifurcation when operated without feedback control. At cooling flowrates less than 153.7 liters/minute, the reactor goes to a high conversion steady state with greater than 95% conversion and a reactor temperature higher than about 410K. Coolant flowrates less than 153.8 liters/minute result in uneconomic operation at low conversion.\n",
"\n",
"For the remainder of this notebook, our objective will be to achieve stable operation of the reactor at a high conversion steady state but with an operating temperature below 400 K, an operating condition that does not appear to be possible without feedback control."
]
},
{
"cell_type": "markdown",
"metadata": {},
"source": [
"## Simulation 2. Fixed Time Grid\n",
"\n",
"Introducing feedback control requires a change in the simulation strategy. \n",
"\n",
"The new approach will be to break the simulation interval up into small time steps of length $dt$. At each breakpoint a PID control calculation will be performed, the coolant flow updated, then `odeint` will be used to simulate the reactor up to the next breakpoint.\n",
"\n",
"The following cell demontrates the simulation strategy assuming a constant coolant flowrate. Note the use of Python lists to log simulation values for later plotting."
]
},
{
"cell_type": "code",
"execution_count": 5,
"metadata": {},
"outputs": [
{
"data": {
"image/png": "iVBORw0KGgoAAAANSUhEUgAABGwAAAEMCAYAAACRPCChAAAABHNCSVQICAgIfAhkiAAAAAlwSFlz\nAAALEgAACxIB0t1+/AAAIABJREFUeJzs3Xd8Tff/wPHXHUlu9iKxySBWECORSvhWlQqlarS0qBmj\nFFVq71UVJEFQu9r6US1VSmlRexatLalNlkR2bnLv74/UrdtcJMgo7+fjkcfNPfeMz33n5nPPeZ/P\nUOj1ej1CCCGEEEIIIYQQothQFnUBhBBCCCGEEEIIIYQxSdgIIYQQQgghhBBCFDOSsBFCCCGEEEII\nIYQoZiRhI4QQQgghhBBCCFHMSMJGCCGEEEIIIYQQopiRhI0Qz9HznHRNJnATQogXl9TxQgjx3yF1\ntmkSl4InCRtRpH744Qfef/99GjRogK+vL127dmXPnj1FXayncuzYMT755JNn3k9iYiJDhgwhKioK\ngBs3buDl5cXevXufed9CCFFcfPrpp3h5eT325/Dhw0VdzAIxb948Nm3aVNTFEEK8RArrnPvw4cN4\neXlx5coVALp27crQoUOf+3EeOHfuHF5eXuzcudNo+e3bt/Hy8qJhw4a5kgqzZ8+mfv36ZGdn5+kY\nP//8M9OnT39uZX7Y474DFy5cCBR8DJ9WaGgoGzduLOpivPDURV0A8XLS6/WMHDmSHTt28N5779G3\nb1+ys7PZsmULffv2ZdKkSbz77rtFXcx82bBhA9euXXvm/Zw/f55t27YxaNAgAFxcXFi3bh0eHh7P\nvG8hhCguBgwYYFTPBwcH06xZMzp27GhY5unpWRRFK1BZWVksWrSIqVOnFnVRhBAvgaI+554wYQJm\nZmYFtn8vLy/s7Ow4ffo0zZo1Myw/ePAg9vb23Lt3j3PnzlG9enXDaydOnKBBgwaoVKo8HWP16tWU\nKFHiuZf9gd69e/P666/nWl66dOkCO+bzsGDBAiZOnFjUxXjhScJGFIl169bxww8/sGLFCho2bGhY\n/uqrr2Jubs706dNp3rw5Tk5ORVjK4sHc3Jw6deoUdTGEEOK5qlChAhUqVDA8V6vVlCpVSuo7IYR4\njor6nLugE+9KpZJ69erx+++/Gy0/ePAgr776KkePHuXAgQOGhE1mZiZ//vknH3/8cYGWKz/KlSsn\n333ikaRLlCgSq1atolmzZkZfHA98+OGHvPvuu6SkpACwdetW2rVrR+3atXnttddYunSpUdNGLy8v\nNm3axIcffkidOnVo1KgR4eHhRvuMj4/nk08+wc/PjwYNGjB48GDu3r1reP3u3bsMGTKEevXqUa9e\nPT755BPu3btneP3TTz9l+PDhLFmyhMDAQGrVqkVwcLBhH59++infffcdp06dwsvLixs3bhAWFkbn\nzp2ZMGECPj4+9OnTB4DLly8zcOBAfH19qVmzJi1atGD9+vVATjPSbt26ARAUFERYWJjJLlEHDx7k\n3XffxcfHh8DAQGbPnk1mZqbh9aZNm7Jy5UpGjRpFvXr18PX1ZcqUKWi12qf7gwkhRBG5c+cOgwcP\npm7dutSvX5+RI0eSmJhoeH348OGMHDmS8PBwXnnlFXx8fBg/fjzJyclMmDCBunXr0qRJE0M9+2Cb\noUOHMn/+fHx9ffHz82PKlClG9aher2fx4sU0bdoUb29v2rdvz9GjRw2vHzhwAC8vL9atW0fDhg1p\n3LgxqampJCQkMGHCBBo3bkyNGjUIDAxk1qxZZGVlkZWVRY0aNQAYO3YsH3zwAQCNGzdm7ty5Ru97\n8ODBhtevXr2Kl5cXa9asITAwEF9fX65fvw7A+vXradGiBTVr1qRVq1bs2LHjucZfCPHflp9zbnjy\neXdmZibh4eE0b96cWrVq0a5dO3799ddHHv/h7jwPukudOHGCDh064O3tTYsWLXJ1Z9q/fz/t2rWj\nVq1avP322/z888+G82tT/Pz8+OOPP9DpdIZlhw4dokGDBvj5+XHw4EHD8rNnz5KRkYGfnx+QU9d/\n8cUXBAUFUbNmTerXr8+HH35oOMfv2rUrR44cYevWrXh5eRn2c/LkSTp37kytWrUIDAwkLCzM6PhN\nmzZl7ty5hlg+z26wsbGxfPrppwQEBFCnTh369Olj6IL2IFaxsbGG9QcOHIiPjw9ZWVmGZe3atWPu\n3LmGv8k333yDv78/AQEBJCcnc//+fSZNmkTjxo2pWbMmAQEBTJ8+3XAt8SAWEydOpGvXrob9fvvt\nt7zxxhvUrFmToKAgtm3b9tze98tKEjai0N29e5fIyEgCAwNNvl62bFlGjx5N+fLl+fLLLxk2bBi+\nvr4sWLCAdu3aMX/+fGbPnm20zdSpU6lQoQKLFi0yJDoe9MvNysrigw8+4MSJE4wZM4bPPvuMqKgo\n+vfvD0BqairdunXj4sWLTJs2jSlTpnDy5EmCg4ON+rb++uuv/PLLL0yaNIlp06Zx8uRJZsyYAeQ0\n7W/SpAmVK1dm3bp1uLi4AHD69Glu3brFwoUL6dmzJ8nJyXTr1o2MjAw+//xzFi5ciLu7O+PGjSMq\nKooaNWowfvx4AObOnWvUNeCBX375hR49elCxYkVCQ0Pp3bs3X331Va7xc8LCwlAoFISFhdGrVy++\n/PJLowsWIYQo7pKTk3n//feJjIxkxowZTJo0iWPHjhEcHGx0Yvzzzz9z9OhRPvvsM/r06cO6det4\n++23yc7OZsGCBdSsWZNJkyZx+/ZtwzZ79+5l+/btTJ8+ncGDB/Ptt98yadIkw+uzZ89mwYIFvPvu\nuyxYsICKFSvSs2dPzp8/b1TG1atXM2vWLEaPHo2VlRVDhw7l8OHDjBo1ii+++IK2bduyfPlyNm3a\nhFqt5quvvgJyuoCNGzcuX/FYsmQJ48ePZ8yYMZQvX561a9cyfvx4mjdvzqJFi2jYsCEfffTRf3Ys\nOCHE85Wfc24gT+fdw4cPZ/ny5bz//vuEh4fj6elJ//79H5u0+bfhw4fz1ltvsXjxYsqUKcOwYcNI\nSEgAcoYGCA4OpmLFioSHhxMYGMjw4cMfu78GDRqQkpLC5cuXAbhy5QrR0dH4+/vj5+fH8ePHDQn5\nkydP4uTkZEg4LF26lPDwcN577z2WL1/OsGHDOHToEJ999hmQ06WrevXqvPLKK6xbt85Qxu7du+Pg\n4EBYWBh9+vRh2bJlua5Pli1bRtu2bfn8889NJswe0Ol0hqT+g5+Hv+MelpyczLvvvsvJkycZNWoU\nn3/+Offu3aNLly7cvn0bf39/1Go1R44cAXISUseOHSM1NZVz584BOTeyz507Z/S5WLlyJTNnzmTM\nmDHY2NgwbNgwDhw4wKhRo1i6dClvv/02q1at4rvvvgMwxKJ3795MmDDBsGzs2LE0bdqURYsW0ahR\nI4YOHcovv/zy2L+feDzpEiUK3YOM9ZP6ZWZnZxMWFkaHDh0YNWoUAAEBASgUChYtWkTv3r0NzTcD\nAgIYMWIEAA0bNuSnn35i7969NGnShN27d3PhwgU2bdpE1apVAXB1dWXQoEFcv36dvXv3cvPmTbZv\n307ZsmUBqF69Oi1btuTXX3819IfNzMxkyZIl2NnZAXDp0iXWrFkD5DTtd3JyIiEhwahJY1ZWFmPG\njKFSpUoAnDlzBjc3N+bOnYutrS0AtWvXxtfXlxMnTtC+fXtD01EvLy9KlSqV625CaGgo/v7+zJo1\nC4DAwEDs7e0ZOXIk586do1q1agB4eHgYBkh75ZVX+PXXX9m7dy9dunTJ419KCCGK1saNG4mOjmbH\njh2UKlUKgKpVq9KqVSv27NnDq6++CuTUtWFhYdjZ2REQEMD//d//oVKpmDJlCgqFggoVKrBz507O\nnTtn+O5JT09n8eLFhguVrKwsZs6caWgmv3r1aoYOHUqvXr2AnLq2c+fOLFy4kNDQUEMZe/bsSZMm\nTYCcGwB6vZ6pU6dSv359APz9/fn11185fvw47du3p3bt2gCUL18+32OTdezY0TDOQVZWFuHh4XTu\n3NlQ5sDAQO7du8f8+fMNZRJCvLzyes4NeTvvjo6OZvv27cyePZs2bdoAOS0Eo6OjmTdvnqFOfpI+\nffrQuXNnQ9neeOMNDh8+TIsWLVi6dCkeHh7MnTsXhUJB48aNSU5O5ssvv3zk/qpVq4atrS2nTp2i\nSpUqHDx4kAoVKlC2bFnUajVpaWmcPHkSPz8/Tp48ia+vLwqFAoDo6GgGDx7Me++9B4Cvry+RkZHs\n2rULyOnSZWNjg4ODg+Ecf9GiRZQvX57w8HDDODiWlpZMmjSJ3r174+zsDEDNmjUNLSUfZ/LkyUye\nPNloWVBQUK6Wl5DTguXOnTv89NNPlCtXDshpYfTaa6+xfPlyxowZQ+3atTl69ChBQUFcunSJtLQ0\n3NzcOHHiBN7e3hw8eBAbGxvq1KnD8ePHAePvsvT0dLKzs5k0aZIh0eTv78/u3bs5fvw4nTp1MsSi\nXLlyeHp6otPpCA0NpWPHjoZrssDAQBISEpg/fz5NmzZ9YhyEadLCRhS6BxXbozLHD0RGRpKQkMAb\nb7xhtDwoKAitVsupU6cMyx6cAAMoFApcXV1JTU0FcjLppUqVMiRrICchs2vXLsqXL8/Ro0epXLky\nrq6uhqx2uXLlqFChAocOHTJs4+bmZkjWQE7SJy0t7bHvQa1WGy4GALy9vVm7di0WFhZcuHCB7du3\ns2TJEoA8dVdKSUnh/PnzJmOiUCgMlS5ArVq1jNZ5OCZCCPFfcPToUapUqUKJEiUM9XPFihUpV66c\n0QxSlSpVMqqfnZycqF69uuGE3MHBAYCkpCTDOrVq1TKqn1977TV0Oh2///47v//+O1qtlsDAQMNx\ns7OzCQgIyDVzlZubm+F3KysrVq5cSb169bh27Rp79uxh8eLFxMfHP5cuqQ8f68qVK8THxxuVMSsr\ni8DAQM6ePWv0XoUQL6e8nnND3s67jx8/jkKhMLnO+fPnSU5OzlO5Hj5vf5CMf3BOfeTIEV599VVD\n/Q3kOt6/qVQq6tWrZ7g2OHjwoCHR4Orqipubm6HufpC4eWDs2LH07NmT2NhYDh8+zNq1azlx4sRj\n6+yjR4/SqFEj9Hq9Ud2r1Wo5ceKEYb2H6+zHCQ4OZsOGDUY/w4YNM7nu8ePHqVmzpiFZA2Bra0tg\nYKDhOqBRo0aGLrxHjx6lVq1a1K1b11C2AwcO8Morr6BW/9N24+GyajQaVqxYgZ+fn+G7bMmSJcTF\nxT0yLlFRUcTGxtK4ceNc30nnz583tKAS+SctbEShe5Dlf7hp+r/duXPHMEbBv0dlf5C1fvhLwcLC\nwmgdpVJp6G+bmJj42IHUEhISOHv2rGFcgYc9nOTRaDSPPMajODg45BqBPjw8nGXLlpGWlkb58uXx\n9fUFeOK+IOdiQ6/XG2LwgLm5OTY2NkYxeZryCiFEcZKQkMCZM2dM1s8xMTGG362trXO9/u868N9K\nlixp9PzB98T9+/cNFzdvvvmmyW0fHuvm3/Xx9u3bmTFjBrdv36ZkyZLUqVMHjUbzXOrfh4/14OS3\nX79+JteNjY01tOQUQryc8nrOXapUqTyddycmJmJra4u5ubnJdR4eC+dxHj5vVypz2g88qHfv3buX\n67z93/WsKQ0aNGDTpk3odDqOHDli1MW1YcOGHD9+nJs3bxIdHW3UPeny5cuMGTOG33//HWtra2rU\nqIGFhcVj6+yEhARWrVrFqlWrcr0WHR2dr3JDzt/J29s7T+vev3/f5H6dnJz4448/AAxj6sTHx3P0\n6FHq1atHhQoVmDdvHpCTsBkwYECu7R+2c+dOpk2bxq1btyhRogR16tR5bFwejP05cOBAk6/HxsYa\nbp6I/JGEjSh0Tk5OVK1alQMHDpicRvDmzZs0bdrUMJbLw4NmPfzc3t4+T8eztbU1GkD4gT179uDt\n7Y2dnR0+Pj6MGTMm1zp5PUZeff/99yxcuJBp06bRvHlzrK2tSU9PZ8OGDXna3sbGBoVCQVxcnNHy\njIwMkpKSnnt5hRCiKNna2lKvXj1D8/yHPeuJ38MDFwOGetXR0dFwB3HlypXY2Njk2vbhu5IPu3Ll\nCh9//DFdu3alV69ehguft99++7FlUSgUue6AP+nC50GLolmzZpnsWlWmTJnHbi+EePHl55z7wQ3E\nx513379/n6SkJDIzM42SNvk9N38cFxeXXOe58fHxT9zO19eXkJAQTpw4QVJSklFSpmHDhmzatIkT\nJ07g4uKCu7s7kJMk6t+/P6VKlWLbtm24ubmhUCiYPXs2165de+SxbG1tad26NW+99Vau1wq67rWz\nszNKCj0QFxdniH/NmjWxt7fnyJEjHD9+nLfffpuKFSsSHR3Nvn37uHXr1iPHNQL466+/GDJkCF26\ndKFPnz6GGxymxtZ8uFwAM2bMoHLlyrlef7hFkMgf6RIlisT7779vGCTy38LCwjAzM6NZs2Y4ODjw\n008/Gb2+bds2VCpVri4/j1K7dm1u377NhQsXDMsuXrxI3759iYyMxMfHh6tXr+Lm5oa3tzfe3t5U\nrlyZsLAwzpw5k+f39OAOweOcPHmSSpUq0a5dO8Md4f379wP/3Fn4d4uch9nY2ODl5WUyJgA+Pj55\nLq8QQhR3Pj4+XLt2DXd3d0P97OHhQWhoqOFO4tM6ffq0UTJ/165dmJmZ4ePjQ61atVCpVCQmJhqO\n6+3tzd69e1m/fv0j6/s///wTrVZLcHCwIVkTHR3NpUuXDHclTW1rZWXFnTt3DM8zMjL4888/H1t+\nDw8PbG1tiYuLMyrjuXPnWLp0qVF3AiHEyyuv59zu7u5PPO+uW7cuer3e5DrVqlV7YsvGvKhXr16u\nAYx37979xO1q1KiBRqPhq6++onLlykYtRvz8/EhLS2Pr1q1G3aHi4+O5du0anTt3xt3d3ZA8P3jw\noFES/d/1to+PD3/99ZdR3atWq5k3b16uZNPzVrduXf744w+jMS6Tk5PZt2+fYVwZpVKJv78/3333\nHbGxsdStW5eKFStSsmRJwsLCqFy5sqErmilnz541fJc9SNbExsZy8eLFR8blwecnNjbWKC4XLlxg\nyZIlebpOEqZJCxtRJNq3b8+uXbvo06cP3bp1w8/Pj5SUFL777jt++eUXpk6diqurKwMGDGDGjBlY\nW1vTuHFjfv/9dxYtWkTXrl3zfHe1adOmVKlShUGDBjFkyBAsLCyYN28edevWpW7dulSpUoVVq1bR\nu3dvevXqhbm5OcuXL+f06dN8+umneX5PdnZ2XLt2jYMHDz4ycVKzZk3WrVvHkiVLqFOnDmfPniU8\nPByFQmHou/ugCfuuXbtydfUCGDRoEAMHDmTkyJG0bt2aqKgo5s2bx+uvv27UhUsIIf7rOnbsyOrV\nqw31s5mZGV988QVnz55l7Nixz7TvtLQ0+vfvT79+/bh27Rrz5s2ja9eu2NnZYWdnR8eOHZkwYQLR\n0dFUqVKFQ4cOERERYRjg15Tq1aujVCqZOXMmb731Fnfu3CEiIgKtVmsYQ0ypVGJjY8OhQ4fw9vam\natWqBAQEsG7dOnx9fSldujQrVqx4Yhcqc3Nz+vbtS2hoKJmZmfj4+HDu3DlCQkJo3759ri4LQoiX\nU17PuYEnnnc7ODjQrFkzJk6cSEJCAm5ubmzZsoXDhw8THh7+XMrbt29f2rVrx7Bhw2jXrh1//vkn\nq1evBh5/c1SlUlG3bl127NiRa4INR0dHvLy8+O2335g4caJhubOzM6VLl2bZsmVYW1uj0+n4+uuv\nOXv2rFHyyc7OjgsXLnD48GF8fX3p168f7733HqNGjSIoKIjExETmzp2LpaVlnseteVrt27dn9erV\n9OrVi48++ghzc3OWLFmCTqeje/fuhvUCAgIYM2YM1atXN7QUrV+/Ptu2baNnz56PPUbVqlVRqVTM\nnDmT9u3bEx0dTUREBBkZGUbjd9rZ2XHkyBF8fHyoWrUqwcHBzJs3D61WS7169Th//jxz586lTZs2\n8p30DCRhI4qEUqkkPDycL7/8ku+//561a9eiVqvx8vJi+fLlNGrUCIDu3btjYWHBihUrWLt2LaVL\nl2bo0KFPrGge9iABM2PGDMaNG4daraZx48aMGjUKpVKJnZ0da9as4bPPPjMkaGrWrMmqVasMTSbz\nolOnTuzcuZO+ffua7NMKOZVsZGQkq1atYtGiRVSoUIHRo0ezefNmw0BplStXplWrVoSGhnLnzp1c\n77VZs2aEh4cTHh7Ojz/+iLOzM++//z4ffvhhnssqhBD/BQ4ODnz11VfMnDmTESNGoFAo8Pb2ZuXK\nlVSsWPGZ9l2nTh38/Pz4+OOPsbS0pHfv3kZ9+seNG0eJEiVYsWIFMTExhulvu3Xr9sh9enp6Mm3a\nNBYuXMjWrVtxdXUlKCgIvV7Pxo0b0el0KJVKBg4cSFhYGFeuXOH7779n0KBBxMfHM336dDQaDe++\n+y5eXl6cPXv2se+hb9++WFlZsWbNGhYsWICLiwt9+vTJNTaBEOLllddzbsjbefecOXOYN28eS5Ys\n4f79+1SpUoVFixbleYaoJ6lcuTILFixg9uzZ9O/fHy8vL4YNG8aMGTOwtLR87La+vr789ttvJqfQ\n9vf35/z580avKRQKQkNDmTJlCoMHD8bOzo4GDRoQEhLC0KFDuXDhAl5eXnTv3p0hQ4bQt29ftm/f\nTp06dVi2bBnz5s1j4MCBWFtbExAQwCeffIKZmdlzicOj2NrasmbNGmbNmsXYsWNRKBTUr1+fdevW\nGWa7hZyEDeS0WHqgQYMGbNu27bHdoSCntcz06dNZsGAB27dvx9XVlTfeeIPmzZuzbt06srOzUalU\n9O/fn/nz53P58mV++OEHevbsiUajMVznuLi40KNHj0eOayPyRqGXUUiFEEII8RIZPnw4N2/e5Ouv\nvy7qogghhHjI/v37sbOzMxqEd926dcyZM4fDhw9Ld0/x0pEWNkIIIYQQQgghitzJkydZs2YNI0aM\noEKFCkRFRRESEkKnTp0kWSNeSpKwEUIIIYQQQghR5IKDg0lJSSE8PJyYmBhcXV354IMP6Nu3b1EX\nTYgiUSRdok6fPs2AAQPYt2+fyde3bNnC3LlziYuLw8/Pj2nTphlmWxBCCCGEEEIIIYR40RXq/Fp6\nvZ4NGzbQs2dPtFqtyXXOnz/PhAkTCAkJ4dChQ5QoUYJRo0YVZjGFEEIIIYQQQgghilShJmwiIiJY\nvXo1/fr1e+Q6P/zwA6+99hq1a9dGo9EwfPhwfvvtN2JjYwuxpEIIIYQQQgghhBBFp1DHsGnfvj39\n+vXjyJEjj1wnMjISHx8fw3NHR0fs7e2JiorKU7eo2NhklPlOQylwdLTi3r1UQCbN+ofExTSJS24S\nE9OePi5OTjYFU6QXWExMUr7WVygUODtbExeXgkyY+A+Ji2kSF9MkLrk9S0xKlrQtoFK9uPJb94N8\nbk2RmJgmcTFN4mJaQdT/hZqwcXFxeeI6aWlpaDQao2WWlpakpaXl6RjOztZPPYK4k5P1U233opO4\nmCZxyU1iYprEpXhSKnO+WJVKyM4u6tIUHxIX0yQupklccpOYFH/yN8pNYmKaxMU0iYtpBRGXYjdL\nlEajIT093WhZWloaVlZWedo+Li4l3y1slEoFDg7WJCSkoNNJhvABiYtpEpfcJCamPUtcpIWNEEII\nIYQQL7dil7Dx8PAgKirK8Dw+Pp7ExEQ8PDzytL1er3/qbJZOpyc7Wy42/03iYprEJTeJiWkSFyGE\nEEIIIUR+Feqgw3nRunVrduzYwbFjx8jIyCAkJITGjRvj6OhY1EUTQgghhBBCCCGEKBTFImEzfvx4\nxo8fD0C1atWYMmUKY8aMwd/fn+joaGbMmFHEJRRCCJFXW7dupWXLlvj4+NCqVSt27twJwJkzZ6hW\nrRo+Pj6Gn4iICCCndeScOXNo2LAhDRo0YOrUqWRLp2ghhBBCCPESK5IuUX5+fhw+fNjwfPLkyUav\nBwUFERQUVNjFEkII8YyioqIYPXo0y5cvp27duhw4cIC+ffuyd+9ezp07R+PGjVm8eHGu7dauXcvu\n3bvZvHkzCoWC4OBgli9fTp8+fYrgXQghhBBCCFH0it0YNkIIUdj0ej1Z2Tq0WToys3IeH/7JzMo2\n/J6VrSMrW0+27sGjnuzsnOXZOr3Razq9HvdyjgTWfPIMeS8KNzc39u/fj7W1NVlZWcTGxmJtbY25\nuTlnz56latWqJrfbtGkT3bt3N8wmGBwczPz58ws0YROTkMbWw9d5pYYLtpbmBXYcIYQQQgghnoYk\nbIQQ/yl6vZ60jGxS0rWkpmeRlpFFemY26dqcx4zM7JznmVkP/f73c202GVodmdpstNk6tFpdzmOW\nrsDK++uJm3iWboirY95munsRWFtbc/36dVq0aIFOp2PixInY2Nhw7tw5zM3Nadq0KTqdjpYtWzJ0\n6FDMzc2JjIzE09PTsA83NzeioqLQ6/UoFIo8HffBNIp5tfXQVX45fpOb0Un0ebN6ft/mC0upVBg9\nihwSF9MkLrlJTIQQQjwvkrARQhQZnV5PSpqW+ymZ3E998JhJUmomKWlZpKRrSUnPIvXvx5Q0LakZ\nWeiLYMIlhQLM1SrUKgVqlRKVSoFamfOoUipRqxSGZTm/K1GrlFSu4EgpZyv0BZcTKpZKly7NqVOn\nOHbsGAMGDKBixYo4Ojri5+fHO++8Q1xcHB999BGhoaEMHz6ctLQ0NBqNYXtLS0t0Oh2ZmZlYWFjk\n6ZjOztZ5Tu4AuJV1gOM3OX0lDnsHa1RycWXEwcG6qItQLElcTJO45CYxEUII8awkYSOEKBAZmdnE\nJ6UTfz+D+PvpxCflPN5LyiAhOScpk5SqRfccsi8qpQKNuQqNuQoLc3XOo5nKsExjrsbCXIXGTIWF\nuQpztRK1WomZWom5WvX349/LVErMzVSYqZSYmT14rkSVn6YbD8qlUuDkZEN8fDLZvFzTeqvVOV8v\n/v7+NG/enF27dhkGGAawsrIiODiYkJAQhg8fjkajISMjw/B6WloaarU6z8kagLi4lHy1sKlS1g6A\n+ymZnPjzFh5l7fO+8QtMqVTg4GBNQkIKOt3L9bl9HImLaRKX3J4lJk5ONgVUKiGEEP9FkrARQjwV\nnU5P3P2M5criAAAgAElEQVR0ou+lEZuYRmJaFtduJxJ9L517SemkpGfle5+WFirsrMyxtTLHxtIM\na40aK82DRzXW/1pmrTHD0kKNmbpYTHgngD179rBixQpWrlxpWKbVatHr9cyaNYuBAwdiY5NzQZKR\nkWFIyHh4eBAVFUXt2rWBnMGL3d3d83VsvV5PfiaWcnW0xNXJirvxqZy8GEulUnb5Ot6LTqfTk50t\nF+D/JnEx7VnjotfruXPnNqVLl3mOpSpa8lkRQgjxrCRhI4R4LG2WjjvxqdyMSeZmbAo3Y1K4HZ9K\nbEIa2Xm8c2hnZYajnQYnWwucbDU42JrnJGaszbG3/vt3KzPMzVQF/G5EQatevTp//PEH33//PW3a\ntOG3335jz549rFu3jn79+qHX6/n444+5desWERERdOrUCYA2bdqwbNkyGjZsiFqtZvHixbRt27ZA\ny6pQKGhQzZUt+6M4HRlHu8b5SxAJ8SIKCKiPhYUFyr+bq+n1ekqUKMn773endeu3Cuy4CxbMB+DD\nD4cU2DGEEEKI/xpJ2AghDJLTtPx15z5Rt5O4fjeJm7Ep3I1Pe2K3JUdbC8q62OBkY0EJew1OdjmJ\nGSc7CxxtLTBTSyLmZVGyZEkiIiKYPn06kydPplKlSixYsABPT08iIiKYOnUqDRs2RKPR8M4779C9\ne3cAunTpQmxsLB06dECr1fLmm2/So0ePAi9vvb8TNlfvJJGYnIG9Td67YAnxolq6dBXu7jmDgGdn\nZ7Nr1w6mTp1AzZq1qVTJrUCOmZiYgL29Q4HsWwghhPivkoSNEC+pTG02f91JIur2faJu3+ev20lE\nJ6Q9cn1zMyVlS1hT5u8fV0crXBwtKelgiZVG/c9YLdL8+6VXv359Nm7cmGu5p6enUVeph6lUKoYO\nHcrQoUMLuHTGvD1LYK5WkpmlY/uR63Rq6vnkjYR4DrKydcTfTy+UYznZaVCrnq7rqEqlonnzloSG\nziEq6gqVKrlx/34i8+d/zuHDh9BoNLz3Xhfat+8M5CRe5s37nDNnTnHvXjzlypXn448/pVatOgB8\n990GvvpqDYmJCVSrVp0RI8bw22+72bFjGwqFgjt3bjN16iyOHj1ERMQCrl+/RpkyZQkOHoC/fwCQ\n0wqoXbuO/PzzT7z3Xje6di345K4QQghRFCRhI8RLIi0jiys3E7lwPYEL1xOIunX/kV2aXJ2sqFTK\nlnIlrSlbwoYyJa0pYa9BmY8ZeIT4L7AwU9HctzxbDlxl+9Fr1KtaEo8yMviwKFhZ2TpGLzlEbGLh\nJGxK2GuY3rfhUyVttFot33//LRkZGdSo4Q3AlCnjsbOzZ/36zSQlJTBy5DA0GhtatnyThQtDAVi7\ndj1KpYr58+cQERHOwoVfcOjQAZYsWcicOaF4eVVj8eIFTJkynoiI5Vy5chl7ewc+/HAIkZFXGDny\nYyZMmEKjRo05evQQ48Z9yuLFK/HwyEmqZmZm8MMPO9BqM59foIQQQohiRhI2QrygdDo9UXfu80dk\nPH9ExhF1O8lk1yYnOwvcStlRqbQtbqXtqFTKFiuNWRGUWIii0TbQjeMXYrgdl8rSH84ytGNtXJ2s\nirpYQhSZfv16oVQqyMzUAnr8/PyZN28RLi6uxMXFcujQAbZs+RlLS0tsbKzo1asXX375FS1bvknf\nvgPQaDSoVGpu376Fra0tMTExAOzcuZ2WLVtRvXpNAHr06MPVq1G5jr9r1w7q129AkyZNAfD3D6BR\no8bs2LGN/v0HAdCsWXPMzMwwM5PvKyGEEC8uSdgI8QJJSddy6nIsZyLj+TMqnuQ0ba51ypa0xqu8\nA14VHKlczh4HGbNDvOTM1Sp6tqrGjDUniL6XxsSVR3k70J3A2qXRmMvXpHj+1Col0/s2LLZdoiIi\nluHu7smtWzcZPfoT7O0dqFEjJ8ly9+4d9Ho977zzzwDEer0eW9ucWdZiY2OZP/9z/voriooVK2Jr\na49erwMgPj4eD4/Khu0sLS2pWrV6ruPfuxdPqVKljZaVKlWamJjof96TU4k8vx8hhBDiv0rORIX4\nj7ufmsnJizEcvxDDuav3cnVzKmGvwdvdmeqVnPCq4ICNpdyNFOLfPMrYM/Sd2iz94Sz3UzL5etcl\nvt8Xibe7M7U8nKnp5oydtXlRF1O8QNQqJS6OxbslV5kyZZk5cw4ffNCF0qXL0L17L5ydS6BSqdi8\neQfm5uaoVApUqmxu3sxJpkyYMJq2bd9mwYKlKBQKtm3bQmTkZSBnUPLY2H+SLikpySxfvpR+/T40\nOq6rayn+/PMPo2W3b9+iZEkXw3PpoSuEEOJlIAkbIf6D0jKyOHY+mkNn73L+2j0e7umkVimoWsER\nb3dnvD2ccXW0RCFntkI8UY1KTkzq0YANe65w+Oxd0jKyOXIumiPnolEA5Vxs8Chrj0cZO9zL2FHK\nyUr+t8QLr1Sp0gwePIzPPptGo0aN8fSsTO3aPixaFEa/fgPRajOZOHE0Dg5OjB07mdTUFCwtNSgU\nCv76K4qvvlpNVlYWAM2bt2T8+E9p0SIId3dPVq9ewdmzfxi6NqWkpADw2mvNWbNmBXv37qZRo0CO\nHDnEvn17WbBgSVGGQgghhCh0krAR4j9Cp9dz4eo99p25w/GL0WRqdYbXzNVKvN2dqedVktqeJbC0\nkH9tIZ6GvY0FvVpVp8P/PDly7i5nIuM4fzWBrGwd16OTuR6dzO6TNwGw1qhxK2NHRVdbKrjaUsHV\nhpIOljI4t3jhBAW9yc8//8SMGZNZsmQlEydOY/78OXTo0Ibs7Gz+978mDB78MQAjRowmNDSEhQvD\nKFmyJK1atWHJkoUkJiZQr14D+vcfzPjxo7l3Lw5v79pMnDgNgFdfbcb48aO4e/c2ISHhzJjxOYsW\nhTFlynhKlSrFhAlTqVatRlGGQQghhCh0Cr3exCik/2ExMUn53kalUsiUxCZIXEwr7LjcT8lkz+83\n2XvqFnH3M/4ph1JBbc8SNKzuire7MxbmqgIvy6PIZ8W0Z4lLyZK2BVSqF1d+6/+8/n0ytNlcuHaP\nSzcSuXIzkajbSWRos02uqzFXUcHF5u8ETk4Sp0wJ66eeUrkoyP+zaRIX0yQuuUndX7jk3P/5kJiY\nJnExTeJiWkHU/3IbXohi6uqdJHYeu87hc3fJeugfvqKrLY28S+FX3RVbKxlTQ4iCZmGmopZHCWp5\n5AxyqtPpuRGTzJVb97l65z5X7yZzMyaZrGw96ZnZXLyRyMUbiYbt1SoFZUpY5yRw/k7mlHexkZZw\nQoj/lNOnTzNgwAD27dsHQGZmJrNmzWLLli0ANGvWjAkTJmBubo5eryckJIT169eTnZ1N27ZtGTVq\nFCpV7ptLiYmJjB49mkOHDmFra8vAgQPp2LFjob43IYQoruRsUYhiRKfXc/JiDDuOXufSQxd8GnMV\nAd6lCaxdhvIuNkVYQiGEUqkwtKCBsgBkZeu4HZfKtbtJXL2bxLW7yVyPTiItI5usbD3X7iZz7W6y\n0X5cHC0fao2T8yiztgkhihu9Xs+3337LzJkzjRIuISEhXLp0ie3btwMQHBzM8uXL6devH2vXrmX3\n7t1s3rwZhUJheK1Pnz659j9u3DisrKw4cOAAFy5coE+fPlSuXJk6deoU2nsUQojiShI2QhQDOr2e\nY+ej+eHAX9yMSTEsd3W05LV65WjkXVruxgtRjKlVSsq72FDexYZG3jnTEev0emIT0nKSNdFJfydt\nkkhIzgQg+l4a0ffSOHYhxrAfO2tzKpWyxbOsPVXKO+BW2hYzddF1dxRCiIiICLZt20a/fv1YunQp\nAFqtlnXr1rF+/XocHBwACA0NNQwwvWnTJrp3746LS87MXsHBwcyfPz9XwiYlJYWdO3eyfft2LCws\nqFWrFq1bt+b777+XhI0QQiAJGyGKlE6n5+jfiZpbsf8kaqpXcqR5g/LUdHeWAUyF+I9SKhS4OFrh\n4mhF/ar/TEd8PyWTa9FJXL+bzNW7SVyPTuZOXCr6v187fSWO01figJzuVJVK2VG5vD013ZypXM7+\nPzUejhDiv699+/b069ePI0eOGJZdvXqV7OxsTp06xYABA0hLS6N169YMGzYMgMjISDw9PQ3ru7m5\nERUVhV6vN5pd7+rVq6jVasqXL2+07o4dO/JcPoVCgTKf1aJSqTB6FBKTR5G4mCZxMa0g4iIJGyGK\ngF6v59TlODbsuWKUqKnp7kSbRm54lrUvwtIJIQqSnbU5Nd2cqenmbFiWkZnNjZicFjhXbt3n0o0E\nYhLSycrWc/lmIpdvJrLt0DU05iqqV3LCp3IJ6lYpKS3vhBAF7kErmYclJCSg1Wr59ddf2bBhAykp\nKQQHB2Nra2tI4Gg0GsP6lpaW6HQ6MjMzsbD4p+tnamqq0XoAGo2G9PT0PJfP2dnaKAmUHw4O1k+1\n3YtMYmKaxMU0iYtpzzMucqYnRCG7eieJdb9c4vy1BMOyWh7OtGnkhnsZuyIsmRCiqFiYq/Aoa49H\nWXterZuz7F5SBpdvJnLpegJnr97jVmwK6ZnZnLgYw4mLMazefoHaniXwr+5KLU9nVPm9xSyEEE/J\n3NwcnU7HkCFDsLOzw87Ojh49erBmzRoGDBiARqMhI+OfmS3T0tJQq9VGyRrISeQ8vB5Aeno6VlZW\neS5LXFzKU7WwcXCwJiEhBZ1OZrgBicmjSFxMk7iY9ixxcXIyPU6pJGyEKCT3UzPZ8OsV9p25bVjm\nVd6Bjq96SqJGCJGLo60FDaq60ODv7lRxiemciYzj98ux/BkVjzZLx7Hz0Rw7H42znQWv1StP49ql\nsdKYFXHJhRAvukqVKqFUKsnMzDQsy87ONvzu4eFBVFQUtWvXBiAqKgp3d/dc+6lYsSJarZZbt25R\npkwZw7oPd6d6Er1ez0OHzhedTi9TEv+LxMQ0iYtpEhfTnmdcJGEjRAHT6fTsPXWLb/dcISU9ZzC+\nUk5WdHrVk9qezk/djFcI8XJxttfwP5+y/M+nLMlpWo5fiObQn3e5cD2BuPsZ/N+vl9m0L4r/+ZSh\nlX8lbCwlcSOEKBh2dnY0a9aMkJAQQkJCSEtLY9WqVbz55psAtGnThmXLltGwYUPUajWLFy+mbdu2\nufZjY2PDa6+9xpw5c5g6dSqXLl1iy5YtLFmypLDfkhBCFEuSsBGiAN2ISWbF1vNE3b4P5EzP/Vag\nO03rlpWBQ4UQT83G0owmdcrSpE5ZbsamsOvYdQ78cYcMbTbbj1xn3+nbvPlKJV6tWw4ztdQ1QhS2\n69evY23tWNTFKFAzZsxg1qxZBAUFodVqeeutt+jZsycAXbp0ITY2lg4dOqDVannzzTfp0aMHALdu\n3aJVq1b8+OOPlClThilTpjBhwgSaNGmClZUVn3zyiaFljhBCvOwUer3+hWrDFBOTlO9tVCoFTk42\nxMcnS5Ouh0hcTMtLXLKydWw7fI3N+6LI/rv/om81F95pWhlHWwuT2/yXyWfFtGeJS8mStgVUqhdX\nfuv/F+1zm5ymZeex6/x05BqZWh0ALg6WdH3DixqVnPK8nxctLs/LyxSXQ4cO8PXXa7h06SJ6vZ5q\n1arTt+8Aqlatnmvd/MRl2bLFREVdYerUz1i9ejlXr0YxbtyUAnkPqampNG/emPXrN1O6dJmn2seJ\nE8cYN24kP/64K1/bXb58gU8+GcLmzT9J3V8I5Nz/+ZCYmCZxMU3iYlpBnPtLCxshnrObMcl8seUc\nV+/mnEA421nQvWVVoxlhhBDiebOxNOOtQHea1CnLpn2R/Hb6NtEJacz55nf+V6cM77xWGQszVVEX\nUxRzmzd/xxdfRDBy5Fj8/PzJzs5m48b1DB7cn4iI5bi7ezyX43Tr1vO57Kc4SklJRqvVFnUxhBBC\nvAAkYSPEc6LX69n9+y2+2XUJbVbO3e3/+ZSl4/88ZOpdIUShcbS14IOW1XitXnlWbD3HX3eS2P37\nLS7dSKT/WzUpU0Km4CwOsnRZ3EtPLJRjOWrsUSuf/D2Unp5OePg8JkyYSqNGgQCo1Wo6d36fhIR7\nXL0ahbu7B/HxcYSGhnD06CEsLDS8+WZrunXrjUplRmpqKosWhbFnzy8AvPJKAB9+OBQbG+PZLx5u\nbTNt2kSsra25ePECly5doEKFSowYMQYvr6ro9XpWrFjKxo3rMTc3p2PHzkREhPHNN9/lq+XMsWNH\nWLYsguvXr5GZqcXXtyFjx05Co9GQlJTEvHmfsX//b6jVZgQFtaZ//8FG26ekJPPRRwOoXLkKI0aM\nITMzg0WLwti9+xf0ej2vv/4GwcEDSU5OYujQwWRmZtC0aQAbNvyAvb1DnssphBBCPEyuIoV4DpLT\ntKzadp7jF2OAnAumXq2qUT0f3RCEEOJ5Ku9iw5hu9fjx4FU27YviZmwKU1cfo1/bGtTyKFHUxXup\nZemymHxoNnHp9wrleM4aR8Y3/OSJSZszZ34nOzsLPz//XK/17z/I8Pvo0Z9QunQZ1q/fTFpaKmPH\njiAzM4vg4EF89tk07t2LZ9Wqb1CpVEydOp7Zs6cxadKMxx77p5+2Eha2mAoVKjB9+iQWLw4nJCSc\nH3/czNatP7Bo0TKcnJyYMmW80WxEeZGWlsaYMZ8wbtxkAgKaEB19lwEDerNz50+0bv0Wn38+nbS0\ndP7v/zaTnZ3FoEH9KFu2POXLVwByElkjRgzF07MyI0aMQaFQEB4+nxs3rrFq1dfodHrGjRvJ6tXL\n6dUrmLlzQxkzZiTbtu2SrgJCCCGeSaGORHj27Fk6dOhAnTp1aNu2Lb///rvJ9VatWkXTpk2pX78+\ngwYNIjY2tjCLKUS+RN2+z8QVRwzJmrpVSjKpp68ka4QQRU6lVNKmkRsjOvtgZ21OemY28zec5teT\nN4u6aKIYSkhIwNbWDrX60Ymdmzdv8Mcfp/noo+FYWVnj4uLCRx99xI8/biEjI53du3fRv/8gHB0d\nsbOz48MPh/DLLzvJyEh/7LEDAhpTuXIVLCw0NG3anOvXrwOwY8c2OnXqTPnyFbC2tmHAgMGP3Y8p\n5ubmLF++loCAJiQnJxMbG4O9vQMxMTFkZmayd+9u+vTpj52dHY6OTsyaFYK/fyMAsrN1jB49HIVC\nwciRY1EoFOj1erZu3Uz//oOwt3fA0dGRXr2C2bz5u3yXTQghhHicQmthk5GRQb9+/ejXrx8dO3Zk\n06ZN9O/fn507d2Jt/U/z7K1bt7JgwQIWL15MzZo1WbhwIf3792f9+vWFVVQh8uy3U7dYs+MiWdk6\nzNRK3n2tMv+rU0am6hZCFCteFRwZ160+od+e5np0Mmu2XyA1XUsr/0pFXbSXklqpZnzDT4pdlyhn\n5xLcv3+frKysXEmb+/fvY2Vlxb178VhaWuLg8E83nzJlyhAfH0dCQgJZWVmUKvVPVyVX19Lo9Xpi\nYmIee2wHh39mVFKr1ej1OV2LY2NjcHFxNdpffqlUKvbv38u6dV8B4OlZmfT0NHQ6HUlJ99Fqtbi4\nuBjWL1u2HAA3blwnOTkJnU7H+fNnuXHjOuXLVyAh4R4ZGRkMGhRs+L7X6/VotVlkZGTku3xCCCHE\noxRawubQoUMolUq6dOkCQIcOHVi1ahV79uwhKCjIsN6OHTvo1KkTPj4+AAwaNIiVK1dy4cIFvLy8\nnngchUKBMp/thpRKhdGjyCFxMU2pVKDN0rHqp/P8cjznLnVJBw2DO9SiguvLObuDfFZMk7iI4sTZ\nXsOn79Ul7NvTnL+WwLd7IgEkaVNE1Eo1Ja2K12D0NWp4Y2am5tCh/QQENDF6bebMKVhZWREcPJC0\ntDQSExMMY7PcuHEDOzt7SpZ0wdzcnDt3bhsSOrdv30KpVBolZPLDxcWVu3fvGJ7HxEQ/ct1p0ybS\ntWsPKlSoSFZWFgAWFhacOXOK5cuXsnTpKkM3p8GD+wE5iSIzMzNiYmIM7+fw4YMkJiZSokQJbGxs\nCQkJ5/PPZzBr1lTCwhZjZ2ePmZkZy5evNSR30tLSiI+Pw8LixZsJUgghRNEptIRNVFQUHh7GMwu4\nubkRGRlptEyn06HRaAzPFQoFCoWCq1ev5ilh4+xs/dStGxwcZCBGUyQuxpJSMxm3+AB/RsYBUNfL\nheHv18PWyryIS1b05LNimsRFFBeWFmqGdKzNwu//4PSVOL7dE4mFmYpm9csXddFEMWBhYUFw8Id8\n9tl0lEoVvr4NyczMYN26rzh27AgREcsoWdKFevV8CQ2dw8cfjyItLYXQ0FBatHgDpVJJ8+YtiYgI\nY+LE6SiVShYunI+/f6Ncgw7nVVDQmyxduohGjRrj7FyCJUsWPnLdyMgr/PzzT3zwQW9+/nkbzs4l\ncHR04uLFC6hUSiwsLMjOzmbHjm2cOnWSGjW8UalUNG36OsuWRTB27CTS09NZsGAeHTt2BkClUqJU\nKunXbxDvvdeBTZs28tZb7Xn99TeIiAhnxIgxqNVqZs+ezp07t1m48AvMzMzJzMxEq9WizEPLJiGE\nEOJRCu1bJDU1FUtLS6NlGo2G9HTjPs1NmzYlJCSEZs2a4e7uzpIlS0hPT89zE9O4uJSnamHj4GBN\nQkIKOp0MDveAxCW3u/dSCfnmFHfiUwFo/UpF2jfxQJueSXx6ZhGXrujIZ8W0Z4mLk9PTXdwI8STm\nZioGtqvJvPWnOXf1Hl/tvISDjQX1q7o8eWPxwnv77Y7Y2NiyYsUSJk8eh1KppHr1GoSFLcbd3ROA\nCROmMH/+53Tq1AaAtm3b0qNHMACDBw9j4cJQund/h4yMTAIDmzB48MdPXZ7XX3+DqKhI+vTpjqWl\nJS1a5LTKNjMzy7Xu8OGfMnv2dL7+eg2lSpVm/PgpKBQKfH0b8uqrzejW7V1UKiVeXtVp2bI1V6/+\nBcDQoSMIDZ3Du+++jUKhoG3bt3nzzbc4ceKYYd92dnYMGjSUOXNm0qhRIEOGDGfhwjC6du1Eeno6\ntWvXYfLknIGVPT0r4+npyRtvNGX58rWUKycJUSGEEE9HodfrC+XqasWKFezfv58vvvjCsGzw4MFU\nrVqVAQMGGK27ZMkSvvnmG7RaLR06dGDnzp0MHz6cJk2a/Hu3ucTEJOW7bCqVAicnG+Ljk2U0/4dI\nXIxdvplI6IbTJKdpUasU9GxVjYbVSxV1sYoF+ayY9ixxKVny5exe9yzyW/+/7J/b9MwsZn99kqjb\nSZiplYzsUhf3MnYvfVweReJiWkHH5dKlizg6OlGiRM7MZn/9FUW3bu/w8897sbDQPGHroiF1f+GS\nc//nQ2JimsTFNImLaQVR/xfaLFHu7u5ERUUZLYuKisLT09NoWXR0NEFBQfzyyy/89ttv9OjRg6tX\nr1KtWrXCKqoQuZy6HMvsr0+SnKbFSqNmct9XaOSd/4EPhXgZbN26lZYtW+Lj40OrVq3YuXOn0es6\nnY6uXbsya9YswzK9Xs+cOXNo2LAhDRo0YOrUqfmeulfkj8ZczeD2tXC2s0CbpSN842kSU17eloKi\neDp06ABTpownNTWVjIx01q5dRZ06dYttskYIIYR4ngotYePv709mZiZr1qxBq9WyYcMGYmNjCQgI\nMFrvwIEDBAcHEx8fT3JyMlOnTqVRo0ZGo/cLUZiOnLtL+MYzaLN0ONtpGNe9Pt6eJYq6WEIUS1FR\nUYwePZpp06Zx8uRJxowZw5AhQ4iPjzess3z5co4dO2a03dq1a9m9ezebN29m69atnDhxguXLlxd2\n8V869jYWfNShNuZmShKSM1m86Q+ydbqiLpYQBu+804XSpUvTqVMb2rRpQVLSfcaNm1zUxRJCCCEK\nRaGNYWNubs7SpUuZOHEiISEhVKxYkUWLFmFlZcX48eMBmDx5Mm3btuXChQsEBQWh0+lo0qQJn332\nWWEVUwgje0/dYtW28+iB0s5WDH/XhxIOcldPiEdxc3Nj//79WFtbk5WVRWxsLNbW1pib5wzKff78\neTZu3Mjrr79utN2mTZvo3r27ITkfHBzM/Pnz6dOnT56Pnd9ZAmUWrxwVS9vSI6gaizf9yflrCWze\n9xe93qr10sfl3+TzYlpBx8XS0oIxY8YXyL4LinxWhBBCPC+FOnR91apV+eabb3Itnzz5nzslCoWC\nkSNHMnLkyMIsmhC5/HzsOl/vvARARVdbhr1TW2aCEiIPrK2tuX79Oi1atECn0zFx4kRsbGzIzMxk\n5MiRTJkyhfXr1xttExkZadRF1s3NjaioKPR6fZ5n/nvaWQJlFi9o3diTazEpbDvwF5v3RdGoTjmq\nVnIq6mIVS/J5MU3ikpvERAghxLOSuQaFMOGXEzcMyZrK5ez5qENtrDTy7yJEXpUuXZpTp05x7Ngx\nBgwYQMWKFdm9ezcBAQHUq1cvV8ImLS0Njeaf1muWlpbodDoyMzOxsLDI0zHzO0ugzG5m7O2ASpy6\nGM2t2FTmfHWcKb19MVerirpYxYZ8XkyTuOQmMwQKIYR4XuQKVIh/2XvqFl/uuAhAlfIODO1YGwtz\nuWgRIj/U6pyvF39/f5o3b8706dNRKpW5EjUPaDQaMjIyDM/T0tJQq9V5TtZAzsDFTzNOsU6nlxkO\nAJVSSZ/WNZi6+hh34lL5v18u0/m1KkVdrGJHPi+mSVxyk5gIIYR4VoU26LAQ/wX7z9xm1bbzAHiW\nteejDrUkWSNEPuzZs4cPPvjAaJlWq6VOnTpcu3aNV155hfr167Nlyxa+/PJLgoODAfDw8DCaSTAq\nKgp3d/fCLLoAKpaypU1AJQB2Hr3BlZuJRVsgIYQQQoiXmCRshPjb8QsxLN96Dj3gVtqOIR1rY2kh\njdCEyI/q1avzxx9/8P3336PT6dizZ48hiXPy5EmOHTvGsWPHaN26Ne+//z6LFy8GoE2bNixbtow7\nd+4QGxvL4sWLadu2bRG/m5dT61cqUaGULXpg5bbzZGXLrFFCCCGEEEVBEjZCABevJ7B485/o9VDB\nxYZh78iYNUI8jZIlSxIREcHq1aupX78+8+fPZ8GCBXh4eDx2uy5dutC0aVM6dOhAq1atqFu3Lj16\n9OUMlrYAACAASURBVCikUouHqVVKBnWsgwK4GZvCruM3irpIQgghhBAvJbkiFS+9GzHJhG44TVa2\njhL2GoZ2qo21xqyoiyXEf1b9+vXZuHHjY9eZOXOm0XOVSsXQoUMZOnRoQRZN5FHVSk4E1i7D3lO3\n2LQviobVXbG3yft4QkIIIYQQ4tlJCxvxUotLTGfu/50iNSMLG0szPn6njlyUCCEE0PFVDywt1KRn\nZvPtnsiiLo4QooidPn2agIAAw/MzZ85QrVo1fHx8DD8RERH/z96dh0Vdrg0c/84wDMuAbK4gIJv7\nvqPmnuZeuVCWRz2ZoGa+2GKnTr6mWZ1KezU74ZJZasf0aFkumbsGLpE7ixuICG4gi7INzMz7BzlF\njDAgMAj357q4hN/8nmfuueUaZu55fvcDwLBhw4ocb9OmDc2aNePmzZvF5i1pHiGEqO1khY2otXLy\nCljy39Ok3c3DxtqK0HHtaOBqb+mwhBCiWqijUfNkLx/+s/ci4WevM7irJx71ZMthIWobg8HA5s2b\n+eCDD7Cy+mMjhpiYGHr37m3sRfZn27dvN36v1+uZNGkSHTp0oEGDBsXOLWkeIYSo7WSFjaiV9AYD\nK3+M5trtLBQKmPZka3wa1bF0WEIIUa306+hBXSdbDMCWQ7LKRoja6H5fspCQkCLHo6Ojad68eanj\nv/76a+7du8fLL79s8nZz5xFCiNpIVtiIWmnzwcucupQCwDP9A2jr52bhiIQQovpRWSl56jFfVm6L\n5uTFFC4nZeDn4WTpsIQQVWj06NGEhIRw/PjxIsdjYmJQq9X0798fvV7PkCFDCA0NRa1WG8/JyMhg\n2bJlfPHFF0VW55R1npIoFAqUZfwIWqlUFPlXSE4eRPJimuTFtMrIixRsRK0TfvY6O49eBaB3O3cG\ndm5s4YiEEKL66tayATuOJZB0O4vNBy/z2rMdUCjkBZoQtUX9+vVNHndxcaFbt24EBQWRmprKrFmz\nWLp0Ka+++qrxnG+++YZ27drRrl27B85vzjwlcXPTlPs5ydlZU65xNZnkxDTJi2mSF9MqMi9SsBG1\nSvz1TL76KRaAZp7OPD+oqbzxEEKIEiiVCkb39mPp5jPEXk0n6sodWvvIqkQhars/Nwa2t7cnODiY\nxYsXFym0bNmyhTlz5jz0PCVJTc0q1wobZ2cN6elZ6PWGsg2uoSQnpkleTJO8mPYweXF1Nd0nUAo2\nota4l5PPv787R4HOQF0nW2Y83QaVlbRxEkKI0rTzd8PPow6XkzLZfDCOlk1cUUqxW4haKyMjg7Cw\nMGbMmIGDQ+GbjLy8PGxs/thp8/Lly6SkpNC7d++Hmqc0BoMBna58j0OvN6DTyZvNP5OcmCZ5MU3y\nYlpF5kXerYpa4X6T4dTMXFRWSmY81QYHO2tLhyWEEI8EhULBmD5+ACTcuMupiykWjkgIYUmOjo7s\n3r2bZcuWkZ+fT0JCAmFhYTz99NPGc06dOkWrVq1K7EVjzjxCCFGbScFG1ArbIq5wNi4VgOcHNcW7\noaOFIxJCiEdLMy8XWni7ALD9SAIGg3yiJkRtpVQqCQsLIzY2lu7duzN+/HieeOIJJk6caDwnKSmJ\nevXqFRubnJxMhw4dSE5ONmseIYSozeSSKFHjRcXfYevheAB6tWnEY20bWTgiIYR4NA0L9CYmIY34\n65nEXk03FnCEEDVft27dOHbsmPFnf39/1qxZ88DzH7SNt7u7OydPnjR7HiGEqM1khY2o0TKztKzc\nFo0B8KzvIE2GhRDiIbTwdsGnUeEKxR1Hrlg0FiGEEEKImk4KNqLGMhgMrN4RQ2aWFhtrK6Y92Rq1\ntZWlwxJCiEeWQqFgaHdvAKKupHHlRqaFIxJCCCGEqLmkYCNqrH0nkjhzubBvzfiBATR0tbdwREII\n8ejr0LSe8fl0x5EEC0cjhBBCCFFzScFG1EjXbt3j232XAOjcvD69pG+NEEJUCKVCwZDuXgD8dv42\nN+5kWzgiIYQQQoiaSQo2osbJL9Cx/McoCnR6XOvYMPGJZtK3RgghKlBgq4a4ONpgAHYelVU2Qggh\nhBCVQQo2osb5/pd4km5noQBeHN4Sja21pUMSQogaRWWlZHAXTwCORN0kM1tr4YiEEEIIIWoesws2\nubm5lRmHEBXicnIGPx27CsDgbl4085ItZ4UQojL0auuOjdqKAp2eg6eSLR2OEEIIIUSNY3bBZsSI\nEcTExFRmLEI8lPwCHau3x2AwQCM3e556zMfSIQkhRI1lb6uiV5vC/mD7T1yjQKe3cERCCCGEEDWL\n2QWb/Px86QMiqrXvDsdzPTUbhQL+PqwF1irZwlsIISrTwE6NUQDp97REnr9l6XCEEEIIIWoUlbkn\nDh8+nMmTJzNs2DA8PT2xtbUtcntQUFCFByeEuS4lZbDreOGlUE9088LP3cnCEQkhRM3XwNWeNn5u\nnLmcyp7Ia3Rv2dDSIQkhhBBC1BhmF2x27NiBnZ0d+/btK3abQqGQgo2wmAKdnjU7YzEYwL2uhid7\nyaVQQghRVR7v7MmZy6nEJWdyOSkDPw8pmAshhBBCVASzCzamCjVlFR0dzdy5c7l06RLe3t688847\ntG/fvth5mzZtIiwsjPT0dAICAvjnP/9J69atH/r+Rc2089hVklMKd4WaPKS5XAolhBBVqGUTF9zr\nakhOyWJ3ZKIUbISoZtLT0zl//jwFBQUYDIYit/Xq1ctCUQkhhDCH2QUbgNTUVDZt2sSVK1d47bXX\nOHbsGAEBAQQEBJQ6Ni8vj5CQEEJCQhg7dixbt25l2rRp7NmzB41GYzwvNjaWjz/+mA0bNuDt7c2q\nVauYNWsWe/fuLfujEzXezTvZ/Bh+BYC+HT3kjYIQQlQxhULBwE6N+XrXeX47f5u0u3m4ONpYOiwh\nBLBlyxbmzZuHVqstdptCoZANRYQQopozu+lwdHQ0gwcP5sCBA2zbto3s7GzCw8MZM2YMR44cKXX8\n0aNHUSqVjB8/Hmtra8aMGUPdunU5ePBgkfMSEhLQ6/XodDoMBgNKpbJYvxwhAAwGA1/vOk+BTo+T\ng5rRvf0sHZIQQtRKga0borFVodMbOHgqydLhCCF+t3TpUsaNG0dkZCSxsbFFvqRYI4QQ1Z/ZK2ze\nf/99Jk6cyMyZM+nQoQMACxcuxMXFhY8//pjNmzeXOD4+Ph4/v6JvqH18fIiLiytyrFevXjRp0oRh\nw4ZhZWWFRqPh66+/NjdMFAoFSrPLUIWUSkWRf0Wh6p6XX87cICYhDYAJg5vhqLGukvut7nmxBMmJ\naZIXUVvYWFvRs00jfv41kUOnkxneowkqqzL+MRZCVLi0tDQmTZqEg4ODpUMRQghRDmYXbKKionj3\n3XeLHQ8KCmLdunWljs/OzsbOzq7IMVtbW3Jzc4scy8vLw9/fn7lz59K0aVNWrlzJSy+9xPbt281a\naePmpin39uPOzprST6qFqmNe7mZr2bD3EgBdWzZkUKBPlW87Xx3zYmmSE9NqW1527NjBp59+yo0b\nN3B3dyc0NJSBAwcSGxvLggULiImJwcHBgaCgIKZPn45CocBgMLB48WI2bdqETqdj1KhR/OMf/8DK\nSnpSPSr6tHfn518TSb+n5fSlVDo1q2fpkISo9Xr06EFERATjxo2zdChCCCHKweyCjZOTE8nJyXh7\nexc5HhUVhaura6nj7ezsihVncnNzsbe3L3Js2bJlNGzYkDZt2gAwY8YMNm7cSEREBP379y/1flJT\ns8q1wsbZWUN6ehZ6vaH0AbVEdc7L1z+d5262FrW1kmf6+5GWllVl912d82IpkhPTHiYvrq6P5qeh\n8fHxvPnmm6xevZqOHTsSERHB1KlTOXjwINOmTWPSpEmsXbuWGzduMG7cOJo3b86AAQNYv349Bw4c\n4IcffkChUBAcHMzq1at58cUXLf2QhJkauWlo4e1CTEIaB05ek4KNENVAq1atWLhwIfv27cPHxwdr\n66KrkWfPnm2hyIQQQpjD7ILNs88+y9y5c3n11VcBOH/+PIcOHeLTTz9l8uTJpY739fUtthInPj6e\n4cOHFzmWnJxcZCWOQqHAysrK7E9ZDQYDOp1Zpxaj1xvQ6eTN5l9Vt7wk3rrHvhPXABge2ARnBxuL\nxFfd8lIdSE5Mq0158fHxITw8HI1GQ0FBASkpKWg0GmxsbNi+fbvx+T0tLQ29Xo+TU2Gj8K1btzJx\n4kTq168PQHBwMEuWLJGCzSOmXwcPYhLSiLqSxs072TRwtS99kBCi0hw7doy2bduSlZXFuXPnitxW\n1SuThRBClJ3ZBZupU6ei0Wj44IMPyMnJ4aWXXqJu3bqEhIQwadKkUscHBgai1WpZu3YtzzzzDFu3\nbiUlJaXYdoJ9+/blk08+YejQoTRr1oy1a9ei0+no1KlTmR+cqHkMBgPrd1/AYID6znYM7upp6ZCE\nEH+h0WhITExk8ODB6PV65s2bV6R/woABA7h27RojRoygY8eOAMTFxeHv7288x8fHh/j4eAwGg9lv\nKsraw0x6DJn2MHnp1Lwezg5q0u9pOXg6mWcHlr6L5KNCfl9Mk7wUV51ysnbtWkuHIIQQ4iGYXbBJ\nTk7m2Wef5bnnniM7OxudToejoyM6nY6zZ88aL2F6ELVazcqVK5k3bx6LFy/G29ubzz//HHt7e+bO\nnQvA/PnzCQoKIjMzk5kzZ5KZmUmLFi1YtWqVNEsTAPwae4sLiekAPDMgAGuV9LcQojpq1KgRp0+f\nJjIykunTp+Pt7U1gYCBQ2OPm5s2bBAcH89lnnzFz5kxycnKK9Cmzs7NDr9ej1WqxsTFvi+jy9jCr\nbT2GzFXevAwObMK3uy8QfvY6U55qi411zXqelt8X0yQvxVkqJ7/88gvdu3dHpVLxyy+/PPA8hUJB\nz549zZ73zJkzTJ8+3Tjn2bNnGTduXJHn7uDgYEJCQjAYDHTq1AmD4Y/VpZ06dWLVqlXF5s3IyODN\nN9/k6NGjODo6MmPGDMaOHWt2XEIIUZOZXbAZMGAA4eHhuLq6Fuk7k5CQwPPPP8/p06dLnaN58+Zs\n2LCh2PH58+cbv1coFEydOpWpU6eaG5qoJfK0Or7dV9houLWvK+383SwckRDiQVSqwj8vgYGBDBo0\niL179xoLNjY2Nnh5eTFlyhTWrFnDzJkzsbW1JS8vzzg+JycHlUpldrEGyt7DTHovmfaweenWvB4b\n91zgbnY+u8Lj6NW2USVEWfXk98U0yUtxlu5fNmXKFMLDw3Fzc2PKlCkPPE+hUJi1tbfBYGDz5s18\n8MEHRVoUxMTE0Lt3b5YvX15sTEJCAgAnTpwotZD+9ttvY29vT0REBOfPn+fFF18kICCA9u3blxqb\nEELUdCUWbP7zn//w2WefAYVP1iNGjCj2pJuVlUVAQM1Z8iyqr+1Hr5B2Nw8rpYJnBwTItddCVJK0\ntDRWrFjBuXPnyM/PL3a7qcL7fQcPHuTLL79kzZo1xmP5+fnk5+czYMAANm/ejLOzs/F4nTp1APDz\n8yM+Pp527doBhT3OfH19yxR3eXuY1aYeQ2VR3rw4a2xo71+XkxdT2PfbNQJbNayE6CxHfl9Mk7wU\nZ6mcxMbGmvy+vMLCwti5cychISGsXLnSeDw6OprmzZubHBMdHU2zZs1Kfa2WlZXFnj172LVrFzY2\nNrRt25bhw4fz/fffS8FGCCEopWAzZswY47L0N998k+DgYBwdHY23KxQK7O3t6d69e6UHKmq3W2nZ\n/HTsKgCPd/GkkZssvRaisrzxxhucPXuWkSNHlvly1JYtW3Lu3Dm+//57Ro4cyeHDhzl48CAbN24k\nJiaGTz75hLfeeovExERWrVrFjBkzABg5ciRffPGFcRn/8uXLGTVqVGU8PFEF+nXw4OTFFC4nZ3L1\n5l28GjiWPkgIUWkMBoPJArxarS517OjRowkJCeH48eNFjsfExKBWq+nfvz96vZ4hQ4YQGhqKWq0m\nJiaGe/fuMWrUKG7dukWXLl146623aNCgQZE5EhISUKlUeHr+0ZPQx8eHn3/+2ezHVtb+ZVC9+gxV\nF5IT0yQvpkleTKuMvJRYsLG2tubJJ58EoHHjxnTs2NG4zF2IqvTtvksU6Aw4adSM6NHE0uEIUaMd\nPXqUr7/+2rjapSzq1atHWFgY7733HvPnz6dJkyZ89tln+Pn5sWTJEt555x169uyJk5MTkyZN4qmn\nngJg/PjxpKSkMGbMGPLz8xkxYoRZOxCK6qmljyv1ne24lZ7DgZNJ/O0J05/CCyEq17Fjx5g/fz5X\nrlxBr9cbj99v6G7OJVH3d+/7KxcXF7p160ZQUBCpqanMmjWLpUuX8uqrr6JWq2nfvj2zZs3CxsaG\nhQsXMnPmTDZu3Fhkjuzs7CI9cABsbW3Jzc01+zGWt38ZSO8lUyQnpkleTJO8mFaReSmx+rJ48WKm\nTZuGnZ0dv/zyS4mNy2bPnl1hQQnxZ1FX7nDyYgoAY/v5YWcjRUMhKpObm1uZesf8VefOndmyZUux\n440aNSIsLMzkGCsrK0JDQwkNDS33/YrqQ6lQ0KeDO5v2X+ZI1E3G9vOX524hLGDu3Ln4+/szZ86c\nYoWRh/Xn53N7e3uCg4NZvHgxr776KjNnzixy7pw5c+jevTu3bt0qUgCys7Mr0r8MIDc3t0i/zNKU\ntX8ZSO8lUyQnpkleTJO8mFYZPcxKfPV08uRJ8vPzsbOz4+TJkw88T3qJiMqiNxj47/7LAPi616F7\nDeuFIER19NJLL7FgwQLeeustvL29sba2LnK7OUvohejVphHfHYojL1/Hkagb9O/Y2NIhCVHr3Lp1\ni7CwMHx8fCp03oyMDMLCwpgxY4bx0tm8vDxjsX/FihX07NmTVq1aAaDVagGKfRjg7e1Nfn4+ycnJ\nuLu7A4U9zPz9/c2Opbz9y0B6L5kiOTFN8mKa5MW0isxLiQWbtWvXmvxeiKoSGXuLhJt3ARjb1w+l\nFAeFqHSLFy8mPT2d0aNHm7zdnCX0Qjjaq+ncvD5Ho26y/2QS/Tp4yAc8QlSxxx9/nIMHD1Z4wcbR\n0ZHdu3djMBh45ZVXSE5OJiwsjHHjxgEQFxfH4cOHWbp0KSqVioULFzJgwACcnJyKzOPg4MCAAQNY\ntGgR7777LhcvXmTbtm2sWLGiQuMVQohHVYkFm5IugfozhUJBz549KyQgIe4r0OnZcigOgDa+bjTz\ncrFwRELUDosXL7Z0CKKG6NfBg6NRN0m6ncXFaxk09XS2dEhC1CqzZ89m5MiRbNu2DU9PT5R/uXZo\n0aJF5ZpXqVQSFhbGu+++S/fu3bG1tSUoKIiJEycC8M9//pOFCxcyZMgQ8vPz6du3LwsWLAAgOTmZ\nYcOGsX37dtzd3VmwYAH/+7//S58+fbC3t+e1114rVw81IYSoiUos2EyZMsWsScxtWiZEWRw+ncyt\ntBwUwOg+ZdveVwhRfl27drV0CKKG8PdwonE9DdduZ3HgZJIUbISoYm+99RYKhYLGjRs/dA+bbt26\ncezYMePP/v7+rFmzxuS5Dg4OvP/++yZvc3d3L9JqwdnZmSVLljxUbEIIUVOVWLCJiYmR5cvCIvK0\nOn4IvwJAt1YNZEtYISrZM888w4oVK6hTpw5BQUElPvdv2LChCiOzHL1Bz683TnI2JZpbOSkU6HXY\nqmyws7LFTmWLs40TzrZOuNg44WzjXPizTR2slFaWDr3aUCgU9OvgwdqfLxB5/hbPZAdQx156IAlR\nVSIjI1m3bh1t2rSxdChCCCHKocSCTY8ePejSpQs9e/akR48eeHp6VlVcopb7OTKRjCwtVkoFTz0m\nq2uEqGy9evUyNhd+7LHHLByN5aXkpLImagPxmQllGqdAQR21A862zr8XcpxwsS0s5rjU0qJO91YN\n2XjgMnlaHeFnrjOku7elQxKi1vD29jY2/BVCCPHoKbFgs2vXLo4ePUp4eDirVq1Cr9fTs2dPAgMD\nCQwMxNlZljaLincvJ5+fjhW+SerXwYN6znYWjkiImu+ll14yfj98+HCaNGliuWAsTG/Qs+rsWhLv\nJQPQ0q0ZvnWaYGNlTY4uj7yCPLIKsknPzSA9L4O0vHTydIVviAwYyNDeJUN7lwQSTc5vLOrYOONi\n6/R7EeevhZ2aU9Sxs1ER2KohB04mceBUEoO7eUkDeSGqyLRp03jjjTeYMGECXl5eqFRFX/r36tXL\nQpEJIYQwR4kFmzp16jBo0CAGDRoEwLVr14iIiOCnn35i/vz5NGrUiB49ehi/hKgI2yKukJOnw0Zt\nxfAeTSwdjhC1zpAhQ2jZsiUjRoxg6NCh1K9f39IhVanIm6eMxZoXWj9Px/ptSx2TU5BDWm4GaXkZ\npOelk3a/mJObXnJR5+6DizqOagdcbJypa+dKfft61LevSz27ujSwr4u9tX3FPeAq0Le9OwdOJnE7\nPZeo+Du08XWzdEhC1AqhoaEAvPfee8Vukx6UQghR/ZVYsPmrxo0bM27cOMaNG4fBYCAqKoqIiAhW\nrFghBRtRIVIycth34hoAT3T1oo5Geh0IUdX27NnDzp072bZtGx999BEdO3ZkxIgRDB48uNiWrDVN\nvi6fHy7/BEDbuq3MKtYA2KnssHOww92h4QPPeVBR535h569FnUztXTIfUNRxUjvi6ehBY0cPPB3c\naezogZutS7XtO+fVwBE/jzpcTspk/4kkKdgIUUViY2MtHYIQQoiHUKaCzZ07d7C3t8fW1pbo6GgO\nHjxI69atmTp1amXFJ2qZrYfjKdAZcLS3ZlAX6ZkkhCV4eHgwZcoUpkyZQmJiIrt27WLz5s28++67\n9OzZk88//9zSIVaaX2+eJC0vHaVCySi/IRU6d3mKOndy07mdncLtnBRuZaeSq8sFKFyhkxrLudQ/\n3ow5WGsIcPYlwMWPpi5+NLSvX60KOP06eHA5KZPTl1NIzcjFzenhdqwRQpg2atQoevToQc+ePena\ntStqtXz4JYQQjyqzCzZ79uxh9uzZhIWF4eHhwfPPP4+7uzurVq1i9uzZTJgwoTLjFLXAtdv3iDh3\nA4ARPZpgZ1OmeqIQohK4uLhQr149GjRowIULF7h165alQ6pUlzOuANDcNYCGmqq/FKykoo7BYOBe\nfhY3s29z7V4y1+4mc+1uEslZN9EZdNzLz+Lk7bOcvH0WADdbV9rVa0X7em3wcfJCqVBW9cMpokvz\n+mzYe4l7OfkcOp3MU72lobwQleHDDz8kIiKCNWvWMHv2bJo3b27sQdmmTZtqVci1lAKdnuspWaRn\nZKPXGSwdTrWgtFKQp1dITv5C8mKa5MU0pZUCxzoVe9m62e+IlyxZwsyZM+nRoweLFi3C3d2d7du3\ns2/fPhYuXCgFG/HQthyMwwDUdbKlbwcPS4cjRK1179499uzZw08//URERATu7u4MHz6c0NBQfHx8\nLB1epbqaWXhJprdj9Vvhp1AU9rVxVDvg7/zH/0OBvoDkrBvEpSdwMf0yF9PiyCrIJjX3DvsSD7Mv\n8TDONk70aNSFHu5dcbG1zIYB1iorerVpxE/Hr3LoTDIjejZBZWXZIpIQNVGzZs1o1qwZkydPRqvV\ncvLkSSIiIpg/fz7Xrl2ja9euBAYG0qNHD7y9a9+ubQU6PW+uOEpKRq6lQxFC1ED1Xe15f2o3FFRM\ncdzsgs2VK1cYPnw4APv372fAgAFA4R+FlJSUCglG1F4XEtM5danw9+ip3r7yIl4ICwoMDMTFxYUh\nQ4YwY8YM2rRpY+mQqoRWp+VGduEKIu86jS0cjflUShVejo3xcmxMX8+e6A16Eu8mcfp2FKdun+Nm\n9i3S8zLYcWUPO6/spXXdFgzy7ouvU5Mqj7VPB3d+On6VjHtaTl1MoXPz2tXQWoiqplar6datG926\ndSM0NJSMjAzjDrCrV69m9+7dlg5RCCFECcwu2DRo0IDo6GjS0tK4dOkS77zzDgAHDhygceNH54Wt\nqH4MBgP/PXgZAM/6DnRr2cDCEQlRu61YsYLu3bvXumXz1+5dR2/QA+Dp+Oiu8lMqlHjX8cS7jicj\n/Z4g+d4Njlz/laPXI8kuyOFsSjRnU6Jp6dqMYb6P06SOV5XF1sDFnlY+rkTF32H/ySQp2AhRBfR6\nPQaDASsrK7Kzs8nJyeH5559n/vz5lg7NIlRWSv41LRCdwor0jCy5nON3SisFzk4ayclfSF5Mk7yY\nprRS4O9dl7uZ2egqKC9mF2z+/ve/M2vWLADat29Pp06dWLZsGWFhYXz44YcVEoyonU5fSuXStQwA\nRvfxQ1nL3iQKUR18++23RX6+evXqA88NCgqq7HAs4urdwsuh6qgdcbapObthuTs0ZHTACEb4PsHJ\nW2fYm3iIpHvXib5znug752lXrzVjA0ZW2aVSfdt7EBV/h5iENK6nZtHITVMl9ytEbRQZGcnLL7/M\nokWL8PPzY8yYMWi1WnJycvjwww8ZOnSopUO0CJWVkvquGmyUhgp7U/Wos7JS4Co5KUbyYprkxTQr\nKwXWqoq9UsTsgs348eNp3749ycnJ9OrVC4BevXoxcOBAmjdvXqFBidpDrzew+ffVNc08nWnj62rh\niISonZYvX27WeQqFosYWbBIzkwDweoRX15REbWVNt0ad6NKwA6dun2N7/G5uZN3k9O1zxN65wEjf\nIfRuHIgVVpUaR/sAN1wcbUi7m8eBk8k8OzCgUu9PiNrsX//6F0OHDqV9+/Z8+eWX2NjYsH//frZt\n28ayZctqbcFGCCEeFWXahqdly5bk5uayfft2Bg8ejEajqZXNykTFORJ1g6SULADG9PWrdZdgCFFd\n7Nu3z9IhWNz9FTZejjX7Ml+lQknH+m1pX681x26c4LtL28jKz2bTxa0cv3mCya2fwdXVodLu30qp\npHc7d7b+Ek/42euM7uOL2rpyi0RC1Fbnz59nyZIl2NnZsW/fPgYOHGjsazNv3jxLhyeEEKIUZq/X\nuXPnDkFBQfztb3/j7bffJi0tjUWLFjFs2DASExMrM0ZRQ+UX6Pj+cBwAnZrWw8+j5lyCIMSjF75g\n0gAAIABJREFULjU1lbCwMN544w1SU1PZsWMHFy9etHRYlUar03I96yYAXo9Qw+GHoVQoCWzUmbe7\nvUqXBh0BSMhM5INjSzmaeKJS77t3O3eUCgXZeQUci7lZqfclRG3m7OxMUlISiYmJREVF0adPHwDO\nnDlD/frSQ0oIIao7sws2CxcuxM3NjWPHjmFjYwMULrP08vJi4cKFlRagqLn2n0giNTMPhQKe7uNr\n6XCEEL+Ljo5m8ODBHDhwgG3btpGdnU14eDhjxozhyJEjlg6vUtzIvoWBwmuwGzu4WziaquWodmBS\nq2eY0e4FHK0dyNXlsThiJZsvbEOn11XKfbo42tChaV0A9kZew2CQ69+FqAxjx45l+vTpBAUF4e3t\nTWBgIGvXrmXOnDlMnDjR0uEJIYQohdkFm4iICP7nf/4HjeaP5oBOTk688cYbREZGVkpwoubKzi1g\n25EEAB5r20iaTgpRjbz//vtMnDiRDRs2YG1tDRQW7SdMmMDHH39s4egqxz1tlvH7OmpHC0ZiOS3d\nmvFG11n4OhVe6rw74SDLTn9BTkFupdzfwE6FK5mu3rrHhcT0SrkPIWq7mTNn8v777zN16lTWrVuH\nUqnEy8uLpUuXMmHCBEuHJ4QQohRmF2x0Oh16vb7Y8bt372JlJdeei7L56XgC93LysVYpGdVLVtcI\nUZ1ERUUxcuTIYseDgoK4fPmyBSKqfFn52QDYqeywUtbev2nONk7M7hzCkIB+AFxIu8SSk8u5q71X\n4ffV1NMZr/qFvXL2/HatwucXQsC4cePw8vJi0qRJ1K1buKqtT58+9O3b17KBCSGEMIvZBZuBAwfy\n0UcfcefOHWNj2EuXLrFgwQIGDBhQaQGKmif9Xh4//1rY92hgp8a4ONpYOCIhxJ85OTmRnJxc7HhU\nVBSurjVzJ7d7+YUrbDTW9haOxPJUShWTO47juRajUaAg8W4Sn5z4nLTcil0Fo1AoGNjZE4ATF26T\nkpFTofMLIeDatWuoVGXaY0QIIUQ1YnbB5s0338TBwYGePXuSnZ3NiBEjGDFiBI0aNeLNN9+szBhF\nDfNj+BW0+XrsbVQMDZRdxoSobp599lnmzp3Lrl27gMJdRtavX8+8efNq7Jbe91fYOFjL5Zn3Pda4\nO5NbPYuVwoqb2bdZ9Nu/uZWdUqH30a1lfRztrTEYYN+JpAqdWwgBzzzzDDNmzGDNmjXs2bOHX375\npciXEEKI6s3skruDgwNLliwhMTGRy5cvU1BQgJ+fHz4+PmbfWXR0NHPnzuXSpUt4e3vzzjvv0L59\n+yLnzJ07lx9//NH4s8FgICcnh48//pgRI0aYfV+ierqZls2h04Wf3A8N9EZja23hiIQQfzV16lQ0\nGg0ffPABOTk5vPTSS9StW5eQkJAa26TyfsFGVtgU1alBe2xVdqw8+zVpeel8emolr3SajrNNxezq\nZ62yok97D7ZFXOHQqWRG9fTBRl17L0kToqL9+9//BuCDDz4odptCoSAmJsbsuc6cOcP06dONhZ6z\nZ88ybtw4bG1tjecEBwcTEhKCVqvlo48+YufOnWi1Wjp27MjcuXNxdy/e1L2keYQQorYrsWATHx9v\n8ri3t3exc0or3OTl5RESEkJISAhjx45l69atTJs2jT179hRpZDx//nzmz59v/HnJkiVERkbyxBNP\nlP5oRLX33aE4dHoDzg5qBnSqHVvnCvEo0Ov1KJV/LLp87rnneO6558jOzkan0+HoWNiI9/bt29Sr\nV89SYVaarN8viZIVNsW1cmvGjHYv8NnpVdzJTePTU6sI7RhSYbnq18GDnUcTyM4r4EjUDfp28KiQ\neYUQEBsb+9BzGAwGNm/ezAcffFCkb2VMTAy9e/dm+fLlxcYsX76cs2fP8v333+Po6MjChQt55ZVX\n+M9//lPs3JLmEUKI2q7Egs2QIUNQKBQP3G7z/m3mVOiPHj2KUqlk/PjxAIwZM4avvvqKgwcPMnTo\nUJNjzp07x9q1a/nxxx+NO5WIR9eVG5kcj7kFwKhePthYy6eoQlQXs2bN4pNPPinW68De/o8VJzt2\n7OCdd97h2LFjVR1epZMVNiULcPFlSusJLD/7FTeybvLv06t5uf2L2KpsSx9cChdHGzo3r8+x6Jvs\njkykd3t3lL/3yhNCPDytVsuuXbu4cuUKEyZM4Pz58/j5+RmbEJcmLCyMnTt3EhISwsqVK43Ho6Oj\nad68uckx2dnZTJ8+3Xgfzz33HE8//XSxDwdKm0cIIWq7Egs2e/furbA7io+Px8/Pr8gxHx8f4uLi\nHjjm/jaEjRo1Mvt+FAoFSrM78xRSKhVF/hWFKjovmw8W/l83crOnTwd3rB7RfMvvS3GSE9Mepbwc\nP36cGTNm8Omnn6JWq4vclpGRwbx589i5cyc9evQoda4dO3bw6aefcuPGDdzd3QkNDWXgwIHcuHGD\n+fPn89tvv6FSqXjiiSeYM2cOarUag8HA4sWL2bRpEzqdjlGjRvGPf/yjynYhlKbDpWtdtwUTWozj\nq+gNJGQmsvLsWqa3+3uF7Kr1eGdPjkXf5HpqNmcvp9LO37w3kkKIkiUmJjJx4kR0Oh0pKSk8+eST\nrF+/nmPHjvHll1/SsmXLUucYPXo0ISEhHD9+vMjxmJgY1Go1/fv3R6/XM2TIEEJDQ1Gr1cyZM6fI\nufv27SMgIKBYsaa0ecwhr/0rhuTENMmLaZIX0yojLyUWbDw8ii9LvnDhApcuXUKv1+Pr62vWEz0U\nVtrt7OyKHLO1tSU3N9fk+b/99huXLl1ixYoVZs1/n5ubxriLVVk5O8tSeFMqIi+nL9wmKv4OABOH\nt6Je3ToPPaelye9LcZIT0x6FvHz99ddMmTKF4OBg/v3vfxufrw8cOMA///lP8vLyWLBgAWPHji1x\nnvj4eN58801Wr15Nx44diYiIYOrUqRw6dIjXXnuNgIAADh06RGZmJjNmzOCzzz4jNDSU9evXc+DA\nAX744QcUCgXBwcGsXr2aF198sSoe/p9W2FT//ytL6tqwI9kFOWy6sJXYtIv89+IPBDV76qHn9XWv\nQ1NPZy4kprPz2FUp2AhRQRYuXEivXr2YN28enTp1AmDx4sW89dZbvP/++6xdu7bUOerXr2/yuIuL\nC926dSMoKIjU1FRmzZrF0qVLefXVV4uct2PHDpYvX/7A1/TmzvMg8tq/YklOTJO8mCZ5Ma0i82J2\n0+HMzExef/11Dhw4gJOTEzqdjnv37tGxY0eWL19u7G/wIHZ2dsWKM7m5uUWW2//Zli1bGDlyZJH+\nNuZITc0qV5Xd2VlDenoWer3py79qo4rKi95g4IsfzgKFL8qbezhy5869igqzysnvS3GSE9MeJi+u\nrg6VFJVpzZo145tvvmHy5Mm88MILfPLJJyxZsoQtW7bQr18/5s2bR4MGDUqdx8fHh/DwcDQaDQUF\nBaSkpKDRaFCr1djZ2TFt2jRsbGyoV68eI0aMYPfu3QBs3bqViRMnGt8YBAcHs2TJkios2MgKG3P1\nbdyT1Jw77Es8zKGkI7g7NOQxj8CHnndINy8uJKZzITGdy8kZ+LlXTGNjIWqz3377jW+//bbIyhaV\nSsW0adN46qmHK7aGhYUZv7e3tyc4OJjFixcXKbSsWLGC5cuXs3TpUrp27VrueUoir/0rhuTENMmL\naZIX0yrjtb/ZBZuFCxdy69Yttm/fbry06eLFi8yZM4cPP/yQBQsWlDje19eXdevWFTkWHx/P8OHD\nTZ6/f/9+li1bZm54RgaDAZ2uzMMA0OsN6HTyC/dXD5uXo9E3iL9+F4AxffzQ6wEe/TzL70txkhPT\nHpW8eHp6sn79el544QX69++Pk5MTH3/88QOfpx9Eo9GQmJjI4MGD0ev1zJs3DwcHh2Kfru7fv9/Y\ntyAuLg5/f3/jbT4+PsTHxxv7pJmjrMvi7y9XLTAUoNXnA1DHxgErq9q9vNec5bxjmg3nZvYtolLP\ns/HCVho5NKCZq98DzzdH+6Z18ainIel2FruOX2Xm6LYPNV9Fk+XfpkleiqtOOVGr1WRmZhY7fu3a\ntTJ/KPpnGRkZhIWFMWPGDBwcCt9k5OXlYWNjAxQ2sp87dy7h4eGsX7/+gT1qSpvHHPLav2JJTkyT\nvJgmeTGtIvNidsFm3759rFq1qkgfmoCAAObOnUtwcHCpBZvAwEC0Wi1r167lmWeeYevWraSkpNCr\nV69i5yYmJpKZmUnr1q3L8FBEdZRfoGfL771r2vq50dzbxcIRCSFK0qBBA9avX8+LL75IXl6eWT1r\nTGnUqBGnT58mMjKS6dOn4+3tTWBg4SoMg8HAwoULiYuL46OPPgIgJyenyJaudnZ26PV6tFqt2S/c\ny7ss3srujz+oHnXr4upctaubqqvSlvO+2nsqb+35kOS7N1l1di3vP/4G9R0e7lKmsQOa8n8bTvLb\n+dvk6sC9XvX7v5Dl36ZJXoqrDjkZOXIkCxYs4J133gEKCyRxcXG88847ZS7G/5mjoyO7d+/GYDDw\nyiuvkJycTFhYGOPGjQNg2bJlHDlyhI0bN5a4s2Bp8wghRG1ndsFGpVKZfNFsa2tLfn5+qePVajUr\nV65k3rx5LF68GG9vbz7//HPs7e2ZO3cugHE776SkJJycnMxuNiaqr30nrpGSkYtCAWP7+Zc+QAhh\nEb/88kuRn1944QUWLFjAhAkTeP3114s0/zVVaP+r+7tNBQYGMmjQIPbu3UtgYCC5ubm8/vrrnD9/\nnrVr1+Lm5gYU/i3Jy8szjs/JyXng350HKeuy+PvLVpNTbhuP6XIU3NE/updsVoSyLOcNbjORfx3/\nlLvaLD48FMZrXWZgbVX+XR3bNHHGxdGGtLt5bNgVy6Sh1WfnGFn+bZrkpbjqdDnsK6+8wuLFixk/\nfjxarZYxY8agUql49tlnmT17drnnVSqVhIWF8e6779K9e3dsbW0JCgpi4sSJFBQUsHr1agoKChg0\naFCRceHh4aSnpzNs2DC2b9+Ou7v7A+cRQghRhoJNjx49+PDDD1m8eDHOzs4A3Llzh48++sjsT2Cb\nN2/Ohg0bih2/X6i5r3v37oSHh5sbmqimsnLz2RZxBYDH2rrjUdfynzQJIUybMmWKyeMpKSkEBwcb\nf1YoFMTExDxwnoMHD/Lll1+yZs0a47H8/HwcHR1JT09nypQp2Nvb8+233xr/lgD4+fkRHx9Pu3bt\ngMJLZn19fcv0GMq7LP5uXpbxe1ulnSzt/Z05y3nr2tZlUqvx/Pv0F1y9m8Sm8z8+VBNiBQoe7+zJ\nxv2XOHzmOiN7+eCkqV4f3sjyb9MkL8VVh5yoVCpef/11Zs2axdWrV9HpdHh5eWFvb09qaqqxaG6O\nbt26cezYMePP/v7+RZ7r/3yfp06deuA89vb2nDx5stR5hBBClKFg88YbbzBx4kT69OlD48aNgcLr\nX/38/HjvvfcqLUDx6NoWcYWs3AJsrK148jEfS4cjhChBbGxshczTsmVLzp07x/fff8/IkSM5fPgw\nBw8eZOPGjcycOZO6devy6aefYm1ddBXGyJEj+eKLL+jevTsqlYrly5czatSoCompNPd+3yHKxkqN\nSmn2n0Xxu1ZuzXjCuz8/JezjUNIR/Jx96Nygfbnn69PenR8jrpCTV8DPv15lbF9ZnSlEebVo0YLw\n8HBcXV0JCAgwHk9MTGTkyJFFCidCCCGqH7NfmdarV48ffviBQ4cOERcXh42NDb6+vvTo0aPcW+mJ\nmut2eg57f7sGwOCunjg7mH9ZgxDi0VWvXj3CwsJ47733mD9/Pk2aNOGzzz4jIyOD48ePY2NjU2Sn\nkJYtW7J+/XrGjx9PSkoKY8aMIT8/nxEjRjB58uQqifne7ztEOciW3uU21OdxLmdc4WJ6HN/E/hdP\nB3caaExvBVwaOxsVAzp5sC0igX2/JfFEVy8c7avXKhshqrPNmzezadMmoHDl4YsvvlisSH779u0H\nbtcthBCi+ijTR4kqlYp+/foV6V9wv3+N9JsRf7blUBwFOgNOGjVPdPOydDhCiCrUuXNntmzZUuz4\n+fPnHzjGysqK0NBQQkNDKzM0k2RL74dnpbRicqvxvP/r/3FXe49V59bxeueZ5e5nM6iLF3sir5Gr\n1fHzr4mM7vNwO1AJUZsMGTKE69evA3Dq1Cm6du1abEcojUZTrL+MEEKI6sfsgs3x48d55513uHLl\nCvrCfZkBjFuultTTQNQu8dczORZ9E4BRj/lgq5ZLDIQQ1dc9beElURpZYfNQnGzq8PdW41l6ciXJ\nWTfYenknY5qOLNdcDnbWDOjUmO1HEtjz2zUGd/XCwa78zYyFqE3s7e156aWXAPDw8GDYsGHywaoQ\nQjyizH4n/fbbb+Pv78+cOXOKbL0qxJ8ZDAY27rsEQCM3ex5r28jCEQkhRMlkhU3FaerizyDvfuxK\n2Mf+a7/Qwq0ZrdyalWuuwV292PPbNfK0OnYdvyqrbIQw07fffstTTz2FWq1Gq9Xy3XffPfDcoKCg\nKoxMCCFEWZldsLl16xZhYWH4+EjzWPFgJy6kcD4xHYCxff2xKsseu0IIYQFZ+bLCpiIN83mc2DsX\nSbibyNqYb3mr62wc1WXfqtjBzpqBv6+y2SurbIQw2/Llyxk0aBBqtZrly5c/8DyFQiEFGyGEqObM\nLtg8/vjjHDx4UAo24oG0+To27L0IQAtvF9r5m79VpBBCWMofTYdlhU1FsFJaManVM7z/6xLuau+x\nLmYTIW0nlWuDgkFdPIv0snm6d9m2eheiNlq1ahXp6emkp6fzxRdfWDocIYQQD8Hsgs3s2bMZOXIk\n27Ztw9PTE+VfVk4sWrSowoMTj5YdRxNIzczFSqlg/ONNZfcwIcQjQVbYVLz69vUYGzCK9bGbOJca\nw+GkI/Ru3KPM8zjaqxnQqTE7jiawOzKRgZ0aU0cjvTiEKMnQoUNRKBTGPpOmSA9KIYR4NJhdsHnr\nrbdQKBQ0btxYetiIYm6l57Dj6FUABnZujEddeeMjhHg0GAs2KjsLR1KzBDbqTHRqLCdvn2XLpW0E\nuPjRSNOgzPM80c2L/SeTyMkr4MfwKzw3qGklRCtEzbF3715LhyCEEKKCmF2wiYyMZN26dbRp06Yy\n4xGPqG/3XqRAp8dJo2ZkT7lsTgjx6MjXFwCUewtqYZpCoeDZ5qOJz7xKel4GX0Z9w2udZ2KtLNvO\ngQ521gzt7sXmg3EcOJXEwM6NaeAql68J8SAeHh6WDkEIIUQFMbsjrLe3N1qttjJjEY+oM5dTOXkx\nBYCx/fyws5FtvIUQjwaDwUDB7wUblVIKNhVNY23PxJZBKFCQdO86P1zeWa55Hu/siYujDTq9gc2H\n4io4SiGEEEKI6snsd9bTpk3jjTfeYMKECXh5eaFSFR3aq1evCg9OVH95Wh3rfj4PgH9jJwJbNbRw\nREIIYb77xRoAa6WVBSOpuZq6+DPQqw+7rx5gX+JhWro1o4Vr2S5rUltb8WQvH77cGUtk7C0uJ2fg\n5+5USRELIYQQQlQPZhdsQkNDAXjvvfeK3SZNy2qvLYfiSMkobDQ8YVAzaTQshHik5P+pYCMrbCrP\ncN9BxKZdJPFuEl9HF2717aAuW6+znm0a8fOviSSlZLFp/2XmjO8gf3OEEEIIUaOZfUlUbGzsA7+k\nWFM7XU7KYE9kIgBDu3vjWd/BwhEJIUTZFOj+XLCRFTaVRaVUMbnls6iV1mRq77IudhMGg6FMcyiV\nCsb09QPgQmI6p36/FFcIIYQQoqYyu2AjxJ/lF+hZvSMGA+BeV8PwHk0sHZIQQpSZVp9v/F6lkP5b\nlamBpj5jAkYCcDYlmsNJR8s8R1s/N5p7OQPwn70X0ebrKjRGIYQQQojqRAo2oly2RVzhemo2CmDy\nkOZYq+RXSQjx6PnzChtruSSq0vVw70q7eq0B2HLpR65n3SzTeIVCwfjHm6JUKEjJyGXH0YTKCFMI\nIYQQolqQd9mizK7cyDS+SH68iyd+HtL4UQjxaNLq/rTCRi6JqnQKhYLxzUfjbONEvr6AL6O+KdJH\nyByN6znweJfGAOw4epWbadmVEaoQQgghhMVJwUaUSU5eAWFbo9DpDdR3tuOpx3wtHZIQQpRb0V2i\nZIVNVXCw1jz0Vt8je/rg7KCmQKfnm90Xy9wPRwghhBDiUSAFG1Em634+z620HKyUCoJHtcJGLZ9I\nCyEeXUV3iZLns6pyf6tvgH2Jh4lJvVCm8XY2KoL6BwBwNi5VGhALIYQQokaSgo0wW/jZ6xyJKuw3\nMLqPHz6N6lg4IiGEeDj5RS6JkqbDVWm47yC8HD0A+CpmA5nau2Ua37VFfWMD4vV7LpCdW7ZLq4QQ\nQgghqjsp2Aiz3LyTzbqfCz8Bbe3jyqCunhaOSAghHt79FTYKFFgpZIVNVVIpVUxqNR61lZq72nt8\nFbUBvUFv9niFQsHzg5qhslJwJzOPjfsvVmK0QogzZ87Qq1cv489nz56lRYsWdOjQwfgVFhYGgMFg\nYNGiRXTv3p0uXbrw7rvvotOZ3tUtIyODGTNm0KlTJ/r27cumTZuq5PEIIcSjQAo2olQ5eQUs23KW\nvHwddTRqXhjeEqVCYemwhBDioeX/vkuUSmmFQp7XqlwD+3o82+xpAGLTLrLryv4yjXevq+HJ33up\nHTp9nbNxqRUeoxC1ncFg4L///S9///vfyc//Y1ViTEwMvXv35uTJk8avkJAQANavX8+BAwf44Ycf\n2LFjBydOnGD16tUm53/77bext7cnIiKCpUuX8vHHH3Pq1KkqeWxCCFHdyfpvUSKd3sDn350jKSUL\npULB1BEtcdKoLR2WEEJUiHx94ZsPlTQctpiuDTtyMe0yEdd/ZXv8z/g5N6Gpi5/Z4wd39eTEhdvE\nJWeyZmcsC17oir2t/H8KUVHCwsLYuXMnISEhrFy50ng8Ojqa5s2bmxyzdetWJk6cSP369QEIDg5m\nyZIlvPjii0XOy8rKYs+ePezatQsbGxvatm3L8OHD+f7772nfvr1Z8SkUCpRl/AhaqVQU+VdITh5E\n8mKa5MW0ysiLFGxEib78MYrTlws/sRz/eAAtm7haOCIhhKg4f15hIyxnbNNRXMlMJDnrBl9GfcM/\nuv4PddSOZo21Uip5YVgL/nf1r6TdzWPD3kv8fViLSo5YiNpj9OjRhISEcPz48SLHY2JiUKvV9O/f\nH71ez5AhQwgNDUWtVhMXF4e/v7/xXB8fH+Lj4zEYDEVWMyYkJKBSqfD09Cxy7s8//2x2fG5umnKv\nkHR21pRrXE0mOTFN8mKa5MW0isyLFGzEAx04mcTWQ5cB6N/Rg/4dG1s4IiGEqFjGgo1C/hxaktpK\nzQutn+dfkUvJ1N7ly3Pf8FL7KViZWUhr5Kbh6d6+bNx/iV/OXqd9QF06Nq1XyVELUTvcXyXzVy4u\nLnTr1o2goCBSU1OZNWsWS5cu5dVXXyUnJwdbW1vjuXZ2duj1erRaLTY2Nsbj2dnZRc4DsLW1JTc3\n1+z4UlOzyrXCxtlZQ3p6Fnq9oWyDayjJiWmSF9MkL6Y9TF5cXR1MHpdXqMKkkxdu8/VP5wFo7evK\nswMDLByREEJUvPuXRFlbyZ9DS2uoqc+zzZ7mq+gNXEi/zPeXdzA6YITZ4wd18eTExdtcupbB6u0x\neNV3oK6zXSVGLETtdr/BMIC9vT3BwcEsXryYV199FVtbW/Ly8oy35+TkoFKpihRroLCQ8+fzAHJz\nc7G3tzc7DoPBwAP6GZdKrzeg08mbzT+TnJgmeTFN8mJaReZFmg6LYk5fSuHf359Dpzfg2cCR6U+1\nxqqsH10IIcQjQFbYVC9dG3akX+PCXWj2JR7m1xsnzR6rVCoIHtEKja2K7LwCPt8aRYHO/F2nhBDm\ny8jI4F//+hf37t0zHsvLyzMWZPz8/IiPjzfeFh8fj6+vb7F5vL29yc/PJzk5uci5f76cSgghajN5\nFy6KOBeXymffnUWnN9DQ1Z53Q3qgkeaNQoga6v623tbSdLjaeMp/GAHOhW/s1sf+l8S7yaWM+IOb\nky1ThrcEIP56Jhv3X6qUGIWo7RwdHdm9ezfLli0jPz+fhIQEwsLCePrpwl3fRo4cyRdffMGNGzdI\nSUlh+fLljBo1qtg8Dg4ODBgwgEWLFpGTk8OZM2fYtm0bI0aYv7pOCCFqsiot2ERHRzNmzBjat2/P\nqFGjHrhlX2RkJE899RQdOnRgxIgRHDlypCrDrLXOxafy6ZazFOgM1He2Y85zHXCtY1v6QCGEeEQV\nGHeJkqbD1YWV0ooXWj+Ps40T+fp8Vp79irvae6UP/F07/7oM6eYFwJ7Ia/x2/lZlhSpEraVUKgkL\nCyM2Npbu3bszfvx4nnjiCSZOnAjA+PHj6d+/P2PGjGHYsGF07NiRyZMnA5CcnEyHDh2Mq2oWLFhA\nQUEBffr04eWXX+a1116jXbt2FntsQghRnVRZwSYvL4+QkBCefvppfv31VyZMmMC0adPIysoqct7N\nmzeZNm0aISEhnDhxguDgYGbOnFmm5mOi7A6fSWbJpjPkF+ip62TLa89KsUYIUT47duxgyJAhdOjQ\ngWHDhrFnz54it6elpTFgwAAuXLhgPGYwGFi0aBHdu3enS5cuvPvuu+jK25SgDLTGXaLkkqjqxFHt\nwNQ2f0OlVJGam8byM1+h1eWbPf6p3r74ezgBsGp7DIm3zC/4CCFM69atG8eOHTP+7O/vz5o1a/jt\nt98IDw/n5ZdfNu7WZGVlRWhoKL/88gvHjh3jn//8J1ZWhYVxd3d3Tp48ibu7OwDOzs4sWbKE48eP\nc+DAAcaMGVP1D04IIaqpKnuFevToUZRKJePHjwdgzJgxfPXVVxw8eJChQ4caz9u6dSs9evRg8ODB\nAAwfPhwfHx+UZvZQUSgU5eoU/+d/axODwcB3h+LY+ssVANzr2vPKM+2p62RXq/NSEslLcZIT02pj\nXuLj43nzzTdZvXo1HTt2JCIigqlTp3Lo0CFcXV2JjIzk7bff5tq1a0XGrV+/ngMHDvBxCk8gAAAg\nAElEQVTDDz+gUCgIDg5m9erVvPjii5Uab4Hu/iVRUrCpbrzreDKhxTi+jPqG+MwE1sZ8y+RW41Eq\nSv8jr7JSEjKqFQu+iiQjS8uS/57m7b91xsnBptSxQgghhBDVRZW9Qo2Pj8fPz6/IMR8fH+Li4ooc\ni4qKokGDBsyYMYPIyEiaNGnCW2+9hVqtNut+3Nw0xup+WdW2feRztQV89t/THPit8I1TG7+6vDmp\nCw72RXNd2/JiLslLcZIT02pTXnx8fAgPD0ej0VBQUEBKSgoajQa1Wk1kZCSzZs3itddeY86cOUXG\nbd26lYkTJxq3jw0ODmbJkiWVXrDRGi+JkoJNddS5QXtScu7wY9xPnLh1hrp2bozyG2LWWNc6trw8\npi3/Wn+CO5l5LN18htfHd8TGWi5/E0IIIcSjocpeoWZnZ2NnV3R7TVtb22KXOmVkZHDo0CE+/fRT\n/u///o+NGzcydepUdu3ahZOTU6n3k5qaVa4VNrVtH/lrt+/x2ZZzJKcUXpLWo3VD/j6sBdpcLXdy\ntUDtzIs5JC/FSU5Me5i8uLo6VFJUlU+j0ZCYmMjgwYPR6/XMmzcPBwcHAgIC2Lt3L7a2tsUKNnFx\ncUV2BfHx8SE+Ph6DwWB2Eb6sKyyVSsUfK2ysrLGyqj0roUpS3VaGDfXtT2puKhHJv/Jzwn7q2rvQ\nu3GgWWP9GzsRPKoVn24+S/z1u6zeHsP0p1ujLMcHO9UtL9WF5KU4yYkQQoiKUmUFGzs7u2LFmdzc\nXOzt7YscU6vV9O7dm169Crf1fO655/jiiy84ceIE/fr1K/V+DAYD5W17UBv2kTcYDBw6ncx/9lxE\nW6BHoYAnH/NleKA3CoXC5OOvDXkpD8lLcZIT02pjXho1asTp06eJjIxk+vTpeHt7Exj44DfZOTk5\n2Nr+0TfLzs4OvV6PVqs1bhNbmvKssLy/S5TGzvaRLpJVhuq0MuylHn8j81Am526d5z8x31Hf2YUe\nXp3NGjuohwOZuTq+2h7Nr7G32HggjmlPt5XVuBVM8lKc5EQIIcTDqrKCja+vL+vWrStyLD4+nuHD\nhxc55uPjw9WrV4sc0+v1GAy1681OZbiTmcu6ny9w6lIKAC6ONgSPbEVTT2cLRyaEqGlUqsI/L4GB\ngQwaNIi9e/eWWLCxtbUlLy/P+HNOTg4qlcrsYg2UfYWlUqkg//dGtvp8A3fuSGNaqL4r5l5o+Ryf\n5Czn6t0klh79kv9v797joqrz/4G/ztyYGe4gqCh3FQQVxBvmrVDTSmUrL/2s/XY1qHbtZ5tb23fX\nNd3t2++x39q2NcV1y76a1X410zbtYqV0MU1MRVRQYVIEkcsAcpvrOb8/gIFpBgEVZmBez11jzmfm\nzHnPh/E9H9/z+ZxjbpIwOmRkl/a9LWkQLpTW4MCxUnxy8CdIVhH3zRzWraKNu/aLq7FfHHnq7Eoi\nIrr5eq1gM3nyZJhMJmzduhX33Xcfdu/ejcrKSttMmlbp6elYsmQJDhw4gOnTp2Pbtm0wGo2YNGlS\nb4Xa74iihP3HSrAjuxBGU/P0o6TYYDw6LwE+GqWLoyOi/iQ7OxubN2/G22+/bWszm83w9fW95n6x\nsbHQ6XS2S7nqdDrExMR069jXM8OydYaNXFB43CyozrjbzDCVTI0nkx7FX3/MwpXGcvwjdwueSnoU\nwwNjO98ZwAOz42AwWXHo1BV8evgiFHIZ7pnevfcY4H794i7YL47YJ0REdKN67bLeKpUKmzZtwp49\nezBx4kS888472LBhA7RaLVatWoVVq1YBABISErBhwwa89tprGDduHD788ENkZWXB25vTSq/HuUs1\n+PPWHGzbdxZGkxXeagUevWskli8cw2INEd10CQkJyMvLw65duyCKIrKzs5Gdne0wm/LnFixYgDff\nfBNlZWWorKzExo0bkZ6e3uPxts6wUQg86XBf4KvywfKxyxCsDoRZtGBD7macr9F1aV+ZTMCjd43E\nuLgQAMDHB3/CR9/qOIOXiIiI3FavjlDj4+Px/vvvO7SvWbPGbnvq1KkOM2+oe8qrG7H9QCGOFlTY\n2lITB+K+mcPhp+3aFbeIiLorJCQEWVlZeOmll7BmzRpERUXhjTfecLhK4M8tXboUlZWVWLhwIcxm\nM+bPn4+HH364x+NtnWHDy3r3HQFe/vh18uP464/rUWuqwxvH/4mMMQ8hPmh4p/vKZTJkLEjEup0n\nkVtYhV3f6lBvMOO+mcOv60TERERERD2JI9R+Rn/VgD2HLuDr46WwtqybHhrigyUzhyExKsjF0RGR\nJxg/fjx27tx5zccUFBTYbcvlcqxYsQIrVqzoydActF4lipf17ltCtMH4vymZeP3YJlQba7AhdzOW\njfolRg3o/Jw2CrkMT909Clm7T+HYuUp8kXMJ9Y1mPHLXSCjkvTbxmIiIiKhTHJn0E/qrBmz9vADP\nb/we+38sgVWU4O+jwsN3xmP1wxNYrCEicsIkNi+J4gybvidUG4IVKZkYoA6CRbTgHye34OiVE13a\nV6mQ48m7R2HamMEAgEOnr+D1HbloMlp6MmQiIiKibuEItY8rLq/HZz9cxOHTV2wzanw0StwxKQJp\nKUPhpZK7OEIiIvfFGTZ9W7AmCCvGPYHXj/0DVxor8NapbdAbqjErYkanV4CSy2R46I54+HmrsOf7\nC8jT6fGnLTn41T2jMTiY580jIiIi1+MItQ+SJAmnL1Tjs8MXkafT29pbCzW3pQyBWsVfLRFRZ1pn\n2LBg03cFePljRcoTyMp9Gz9dvYhdhXtR0VSFJSN+Abns2l9aCIKAe2fEItDXC+/uO4fLVY3405Yc\nLJuXiOThA3rpFRARERE5xxFqH2KxijiSX47PDl/ExfJ6W3togAa3TwzHlFGDOaOGiKgbOMOmf/BV\n+eDpsY/jf06/j+MVefiu9DD0hmo8lPh/4KPsfLZMWspQDBngjfW78lDXaMbrH+Ri3i2RWDAlmue1\nISIiIpfhCLUPaDJakH28FPtyilFdZ7S1x4T5Ye7ECKSMCIFMxqtbEBF1V+tVoliw6ftUchUeHfUA\ndp3fiy+Lv8YZ/Vn8vyOv47FRDyDSL7zT/eMiAvHHhyZg3c6T+KmsDh8fvIC8Ij2WzU/gEikiIiJy\nCY5Q3Vh1nRH7coqRfbwETUYrAEAAkDx8AOZOisCwIf6drtEnIqKOma086XB/IhNkuGf4PAzyDsW/\nzu6C3lCNV4+ux8IR6ZgaNqnTz8wgPzV+90AK/nd/Ib48egk/ldXhxc1HsDhtGGaOH9pLr4KIiIio\nGUeobqiksgGfHr6AQ6faTiSskMswdfQgzJ4Qzm/6iIhuAlESYZVEAJxh09/cEjYRQ33D8M+T76DK\noMf7BTtRUH0e9424Gz6qa3+GKhVy3D97BJJig/Hm3jOorTfhnc/P4tCpK/j1krEI0HDpMREREfUO\njlDdyNniGnx6+CKOn6+0tflolEhLGYK0lKHw81a5MDoiov7FIrZdwlkh8OOwv4nwHYrnJyzH/5z+\nF/KqzuBYeS7O1xTh/viFGD0godP9R8UEY+2jk7D1swIcyS/H+ZJarPjrAcwcPxTpU2KgVfM9Q0RE\nRD2Low0XEyUJJ85V4pPDF3G+pNbWHuynxpyJ4Zg2JownEiYi6gHmdgUbpZwfh/2RVqlF5piH8E3J\n9/jw/B7UmeqRlfs2UgePx92xd3U628ZHo8QTvxiFqUVV2LbvLMqrm7DvyCUcPFmGuyZHIS1lCFRK\nfkYTERFRz+AI1UUkScKPZyux+1sdLlW0XfEpPNQHd6RGYEJ8KOQyXpmCiKincIaNZxAEAdOH3oL4\noBHYeuZfKKq9gEOXc3Cy4jQWxM7FLWETIROu/Xk7OiYYf358EvYfL8P2L8+iwWDB/+4/j305xZg/\nJQpTRg2GUsHPbCIiIrq5OELtZZIk4fi55kJN+0tzj4wMxB2pEUiMCuKJhImIeoHdDBuew6bfC9UO\nwIqUJ7C/+Fvs0X2OBksj3ivYiYOXj2Dh8PmI8Y+65v4qhRz/5/Y4TBgRjF3f6PDNicuorjNiy6cF\n2P2tDrePD8eM5CFcKkX9Vm5uLp588kl8++23du2iKOLBBx/EqFGj8NxzzwEA7rrrLpSWltoeY7FY\nYDKZ8PXXX2PgwIF2+588eRKLFy+GWq22tWVkZCAzM7MHXw0RUd/AUUUvyiuqwgdfF+FCWZ2tLSEq\nEL+YGoNhQ/1dGBkRkeexm2HDgo1HkAkyzIyYjnEDk7Dz3Mc4Wn4CF64W45Wj6zF6wEjMj5mLIT6D\nr/kcQX5qPDg3HnMmRmDXN0U4cqYctfUmbD9QiH8f/AlTRw/GjOQwDAnx6aVXRdSzJEnCBx98gJdf\nfhlyueMSwLfeegs5OTkYNWqUrW3Pnj2226Io4qGHHsLYsWMdijUAcObMGUyfPh0bN27smRdARNSH\ncYTaCy5V1ON/vzqPPJ3e1jYyMhDpU6MxIjzAhZEREXkuFmw8V4CXPx4ZdT9u0U/EzvMfo6T+Mk5W\nnkFeZT7GDUzC7ZG3dVq4GRSkRWb6KNw9vRGf/1CMb09ehsFkxRdHL+GLo5cwbIg/pieFYVxcCDRe\nfH9R35WVlYVPPvkEmZmZ2LRpk919+fn52LlzJ2bPnt3h/lu2bEF9fT2WL1/u9P7Tp08jPj7+psZM\nRNRfcATRg2rrjfjwGx2+yS2F1Hx1bgwb4o97Z8QgLiLQtcEREXk4Lomi+KDheH7C0zh65QQ+LvoM\nlQY9cq4cR86V40gIisPsyFsxPCDmmkuVBwZq8cs5cUifGo0Dx0vwzYlSVF014nxJLc6X1GLLZwVI\nig3GxISBGB0TBLWK7zXqW+69915kZmbihx9+sGs3mUx47rnnsHbtWmzfvt3pvrW1tVi3bh3efPNN\np7NzgOYZNiqVCmlpaRBFEXfccQdWrFgBlaprV0cVBAHdPe2jTCbY/ST2SUfYL86xX5zriX7hqKEH\nWKwiPvvhIj7+/gKMJisAICRAjUW3DsO4uBCeo4aIyA1whg0BzcukJgwai7Gho/H95Rx8ceEAKg16\nnNYX4LS+AGHegzBtSCpSh4wD0PEyJz9vFRZMica8yVE49ZMeB46VILewChariKNnK3D0bAUUcgFx\n4QEYHTsAY2KDMTBQwzEBub3Q0FCn7a+88gqmTp2KcePGdViweffdd5GUlISkpKQOnz8wMBCTJk3C\nkiVLUFVVhaeffhqvv/46nn322S7FFxzsfd1/jwICrn2lOE/EPnGO/eIc+8W5m9kvHKHeZOcu1WDL\npwUoqWwAAGi9FJh3SxRmjhvKK0gQEbkRMws21I5CpsC0IamYEjYRx8pPYt/FAyiuK0FpQxn+dXYX\ndhXuxS0R45EUNBqxftEdXllKJhMwOiYYo2OC0WAw48eCChw+cwVnLlTDYpVw6qdqnPqpGu9/eQ6h\nARokRAVi2FB/DBsagBB/NQs41Cd8//33OHToUIeFmlY7d+60nYi4I1lZWbbbWq0WGRkZePXVV7tc\nsKmqariuGTYBAd6oqWmAKErd27mfYp84x35xjv3i3I30S1CQ8y+FOEK9SRoMZuw4UIjs481nxBcA\n3Dp2CO6eHgMfjdK1wRERkYPWGTYyQdbpZZ3Jc8gEGcYNTEJK6BgU1v6Eb0sO41j5CRitJuzXHcR+\n3UH4q/wwbmASkkJGIdovAnKZ86Ue3molpiWFYVpSGOqbzMgrqkJuURXyivSobzKjvKYJ5cebcKBl\n7ODvo8LwIf6ICfNHxEAfRAz05RiC3NLevXtx8eJF3HLLLQAAg8EAQRBQVFRkO3lwYWEhKisrMX36\n9A6fp7a2FllZWXjqqafg49P8jxWj0QgvL68uxyJJEqzW63sdoijBauU/NttjnzjHfnGO/eLczewX\nFmxukCRJOJJfjne/OIerDSYAwNAQbzw4Nx6xQ3jlJyIid9U6w4bnryFnBEHAsIBoDAuIxsLh83Gk\n/CiOlp+ArqYYtaar+Kr4G3xV/A28FVokBMdh1ICRSAiKg1apcfp8PholUhMHITVxEERRgu7yVZws\nqsLZ4hoUlV6FySKitt6EnIIK5BRU2PYL9PVCeKgPwkN9MChIi0FBWgwM0rKQQy61du1arF271rb9\n/PPPIzAw0G42zfHjx5GYmHjNc9H4+vpi3759kCQJv/nNb1BaWoqsrCwsXry4R+MnIuorOEq9AfVN\nZmz5rAA5+eUAAJVChvSp0Zg9IRwKOb+tJSJyZ60zbLgcijrjo/LGrMgZWDz2Lpy6WIjDl4/hWHku\nrjRWoMHSiCNXjuHIlWOQCTJE+A61FXpi/aOgVWodnk8mExA7xN/2xY7FKqK4vB7nLtXi/KUa/FRW\nh8paAwCgus6I6jojcgur7GPSKDEwSINgPzWC/dQI8lMjyM/LdttbreDyKnKpkpIShISEOLSXlpbi\nrrvuwp49exAWFoasrCz86U9/QmpqKtRqNZYsWYIHH3zQBRETEbkfQZKkfjWHqaKirtv7yOUCgoJ8\noNfXd3nq0smiKry19wxq65tn1SRGBeKXc+MRGuD8m7W+6Hr6xROwXxyxT5y7kX4JCfHtoaj6r+7m\n/0NlR7D19Hb4e/nhpSm/76Go+h7+fXbOWb+UN1Yir+oM8irP4FxNEURJtNtHgIAwn0GI9otAuO8Q\nhPsOQZj3ICjlnc+OaTSYUVxej4vl9Si+Uo+SynqU6ZvQZLR0ui/Q/CWSn7cKvloV/LRK+GpV8PVW\nwk+rgq9WCa2XElq1AhovBTRecmi9lFB7ySHrZpGH7xdHzP29q7fG/v0d+8Q59otz7BfneiL/82vF\nbrJYRew4UIjPjxQDaB4QLU4bhtvGDuE3WUREfQiXRNGNCtUOQJp2GtLCp6HJ0oSC6kKcrynC+Rod\nLtWVQoKEkvrLKKm/bNtHJsgw2HsghvgMxiBtKAZ6h2KQNgQDNMF2s720aiXiIgIRFxFoa5MkCXWN\nZpTpG3FF34gr1U3Q1xmgrzVA3zITx9pykkOTRURlrcE2U6crBABqLwW0XnKovRRQKeTwUsqgUsqh\nUsrhpWi9LYNXS5taJYe/nwYGgwkyQYBCLoNCJkAul0Ehb96WywUoZD/blssgEwQIQvOMI5kgOGwL\nAji2IiIij8ZRajfU1BuxYVcezl2qBQBED/bFY/MSMDiYlzMjIupruCSKbiaNQoPkkFFIDhkFAGiy\nNKGo9gLO1+hQXFeCi3WX0GBuhCiJDkUcoLmQE6wORLA6CEHqwJY/AbbbAV5+kMvk8PNWwc9bhRHh\nAQ4xiKKE2gYT9HUGVF81oq7RhKuNZlxtNKGuofl2XaMJdY1mNBkttuJOKwlAk9HSMovH2FNd1S2C\ngJbijQCZrN1tJ4Wd1tqO0PJf27atXUDL/wFBgNDuvuYmoeW+1udoe5629rY72/Zv95iWOwYEaLEk\nLRYB3l0/eS4REdHPcZTaRQUXq7Fh9ynbiYVvnxCOhbfG8lw1RER9lIUzbKgHaRQaJAbHIzE4HkDz\n7JgaYy0u1pWguK4Elxuu4EpjOcobK2GVrBAlERVNVahoqnL6fAIE+Ci94avygZ/KF74qH9ttH5UP\ntAoNtAo1tEotAoPUGBLqBy+5V4czVCRJgsksotFoQWNLkabRYLEVbJpMFpjMIkxmK0xmEUaL1Xbb\nZGlpM1thslghSYDJbIXFKsFiFWGxSrBaRdyMSfKSBFglCYAEXOeVgFyhsOQqxsQEYXLiIFeHQkRE\nfRhHqZ2QJAmfHynG9v2FECUJXio5HrlzJCbEh7o6NCIiugFmzrChXiQIAgLVAQhUByApJNHWbhWt\nqDLocaWxAuWNldAbqqE31LT8rEajpQkAIEFCnbkedeZ6lDaUdemYMkEGjUINrUIDtdwLKrkKKrkK\nXi0/VXIVvGTt2rxUUGmU8JYp4CeTQylTQC7IoZApoJCpoBAUkMtatlvaVQoFQoP9UVtrgGRtPqYg\nCJAkCaIk2Yo3lpZLnDYXdJqLOqIoQYIEUQREqWVbkiBKzduSKLW0t2y33mdrlyBJbfc191Pzf5p/\ntrW1nrFRkqS2QlK7x7W22R5nu7N1f8l2f9v+kv3xbL9rYHCoL1Jig7r0eyIiIuoIR6nX0GS0YPMn\n+barQA0O1uKpu0cjbACXQBER9XVcEkXuQC6TI1QbglCt49V0AKDJYoDeUI0aYy2umupRZ6pDnake\nV011tu16UwOaLE2wSPZTUERJRIO5EQ3mxt54KTYCBMiE1nPUyCCDrOUcNc3FnObttrbmdhlk7fZr\nfhbBtgzJfpmT/Xa7rbalTq37Cu3vc3wuATK7JVJtz/bzF2Xf+vPHtN9LEACZOggmaQY0cLxKGBER\nUVdxlNqB0soGvPHhSVyuah7kjI8PxcN3xEPjxS4jIuoPeNJh6gs0CjWG+AzGEJ/B13ycJEkwixY0\nWhrRZDGg0dyEJksTGi1NaDQ3wWA1wmQ1wSSaYLKaYLSaYLKa226LzT/NVhMsohUWyQKLaIVV6v46\nJAkSrJIVHn3hkCtAiDIEkwaNd3UkRETUh/XqKPX06dNYtWoVzp8/j8jISLz44otITk52eFxGRga+\n//57yOVyW9uxY8d6Lc6c/HK8ufcMjCYrZIKAxbfFYvaEcF6pgIioC/bu3Yu///3vKCsrQ1hYGFas\nWIFZs2ahtrYWL7zwAg4dOgRfX1889dRTWLRoEYDmf2y++uqr2L59O6xWK9LT0/G73/3O7nPgZuMM\nG+pPBEGASq6ESu6PAC//m/a8kiTBIllhFS32hRzRAotkhQQrtL4q6GvqYLGKEKXmPxKkltvNPyVJ\nhFUSIUKCJIlt90FsWeoktvvTfM6a5v+h3ZIl+21bq9R2b+tSJrs9JLQ9F6SWZU/tnkP6+X12PeDk\nVuu25OxhEGRAiF8QkkITQUREdCN6bZRqNBqRmZmJzMxMLFq0CLt378YTTzyBL774At7e9kuMTp8+\njW3btmH06NG9FR4AwGCy4N1955B9vBQA4O+tQmZ6ot0lNYmIqGM6nQ4vvPAC3nrrLaSkpODgwYN4\n/PHH8fXXX2P16tXQarU4ePAgCgoKsGzZMgwfPhzJycnYtm0bDhw4gI8++giCICAjIwNvvfUWli1b\n1mOxsmBD1DlBEKAUFB3ORJPLBQQF+UAvq4fVo6fUtLH1iZ59QkREN6bXLnF06NAhyGQyLF26FEql\nEgsXLsSAAQOQnZ1t97iqqiro9XqMGDGit0KD/qoB/9ydh9+u/95WrBk+1B9/fHgCizVERN0QHR2N\n7777DikpKbBYLKisrIS3tzdUKhW++OILLF++HF5eXhgzZgzmzZuHXbt2AQB2796NBx98EKGhoQgJ\nCUFGRgY+/PDDHo2VS6KIiIiIyJ312ihVp9MhNjbWri06OhpFRUV2badPn4a3tzcyMjKQn5+PqKgo\nPPfccxg7dmyXjiMIAmTdLENt2JWHs8W1AAC5TMDd02Nw5+QIyLv7RP2MTCbY/aRm7BdH7BPnPLVf\nvL29UVxcjDlz5kAURaxevRoXL16EQqFAeHi47XHR0dH4/PPPAQBFRUUYNmyY3X06nQ6SJHV5OWp3\n83/rcgYvuQpyuWf9jq7FU9+3nWG/OMd+ccQ+ISKim6XXCjaNjY3QaDR2bWq1GgaDwa7NaDQiOTkZ\nK1euRGRkJHbs2IFly5bhk08+QUiI8ysotBcc7N3tc81MTR4KQZAhOS4EaePCERbi0639+7uAAF4V\nyxn2iyP2iXOe2C+DBw/GiRMnkJOTgyeffBKPPvoo1Gq13WPafwY0NTXZ3a/RaCCKIkwmE7y8vLp0\nzO7m/zlx03DVfBW3x01DUBDz/s954vu2K9gvzrFfHLFPiIjoRvVawUaj0TgUZwwGA7Ra+8sdzpo1\nC7NmzbJtL126FO+99x4OHz6MefPmdXqcqqqGbs+wuS15MO6+dRhqahogihL0+vruPUE/JZMJCAjw\ntvULNWO/OGKfOHcj/dLXCwgKRfPHy+TJk3H77bcjLy8PRqPR7jHtPwPUarXd/U1NTVAoFF0u1gDd\nz/+R6kj8adZK1NQ0MO+3w7/PzrFfnGO/OPLk3E9ERDdXrxVsYmJi8M4779i16XQ6hyLMp59+ClEU\nceedd9rajEZjlwftkiTB2v0rUAIARFHiyeGcYL84x35xxD5xzpP6JTs7G5s3b8bbb79tazObzYiI\niMDXX3+N0tJShIWFAWj+DGhdBhUbGwudToekpCTbfTExMd069vXmf0/6/XQH+8U59otz7BdH7BMi\nIrpRvXaSlsmTJ8NkMmHr1q0wm83YsWMHKisrMXXqVLvHNTY24s9//jPOnz8Ps9mMf/7znzAYDJgy\nZUpvhUpERNcpISEBeXl52LVrF0RRRHZ2NrKzs7FkyRLMnDkTr7zyCpqampCbm4uPP/4Y8+fPBwAs\nWLAAb775JsrKylBZWYmNGzciPT3dxa+GiIiIiMh1em2GjUqlwqZNm7B69Wq8+uqriIyMxIYNG6DV\narFq1SoAwJo1a3DPPfegoqICjz32GGpqapCQkIBNmzY5LJ0iIiL3ExISgqysLLz00ktYs2YNoqKi\n8MYbbyA2NhZr167FH//4R8yYMQNarRYrV660zahZunQpKisrsXDhQpjNZsyfPx8PP/ywi18NERER\nEZHrCJIk9au5mhUVdd3eRy4XEBTkA72+nlNX22G/OMd+ccQ+ce5G+iUkxLeHouq/upv/+b51jv3i\nHPvFOfaLI+b+3sWx/83BPnGO/eIc+8W5nsj/nn3daiIiIiIiIiIiN8SCDRERERERERGRm2HBhoiI\niIiIiIjIzfS7c9gQEREREREREfV1nGFDRERERERERORmWLAhIiIiIiIiInIzLNgQEREREREREbkZ\nFmyIiIiIiIiIiNwMCzZERERERERERG6GBRsiIiIiIiIiIjfDgg0RERERERERkZthwYaIiIiIiIiI\nyM2wYENERERERERE5GY8vmBz+vRpLFy4EMnJyUhPT8fx48ddHZJbyMnJwaJFixPSbP0AAAoUSURB\nVDBu3DjMmjUL77//vqtDchuVlZWYPHky9u/f7+pQ3EJZWRkyMjKQkpKC6dOnY8uWLa4OyS38+OOP\nuOeee5CSkoI5c+bg3//+t6tDonaY+51j7u8Yc7895n7nmPvdH/O/I+b+a2P+t8f876hHc7/kwQwG\ngzRt2jRp27ZtkslkkrZv3y6lpqZK9fX1rg7NpWpqaqQJEyZIH330kWS1WqW8vDxpwoQJ0nfffefq\n0NzC448/LsXHx0tfffWVq0NxOVEUpbvvvlt6+eWXJZPJJJ09e1aaMGGCdPToUVeH5lIWi0VKTU2V\nPvnkE0mSJOnIkSNSQkKCVFxc7OLISJKY+zvC3H9tzP1tmPudY+53f8z/jpj7O8f834b531FP536P\nnmFz6NAhyGQyLF26FEqlEgsXLsSAAQOQnZ3t6tBcqrS0FDNmzMD8+fMhk8mQmJiISZMm4ccff3R1\naC733nvvQaPRYPDgwa4OxS2cOHEC5eXlePbZZ6FUKjF8+HC8//77iI6OdnVoLnX16lXo9XpYrVZI\nkgRBEKBUKiGXy10dGoG5vyPM/R1j7rfH3O8cc7/7Y/53xNx/bcz/9pj/HfV07vfogo1Op0NsbKxd\nW3R0NIqKilwUkXsYOXIk/vKXv9i2a2trkZOTg/j4eBdG5Xo6nQ6bN2/G6tWrXR2K2zh16hSGDx+O\nv/zlL5gyZQrmzJmDEydOIDAw0NWhuVRgYCCWLl2KZ555BomJibj//vvxhz/8gR/2boK53znmfueY\n+x0x9zvH3O/+mP8dMfd3jPnfEfO/o57O/Yqb8ix9VGNjIzQajV2bWq2GwWBwUUTup66uDpmZmUhM\nTERaWpqrw3EZi8WC3/72t/jP//xPBAQEuDoct1FbW4vDhw8jNTUV+/fvR15eHh577DGEh4dj/Pjx\nrg7PZURRhFqtxt/+9jekpaXh4MGD+M1vfoPExEQOgNwAc3/nmPubMfc7x9zvHHO/+2P+vzbm/jbM\n/84x/zvq6dzv0TNsNBqNQ4I2GAzQarUuisi9FBcX47777oO/vz/WrVsHmcxz3y7r16/HyJEjMWPG\nDFeH4lZUKhX8/f2RkZEBlUplO9HWl19+6erQXOrzzz9Hbm4u5s6dC5VKhVtvvRW33nordu3a5erQ\nCMz9nWHub8Pc7xxzv3PM/e6P+b9jzP32mP+dY/531NO536P/JsbExECn09m16XQ6DBs2zEURuY9T\np05h8eLFmDp1KtavXw+1Wu3qkFxq79692LNnD8aPH4/x48ejtLQUzzzzDP7xj3+4OjSXio6OhtVq\nhdVqtbW1rt/0ZJcvX4bJZLJrUygUPI+Bm2Du7xhzvz3mfueY+51j7nd/zP/OMfc7Yv53jvnfUY/n\n/pty6uI+ymg0SlOnTpW2bNlid6b4hoYGV4fmUhUVFVJqaqq0ceNGV4fitm677TaeKV6SpKamJmna\ntGnSa6+9JpnNZuno0aNScnKydOzYMVeH5lL5+flSYmKitGPHDkkURenw4cPS2LFjpdzcXFeHRhJz\nf0eY+zvH3N+Mud855n73x/zviLm/a5j/mzH/O+rp3C9IkgeXwwDk5+dj9erVKCgoQGRkJFavXo3k\n5GRXh+VSWVlZ+Otf/+owPfQ//uM/sGLFChdF5V7S0tLwhz/8AbfddpurQ3G5CxcuYM2aNTh58iR8\nfHzw1FNP4d5773V1WC731Vdf4W9/+xuKi4sRFhaGp59+GrNnz3Z1WNSCud8Rc3/nmPvbMPc7x9zv\n/pj/7TH3dw3zfxvmf0c9mfs9vmBDRERERERERORuPPocNkRERERERERE7ogFGyIiIiIiIiIiN8OC\nDRERERERERGRm2HBhoiIiIiIiIjIzbBgQ0RERERERETkZliwISIiIiIiIiJyMyzYUJ/y/PPPIy4u\nrsM/f//733H48GHExcXBaDT2eDyXLl2yHXv58uXXvX9hYWGnj3322Wdtx+rK44mI+gvmfuZ+IvJM\nzP/M/55OkCRJcnUQRF1VV1cHg8EAADh27Bh+/etf46uvvoJKpQIAaLVaKJVK1NbWIiQkpMfjuXTp\nEmbOnIlt27ZhxIgR8PPz69b+VqsVer0eQUFBkMvl13xsXV0ddDodFi1ahL179yI2NvZGQici6jOY\n+5n7icgzMf8z/3s6hasDIOoOX19f+Pr6AgD8/f0BAAMGDICXl5fd43ojYbcXGBjY7YQNAHK5vMux\n+vr6IigoqNvHICLq65j7mfuJyDMx/zP/ezouiaJ+5+fTIuPi4rB3717Mnz8fY8aMwSOPPILLly9j\n5cqVSE5Oxu23345Dhw7Z9r9y5QqWL1+OsWPHYtq0aVi9ejUaGhq6dOzWaY4HDhzA7NmzkZSUhBUr\nVqC0tBSPP/44kpKSkJ6ejvz8fLvHt05zTEtLw9atW/HAAw9g9OjRmDNnDrKzs29yDxER9T/M/URE\nnon5n/ozFmzII7zyyitYtWoV3nnnHZw6dQoLFizAyJEj8cEHH2DYsGF48cUXAQCSJOFXv/oVlEol\ntm/fjnXr1iE/Px8vvPBCt473+uuv47XXXsP69euxb98+3Hvvvbjzzjuxfft2qNVq/Pd///c19126\ndCn27NmDuLg4vPDCCzCbzTf0+omIPBFzPxGRZ2L+p/6CS6LII/zyl7/EhAkTAACpqakoLi7GI488\nAgBYsmQJMjMzYbVa8cMPP0Cn0+Hdd9+FUqkEAPzXf/0X5s6di7KyMgwaNKhLx3viiSeQmJgIAIiP\nj0dYWBh+8YtfAADS09OxadOmDvedP38+7rzzTgDAk08+ifT0dJSVlSE8PPz6XjwRkYdi7ici8kzM\n/9RfsGBDHiEiIsJ2W6PR2CVAtVoNURRhsVhQWFiI+vp6TJw40eE5dDpdl5P2z483dOhQu+OZTKYO\n942KirLd9vHxAQBW2YmIrgNzPxGRZ2L+p/6CBRvyCAqF/VtdJnO+GtBisSAiIsJpFbw7JzPr6vGc\naa3ut8eLuRERdR9zPxGRZ2L+p/6C57Ahaic2NhZlZWXw9fVFZGQkIiMjYbFY8PLLL6O+vt7V4RER\nUQ9g7ici8kzM/+TuWLAhamfKlCmIjY3FM888g1OnTiEvLw8rV65EdXU1QkNDXR0eERH1AOZ+IiLP\nxPxP7o4FG6J2ZDIZ1q9fDx8fHzzwwAN45JFHEBUVhXXr1rk6NCIi6iHM/UREnon5n9ydIHGBHNF1\nu3TpEmbOnIm9e/ciNja23x2PiIgcMfcTEXkm5n/qbZxhQ3QTVFdX4+rVqz16jLq6Ouj1+h49BhER\ndR1zPxGRZ2L+p97Cgg3RTXD//ffj97//fY8e48UXX8SiRYt69BhERNR1zP1ERJ6J+Z96C5dEERER\nERERERG5Gc6wISIiIiIiIiJyMyzYEBERERERERG5GRZsiIiIiIiIiIjcDAs2RERERERERERuhgUb\nIiIiIiIiIiI38/8BIsntY+5UdXEAAAAASUVORK5CYII=\n",
"text/plain": [
""
]
},
"metadata": {},
"output_type": "display_data"
}
],
"source": [
"# set initial conditions and cooling flow\n",
"IC = [C0,T0,Tcf]\n",
"\n",
"# do simulation at fixed time steps dt\n",
"dt = 0.05\n",
"ti = 0.0\n",
"tf = 8.0\n",
"\n",
"# create python list to log results\n",
"log = []\n",
"\n",
"# start simulation\n",
"c,T,Tc = IC\n",
"qc = 153.8\n",
"for t in np.linspace(ti,tf,int((tf-ti)/dt)+1):\n",
" log.append([t,c,T,Tc,qc]) # log data for later plotting\n",
" c,T,Tc = odeint(deriv,[c,T,Tc],[t,t+dt])[-1] # start at t, find state at t + dt\n",
"\n",
"def qplot(log):\n",
" log = np.asarray(log).T\n",
" plt.figure(figsize=(16,4))\n",
" plt.subplot(1,3,1)\n",
" plt.plot(log[0],log[1])\n",
" plt.title('Concentration')\n",
" plt.ylabel('moles/liter')\n",
" plt.xlabel('Time [min]')\n",
"\n",
" plt.subplot(1,3,2)\n",
" plt.plot(log[0],log[2],log[0],log[3])\n",
" if 'Tsp' in globals():\n",
" plt.plot(plt.xlim(),[Tsp,Tsp],'r:')\n",
" plt.title('Temperature')\n",
" plt.ylabel('Kelvin')\n",
" plt.xlabel('Time [min]')\n",
" plt.legend(['Reactor','Cooling Jacket'])\n",
"\n",
" plt.subplot(1,3,3)\n",
" plt.plot(log[0],log[4])\n",
" plt.title('Cooling Water Flowrate')\n",
" plt.ylabel('liters/min')\n",
" plt.xlabel('Time [min]')\n",
" plt.tight_layout()\n",
"\n",
"SS = log[-1]\n",
"qplot(log)\n"
]
},
{
"cell_type": "markdown",
"metadata": {},
"source": [
"## Simulation 3. Proportional-Integral-Derivative Control\n",
"\n",
"Proportional-Integral-Derivative (PID) control is the workhorse of the process control industry. In standard form, the PID algorithm would be written\n",
"\n",
"$$q_c(t) = \\bar{q}_c - K_c\\left[(T_{sp}-T) + \\frac{1}{\\tau_I}\\int_0^t (T_{sp}-T)dt' + \\tau_D\\frac{d(T_{sp}-T)}{dt} \\right]$$\n",
"\n",
"For the control reactor temperature, note the controller is 'direct-acting' such that a positive excursion of the reactor temperature $T$ above the setpoint $T_{sp}$ is compensated by an increase in coolant flow, and vice-versa. Thus a negative sign appears before the term $K_c$ contrary to the usual textbook convention for negative feedback control.\n",
"\n",
"The practical implementation of PID control is generally facilitated by a number of modifications."
]
},
{
"cell_type": "markdown",
"metadata": {},
"source": [
"### Independent Parameters\n",
"\n",
"A common practice is to introduce an independent parameterization for each fo the P, I, and D terms. Rewriting, the control equation becomes\n",
"\n",
"$$q_c(t) = \\bar{q}_c - \\left[k_P(T_{sp}-T) + k_I\\int_0^t (T_{sp}-T)dt' + k_D\\frac{d(T_{sp}-T)}{dt} \\right]$$\n",
"\n",
"where\n",
"\n",
"\\begin{align*}\n",
"k_P & = K_c \\\\\n",
"k_I & = \\frac{K_c}{\\tau_I} \\\\\n",
"k_D & = K_c\\tau_D\n",
"\\end{align*}"
]
},
{
"cell_type": "markdown",
"metadata": {},
"source": [
"### Setpoint Weighting\n",
"\n",
"Step changes in setpoint $T_{sp}$ can produce undesired 'kicks' and 'bumps' if PID control is implemented directly using in standard form. It is common practice to introduce setpoint weighting factors for the proportional and derivative terms. This can be written as \n",
"\n",
"$$q_c(t) = \\bar{q}_c - \\left[k_Pe_P(t) + k_I\\int_0^t e_I(t')dt' + k_D\\frac{e_D(t)}{dt} \\right]$$\n",
"\n",
"where\n",
"\n",
"\\begin{align*}\n",
"e_P(t) & = \\beta T_{sp}(t) - T(t) \\\\\n",
"e_I(t) & = T_{sp}(t) - T(t) \\\\\n",
"e_D(t) & = \\gamma T_{sp}(t) - T(t)\n",
"\\end{align*}\n",
"\n",
"Common practice is to set $\\gamma = 0$ which eliminates derivative action based on change in the setpoint. This feature is sometimes called 'derivative on output'. This almost always a good idea in process control since it avoids the 'derivative kick' associated with a change in setpoint.\n",
"\n",
"In practice, the term $\\beta$ is generally tuned to meet the specific application requirements. In this case, where setpoint tracking is not a high priority, setting $\\beta = 0$ is a reasonable starting point."
]
},
{
"cell_type": "markdown",
"metadata": {},
"source": [
"### Discrete Time Implementation\n",
"\n",
"The simulation strategy adopted here requires a discrete time implementation of PID control. For a sampling time $dt$, the PID algorithm becomes\n",
"\n",
"$$q_c(t_k) = \\bar{q}_c - \\left[k_Pe_P(t_k) + k_Idt\\sum_0^{t_k} e_I(t_{k'}) + k_D\\frac{e_D(t_k)-e_D(t_{k-1})}{dt} \\right]$$\n",
"\n",
"Implementation is further streamlined by computing changes is $q_c(t_k)$\n",
"\n",
"$$\\Delta q_c(t_k) = q_c(t_k) - q_c(t_{k-1})$$\n",
"\n",
"Computing the differences\n",
"\n",
"$$\\Delta q_c(t_k) = -\\left[k_P(e_P(t_k)-e_P(t_{k-1})) + k_I\\ dt\\ e_I(t_k) + k_D\\frac{e_D(t_k) - 2e_D(t_{k-1}) + e_D(t_{k-2})}{dt}\\right]$$"
]
},
{
"cell_type": "markdown",
"metadata": {},
"source": [
"### Bounded Control\n",
"\n",
"A final consideration is that the coolant flows have lower and upper bounds of practical operation. \n",
"\n",
"$$q_c = \\max(q_{c,min},\\max(q_{c,max},q_c)) $$"
]
},
{
"cell_type": "code",
"execution_count": 6,
"metadata": {
"scrolled": false
},
"outputs": [
{
"data": {
"image/png": "iVBORw0KGgoAAAANSUhEUgAABGwAAAEMCAYAAACRPCChAAAABHNCSVQICAgIfAhkiAAAAAlwSFlz\nAAALEgAACxIB0t1+/AAAIABJREFUeJzs3XdcleX7wPHPGewpDsSNirgFB0iCppmlVqammZV+zYHm\nyL1wz8xyoqLmzvqq/Sqrr6aZOco9KhU3uAcge3M45/fHkSeODAERUK/36+WLc57zjPtcyH3ucz33\nUBkMBgNCCCGEEEIIIYQQosRQF3cBhBBCCCGEEEIIIYQpSdgIIYQQQgghhBBClDCSsBFCCCGEEEII\nIYQoYSRhI4QQQgghhBBCCFHCSMJGCCGEEEIIIYQQooSRhI0QT1FhLsImC7oJIcTzQ+p0IYQo2aSe\nzp7EpWhJwkaUKD/99BMffPABzZo1w8vLiw8//JD9+/cXd7EK5MSJE4wZM+aJzxMTE8Pw4cMJDQ0F\n4NatW7i7u3PgwIEnPrcQQhSX8ePH4+7unuu/o0ePFncxn4pFixaxffv24i6GEOI5V1Tt6qNHj+Lu\n7s7Vq1cB+PDDDxkxYkShXyfD+fPncXd3Z8+ePSbb7969i7u7O82bN8+SVJg/fz5NmzYlPT09T9f4\n9ddfmTNnTqGVObPcPveWL18OPP0YFtSSJUv47rvvirsYLxRtcRdACDBmaseNG8fu3bt5//33GTBg\nAOnp6fz8888MGDCA6dOn06NHj+IuZr58++233Lhx44nPc+HCBXbu3MnQoUMBKFeuHFu2bKFGjRpP\nfG4hhCguH3/8sUm97u/vT9u2benWrZuyrWbNmsVRtKdKp9OxYsUKZs2aVdxFEUI8p4q7XT116lTM\nzMye2vnd3d2xt7fnn3/+oW3btsr2w4cP4+DgQFRUFOfPn6du3brKa6dOnaJZs2ZoNJo8XWPjxo2U\nKVOm0MueoV+/frz66qtZtru4uDy1axaGZcuWMW3atOIuxgtFEjaiRNiyZQs//fQT69ato3nz5sr2\n1q1bY25uzpw5c2jXrh1OTk7FWMqSwdzcHA8Pj+IuhhBCPJEqVapQpUoV5blWq6V8+fJSvwkhxBMq\n7nb10062q9VqmjRpwl9//WWy/fDhw7Ru3Zrjx49z6NAhJWGTmprKuXPnGDVq1FMtV35UqlRJPu9E\nnsiQKFEibNiwgbZt25p8qGQYMmQIPXr0ICEhAYAdO3bQuXNnGjVqxCuvvMLq1atNuj26u7uzfft2\nhgwZgoeHBy1atCAwMNDknJGRkYwZMwZvb2+aNWvGsGHDuH//vvL6/fv3GT58OE2aNKFJkyaMGTOG\nqKgo5fXx48czevRoVq1ahZ+fHw0bNsTf3185x/jx4/n+++/5+++/cXd359atWyxdupT33nuPqVOn\n4unpSf/+/QG4cuUKgwcPxsvLi/r16/Paa6+xbds2wNjFtFevXgB06NCBpUuXZjsk6vDhw/To0QNP\nT0/8/PyYP38+qampyutt2rRh/fr1TJgwgSZNmuDl5cXMmTNJS0sr2C9MCCGKyL179xg2bBiNGzem\nadOmjBs3jpiYGOX10aNHM27cOAIDA3nppZfw9PRkypQpxMfHM3XqVBo3bkyrVq2UejXjmBEjRrB4\n8WK8vLzw9vZm5syZJvWmwWBg5cqVtGnThgYNGtC1a1eOHz+uvH7o0CHc3d3ZsmULzZs3p2XLliQm\nJhIdHc3UqVNp2bIl9erVw8/Pj3nz5qHT6dDpdNSrVw+ASZMm8Z///AeAli1bsnDhQpP3PWzYMOX1\n69ev4+7uzqZNm/Dz88PLy4ubN28CsG3bNl577TXq169Px44d2b17d6HGXwjx7MlPuxoe37ZOTU0l\nMDCQdu3a0bBhQzp37szvv/+e4/UzD+fJGC516tQp3nnnHRo0aMBrr72WZTjTn3/+SefOnWnYsCFd\nunTh119/VdrQ2fH29ubs2bPo9Xpl25EjR2jWrBne3t4cPnxY2R4cHExKSgre3t6AsX7/8ssv6dCh\nA/Xr16dp06YMGTJEacd/+OGHHDt2jB07duDu7q6c5/Tp07z33ns0bNgQPz8/li5danL9Nm3asHDh\nQiWWhTn0NSIigvHjx+Pr64uHhwf9+/dXhqBlxCoiIkLZf/DgwXh6eqLT6ZRtnTt3ZuHChcrv5L//\n/S8+Pj74+voSHx9PbGws06dPp2XLltSvXx9fX1/mzJmjfF/IiMW0adP48MMPlfP+3//9H6+//jr1\n69enQ4cO7Ny5s9Det5CEjSgB7t+/T0hICH5+ftm+XrFiRSZOnEjlypX56quvGDlyJF5eXixbtozO\nnTuzePFi5s+fb3LMrFmzqFKlCitWrFASHRljdnU6Hf/5z384deoUAQEBfPbZZ4SGhjJo0CAAEhMT\n6dWrF5cuXWL27NnMnDmT06dP4+/vbzLu9ffff2fv3r1Mnz6d2bNnc/r0aebOnQsYu/q3atUKNzc3\ntmzZQrly5QD4559/uHPnDsuXL+ejjz4iPj6eXr16kZKSwueff87y5cupXr06kydPJjQ0lHr16jFl\nyhQAFi5caDJUIMPevXvp06cPVatWZcmSJfTr14+vv/46y/w5S5cuRaVSsXTpUvr27ctXX31l8gVG\nCCFKmvj4eD744ANCQkKYO3cu06dP58SJE/j7+5s0kn/99VeOHz/OZ599Rv/+/dmyZQtdunQhPT2d\nZcuWUb9+faZPn87du3eVYw4cOMCuXbuYM2cOw4YN4//+7/+YPn268vr8+fNZtmwZPXr0YNmyZVSt\nWpWPPvqICxcumJRx48aNzJs3j4kTJ2Jtbc2IESM4evQoEyZM4Msvv6RTp06sXbuW7du3o9Vq+frr\nrwHjELDJkyfnKx6rVq1iypQpBAQEULlyZTZv3syUKVNo164dK1asoHnz5nzyySfP7NxvQognl592\nNZCntvXo0aNZu3YtH3zwAYGBgdSsWZNBgwblmrR51OjRo3n77bdZuXIlFSpUYOTIkURHRwPG4f/+\n/v5UrVqVwMBA/Pz8GD16dK7na9asGQkJCVy5cgWAq1evEhYWho+PD97e3pw8eVJJwp8+fRonJycl\n4bB69WoCAwN5//33Wbt2LSNHjuTIkSN89tlngHFIV926dXnppZfYsmWLUsbevXvj6OjI0qVL6d+/\nP2vWrMnyHWTNmjV06tSJzz//PNuEWQa9Xq8k8jP+Zf5cyyw+Pp4ePXpw+vRpJkyYwOeff05UVBQ9\ne/bk7t27+Pj4oNVqOXbsGGBMSJ04cYLExETOnz8PGG9Wnz9/3uT/xfr16/n0008JCAjA1taWkSNH\ncujQISZMmMDq1avp0qULGzZs4PvvvwdQYtGvXz+mTp2qbJs0aRJt2rRhxYoVtGjRghEjRrB3795c\nf38i72RIlCh2Gdnsx43ZTE9PZ+nSpbzzzjtMmDABAF9fX1QqFStWrKBfv35K105fX1/Gjh0LQPPm\nzfnll184cOAArVq1Yt++fVy8eJHt27dTu3ZtAJydnRk6dCg3b97kwIED3L59m127dlGxYkUA6tat\nS/v27fn999+VsbKpqamsWrUKe3t7AC5fvsymTZsAY1d/JycnoqOjTbo76nQ6AgICqFatGgBnzpzB\n1dWVhQsXYmdnB0CjRo3w8vLi1KlTdO3aVelW6u7uTvny5bPcaViyZAk+Pj7MmzcPAD8/PxwcHBg3\nbhznz5+nTp06ANSoUUOZPO2ll17i999/58CBA/Ts2TOPvykhhCha3333HWFhYezevZvy5csDULt2\nbTp27Mj+/ftp3bo1YKxbly5dir29Pb6+vmzduhWNRsPMmTNRqVRUqVKFPXv2cP78eeWzJjk5mZUr\nVypfWnQ6HZ9++qnSZX7jxo2MGDGCvn37Asa69b333mP58uUsWbJEKeNHH31Eq1atAGPC32AwMGvW\nLJo2bQqAj48Pv//+OydPnqRr1640atQIgMqVK+d7LrJu3bopcx7odDoCAwN57733lDL7+fkRFRXF\n4sWLlTIJIV4seW1XQ97a1mFhYezatYv58+fz1ltvAcZegWFhYSxatEiphx+nf//+vPfee0rZXn/9\ndY4ePcprr73G6tWrqVGjBgsXLkSlUtGyZUvi4+P56quvcjxfnTp1sLOz4++//6ZWrVocPnyYKlWq\nULFiRbRaLUlJSZw+fRpvb29Onz6Nl5cXKpUKgLCwMIYNG8b7778PgJeXFyEhIfz222+AcUiXra0t\njo6OSjt+xYoVVK5cmcDAQGUeHCsrK6ZPn06/fv0oXbo0APXr11d6R+ZmxowZzJgxw2Rbhw4dsvS2\nBGMPlnv37vHLL79QqVIlwNjD6JVXXmHt2rUEBATQqFEjjh8/TocOHbh8+TJJSUm4urpy6tQpGjRo\nwOHDh7G1tcXDw4OTJ08Cpp9fycnJpKenM336dCXR5OPjw759+zh58iTdu3dXYlGpUiVq1qyJXq9n\nyZIldOvWTfne5efnR3R0NIsXL6ZNmzaPjYN4POlhI4pdRqWXU1Y5Q0hICNHR0bz++usm2zt06EBa\nWhp///23si2jQQygUqlwdnYmMTERMGbZy5cvryRrwJiQ+e2336hcuTLHjx/Hzc0NZ2dnJeNdqVIl\nqlSpwpEjR5RjXF1dlWQNGJM+SUlJub4HrVarfDkAaNCgAZs3b8bCwoKLFy+ya9cuVq1aBZCn4UoJ\nCQlcuHAh25ioVCqlQgZo2LChyT6ZYyKEECXR8ePHqVWrFmXKlFHq46pVq1KpUiWTFaSqVatmUh87\nOTlRt25dpXHu6OgIQFxcnLJPw4YNTerjV155Bb1ez19//cVff/1FWloafn5+ynXT09Px9fXNsnKV\nq6ur8tja2pr169fTpEkTbty4wf79+1m5ciWRkZGFMgQ187WuXr1KZGSkSRl1Oh1+fn4EBwebvFch\nxIsjr+1qyFvb+uTJk6hUqmz3uXDhAvHx8XkqV+a2eUYCPqPdfOzYMVq3bq3U2UCW6z1Ko9HQpEkT\npf1/+PBhJdHg7OyMq6urUl9nJG4yTJo0iY8++oiIiAiOHj3K5s2bOXXqVK719PHjx2nRogUGg8Gk\nvk1LS+PUqVPKfpnr6dz4+/vz7bffmvwbOXJktvuePHmS+vXrK8kaADs7O/z8/JS2fosWLZRhu8eP\nH6dhw4Y0btxYKduhQ4d46aWX0Gr/7a+RuayWlpasW7cOb29v5fNr1apVPHjwIMe4hIaGEhERQcuW\nLbN8Dl24cEHpQSWejPSwEcUu4w5A5q7qj7p3754yZ8GjM7ZnZLQzf2BYWFiY7KNWq5WxuDExMblO\nshYdHU1wcLAyz0BmmZM8lpaWOV4jJ46Ojllmpw8MDGTNmjUkJSVRuXJlvLy8AB57LjB++TAYDEoM\nMpibm2Nra2sSk4KUVwghilN0dDRnzpzJtj4ODw9XHtvY2GR5/dE671Fly5Y1eZ7xuRAbG6t80Xnz\nzTezPTbzXDeP1r+7du1i7ty53L17l7Jly+Lh4YGlpWWh1LeZr5XREB44cGC2+0ZERCg9N4UQL468\ntqvLly+fp7Z1TEwMdnZ2mJubZ7tP5rlwcpO5ba5WG/sMZNS1UVFRWdrmj9at2WnWrBnbt29Hr9dz\n7Ngxk2GtzZs35+TJk9y+fZuwsDCT4UlXrlwhICCAv/76CxsbG+rVq4eFhUWu9XR0dDQbNmxgw4YN\nWV4LCwvLV7nB+Htq0KBBnvaNjY3N9rxOTk6cPXsWQJlTJzIykuPHj9OkSROqVKnCokWLAGPC5uOP\nP85yfGZ79uxh9uzZ3LlzhzJlyuDh4ZFrXDLm9xw8eHC2r0dERCg3TETBScJGFDsnJydq167NoUOH\nsl1i8Pbt27Rp00aZyyXzhFqZnzs4OOTpenZ2diYTCGfYv38/DRo0wN7eHk9PTwICArLsk9dr5NUP\nP/zA8uXLmT17Nu3atcPGxobk5GS+/fbbPB1va2uLSqXiwYMHJttTUlKIi4sr9PIKIURRsrOzo0mT\nJkpX/cyetBGYeeJiQKlHS5UqpdxNXL9+Pba2tlmOzXyHMrOrV68yatQoPvzwQ/r27at8CerSpUuu\nZVGpVFnuhj/uS1BGj6J58+ZlO7SqQoUKuR4vhHg+5addnXGTMLe2dWxsLHFxcaSmppokbfLb/s5N\nuXLlsrRlIyMjH3ucl5cXCxYs4NSpU8TFxZkkZZo3b8727ds5deoU5cqVo3r16oAxSTRo0CDKly/P\nzp07cXV1RaVSMX/+fG7cuJHjtezs7HjjjTd4++23s7z2tOtbe3t7k6RQhgcPHijxr1+/Pg4ODhw7\ndoyTJ0/SpUsXqlatSlhYGH/88Qd37tzJcV4jgGvXrjF8+HB69uxJ//79lZsa2c2fmblcAHPnzsXN\nzS3L65l7BImCkyFRokT44IMPlEkjH7V06VLMzMxo27Ytjo6O/PLLLyav79y5E41Gk2XIT04aNWrE\n3bt3uXjxorLt0qVLDBgwgJCQEDw9Pbl+/Tqurq40aNCABg0a4ObmxtKlSzlz5kye31PG3YPcnD59\nmmrVqtG5c2flDvGff/4J/HvX4dEeOZnZ2tri7u6ebUwAPD0981xeIYQoaTw9Pblx4wbVq1dX6uMa\nNWqwZMkS5a5iQf3zzz8myfvffvsNMzMzPD09adiwIRqNhpiYGOW6DRo04MCBA2zbti3H+v3cuXOk\npaXh7++vJGvCwsK4fPmycocyu2Otra25d++e8jwlJYVz587lWv4aNWpgZ2fHgwcPTMp4/vx5Vq9e\nbTK0QAjxYslru7p69eqPbVs3btwYg8GQ7T516tR5bG/GvGjSpEmWCYz37dv32OPq1auHpaUlX3/9\nNW5ubiY9Rry9vUlKSmLHjh0mw6EiIyO5ceMG7733HtWrV1cS5ocPHzZJnD9aV3t6enLt2jWT+lar\n1bJo0aIsyabC1rhxY86ePWsyj2V8fDx//PGHMq+MWq3Gx8eH77//noiICBo3bkzVqlUpW7YsS5cu\nxc3NTRmKlp3g4GDl8ysjWRMREcGlS5dyjEvG/5+IiAiTuFy8eJFVq1bl6buQeDzpYSNKhK5du/Lb\nb7/Rv39/evXqhbe3NwkJCXz//ffs3buXWbNm4ezszMcff8zcuXOxsbGhZcuW/PXXX6xYsYIPP/ww\nz3db27RpQ61atRg6dCjDhw/HwsKCRYsW0bhxYxo3bkytWrXYsGED/fr1o2/fvpibm7N27Vr++ecf\nxo8fn+f3ZG9vz40bNzh8+HCOiZP69euzZcsWVq1ahYeHB8HBwQQGBqJSqZRxvRld2n/77bcsQ70A\nhg4dyuDBgxk3bhxvvPEGoaGhLFq0iFdffdVkCJcQQjxrunXrxsaNG5X62MzMjC+//JLg4GAmTZr0\nROdOSkpi0KBBDBw4kBs3brBo0SI+/PBD7O3tsbe3p1u3bkydOpWwsDBq1arFkSNHCAoKUib4zU7d\nunVRq9V8+umnvP3229y7d4+goCDS0tKUOcPUajW2trYcOXKEBg0aULt2bXx9fdmyZQteXl64uLiw\nbt26xw6hMjc3Z8CAASxZsoTU1FQ8PT05f/48CxYsoGvXrlmGLwghXhx5bVcDj21bOzo60rZtW6ZN\nm0Z0dDSurq78/PPPHD16lMDAwEIp74ABA+jcuTMjR46kc+fOnDt3jo0bNwK53wDVaDQ0btyY3bt3\nZ1lEo1SpUri7u3Pw4EGmTZumbC9dujQuLi6sWbMGGxsb9Ho933zzDcHBwSbJJ3t7ey5evMjRo0fx\n8vJi4MCBvP/++0yYMIEOHToQExPDwoULsbKyyvO8NQXVtWtXNm7cSN++ffnkk08wNzdn1apV6PV6\nevfurezn6+tLQEAAdevWVXqHNm3alJ07d/LRRx/leo3atWuj0Wj49NNP6dq1K2FhYQQFBZGSkmIy\nR6e9vT3Hjh3D09OT2rVr4+/vz6JFi0hLS6NJkyZcuHCBhQsX8tZbb8nnUCGRhI0oEdRqNYGBgXz1\n1Vf88MMPbN68Ga1Wi7u7O2vXrqVFixYA9O7dGwsLC9atW8fmzZtxcXFhxIgRj62EMstIwMydO5fJ\nkyej1Wpp2bIlEyZMQK1WY29vz6ZNm/jss8+UBE39+vXZsGGD0p0yL7p3786ePXsYMGBAtuNdwVgB\nh4SEsGHDBlasWEGVKlWYOHEiP/74ozKJmpubGx07dmTJkiXcu3cvy3tt27YtgYGBBAYG8r///Y/S\npUvzwQcfMGTIkDyXVQghSiJHR0e+/vprPv30U8aOHYtKpaJBgwasX7+eqlWrPtG5PTw88Pb2ZtSo\nUVhZWdGvXz+T8f2TJ0+mTJkyrFu3jvDwcGUp3F69euV4zpo1azJ79myWL1/Ojh07cHZ2pkOHDhgM\nBr777jv0ej1qtZrBgwezdOlSrl69yg8//MDQoUOJjIxkzpw5WFpa0qNHD9zd3QkODs71PQwYMABr\na2s2bdrEsmXLKFeuHP37988yT4EQ4sWS13Y15K1t/cUXX7Bo0SJWrVpFbGwstWrVYsWKFXleIepx\n3NzcWLZsGfPnz2fQoEG4u7szcuRI5s6di5WVVa7Henl5cfDgwWyX0Pbx8eHChQsmr6lUKpYsWcLM\nmTMZNmwY9vb2NGvWjAULFjBixAguXryIu7s7vXv3Zvjw4QwYMIBdu3bh4eHBmjVrWLRoEYMHD8bG\nxgZfX1/GjBmDmZlZocQhJ3Z2dmzatIl58+YxadIkVCoVTZs2ZcuWLcqKtmBM2ICxx1KGZs2asXPn\nzlyHQ4Gxt8ycOXNYtmwZu3btwtnZmddff5127dqxZcsW0tPT0Wg0DBo0iMWLF3PlyhV++uknPvro\nIywtLZXvMuXKlaNPnz45zmsj8k9lkFlHhRBCCPECGT16NLdv3+abb74p7qIIIcQL788//8Te3t5k\nEt4tW7bwxRdfcPToURniKV5o0sNGCCGEEEIIIUSxOH36NJs2bWLs2LFUqVKF0NBQFixYQPfu3SVZ\nI154krARQgghhBBCCFEs/P39SUhIIDAwkPDwcJydnfnPf/7DgAEDirtoQhQ7GRIlhBBCCCGEEEII\nUcLIWltCCCGEEEIIIYQQJYwkbIQQQgghhBBCCCFKmOduDpvw8LgCHadSqShd2oYHDxKQUWKmJDY5\nk9jkTuKTsyeJTdmydk+pVM+Wgtb3+SX/jyUGIDEAiQEUfQykvjcqSH0v/19zJrHJncQnZxKbnD1p\nbHKq76WHzUNqtTHIaolIFhKbnElscifxyZnE5tkhvyuJAUgMQGIAEoNnifyuciaxyZ3EJ2cSm5w9\nrdhIqIUQQgghhBBCCCFKGEnYCCGEEEIIIYQQQpQwkrARQghRKCIiIvDx8eH3338H4MyZM9SpUwdP\nT0/lX1BQEAAGg4EvvviC5s2b06xZM2bNmkV6enpxFl8IIYQQQogSRRI2QgghCkVAQADR0dHK8/Pn\nz9OyZUtOnz6t/Bs4cCAAmzdvZt++ffz444/s2LGDU6dOsXbt2uIquhBCiDzYsWMH7du3x9PTk44d\nO7Jnzx4AYmJiGDx4ME2aNOHll19m27ZtyjGSoBdCiIJ77laJEkIIUfS++eYbrKyscHFxUbYFBwdT\nu3btbPffvn07vXv3ply5cgD4+/uzePFi+vfvn+drFtWkd2q1yuTni0hiIDEAiQG82DEIDQ1l4sSJ\nrF27lsaNG3Po0CEGDBjAgQMHmDZtGtbW1hw6dIiLFy/Sv39/3Nzc8PDwMEnQq1Qq/P39Wbt2bb7q\neyGEeFEVacImODiYKVOmcOXKFapWrcr06dPx8PDIsp+/vz+HDx9Go9Eo206fPl1k5dQbDFy7G8c/\nVyPQG8C1vB31qzthptU8/mAhhHjBhIaGsm7dOrZu3UqXLl2U7efPn8fc3Jw2bdqg1+tp3749I0aM\nwNzcnJCQEGrWrKns6+rqSmhoKAaDAZUqb1+ESpe2yfO+hcHR0aZAx12/G8vBv27zVssa2NuYF3Kp\nilZBY/A8kRhIDODFjIGrqyt//vknNjY26HQ6IiIisLGxwdzcnD179rBr1y4sLCxo2LAhb7zxBj/8\n8AMeHh6FkqAvSqcvhfP31QfKc41GRcuGFahaXpZYF0IUvSJL2KSkpDBw4EAGDhxIt27d2L59O4MG\nDWLPnj3Y2Jh+6AUHB7N582YaNGhQVMUDQK83cDT4Pt8fCOHug0ST1yqVtWX8+55YW5oVaZmEEKIk\n0+l0jB07loCAABwdHU1eK1WqFN7e3rz77rs8ePCATz75hCVLljB69GiSkpKwtLRU9rWyskKv15Oa\nmoqFhUWerv3gQUKR9bBxdLQhOjoBvd6Q7+OnrDxEZFwK50IiGPOe51Mo4dP3pDF4HkgMJAZQ9DFw\ncrJ96tfIDxsbG27evMlrr72GXq9n2rRp3LhxA61WS+XKlZX9XF1d2b17N0ChJOgL0qOyIL2hdOl6\nVv54jlSd3mT7zbB4Jvdumr8ClGAvck+xvJD45Exik7OnFZsiS9gcOXIEtVpNz549AXjnnXfYsGED\n+/fvp0OHDsp+Dx48IDIyklq1ahVV0UhMTuP7g6GcvhxOZGyKsr2soyWW5lpuhcVzKzyexd/+w6h3\nPTA3k542QggBsHz5curUqUOrVq2yvJYxwTCAtbU1/v7+LFiwgNGjR2NpaUlKyr/1bVJSElqtNs/J\nGjDOi1CU0yDo9QbS0/P/BS0yzvg+z4ZEFuj4kqSgMXieSAwkBvBix8DFxYW///6bEydO8PHHH9O3\nb1+TBDyApaUlycnJAIWSoH+SHpX56Q0VE5+iJGsauZUhISmNK7diiElILXHJs8LwIvYUyw+JT84k\nNjkr7NgUWcImNDSUGjVqmGxzdXUlJCTEZFtwcDA2Njb4+/tz4cIFqlWrxrhx4/D0zNtdyYJk4Pf9\ndYffTt5SnjesUZq3/VypXsEelUrFgb/usOZ/57l8K4Yf/gjlvbZu+bvAM04yqTmT2ORO4pOz5yU2\nO3bsIDw8nB07dgAQHx/PyJEjGThwINHR0QwePBhbW2MjNyUlRWmc16hRg9DQUBo1agQYPyOqV69e\nPG+iCKXr9WiKoluQEEI8JVqt8euDj48P7dq14+zZsyYJeIDk5GSsra0BCiVBX5AelQXpDRUWlaQ8\n7vmKGzfzoWREAAAgAElEQVTC4rhyK4a4hFQiI+PzV4ASTHrL5U7ikzOJTc6eNDY5JYWLLGGTmJiI\nlZWVybbM2fcMKSkpeHh4MGbMGKpWrcq3335L//792blzJ2XLln3sdQqSgW/fojqR8alUKGtD8/ou\nVC1vb/L6221qEZmQxvf7rvDnmbsM6NLwhZzPRjKpOZPY5E7ik7NnPTa//PKLyfM2bdowefJkWrVq\nRbt27TAYDIwaNYo7d+4QFBRE9+7dAXjrrbdYs2YNzZs3R6vVsnLlSjp16lQcb6FI3Q5PoIqzzIMg\nxOMYDAbu3buLi0uF4i6KeGj//v2sW7eO9evXK9vS0tKoUqUKBw4c4M6dO1SoYPx9hYaGKsOgCiNB\n/yQ9KvPTGyoxWac81mrUWJkbvyolp6aTkpqOVvN8Jdxf5J5ieSHxyZnEJmeFHZsiS9hYWVllSc5k\nzr5naNu2LW3btlWe9+zZk2+++YajR4/yxhtvPPY6BcnAa4E+7d2VjFh2GXS/+s78sO8KcYlp7D12\nnWa1y+XvIs8wyaTmTGKTO4lPzp4kNs9Ct2y1Wk1QUBCzZs2iefPmWFpa8u6779K7d2/AWLdHRETw\nzjvvkJaWxptvvkmfPn2KudSFT5duOg9CyN1YSdiIZ4qvb1MsLCxQZ2pclS5dhg8+6M0bb7z91K67\nbNliAIYMGf7UriHyp27dupw9e5YffviBt956i4MHD7J//362bt3K3bt3+eKLL5g1axaXL1/m559/\nZtWqVcCzlaBPTv03YWNprsEm09yVCck6HJ7xieOFEM+eIkvYVK9ena+++spkW2hoaJYkzC+//IJe\nrzeZ1yZzN/rHedI5DXLKiDnaWlCnWimCr0Vx4K87NHZ7fG+f541kUnMmscmdxCdnz1ts9u7dqzyu\nWbOmyZ3YzDQaDSNGjGDEiBFFVLLikZiiM3keeieWlz0qFlNphCiY1as3UL16TTQaFQ4OVmzd+n/M\nmDGV+vUbUa2a61O5ZkxMNA4Ojo/fURSZsmXLEhQUxJw5c5gxYwbVqlVj2bJl1KhRg5kzZzJ16lRa\ntWqFtbU1Y8aMUXrUPEsJ+uTUf79EWJhrsLH696tSYnKaJGyEEEWuyBI2Pj4+pKamsmnTJnr06MH2\n7duJiIjA19fXZL/ExES++OILatWqRdWqVdmwYQPJycm0aNGiqIqaI98GLgRfi+JMyAOi41NwtM37\n2FshhBAvnszd68HYw0aIR+nS9UTGJj9+x0LgZG/5RMM6NBoNr73WgUWLviA09CrVqrkSGxvD4sWf\nc/ToESwtLenUqSsffNAblUpFTEw0ixZ9zpkzfxMVFUmlSpUZNWo8DRt6APD999/y9debiImJpk6d\nuowdG8DBg/vYvXsnKpWKe/fuMmvWPI4fP0JQ0DJu3rxBhQoV8ff/GB8fYxvS17cpnTt349dff+H9\n93vx4YclMxnwPGjatCnfffddlu2Ojo4sXrw422OepQR9RsLGwlyDWqUy7WGTpMvpMCGEeGqKLGFj\nbm7O6tWrmTZtGgsWLKBq1aqsWLECa2trpkyZAsCMGTPo0qUL4eHh9OvXj+joaOrWrcvq1auzDJ0q\nDo1rlcXKQkNSSjqnL0fQ2lPukgohhMhZQnKayfM74Qkkpeiwsiiyj19RwunS9UxcdYSImKJJ2JRx\nsGTOgOYFTtqkpqaydes3pKSkUK9eAwBmzpyCvb0D27b9SHR0FGPHDsfJyYmOHd9i+fIlAGzevA21\nWsPixV8QFBTI8uVfcuTIIVatWs4XXyzB3b0OK1cuY+bMKQQFreXq1Ss4ODgyZMhwQkKuMm7cKKZO\nnUmLFi05fvwIkyePZ+XK9dSoUfNhuVL46afdpKWlFk6gxAspY0iU5cMVYS0fJm70BgPxj9TnQghR\nFIq0xVi7dm3++9//Ztk+Y8YMk+f+/v74+/sXVbHyzNxMQ40KDpwNjeRm2PMzU7wQQoin49EeNgbg\n6u0Y6lcvXTwFEqIABg7si1qtIjU1DTDQvLkPixatoFw5Zx48iODIkUP8/POvWFlZYWVlRc+evdi+\n/Ts6dnyLAQM+xtLSEo1Gy927d7CzsyM8PByAPXt20b59R+rWrQ9Anz79uX49NMv1f/ttN02bNqNV\nqzYA+Pj40qJFS3bv3smgQUMBaNu2HWZmZpiZmWU5Xoi8yuhhY2luTNioVCpsrLTEJaaRkCQJGyFE\n0ZNbfPlUqawtZ0MjuSUJGyGEEI+R0cPG2kKLk70lt8LjOX89ShI2QqHVqJkzoHmJHhIVFLSG6tVr\ncv/+HQICxuLg4Ei9esYky/379zAYDLz77r8TEOv1BuztjStuRkREsHjx51y7FkrVqlWxs3PAYDBO\nxh0ZGUmNGm7KcVZWVtSuXTfL9aOiIilf3sVkW/nyLoSHh/37vpzK5Os9CZGdfxM2/35FsrE0MyZs\nkmVIlBCi6EnCJp8qlTMuwXsrPB6DwZDvJcSFEEK8ODJ62FhbaqlTtZSSsBEiM61GTblSxT/0+3Eq\nVKjI8uXL6dSpE+XLV6B3776ULl0GjUbDjz/uxtzcOCFrbGwsiYmJAEydOpFOnbqwbNlqVCoVO3f+\nTEjIFcA4iW1ExL9Jl4SEeNauXc3AgUNMruvsXJ5z586abLt79w5ly/67Yqc0x0RhUIZEPexhAygT\nDyfKkCghRDEo+KxzL6hKZY3L6SanpvOgiMabCyGEeDZl3JG1sTSjTrVSAFy/F5dlbhshnhUVK1bk\nk09Gsm7daq5cuYyzc3kaNfJkxYqlpKQkExsbw+TJ41i9ehkAiYkJWFlZolKpuHYtlK+/3ohOZ/y7\naNeuPb/88j8uXbqATqdj48Z1BAefVYY2JSQkAPDKK+04deo4Bw7sIz09ncOH/+SPPw7Qtm27YouD\neD6lPDIkClAmHpZJh4UQxUESNvnkUtoG9cPbOLfCE4q5NEIIIUqyjDkPrC21uFd2RK1SYQAu3ogu\n3oIJ8QQ6dnwLT88mzJ07g/T0dKZNm01k5APeeectevToQpkyZRg5chwAY8dO5OuvN9GuXSsCAsbQ\nvv0bREdHERMTTZMmzRg0aBhTpkykY8dXuHr1MtOmzQagdeu27Nv3GyNHDqFSpcrMnfs569ev5vXX\nW7N8+WKmTp1FnTr1ijMM4jmUeZWoDDaWxh42kmgXQhQHGRKVT2ZaNeVLW3MnIoGb4fF4uMmYaSGE\nENlLVHrYaLGy0OLqYsfVO7Gcvx5F41pli7l0QjzeH3+cyHb7woXLlMdOTqWZPn1Otvv5+rbC17eV\nybaePXspj994oxNvvNEpy3HNmnmzc+feTM+b06xZ83yVUYj8ymkOG0BWiRJCFAvpYVMAlcoa57G5\nHS4TDwshhMiZMunwwwZ/7arGYVFnQh5gMBiKrVxCCCGyyn4OGxkSJYQoPpKwKYCMeWxkaW8hhBC5\nydzDBsCjprFXZlhUEnceJBZbuYQQQmSV/Rw2MumwEKL4SMKmADISNvcjk0jTpRdzaYQQQpRUCZlW\niQJwrWCPg41xJZ3Tl8KLrVxCCCGyym1IlCzrLYQoDpKwKYCMIVF6g4F7kUnFXBohhBAlVWKK8Y5s\nRoNfrVLh+XDus9OXJWEjhBAlSW7Leickp6GXoaxCiCImCZsCcLSzUB7HJqYWY0mEEEKUZI/2sAHw\nfDjZcOjdOCJjk4ulXEIIIbJKzmVZb4MBklOkl40QomhJwqYAtBo1VhbGxnd8ooxnFUIIkZUuXa/M\nh5DR4AeoXaWU8mXgpAyLEkKIEiPbIVFW/9bf8TIsSghRxCRhU0B2Dyvv+CRJ2AghhMgqMdOd2Mw9\nbMy0ajzdjL1sjpy7V+TlEkIIkVWaTk+63jjkySKbSYdBJh4WQhQ9SdgUkK21MWETJ0OihBBCZCMx\n053YzHdoAXzqOwPGYVF3HyQUabmEEEJklTF/DZgOicqccJelvYUQRU0SNgVk+7DxHSc9bIQQQmQj\nIdOd2Mx3aAHqVC2lrBZ15Nz9Ii2XEKLo3blzu7iLIB4jYzgUmCZsNGo1VhbG5wnSw0YIUcS0j99F\nZEcZEiVz2AghhMhGRg8bFSjznmXQqNV413Vm9/GbHD53j05+rqhVqmIopRCPd+TIIb75ZhOXL19C\npYLatevQv//H1K5d94nOu2bNSkJDrzJr1mds3LiW69dDmTx5ZiGV2lRiYiLt2rVk27YfcXGpUKBz\nnDp1gsmTx3Hs2LF8HXfp0gXGjPmE7dt3Fei6omikmCRsTOtsG0szklLSSZAbtc8tvd6ALv3fYXFq\nsxSi41LQpRswGAzo9Qb0gEFvQG8woDeAwWDAYDAea8CAXm/cpn+4/dHjDBgnrzYyKI8zbcKA6cac\njsl0CIZHNhpyOM+/j7M5Tx4XQFOrwcbGkoSEZPT6zOUohBXUnvAUxb2Gm0oF1SqVomoZq0I9ryRs\nCihjSJTMYSOEECI7GXdirSy02SZjXqpfnt3HbxIRk8z5a1HUc3Uq6iIK8Vg//vg9X34ZxLhxk3jp\nJR8cHKxZvXotw4YNIihoLdWr1yiU6/Tq9VGhnKckio+PR6eToTQlXU49bMCYsImISc510uH9f91m\nz4lb2FmbUc3Fnk4tXE3mwhH5k6bTk5SqIynl4b9kHYkp6SSn6khNSyclTU+aLp1UnZ7UND2punTl\nZ5pOT2qa8acu3UC6/tGfBtLT9ej0BtLTjY+L+8u+eH5M+KAxbpUcC+18krApIGVIlPSwEUIIkY3U\nNOOtp5wa7FWc7XB1sSf0biy/n74tCZsXmE6vIyo5pkiuVcrSAa06b82/5ORkAgMXMXXqLFq08EOj\nUWFhYUHPnh8SGRnF9euhVK9eg8jIByxZsoDjx49gYWFJ27bt6NdvEObm5iQmJrJixVL2798LwEsv\n+TJkyAhsbW1NrpW5t83s2dOwsbHh0qWLXL58kSpVqjF2bADu7rUxGAysW7ea777bhrm5Od26vUdQ\n0FL++9/v89Vz5sSJY6xZE8TNmzdITU3Dy6s5kyZNx9LSkri4OBYt+ow//zyIVmtGhw5vMGjQMJPj\nExLiGTJkEG5utRg7NoDU1BRWrFjKvn17MRgMvPrq6/j7DyY+Po7Roz8hNTWFV1/149tvf8LBofAa\n8qLwZJ7D5tF628bK+DeTUw+bC9ej2LjrotJL4cKNaEJuxzC8e6MsvXVeVInJOqLikomKTyEuIY24\nxFTikh7+TEwz/ktKIyk5jcSUdHTp+sef9BmlyvRA9fBZ5vs6xsem2zNtMh6Tabsq8wmzbANVpicm\n53u44ZHDH1t2tVr1sFdRDu/rCaiKubfxk16+fGkbKpSxKZzCPCQ1SAHZWRvnHohPkkmHhRACICIi\ngjfffJM5c+bQunVr7t27x4wZMzh58iRarZbXX3+dcePGYW5ujsFgoEmTJibdeJs0acKXX35ZjO+g\ncGU0NrWanD/9W3tWJPRuLH9djiAqLoVSdhZFVTxRQuj0OmYcmc+D5KgiuV5py1JMaT4mT0mbM2f+\nIj1dh7e3T5bXBg0aqjyeOHEMLi4V2LbtRxISEpg4cTRr1qxk0KChfPbZbKKiItmw4b9oNBpmzZrC\n/PmzmT59bq7X/uWXHSxdupIqVaowZ850Vq4MZMGCQP73vx/ZseMnVqxYg5OTEzNnTiE9PT3Xcz0q\nKSmJgIAxTJ48A1/fVoSF3efjj/uxZ88vvPHG23z++RySkpLZuvVH0tN1DB06kIoVK1O5chXl+NGj\nh1OzphtjxwagUqkIDFzMrVs32LDhG/R6A5Mnj2PjxrX07evP558vZvLkcfzvf7/lq5yiaGX0sLEw\n02TpFWn/cM6xqLiULMfFxKew8sdzGAzgUtoat0oOHPj7LpduxbBo69+M6uGJmfb5njbUYDAQn5TG\n/agk7kcmEh6dRGRcClFxKUTGJhMVl2LSg6mgzLRqLM01WJhpMDfTYK5VG/+ZaTDTqh9uV2OmffhT\no0arUaPRqNCqH/7UqNGoVSbbNGo1Wo3q4XbjOUuVsiE2NhGDAdQqFSrVw59qFWqMSQtV5u2qh9v4\n97FaZUxAZLxe3MmIwqDRqHBysiUyMp70dOmXlNnTio0kbArINtOy3gaD4bn4AxRCiCcREBBAdHS0\n8nzMmDG4ublx4MABYmNjGTx4MMuWLWPEiBFcv34dgFOnTj239adOl5Gwybmh7lWnHFv2XiYhWcf+\nv27ztl/1oiqeEI8VHR2NnZ09Wm3OzcXbt29x9uw/fPrpAqytbbC2tqFfv0HMnj2Njz7qz759vxEU\ntJZSpUoBMGTIcN5/vxsTJ07N9dq+vi1xc6sFQJs27QgMXATA7t076d79PSV58vHHw/jjjwP5el/m\n5uasXbuZihUrER8fT0REOA4OjoSHh5OamsqBA/tYtWoD9vb2AMybtwAzMzNu3bqJXq9nyJAhqFRq\nxo2bhEqlwmAwsGPHj6xYsUbpPdO3rz/TpgXQt69/vspW0p04cYJ58+YREhJCqVKl6NevHz169ODM\nmTN0794dS0tLZV9/f38GDhyIwWBgwYIFbNu2jfT0dDp16sSECRPQaErWcKGMhMKjw6EAXJysAbJd\n1e+nQ9eISUjFwkzDkC4NcCltQ+Vydmz+9RKXbsXw1e6L/Kd97efisy5dr+d+ZBK3wuO5E5FAWFQS\n96MSuR+ZRGJK3of92VhqsbU2x87aDDsrM+wyHlubY2OpxdJci7WFBitLLVYWD/+Za4ss8aV88bbS\nSFJCFDtJ2BSQ3cM5bHTpBpJT07NMKCmEEC+Sb775BisrK1xcXABITU3FysqKQYMGYWFhQdmyZXnz\nzTf59ddfAQgODsbd3f2JGrAqlQp1EbTd1GqVyc+8Sn/Ye8hMa7yDlx0rjRbfhi7sOnaTA3/fpZOf\na64JnuJS0Bg8T55WDDQaM6a3GEtUcvTjdy4EpSwd8zwkqmzZMsTGxmIw6NBqzUxiEBsbi7W1NTEx\nUVhZWVG6dCnluAoVXIiMfEBcXAw6nY6KFSsofwMVKlTAYDDw4EGEcodao1GZPFapwNGxlHKMubkW\ng0GPRqMiIiKc8uXLm5wPjF+wcvo7y3g946e5uZZDhw6yZctmAGrWrEVychJgIDExjrS0NFxcnJVj\nqlSpDMDdu7eIi4tDr9dz/vw57t69ReXKVYiMjCIlJYWhQ/2VOs1gMJCWpkOnSzW59rMsJiaGjz/+\nmMmTJ9OxY0fOnz9Pnz59qFKlCrdu3aJly5asXLkyy3GbN29m3759/Pjjj6hUKvz9/Vm7di39+/cv\nhneRs4whUdkNY80Y4nAvMpF0vR7Nww+fNJ2eo8HGlf46+FTFpbRxv1eaVCI2IZWfDl3j4D93qeZi\nT2vPikXxNgqNLl3PzbB4rtyK4cb9OG6Gx3MnIvGxQ5WsLbSUK2VFaXtLStlZ4PTwp/GxBY62FiXy\nc06IkkqyDAWU0cMGjL1sJGEjhHhRhYaGsm7dOrZu3UqXLl0A4x3sVatWmez3+++/U7t2bQDOnz9P\nfHw8nTp1IiwsjGbNmhEQEICzs3Oer1u6tE2R3rF0dMzfmGQzM+PngqWFFicn2xz3e7u1G7uO3SQ6\nPoWr9+LxaVCwFWyKQn5j8Dx6WjEoR8mb16Rly5cwNzfjzJlTvPLKK8p2R0cbJk0ah42NDSNHjiQp\nKQmVKk3pRXPmTCSOjo64u1d/OI9NDNWrG5MeV6/eR61WU716JayszDE3N/59ZH5sYWGGlZW58ndj\nZ2eFRqPGycmWSpUqEhcXpbx2/foDABwcrLP8nY0fPx5/f39cXV3RaIy9J5ydnbh27RLr1q1m27Zt\nVKtWDYBevXphZWVO9eqVMTMzIyUlHienSgAcPHiQ6OhoypUrh729PWvWrGHatGnMnz+HTZs24eBg\nhZmZGdu3b6dyZeP7TExMJCIiAheX0ty4YYVKpcq1HngW3Llzh1atWvHmm28CUK9ePby9vTl16hQR\nERFK/f6o7du307t3b8qVKwcYe94sXrw4XwmbgiTo85tkTX3YK9LKXJsluVapnPF3p0s3EBmbQvnS\nxh43py5FkJCsQwX4NXIxOa7Ly9W5cT+Ov68+YPPuS5R1tKRRzTL5exNPSXaxSUzWcfV2DJduRnP5\nVgxX78Qoc7E9ykyrpkJpG8qXtsK5lDXOTtaUd7LG2ckKWyuzZ743kdykyJnEJmdPKzaSZSigjDls\nwJiwKetYuMt3CSHEs0Cn0zF27FgCAgJwdMz+C6fBYGD27NmEhIQwf/58wJjQ8fDw4JNPPsHCwoLZ\ns2czdOhQtm7dmudrP3iQUGQ9bBwdbYiOTkCvz3vX6Nj4ZOMDA0RGxue4n7VWRd1qpQi+FsX2/Vdx\nr2j/pEUudAWNwfPkRY3BwIGDmTRpMomJqfj4vISlpYagoNX8+echVq1ai5mZDU2bejFt2gzGjJlA\nfHw8Cxcu4tVXXyM6OpHXXmvPp59+xowZs1GrNcyaNYeXXvIlLU1FUlIqqak6IiPjTR6npKSRlJSq\n/N3ExSWRnq4nMjKedu3as3LlCjw9vSlTpgzz5hnrlJiYRKytTf/Ozp+/wNat/8dHH/Xnu+++pUyZ\nMqhUFty9G4FKpSI5OZ3w8Bh27drJiRMnqF27HjExSbzySju++GIBU6bMIDk5mTlz5vLuu+9hZWWH\nSqVGrVbTr98gunfvwtq1G+ncuSvt2rVnzpxPGT9+ElqtlnnzZnPv3l2CgtaQnJxOSkoK9+9HYWZm\nliXGuSlJSZ46deoodTgYe9ycOHGCTp06sXr1aszNzWnTpg16vZ727dszYsQIzM3NCQkJoWbNmspx\nrq6uhIaG5mtKgSdJ0Oc5yfrwA8XWxjxL3O0drNGoVaTrDcQkp1P34etHL5wFoKFbGdyqZU3GjO/j\nzfjAg1y/F8ey788yy/8lalcrGRPMh0UlEhwaxfnQB5y/Fsm1u7HZLu3sZG+BW+VSVHOxp1oFe6q5\n2ONSxhbNC/CFXW5S5Exik7PCjo0kbArI2kKLSmVcs15WihJCvKiWL19OnTp1aNWqVbavJycnM3bs\nWC5evMimTZsoXbo0AEOHDjXZb9y4cTRv3pywsDDlLuzjGAwG8jnX6BPRP1z+M68y7kxqNarHHvey\nR0WCr0VxLjSS2+EJlH84X0JJk98YPI9etBi8/XY3rK3tWLNmFdOnT0aj0VCnTj2WLl1JtWo1SE83\nMGXKTBYv/pyuXY09L9q1a4+//xDS0w0MHTqS5cuX8MEH75KSkoqfXyuGDRtFerrBuMqIwZDlscFg\nbF9lxFn/8CZ/erqBV155natXQ+jbtxdWVla89loHANRqbZbfy6hR45k/fw5ff72J8uVdmDx5Jno9\nNG3qTevWbXn//XfRaNS4u9elffs3CA0NJT3dwPDhY1iy5Au6deuMSqWiU6cudOz4NqdOnYCH66LY\n2toxdOgIvvjiU3x8fPnkk1EsX76Unj27kZycTKNGHkyfPpf0dAOurjWpVq06r73WhnXrNlOpUuWi\n+eU9RXFxcQwcOJB69erRpk0bvv32W7y9vXn33Xd58OABn3zyCUuWLGH06NEkJSWZzG1jZWWFXq8n\nNTUVC4u8TbRekAR9fpOs0bFJAGjV2SfZnZ2suRORwKVrD3CvaEdMfAonz4cB4F2nXI6J+eHdGjJz\n/Qki41KYuPxP+r5RB5/65fP3Zp6QXm/gZli80nvm8q1oImOzTqAMULGsDW6VHKhV2RG3So6UdbTM\nkiyLic46l8/z5EVN0OeFxCZnTxqbnBL0KoMhu1zqsys8PK5AxxVkVudhiw8Sn5RG3451aNHApUDX\nfRbIbOA5k9jkTuKTsyeJTdmydk+pVPn3+uuvEx4erjTm4uPjlblrunfvTr9+/bC2tmbJkiUmPXBW\nrVpFixYtqFevHgD379+nZcuWHDt2DAcHhzxdu6D1fX4V9Hf11e6L7D11m4Y1SjO8W6Nc99Wl6xkX\ndJiouBRaeVSg9+vZDy0oLvK3LDGAkhGDy5cvUaqUE2XKGHszXLsWSq9e7/LrrwewsLB8zNFPrqhj\nUJLq+ww3b95k4MCBVK5cmUWLFpkkYzLs2rWLBQsWsGvXLho3bsy6deto1MhYD166dInOnTtz7ty5\nPF+zIPV9fn9Xa34O5s+z9/CqU46BnepneX3592c4cTEcn3rO9H+zHruP3eC/e69gYa5h0RDfbOe+\nyXAnIoFF2/4mIsbY89LTrQyd/aorQ60KW3KqjpA7sVx5mJy5eic221WazDRqXF3scKvsSM2KDtSo\n6GAy7cOLqiTUdSWVxCZnTxqbnOp76WHzBOyszYhPSiM+SXrYCCFeTL/88ovJ8zZt2jB58mRefvll\nevXqRZkyZVi6dGmWYQAhISEcPHiQJUuWoNVqmT17Nq+88kqekzXPgoyJGc3yMLmiVqPm1aaV2fr7\nFf48c5dOvq442soS30I86siRQ5w4cYy5cz9Ho1GzefMGPDwaF0myRsC5c+fo168fb731FuPGjUOt\nVhMTE0NQUBCDBw/G1taYgEhJSVF6z9SoUYPQ0FAlYRMaGkr16iVvRbzcVomChxMPXwznTkQiAH+e\nvQdAU/eyuSZrMo6d1Lspy78/y6Wb0Zy+HMHpyxFUKmtDfdfSVHG2pbKzHS5O1vme/8JgMBAWnUTo\n3VhCbsdy+XYMN+/Ho8/mnrytlZmx90wVR5rWdaGUtTbLEuZCiJKlSBM2wcHBTJkyhStXrlC1alWm\nT5+Oh4dHjvsfPnyYPn36cPLkSWxsSt44ucxLewshhPjX6dOnOXbsGBYWFnh5eSnb69aty+bNm5k0\naRKzZ8+mffv2pKWl8fLLLzNz5sxiLHHh0z28u6LN4zKkrTwq8POhaySm6Pj1+E26ta75+IOEeMG8\n+25Pbt++Sffub5GWloanZxMmT55R3MV6IURERNCvXz/69OnDgAEDlO12dnb8+uuvGAwGRo0axZ07\ndwgKCqJ79+4AvPXWW6xZs4bmzZuj1WpZuXIlnTp1Kq63kaOMVaIszbP/epSxAtTdyASu34vjZphx\nCHf/oE0AACAASURBVFSL+nnrZW9vbc7Y9zw5Gnyf7w+GEBGTzK3wBG6F/zu8yEyrpqyjFeUcrSjr\naEUpOwusLDRYW5phaW5cYjoxJY0HMcmExyQTEZ3EzbB4EpKzX1Lb2ckat0oOuFV0wK2yI86lrJTV\n2KSXhBDPhiJL2KSkpDBw4EAGDhxIt27d2L59O4MGDWLPnj3ZJmNiYmKYOHEiJXnEVkbCRuawEUII\no7179yqPL168mON+tra2zJ07tyiKVGwyetho87iUr5WFljZNKvHzoWvs++s2HX2qYW0pHWGFyMzc\n3Jzx4ycDk4u7KC+cb7/9lsjISFasWMGKFSuU7b169SIoKIhZs2bRvHlzLC0teffdd+nduzcAPXv2\nJCIignfeeYe0tDTefPNN+vTpU1xvI0fJaXnoYYNxfrKfDl0DoLS9JbWq5H2FN7VahU/98njXdeby\nrWhOXgon9E4sN8PjSU3Tk6bTcycigTsR+Z8jxkyrpqqzHTUrOuBWyYEalRywz7RIihDi2VRkLcEj\nR46gVqvp2bMnAO+88w4bNmxg//79dOjQIcv+06ZNo0OHDnz55ZdFVcR8s7OWHjZCCCGyl6bL+5Co\nDG2bVmL3sRskpaTz++lbdPSp9pRKJ4QQ+ZNx4zUn69evz3a7RqNhxIgRjBgx4imVrHCkpBrrbHOz\n7BM25Z2slAVHTl0KB+Cl+uULNKRIrVbhXqUU7lVKAcZJge9HJXIrPIGwqETCo5MIi0oiLjGNxBQd\niSk6UlLT0ahVWJhpKO1gSRkHS8o4WOFSxprqLvZUKGODNh+fN0KIZ0ORJWxCQ0OpUaOGyTZXV1dC\nQkKy7Pvjjz8SGxvLqFGj8p2wUalUBVrmtSDrptvbGLPW8UlpaPJ4B/VZ9LTWlH8eSGxyJ/HJmcTm\n+ZcxJEqTjwa0vbU5fg0r8NupW/x64hbtmlXGTJv73AhCCCGeXMacL9ocPpfNtBpqVHTgyq0Y436a\n/2fvvsOjqtIHjn+nZJKZJJBCEnoIRTpEULqigChKWZWirIgoQgDRDRZc+IlIscMusK4BFF2BtaLi\nInYFFAREmpKAlAAJoSUhIZmUaff3x2SGjClM6swk7+d58pice+/cM2/inct7z3mPiv7dqmfREbVa\nRZPwQOe0q9JUZBl0IUTdUWsJm7y8PPR6vUtbQEAABQUFLm1paWksW7aM//73v5jNFR+5Eh4eWKWL\nWUXWTY9qZC+slm+ylLkMV11S3WvK1yUSm/JJfMomsam7KlJ0uLhbe7Xgh31nuGw08ePBswzq0bwm\nuieEEKIYa9E1u7wHKfFjunM0NYsCk5Um4YFEhujL3Le6SbJGiPqp1hI2er2+RHKmoKAAg8Hg/Nlm\nszF79mzi4+OJiooiNTW1wufJyDBWeoRNRddNVxdl4rNyCsnMzK34SX1EVdeUr8skNuWT+JStKrGp\nDwniusDsqGGjrdhNdqMQPb07RfHzoXP8b8dJ+ndtgn8ZQ/SFEEJUD2vRZ7GmnISN3l9LtzaNaqtL\nQghRewmb1q1bs27dOpe25ORkhg8f7vz53LlzHDhwgKSkJObPn4/NZr/ZHThwIAkJCVx33XVXPY+i\nKFitle+nzaa4XS1dX1RFPq/AUi8qrFckNvWNxKZ8Ep+ySWzqLovFUXS44k8RRt0Qw+6k82Tnmvju\n11Ru7xNd3d0TQghRjGNKlExVFkJ4k1qrTNW3b19MJhNr167FbDbz0UcfkZ6ezoABA5z7NG3alIMH\nD7Jnzx727NnDZ599BsDWrVvdStbUNsfKH1ab4tWrWQkhhKh9lZ0SBRAZomdgbFMANv98SorbCyFE\nDXOMsJGEjRDCm9Rawkan07F69Wo+//xzevXqxbp163j99dcxGAzMmzePefPm1VZXqk3xIZNWme4h\nhBCiGEfR4cqu2jGiXyv8/TTkFVr45MeSBfqFEEJUH8f0ZG1laisIIUQNqbUpUQAdOnTgvffeK9G+\nYMGCUvdv3rw5R44cqeluVVrxlT+sNgVZyEMIIYSDxVnDpnI3/w2D/BneL5oNW0+wZd8ZBnZvSsuo\n4OrsohBCiCI2GWEjhPBCkkKuguIXdKlBIYQQojhn0eEq3PwPvb4lkSF6FAXWff2Hs8aCEEKI6uVO\n0WEhhKhtkrCpAtcpUTYP9kQIIYS3cRYdruQIGwA/rZrxt1wDwLEz2fyw90y19E0IIYQrGWEjhPBG\nkrCpAqlhI4QQoiyOGjaVKTpcXLc24fTpHAXAR1uOczErv8p9E0II4UqKDgshvJEkbKrApYaNTIkS\nQghRjLOGTRUTNgD3Dm5HsMGPQrOVVf87JKM6hRCiGimKIlOihBBeSRI2VaCVKVFCCCFKYSt286/V\nVv3mP9ig44FhHQA4fuYyG386WeXXFEIIYVe8PJiMsBFCeBNJ2FSBTIkSQghRGkf9Gqj6lCiHa9tF\ncHOPZgB8vuMkB46lV8vrCiFEfVf8Pl6jkoSNEMJ7SMKmCmRKlBBCiNJYin0mVMeUKIdxN7clOioY\nBVj1v0OczTBW22sLIUR9ZSuesNFIwkYI4T0kYVMFMsJGCCFEaRz1a6B6EzY6Pw2P3NWVIL0f+YVW\nln14kMtGU7W9vhBC1EfF7+NlSpQQwptIwqYKimfgLVLDRgghRBHXhE313vyHNwxgxp1d0GpUXMjK\nZ9lHB8gvtFTrOYQQoj6xFStiI0WHhRDeRBI2VeAywkamRAkhhChiLp6w0Vb/R237lqFMHt4JgOSz\nOSx5fz95BeZqP48QQtQHLiNspIaNEMKLSMKmCjTqYjVsZEqUEEKIIjVRdPjPenWM4v7b2gNwIu0y\nr7y3n9x8SdoIIURFudSwkRE2QggvIgmbKig+JUqW9RZCCOFQU0WH/+ym2GZMGtYBFXDqXA6vvLuP\nbKlpI4QQFWItNipSatgIIbyJJGyqQK1S4Rg1KVOihBD1XXp6On379uWHH34AIDs7mxkzZtCzZ09u\nuukmPvzwQ+e+iqKwZMkS+vTpw/XXX8+iRYuwWq2e6nq1M9dQ0eHS3NC9KZOHd0KlgpQLuSx4+xeS\nz16u0XMKIeqnPXv2MGbMGHr27MmQIUN47733AN+/3lulho0QwktJwqaKHNOiZEqUEKK+mzt3LllZ\nWc6fn3nmGQwGAzt27GD58uW8+uqr7N+/H4D169ezZcsWPvvsMzZv3szevXtZs2aNp7pe7Yo/rfXT\n1vzNf98ujZk2qgs6rZpLOYW8sG4v2387W+PnFULUH9nZ2UyfPp3777+fX375hWXLlrF06VJ27Njh\n89d7m6wSJYTwUlpPd8DXaTQqLFZJ2Agh6rd3330XvV5PkyZNADAajXz77bd89dVX+Pv7061bN4YP\nH86nn35KbGwsGzduZOLEiURGRgIwdepUli1bxsMPP+z2OVUqFepaeOzguHmvyE188ae1Op2mVopY\n9u4cReNwA8s/Okh6dgFvfp7EsTPZ3DukHXr/qn3cVyYGdY3EQGIA9TsGaWlpDBw4kBEjRgDQuXNn\nevfuzd69e2v8el/TrC41bOR5thDCe7h9B1dQUEBAQEBN9sUnadUqCnF9miqEEPVJcnIyb731Fh98\n8AF33XUXAKdOnUKr1dKiRQvnfjExMXz99dcAnDhxgrZt27psS05ORlEUVG4mN8LDA93etzqEhAS6\nva/+bC5gH1rfKDy4prpUQlhYEP+cFcbLa/dw8Fg6W/encejkJWaOiaVHh8gqv35FYlBXSQwkBlA/\nY9CxY0deeeUV58/Z2dns2bOH9u3b1/j1vjIJ+som13R+apc6lXVRfU48ukPiUzaJTdlqKjZuJ2xG\njBjB8uXL6dixY7V2wNc55rnKCBshRH1ksVh46qmnmDt3LiEhIc72vLy8Ekn+gIAACgoKAMjPz3fZ\nrtfrsdlsmEwm/P393Tp3Roax1kbYhIQEkpVldBk2X55LWUbAXr8mMzO3JrtXqr+N6coXO0/zybYT\npGfl8+zqn+nXpTF339SaRg31FX69ysSgrpEYSAyg9mMQFhZU4+eojJycHOLi4pyjbN555x2X7dV9\nva9Kgt6d5FqG8coKe+HhQQQbdJU6l6+pj4nHipD4lE1iU7bqjo3bCRuz2VyrTzJ9hUYjNWyEEPXX\nv//9bzp27MjAgQNd2vV6PYWFhS5tBQUFGAwGwH4zX3x7fn4+Wq3W7Zt3sBeyrM26lTab4naB+UKz\nfdSlVqPyUFF6FcN6R9OtTSPe2pzEibTL7Pj9HLuTzjOoR3OG92tFkN6vwq9akRjUVRIDiQHU7xik\npKQQFxdHixYt+Oc//8nx48dr/HpfmQR9RZJrl7LynN9fzs7DXFC3V9uT5Gv5JD5lk9iUraqxKStB\n73bCZvjw4UyaNIk77riDFi1alHhyOm7cuAp3qi5wjrCRKVFCiHpo8+bNXLx4kc2bNwOQm5vLrFmz\nmDx5MmazmbS0NJo2bQrYp045hsW3adOG5ORkunfv7tzWunVrz7yJGmCxFCVstJ6thdCsUSBz7uvJ\nlv1n+OynZC7nmfn6lxS2Hkjjhm5NGHJdCyJDKj7iRghRPx06dIjJkyczcuRIZs+ejVqtJjo6usav\n91VJ0LuTXDObr9zHK0r9Wf21Pice3SHxKZvEpmzVHRu3EzabN29Gr9fz/fffl9imUqnqfcLGIhlG\nIUQ99OWXX7r8PGjQIJ555hluvvlmDh8+zJIlS1i0aBFHjx5l06ZNrFq1CoCRI0fy5ptv0qdPH7Ra\nLStXrmTUqFGeeAs1wlKUxPer4SW93aFWqxjUozl9Ozfm619S+HL3aQpNVr7dk8p3e1KJbdeIG7o3\npUtMWI0vQS6E8F3p6elMnjyZSZMmMWXKFGd7UFAQgwcP9unrvWvRYZlRIITwHm4nbEpL1IhiU6Ik\nwyiEEC4WLlzIs88+y8CBAzEYDDz55JPOJ6zjx48nPT2d0aNHYzabGTFiBJMmTfJwj6uPuegzwZsS\nIHp/LaMGxDCoRzO27DvD93vPkG00se9oOvuOphNs8KNXxyiuax9B2+YNZaUUIeqorKwsjhw5gsVi\nQVFc718HDBhQ5nEfffQRmZmZvP7667z++uvO9vvvv9/nr/eyrLcQwltVaJ3PjIwMPvzwQ06ePMmT\nTz7Jrl27aNeuHe3ataup/nm9K0WHZUqUEEIUT+6HhISwbNmyUvfTaDTEx8cTHx9fW12rVY5pst6U\nsHEINugY0T+G23pHszvpPNsOpHE0NZucPDPf/ZrKd7+mEhigpWvrcDrHhNGuRQiNw2TalBB1wccf\nf8z8+fMxmUrWaFGpVCQlJZV5bFxcHHFxcWVu9+XrvWOEjQpQS81OIYQXcTthk5iYyP3330/btm35\n/fffmTFjBtu3b+fvf/87CQkJ9O3btyb76bVklSghhBB/ZnZMidJ6742/n1ZN/65N6N+1CRey8tl5\n6By7Es9zNiMPY4GFnYnn2Zl4HoCQIH+6tm1EdGQg0VHBNIsIJEBXoWc+QggvsHz5csaOHcvf/vY3\ngoK8cwUqT3CMsJHRNUIIb+P23dYLL7zAxIkTmTlzJtdeey0AixcvJjQ0lFdffZUNGzbUWCe9mUbj\nKDosCRshhBB2jho2Gi8cYVOayBA9I/vHMLJ/DBcu5XHgWAYHj6dz9Ew2JrONrNxCftx/hh//dEyz\niECaRwTROMxARIieiFA9DQx+sqqkEF7q0qVLPPDAA5Ks+RPHg1fHfb0QQngLtxM2hw4dYtGiRSXa\nx40bx7p166q1U77EMcdfRtgIIYRwsFjsnwneUHS4oiJDDdxyvYFbrm+BxWoj5UIux85kc/J8LodO\npHPZaAbgQlY+F7Ly2Xc03eV4fz8NjUICCA3yJyTIn4ZBOkKC/AkJ0tEwyJ9ggx+BAX4Y/LXyNFuI\nWtavXz927NjB2LFjPd0Vr2IrquUjBYeFEN7G7YRNw4YNSUtLIzo62qX90KFDhIWFVXvHfIXUsBFC\nCN+iTj4BgBISghIadtU2VXgohAW57tewIUpYeJlthrTTNM46S3DklY9Z9clkUBTXY8tra9AQJbwa\n2k6dBJvN/bbgBiiNGgGgSz1NG5uNdm0aEDqsExkZOeQc+oNz6UZO56s4btJx5qIR5eRJTIUm8nQG\nLhsacuaiEfOxE6QrNmcbQGT2edTF2vT+GlrmZRCoU6NqEAwRkQT4a4jIOo9ODargYGwREfj7aQhJ\nP4u/xt6mjopCq1VjOJeKRlHsx0ZGolWr8D97Bq1iRd2gAaqoSDRqFZrUFLBYUAKDUCIj7e835XSF\n2lQNgq/8HaSmgNnsul9F2wyBKFFRZbedSQWTqUptGAzYohpXvS3tDBQWogoKhLC2Lm2l7Ydej61x\nk4q1nU2DgoKqtQUEYGvStGJt585Cfr7bbSp9AIS1d93P3x9b02bltjn66mmdO3dm8eLFfP/998TE\nxODn5+eyfdasWR7qmWc57uOlfo0Qwtu4nbC59957mTdvHk888QQAR44cYdu2baxYscLtSu+JiYnM\nmzePY8eOER0dzXPPPUdsbKzLPoqisHz5cj788EOMRiNdunRh3rx5XlvYWKZECSGEbwnvbf/cMT4+\nm7zZc13bZj1F3tP/B0BY3x6obDbyH38KXn3J3tavJyqrFWP8E+T9fZ69rf91qCwW8h57HOPcZwG4\n/8nRTLJa2HbrffDgjfb9buiFqrCQvJnxGJ95zt52Y29UBQXkzXgM47ML7W0D+6DKzydv+qMY59tH\ntobd1A9VnpG8aTMxPrcYgNCb+6M25pIX9wjGBc/b2wYNQJ2bQ97U6RgXvnilLecyeVOmYVxkfx+h\nQ25EnZ1F3sNxGBe/bG+75UbUWVnkPzSF3BdetbcNHYj60iUKHpoCb6xEpVLR8s6htMrMpPukyeS+\ntNQev47jUGekc37M/ey+5//IyC5gzIQHCbycyY6+I1h92wyyjSZefXc2oXlZfNl1KK/dMp38Qit/\nfyOecOMlvux6C6/dMgOAt1c+SLgxk6+7DGHF0EcAeGvVQzTKzeDrzoNZcetMANaseoiI3Ay+6TyY\n5UVtb65+mMici3zbeRDLbn3U3vbGFCIvX2Brl0G8PnIWKpWKZf96kMjs8/zUbTCr73oCtUrFS/+c\nROSls/zcfTBvj3salQoWvDqRyIw0frl2MO9PnIfVamPOC/cRkX6GvT0G898JzwAw+/n7iLiYyr5r\nB/HuA/a/gycWTyDywmkO9ChqU6mYteg+Is+f5uC1N/PuQ8+hQsXfFt1H5LlT/HbtTbw/2f53MHPR\n/USdTebQtTfx/hT738GMhfcTlXaCxGsH8sHU50EF0xbcT9SZ4yTF3sgH019EBUxZcD+NU49xuPsN\nfDDD/jt/eOFEmqQc5Uj3Abw/4+WitgdokvIHf3Trz3uPvALA5EWTaHr6CH907cd7M+1/B5MXP0jT\nU4c52rUvq59cjslkZtKih2h2MomjXfry3mNLAHjw+ck0S07kWOfevPu3fwAw6fmHaZ58iOOdevHf\n+H8C8MCLU2hx/HeOd7qe/8YvK2qbSovjv3Gi43Wsn7UcgIkvxdHy2EGSO/Rk3eMritqm0fLYAZLb\n92DdE/+y///2ynSi/9jPyfY9WFvUNuGVGbT6Yx+nroll7VP2FY3ue2UGrY7s5XS7WN6ZbW/766uP\nEHP4V0636847sxPsbUtmEpO0h5S23fjP0ysBGL/kUVon/cKZtl3J+GEbYQYtwTPj0G39AXPP68n6\n4jsAgh+bju6H7zD3vI6sL+xF2IP/NoPs9z7GG+zatYtu3bphNBr5/fffXbbV56mMjho2MsJGCOFt\n3E7YTJkyhcDAQF588UXy8/N55JFHaNSoEXFxcTzwwANXPb6wsNBZXX7MmDFs3LiRadOm8e233xIY\nGOjc76OPPuKrr75iw4YNREREsGLFCp566ik++eSTSr3BmqZ1TIlSJGEjhBDCVb25+S96mw0C/ejT\nyT7aQq/TAHBtuwhentYPRVFo8JYO8qDHNRHMGtedvAILhre1YIQWEUHc0K0JBSYrflr7Z2uwQUez\niEAKTVYcoaxMRBXsn9EWm4KxwGJvK/rcNpmtZF4uBK48ZS8wWUhLN9qPsdjbcvMtHDl9CQBzUVuO\n0cSRlKyi17G3Xc4zk3TqkvO1AbKNJg6dtLcVOvYzFvL7icyi8xXtl2viwPEMl7as3ELntLP8Qnvf\ns3IK+fWPiwDkFWvbc/gCABOK3mNWbiG/FLX9tajtUq7pSlthyf3GF5idfXG03VvUlpVr4sf9ZwAY\nm28p2q+Q3UkXitquHOtoG+NoM15pG51nLorBlba780tpK2W/u/JNzjg72u40ltzvL3mmot+RmV1F\nxbNHGh3HmpxtI4racoq1DXe2XTn2DsexRhMbvz/KlOEd8UVr1671dBe8klWKDgshvJRKUdzLNKSl\npdG4cWPUajV5eXlYrVaCg4OxWq0kJibStWvXco/funUrzz77LFu2bHG2jRgxgmnTpnH77bc72xRF\nIT8/H4PBQG5uLv/85z85ceIEa9ascesNXbyY49Z+f6bRqAgLCyIzM7dCo2Ve+/g3fv3jIjd2b8oD\nwzpU6tzerrKxqQ8kNuWT+JStKrGJiAiuoV75lspe7ys6JUodHkpom5ZkZuaiHDtu3+8qU6LeX/0l\nB45n0Dm2NX+9x76Koi9OiXK0qUMaEHpNjD0GJ5JL7nf6FFitVWsLCkaJiLhqmy0wCHNYOBarAidP\nYjVbMOsNmEIbYbHaUKekYDWbMAcEUtAwDItNQXsmBcVkxhRgoCA0HMUG/udSwWLBHGAgPyQcRYGA\ns6lgtWD2N5DXMBwFBf25M6gsVsx6AzRujDGvEP35NFQWCyb/omNRCEo/i9pswVT0eigQmJ6Guth+\nAIEXz6KxWjD5B5DX0B6DoPRzqK0WzLoAjI79Ms6hsTja7PsFp59DZTFj9g/AWHRscMY51BYzZp2e\n3KJjgzPOo7GaXY+tapvFjMU/AEtUEwoLzQQVtbnsl1m0n58/uaERbrXlhBbF4NIFtGaTy34Vb9OR\nG2qfehaUWUpbaftduojWXOh2GwH+DB19AyF6DcqZtBqfElUd1/uffvqJPn36oNVq+emnn8rcT6VS\n0b9//yqfryZU5npfkc/ZLfvP8M6XRwhv4M8r070zBtVJ7s/KJ/Epm8SmbFWNTVnXe7dH2AwePJjt\n27cTFhaGwWBwtp86dYr77ruPAwcOlHt8cnIybdq0cWmLiYnhxIkTLm0qlQqDwcDHH3/MnDlzCAoK\ncjtZ4zheXYkaj46MekUz69qiJVttilJnK8tXNjb1gcSmfBKfsklsPMcW07pCbapi13Z3j00Pb8a5\nDB3XNAy9sl+rmJLH1kZbdKsqt7nEoLT9WkbXapsf4KcF2rctsR8RpYx8aB1esq1L45Jt1zYr2Yb9\nvK43YiV/51BKX0ptK22K9zWltLV3s620h0Wljf6oeptrDKr7HJ081FYxxWNQWhLG3bbaNHnyZLZv\n3054eDiTJ08ucz+VSkVSUlIt9sx7yLLeQghvVW7C5t133+W1114D7CNfRowYUWJ+q9FodKu+TF5e\nHnq93qUtICCAgoKCUvcfPnw4w4cPZ+3atUyePJmvv/6akJCQq54nPDywSnNwQ0ICr75TMXq9DgCt\nVkNYWN1eIrGisalPJDblk/iUzRtic+nSJVatWsXvv/+O2Wwusf29997zQK98m7loWW9tHU3kCyF8\nx+HDh0v9XlxxZUqU763sJ4So28pN2IwePRq9Xo/NZmPOnDlMnTqV4OArQ3Uco2H69Olz1RPp9foS\nyZmCggKX0TrF6XT2RMhDDz3E+vXr2b17N0OHDr3qeTIyjJUeYRMSEkhWltGZZXeHtWh+el6+iczM\n3Iqf2AdUNjb1gcSmfBKfslUlNtWdHH766af57bffGDlyJEFBdTvxXFsszoSN3PwLIbyPoiilJugd\n99/1jeNzWCsjbIQQXqbchI2fnx9/+ctfAGjevDk9evRAq3V7FpWL1q1bs27dOpe25ORkhg8f7tK2\nfPlyrFYr8fHxgP0DxWQyuSSKyqMoClZrpboI2C/YFZlz5hg6abFW7DhfVNHY1CcSm/JJfMrmDbHZ\nuXMn77zzDt27d/doP+oSR6FaR/FcIYTwBrt27WLBggWcPHkSW1GRbbDfP8uUKJkSJYTwPuVmX5Yu\nXcq0adPQ6/X89NNP5RYqmzVrVrkn6tu3LyaTibVr13LPPfewceNG0tPTGTBggMt+3bt358knn2TY\nsGG0bt2alStXEhQURI8ePSrwtmqPYwUQq9V2lT2FEMI7hYeH4+/v7+lu1CnmoiScjLARQniTefPm\n0bZtW2bPnk1AQICnu+M1ZJUoIYS3Kjdhs2/fPsxmM3q9nn379pW5nzs1Y3Q6HatXr2b+/PksXbqU\n6OhoXn/9dQwGA/PmzQNgwYIFDBw4kFmzZjFjxgxycnK49tpreeONN7z2HxOOC7tVpnsIIXzUI488\nwsKFC5k7dy7R0dH4+fm5bK+vQ+SrwiI1bIQQXujChQskJCQQE1OyQHl95hhho5GEjRDCy5SbsFm7\ndm2p31dWhw4dSi1euWDBApef77nnHu65554qn682aIsK5kjCRgjhq5YuXUpWVhZ33313qdvr6xD5\nqnAkbPxkhI0QwovccsstbN26VRI2fyIjbIQQ3qrchE15U6CKU6lU9O/fv1o65GscS3nLlCghhK9a\nunSpp7tQ55iLathopYaNEMKLzJo1i5EjR7Jp0yZatGhRYlWkJUuWeKhnnuVI2GiqsNKsEELUhHIT\nNpMnT3brRepzkTKNTIkSQvi4Xr16eboLdY7jM0Fq2AghvMncuXNRqVQ0b95catgUI0WHhRDeqtyE\nTVJSklv1aeozR8LGIgkbIYQPueeee1i1ahUNGjRg3Lhx5V7rS5vKKsrnHGEjNWyEEF5kz549rFu3\njq5du3q6K17FKjVshBBeqtyETb9+/bj++uvp378//fr1o0WLFrXVL5+hKXp66ulleYUQoiIGEFhD\nBgAAIABJREFUDBjgLC58ww03eLg3dc+VosMywkYI4T2io6MxmUye7obXkRE2QghvVW7C5quvvmLn\nzp1s376dN954A5vNRv/+/enbty99+/YlJCSktvrpta5MiZIaNkII3/HII484vx8+fDitWrXyXGfq\nICk6LITwRtOmTePpp59mwoQJtGzZEq3W9Z8CAwYM8FDPPMuqyAgbIYR3Kjdh06BBA4YOHcrQoUMB\nSE1NZceOHXz55ZcsWLCAJk2a0K9fP+dXfSQ1bIQQvm7YsGF06tSJESNGcPvttxMZGenpLvk0s8WG\npWjUpU6n8XBvhBDiivj4eACef/75Etvqc01KW9GDV0nYCCG8TbkJmz9r3rw5Y8eOZezYsSiKwqFD\nh9ixYwerVq2qvwkbmRIlhPBx3377LV988QWbNm3ilVdeoUePHowYMYJbb72Vhg0buvUamzdvZsWK\nFZw7d46mTZsSHx9Pp06duOOOO1z2M5lMNG/enK+++gpFUejZsyeKcuX62bNnT954441qfX+1LSu3\n0Pl9SJC/B3sihBCuDh8+XC2vc/DgQaZPn+5cUfa3335j7NixLoWMp06dSlxcHIqisHTpUj788EOs\nViujRo3i73//OxqN9yS0ZVlvIYS3qlDCJjMzE4PBQEBAAImJiWzdupUuXbowZcqUmuqf15MpUUII\nX9esWTMmT57M5MmTSUlJ4auvvmLDhg0sWrSI/v378/rrr5d7fHJyMnPmzGHNmjX06NGDHTt2MGXK\nFLZt28a+ffuc+128eJE777yTuXPnAnDq1CkA9u7dW6cK3F/KuZKwCZWEjRDCw0aNGkW/fv3o378/\nvXr1QqfTVfq1FEVhw4YNvPjiiy4Jl6SkJG688UZWrlxZ4pj169ezZcsWPvvsM1QqFVOnTmXNmjU8\n/PDDle5HdbNJ0WEhhJdye3L9t99+y0033cTevXs5deoU9913H5s3b+Zvf/sba9eurck+ejWZEiWE\nqEtCQ0OJiIggKioKjUbDhQsXrnpMTEwM27dvp0ePHlgsFtLT0wkMDCzxj4Jnn32WYcOGceONNwKQ\nmJhI+/bt61SyBq6MsPH306D3954nyEKI+unll18mMjKSt99+mwEDBnD//fezcuVKDh486DLC0R0J\nCQm88847xMXFubQnJibSoUOHUo/ZuHEjEydOJDIykoiICKZOnconn3xS6fdTE2SEjRDCW7k9wmbZ\nsmXMnDmTfv36sWTJEpo2bcrnn3/O999/z+LFi5kwYUJN9tNraYqWbJUpUUIIX5Wbm8u3337Ll19+\nyY4dO2jatCnDhw8nPj6emJgYt14jMDCQlJQUbr31Vmw2G/PnzycoKMi5/eeff2bv3r288sorzrak\npCRyc3MZNWoUFy5c4Prrr2fu3LlERUW5dU6VSoW6Fmr6Om7g3b2RzzbaV2AJDfZHq60bRYcrGoO6\nSGIgMQDfjEH79u1p3749kyZNwmQysW/fPnbs2MGCBQtITU2lV69e9O3bl379+hEdHV3ua919993E\nxcWxe/dul/akpCR0Oh2DBg3CZrMxbNgw4uPj0el0nDhxgrZt2zr3jYmJITk5GUVR3E7YV+Z6X5Hf\nla0ocaXVqJ339nWZL/4d1yaJT9kkNmWrqdi4nbA5efIkw4cPB+CHH35g8ODBgP1DID09vVo75Uu0\nRZ8eMsJGCOGr+vbtS2hoKMOGDWPGjBl07dq1Uq/TpEkTDhw4wJ49e5g+fTrR0dH07dsXgFWrVvHg\ngw8SGBjo3F+n0xEbG8tjjz2Gv78/ixcvZubMmXzwwQdunS88PLBWR+eEhARefScg32yfIhsZZiAs\nLOgqe/sWd2NQl0kMJAbguzHQ6XT07t2b3r17Ex8fT3Z2tnNF2DVr1vDNN9+Ue3xZRelDQ0Pp3bs3\n48aNIyMjg8cee4zly5fzxBNPkJ+f71LbRq/XY7PZMJlM+Pu7N220Ktd7d35XjuldBr2uzl23y+Or\nf8e1ReJTNolN2ao7Nm4nbKKiokhMTOTSpUscO3aM5557DoAtW7bQvHnzau2UL5EaNkIIX7dq1Sr6\n9OlT5eSHY3nYvn37MnToUL777jv69u3L2bNn+eWXX1iyZInL/jNnznT5efbs2fTp04cLFy64tVJV\nRoax1kbYhIQEkpVldNY5KM/ZizkABOm1ZGbm1nT3akVFY1AXSQwkBlD7MaiJ5IHNZkNRFDQaDXl5\neeTn53PfffexYMGCSr9mQkKC83uDwcDUqVNZunQpTzzxBAEBARQWXqntlZ+fj1ardTtZA5W73lfk\nd1VQaAbAbLbUmet2eeT/5fJJfMomsSlbVWNT1vXe7YTNgw8+yGOPPQZAbGwsPXv25F//+hcJCQm8\n/PLLFe5QXSFTooQQvuj99993+fn06dNl7jtu3LhyX2vr1q289dZbvP322842s9lMcHAwYB+V2atX\nL8LCwlyOW7VqFf3796dz586AfQUpwO2beEVRsFrd2rVa2GyKW9f6zMv2f5iEBPrXuc8Gd2NQl0kM\nJAbguzHYs2cPjz76KEuWLKFNmzaMHj0ak8lEfn4+L7/8MrfffnuFXzM7O5uEhARmzJjhnApbWFjo\nvJa3adOG5ORkunfvDtgL1bdu3bpC56jK9d6d35Vju1ql8snfa2X56t9xbZH4lE1iU7bqjo3bCZvx\n48cTGxtLWloaAwYMAGDAgAEMGTKkzCJj9YGmKN2vYP/lyHw+IYQvKG0lj9KoVKqrJmw6derE77//\nzqeffsrIkSP58ccf2bp1q3Nq04EDB4iNjS1x3IkTJ/jxxx9Zvnw5Wq2WxYsXM3jwYLeXEvdWjlWi\nQoNlhSghhHd56aWXuP3224mNjeWtt97C39+fH374gU2bNvGvf/2rUgmb4OBgvvnmGxRF4fHHHyct\nLY2EhATGjh0LwMiRI3nzzTfp06cPWq2WlStXMmrUqOp+a1UiRYeFEN6qQst6d+rUiYKCAj7//HNu\nvfVWAgMDr1qcrK4rvvyf1WZDrZYVQYQQ3u/777+vtteKiIggISGB559/ngULFtCqVStee+012rRp\nA8CZM2dKTdj83//9H4sXL2bYsGGYzWZuuukmFi5cWG398gRFUcjKtY8UCpElvYUQXubIkSMsW7YM\nvV7P999/z5AhQ5x1bebPn1+p11Sr1SQkJLBo0SL69OlDQEAA48aNY+LEiYD9oW96ejqjR4/GbDYz\nYsQIJk2aVI3vqupkWW8hhLdyO2GTmZnJtGnTOHToEDabjV69erFkyRKOHz/OmjVraNGiRU3202sV\nryRvsSr4VSgFJoQQ3iEjI4MPP/yQkydP8uSTT7Jr1y7atWtHu3bt3Dr+uuuu4+OPPy5127p160pt\nDwoK4oUXXqh0n71Rbr4Zi9Ve00xG2AghvE1ISAhnzpzBarVy6NAh4uPjATh48KBbtcMcevfuza5d\nu5w/t23b1mVabHEajYb4+HjnubyRVbkyJUoIIbyJ2+W7Fi9eTHh4OLt27XLOSX3ppZdo2bIlixcv\nrrEOejtNsQposlKUEMIXJSYmcuutt7JlyxY2bdpEXl4e27dvZ/To0fz888+e7p5PcUyHAknYCCG8\nz5gxY5g+fTrjxo1zruS3du1aZs+e7RwRUx/JCBshhLdyezzIjh07+M9//uOyJGvDhg15+umnuffe\ne2ukc77AdUqUJGyEEL7nhRdeYOLEicycOZNrr70WsCfpQ0NDefXVV9mwYYOHe+g7snLtCRuVChoE\n+nm4N0II4WrmzJl07NiR1NRUhg8fjlqtpmXLlixfvpybbrrJ093zGGfRYUnYCCG8jNsjbKxWK7ZS\nlq7OyclBo6m/dVuKT4myWmVpbyGE7zl06BAjR44s0T5u3DiOHz/ugR75LscIm4aBOpcRmEII4Q3G\njh1Ly5YteeCBB2jUqBEAAwcOrNfJGrjy0FVG2AghvI3bd5NDhgzhlVdeITMzE1XR/M5jx46xcOFC\nBg8eXGMd9HYywkYI4esaNmxIWlpaifZDhw6VWIpblE9WiBJCeLPU1FS0Wim4+Gc2RUbYCCG8k9tX\n7Dlz5jB37lz69++PoiiMGDGCwsJCbr75ZubMmVOTffRqGo3UsBFC+LZ7772XefPm8cQTTwD2VUS2\nbdvGihUrvG4lD2+XcbkAgNDgAA/3RAghSrrnnnuYMWMG48aNo3nz5gQEuF6rBgwY4KGeeZYs6y2E\n8FZuJ2yCgoJYtmwZKSkpHD9+HIvFQps2bYiJianJ/nk9rVqmRAkhfNuUKVMIDAzkxRdfJD8/n0ce\neYRGjRoRFxdXr4tQVkbKhVwAmjYyeLgnQghR0r///W8AXnzxxRLbVCoVSUlJtd0lr+Ao+6CVhI0Q\nwsuUm7BJTk4utT06OrrEPvU1cSNTooQQvshms6EuVmPlr3/9K3/961/Jy8vDarUSHBwMwMWLF4mI\niPBUN32KxWojLd0IQMvIYA/3RgghSjp8+LCnu+CVbDLCRgjhpcpN2AwbNgyVSoWilJ6IcGyrzxl5\nmRIlhPBFjz32GP/4xz9K1DIwGK6MDNm8eTPPPfccu3btqu3u+aS0dCOWopVGWjYuP2GTkpPGqcun\nub5xD/w1utronhBCAGAymfjqq684efIkEyZM4MiRI7Rp08ZZhLg+kilRQghvVW7C5rvvvqutfvgs\nlxE2VknYCCF8w+7du5kxYwYrVqxAp3NNGGRnZzN//ny++OIL+vXr56Ee+p7T5+3TofT+WiIall3D\nZkfabt478glWxcrXp7ZwX8cxXBPapra6KYSox1JSUpg4cSJWq5X09HT+8pe/sH79enbt2sVbb71F\np06dPN1Fj7DJKlFCCC9V7ipRzZo1K/FlNBo5cOAA+/btIzs729leX7lOiZIaNkII3/DOO++QmJjI\n1KlTyc/Pd7Zv2bKFO+64g59++omFCxeyZs0aD/bSt5w+nwNAy8gg52qKf7b73F7WH/4Iq2IFIKMg\nk38feJMLeRdrrZ9CiPpr8eLFDBgwgB9++MGZrF+6dCk33XQTL7zwgod75zkywkYI4a3cXtb78uXL\nxMXFMXLkSJ577jnmz5/PXXfdxfjx48nJyXHrNRITExk9ejSxsbGMGjWK/fv3l7rfBx98wNChQ+nR\nowd33303e/bscbebtU6juXJht8iUKCGEj2jfvj3//e9/SUlJ4aGHHuL8+fPMmTOHuLg4unbtyqZN\nmxgzZoynu+lTHAmbFlFBpW632qxsOvEVAG0atiK+xzQa6oIx2yy8e+STMqcfCyFEdfn111954IEH\nXGqYabVapk2bxu+//+7BnnmW1TnCxu1/GgkhRK1w+6q0ePFiLly4wOeff86uXbvYs2cP//vf/ygo\nKODll1++6vGFhYXExcVx11138csvvzBhwgSmTZuG0Wh02W/nzp0sXbqUZcuWsWfPHu677z7i4uK4\ndOlSxd9dLVCrZEqUEMI3tWjRgvXr13P58mUGDRrEli1bePXVV3n99deJiorydPd8ik1ROF20QlR0\nVOn1a3af30dGgf2z7J72d9E2JIYx1/wFgD8uHWPnuV9rp7NCiHpLp9Nx+fLlEu2pqakEBgZ6oEfe\nwVl0uIzRkUII4SluJ2y+//57nn32Wdq0uTLPvl27dsybN4+vv/76qsfv3LkTtVrN+PHj8fPzY/To\n0TRq1IitW7e67Hfu3DkeeughOnbsiFqt5s4770Sj0XDs2LEKvK3ao1KpnNOiZEqUEMLXREVFsX79\nejp37kxERITUrKmki1n5FJjs05xalpKwsSk2vj75PQCxEV1oGtTY+X23Rp0B+Oz4FxRYCmqpx0KI\n+mjkyJEsXLjQOZomOzubrVu3Mm/ePIYPH+7h3nmOVWrYCCG8VLlFh1121Grx9/cv0R4QEIDZbL7q\n8cnJyS7JHrAvBX7ixAmXtr/85S8uP//6668YjcYSx5ZFpVJRmdGMjjmrlZm7qtGosNoUFFynSNUV\nVYlNXSexKZ/Ep2yejs1PP/3k8vNDDz3EwoULmTBhAk899RQajca5bcCAAbXdPZ+TdNI+csZfp6FJ\nuKHE9r3nD3AhPx2A21oNdrarVCrubjecxIzDXDbl8M2pLYxoc1vtdFoIUe88/vjjLF26lPHjx2My\nmRg9ejRarZZ7772XWbNmebp7HuMsOlwH7+OFEL7N7YRNv379ePnll1m6dCkhISEAZGZm8sorr7j1\nRDYvLw+9Xu/SFhAQQEFB2U8Tjx07xqOPPsqjjz5KWFiYW/0MDw8ss9ijO0JCKj4c1E+jxmS2odfr\nCAsrvXZBXVCZ2NQXEpvySXzK5qnYTJ48udT29PR0pk6d6vxZpVKRlJRUW93yWfuP2ZMxXVqFodW4\nPjWwKTa+PGUfXdMlvCMtgl0L9TfSh3Nzixv45vQWvkvZRt+mvWikd+8zTwghKkKr1fLUU0/x2GOP\ncfr0aaxWKy1btsRgMJCRkUF4eLinu1jrbIr9oSvIAyYhhPdxO2Hz9NNPM3HiRAYOHEjz5s0B+3zX\nNm3a8Pzzz1/1eL1eXyI5U1BQgMFQ8kkk2J/+xsfHM2nSJKZMmeJuN8nIMFZ6hE1ISCBZWUZnlt1d\njgRR9uV8MjNzK35yL1eV2NR1EpvySXzKVpXYVEdi+PDhw1V+DWFXaLaSdMo+wqZ720Ylth+4eIiz\nxvMADIsZXGI7wK2tbmbn2T3kmHNZf/gjZsZORq2S4pdCiOrVsWNHtm/fTlhYGO3atXO2p6SkMHLk\nSPbt2+fB3nlG8c9gjdSwEUJ4GbcTNhEREXz22Wds27aNEydO4O/vT+vWrenXr59bI1pat27NunXr\nXNqSk5NLnS+7YcMGFi9ezIIFCyo8n1ZRFKzWCh3iwmZTKlw82DHf1Wy21enCw5WJTX0hsSmfxKds\nEhvfl3gyE7PFhgro1qbk0+kdZ3cD0CG0Ha0atCz1NfRaPePa38kbv6/lj0vH+OnMTm5sLvWEhBBV\nt2HDBj788EPAfp/88MMP4+fn57LPxYsXiYyM9ET3PM5aLGEjI2yEEN7G7YQN2IdR3nzzzS71DBz1\na3Q6XbnH9u3bF5PJxNq1a7nnnnvYuHEj6enpJWoj/Pzzzzz33HOsWbOG6667riLd8xjHfFerjCAQ\nQoh650DRdKjWzRrQILDkZ2FmQRYAHcLaldhW3LWRXekR2Y29Fw7y0dH/EWmIuOoxQghxNcOGDePs\n2bMA7N+/n169epVYESowMJChQ4d6onse5zLCRhI2Qggv43bCZvfu3Tz33HOcPHkSW7HVkBRFcavG\ngU6nY/Xq1cyfP5+lS5cSHR3N66+/jsFgYN68eQAsWLCA1atXYzabefjhh12OX7ZsGTfeeGNF3lut\nubJKlCRshBCiPik0Wfn1yEUAYkuZDgWQY8oBoIGu9OW+ixvX/k5Sc9K4kJ/Oqt/+w7Ruk2gX6l7R\nfSGEKI3BYOCRRx4BoFmzZtxxxx1XfdBan8gIGyGEN3M7YfPMM8/Qtm1bZs+eTUBAQKVO1qFDB957\n770S7QsWLHB+v2bNmkq9tidpiormWK2yrLcQQtQnPx5Mw1hgQaNW0a9LkxLbrTYrRnMeAMG6q9ce\nCvIL5JHYySz59TWyTTks37+aka1vY1CLG9CoNVc9Xggh/uz999/nzjvvRKfTYTKZ+OSTT8rcd9y4\ncbXYM+9glRE2Qggv5nbC5sKFCyQkJBATE1OT/fFJzilRioywEULUT5s3b2bFihWcO3eOpk2bEh8f\nz5AhQ/jtt98YO3asS6J/6tSpxMXFoSgKS5cu5cMPP8RqtTJq1Cj+/ve/uywp7s2sNhtf/5ICQJ/O\nUYQG+5fYJ8d8pRB9sBsjbADC9WE8du1UVv32DufyLvDp8c3sPPcrI1vfRtdGHaUYsRCiQlauXMnQ\noUPR6XSsXLmyzP1UKpXbCZuDBw8yffp0fvrpJwCys7OZM2cOO3fuJDg4mBkzZjBmzBgAr7/W22SE\njRDCi7mdsLnlllvYunWrJGxK4ZwSJYVDhRD1UHJyMnPmzGHNmjX06NGDHTt2MGXKFLZt20ZSUhI3\n3nhjqf9IWL9+PVu2bOGzzz5DpVIxdepU1qxZU2JKrLfaeeg86dn21Q9vub45G49/AcCgFjc4R9Pk\nmIzO/YN17i/hHhUYyZPXzeTDoxvZeXYP54znWfXbf2gSGMXQ6JvpGdldRtwIIdzyxhtvkJWVRVZW\nFm+++WaVXktRFDZs2MCLL77oknB55plnMBgM7NixgyNHjvDwww/Trl07YmNjvf5aby1W6kFG2Agh\nvI3bCZtZs2YxcuRINm3aRIsWLVD/ae3sJUuWVHvnfIVzSpTUsBFC1EMxMTFs376dwMBALBYL6enp\nBAYGotPpSExMpEOHDqUet3HjRiZOnOhcmWTq1KksW7bMa27iHY6dySbraAbXtgkF7DfzGdkF/Pfb\no4C9ds1F5QRfn/oBgC2p23mw83i6NurkrF8DEOxXseXYA7T+TOg4lhua9WHj8S/549IxzhrP85/E\n99h04muGtBxI3ybX4afxu/qLCSHqrdtvvx2VSuWsO1kad2tSJiQk8MUXXxAXF8fq1asBMBqNfPvt\nt3z11Vf4+/vTrVs3hg8fzqeffkpsbKzXX+ul6LAQwpu5nbCZO3cuKpWK5s2bV7qGTV11ZZUoqWEj\nhKifAgMDSUlJ4dZbb8VmszF//nyCgoJISkpCp9MxaNAgbDYbw4YNIz4+Hp1Ox4kTJ2jbtq3zNWJi\nYkhOTi73HxXFqVQq1LUwO2j1Z4mcy8zj5h7NmHhbe/IKLazedIj8QguBAVomDmvPeyfede5vsprY\nnraL2KjOGC32KVGBfgZ0fhVamNGpTWg0s66bSnL2ab5M/p4DFw+RUZDJ+398whcnv2FQyxu4oXkf\nAv0M1fJ+S+OYJlCfpwtIDCQG4Jsx+O6776rtte6++27i4uLYvXu3s+3UqVNotVpatGjhbIuJieHr\nr78GqPK1Hip3vXf7d1Vss5+f2nlfX5f54t9xbZL4lE1iU7aaio3bd4979uxh3bp1dO3atVo7UBdo\nZUqUEELQpEkTDhw4wJ49e5g+fTrR0dGEhobSu3dvxo0bR0ZGBo899hjLly/niSeeID8/3+UBgF6v\nx2azYTKZ8PcvWQ/mz8LDA92+2a+KG3s054Nv/+CHvWfIybdw+txlLlzKB2Dm2Gtp3ETPoV+OANA0\nOIq0nPNkmy8TFhaE5YIJgFB9Q8LCKjbC5s/CwjrRM6YTKdlpbEz6mp9O/8JlUy6fHvuCL5K/48ZW\nvRl2zc00b1Cy+HF1CQlxf1pXXSUxkBiAb8WgWbNm1fZajlEyxeXl5ZV4mBsQEEBBgX3KaFWv9VC1\n6/3Vflc5pisPXMPDgghrqK/UeXyRL/0de4LEp2wSm7JVd2zcTthER0djMpmq9eR1hSzrLYQQoNXa\nP1L69u3L0KFD+e6770hISHBuNxgMTJ06laVLl/LEE08QEBBAYWGhc3t+fj5ardbtG/iMDGOtjLC5\no08LUi/ksOPgWfYknQdAq1Exfsg1dGzRgO+O/IzVZsVPreWmZv357+GPuWDMIDMzl3NZGQAEagxk\nZuaWdxq3BdKA8deMZmiLQXxzcgs/p+2h0Grim+M/8s3xH2kT0oo+Ta7juqhu6P2q5x8earWKkJBA\nsrKMLtMH6hOJgcQAaj8GVU301ga9Xu9yLQcoKCjAYLCP+qvqtR4qd71393d16dKVWmOXs/NQWa0V\nO5EPkv+XyyfxKZvEpmxVjU1Z13u3EzbTpk3j6aefZsKECbRs2dJ5Y+4wYMCACneqrtBoHMt6yx+t\nEKL+2bp1K2+99RZvv/22s81sNqMoCi+99BIzZswgKMj+IVRYWOi8SW/Tpg3Jycl0794dsBcvbt26\ntdvnVRSF2riv1mhUzBrfk2bhSaReyEWjVjO4Z3OiGwdjtSrsPrsPgC6NOtHY0BiAAksBOQV5XC60\nJ2mC/IKq/TMiVBfK2GvuZHjMrWxP283W1B1cKszieNZJjmed5IMjn9IprD1dIzrTNbwjQRUoelwW\nm02p9591EgOJAUgMiouOjsZsNpOWlkbTpk0B+/XcMQ2qqtd6qNr1/mq/K5O5eEkDVb36vcrfcfkk\nPmWT2JStumPjdsImPj4egOeff77ENneKlNVlV0bYSA0bIUT906lTJ37//Xc+/fRTRo4cyY8//sjW\nrVt5//33nct3P/7446SlpZGQkMDYsWMBGDlyJG+++SZ9+vRBq9WycuVKRo0a5eF3Uzp/Pw3D+7Uq\n8QF8qSCLo1knALg+KpawgBDntsyCS+SY7AmbBm4u6V0ZBj8Dt0TfxKAWN5CU+Qc7z/3KbxcPYbZZ\nOJB+iAPph1ChonXDVnSL6ESnsPY0CYyqlelkQoi6LygoiMGDB7NkyRIWLVrE0aNH2bRpE6tWrQK8\n/1rvsqy3XBeFEF7G7YTN4cOHa7IfPs2RsLHIsDAhRD0UERFBQkICzz//PAsWLKBVq1a89tprtG3b\nloSEBBYtWkSfPn0ICAhg3LhxTJw4EYDx48eTnp7O6NGjMZvNjBgxgkmTJnn43VTMnvP7AdBr9XQK\n74BGpUatUmNTbGQWXOJy0SpRjmW+a5JGraFLo450adSRPHMe+y7+xsGLiRy5dBSzzcLx7GSOZyfz\nCZ8TrAuifWhb2oe2o31oW8L1oTXePyFE3bVw4UKeffZZBg4ciMFg4Mknn3SOqPH2a71VVokSQnix\nyi1ZIVzIlCghRH133XXX8fHHH5dob9u2rctUqeI0Gg3x8fHOEZy+6Jfz9ulQPSK74qe2f6SG+oeQ\nUZBJZkGWc4RNbSRsijP4GejftDf9m/am0GoiKfMPDl48xKGMw+SajeSYctlzfr8z4RTi35CYhtG0\nbtCSmIbRNA9u5nw/QgjxZ71792bXrl3On0NCQli2bFmp+3r7td5lhI0kbIQQXkbuxqqBTIkSQoj6\nJy33HGdyzwJwXdS1zvawAHvCJqMgk1yzvZhlbSdsivPX6IiN6EJsRBdsio2zxvMczjzc5VojAAAg\nAElEQVTK4UtHOZaVjMlqIqswm30XDrLvwkEANCoNjQMjaRbUhGZBTWjRoCmd9W1QlFqo8iyEELXI\nqhQbYVMPlvQWQvgWSdhUA1klSggh6p99F38D7KNT2obEONvDAuzTi1JyzmBT7In8mqxhUxFqldqZ\nhBnc8kYsNgunc86QnH2K5OxTnMg+RbbpMlbFypncs86EFAB7wV/jT4Q+3P5laESEvhGRhkaE+jek\noX8DtDIqRwjhYxwjbFRIDRshhPeRO6tqIFOihBCi/knKOAJAt0adUKuujDxxJGxO5aQ62zw5wqY8\nWrWW1g2jad0wGrCvxHKpMIvTl1OdCZvU3LNkFGQCUGgtJDU3jdTctFJfL9gviBD/BoQENKShf0Ma\n6oIJ9Ask0M9w5UsbSJAuEJ3aTwofCyE8zvHAVaZDCSG8kSRsqoGMsBFCiPol12zk5OUUADqFt3fZ\n5kjYmKwmZ1uwn3cmbP5MpVIRFhBKWEAosZFdne0mpZACrZFjZ09zzniRi/kZXMxL52J+hnPaF0CO\nOZcccy4pZSR0itOqtRi0egI0/vhrdPhr/fEv+t7e5l/UpsNP7YdWrUWr1uKn0ji/v/Klse9TbJu6\nqAC08wsVapUalUrlkmATQtRvjhE2UnBYCOGNJGFTDaSGjRBC1C9HMo+ioKBRaWgX0sZlW/GlvQH0\n2gD8NH612b1qp9cG0CysEaGElxhNmm/JJ6vwMlkF2WQVun7lmIzkmo0YzXkUWAtcjrPYLFw25XCZ\nnNp8KwCoUDkTN45EjuNLpVKhxvGzChUqKPqvVqMuVqBUhQqKtjl/chk1pPrTPs4WVbFtqLAf4vqz\n46fKvr+KH3T1Y1Qq8NNqMFusOMp+VLaXVXl/FT5XNY7k8tNoubvrbURpmlTbawrPsskIGyGEF5OE\nTTVwFCiTKVFCCFE/JGb8AUCbkBgCtP4u2xwjbBx6RHartX55gl6rR6/V0yQwqtz9rDYrRkseuSZ7\nAsdoySPPnE+htZBCq6nov4UUWkwUOL63FlJgKcRis2C2WbAoFiw2KxabBYvNgkLlPncVFBRFcdYY\nEqIiAo/qmdjhHk93Q1QTS9EDVxlhI4TwRpKwqQZ+RTVsLFa58RNCiLpOURQSM+31azqFXVNie2ix\nETah/iGMaTeq1vrmzTRqDQ10wdVagNlqs2JRriRwnF+KFatiRVEUrIoNm2JDUWzYFAUbRf9VrNgU\nxd6OPXlz5cveblVsUJQWUqnAYNBhzCvEZlPsrYp9myNxpBRbbca+vSg55GhRHFuK2pRi24r9XJlE\nVPFz1wQFBZUK9Hod+fkmrna6Cr+HCu9e8++3NDqNH8M6DQRzjZ5e1CIZYSOE8GaSsKkGflp7wsZk\nkYSNEELUdRfy07lssk/j6VBKwsZPreXudiM4ffkMo9uN8PnpUN5Mo9agQYO/Rlfz59KoCAsLIjMz\nt96OqJUYFMUg2B4DUTdI0WEhhDeThE010PlpADCZrR7uiRBCiJqWmnMGsCdmmpYxDWhQixtqs0tC\nCCEqyTHCRisJGyGEF5JlEqqBvyNhIyNshBCizkvJsa+A1DSwCRq1xsO9EUIIURUyJUoI4c0kYVMN\nnFOiZISNEELUealFS1Y3D27q4Z4IIYSoqitTouSfRUII7yNXpmqg08oIGyGEqA8URSGlaEpUC0nY\nCCGEz3OMsJFVooQQ3kgSNtVA52cPo9liX1lCCCFE3ZRtukyu2QhA86BmHu6NEEKIqrIW3burVZKw\nEUJ4H0nYVAOd9koYzTLKRggh6izH6BoVKpoFNfZwb4QQQlSVY8UzGWEjhPBGkrCpBo5VokDq2Agh\nRF2WWlRwOCowEl0tLCUthBCiZknRYSGEN5OETTWQETZCCFE/pOaeBaBFkNSvEUKIusAqNWyEEF6s\nVhM2iYmJjB49mtjYWEaNGsX+/fvL3f/tt9/m0UcfraXeVV7xETaFMsJGCCHqrIz8DACiDBEe7okQ\nQojq4Kg/KSNshBDeqNYSNoWFhcTFxXHXXXfxyy+/MGHCBKZNm4bRaCyxb15eHi+//DIvvvhibXWv\nSmSEjRBC1A+ZhVkAhAWEergnQgghqoOMsBFCeDNtbZ1o586dqNVqxo8fD8Do0aP5z3/+w9atW7n9\n9ttd9n3kkUfQ6/WMGzeOS5cuVeg8KpUKdSXSUI6semWy6/qAK2G02GxoNHXrgl+V2NR1EpvySXzK\nJrHxPYVWE0ZzHgChASEe7o0QQojqIMt6CyG8Wa0lbJKTk2nTpo1LW0xMDCdOnCix7wsvvEBUVBQr\nVqyocMImPDwQVRWW5QsJCazwMRbrlVE1AQH+hIUFVfr83qwysakvJDblk/iUTWLjOy4VZDm/D5OE\njRBC1AlWKToshPBitZawycvLQ6/Xu7QFBARQUFBQYt+oqKhKnycjw1jpETYhIYFkZRmdmfaK0KhV\nWG0K6Zm5ZGbqr36AD6lqbOoyiU35JD5lq0psvDEpvHnzZlasWMG5c+do2rQp8fHxDBkyhHPnzrFg\nwQJ+/fVXtFott912G7Nnz0an06EoCj179kRRrrz/nj178sYbb3jwnZQts+DKA4SG/g092BMhhPBO\nb775Jv/4xz/w8/Nztq1evZp27doxZ84cdu7cSXBwMDNmzGDMmDEe7OkVMsJGCOHNai1ho9frSyRn\nCgoKMBgM1XoeRVGwVqHur82mYLVW/B+WOj81+YVWCkzWSh3vCyobm/pAYlM+iU/Z6kJskpOTmTNn\nDmvWrKFHjx7s2LGDKVOmsG3bNp588knatWvHtm3buHz5MjNmzOC1114jPj6eU6dOAbB3794qjYys\nLZn59hE2DXTB+Klr7eNTCCF8RmJiIvHx8Tz00EMu7Y8++igGg4EdO3Zw5MgRHn74Ydq1a0dsbKyH\nenqFuWikvEYji+cKIbxPrV2ZWrduTXJysktbcvL/t3fvUVHW+R/A33O/AAICKiQigoGwKSIq/NRU\nLC1XZXfTXVfb3ZOVt1Y7utqWlaGuq+e4Xigz05O2XnbdbPenpdSaadRPV4syDQq6MJsYkgJyZ+7P\n749hRkYdGGCGeWDer3M8Mzw8M8+XD+PnO3zm83wfHeLj47tqCF6lkNuuFGU0cdFhIvIvsbGxOHPm\nDFJTU2E2m1FRUYGAgAAolUpoNBosWrQIKpUKERERmD59Oi5cuADA9sY+ISGhWxRrAKCq+ZQorl9D\nRHRnX331FYYMGeK0raGhASdPnsTSpUuhUqkwdOhQTJs2DUeOHPHRKJ3pDWYAgEYpa2NPIqKu12Uf\nEWZkZMBoNGL//v2YPXs2jh49ioqKCowdO7arhuBV9itFGc28rDcR+Z+AgACUlpZiypQpsFqtyM7O\nRmBgIHbt2uW03+nTp5GYmAjA9sa+vr4eWVlZuHbtGkaOHIlnn33W7dNiO7rIfHvZ1zW40XyFqDB1\naI9bXL4tXCSbMQAYA4AxaE1TUxN0Oh327duHlStXolevXnj00UeRlJQEuVyO6Ohox76xsbE4ceKE\n28/dkXzv7u9Kb7K9d9eq5X6T2/k6bh3j4xpj45q3YtNlBRulUondu3cjOzsbW7ZsQUxMDF555RVo\ntVqsXr0aALB27dquGo7HKRXssCEi/xYZGYmLFy8iPz8fixcvRkxMDDIyMgDYTlddv349SkpKsGnT\nJgC2eSElJQVPPvkkVCoV1q9fjyVLluCNN95w63idXWS+verMdQCAqJAIUa4j1BW4SDZjADAGAGNw\nJxUVFRgxYgR+/etf48UXX8SlS5ewcOFCPPLII1Cr1U77ulrH0pXO5Pu2flem5tOSe4do/S6383Xc\nOsbHNcbGNU/HpktPwk9MTMShQ4du2+6qULNkyRJvD8lj2GFDRP5OLrdNKRkZGZg8eTLef/99ZGRk\nQK/X46mnnkJxcTH279+PsLAwALfn+D/+8Y9IT0/HtWvX0KdPnzaP19FF5tvLvkD0tfpKAIBGEoCq\nqnrvH1hEuIA4YwAwBkDXx6A7FRCio6Nx4MABx9dpaWnIyspCfn4+DAaD077tXceyI/ne3d9VXYPR\ndsdq9Zvczv/LrWN8XGNsXOtsbFzle66a6CHssCEif5WXl4e9e/fi9ddfd2wzmUwICgpCdXU1Hnvs\nMWi1WvzjH/9ASMjN9V927dqFMWPGIDk5GQBgNNreNKtUKreO29lF5tvDKlgda9iEKEO6/ULRHdUT\nFsnuLMaAMQAYgzspLCzEmTNnMH/+fMc2g8GAyMhImEwmlJWVISoqCkD717HsTL5v63fV1LyGjUoh\n87vfKV/HrWN8XGNsXPN0bLgcuoeww4aI/FVSUhIKCgpw5MgRWK1W5OXlIS8vD9OmTcOSJUsQHh6O\n1157zalYAwAlJSXYuHEjbty4gbq6Oqxfvx6TJk1CcLD4Lplda6iH2Wp7U99bxUWHiYhupdVqsX37\ndrz77ruwWq34z3/+g+PHj2Pu3LmYNGkSNm/ejKamJly6dAnHjh3D9OnTfT1kAIDeaMvtai46TEQi\nxA4bD2GHjTgJggCLtfmfxQpzc8XTbLHCYm2+tQgwW62wWgUIAppvBVgFNN+2uG+9uU0QAEgArVaF\n+no9zGYrBDg/3ioIQHOBVbg5KMd9oUXxVcDt+wotdmjxVE7P0fIxt+5/89vC7cfzYFG8xWicSCUS\nqNUK6PUmWyzcf0KP8Wjt34NPJpECacmRiO/XfdrdXYmIiMDOnTvx5z//GWvXrsXAgQPx8ssvo6am\nBh9//DFUKhVGjRrl2D8pKQkHDx7Ec889h/Xr1+PBBx+EyWTChAkTsG7dOh/+JK5VNFQ57vMqUURE\nt4uNjcW2bduwdetWPP300+jbty82bNiA5ORkrFu3Di+88ALGjx8PrVaLlStXYtiwYb4eMqyCAL3B\n9mGrRsU/i4hIfJiZPMTeYWNih027CYIAk9mKRoMZTQYzGvVmNDbfNhls9/VGM4wmK4wmCwwmK4xm\ni+Nr+32DyQKj2WorzFgEWKy2WyKxeu+TK9i5Yjyk3eSy1q1JS0vDv/71r9u2FxcXu3xMYGAgNmzY\n4M1heUxl0w0AgFwqR6CCC+0REd1JZmYmMjMzb9seEhKCnJwcH4yodQajxfFZjEbJP4uISHyYmTxE\nqWg+JYodNg4mswWVtQZU1uhRXW9AbaMRtQ1G1DaYUNtoRF2DETWNRjQ0mURdWJHAtoiURGLrGLFf\nWlICCaRSie0SkAJs35dKmrfbLkEpkUggge17Ts/ZvN1+AEmLo9n3df6+5LZ9bftJWtxv/spxX9Li\nfsvnk9zc5uU6gUQigUIhg8lkcer+cfexYuSpYUkkEqQl9YNCLuU5wN1AncG2EGWQIlC0r00iImof\nvfHmB61qFU+JIiLxYcHGQ5RyW5I3+FGHjSAIqG004WpFA65WNeJ6dRMqa/SorNWjskaPGvuq+x2k\nUcmhVcmgUSmgVsmgUsiglEtttwoplHIZlC3uqxRSKOQyyOUSyKVSyGQSyGVSyKUSyGTNXzdvl0lt\n33PcyiSQSmwFGKnE9se0rTjTeuFAJpOgd+9AVFXV84/uO2B8XGsZGxK/emMjAECr0Ph4JERE5CmN\nzQsOA+ywISJxYmbyEEVzh42ph3bYGEwWfPtDDUp+qMXlH+twtbIRVysb0KA3t/1gAIEaBYIDlOgV\noESQVoFeAUr00tq+DtQoEKCWNxdo5NCq5VAr5ZBK+Sk2EYlDg71gI2fBhoiop9C3LNhwDRsiEiFm\nJg9RNXfY9ISrRAmCgPKqRhSXVqPkh1r898c6lFW0fj354EAl+oZoEBasRliwBuHBaoT1Utu+7qWC\nQs42UyLqvupZsCEi6nGajDcLNrxKFBGJEQs2HmLvsDGau1+Hjb1AU3S5GsWXb6D4crXL05l6BSgx\nsF8QosIDEBmmRVSY7VarVnTxqImIuo6jw0ah9fFIiIjIU+xXiFIpZezsJiJRYsHGQ+xr2BhN3aPD\nxmyx4pvSanz+bSUufluBa9VNt+2jUckQf1cIBkUF4Z7BfRAWqEAvrZILbhKR32kwNQBghw0RUU/S\n1HxKlIbdNUQkUizYeIiyG3TYmMxWXPquAp8UXcMXJVWOScpOq5Lj7ugQJAwIQeKAUET3CXRcBYkL\nxxKRP6s3cNFhIqKepqn5KlFcv4aIxIrZyUNudtiIq2BjFQR8U1qN/xT+iPyia06r4QNA/4gADIsP\nR0p8OGIje7EdlIjoDupNXMOGiKinsS86rOYVoohIpJidPMTRYSOSU6Iqa/TIu/gDzhaUo6rW4Ngu\nAZAwIAQjEvpgWFwYwkP4xwcRUVu4hg0RUc9jX3RYo+IpUUQkTizYeIi9w8ZiFWCxWiGTSrt8DFZB\nwJe6Kpz67Adc/K4CQouzl/pHBCLjJ30xekhf9O6l7vKxERF1V1bBikaTbZ0vdtgQEfUcTc2LDmvY\nYUNEIsXs5CH2DhvAdlqURtV1BZtGvRkfXSrD6Qs/4NqNm4sHB2oUGHtPJDJ+0g/RfQK7bDxERD2J\nvVgDcA0bIqKeRN/cYaNmhw0RiRQLNh5i77ABbIv7alTeP2Z1vQHv5Zfigws/OD4hAIC4u3ohc3h/\npCVGQCHnBERE1BmN5hYFGzlPiSIi6inYYUNEYsfs5CHOHTbeXcemvKoR757/HmcLymFuvmqTXCbF\n//ykLyYO74+YfkFePT4RkT9hhw0RUc/U5Oiw4Z9ERCROzE4e0rLDxuClS3uXlNXinXPf47Ovr8O+\nPI1WJUfmiLswaUQ0ggOUXjkuEZE/azQ3Ou5zDRsiop7DfpUoLjpMRGLFgo2HKFp02JjMnuuwEQQB\nX5RU4d3z36PocrVje2iQClNGRmPcsCho+KkAEZHXNDR32KjlakglXb+gPBEReUeTo2DD99JEJE7M\nTh6iatFhYzR1vsPGbLHik6JreOfcZVy5Xu/YHhUegAdHD8DopL6Qy/iHAxGRtzWabB02AeyuISLq\nUbiGDRGJHbOTh7TssDF2osOmyWDG/126ihOflKKyVu/YPrh/MB5Mj8HQuDBIJZJOjZWIiNxnX3RY\nq+CCw0REPYUgCI41bHhKFBGJFQs2HiKVSCCXSWG2WDvUYVNZo8f7n15B3kXnKz6lxIdjanoM4vsH\ne3K4RETkJvuiw1xwmIio5zCarBCaF4VUs8OGiESK2cmDVAp7wca9DhtBEFBSVov38kuRX3QdVsF+\nxScJ0pP6YcroAbgrPMCbQyYiojY0NJ8SxQWHiYh6Dnt3DcA1bIhIvJidPEght50WZWzjKlE36gw4\nV1iO//viKq5W3rz6SKBGgczUuzAxtT+v+EREJBL2U6ICeEoUEVGPYV9wGAA0Sp4SRUTixIKNBykV\ntmR/pw6bipomXPimAp9/U4GiyzccLZgAEBmmxeSR0chI7ud4DiKi7iQ3NxcvvfQSysvLERUVhWXL\nluG+++5DTU0NVq1ahXPnziEoKAhPPPEEZs2aBcDWZbhlyxYcPnwYFosFWVlZeOaZZyCTiSsP8pQo\nIqLO+/LLL7F69Wp8++23iImJwZo1a5CSkuKz8eiNN9+vq9lhQ0Qi1aXZyd1EfezYMWzduhWVlZUY\nPXo01q9fj/Dw8K4caocomztsTGbbaVHfXKnBl/+tQqGuCpev1Tvtq1HJMGpIX4y5JxJxUb0g4ULC\nRNRN6XQ6rFq1Cnv27EFqairOnj2L+fPn48MPP0R2dja0Wi3Onj2L4uJiPP744xg8eDBSUlJw8OBB\nfPDBB3jrrbcgkUiwYMEC7NmzB48//rivfyQnPCWKiKhzDAYDFi5ciIULF2LWrFk4evQoFi1ahJMn\nTyIgwDen/7fssFGzw4aIRKrLCjbuJuqioiK88MIL2LNnDxISErBu3To888wz2L17d1cNtcPs3THv\n5Zfifz/SwWxxPjVKo5JjWFwYht8dgWFxYeymIaIeITY2FmfOnEFAQADMZjMqKioQEBAApVKJkydP\n4t///jdUKhWGDh2KadOm4ciRI0hJScHRo0fxu9/9Dn369AEALFiwADk5OaIr2Ny8ShQLNkREHXHu\n3DlIpVLMmTMHADBz5kz89a9/RV5eHqZOneqVY3713yrUFVegocHgWCeypdLmD1OVcinkMult3yci\nEoMuK9i4m6jffvttTJo0CcOGDQMArFixAhkZGaioqBB9l42quQBTXW90bOsfEYCkgb1xT1wYEqJD\nOCEQUY8UEBCA0tJSTJkyBVarFdnZ2bh8+TLkcjmio6Md+8XGxuLEiRMAgJKSEsTHxzt9T6fTQRAE\nt7oOJRIJpF2QUo0WW04PUgZAJvPPbkipVOJ0648YA8YAYAw6SqfTIS4uzmlbbGwsSkpK3Hp8e/N9\n6bV6bDx4wa19NSq53+V2vo5bx/i4xti45q3YdFnBxt1EXVJSguHDhzu+Dg0NRXBwMHQ6nVsFm46+\ngfdEgCem3oW6JhMG9gtCcmxvJA0MRUigqsPPJxb8j+kaY9M6xse1nhibyMhIXLx4Efn5+Vi8eDEe\nffRRqNVqp33UajX0ej0AoKmpyen7Go0GVqsVRqMRKlXbuTMsLKBLTif9acIkfHX9a/xPXCo0CnXb\nD+jBQkJ45ULGgDEAGIP2amxshEbj3KXYcj5oS3vzvSZAjWGDw1FW0dDqflKJBNPHDULv3oFuP3dP\nwtdx6xgf1xgb1zwdmy4r2LibqG99Aw/Y3sQ3NTW5dZzOvoHvTIAfHBuIB8fGtb1jN8X/mK4xNq1j\nfFzrSbGRy21TSkZGBiZPnoyCggIYDAanffR6PbRa29WW1Gq10/ebmpogl8vdKtYAQGVlQ5d02Nx3\n13jMTJ6K6uoGNFnr235ADySVShASEoDq6gZYrbefWuAPGAPGAOj6GPSUQoJGo7ntPX/L+aAtHcn3\nK2anuP27qqryr9zO/8utY3xcY2xc62xsXOX7LivYuJuoXRVxvJnQAb74WsPYuMbYtI7xca0zsRHb\nG/i8vDzs3bsXr7/+umObyWTCgAED8OGHH6KsrAxRUVEAbN2W9tOg4uLioNPpHKfA6nQ6DBo0yO3j\nCoIAy+0X5fMaq1WAxeLfr2PGgDEAGAOAMWivQYMG4cCBA07bdDodpk2b5tbjO5Pv+btyjbFpHePj\nGmPjmqdj02UFG3cTtf0NvF1VVRVqampuO53Klc6+geeLzzXGxjXGpnWMj2s9ITZJSUkoKCjAkSNH\nMGPGDHz00UfIy8vDG2+8gatXr2Lz5s3405/+hG+++QbHjh3Drl27AAAzZszAa6+9hvT0dMjlcrz6\n6qvIysry8U9DRESelpGRAaPRiP3792P27Nk4evQoKioqMHbsWF8PjYhI1LpsBdyWidpkMuHNN9+8\nY6KeNm0aTpw4gfz8fBgMBmzZsgX33nsvQkNDu2qoRETUDhEREdi5cyf27duHtLQ05OTk4OWXX0Zc\nXBzWrVsHs9mM8ePHY+nSpVi5cqWjo2bOnDnIzMzEzJkz8dOf/hSpqal45JFHfPzTEBGRpymVSuze\nvRvHjx/HqFGjcODAAbzyyitud9ATEfkriSDc4Tp3XlJUVITs7GwUFxcjJiYG2dnZSElJwerVqwEA\na9euBQDk5uYiJycH169fR1paGjZs2ICwsDC3jnH9el2HxiaTSdC7dyCqquq7/afdnsbYuMbYtI7x\nca0zsYmICPLSqLqXjub79uLrmDEAGAOAMQC6PgbM9zYdyfd8vbrG2LSO8XGNsXGts7Fxle+77JQo\nAEhMTMShQ4du224v1NhNnTrV6VLfRERERERERET+pMtOiSIiIiIiIiIiIvewYENEREREREREJDJd\nuoYNERERERERERG1jR02REREREREREQiw4INEREREREREZHIsGBDRERERERERCQyLNgQERERERER\nEYkMCzZERERERERERCLDgg0RERERERERkciwYENEREREREREJDIs2BARERERERERiQwLNkRERERE\nREREIuP3BZsvv/wSM2fOREpKCrKysvD555/7ekiikp+fj1mzZmHEiBG47777cOjQIV8PSXQqKiqQ\nkZGB06dP+3ooolFeXo4FCxYgNTUV9957L/bt2+frIYnKZ599hl/84hdITU3FlClT8Pbbb/t6SOQC\n5wjOA3b+nuuZ15m7uxPmbteY09vm7/neFc4Drnl1fhD8mF6vF8aNGyccPHhQMBqNwuHDh4X09HSh\nvr7e10MTherqamHkyJHCW2+9JVgsFqGgoEAYOXKkcObMGV8PTVTmz58vJCYmCqdOnfL1UETBarUK\nP//5z4WNGzcKRqNR+Prrr4WRI0cKn376qa+HJgpms1lIT08X3nnnHUEQBOGTTz4RkpKShNLSUh+P\njG7FOYLzQEv+nOuZ15m7uxPmbteY093jz/neFc4Drnl7fvDrDptz585BKpVizpw5UCgUmDlzJsLD\nw5GXl+froYlCWVkZxo8fj+nTp0MqlSI5ORmjR4/GZ5995uuhicbf//53aDQaREZG+nooonHx4kVc\nu3YNK1asgEKhwODBg3Ho0CHExsb6emiiUFtbi6qqKlgsFgiCAIlEAoVCAZlM5uuh0S04R3AesPP3\nXM+8ztzdnTB3u8ac3jZ/z/eucB5wzdvzg18XbHQ6HeLi4py2xcbGoqSkxEcjEpchQ4Zg06ZNjq9r\namqQn5+PxMREH45KPHQ6Hfbu3Yvs7GxfD0VUCgsLMXjwYGzatAljxozBlClTcPHiRYSGhvp6aKIQ\nGhqKOXPmYPny5UhOTsbcuXPx/PPP842BCHGO4DwAMNcDzOsAc3d3wtztGnN665jvXeM84Jq35we5\nR56lm2psbIRGo3HaplarodfrfTQi8aqrq8PChQuRnJyMzMxMXw/H58xmM5566ik8++yzCAkJ8fVw\nRKWmpgbnz59Heno6Tp8+jYKCAjz22GOIjo5GWlqar4fnc1arFWq1Gjk5OcjMzMTZs2fxhz/8AcnJ\nyXzDJDKcI5z54zzAXG/DvM7c3Z0wd7vHH3N6a5jvW8d5wDVvzw9+3WGj0WhuS7GzLA0AAAcXSURB\nVN56vR5ardZHIxKn0tJSzJ49G8HBwdi+fTukUr9+2QAAduzYgSFDhmD8+PG+HoroKJVKBAcHY8GC\nBVAqlY7Ft95//31fD00UTpw4gUuXLuGBBx6AUqnEhAkTMGHCBBw5csTXQ6NbcI64yV/nAeZ6G+Z1\n5u7uhLm7bf6a01vDfN86zgOueXt+8Ov/nYMGDYJOp3PaptPpEB8f76MRiU9hYSF++ctfYuzYsdix\nYwfUarWvhyQKubm5OH78ONLS0pCWloaysjIsX74cu3bt8vXQfC42NhYWiwUWi8WxzX5OJwFXr16F\n0Wh02iaXy7kOgghxjrDx53mAud6GeZ25uzth7m6dP+f01jDft47zgGtenx88snRxN2UwGISxY8cK\n+/btc1pFvqGhwddDE4Xr168L6enpwquvvurroYjexIkTuZJ8s6amJmHcuHHCtm3bBJPJJHz66adC\nSkqKcOHCBV8PTRSKioqE5ORk4c033xSsVqtw/vx5Yfjw4cKlS5d8PTS6BecIzgO38tdcz7zO3N2d\nMHe7xpzuPn/N965wHnDN2/ODRBD8uyxWVFSE7OxsFBcXIyYmBtnZ2UhJSfH1sERh586d2Lp1620t\npL/97W+xbNkyH41KnDIzM/H8889j4sSJvh6KKHz//fdYu3YtvvjiCwQGBuKJJ57AQw895Othicap\nU6eQk5OD0tJSREVF4cknn8T999/v62HRHfj7HMF5wJk/53rmdebu7sTfc7crzOnu8+d87wrnAde8\nOT/4fcGGiIiIiIiIiEhs/HoNGyIiIiIiIiIiMWLBhoiIiIiIiIhIZFiwISIiIiIiIiISGRZsiIiI\niIiIiIhEhgUbIiIiIiIiIiKRYcGGiIiIiIiIiEhkWLChbu3pp59GQkKCy38vvfQSzp8/j4SEBBgM\nBq+P58qVK45jL126tMOP/+6779rcd8WKFY5jubM/EVF3xVzPXE9E/oH5nvmenEkEQRB8PQiijqqr\nq4NerwcAXLhwAUuWLMGpU6egVCoBAFqtFgqFAjU1NYiIiPD6eK5cuYJJkybh4MGDuPvuu9GrV692\nPd5isaCqqgq9e/eGTCZrdd+6ujrodDrMmjULubm5iIuL68zQiYhEi7meuZ6I/APzPfM9OZP7egBE\nnREUFISgoCAAQHBwMAAgPDwcKpXKab+uSOgthYaGtjuhA4BMJnN7rEFBQejdu3e7j0FE1N0w1zPX\nE5F/YL5nvidnPCWKerxb2yYTEhKQm5uL6dOnY+jQoZg3bx6uXr2KlStXIiUlBZMnT8a5c+ccj//x\nxx+xdOlSDB8+HOPGjUN2djYaGhrcOra9DfKDDz7A/fffj2HDhmHZsmUoKyvD/PnzMWzYMGRlZaGo\nqMhpf3sbZGZmJvbv34+HH34Y99xzD6ZMmYK8vDwPR4iIqPtjrici8g/M9+RPWLAhv7R582asXr0a\nBw4cQGFhIWbMmIEhQ4bgn//8J+Lj47FmzRoAgCAI+P3vfw+FQoHDhw9j+/btKCoqwqpVq9p1vBdf\nfBHbtm3Djh078N577+Ghhx7C1KlTcfjwYajVavzlL39p9bFz5szB8ePHkZCQgFWrVsFkMnXq5yci\n8gfM9URE/oH5nnoqnhJFfuk3v/kNRo4cCQBIT09HaWkp5s2bBwD41a9+hYULF8JiseDjjz+GTqfD\n3/72NygUCgDAhg0b8MADD6C8vBz9+vVz63iLFi1CcnIyACAxMRFRUVH42c9+BgDIysrC7t27XT52\n+vTpmDp1KgBg8eLFyMrKQnl5OaKjozv2wxMR+QnmeiIi/8B8Tz0VCzbklwYMGOC4r9FonBKkWq2G\n1WqF2WzGd999h/r6eowaNeq259DpdG4n9VuP179/f6fjGY1Gl48dOHCg435gYCAAsApPROQG5noi\nIv/AfE89FQs25JfkcueXvlR657MDzWYzBgwYcMcqeXsWO3P3eHdir/63xIu7ERG1jbmeiMg/MN9T\nT8U1bIhaERcXh/LycgQFBSEmJgYxMTEwm83YuHEj6uvrfT08IiLyAOZ6IiL/wHxP3Q0LNkStGDNm\nDOLi4rB8+XIUFhaioKAAK1euxI0bN9CnTx9fD4+IiDyAuZ6IyD8w31N3w4INUSukUil27NiBwMBA\nPPzww5g3bx4GDhyI7du3+3poRETkIcz1RET+gfmeuhuJwBPmiDzmypUrmDRpEnJzcxEXF9fjjkdE\nRMz1RET+gvmefI0dNkRecOPGDdTW1nr1GHV1daiqqvLqMYiIyDXmeiIi/8B8T77Cgg2RF8ydOxfP\nPfecV4+xZs0azJo1y6vHICIi15jriYj8A/M9+QpPiSIiIiIiIiIiEhl22BARERERERERiQwLNkRE\nREREREREIsOCDRERERERERGRyLBgQ0REREREREQkMizYEBERERERERGJzP8DD1rEo6WqsowAAAAA\nSUVORK5CYII=\n",
"text/plain": [
""
]
},
"metadata": {},
"output_type": "display_data"
}
],
"source": [
"# setpoint\n",
"Tsp = 390\n",
"\n",
"# set initial conditions and cooling flow\n",
"IC = [C0,T0,Tcf]\n",
"\n",
"# do simulation at fixed time steps dt\n",
"dt = 0.05\n",
"ti = 0.0\n",
"tf = 8.0\n",
"\n",
"# control saturation\n",
"qc_min = 0 # minimum possible coolant flowrate\n",
"qc_max = 300 # maximum possible coolant flowrate\n",
"def sat(qc): # function to return feasible value of qc\n",
" return max(qc_min,min(qc_max,qc))\n",
"\n",
"# control parameters\n",
"kp = 40\n",
"ki = 80\n",
"kd = 0\n",
"beta = 0\n",
"gamma = 0\n",
"\n",
"# create python list to log results\n",
"log = []\n",
"\n",
"# start simulation\n",
"c,T,Tc = IC\n",
"qc = 150\n",
"\n",
"eP_ = beta*Tsp - T\n",
"eD_ = gamma*Tsp - T\n",
"eD__ = eD_\n",
"\n",
"for t in np.linspace(ti,tf,int((tf-ti)/dt)+1):\n",
" # PID control calculations\n",
" eP = beta*Tsp - T\n",
" eI = Tsp - T\n",
" eD = gamma*Tsp - T\n",
" qc -= kp*(eP - eP_) + ki*dt*eI + kd*(eD - 2*eD_ + eD__)/dt\n",
" qc = sat(qc)\n",
" \n",
" # log data and update state\n",
" log.append([t,c,T,Tc,qc])\n",
" c,T,Tc = odeint(deriv,[c,T,Tc],[t,t+dt])[-1] # start at t, find state at t + dt\n",
" \n",
" # save data for PID calculations\n",
" eD__,eD_,eP_ = eD_,eD,eP\n",
"\n",
"qplot(log)"
]
},
{
"cell_type": "markdown",
"metadata": {},
"source": [
"## Simulation 4. Interactive Controller Tuning\n",
"\n",
"In this example, PID control is used to stabilize an otherwise unstable steady state, thereby allowing the reactor to operate at temperature and conversion that would not be possible without control. The acheivable operating conditions are limited by the controller tuning and the limits on the available control action.\n",
"\n",
"The following simulation provides for the interactive adjustment of reactor setpoint temperature and the proportional, integral, and derivative control gains. Adjust these in order to answer the following questions:\n",
"\n",
"1. What is the minimum achieveable temperature setpoint for the reactor with conversion greater than 80%? What limits the ability to reduce the temperature setpoint even further?\n",
"\n",
"2. Adjust the temperature setpoint to 420K. Adjust the controller gains for satisfactory closed-loop performance. Repeat the exercise for a setpoint of 390K. How is the behavior different? What happens when when the proportional gain is set too small? Explain what you see."
]
},
{
"cell_type": "code",
"execution_count": 7,
"metadata": {},
"outputs": [
{
"data": {
"image/png": "iVBORw0KGgoAAAANSUhEUgAABGwAAAEMCAYAAACRPCChAAAABHNCSVQICAgIfAhkiAAAAAlwSFlz\nAAALEgAACxIB0t1+/AAAIABJREFUeJzs3XdcleX7wPHPGRz2EPcCceEWF0iKfjXTstLUNLXULBVN\nszQ3uVdlTlARUzMzv5bfyurnzFm5RzlwQ5oKIiJ7nvH743gO58gQUYH0er9evoRn3ucGnvOc67nu\n61YYDAYDQgghhBBCCCGEEKLEUBZ3A4QQQgghhBBCCCGENQnYCCGEEEIIIYQQQpQwErARQgghhBBC\nCCGEKGEkYCOEEEIIIYQQQghRwkjARgghhBBCCCGEEKKEkYCNEE/Q45yETSZ0E0KIp4dc04UQomST\n63TupF+KlgRsRIny888/89Zbb9GiRQt8fX3p168f+/btK+5mFcqxY8cYO3bsIx8nISGBDz/8kMjI\nSACuX7+Ot7c3+/fvf+RjCyFEcZkwYQLe3t75/jt8+HBxN/OJWLRoEZs3by7uZgghnnJFdV99+PBh\nvL29uXLlCgD9+vVj1KhRj/08JufOncPb25tff/3VanlUVBTe3t60bNkyR1Bh3rx5NG/eHJ1OV6Bz\n7Ny5kzlz5jy2NlvK731v2bJlwJPvw8JasmQJ33//fXE345miLu4GCAHGSO348ePZsWMHb775JkOG\nDEGn0/HLL78wZMgQpk+fTu/evYu7mQ9l06ZNXLt27ZGPc/78ebZu3cr7778PQLly5di4cSM1atR4\n5GMLIURxee+996yu64GBgXTo0IGePXual9WsWbM4mvZEabVali9fzqxZs4q7KUKIp1Rx31dPnToV\nGxubJ3Z8b29vXFxcOHXqFB06dDAvP3jwIK6urty9e5dz585Rr14987oTJ07QokULVCpVgc7x1Vdf\nUaZMmcfedpNBgwbxwgsv5FhesWLFJ3bOx2Hp0qVMmzatuJvxTJGAjSgRNm7cyM8//8yaNWto2bKl\neXm7du3QaDTMmTOHjh074u7uXoytLBk0Gg0+Pj7F3QwhhHgkHh4eeHh4mL9Xq9VUqFBBrm9CCPGI\nivu++kkH25VKJc2aNePPP/+0Wn7w4EHatWvH0aNHOXDggDlgk5mZydmzZ/noo4+eaLseRpUqVeT9\nThSIDIkSJcLatWvp0KGD1ZuKyYgRI+jduzcpKSkAbNmyhW7dutG4cWOef/55Vq5caZX26O3tzebN\nmxkxYgQ+Pj60atWKkJAQq2PGxcUxduxY/Pz8aNGiBSNHjuTWrVvm9bdu3eLDDz+kWbNmNGvWjLFj\nx3L37l3z+gkTJjBmzBjCwsIICAigUaNGBAYGmo8xYcIEfvjhB/766y+8vb25fv06wcHB9OnTh6lT\np9KkSRMGDx4MwOXLlxk+fDi+vr40aNCATp068d133wHGFNP+/fsD0LlzZ4KDg3MdEnXw4EF69+5N\nkyZNCAgIYN68eWRmZprXt2/fni+//JKJEyfSrFkzfH19mTlzJllZWYX7gQkhRBGJjo5m5MiRNG3a\nlObNmzN+/HgSEhLM68eMGcP48eMJCQnhueeeo0mTJkyZMoXk5GSmTp1K06ZNadu2rfm6atpn1KhR\nLF68GF9fX/z8/Jg5c6bVddNgMLBixQrat29Pw4YN6dGjB0ePHjWvP3DgAN7e3mzcuJGWLVvSpk0b\nUlNTiY+PZ+rUqbRp04b69esTEBDAp59+ilarRavVUr9+fQA+/vhj3n77bQDatGnDwoULrV73yJEj\nzeuvXr2Kt7c369atIyAgAF9fX/755x8AvvvuOzp16kSDBg14+eWX2bFjx2PtfyHEv8/D3FfDg++t\nMzMzCQkJoWPHjjRq1Ihu3bqxZ8+ePM9vOZzHNFzqxIkTvP766zRs2JBOnTrlGM70xx9/0K1bNxo1\nakT37t3ZuXOn+R46N35+fpw5cwa9Xm9edujQIVq0aIGfnx8HDx40Lw8PDycjIwM/Pz/AeH3/4osv\n6Ny5Mw0aNKB58+aMGDHCfB/fr18/jhw5wpYtW/D29jYf5+TJk/Tp04dGjRoREBBAcHCw1fnbt2/P\nwoULzX35OIe+xsbGMmHCBFq3bo2Pjw+DBw82D0Ez9VVsbKx5++HDh9OkSRO0Wq15Wbdu3Vi4cKH5\nZ/Lf//4Xf39/WrduTXJyMomJiUyfPp02bdrQoEEDWrduzZw5c8yfF0x9MW3aNPr162c+7v/+9z9e\nfPFFGjRoQOfOndm6detje91CAjaiBLh16xYREREEBATkur5y5cpMmjSJqlWr8vXXXzN69Gh8fX1Z\nunQp3bp1Y/HixcybN89qn1mzZuHh4cHy5cvNgQ7TmF2tVsvbb7/NiRMnCAoK4rPPPiMyMpJhw4YB\nkJqaSv/+/bl48SKzZ89m5syZnDx5ksDAQKtxr3v27GH37t1Mnz6d2bNnc/LkSebOnQsYU/3btm1L\nrVq12LhxI+XKlQPg1KlT3Lx5k2XLlvHOO++QnJxM//79ycjI4PPPP2fZsmVUr16dyZMnExkZSf36\n9ZkyZQoACxcutBoqYLJ7924GDhyIp6cnS5YsYdCgQXzzzTc56ucEBwejUCgIDg7m3Xff5euvv7b6\nACOEECVNcnIyb731FhEREcydO5fp06dz7NgxAgMDrW6Sd+7cydGjR/nss88YPHgwGzdupHv37uh0\nOpYuXUqDBg2YPn06UVFR5n3279/P9u3bmTNnDiNHjuR///sf06dPN6+fN28eS5cupXfv3ixduhRP\nT0/eeecdzp8/b9XGr776ik8//ZRJkybh4ODAqFGjOHz4MBMnTuSLL76ga9eurF69ms2bN6NWq/nm\nm28A4xCwyZMnP1R/hIWFMWXKFIKCgqhatSrr169nypQpdOzYkeXLl9OyZUs++OCDf23tNyHEo3uY\n+2qgQPfWY8aMYfXq1bz11luEhIRQs2ZNhg0blm/Q5n5jxozhtddeY8WKFVSqVInRo0cTHx8PGIf/\nBwYG4unpSUhICAEBAYwZMybf47Vo0YKUlBQuX74MwJUrV4iJicHf3x8/Pz+OHz9uDsKfPHkSd3d3\nc8Bh5cqVhISE8Oabb7J69WpGjx7NoUOH+OyzzwDjkK569erx3HPPsXHjRnMbBwwYgJubG8HBwQwe\nPJhVq1bl+AyyatUqunbtyueff55rwMxEr9ebA/mmf5bva5aSk5Pp3bs3J0+eZOLEiXz++efcvXuX\nvn37EhUVhb+/P2q1miNHjgDGgNSxY8dITU3l3LlzgPFh9blz56x+L7788ks++eQTgoKCcHJyYvTo\n0Rw4cICJEyeycuVKunfvztq1a/nhhx8AzH0xaNAgpk6dal728ccf0759e5YvX06rVq0YNWoUu3fv\nzvfnJwpOhkSJYmeKZj9ozKZOpyM4OJjXX3+diRMnAtC6dWsUCgXLly9n0KBB5tTO1q1bM27cOABa\ntmzJtm3b2L9/P23btmXv3r1cuHCBzZs3U6dOHQDKly/P+++/zz///MP+/fu5ceMG27dvp3LlygDU\nq1ePl156iT179pjHymZmZhIWFoaLiwsAly5dYt26dYAx1d/d3Z34+HirdEetVktQUBDVqlUD4PTp\n03h5ebFw4UKcnZ0BaNy4Mb6+vpw4cYIePXqY00q9vb2pUKFCjicNS5Yswd/fn08//RSAgIAAXF1d\nGT9+POfOnaNu3boA1KhRw1w87bnnnmPPnj3s37+fvn37FvAnJYQQRev7778nJiaGHTt2UKFCBQDq\n1KnDyy+/zL59+2jXrh1gvLYGBwfj4uJC69at+fbbb1GpVMycOROFQoGHhwe//vor586dM7/XpKen\ns2LFCvOHFq1WyyeffGJOmf/qq68YNWoU7777LmC8tvbp04dly5axZMkScxvfeecd2rZtCxgD/gaD\ngVmzZtG8eXMA/P392bNnD8ePH6dHjx40btwYgKpVqz50LbKePXuaax5otVpCQkLo06ePuc0BAQHc\nvXuXxYsXm9skhHi2FPS+Ggp2bx0TE8P27duZN28eXbp0AYxZgTExMSxatMh8HX6QwYMH06dPH3Pb\nXnzxRQ4fPkynTp1YuXIlNWrUYOHChSgUCtq0aUNycjJff/11nserW7cuzs7O/PXXX9SuXZuDBw/i\n4eFB5cqVUavVpKWlcfLkSfz8/Dh58iS+vr4oFAoAYmJiGDlyJG+++SYAvr6+REREsGvXLsA4pMvJ\nyQk3Nzfzffzy5cupWrUqISEh5jo49vb2TJ8+nUGDBlG6dGkAGjRoYM6OzM+MGTOYMWOG1bLOnTvn\nyLYEYwZLdHQ027Zto0qVKoAxw+j5559n9erVBAUF0bhxY44ePUrnzp25dOkSaWlpeHl5ceLECRo2\nbMjBgwdxcnLCx8eH48ePA9bvX+np6eh0OqZPn24ONPn7+7N3716OHz9Or169zH1RpUoVatasiV6v\nZ8mSJfTs2dP8uSsgIID4+HgWL15M+/btH9gP4sEkw0YUO9NFL6+osklERATx8fG8+OKLVss7d+5M\nVlYWf/31l3mZ6YYYQKFQUL58eVJTUwFjlL1ChQrmYA0YAzK7du2iatWqHD16lFq1alG+fHlzxLtK\nlSp4eHhw6NAh8z5eXl7mYA0Ygz5paWn5vga1Wm3+cADQsGFD1q9fj62tLRcuXGD79u2EhYUBFGi4\nUkpKCufPn8+1TxQKhfmCDNCoUSOrbSz7RAghSqKjR49Su3ZtypQpY74ee3p6UqVKFasZpKpVq2Z1\nPXZ3d6devXrmm3M3NzcAkpKSzNs0atTI6nr8/PPPo9fr+fPPP/nzzz/JysoiICDAfF6dTkfr1q1z\nzFzl5eVl/trBwYEvv/ySZs2ace3aNfbt28eKFSuIi4t7LENQLc915coV4uLirNqo1WoJCAggPDzc\n6rUKIZ4dBb2vhoLdWx8/fhyFQpHrNufPnyc5OblA7bK8NzcF4E33zUeOHKFdu3bmazaQ43z3U6lU\nNGvWzHz/f/DgQXOgoXz58nh5eZmv16bAjcnHH3/MO++8Q2xsLIcPH2b9+vWcOHEi3+v00aNHadWq\nFQaDwep6m5WVxYkTJ8zbWV6n8xMYGMimTZus/o0ePTrXbY8fP06DBg3MwRoAZ2dnAgICzPf6rVq1\nMg/bPXr0KI0aNaJp06bmth04cIDnnnsOtTo7X8OyrXZ2dqxZswY/Pz/z+1dYWBh37tzJs18iIyOJ\njY2lTZs2Od6Hzp8/b86gEo9GMmxEsTM9AbBMVb9fdHS0uWbB/RXbTRFtyzcMW1tbq22USqV5LG5C\nQkK+Rdbi4+MJDw831xmwZBnksbOzy/MceXFzc8tRnT4kJIRVq1aRlpZG1apV8fX1BXjgscD44cNg\nMJj7wESj0eDk5GTVJ4VprxBCFKf4+HhOnz6d6/X49u3b5q8dHR1zrL//mne/smXLWn1vel9ITEw0\nf9B59dVXc93XstbN/dff7du3M3fuXKKioihbtiw+Pj7Y2dk9luut5blMN8JDhw7NddvY2Fhz5qYQ\n4tlR0PvqChUqFOjeOiEhAWdnZzQaTa7bWNbCyY/lvblSacwZMF1r7969m+Pe/P5ra25atGjB5s2b\n0ev1HDlyxGpYa8uWLTl+/Dg3btwgJibGanjS5cuXCQoK4s8//8TR0ZH69etja2ub73U6Pj6etWvX\nsnbt2hzrYmJiHqrdYPw5NWzYsEDbJiYm5npcd3d3zpw5A2CuqRMXF8fRo0dp1qwZHh4eLFq0CDAG\nbN57770c+1v69ddfmT17Njdv3qRMmTL4+Pjk2y+m+p7Dhw/PdX1sbKz5gYkoPAnYiGLn7u5OnTp1\nOHDgQK5TDN64cYP27duba7lYFtSy/N7V1bVA53N2drYqIGyyb98+GjZsiIuLC02aNCEoKCjHNgU9\nR0H9+OOPLFu2jNmzZ9OxY0ccHR1JT09n06ZNBdrfyckJhULBnTt3rJZnZGSQlJT02NsrhBBFydnZ\nmWbNmplT9S096k2gZeFiwHwdLVWqlPlp4pdffomTk1OOfS2fUFq6cuUKH330Ef369ePdd981fwjq\n3r17vm1RKBQ5noY/6EOQKaPo008/zXVoVaVKlfLdXwjxdHqY+2rTQ8L87q0TExNJSkoiMzPTKmjz\nsPff+SlXrlyOe9m4uLgH7ufr68uCBQs4ceIESUlJVkGZli1bsnnzZk6cOEG5cuWoXr06YAwSDRs2\njAoVKrB161a8vLxQKBTMmzePa9eu5XkuZ2dnXnnlFV577bUc65709dbFxcUqKGRy584dc/83aNAA\nV1dXjhw5wvHjx+nevTuenp7ExMTw+++/c/PmzTzrGgH8/ffffPjhh/Tt25fBgwebH2rkVj/Tsl0A\nc+fOpVatWjnWW2YEicKTIVGiRHjrrbfMRSPvFxwcjI2NDR06dMDNzY1t27ZZrd+6dSsqlSrHkJ+8\nNG7cmKioKC5cuGBedvHiRYYMGUJERARNmjTh6tWreHl50bBhQxo2bEitWrUIDg7m9OnTBX5NpqcH\n+Tl58iTVqlWjW7du5ifEf/zxB5D91OH+jBxLTk5OeHt759onAE2aNClwe4UQoqRp0qQJ165do3r1\n6ubrcY0aNViyZIn5qWJhnTp1yip4v2vXLmxsbGjSpAmNGjVCpVKRkJBgPm/Dhg3Zv38/3333XZ7X\n97Nnz5KVlUVgYKA5WBMTE8OlS5fMTyhz29fBwYHo6Gjz9xkZGZw9ezbf9teoUQNnZ2fu3Llj1cZz\n586xcuVKq6EFQohnS0Hvq6tXr/7Ae+umTZtiMBhy3aZu3boPzGYsiGbNmuUoYLx3794H7le/fn3s\n7Oz45ptvqFWrllXGiJ+fH2lpaWzZssVqOFRcXBzXrl2jT58+VK9e3RwwP3jwoFXg/P5rdZMmTfj7\n77+trrdqtZpFixblCDY9bk2bNuXMmTNWdSyTk5P5/fffzXVllEol/v7+/PDDD8TGxtK0aVM8PT0p\nW7YswcHB1KpVyzwULTfh4eHm9y9TsCY2NpaLFy/m2S+m35/Y2Firfrlw4QJhYWEF+iwkHkwybESJ\n0KNHD3bt2sXgwYPp378/fn5+pKSk8MMPP7B7925mzZpF+fLlee+995g7dy6Ojo60adOGP//8k+XL\nl9OvX78CP21t3749tWvX5v333+fDDz/E1taWRYsW0bRpU5o2bUrt2rVZu3YtgwYN4t1330Wj0bB6\n9WpOnTrFhAkTCvyaXFxcuHbtGgcPHswzcNKgQQM2btxIWFgYPj4+hIeHExISgkKhMI/rNaW079q1\nK8dQL4D333+f4cOHM378eF555RUiIyNZtGgRL7zwgtUQLiGE+Lfp2bMnX331lfl6bGNjwxdffEF4\neDgff/zxIx07LS2NYcOGMXToUK5du8aiRYvo168fLi4uuLi40LNnT6ZOnUpMTAy1a9fm0KFDhIaG\nmgv85qZevXoolUo++eQTXnvtNaKjowkNDSUrK8tcM0ypVOLk5MShQ4do2LAhderUoXXr1mzcuBFf\nX18qVqzImjVrHjiESqPRMGTIEJYsWUJmZiZNmjTh3LlzLFiwgB49euQYviCEeHYU9L4aeOC9tZub\nGx06dGDatGnEx8fj5eXFL7/8wuHDhwkJCXks7R0yZAjdunVj9OjRdOvWjbNnz/LVV18B+T8AValU\nNG3alB07duSYRKNUqVJ4e3vz22+/MW3aNPPy0qVLU7FiRVatWoWjoyN6vZ4NGzYQHh5uFXxycXHh\nwoULHD58GF9fX4YOHcqbb77JxIkT6dy5MwkJCSxcuBB7e/sC160prB49evDVV1/x7rvv8sEHH6DR\naAgLC0Ov1zNgwADzdq1btyYoKIh69eqZs0ObN2/O1q1beeedd/I9R506dVCpVHzyySf06NGDmJgY\nQkNDycjIsKrR6eLiwpEjR2jSpAl16tQhMDCQRYsWkZWVRbNmzTh//jwLFy6kS5cu8j70mEjARpQI\nSqWSkJAQvv76a3788UfWr1+PWq3G29ub1atX06pVKwAGDBiAra0ta9asYf369VSsWJFRo0Y98CJk\nyRSAmTt3LpMnT0atVtOmTRsmTpyIUqnExcWFdevW8dlnn5kDNA0aNGDt2rXmdMqC6NWrF7/++itD\nhgzJdbwrGC/AERERrF27luXLl+Ph4cGkSZP46aefzEXUatWqxcsvv8ySJUuIjo7O8Vo7dOhASEgI\nISEh/N///R+lS5fmrbfeYsSIEQVuqxBClERubm588803fPLJJ4wbNw6FQkHDhg358ssv8fT0fKRj\n+/j44Ofnx0cffYS9vT2DBg2yGt8/efJkypQpw5o1a7h9+7Z5Ktz+/fvnecyaNWsye/Zsli1bxpYt\nWyhfvjydO3fGYDDw/fffo9frUSqVDB8+nODgYK5cucKPP/7I+++/T1xcHHPmzMHOzo7evXvj7e1N\neHh4vq9hyJAhODg4sG7dOpYuXUq5cuUYPHhwjjoFQohnS0Hvq6Fg99bz589n0aJFhIWFkZiYSO3a\ntVm+fHmBZ4h6kFq1arF06VLmzZvHsGHD8Pb2ZvTo0cydOxd7e/t89/X19eW3337LdQptf39/zp8/\nb7VOoVCwZMkSZs6cyciRI3FxcaFFixYsWLCAUaNGceHCBby9vRkwYAAffvghQ4YMYfv27fj4+LBq\n1SoWLVrE8OHDcXR0pHXr1owdOxYbG5vH0g95cXZ2Zt26dXz66ad8/PHHKBQKmjdvzsaNG80z2oIx\nYAPGjCWTFi1asHXr1nyHQ4ExW2bOnDksXbqU7du3U758eV588UU6duzIxo0b0el0qFQqhg0bxuLF\ni7l8+TI///wz77zzDnZ2dubPMuXKlWPgwIF51rURD09hkKqjQgghhHiGjBkzhhs3brBhw4bibooQ\nQjzz/vjjD1xcXKyK8G7cuJH58+dz+PBhGeIpnmmSYSOEEEIIIYQQolicPHmSdevWMW7cODw8PIiM\njGTBggX06tVLgjXimScBGyGEEEIIIYQQxSIwMJCUlBRCQkK4ffs25cuX5+2332bIkCHF3TQhip0M\niRJCCCGEEEIIIYQoYWSuLSGEEEIIIYQQQogSRgI2QgghhBBCCCGEECXMU1fD5vbtpELtp1AoKF3a\nkTt3UpBRYtakb/ImfZM/6Z+8PUrflC3r/IRa9e9S2Ov9w5LfY+kDkD4A6QMo+j6Q671RYa738vua\nN+mb/En/5E36Jm+P2jd5Xe8lw+YepdLYyUrpkRykb/ImfZM/6Z+8PY19Exsbi7+/P3v27AHg9OnT\n1K1blyZNmpj/hYaGAmAwGJg/fz4tW7akRYsWzJo1C51OV5zNz9PT+LN6WNIH0gcgfQDSB/8m8rPK\nm/RN/qR/8iZ9k7cn1TdPXYaNEEKI4hEUFER8fLz5+3PnztGmTRtWrFiRY9v169ezd+9efvrpJxQK\nBYGBgaxevZrBgwcXZZOFEEIIIYQosSQ2JoQQ4pFt2LABe3t7KlasaF4WHh5OnTp1ct1+8+bNDBgw\ngHLlylG2bFkCAwP54Ycfiqq5QgghhBBClHiSYSOEEOKRREZGsmbNGr799lu6d+9uXn7u3Dk0Gg3t\n27dHr9fz0ksvMWrUKDQaDREREdSsWdO8rZeXF5GRkRgMBhQKRYHOW1QpuUqlwur/Z5H0gfQBSB+A\n9IEQQoiiJQEbIYQQhabVahk3bhxBQUG4ublZrStVqhR+fn688cYb3Llzhw8++IAlS5YwZswY0tLS\nsLOzM29rb2+PXq8nMzMTW1vbAp27dGnHAgd3Hgc3N8ciO1dJJX0gfQDSB/Ds9sGWLVsIDg4mOjqa\nSpUqMWrUKDp06EBCQgKTJk3i0KFDODs7M3z4cHr27AkYa5YtWLCA7777Dp1OR9euXZk4cSIqlaqY\nX40QQpR8ErARQghRaMuWLaNu3bq0bds2xzpTgWEABwcHAgMDWbBgAWPGjMHOzo6MjAzz+rS0NNRq\ndYGDNQB37qQUWYaNm5sj8fEp6PXP5owI0gfSByB9AEXfB+7uTk/8HAUVGRnJpEmTWL16NU2bNuXA\ngQMMGTKE/fv3M23aNBwcHDhw4AAXLlxg8ODB1KpVCx8fH6lZJoQQj6BIAzbh4eFMmTKFy5cv4+np\nyfTp0/Hx8cmxXWBgIAcPHrSKvJ88ebLI2qk3GPg7KolTV2LRG8CrgjMNqrtjo5YnAUIIYWnLli3c\nvn2bLVu2AJCcnMzo0aMZOnQo8fHxDB8+HCcn4weOjIwMc0CmRo0aREZG0rhxY8D4QaB69eoPdW6D\nwUBRTiyl1xvQ6R7+A9r128kcDr9FJ18PnOxtnkDLik5h++BpIn0gfQDPZh94eXnxxx9/4OjoiFar\nJTY2FkdHRzQaDb/++ivbt2/H1taWRo0a8corr/Djjz/i4+NjVbMMjPf5ixcvLnEBm6vRSew/dTPH\nz1WlUtCmUSU8K2RPuXvwTDRanZ6AxpVyHOd0xB2u306mk68HyiLMAhVCPJ2KLGCTkZHB0KFDGTp0\nKD179mTz5s0MGzaMX3/9FUdH67TS8PBw1q9fT8OGDYuqeYDxzffIuVt8vy+CqDupVuuqlHVi/JtN\ncLT7d99sCyHE47Rt2zar79u3b8/kyZNp27YtHTt2xGAw8NFHH3Hz5k1CQ0Pp1asXAF26dGHVqlW0\nbNkStVrNihUr6Nq1a3G8hCduzZbzREYlciYijqkDWxR3c4QQotAcHR35559/6NSpE3q9nmnTpnHt\n2jXUajVVq1Y1b+fl5cWOHTsAiq1m2cPWG/rm14tcup6Q67obt1MI6t8MgLjEdFb+Eg6AZwVnvCq5\nmLczGAys+OksqelaalRyoW4194drdBGRWkz5k/7Jm/RN3p5U3xRZwObQoUMolUr69u0LwOuvv87a\ntWvZt28fnTt3Nm93584d4uLiqF27dlE1jdR0LT/+HsGJi7HEJaabl5d1s8Neo+afmGSu305m8aZT\nfPSGD7Y2kmkjhBD5USqVhIaGMmvWLFq2bImdnR1vvPEGAwYMAKBv377Exsby+uuvk5WVxauvvsrA\ngQOLudVPRmRUIgBXbyWRmaVDI+8hQoh/sYoVK/LXX39x7Ngx3nvvPd59912rmmQAdnZ2pKcb76mL\nu2ZZQeuxGgPtAAAgAElEQVQNpWZoAfCq5ELFMsZ9bt5O4e+oRDK1evPwtORMvXmf20mZNLMYtpae\nqSU13XgcnUJZooa05eZZrcVUUNI/eZO+ydvj7psiC9hERkZSo0YNq2VeXl5ERERYLQsPD8fR0ZHA\nwEDOnz9PtWrVGD9+PE2aNCnQeQoTgd/75w1+PXbd/H3D6qXp1saL6pVcUCgU/PbXTb745RyXryfw\n428R9H2h6IJJJYFEUvMmfZM/6Z+8Pa19s3v3bvPXNWvW5Msvv8x1O5VKxahRoxg1alQRtaz4lHK2\n5W6SsV7PyUux+NUrX8wtEkKIwlOrjR8f/P396dixI2fOnLGqSQaQnp6Og4MDQLHVLHvYekPpGcYx\ntm0bV6Jd08oAbDt8zRiwydIRF5cMQGpK9muJuZNsXg4Qn5S9LjYuxWpdSSL1qPIn/ZM36Zu8PWrf\n5BXgLbKATWpqKvb29lbLLKPvJhkZGfj4+DB27Fg8PT3ZtGkTgwcPZuvWrZQtW/aB5ylMBP7FVtW5\nk5RBpbJO+DeoiGdFF6v1XdvVJj5Vy6bdl/j9VBSDXmuEne2zV69ZIql5k77Jn/RP3qRvnn5O9jbm\ngM0fp6MkYCNEARgMBqKjo6hYMWeNEFE89u3bx5o1a6wC8VlZWXh4eLB//35u3rxJpUrGn1dkZKR5\nGFRx1ywraL2hLK3xBCqlInt703+G7GNotdkZNkmpWVbHTkrNMn+dlqGzWqfXG0rcQ5pnsRbTw5D+\nyZv0Td4ed98UWdTB3t4+R3DGMvpu0qFDBzp06GD+vm/fvmzYsIHDhw/zyiuvPPA8hYnA2wDvdK5j\njojlFg0PaFieH/ZeJiVdy7Y/Imjj8+zcQEgkNW/SN/mT/snbo/RNSU+xFta0uuyb+7N/x3E3KYNS\nzgV/sixEcWrdujm2trYoLW6uSpcuw1tvDeCVV157YuddunQxACNGfPjEziEeTr169Thz5gw//vgj\nXbp04bfffmPfvn18++23REVFMX/+fGbNmsWlS5f45ZdfCAsLA/49Ncuy7l2rbdTZv+umZ8CW79N6\nQ/bXKWnZARqAtHvDqgAyMrO/PnAmii+3nqdPh9q0a1L5sbZbCPF0K7KATfXq1fn666+tlkVGRuYI\nwmzbtg29Xm9V18ZyZpEHedRZQ/KKiDnba/CpVYbjF26z+8QNWjWsWPiT/EtJJDVv0jf5k/7Jm/TN\n009ncaNvMMDR8zF0bFE1nz2EKFlWrlxL9eo1UakUuLra8+23/2PGjKk0aNCYatW8nsg5ExLicXV1\neyLHFoVTtmxZQkNDmTNnDjNmzKBatWosXbqUGjVqMHPmTKZOnUrbtm1xcHBg7Nix5oyaf0vNsqx7\nmTMai1lhTRkxFjEaLJ+xpKRnB2Uguw4OQHpW9geSr3dcRKszsG77Bf7jU6nQ9XiEEM+eIgvY+Pv7\nk5mZybp16+jduzebN28mNjaW1q1bW22XmprK/PnzqV27Np6enqxdu5b09HRatWpVVE3N0398KnP8\nwm0ioxK5disJj/LOD95JCCHEM01nkWEDcDj8lgRshBWtTm816cGT5O5ih1r1kKnIFlQqFZ06dWbR\novlERl6hWjUvEhMTWLz4cw4fPoSdnR1du/bgrbcGoFAoSEiIZ9Gizzl9+i/u3o2jSpWqfPTRBBo1\n8gHghx828c0360hIiKdu3XqMGxfEb7/tZceOrSgUCqKjo5g161OOHj1EaOhS/vnnGpUqVSYw8D38\n/Y33kK1bN6dbt57s3LmNN9/sT79+JS8Y8LRo3rw533//fY7lbm5uLF68ONd9/g01y/QGA9p7D08s\nM2xM03JbZtUY9AXNsMkO2KgshkL9HZ2E133lF4QQIi9FFrDRaDSsXLmSadOmsWDBAjw9PVm+fDkO\nDg5MmTIFgBkzZtC9e3du377NoEGDiI+Pp169eqxcuTLH0KniULdaKUq72HEnMZ2/LsdKwEYIIcQD\nmT4E+NYtx5FzMURGJRJzN5VypYr/fU0UP61Oz6SwQ8QmFE3ApoyrHXOGtCx00CYzM5Nvv91ARkYG\n9es3BGDmzCm4uLjy3Xc/ER9/l3HjPsTd3Z2XX+7CsmVLAFi//juUShWLF88nNDSEZcu+4NChA4SF\nLWP+/CV4e9dlxYqlzJw5hdDQ1Vy5chlXVzdGjPiQiIgrjB//EVOnzqRVqzYcPXqIyZMnsGLFl9So\nUfNeuzL4+ecdZGVlPp6OEs8Uy7o0uQ6JMuQ+JCo53Tpgk5qee8CmtKsdKenGkguHw29JwEYIUWBF\nWjm3Tp06/Pe//82xfMaMGVbfBwYGEhgYWFTNKjClQoG3hxsHzkQTcTOxuJsjhBDiX8A0JKqBV2nC\n/75LcloWv52KokfbGg/YU4iSYejQd1EqFWRmZgEGWrb0Z9Gi5ZQrV547d2I5dOgAv/yyE3t7e+zt\n7enbtz+bN3/Pyy93YciQ97Czs0OlUhMVdRNnZ2du374NwK+/buell16mXr0GAAwcOJirVyNznH/X\nrh00b96Ctm3bA+Dv35pWrdqwY8dWhg17H4AOHTpiY2ODjY1N0XSKeKpk5hGwMWXYWA6Jsvw6vwwb\nyyFRKWnZy4+ej6FX+5rmYwshRH6evamOHlGNSi4cOBPNlZuJGAwGGYMqhBAiX6aiwxobJS3qlGPP\nyRv838GrXLuVzJAu9XC0kw+YzzK1SsmcIS1L9JCo0NBVVK9ek1u3bhIUNA5XVzfq1zcGWW7disZg\nMPDGG9kFiPV6Ay4uxgyC2NhYFi/+nL//jsTT0xNnZ1cMBuPfRFxcHDVq1DLvZ29vT5069XKc/+7d\nOCpUsK4dWKFCRW7fjsl+Xe5lHuo1CWEpyzJgo7LMsLk3JCqvosP51LCxzLBJscjEuZuUwaV/4vH2\nKAUY3yOOnY/B26OUFKQXQuQgAZuHVL2SKwDJaVncjk+TlHYhhBD5MmXYqFVKugZ48U9MMpdvJHA6\n4g7//fUS776S8wOqeLaoVcp/xf1EpUqVWbZsGV27dqVChUoMGPAupUuXQaVS8dNPO9BoNAAkJiaS\nmpoKwNSpk+jatTtLl65EoVCwdesvRERcBoxFbGNjs4MuKSnJrF69kqFDR1idt3z5Cpw9e8ZqWVTU\nTcqWLWf+Xp6fiUeRpcsjw+belwbLIVEWwZssrd7qAW5uRYe1Oj3pmdYzopyJjDMHbL7cep4DZ6Lx\nrODMlAHN5WGwEMJK4avOPaOqlHNEc+9CfkWGRQkhhHgAU4aNSqnAxUHDxLea0i3AOLPOH2eiOXf1\nbnE2T4iHUrlyZT74YDRr1qzk8uVLlC9fgcaNm7B8eTAZGekkJiYwefJ4Vq5cCkBqagr29nYoFAr+\n/juSb775Cq3W+KG2Y8eX2Lbt/7h48TxarZavvlpDePgZ89CmlJQUAJ5/viMnThxl//696HQ6Dh78\ng99/30+HDh2LrR/E0yUrzxo2pqLD2dsa7pvYMS0jOxiTlksNG8ssnOqVjJlnl68nAPDH6SgOnIkG\n4Gp0EuevxT/KyxBCPIUkYPOQVEol1e4VCou4IQEbIYQQedPrDeabe9MwFIVCwcv+1fCsYCxcv277\nBasUeyFKupdf7kKTJs2YO3cGOp2OadNmExd3h9df70Lv3t0pU6YMo0ePB2DcuEl88806OnZsS1DQ\nWF566RXi4++SkBBPs2YtGDZsJFOmTOLll5/nypVLTJs2G4B27Tqwd+8uRo8eQZUqVZk793O+/HIl\nL77YjmXLFjN16izq1q1fnN0gniKWRYfVudawyX1IFEBSWnah69yGRFnWuWlc0zh0LzIqkZj4NNbt\nuGB1rB1HrhX6NQghnk4yJKoQalRy4eI/8Vy5mVDcTRFCCFGC6fTZHwIsp3VVKhX06+jNrK+OER2X\nalXPQIiS5Pffj+W6fOHCpeav3d1LM336nFy3a926La1bt7Va1rdvf/PXr7zSlVde6ZpjvxYt/Ni6\ndbfF9y1p0aLlQ7VRiILKu4aN8X99HkOiAJJSsyh/7/Kd25CoZIuAjU/NMvywP4JMrZ5vdl4kM0uP\nk70Nrz5XjQ27LvHXlTtE3UmhYmnHx/bahBD/bpJhUwimdMZ/YpLJzNI9YGshhBDPKtOU3kCOQq/V\nK7lQpazxpvxw+K0ibZcQQohsWdrs+3mNTc4MG+shUfcHbLIzbNJyy7C5V3BYpVRQpawjrk7GWk+n\nrtwBoGX98rRrWtlccPjH33LOlCaEeHZJwKYQTIWHdXoDN2JTirk1QgghSiqdxV2+SpWzkKRfvfKA\ncZpXrUXRSyGEEEXHVHRYqVCgUuasYWOwmiXKet+k1OwMmlTLGjZZOvQGg3lKb0d7GxQKBbUqu1rt\n/1yDCqhVSl5tVQ0wvh+cjrjz6C9KCPFUkIBNIbg5acyFh+8kFM00nEIIIf59LIMwlkOiTPzqGgM2\nKelazkbGFVm7hBBCZDMNibIsOAzZs0Tp86lhYznkyTLDBiAzS2de72hnrERRs4qbeX3F0g54ljfW\nM2vTuBI1Khuz+Ndtv8C5q3ethtUKIZ5NErApBIVCQWlXOwBiJWAjhBAiDzqdZYZNzrfcMm721Kxi\nfNr626moImuXEEKIbJl5BGzMGTYFGBKl1xtyTN+dkakzD4lytLcBoFaV7Awb//oVzOdQKhQM6FQH\nlVJBbEI68zacZOKKQ0TIrLRCPNMkYFNI2QGbtGJuiRBCiJJKa/F0VJ3LkCiANo0qAXDy4m1uxaUW\nSbuEEEJkyzPDxlTDxnJI1H1JL6YhUWmZ1tk1YCw8bJrW28nOGLCpWs6JKmWdcHXU0KphRavtq5Rz\nYkiX+ub6ZrEJ6cz9+jj7/7pZ2JcmhPiXk1miCqmMqz0gGTZCCCHyZplho1bm/oykZf3yfL//CvHJ\nmWw/+g/9O3kXVfOEEEJgEbBR5T4kyoAxs0ahUOSc1vtewMayfo1JRqbOPK23o73xY5dapWTqwObo\n9QZs1Koc+7SoU44Wdcrxd3QioT+eJSY+jbXbzlPOzZ46njKboBDPGsmwKaQy9zJs7iRKwEYIIUTu\nrGrY5JFho1YpeaFFVQB+PxVFQkpmrtsJIYR4MvLKsDENV4LsYVF5DYm6v34NQHqmZQ0bG/NylVKZ\na7DGUrUKLkx5uwWVyzpiMMCKn87K+4MQzyAJ2BRSaZfsGjb3X7iFEEIIsJ4l6v5pvS21bVwZe1sV\nWp2e7UeuFUXThBBC3GOaJUqdx5AoyC42nNcsUblm2GRl17BxsrfJsf5BHOzUDOvaAFsbFQkpmQT/\n71SugSEhxNNLAjaFZMqwMRYTkwunEEKInKyKDucyS5SJg52aF5obs2x2n7hOojxFFeKpcvPmjeJu\ngsiHKcNGkyNgk/216QGt/r6IjSkgYwqkqFVKbDXG7BnjkKjsab0Lo1IZR95+qQ4AETcTWbxJgjZC\nPEukhk0hmQI2YCw8XJiouRBCiKebrgBFh01eaFGVncf+IS1Dx7bD1+jVvuaTbp4QBXLo0AE2bFjH\npUsXUSigTp26DB78HnXq1Huk465atYLIyCvMmvUZX321mqtXI5k8eeZjarW11NRUOnZsw3ff/UTF\nipUKdYwTJ44xefJ4jhw58lD7Xbx4nrFjP2Dz5u2FOq948rQFGBKlz2NIlPZeYD71XhDFwVaFQqEg\nI1NnHBKVbj2td2H41StPanoW63Zc5OI/8UxaeYg+z9eiRZ1yVm38N9HrDej0BnR6PXq9Aa3eYFym\nM6AzGNDp9BbbGNfpDQYMBovgmbG4EHqAe8vvLbL42gAG08/v3v73lltuh8WPVaEEJyc7kpPTMejB\nQN6jKQo70CK//fI7XyFXPTYKBTg62pGSkl7o1/60UiigWpVSeJaxf6zHlYBNIbk4arBRK8nS6rmT\nkE61Ci7F3SQhhChWsbGxvPrqq8yZM4d27doRHR3NjBkzOH78OGq1mhdffJHx48ej0WgwGAw0a9bM\n6sa3WbNmfPHFF8X4Ch4/rVWGTf5JrY52NnRoVpWfD/zNnj9v8Gqratjbytu0KF4//fQDX3wRyvjx\nH/Pcc/64ujqwcuVqRo4cRmjoaqpXr/FYztO//zuP5TglUXJyMlqtZESUZHkWHbYM2OhNQQLrT6mm\nwLwpYGNvZ2N8b0sxZt9k3Jvqu7AZNibtmlYBYMOuyyQkZxK6+Sy/n4rizY61KV/K4ZGO/TD0BgOJ\nKZncTcogJT2L1HQtKWlZpKRrSUk3/p+WriVDqyMzS0/Wvf8zsnRkafVk3vted//YMiGeEhPfakqt\nKm6P7XhyJ1hICoUCdxc7bsWlykxRQggBBAUFER8fb/5+7Nix1KpVi/3795OYmMjw4cNZunQpo0aN\n4urVqwCcOHHiX/t0sCBMN/IKBSjzGRJl8nyzKmw5dJWMTB2Hw2/xnyaVn3QTRQmg1Wu5m55QJOcq\nZeeKWlmw27/09HRCQhYxdeosWrUKQKVSYGtrS9++/YiLu8vVq5FUr16DuLg7LFmygKNHD2Fra0eH\nDh0ZNGgYGo2G1NRUli8PZt++3QA891xrRowYhZOTk9W5LLNtZs+ehqOjIxcvXuDSpQt4eFRj3Lgg\nvL3rYDAYWLNmJd9//x0ajYaePfsQGhrMf//7w0Nlzhw7doRVq0L5559rZGZm4evbko8/no6dnR1J\nSUksWvQZf/zxG2q1DZ07v8KwYSOt9k9JSWbEiGHUqlWbceOCyMzMYPnyYPbu3Y3BYOCFF14kMHA4\nyclJjBnzAZmZGbzwQgCbNv2Mq+vju5EXj0em1hhUyZFhY/GtKU5z/7TeBoMxmJOWnp1hYxoOG5eY\nYd7Oye7Rs/HbNa1CPS93vt5xkbORcZyJjGPyF4fp0LwqL/t7WhU2LiyDwUBSWhax8enEJqQRm5Bu\n/Bef/bVlQf2SSIHxs5pCYXz/BYVxeJsCFGQvN30N1tlUxvUKcxaOaVl+58t7Zd5r89+vcOd7lHuq\ngu6qVCrR3/+HIACoUNqRSmUcH+sxJWDzCMq4SsBGCCEANmzYgL29PRUrVgQgMzMTe3t7hg0bhq2t\nLWXLluXVV19l586dAISHh+Pt7f2INxYKHpC08liYAi0FCbjcz/QAUa1S5jlLlKVSLra0qFuOQ2dv\nsffkDdo3q1wiAlqP0gdPiyfVB1q9lhmH5nEn/e5jPW5eStuVYnqrcQUK2pw9+xc6nZbnnnsOlUph\n1QcjRmQHMCZNGkulSpX43/9+JiUlhQkTxrBmzQree28k8+bNJi4ujq+/3ohKpWL69Ml8/vkcZs6c\ni1KpQKFQmI9t+lqhgO3bt7B0aRhVq3owe/Z0wsJCWLRoKT///BNbtvxMWNhq3N3dmT59CjqdDpVK\nke/fmGmdSqUgMzOdoKCxTJ06k4CAtsTE3CIw8F127dpOly6vMX/+XNLS0vjf/35Gq9UyYkQgVat6\n4OHhAShIS0tjzJgPqVWrFhMnfoxCoWDhwsVcv36Nr7/+L3q9gaCg8axbt5rBg4eycOESJk0ax7Zt\nux/thyeeGFPR4fsDNrkVHc5tshGdXp+dYWOrNmfsWM4ma5rW+1GVL+XA6F6NOXo+hg27LpGQnMm2\nw9fYffw6vnXL06hGaapXcsHN2daq/ZavIyUli7up8Vy5FkfM3bQcwZmMLF2B22Nvq8LB1gZHezWO\ndjY42qlxsFOjsVGhUavQ2Chz/d9GrUR9729fpVSiUpq+zv6nVCpQqe6tu/eer1Ao7gVksoMTSmP0\nxRyoeVQqlQJ3dyfi4pKtatEJ6Zv8PKm+kYDNIzBP7S0BGyHEMywyMpI1a9bw7bff0r17dwA0Gg1h\nYWFW2+3Zs4c6dYyFE8+dO0dycjJdu3YlJiaGFi1aEBQURPny5Qt83tKlHYs0mOHm9vBPTOzsjVkT\napUSd3enB2xt9Np/anHo7C2uxSRzOymLOtXcH/q8T0ph+uBp87j7QKvTosxnBrHHTalS4l7KCbXq\nwbeAWm06rq6ulCtnnRFi2QfXrl3jzJlTrFy5And34+/qRx+NYsKECYwZM5q9e3ezYcMGatQwFtWe\nPDmIzp07M3/+POztNWg0atzdnay+trW14fnnn8fPrykAr73WhU8++QR3dyd27drOwIFv07ixsX7O\npEkTeOmlfbi6OuT7N2Zra7xWuLo6UKGCOz/++CMeHh4kJSWRmZlC6dLuJCfH4+SkYd++PWzatIlq\n1YwZO2FhK9BoNFy9ehWDQc+IESPQaGyYN+9T81P4LVt+ZsOGDXh5GYetjB79IaNHj2b8+DE4O9sb\nM7MLeA0oyY4dO8ann35KREQEpUqVYtCgQfTu3ZvTp0/Tq1cv7OyyazwGBgYydOhQDAYDCxYs4Lvv\nvkOn09G1a1cmTpyISpX/tNZFKa9pvXOfJSrnBzGtzmBRw0ZNutIY8LAK2DyG7BcThUJhDs5sOXSN\nHUeukanV8/vpKH4/HQUY33dcHTXY26pRqRTodAaS0zJJTMnK9TXkRqNWUsbNnjKudvf+Gb8u62ZP\nKWdbHO3VDxzuK4R4NBKweQSmgI1k2AghnlVarZZx48YRFBSEm1vuaf4Gg4HZs2cTERHBvHnzAGNA\nx8fHhw8++ABbW1tmz57N+++/z7ffflvgc9+5k1JkGTZubo7Ex6fkmB3kQRIS0wDjDFFxcckF2qeC\nq4bKZR25cTuFH/ZeIrBL/Ydu8+P2KH3wtHiSfTC15Rjupsc/eMPHoJSdG4kFvG+xtXUkPj6BmJi7\nqNU2Vn0QH5+Ag4MDkZHXsbe3BzTm33FHRzdiY2OJjLxOVlYWjo5u5nX29q4YDAYuXIgkLS2TzEwt\ncXHJVl9nZGTh6pq9T1paFlqtjri4ZKKionF2LmV1PICEhFQcHPL+G0tNTbXYLo1fftnGxo3rAahZ\nszbJySmkpmZw9epNsrKysLV1Mp/DyckYiEpKSiMxMRG9Xs+pU6c4deocVat6EBcXR3p6Om+99ZY5\niGwwGMjK0hIVdYekpDQMBkOBrwGWSlKQJyEhgffee4/Jkyfz8ssvc+7cOQYOHIiHhwfXr1+nTZs2\nrFixIsd+69evZ+/evfz0008oFAoCAwNZvXo1gwcPLoZXkbvsGjbWQSTLZwLmIVG5/Pnr9AbzzE2W\ntcfi7gVsVEoFdprHH6Cy06jp3qY6HVtU5cDpKI6cj+FqdBI6vQGtTm8VMMqNWqWgtIsdZdzsKetq\nR+l7wZjSrnaUdbXH2cGmRGR5CvEsk4DNIyjtci/D5t4NuRBCPGuWLVtG3bp1adu2ba7r09PTGTdu\nHBcuXGDdunWULl0agPfff99qu/Hjx9OyZUtiYmIoV65cgc5tMBjQFTxr+5GZZq94GJn30spVSsVD\n7fsfn8qs33mRI+Ex9G5fq8TMRFiYPnjaPIk+UKDC3bb0Yz1mngwUuP116zbExkbNH3/8QevW2X/j\ner2B2bNn4ODgQGDgcNLS0oiLu2uuzXL9+g1cXFxxdy+LRqPhxo0onJ2z1ymVSlxc3NDrDff+jg1W\nXxtnbslup6lUgk5noFy58kRFRZnXRUffMq+7/3XNnj2Nfv0G4uHhSUaGcaYetVrDn3/+yapVYaxc\nuZaqVT0AGDlyKHq9AWdnN2xsbIiOjsHJyRgMOnz4IAkJCZQpUwZnZ2dWrVrFhAlBzJkzk+DgFTg5\nuWBjY8Pq1eupXNmYYWPskzuo1Rpzu/7tfzs3b96kbdu2vPrqqwDUr18fPz8/Tpw4QWxsrDmD8n6b\nN29mwIAB5mt7YGAgixcvfqiATWGGwD7MMEZTTRaNxnr4qtoi40ahMA55yP1o2QEbR3sb82w9SanG\n3zsnexurYz1urk4aXvL35CV/TzKzdNyMTSE2IZ3ElEzSMrToDcb2O9nb4OZki5uzLZ6V3VDodUUz\ntdC/jAwDzpv0Td6eVN9IwOYRODtqAEjL0KHV6VEXYUqzEEKUBFu2bOH27dts2bIFMM6GMnr0aIYN\nG0avXr0YNGgQDg4ObNy40SoDJywsjFatWlG/vjF7JDMzEwBbW9uifxFPkGkWjAdN6X0///oV+G7v\nZTKz9Px+KooX/TyeRPOEyJetrS2BgSP47LM5KJUq/P39SU5OZtWqMI4dO0Jo6CrKli1Hs2a+LFky\nn48+mkhKSjKrVq2gY8cXUSqVdOz4EqGhwUybNgelUsmyZYvx92+Vo+hwQXXu/CorVy6nVas2lC5d\nhrCwZXluGxFxhZ07t/H224PYuXMrpUuXoVQpdy5evIBKpcTW1hadTseOHVv566+T1K/fEJVKRfv2\nL7BqVSgffzyd9PR0li5dRM+efQBjsU2lUsl7771P79492Lz5e157rQcvvPAioaEhjBsXhFqtZt68\nOURHR7Fs2RdoNBoyMzPJysrCxqZkBF8Lo27duuYsSTBm3Bw7doyuXbuycuVKNBoN7du3R6/X89JL\nLzFq1Cg0Gg0RERHUrFnTvJ+XlxeRkZEYDIYCZ288yhDYggxjNNwLw7g42VllNSVnZhdWdXV1wN3N\nHjt7TY79nZwdzJk3ri52qNXW2TRl3fMfsve4VSjvWmTneprJMOC8Sd/k7XH3jQRsHoGDRcpjeqYO\nJ3sJ2Aghni3btm2z+r59+/ZMnjyZ//znP/Tv358yZcoQHByc40NKREQEv/32G0uWLEGtVjN79mye\nf/55XF2frptM0xP1hx3j72CnpmW9Cuz/6yZ7/7xBR9+quRaPFOJJ6969J05OzqxZE8bMmZNRqVTU\nrVuf4OAVVK9u/BA+depMFi/+nF69ugDQseNLBAaOAGDkyNEsW7aEAQPeICMjk4CAtowc+VGh2/PC\nCy8SGRnB4MEDsLe3p1OnzgC5BkLGjJnAvHlz2LBhHRUqVGTKlJnG2h++LWnXrgP9+/dGpVLi7V2P\nl156hatX/wZg1KhxLFkyn969u6NQKOjatTuvvvoaJ04cMx/bxcWF998fxfz5n9CqVQAffjiGZcuC\n6devF+np6TRu7MOMGXMBqFGjFtWqVadz5+dZs2Y9VapULfTrLymSkpIYOnQo9evXp3379mzatAk/\nP/kVEQ8AACAASURBVD/eeOMN7ty5wwcffMCSJUsYM2YMaWlpVrVt7O3t0ev1ZGZmFjhIX5ghsA8z\njDEt3ZgJo7s39M4k0SKLPu5uMkq9juQU4zAjpUJhrgVzJy6Z9HsZNtpMLQaD9Qw6HmUdCzUk7kmR\nYa75k/7Jm/RN3h61b/IK6ioMuZU6/xe7fTupUPsVpqpzdFwqk8IOAfDJUH/KudkX6twlnVQDz5v0\nTf6kf/L2KH1TtqzzE2rVozMFbFxdXenTpw+2trZWhSXr1avH+vXrSU5OZvbs2ezZs4esrCz+85//\nMGXKlIcK2BT2ev+wHuVntePINf67+zIVSzswe3DLh9r3anQS0788CsCI7g1pWrvsQ+3/OMnfsvQB\nlIw+uHTpIqVKuVOmTBkA/v47kv7932Dnzv3Y2to9YO9HV9R9UBKv9//88w9Dhw6latWqLFq0yCoY\nY7J9+3YWLFjA9u3badq0KWvWrKFx48YAXLx4kW7dunH27NkCn7Mw1/uH+VlN/uIwN2JTeKN9TTr5\nZmc03opLZeK9e/1Ph/pT1s2e7UeusXH3ZWxtVObZlGYP9mP5j2e5fjuZnv+pgVZv4If9EebjDOxc\nh4BGBZ92/kkrCX/LJZn0T96kb/L2qH2T1/VeMmwegWVRsbR0bTG2RAghSobdu7Onrb1w4UKe2zk5\nOTF37tyiaFKx0uoLl2ED4FnBmXrVShH+911+PvA3TWqVkeKP4pl36NABjh07wty5n6NSKVm/fi0+\nPk2LJFgj4OzZswwaNIguXbowfvx4lEolCQkJhIaGMnz4cPNQt4yMDHP2TI0aNYiMjDQHbCIjI6le\nvXqxvYbc5DVLlOUl9/5ZomzUSnPARqc3oLtXbEmlUnLfiCiqV3R5Es0WQjwDZAzPI3Cwzb4amwqN\nCSGEECa6e4UsH7aGjcmrz1UDjNk2ZyLjHlezhPjXeuONvlSsWJFevbrQpUsnkpISmTx5RnE365kQ\nGxvLoEGDGDhwIBMnTkR5LxDt7OzMzp07CQkJISsri6tXrxIaGkr37t0B6NKlC6tWrSI6OprY2FhW\nrFhB165di/Ol5JCpNQZebFR5T+ttniVKnx2wMdHpDObCxWqVAluLGaFsbVRULC31PoQQhVOkGTbh\n4eFMmTKFy5cv4+npyfTp0/Hx8clz+4MHDzJw4ECOHz+Oo2PJu9DZqFWoVQq0OgOpErARQghxH63O\nVHS4cM9Hald1o1YVVy5dT+DnP/6mgZe7ZNmIZ5pGo2HChMnA5OJuyjNn06ZNxMXFsXz5cpYvX25e\n3r9/f0JDQ5k1axYtW7bEzs6ON954gwEDBgDQt29fYmNjef3118nK+n/27jy+qSpt4PjvJmnaJi3d\nKDuUssgmgohAgREVxQFZxhFBGR1kBCngMsUdRkQWd5gXHIeCiBuMOoyOOIjLIIICAjLiwqpIWQuU\n7nuz3fePLE3aJk3bdH++H/k0Pbn35uQgNzfPfc5zzIwbN45p06bV19uokPcMm9LzrTNQ4yxN4R7c\nsdhsHud79yW849qEy4o6Qohqq7OATUlJCYmJiSQmJnLbbbexadMmZs2axdatWysMxuTk5DBv3jwa\neomd0GAdeYVmybARQghRjsWZIl/Ni3VFURg3rDPL3/uB4+dyOHo6m15xUYHsohBC+MV5He/NG2+8\nUWG7VqslKSmJpKSkWupZzZmtFQds3AMtzqlQqlpxho3VNQVWITioNGAT37bh1SESQjQedRaw2bNn\nDxqNhilTpgAwceJE3nzzTXbs2MGYMWPKbb9w4ULGjBnD2rVrq/Q6iqJUuYo8VH/ddIMjYFNstqKt\nZsp7Q1dba8o3BTI2vsn4eCdj0zxYXXdcq//33KdzNPFtw0k5n8fm3SclYCOEEAGkqqpfNWzKTonS\nuQdsbKprCqxWq3hkVXaIrbvlvIUQTU+dBWxSUlLo2rWrR1t8fDwnTpwot+1HH31Ebm4uDz30UJUD\nNjExxhqli1d13fRwo56LWUWg0XhdiqupCPSa8k2JjI1vMj7eydg0ba5lvas5JQrsNyLGDu3My+//\nxJFTWRw/m0O3Dk1r+XMhhKgvVpvqCsb4qmHja0qU1eo2JUqjKZNhIwWHhRDVV2cBm8LCQkJDPZe9\nDgkJobi42KMtNTWVFStW8I9//AOz2Vzl18nIKKh2hk111k3XO6LrGVmFZGbmV/2FG4GarinflMnY\n+Cbj411NxqapB4ebEmsNp0Q59evWkg6xYZy9lM/mb07y59v6BaB3QgghnNk1AEFBnss7eWTY4Dkl\nSqPYz+1Wm4rF5l50WEPX9i347eBOtDDoaddSbswIIaqvzgI2oaGh5YIzxcXFGAwG1+82m43HHnuM\npKQkWrduzdmzZ6v8OqqqYrVWv582m1qlddND9PYhLCw2N/m16Ks6Ns2JjI1vMj7eydg0bZYAZNiA\n/S7v2KFxJG86xI+/ZnDqQh5xbaQughBC1JSzfg1UkGHjXsPGsZmzlo1Go7gCNu41bHRaBUVRmHRd\nt1ruuRCiOaizZb27dOlCSkqKR1tKSgrdupWezC5cuMAPP/zAwoULGThwIOPHjwdgxIgR7N+/v666\nWiWhjqW9C0tqECUSQgjRJDkzbGpSw8ZpYI9WtIm23+T4z+6TNT6eEEIIsLhn2JQtOuyxrLcjw8ax\nuaIorvqVzmXBoeYZlUII4a7OAjYJCQmYTCbefvttzGYz//rXv0hPT2f48OGubdq1a8ePP/7I/v37\n2b9/Px999BEAO3bsYODAgXXV1SoJDbZn2MgqUUIIIcpyZdgE4AJeo1G4OSEOgO9+vkTK+dwaH1MI\nIZo7k4+AjfuUKGdmjc1jSpR9+xKTW8CmhhmVQgjhrs7OKHq9nldffZWPP/6YQYMGsX79elatWoXB\nYGDBggUsWLCgrroSUAYJ2AghhPCiNEU+MB+3Q/q0pm2MPcvmX9t/dd3xFUIIUT1mvzNs7D+dARtF\nU5phU2IuDdgE6nwvhBBQhzVsAHr27Mm7775brn3RokUVbt+hQweOHTtW292qEcmwEUII4Y2rCGV1\nquFXQKvRMHFEV17+wL5i1MGUTPp2iQnIsYUQojnyFbBRKlglyjklSqMo6BzZkyaPgI1MiRJCBI6E\ngGvIGbAplICNEEKIMqyOgI02gBfw/bu3dC3r/c7WXzy+bAghhKgas1v9mfJFh0sfOzMabTinRCml\nU6LMpedhqWEjhAgkCdjUkEyJEkII4Y1zSlQgAzaKojDlhu4oClzILOTTfacDdmwhhGhu3FeJ0vnK\nsHFMiVIdDxSl9NzuXsNGpkQJIQJJzig15MywMZltrtR3IYQQAtyLDnt+3JZYTVwoSKv2cTu3acH1\nV3YAYPPuk6RlF1W/k0II0Yw5sxR1Wo1HzRrwrGFTruiwxj3Dxr3osGTYCCECRwI2NWQIKS0DVGyS\npb2FEEKU8rasd/KPb7B470v8nPVrtY99yzXxtDDqMVts/OO/P0sBYiGEqAZnwKZs/RonZ8zGNSXK\ncarVKFJ0WAhR++SMUkPODBuQOjZCCCE8eVvW+0zeWQDOF1ys9rENIUHcfn03AH78NYPvfk6v9rGE\nEKK5qixg48yyccTfPaZEOYsOe2TYSA0bIUQAScCmhtwDNkXFErARQghRqqJlva02K0WWYgBMVlON\njj+4d2t6xUUBsP6/x8gvMtfoeEII0dw4a9iULTjs5Kxjo1Y0Jcqxj0kybIQQtUTOKDVkCNa6Hkvh\nYSGEEO5cy3q7XcDnmwtdj822mgVYFEXhrpt6oNdpyMk3sf7zYzU6nhBCNDdmcyUZNo5mZ6BGdZ8S\nVUGGjSzrLYQIJAnY1FCQTus6MUvARgghhDtrBVOiCswFrsdmW80/N9pEG7jtOvvUqH1H0vjm0IUa\nH1MIIZoLV4ZNJVOinIEaZ+DGY5Uoj2W95euVECJw5IwSAM5pUVLDRgghhDuLo+iB+6oh+e4BG2tg\npjBdN6A9fTrbp0a99ekxzqUXVLKHEEII8KfosKOGjXNKlGOqq0ZR0DlXiTJZXG0aqWEjhAggCdgE\ngDNgIxk2QojmLD09nYSEBL788ksAcnJymDNnDldddRXXXnstGzdudG2rqirLli1jyJAhXH311SxZ\nsgSrtemttOfMsNG53XEtcJsSZbLVrIaNk0ZRmD6uDxFhekrMVv7+758olLpqQghRKVfAxkvtGWf8\npdyUKI1SLsNGlvQWQgSaBGwCQAI2QggB8+fPJzs72/X7k08+icFgYPfu3axcuZKXXnqJ77//HoAN\nGzawfft2PvroI7Zs2cJ3333HunXr6qvrtcZaSYaNyRq4z40Io55ZEy5Hoyiczyhk1aaDrtcXQghR\nMX8zbFTH6bR0SlRpDRtn0WGpXyOECDQJ2ASAwRWwaXp3h4UQwh/vvPMOoaGhtG3bFoCCggK2bt3K\nAw88QHBwMFdccQVjx47lww8/BGDTpk1MnTqVVq1aERsby8yZM/n3v/9dn2+hVpQu6+2eYeNewyaw\nqzpd1jGSO0ddBsChlEze+vSYa2UTIYSoqf3793Pbbbdx1VVXccMNN/Duu+8CjTujstIaNhpvU6JK\nz+0mR9BH6tcIIQJNV/kmojJSw0YI0ZylpKTw+uuv889//pPf//73AJw6dQqdTkfHjh1d28XHx/P5\n558DcOLECbp16+bxXEpKCqqquu5mVkZRFOri2th5sV6dugTOKVH6II0ry6bA4rlKVKBT6EcO7EBa\ndhGf7j3N1z+exxCi444buvs9rhWpyRg0FTIGMgbQvMcgJyeH2bNn8+STT3LzzTdz5MgRpk2bRqdO\nnXj33XddGZXHjh1jxowZdO/enf79+3tkVCqKwsyZM1m3bh0zZsyo77cEgMVSfjU/d0rZKVGOdo2i\nlDt/S4aNECLQJGATAKGOpb1lSpQQormxWCw8+uijzJ8/n8jISFd7YWEhISEhHtuGhIRQXFwMQFFR\nkcfzoaGh2Gw2TCYTwcHBfr12TIyxRkGIqoqMNFZpe5tNdV3gR0YaiI4OA8CslJRupLW52gNp1sT+\nmKwq2/af4bN9ZwgJ0XPP+D41Hq+qjkFTJGMgYwDNcwxSU1MZMWIE48aNA6BPnz4MHjyY7777jq1b\nt/LZZ5+Vy6js37+/R0YlwMyZM1mxYkXDCdjYfAdsyq0SZSudElV2H8mwEUIEmt8Bm+Li4nIX38JO\natgIIZqrv//97/Tq1YsRI0Z4tIeGhlJSUuLRVlxcjMFgAOzBG/fni4qK0Ol0fgdrADIyCuoswyYy\n0kh2doHrQt0fzroIAEWFJWRm5gOQmZ/jai8sKXa1B9qdN3Yjr6CEb4+ksemrX8nMLuTuMT29finx\npbpj0JTIGMgYQN2PQW0EdKurV69evPjii67fc3Jy2L9/Pz169GiQGZX+ZkNZHX+PQTpNhRmPGrc+\narWKK8PGvm2ZDBtd+aybhqg5Z4r5Q8bHOxkb72prbPwO2IwbN46VK1fSq1evgHagKQjR2zNsis0N\nZz6uEELUhS1btnDp0iW2bNkCQH5+PnPnzmX69OmYzWZSU1Np164dYJ865bxo79q1KykpKfTr18/1\nXJcuXar02qqqUpdlEGw21TXFyR8lptLOaVBc++aZSmvYlFhMVTpm1SjcO643eq2GXQcv8PWP50nL\nKmL2LZcTbtBX64hVHYOmSMZAxgBkDPLy8khMTHRl2bz11lsezzekjMrKsqE0jkiQ0aivMECmc9S2\nCTXYn9c6gt5Gg94V7HEK1usaVJCtMs0xU6wqZHy8k7HxLtBj43fAxmw212nqeWPiTH9srnebhBDN\n16effurx+/XXX8+TTz7Jddddx9GjR1m2bBlLlizhl19+YfPmzaxZswaA8ePH89prrzFkyBB0Oh2r\nV69mwoQJ9fEWao37hbz7HdfaLDpcllajYdrNvYgIC2bLnlMcO5PNU+v2MWNcH3rFRdXqawshmqYz\nZ86QmJhIx44d+b//+z9+/fXXBplR6W82VGGR/TxsNVsrzHh0Fm7Pz7dnRJpM9oz64hJzaUEbB0Wl\n1rImA0my5XyT8fFOxsa7mo6Nt2Cv3wGbsWPHMm3aNG6++WY6duxYbnrU5MmTq9yppsKZ9lQ2yi6E\nEM3Z4sWLeeqppxgxYgQGg4FHHnnElVEzZcoU0tPTmThxImazmXHjxjFt2rR67nFgWa2lU6K0bumx\n+R4Bm9qfSqtRFCZe25X2LY28+dlRsvNNvPjOAa67sj23juiKIUTK2Qkh/HPo0CGmT5/O+PHjeeyx\nx9BoNMTFxTXojMrKsqEsjnO1RqNUuJ3zhrXFaj+O83pfcXvOydsxGqrmnilWGRkf72RsvAv02Ph9\nlbZlyxZCQ0PZtm1buecURWnWARvnhbhEGYUQzZ37Z0RkZCQrVqyocDutVktSUhJJSUl11bU655lh\nY781bLZZKLGaXO0mt8e1LeHyNsS1CWf1R4c4k5bPlwfO8e3RNEYP6cT1AzoQHKSts74IIRqf9PR0\npk+fzrRp07j33ntd7WFhYYwcObLRZlQ6AzbeVnhyxtudRYedPzWKUq5WhawSJYQINL8DNhUFaoSd\nsxiZBGyEEEI4WdwybJwX8e7ToaD2p0SV1a6lkSenDuTTvaf5aNdJ8ovMbPzyVz7/9gy/HdSJoZe3\nqXZ9GyFE45Cdnc2xY8ewWCyu6T5Ow4cP97rfv/71LzIzM1m1ahWrVq1ytf/xj39s1BmVFsed8MpW\niXJe5ztX/1MUxSN70tcxhBCiuqqUB52RkcHGjRs5efIkjzzyCHv37qV79+507969tvrXKGhlSpQQ\nQogyLG7psM7PiQJzocc2ZpsFm2pDo9TdRb5Oq2Hs0M4M69uW/+w+ydc/pJKTb+K9bcf51/ZfubJ7\nS4Ze3pZenaMk60aIJuaDDz5g4cKFmEzls/sUReHIkSNe901MTCQxMdHr8401o9Jq9b2st3Pak+oo\nWOMM3GiU8st4N4YVooQQjYvfAZvDhw/zxz/+kW7dunHw4EHmzJnDrl27eOKJJ0hOTiYhIaE2+9mg\naWRKlBBCiDLcg/jOLwL5poJy21lsFvTaus9qiQoP5o839eC3gzvx8e6T7D1yEZPZxv5jl9h/7BI6\nrYbLOkbQJz6a7h0i6G8MqfygQogGbeXKlUyaNIk///nPhIU1ntWMapPFca7WeVmK19lcNsNGo1HK\nTYHSVbUyshBCVMLvgM2zzz7L1KlTuf/++7nyyisBWLp0KVFRUbz00ku8//77tdbJhk4ybIQQQpTl\nOSXKEbAxlw/YmGzmegnYOLWKDGXamF7cPrI73x5NY+dP5zl+NgeL1cbhk1kcPpkFgKJAm2gDbaIN\ntIwIJTYyhJaRocRGhNDCqMcYElSunoMQomHJysri7rvvlmCNG4vFfq7Wesuwcd6YLVPDRlGUcvtI\nho0QItD8DtgcOnSIJUuWlGufPHky69evD2inGhtXho0qARshhGjoNCknAFAjI1GjoittU2KiwLHU\nomu7iAjU6BifbbqLeYQX5ZIX2gKtRkFzMgXO/0JYXjH54fZslVYXc+HXX1BadSzd92QKqCpqiwjU\nmAC0nToJNlulbcYLZ7m2hY0Rv40jO7QFh1MyObPvICnnsjhv1pFriOB8RiG2EymcV20U6g3kGiLs\n7yPnIlrVhhreAjU2lrBQHW1z0wjWKRAejjUmluAgDTFZF9ArYA0LxxIdg06jEH4pFS0qalgYtphY\ntFoF48VUtNhQwsIwR7dEURRCz58FmxWbYzuAkNSzKKoVmyEMW8tYUECfehatzYrVGIbaMhZFgaDU\ns2isVmxGA9aWrQDQnTuDYrG32Vq2dmuzYDMascW28mgjPAyrEk9ObjHKmTMoZjM2oxFry1YoioIu\n9axHG1BhW1DqWRSLGZvBgDXW+br2NtW9LfUcVNRmNoFbmzb1HIrZZN+uVZm20FCsrdrUvO18Koqp\nBI3BgFbfldwCE5w7h2IqqXA7NSQEa+u2VWu7cB6lpLhmbcEhWNtUse3ieZRi/9uCDCEQfbn939GF\n81BUBMHB2Nq199lmcxynvg0dOpTdu3czadKk+u5Kg2Gx+Vt0uEyGjdSwEULUAb8DNhEREaSmphIX\nF+fRfujQIaKjowPescZEMmyEEKLxiBncH4CChx6j8LH5nm1zH6Xw8b8AEJ0wAMVmo+ihR+Gl5+1t\nQ69CsVopSHqYwicW2NuGDUSxWCh88CEK5j9lbxt+NTFmM7dcfStv/eYudFqF6N8M4rclJRSP78+m\nP15PgaWQ5Q+/h978DwrnPEjBU4vt+44YglJUROHsByhYaL9REn3tUJTCAgpn3U/B00sBiLpuGJqC\nfAoT76Ng0TP2tuuHo8nPo3DmbAoWP1falpdL4b2zKFhifx9RN1yDJiebwhmJFCx9wd524zVosrMp\nuude1GdfIuHyNoy9dTCarCxyp97DLw8u5eAvl7htylQM+Tl8cfVYVl4zA5uqsuwfjxJZlMOWfr9l\n1chELgJPJc8kqjCHT664ib/fMAuAt5LvJqowm0/7juKVG2cD8MbqacQUZPFp3xv5+41zHG1/IqYg\nk88vv4GXR90HwOtr7qFlfgaf9xnJyzfdD8C6NfcQm5/Bf/uMZKWj7bVXZ9Aq7xJb+1zPipseAGDt\n2hm0zr3EF72v4/9++6Cj7V5a56axrde1/HX0nwFY89pM2uZc5MteI1g+2l5vY/VribTLucCXPUew\nfIyjbd0s2mWfZ0eP3/DSzQ8BkLxuNu2zUz3aVr0+hw5Z5/iqx3BevPlhAP7+xn10zDzLzsuG8vzY\nRwF45Y376JR5lp3dh/L8OHvb3958gLiM0+zqnsBz4x4D4OU3H6Bzxml2dxvCs+MfB2DlWw/SOf0U\n33QdzDMTnnC0/ZnO6SfZ03UQSyfMA+D/3k6i66UUz7b1c+madoJ9XQay+Hf2/+//un4u3dJO8G38\nQBbdYm9bvuEhul/8lW/jr2L2LU8CsGzDI1x28Rf2dx7A07+3/1t46R+P0OPCL/wv7koW3mr/t/Di\nPx6l54Wf+S6uP0/duhCAF955jF7nj3Egrh8Lbn0agOfffZzeqUf5vtMVPDlxkaPtCXqnHuGHjn35\ny232fx/PvfcEfc55tj373nwuP3eIHztczvxJ9n8zz/xzPn3PHuJg+z48Mdn+b2bpP//CFWcPcrB9\nb56YbP83s2Tjk/Q78xOH2vfi8cnPArD4Xwvof/pHDrfrxWO329sW/esprjz9A0fa9uDrtzZx44D2\nhN+fiH7Hl5ivuprsT74AIPzB2ei//ALzVQPJ/sS+YEf4n+eQ8+4HNAR9+vRh6dKlbNu2jfj4eIKC\ngjyenzt3bj31rP5UueiwWw2b8lOiJMNGCBFYfgds7rjjDhYsWMDDD9svOI4dO8ZXX33Fyy+/7Hel\n98OHD7NgwQKOHz9OXFwcTz/9NP379/fYRlVVVq5cycaNGykoKODyyy9nwYIFDbqwsdSwEUII4UvZ\nwpTh+jAKLIVetm549DotV/VsTXwrIyF6eyHioZe3oc8jI8jKK8H4ug6KoFdcNLeP7E5+kZngtfbt\nWkeFcmX3lhSbrK7pAoaQIFpHG7Baba4vQ0FaDcF6rasAqBANVW5++YK9jcXevXu54oorKCgo4ODB\ngx7POYvrNjfOc4636UyuosMVTYkqV3RYMmyEEIGlqGXX8/Nhw4YNrF27lvPnzwPQsmVLpk+fzt13\n313pviUlJdx4440kJiZy2223sWnTJpYtW8bWrVsxGo2u7TZu3Mjrr7/O66+/TmxsLC+//DLbt2/n\n3//+t199vHQpz9+340GrVYiODiMzMx+rtWqBl31HLpK86RChwTpeSbqmWq/fkNVkbJo6GRvfZHy8\nq8nYxMaG11KvGpfqnu+rOiVKExNFVNdOZGbmox7/1b6dH1Oijp3O4m/bzpBvaMFrj12P5mQKW1I+\n58vcQ7Tq0JMTOSdpdTGXGX3upH2HXvU+JcrVFt4CtWVLjzZNZAuiLou3j8GJlPLbnT4FVmvN2sLC\nUWNjXW2qxYLFYMQaE4vq3M5in+pkbdkSVQXt6VOoVis2gxFLy1h729nTYLZgNRqxxMSiqiq6s2dQ\nLRbXdgBB586guO3r3mYNLd9GeBjGLp3IySlEc/asfeqUwYglxv4+7FOdSl9DraANFYLOn0UxW7AZ\nDFicU6e8tpmxhZa2BV04V3GbyYy1TJvGbMYWEorZMXWqZm2paMwmMBowdO1Mbm4RmnPn0JhNFW5n\nCw7B7Jgm5XfbxfNoTCU1a9MHY3ZMk/K7Le0CmpJiv9t0xhB6XnMlWVkFqOdSa31KlJzv7apzvvf3\nc3bWsh2UmK3MueVyrurRqtzzz63/Hz+fzeHmhDhuHdGVx1d/Q1pWEX+48TIijHr+/mFp4Ou6Ae25\na1SPKve1rsn1mW8yPt7J2HhX07Hxdr73O8MmNTWVO+64gz/84Q8UFhZitVoJDw/HarXy008/0bdv\nX5/779mzB41Gw5QpUwCYOHEib775Jjt27GDMmDGu7SZOnMjNN9+MwWAgPz+fvLw8oqKi/O1mvSib\nKimEEKLhssV3qVKb4nbXtSr75psvkRea41o1xNY5nvSiaPLVELoE2W9UpLVuQX7HNqhRMaX7do4v\nf7yatMV1rnGbxxhUtF2nuFpp0zr+ANDVPs4eEzh6dCu3L70qyMiNquALVExP/9pa2ttcF2J6Ddbo\nCo4XW8G+FbW1qkFbaz/b2gS6zf5+Sy9GdVhjKxiDNjVpq+BCtQG2abWKK+OioiCMv211aefOnQwZ\nMgSdTsfOnTu9bqcoCsOGDavDnjUMFmslRYfLZNh4LOstq0QJIWqZ3wGbkSNHsmvXLqKjozEYDK72\nU6dOceedd/LDDz/43D8lJYWuXbt6tMXHx3PixAmPNkVRMBgMfPDBB8ybN4+wsDDWrVvnbzdRFIXq\nnCud05qqs8JFkM5xMW5Tm2R1+JqMTVMnY+ObjI93MjZNn2upWLfPBbPNDECoLgQFBRUVk9VcJEnO\nwwAAIABJREFUL/0TQjQP06dPZ9euXcTExDB9+nSv2ymKwpEjR+qwZ/VPVVVXDUqvRYfLLC7iPiWq\nbN0bb8cQQojq8hmweeedd3jllVcA+wlt3Lhx5ea3FhQU+FVfprCwkNDQUI+2kJAQiouLK9x+7Nix\njB07lrfffpvp06fz+eefExkZWenrxMQYazQHNzLSWPlGZURE5ANgVVWio5vuMonVGZvmQsbGNxkf\n7xrC2GRlZbFmzRoOHjyI2Vw+ePDuu+/WQ68aP9ddW7egnDM4o9fqCdIGYbKaMNkkYCOEqD1Hjx6t\n8LEoLTgM3rNjnF8ryq0SpSm/SpTUsBFCBJrPgM3EiRMJDQ3FZrMxb948Zs6cSXh4aZqoMxtmyJAh\nlb5QaGhoueBMcXGxR7aOO71eD8A999zDhg0b2LdvH6NGjar0dTIyCqqdYRMZaSQ7u6DKU5sKC0oA\ne4ZNRkZekyvaVpOxaepkbHyT8fGuJmMT6MDw448/zk8//cT48eMJC2u6Qee6ZjJbAdAHaUvbrPZi\npUEaHXqNPWBjlgwbIUQdU1W1wgC98/q7ubC4FTmvfJUo++/OgI2iUH5Zb8maFUIEmM+ATVBQEL/7\n3e8A6NChAwMGDECn83sWlYcuXbqwfv16j7aUlBTGjh3r0bZy5UqsVitJSfalM1VVxWQyeQSKfFFV\nFau1Wl0E7EGXmhRQMlts5SrGNxU1HZumTMbGNxkf7xrC2OzZs4e33nqLfv361Ws/mpq8QvuXofDQ\n0qorzmwavVZPkCbIo00IIWrb3r17WbRoESdPnsRmKw1WqKraLKdEWd1umHgra+BtSpRGUcpl1DTF\n0ghCiPrlM/qyfPlyZs2aRWhoKDt37vRZqGzu3Lk+XyghIQGTycTbb7/N7bffzqZNm0hPT2f48OEe\n2/Xr149HHnmE0aNH06VLF1avXk1YWBgDBgyowtuqW+7RdZtNRbIhhRCNSUxMDMHBwfXdjSYnt9Ce\nTRNuLL1j7axho9cEodcGebQJIURtW7BgAd26deOxxx4jJCSkvrtT79wzbIK8FR12/HRNiXIVHVbK\n1azxlqUjhBDV5TNgc+DAAcxmM6GhoRw4cMDrdv5MAdLr9bz66qssXLiQ5cuXExcXx6pVqzAYDCxY\nsACARYsWMWLECObOncucOXPIy8vjyiuvZO3atQ36y4R70VCrTfVcwUIIIRq4++67j8WLFzN//nzi\n4uIICvI8izW3FPlAcWbYtDC4Zdi41bDROzJsZEqUEKKupKWlkZycTHx8+RXlmiP3gE3lGTb2352B\nG0VDuax6CdgIIQLNZ8Dm7bffrvBxdfXs2bPC4pWLFi3y+P3222/n9ttvr/Hr1RVNmQwbIYRoTJYv\nX052dja33nprhc83txT5QMktcGTYGEoDXu41bIK0MiVKCFG3brzxRnbs2CEBGwf3Kcnegi2ly3o7\niw7b2zVKBUWHpYaNECLAfAZsfE2BcqcoCsOGDQtIhxojbZkMGyGEaEyWL19e311okvIcU6JaVDQl\nyq2GjWTYCCHqyty5cxk/fjybN2+mY8eOaMpkiCxbtqyeelY/zH4VHbb/dN6Uda0SpSjlsnKkho0Q\nItB8BmymT5/u10GaY5EydxpFMmyEEI3XoEGD6rsLTVKus+iwoYKiw1LDRghRD+bPn4+iKHTo0EFq\n2OCZYeMt2FKaYWP/XbWprvayS4HLlCghRKD5DNgcOXKkyS1RXRskw0YI0djcfvvtrFmzhhYtWjB5\n8mSf5/qKprIK32yqWpph45gSpaqqK5smyH2VKMmwEULUkf3797N+/Xr69u1b311pEDyW9fayymvZ\nVaJKp0SVD/JIwEYIEWg+AzZDhw7l6quvZtiwYQwdOpSOHTvWVb8aFalhI4RobIYPH+4qLvyb3/ym\nnnvT9BQUmV13Y51Toiw2Cyr2Rr1GVzolSjJshBB1JC4uDpPJVN/daDA8AjZeM2zsP1XXst7OosPl\na9jopIaNECLAfAZsPvvsM/bs2cOuXbtYu3YtNpuNYcOGkZCQQEJCApGRkXXVzwbNI8NGlYCNEKLh\nu++++1yPx44dS+fOneuvM02QczoUlE6Jci8urNfq0Wv15dqFEKI2zZo1i8cff5y77rqLTp06odN5\nfhUYPnx4PfWsflhslRcddpY+cG7qvqx32VWipIaNECLQfAZsWrRowahRoxg1ahQAZ8+eZffu3Xz6\n6acsWrSItm3bMnToUNef5sr9ZC0ZNkKIxmb06NH07t2bcePGMWbMGFq1alXlY2zZsoWXX36ZCxcu\n0K5dO5KSkujduzc333yzx3Ymk4kOHTrw2WefoaoqV111letuJcBVV13F2rVra/ye6lteQekdbOcq\nUe6ZNEGaIII09o9gKToshKgrSUlJADzzzDPlnmuONSmtjgwbjaJ4ZMy7c8+wUVVnniRoNOWzcrQy\nJUoIEWA+AzZldejQgUmTJjFp0iRUVeXQoUPs3r2bNWvWNOuAjUZq2AghGrGtW7fyySefsHnzZl58\n8UUGDBjAuHHjuOmmm4iIiKh0/5SUFObNm8e6desYMGAAu3fv5t577+Wrr77iwIEDru0uXbrELbfc\nwvz58wE4deoUAN99912Tq5eW66hfE6zXEhykBUqX9AZHho1GlvUWQtSto0ePBuQ4P/74I7Nnz3at\nKPvTTz8xadIkj0LGM2fOJDExEVVVWb58ORs3bsRqtTJhwgSeeOIJtFptQPpSExZH0WFv06HALcPG\npuKeSK9UsEqUTIkSQgRalQI2mZmZGAwGQkJCOHz4MDt27ODyyy/n3nvvra3+NQpaqWEjhGjE2rdv\nz/Tp05k+fTpnzpzhs88+4/3332fJkiUMGzaMVatW+dw/Pj6eXbt2YTQasVgspKenYzQa0ev1Hts9\n9dRTjB49mmuuuQaAw4cP06NHj2oHaxRFwUuNyIByBuW93X2tSH6RPQjTwqB3XdBbsbieDwkKIjjI\nWdvGjFarcC7/AjklufSOuSxQXQ+Y6oxBUyNjIGMAjXMMJkyYwNChQxk2bBiDBg0qd26uClVVef/9\n93nuuec8Ai5HjhzhmmuuYfXq1eX22bBhA9u3b+ejjz5CURRmzpzJunXrmDFjRrX7ESjOGja+MmNK\niw6XFh4GR1ZOmc8vKToshAg0vwM2W7duZe7cuSQnJ9O+fXvuvPNO2rVrx9q1a5k7dy533XVXbfaz\nQZMMGyFEUxEVFUVsbCytW7fm559/Ji0tza/9jEYjZ86c4aabbsJms7Fw4ULCwsJcz3/zzTd89913\nvPjii662I0eOkJ+fz4QJE0hLS+Pqq69m/vz5tG7d2q/XjIkx1mlmTmSk0e9tzY6PguiIEKKj7eOQ\nbiv9yG3dMoqIXHu7VbESERnKIzuSyTMV8NJNf6FTZPvAdTyAqjIGTZWMgYwBNK4xeOGFF9i9ezdv\nvPEGc+fOpWfPnq6alH379q3SeTQ5OZlPPvmExMREXn31VVf74cOH6dmzZ4X7bNq0ialTp7qm286c\nOZMVK1Y0qICNrwyb0mW9VY8pvBrFsbS3VnFl6kgNGyFEoPkdsFmxYgX3338/Q4cOZdmyZbRr146P\nP/6Ybdu2sXTp0mYdsJEMGyFEY5afn8/WrVv59NNP2b17N+3atWPs2LEkJSURHx/v93Hatm3LDz/8\nwP79+5k9ezZxcXEkJCQAsGbNGv70pz9hNJZ+ydHr9fTv358HH3yQ4OBgli5dyv33388///lPv14v\nI6OgzjJsIiONZGcX+H2OT0svAMCg15KZmQ9AelaO6/mCHBOWEvsXhSJTCSnnU8kz2fc5dO44YbbK\np6LVpeqMQVMjYyBjAHU/Bs6Ab0306NGDHj16MG3aNEwmEwcOHGD37t0sWrSIs2fPMmjQIBISEhg6\ndChxcXE+j3XrrbeSmJjIvn37PNqPHDmCXq/n+uuvx2azMXr0aJKSktDr9Zw4cYJu3bq5to2Pjycl\nJQVVVf0OFlUno9KfbCjnX6FOq/EabHFe56sqHv3V6ez7aDUaLFYrAPogbaMI2jTGTLG6JOPjnYyN\nd7U1Nn4HbE6ePMnYsWMB+PLLLxk5ciRg/xBIT08PaKcaG8mwEUI0ZgkJCURFRTF69GjmzJlD3759\nq3Uc52ojCQkJjBo1ii+++IKEhATOnz/Pt99+y7Jlyzy2v//++z1+f+yxxxgyZAhpaWl+FT5WVRXH\nNXKdsNlUrFb/zvE5jqLD4YYg1z7FFnubVtGCqkGHY1lvq5n0wmzXvumFWX6/Tl2ryhg0VTIGMgbQ\neMdAr9czePBgBg8eTFJSEjk5Oa4VYdetW8d///tfn/t7OzdHRUUxePBgJk+eTEZGBg8++CArV67k\n4YcfpqioyKO2TWhoKDabDZPJRHBwsF/9rklGpa9sqOAQ+3k4WK/1GhwLDbVvowvSEhFpKD1uhIHo\n6DB0Og0lZvuHUUy0MSBBtrrSmDLF6oOMj3cyNt4Femz8Dti0bt2aw4cPk5WVxfHjx3n66acB2L59\nOx06dAhopxobybARQjRma9asYciQIdW+GN6xYwevv/46b7zxhqvNbDYTHh4O2IP8gwYNIjo6utzr\nDhs2jD59+gD2FaQAvy/gGzJn0WHnClEAJsdqUEGOYsNBWkfAxmYmq6Q0YJNZnFVX3RRCNEM2mw1V\nVdFqtRQWFlJUVMSdd97JokWLqn3M5ORk12ODwcDMmTNZvnw5Dz/8MCEhIZSUlLieLyoqQqfTVelc\nX52MSn+yoXJziwBQwJUNWZbJZK8/VlJiJsNtm7y8IjIz83G/mV6QX0ymruFnHki2nG8yPt7J2HhX\n07HxFuz1O2Dzpz/9iQcffBCA/v37c9VVV/G3v/2N5ORkXnjhhSp3qCnxyLBR5X9cIUTD995773n8\nfvr0aa/bTp482eexevfuzcGDB/nwww8ZP348X3/9NTt27HBNbfrhhx/o379/uf1OnDjB119/zcqV\nK9HpdCxdupSRI0f6tTJVQ+dc1ruFe8DGsRqU3hGocV8lKqvYPWBT+lgIIQJp//79PPDAAyxbtoyu\nXbsyceJETCYTRUVFvPDCC4wZM6bKx8zJySE5OZk5c+a4apeVlJS4AjJdu3YlJSWFfv36AfaVBbt0\n6VKl16hJRqWvbCiTxVF0WKPxnjGllh7HYindRlXBalU9Cw072hqLxpopVldkfLyTsfEu0GPjd8Bm\nypQp9O/fn9TUVIYPHw7A8OHDueGGG7wWGWsuNIqCgv18LpFGIURjUNFKHhVRFKXSgE1sbCzJyck8\n88wzLFq0iM6dO/PKK6/QtWtXAM6dO1dhwOYvf/kLS5cuZfTo0ZjNZq699loWL15c9TfTAOUW2oMz\n4cYgV5vZsay3M1ATorNPEbCpNs4XXHRtJxk2Qoja8vzzzzNmzBj69+/P66+/TnBwMF9++SWbN2/m\nb3/7W7UCNuHh4fz3v/9FVVUeeughUlNTSU5OZtKkSQCMHz+e1157jSFDhqDT6Vi9ejUTJkwI9Fur\nFoul8qLDHqtEuV3nO1eIcs+0l1WihBCBVqVlvXv37k1xcTEff/wxN910E0ajsdLiZM2FRqNgtalS\nw0YI0Shs27YtoMcbOHAgH3zwQYXPrV+/vsL2sLAwnn322YD2oyHILzJTVGJPoY8KK035L82wsWfd\ntDbEup47nHnM9TizOLtKxTiFEMJfx44dY8WKFYSGhrJt2zZuuOEGV12bhQsXVuuYGo2G5ORklixZ\nwpAhQwgJCWHy5MlMnToVsN/0TU9PZ+LEiZjNZsaNG8e0adMC+K6qz7m6k69Ai/NUrKqq57LemooC\nNnLeFkIElt8Bm8zMTGbNmsWhQ4ew2WwMGjSIZcuW8euvv7Ju3To6duxYm/1s8LSOgI1k2AghGqOM\njAw2btzIyZMneeSRR9i7dy/du3ene/fu9d21RudsWmmNgw6tSucjm8vUsAnXhxEWZCTfXECeqXQf\ns81MvrmAcH3jKVwphGgcIiMjOXfuHFarlUOHDpGUlATAjz/+6Fexd6fBgwezd+9e1+/dunXzqGPm\nTqvVkpSU5HqthsRi8yPDxhGxsdlU3CsfOAM5Wrdgj1YybIQQAeb3WWXp0qXExMSwd+9e15zU559/\nnk6dOrF06dJa62Bj4UqXlICNEKKROXz4MDfddBPbt29n8+bNFBYWsmvXLiZOnMg333xT391rdM44\nAjbRLYIxhpROiSpbwwagrbF1hceQaVFCiNpw2223MXv2bCZPnkxcXBwJCQm8/fbbPPbYY66MmObE\nWWfCV6DFme3obUqUzi3DRitLHQshAszvDJvdu3fz5ptvYjSWLlMVERHB448/zh133FErnWtMnCdo\nmRIlhGhsnn32WaZOncr999/PlVdeCdiD9FFRUbz00ku8//779dzDxsUZsOkY65khY3LUsAnyCNi0\n4ZfsE+WOkVmcTVyL5p25KoQIvPvvv59evXpx9uxZxo4di0ajoVOnTqxcuZJrr722vrtX5yxWe4ZN\nkI+AjaayKVFaCdgIIWqP3xk2VqsVmyNt0F1eXh5arTagnWqMJMNGCNFYHTp0iPHjx5drnzx5Mr/+\n+ms99KhxcwVsWnsGbMzODBtN6cpR7cI8M2x0iv3zVDJshBC1YdKkSXTq1Im7776bli1bAjBixIhm\nGayB0ho2Wr+KDquobgEb15Qox3rjOq0itceEEAHnd8Dmhhtu4MUXXyQzM9N1Mjp+/DiLFy9m5MiR\ntdbBxkIjGTZCiEYqIiKC1NTUcu2HDh0iOjq6HnrUeFltNs6lFwDQsVW4x3OmMjVswJ5h466TI6sm\nS5b2FkLUgrNnz6LTVWnNkSbNmWHju+iw25Qot8v8sqtESf0aIURt8PuMPW/ePObPn8+wYcNQVZVx\n48ZRUlLCddddx7x582qzj42C1i36LoQQjckdd9zBggULePjhhwH7KiJfffUVL7/8coNZyaOxuJBR\n6PoC0LFVmSlRldSwMQYZaGOI5UTOScmwEULUittvv505c+YwefJkOnToQEhIiMfzw4cPr6ee1Q9X\nwMbHVCbXlKgyi4u4athoy9eyEUKIQPE7YBMWFsaKFSs4c+YMv/76KxaLha5duxIfH1+b/Ws0nCdt\nybARQjQ29957L0ajkeeee46ioiLuu+8+WrZsSWJiYrMsQlkTzulQ+iANrSJDPZ5z1rBxD9gYgwxE\n6MPJMeURHRxJdEgUIFOihBC14+9//zsAzz33XLnnFEXhyJEjdd2leuVP0WGNW4ZNhVOiHPtKho0Q\nojb4DNikpKRU2B4XF1dum+YeuNFKDRshRCNis9nQaEovLv/whz/whz/8gcLCQqxWK+Hh9uk8ly5d\nIjY2tr662eg4AzYdYsNcU2WdnFOi3GvYgH1aVI4pj8iQ0oDNpaJMVFWVeghCiIA6evRofXehQSmd\nEuX9XOs8D9uLDpe2u4oOazwzbYQQIpB8BmxGjx6Noige0WR3zueaY0S+LKlhI4RoTB588EH++te/\nlqtlYDAYXI+3bNnC008/zd69e+u6e43WsTP22jPxbVqUe85ZdNi9hg3A1W2u5ETOSa6M7Utroz04\nVmwtJteUT0RweLnjCCFETZhMJj777DNOnjzJXXfdxbFjx+jataurCHFzYnFct/uqYeOMvdvUiqdE\nuQI2GsmwEUIEns+AzRdffFFX/Wj0JMNGCNGY7Nu3jzlz5vDyyy+j13tmfOTk5LBw4UI++eQThg4d\nWk89bHwKis2knM8FoHfnqHLPV1TDBmBI24EMajMAjaKhyFLkak8rTJOAjRAioM6cOcPUqVOxWq2k\np6fzu9/9jg0bNrB3715ef/11evfuXd9drFNWf4oOa9ymRFF+SpTONSVKMmyEEIHnMxTcvn37cn8K\nCgr44YcfOHDgADk5Oa725q40w6b80udCCNHQvPXWWxw+fJiZM2dSVFQaJNi+fTs333wzO3fuZPHi\nxaxbt64ee9m4HD2Vhara77r2jCsfsDFXUMPGSaPYP45DdaGE6+3Fii8WXqrF3gohmqOlS5cyfPhw\nvvzyS1ewfvny5Vx77bU8++yz9dy7uudc1tvXdCaN+5Qot8v8slOitJJhI4SoBX6fWXJzc0lMTGT8\n+PE8/fTTLFy4kN///vdMmTKFvLw8v45x+PBhJk6cSP/+/ZkwYQLff/99hdv985//ZNSoUQwYMIBb\nb72V/fv3+9vNeiMZNkKIxqRHjx784x//4MyZM9xzzz1cvHiRefPmkZiYSN++fdm8eTO33XZbfXez\nUTmUkglAl/YtCA0un8DqrYZNWa0N9mlRErARQgTa//73P+6++26PGmY6nY5Zs2Zx8ODBeuxZ/TA7\nMmx8Fx22/7TZVI/VYF1TorRSw0YIUXv8DtgsXbqUtLQ0Pv74Y/bu3cv+/fv5z3/+Q3FxMS+88EKl\n+5eUlJCYmMjvf/97vv32W+666y5mzZpFQUGBx3Z79uxh+fLlrFixgv3793PnnXeSmJhIVlbDXjFD\natgIIRqbjh07smHDBnJzc7n++uvZvn07L730EqtWraJ169aVH0B4OHTSHrDp0zm6wuddNWwqyLBx\n5wzYpEnARggRYHq9ntzc3HLtZ8+exWg01kOP6pe1ikWHfa0S5WtalRBCVJffZ5Zt27bx1FNP0bVr\nV1db9+7dWbBgAZ9//nml++/ZsweNRsOUKVMICgpi4sSJtGzZkh07dnhsd+HCBe655x569eqFRqPh\nlltuQavVcvz48Sq8rbqndS35JwEbIUTj0bp1azZs2ECfPn2IjY2VmjXVdDGrkEvZxUDFARtVVUtr\n2Gh8B2xaSYaNEKKWjB8/nsWLF7uyaXJyctixYwcLFixg7Nix9dy7uueaEuVjOpPGrYaNz6LDkmEj\nhKgFPosOe2yo0xEcHFyuPSQkBLPZXOn+KSkpHsEesC8FfuLECY+23/3udx6//+9//6OgoKDcvt4o\nikJ1ppA6T8Zll2H1lzO6rqpNr+hYTcemKZOx8U3Gx7v6HpudO3d6/H7PPfewePFi7rrrLh599FG0\nWq3rueHDh9d19xqd/x2zB1eMITri25UvFGxVrdhU+53cimrYuHNm2GQUZ3GxIA2dJoiY0PI1cYQQ\noqoeeughli9fzpQpUzCZTEycOBGdTscdd9zB3Llz67t7dc6/Zb3tP21elvWODg8BIMrxUwghAsnv\ngM3QoUN54YUXWL58OZGRkQBkZmby4osv+nVHtrCwkNDQUI+2kJAQiouLve5z/PhxHnjgAR544AGi\noytOMS8rJsboSl2sjsjI6qWDBjvqFQTpdURHh1X79Ruy6o5NcyBj45uMj3f1NTbTp0+vsD09PZ2Z\nM2e6flcUhSNHjtRVtxqtfYcvAjCwZ6sKC08669cABPlZw8am2li09yW0ipb7+k/nsij/blwIIYQ3\nOp2ORx99lAcffJDTp09jtVrp1KkTBoOBjIwMYmJi6ruLdcrq17LezilRVDgl6voB7WkZEUKPTpG1\n11EhRLPld8Dm8ccfZ+rUqYwYMYIOHToA9vmuXbt25Zlnnql0/9DQ0HLBmeLiYgwGQ4Xb79y5k6Sk\nJKZNm8a9997rbzfJyCiodoZNZKSR7OyCahUOtjki9IWFJjIz86vegQaspmPTlMnY+Cbj411NxiYQ\nQeGjR4/W+BjC7nxGAafT7Of9Qb0qrv3jrF8DlWfYxIR43qCwqlbW/vQ2Dw+c45ouJYQQ1dGrVy92\n7dpFdHQ03bt3d7WfOXOG8ePHc+DAgXrsXd2z+LGstzNg463osD5Iy8CerWqxl0KI5szvgE1sbCwf\nffQRX331FSdOnCA4OJguXbowdOhQvzJaunTpwvr16z3aUlJSKpwv+/7777N06VIWLVpU5fm0qqpi\ntVZpFw82m4rVWvUvls6TtsVqq9b+jUF1x6Y5kLHxTcbHOxmbxm+vI7smIkxPj44V32EttJQunR6i\nLT+92J1WoyUsyEi+2V6U36gzUGApZOMvHzGn3z0B6rUQorl4//332bhxI2C/Tp4xYwZBQZ6B40uX\nLtGqVfMLOlhcq0RVPiXKfVlvBWqU0S+EEP7yO2AD9jTK6667zqOegbN+jV7vO8U7ISEBk8nE22+/\nze23386mTZtIT08vVxvhm2++4emnn2bdunUMHDiwKt2rVxpZ1lsIIZodm03lm0MXALi6ZyuvNYmy\nirMBUFCICG5R6XHHdbmJL8/uYlL3CWSVZPP2kX/yc+Zxii0lhOh8B3yEEMLd6NGjOX/+PADff/89\ngwYNKrcilNFoZNSoUfXRvXrlKjrsK8PGreiwc0qUBGuEEHXF74DNvn37ePrppzl58iQ2Z3gZ+4nL\nnxoHer2eV199lYULF7J8+XLi4uJYtWoVBoOBBQsWALBo0SJeffVVzGYzM2bM8Nh/xYoVXHPNNVV5\nb3VKlvUWQojm538/X3KtDvWbK9p53S67JAeAcH0YOk3lH73D2w9hePshAOSbC1BQsKhWjmUdp19s\nnwD0XAjRXBgMBu677z4A2rdvz80331zpjdbmwr+iw6XLejunRFWn/IIQQlSH3wGbJ598km7duvHY\nY48RElK9Kug9e/bk3XffLde+aNEi1+N169ZV69j1TSsZNkKIZmzLli28/PLLXLhwgXbt2pGUlMQN\nN9zATz/9xKRJkzw+N2bOnEliYiKqqrJ8+XI2btyI1WplwoQJPPHEEx4rVDVkqqqyZc8pAC7vEk3H\nVt5rCzkzbCKDI6r8OmFBRuIjOnEi5xSHMo5IwEYIUSXvvfcet9xyC3q9HpPJxL///W+v206ePLkO\ne1a/bDYVZ0karc8aNo7t3aZEaSTDRghRR/wO2KSlpZGcnEx8fHxt9qfRcp64JcNGCNHcpKSkMG/e\nPNatW8eAAQPYvXs39957L1999RVHjhzhmmuuYfXq1eX227BhA9u3b+ejjz5CURRmzpzJunXrymVY\nNlSHT2Zx6kIeAKMHx/nc1plhExVSvVVE+sT0dARsjrkyW4UQwh+rV69m1KhR6PX6Cs91XHvaAAAg\nAElEQVTFToqi+B2w+fHHH5k9ezY7d+4EICcnh3nz5rFnzx7Cw8OZM2cOt912G0CDDc47s2sAgnwE\nbBRX0WGZEiWEqHt+B2xuvPFGduzYIQEbL7QyJUoI0UzFx8eza9cujEYjFouF9PR0jEYjer2ew4cP\n07Nnzwr327RpE1OnTnUVupw5cyYrVqxocAGbnPwSsousRIaWfrkoKrHw1mf2lbbi27agZyXLuWY5\nAjbVybAB6BPTi/+c+IzskhxO550lrkXHah1HCNH8rF27luzsbLKzs3nttddqdCxVVXn//fd57rnn\nPAIuTz75JAaDgd27d3Ps2DFmzJhB9+7d6d+/f4MNzlvcCv77KjqskSlRQoh65HfAZu7cuYwfP57N\nmzfTsWNHNGXOVMuWLQt45xoTKToshGjOjEYjZ86c4aabbsJms7Fw4ULCwsI4cuQIer2e66+/HpvN\nxujRo0lKSkKv13PixAm6devmOkZ8fDwpKSl+Z5AoilInF80vvvM9Z9Ly+eNvezDyqg7YVJV3vviF\nS9nFaDUK08b0RKfz3RFnhk10aITPLwbexEW0IzokisziLN44/A6PXn0fYXpj5TsGiPMzzltR5eZA\nxkDGABrnGIwZMwZFUXyeW/2tSZmcnMwnn3xCYmIir776KgAFBQVs3bqVzz77jODgYK644grGjh3L\nhx9+SP/+/RtscN7iVpNT5+PDRHGfEuUM2EiGjRCijvgdsJk/fz6KotChQ4dq17BpyiTDRgjR3LVt\n25YffviB/fv3M3v2bOLi4oiKimLw4MFMnjyZjIwMHnzwQVauXMnDDz9MUVGRx+dJaGgoNpsNk8lE\ncHDlKyHFxBjrJC09vn0EZ9LyWf/ZMWyKwtGTWew/Yl/K+w+/7cmVvdtWeoxskz1g0zGmDdHR3mvd\n+HLfkKks/epl0grTWbJ3OZe37snEPmNoF966WserjsjIugsSNVQyBjIG0LjG4IsvvgjYsW699VYS\nExPZt2+fq+3UqVPodDo6dizN/IuPj+fzzz8HqHFwHqoXoK8suOac3gSg12u8BtOdAXlVLZ0KpdEo\n1Qq+NxSNMfBYl2R8vJOx8a62xsbvgM3+/ftZv349ffv2DWgHmorSJf8kYCOEaJ50OvtHSkJCAqNG\njeKLL74gOTnZ9bzBYGDmzJksX76chx9+mJCQEEpKSlzPFxUVodPp/ArWAGRkFNRJhs2dN3bn9IU8\nTp7PZf0nR13tw/q24bp+bcnMzPe5f5GlmCKzfSWpIEtwpdt70y6oPX/sPYk3Dr5LdnEuO0/t48fz\nh5k7cBZtjK2qdUx/aTQKkZFGsrMLmm0mqYyBjAHU/RhUN8Drrn379gHoiZ0zS8ZdYWFhuZu5ISEh\nFBfbz3s1Dc5DzQL03oJrJrX0eC2jw7yOdcSlQtfj0FD76lpajSYgfzf1rTEFHuuDjI93MjbeBXps\n/A7YxMXFYTKZAvriTYlk2AghmqsdO3bw+uuv88Ybb7jazGYzqqry/PPPM2fOHMLC7Be2JSUlrgv0\nrl27kpKSQr9+/QB78eIuXbr4/bqqqmK1Bu59eBMcpOWp6UNY+e53nL2Uj1ajYfTgTgy/oi2qClar\n7/N+RmG263FEUESl2/sysNWVtBvUlkMZR/n05DZyTfks35/Mn6+cSetaDtqAfdpvTfrfFMgYyBiA\njIG70NBQj+A7QHFxMQaDAaDGwXmoXoC+suBaekaB63F+fhGZXmog5+cXux7n5BY5HqnVDr43BBJ8\n9U3GxzsZG+9qOjbegsB+B2xmzZrF448/zl133UWnTp1cd1Kdhg8fXuVONSVSw0YI0Vz17t2bgwcP\n8uGHHzJ+/Hi+/vprduzYwXvvvedavvuhhx4iNTWV5ORkJk2aBMD48eN57bXXGDJkCDqdjtWrVzNh\nwoR6fjcVaxkZyp8n9avwC1p6UQYnc04zoHU/NEr5bxTOJb0VFCKCW9S4L+3C2tAurA3dIuP52/dr\nyTXlseLAau7rP4N2YW1qfHwhhKiKuLg4zGYzqamptGvXDrAH4J3ToGoanIeaBei9BddM5tIaNhoU\n7wE4t2azxb6PovjYvhGRwKNvMj7eydh4F+ix8Ttgk5SUBMAzzzxT7jl/ipQ1dZJhI4RormJjY0lO\nTuaZZ55h0aJFdO7cmVdeeYVu3bqRnJzMkiVLGDJkCCEhIUyePJmpU6cCMGXKFNLT05k4cSJms5lx\n48Yxbdq0en43VWOxWVh5YA0ZxVnkmPIY2emacts4Cw6H68PQafz+2K1UfEQcc/rfw9++X0uOKY9n\n9v2VPjE96N/qCvrG9KrTosRCiOYrLCyMkSNHsmzZMpYsWcIvv/zC5s2bWbNmDdBwg/Puy3prfS7r\nXfrY+SVMyncIIeqK31eOR48erXyjZsxZLV4ybIQQzdHAgQP54IMPyrV369bNY6qUO61WS1JSkuuG\nQGP0zflvySjOAuCL019xTYehBJUJyjgzbKq7pLcvXSI6M7vfPbx2cD25pjwOZhzlYMZRtIqWvi17\nk9B2IL1jelSY+SOEEIGyePFinnrqKUaMGIHBYOCRRx5xZdQ01OC8+x1wna9lvd2iM84bs3VR8F4I\nIaAKARvhm2TYCCFE82K2Wfj05DbX7zmmXPZfOEBCu6s9tnNm2ETVQsAGoFtkPIuGPsH3aT+x/+IB\njmUdx2yz8P2ln/j+0k+EB4XROaIjncI70Cm8A3EtOhKub/zFMoUQ9Wfw4MHs3bvX9XtkZCQrVqyo\ncNuGGpw3u2XY6Hxm2JQGZ5xZObJCjhCirkjAJkCkho0QQjQv3144QHZJDgoKnVt0IiX3FFtP72BI\n24EeF/hZjoBNZEhkrfUlSKPj6jZXcnWbKym2lPBd2o98c/5bTuScJM+cz0/pR/gpvXTqcoS+BW2N\nrWkb1pq2BvvPVoZYjDqD3DkWQjQLVvcpUT4CMBpFMmyEEPVHAjYBIhk2QgjRvPyYfgiAvi17M7LT\nNfz1u1VcKEwjuySHKLfgTFYtZ9iUFaILZmi7qxna7mouFKRxNOsXTuee5XTeWS4UpKGikmPKJceU\ny9GsXzz2DdWFEBsaQ0vHH+fj1mEtiVQNddJ/IYSoCxbHlCitRvEZgPGoYWNzZNhIvEYIUUckYBMg\nrgwbVQI2QgjR1FlsFn7OOg5An5gedApvj4KCisqFgjSPgE12cd0GbNy1Mbaijdty38WWEs7mp5Ka\nf57zBRddf/LN9uVtiyzFnM47x+m8c+WOpVU0tNCH0yK4BZH6FrQIbkGEvgURweEYggwYdCGE6uw/\nDUGhBGuDpXaOEKLBck5v8jUdCjwzbJxBHpkSJYSoKxKwCRCNZNgIIUSzkZJzihKrCYBe0T3Qa/VE\nh0SRUZzJ+cKL9Iq5DLAHQIqtxUDtTonyV4gumG6R8XSLjPdozzPlc6konUuFGaQXZXCpKJP0Ivvj\nPHM+AFbVRlZJDlklOZzy47UUFIK1eoI0QQRpgwjSBKF3/tQEEaTVodMEoVU0aBQNGhw/NZrSNkWD\nVtGiQXH8rkVRFBTH8e3/2T9/nXfIFWeLopQ+57atq0VxPVPmWM4t8Li1rtFAWE4I+QXF2EpnUgRM\nXX79U6r5ahoNGHNCKKilMaipupimotfpGB5+Va2/jqh9FpszYOP7/5uKVomq7r8hIYSoKgnYBIjW\nVcOmAV7BCCGECKjDmT8D0NrQipjQKADaGlvZAzb5F13bOQsOQ/1k2PgrXB9GuD6MLhGdyz1XbCkm\n05SJWVfCuYw0sopy7FOqSvLIMeWSW5JHoaUIs83ssZ+KSrG1hGJrCZjLHVaIRutYzi9MuWxifXdD\n1JAz+OJrSW8ou0qUs+hw7fVLCCHcScAmQKTosBBCNB9HHAGb3tGXudraGttwMOMoFwpLAzbOJb0B\nIoJb1F0HAyhEF0LH4PZER4fRJTTfYylcd2armUJLMUWWIgotRRSaCymxmjDbzPY/VjMmm8XtsRmL\nzYJNtbn+WFUbquNnaZvV8Vi1/8QGqoqKPSgEoDqmI6vOFtX1jL1F9fac577ux4Ky71NBo1Ecn/PV\n+6yvrSsEtbaOXH4I0CiKffp3NV+y1vpaR3QaHVe06VXf3RABYDJbAdDrfEdflAqKDmuk6LAQoo5I\nwCZAtIpMiRJCiOagwFzIGUeNF+fUJ8BVK+Z8QRqqqqIoiivDJlwfhk7TtD9yg7RBRGiDiAgOr++u\n1AqtViE6OozMTO9Bq6ZOxsBzDETjVmK2Z8sE67U+t9NUNCVKAjZCiDoiCX0BIhk2QgjRPJxxK8jb\nJSLO9bitsTUARZYick15QGmGTVRw/devEUIIUarYZAEgOKiSgI1MiRJC1CM53QSILOsthBDNw9n8\nVABahkQTqgt1tbc2lK7GdL7APi0qu46X9BZCCOEfkzPDppKAjVLRKlGSYSOEqCMSsAkQWdZbCCGa\nh7N59oBNh/B2Hu0humCiQ+wFiJ0BmyxHwKYhrBD1/+3deXhU1f0/8PfsW0IWEjCRECYhBkiFBBJM\nCgiCglIgrYRKQdvHjVXwhxW1qBhBik8tIoKoUNGytFRav6BCKyIaKIjKXkBAyYjBEDFkn2T2+/tj\nMkOGZJJJMpn1/XoeHoabe+eeHPBz4nvOOZeIiK4xNu5h0+YMm6ZLomxcEkVEvsXAxks4w4aIKDw4\nZtj0irix2deu7WPjGthwhg0RUWBxBjZt7mHTZEmUtXFJFPMaIvIRBjZewj1siIhCn8lqxo/1PwEA\nekUmNPt6UmOIo6u+CACoatzDJpqBDRFRQDGaHDNsPH9KlMXxlCgmNkTkIwxsvIQzbIiIQt9lfRls\ngv0T1l4Ric2+3jdaCwAo1ZehvKECBqsRABDDJVFERAHFMcNG3p4lUY0zbLgkioh8hYGNlziSdkHg\nPjZERKHKsX+NRqZucdZMSlQyxCL70Hr4x+PO45xhQ0QUWDzdw0bk8pQobjpMRL7FwMZLJE0KN5dF\nERGFJsf+NUkRN7b4CatSqnQui/rqx2PO49GKbr5pIBERecQR2Cjbs4eNM7DpunYRETXFwMZLmq5l\nZWBDRBSaHJsJJ0bc4PacvjH2ZVFljefGKKIhFUu7vnFEROQxY+Njvdu3JIpPiSIi3/JpYHPmzBkU\nFBQgMzMT+fn5OH78eKvnv/POO5g/f76PWtc5EvG1ruQ+NkREoamycRNhx+O7W5IWneLy5/HaO7q0\nTURE1H5GkwWAB0uiXGbYND4lilNsiMhHfBbYGI1GzJo1C3fffTe++uor3HfffZg9ezb0en2zc+vr\n6/GnP/0JL774oq+a12kuM2y4hw0RUcixCTbnY7pjW9lEODVKCxHsY0J2z0zkJWT7pH1EROQ5xwyb\ntgKbpkuiLFYuiSIi3/JZYHPo0CGIxWJMmzYNMpkMBQUFiIuLQ1FRUbNzH3nkEVy8eBH33HOPr5rX\naZIWNiQjIqLQUWuqg1Ww73kQo3Af2KhlKtyT/ksMT7wFU9Pv5tR5IqIAZPJw0+Emk+idM2xY14nI\nV3y2qF6n0yE1NdXlmFarRXFxcbNzly9fjp49e2L16tWorKxs131EIpFLYfWUY4ZMR6c4SqXXrhMB\nkEhCp5B3tm9CGfumdewf90Ktb3bt2oXVq1ejrKwMiYmJWLBgAW6//XaUlZVhyZIlOHLkCKRSKe68\n8048+eSTkMvlEAQBQ4YMgdBkVuKQIUPwl7/8xY/fiXsVjcuhgLYf0z3ixryubg4REXWQxWpzfsCq\naGPTYVFLmw6HyNhNRIHPZ4FNfX09VCqVyzGlUgmDwdDs3J49e3b4Pt27azqVekdHazp0XZ3J5nwd\n2U2N2BhVK2cHp472TThg37SO/eNeKPSNTqfDokWLsGHDBgwePBgHDx7EjBkzsG/fPixcuBBpaWnY\nt28fampqMHfuXLz22mtYsGABLl68CAA4evRoUHxa6di/RiaWIkIW/H9vRETe9tZbb2HlypWQyWTO\nY+vXr0daWhoWLVqEQ4cOITIyEnPnzsWUKVP81k6Dyep83Z4lUVYuiSIiH/NZYKNSqZqFMwaDAWq1\n2qv3uXpV3+EZNtHRGlRV6Tv0lKe62mvfW0VlHSSCtZWzg0tn+yaUsW9ax/5xrzN9Exsb0UWt6hit\nVosDBw5Ao9HAYrGgvLwcGo0GcrkcKpUKs2fPhkKhQHx8PCZOnIiPP/4YgH0j+vT09KAIawCg0mDf\nvyZGER00bSYi8qUzZ85gwYIFePDBB12Oz58/H2q1GgcPHsS5c+fw8MMPIy0tDZmZmX5pp2M5FAAo\nZK3/j0PTcm+xckkUEfmWzwKblJQUbN682eWYTqfDhAkTvHofQRBg7URWYrMJzvS8Xfdt8tpstnXo\nPQJdR/smHLBvWsf+cS9U+kaj0aCkpATjxo2DzWZDYWEhIiIisG7dOpfzPv30U/Tr1w8A8PXXX6Ou\nrg75+fm4cuUKcnJy8PTTT3s8y7KjS2DbyzH1vdJon2ETo4oOqWWvngi1JXwdwT5gHwDsg7Z8/fXX\nmDx5sssxvV6PPXv24KOPPoJCocDAgQMxYcIEbN++3W+BjdHs+Qwbkci+jbyAJkuiGNgQkY/4LLDJ\ny8uDyWTCpk2bMHXqVOzYsQPl5eUYPny4r5rQpSQtrG8lIgonCQkJOHHiBA4fPow5c+YgOTkZeXn2\nvVwEQcCyZctQXFyMl156CQAgl8uRmZmJRx99FAqFAsuWLcO8efPw7rvvenS/zi6Bba86ax0AIKFb\nfMDNcvKVUFjC11nsA/YBwD5oSUNDA3Q6HTZu3IiFCxeiW7duePDBBzFgwABIpVIkJSU5z9Vqtdi9\ne7fH792RgL61cM1svbaVgVolbTOEF4lE9g+FG3/Gl0hEQR3cM3hsHfvHPfaNe13VNz4LbORyOdav\nX4/CwkK8/PLLSE5Oxuuvvw61Wo3FixcDAJYsWeKr5nidy2O9GdgQURiSSu1DSl5eHsaOHYtPPvkE\neXl5MBgMeOKJJ3Du3Dls2rQJ3bt3BwDMmzfP5fonn3wSubm5uHLlCnr06NHm/Tq6BLa9HMvXymp/\nAgCoRRpUVNR1/Y0DCJc3sg8A9gHg+z4IpnC4vLwcQ4YMwW9+8xu8+uqrOHnyJGbNmoX7778fSqXS\n5Vx3+1i605mAvqVw7YfKa/e+oWeUB0+KEsFmvRbYKJWyoPq7cYfBY+vYP+6xb9zzdt/4LLABgH79\n+mHr1q3NjrsLaq7/YT6Q8bHeRBSuioqK8Pbbb+Odd95xHjObzYiMjERVVRUeeughqNVq/OMf/0B0\n9LWnK61btw7Dhg1DRkYGAMBkMgEAFAqFR/ft7BLY9qpssO9hE62ICollbB0RKkv4OoN9wD4A2Act\nSUpKctn+IDs7G/n5+Th8+DCMRqPLue3dx7IjAX1r4Vr5VXvoLgJQV1MPfRthkOPLjvcxmyxBHdwz\nfG0d+8c99o17ne0bdyGwTwObUOYyw0bgP14iCh8DBgzAqVOnsH37dkyaNAn79+9HUVER3n33Xcyb\nNw9xcXFYvXq1y1NDAKC4uBj79+/Hq6++CqlUimXLlmHMmDGIiory03finsVqQY2pFoB902EiInJ1\n+vRpHDhwADNmzHAeMxqNSEhIgNlsRmlpKRITEwHY97Hs27evx+/dmYC+pXCtwWh/M7lcApsNcN2N\nsrnr96wRQRQSgR2Dx9axf9xj37jn7b7xwWTy8MAZNkQUruLj4/HGG29g48aNyM7OxqpVq/Daa6+h\nuroaX375JQ4ePIihQ4ciKysLWVlZmD59OgDgmWeeQa9evXDXXXdh1KhRkMlkWL58uZ+/m5ZVNFRB\naPyBPlbJwIaI6HpqtRpr1qzBf/7zH9hsNnz++efYuXMnpk+fjjFjxmDFihVoaGjAyZMn8eGHH2Li\nxIl+a6ux8bHebS2Fcrh+Ao6I/wdFRD7CGTZewj1sup4gCLAJ9sTSarO/FgT7cQEAmrx2HEeT14L9\nFPtx4dprQWj5eOttsf8ukYhQWW9BTU2Dx0mq0MqnOJ2ZnNXWta3dt40PljpMLBGhW60JNTUNsDGF\ndyGWiPAztWdLf4JBdnY23nvvvWbHz5075/aaiIiIgA1orldeX+F8Hc0ZNkREzWi1WrzyyitYuXIl\nnnrqKfTs2RPLly9HRkYGli5diueeew4jR46EWq3GwoULMWjQIL+11fGUKKWHgc31M2z4lCgi8hUG\nNl7CGTaAxWpDjd6EugYz6hrM0Bss0DeYoTeYYTBZYTRbYTLbYLI0/m62wmS2wmixv7ZYbfYpZI2/\nbDYBlsbfHceJQklsNwVemvNz2B8YSoGsonH/GpVUCaU0dII2IiJvGj16NEaPHt3seHR0NFatWuWH\nFrXMEdjIPQ1sxAxsiMg/GNh4SbjMsKmtN6G0XI/Scj1+rGxARY0BFbVGVNQYUK03dWqGCFG4iY9W\nQywWQbC1fS75V51JDwDQSD3fJJOIiAKTc0mU3LO1TVwSRUT+wsDGS8Qi+2fkAkJnho3RbMV3l2tQ\nfLkG31/R4+x3FajWmzy+XiGXIEIphUYlg1IuhUImgVwmhlwqgUImhlwmgUxq/10uFUMmFUMsFkHS\n+EssFkEqdj3mOC5pPC4S2QdREUTOwVQkajxu/wPE7o4DQJNrHY+LFIvsX/fksxOJRISYGA0qK/Ut\n/r239R4dfUSl63t4cI43zuhAU136h0uiXEgkIiTeEGXvm65ak0ZeozfVAwDUMpWfW0JERJ3lmGHj\n6R42XBJFRP7CwMaLxGKRcylPsLpSWY+TF67iZPFVnPu+CmZLyx/9R0XIkRCrRvduSsR0UyK2mwKx\nkUrERioQqZZBo5JBKgn9jx8kEhGiIhSwmswMJFogkYigVspgUEjZP9eRSEReCezIN5yBDWfYEBEF\nPVM7A5tmM2w4fhORjzCw8SJJY2ATbDNsqvUmfHHmR3x+qgwXf6xt9vUeMSoM0HZHrzg1knpEIDFO\nA41S1sI7ERGFpjqzPbBRcYYNEVHQMzgCG3lH97DxepOIiFrEwMaLHMXcFgQbuQiCgDMXK7HnqxL8\nr7jCpc0apRQZ2lgMTO2ODG13++yZ2AhUVNRxlgQRhaVrM2wY2BARBbt2P9YbXBJFRP7BwMaLHE+K\nstoCdwdRs8WGQ2fK8PFXJbj0k955XCGXIPumeOT97Aak946GRBz6y5mIiDzFwIaIKHS0d0nU9T8W\nizjFhoh8hIGNFzkCm0Dcw8ZiteHA/y7jg4PfoaLG6DyuTYjE7UOSMDg93uNBi4go3NRx02EiopBh\nNNs/XPV8DxsuiSIi/2Bg40Vi5wybwAlsbIKAQ6fLsOO/OvxUZQBg3zhtyE3xGJvTG6k3duPGaURE\nbeAMGyKi0GEwtXMPGz4lioj8hIGNFwXaDBvd5Rps3n0euss1AOwPbs7p3wP5w7VI6K7xb+OIiIKI\nY9NhtYxPiSIiCnZ8ShQRBQsGNl4kDpDAprbehH8VFWP/iVI4WpKVFodfjUhBrx4Rfm0bEVGwsdqs\nMFrsS0k5w4aIKPgZGwMbucyzPRubPSWKa6KIyEcY2HiRuHFHMn8tibLZBHx2/Af8375i6A0WAEBC\ndzWm3XETMvrE+qVNRETBTm9pcL5mYENEFPyM7d10+LoZNZxgQ0S+wsDGi/y5JOqbS1XYsvs8vr9S\nB8C+Jjd/mBa3Z/eCVMInPhERdVR943IogJsOExEFO0EQnI/1Vnq4h831AQ33sCEiX2Fg40WO4m0V\nfBfYVNUZse3TC/j8dJnzWF5GT0y5rS+iIxQ+awcRUaiqN3OGDRFRqDBbbM4tAzo6w4ZLoojIVxjY\neJEvZ9hYrDZ8cuQSdvxX59zpvld8BO4dexNuSoru8vsTEYWL+sYlUSKIoJQq/dwaIiLqDMdyKACQ\nd/Cx3pxgQ0S+wsDGi3z1WO+TF67iH3u/weWrjU8tUUjxq1tTMCorERIxlz8REXmTY4aNSqqEWMQa\nS0QUzJoGNh7PsLmu9HNJFBH5CgMbL+rqGTY/lOvxj73f4FRxBQD7Y7pHDErA3SNT0U0t75J7EhGF\nu3qL45HeXA5FRBTs9A0W52vP97BxDWg8DXqIiDqLgY0XddUMm4oaAz44+B32n7gMW+P+OH17ReE3\nY9KgTejm1XsREZErxwwb7l9DRBT8viurAQBolFJER3q23+P1M2qSb4j0eruIiFrCwMaLHDNsLFab\nV96vtt6EXYcu4pMjPzjfs3s3Jabcloqcfj2apf1EROR9zsCGM2yIiILehVJ7YKNN7Obx0qamewyr\nFVL0iOF4QES+wcDGi+Kjlfj6InDhh5pOvU/JlTrsPXoJn58ug8lsD2oiVDKMz03G6ME3erxBGhER\ndZ5j02G1VO3nlhARUWcVNwY2qYlRHl/T9ENSbUIk97AhIp9hYONFg/rGYd+Jy7jwQzWq9SZEaTzf\nV8ZgsuDo+Z+w78RlnC+pch5XyCUYl5OEcUN7Q6XgXxcRka/pzdzDhogoFNQbzCgt1wMAUhM931ag\n6QwbbTuuIyLqLD7uwosG9ImFXCqGAODkt+Vtnt9gtODkhXKs/+AM/t/q/+IvH37tDGviopT49W19\n8ec5P8cvR6QwrCGigLZr1y7cddddyMrKwi9+8Qvs2bMHAFBdXY25c+diyJAhGDVqFLZt2+a8RhAE\nrFixArm5ucjJycELL7wAq9Xq7hZ+45hho5Fxhg0RUUedOXMGBQUFyMzMRH5+Po4fP+7zNugu1zpf\ntyd4sViv7U+ZkuD5zBwios5iCuBFCpkEA/rE4vi35Tj2TTlGDEp0fk0QBNQ2mPHtpWqcL6nC+ZIq\nXPyxFkKT/YklYhF+po3FyKwbMTClu3MTYyKiQKbT6bBo0SJs2LABgwcPxsGDBzFjxgzs27cPhYWF\nUKvVOHjwIM6dO4eHH34YaWlpyMzMxJYtW/DZZ5/h/fffh0gkwsyZM7FhwwY8/PDD/v6WXHDTYSKi\nzjEajZg1axZmzZqFKVOmYMeOHZg9ezb27NkDjUbjs3ZcKK0GACR0V0OjlHl8XbBiTFwAABC9SURB\nVMmVOudrbQI3HCYi3/FpYHPmzBksXrwY3377LZKTk/H8888jMzOz2XkffvghVq5ciatXr+KWW27B\nsmXLEBcX58umdlhWWhyOf1uO099V4NV/nsTVGgPqGsyorTe73YxYmxCJvIwbMHRATz6em4iCjlar\nxYEDB6DRaGCxWFBeXg6NRgO5XI49e/bgo48+gkKhwMCBAzFhwgRs374dmZmZ2LFjB373u9+hR48e\nAICZM2di1apVgRfYWLjpMBFRZxw6dAhisRjTpk0DABQUFOCvf/0rioqKMH78+C6559ffVaD2XDn0\neqPzKavHvrHPgE9p51NW643XHgUeFeHZk6WIiLzBZ4GNp8n62bNn8dxzz2HDhg1IT0/H0qVL8Yc/\n/AHr16/3VVM7ZVDfOIgAmC02HG9hWZRYJELyDRG4KSkaNyVFI61XNCJUnif8RESBSKPRoKSkBOPG\njYPNZkNhYSG+//57SKVSJCUlOc/TarXYvXs3AKC4uBh9+/Z1+ZpOp4MgCB49BU8kEkHsg4W9JqsJ\nABAhV0MiCc+Zj44Zn+E885N9wD4A2AcdpdPpkJqa6nJMq9WiuLjYo+vbW+9LrtThxS3H3H49tVdU\nh+t5KIwD/HfcOvaPe+wb97qqb3wW2HiarH/wwQcYM2YMBg0aBAB4/PHHkZeXh/Ly8qCYZdNNI8ek\n4VocO/8TboyPQGKcGpFqOSJUMkSqZegVH8H9aIgoJCUkJODEiRM4fPgw5syZgwcffBBKpdLlHKVS\nCYPBAABoaGhw+bpKpYLNZoPJZIJC0fYnmN27azwKdjrrF+lj8PVP5/Hz1MFQyZRtXxDCoqN9t3Qh\nULEP2AcA+6C96uvroVK5zlJsOh60pb31XqVRYlBanHOD4aZ6xKhx57AURLZjVnvhw7n420dn8eCk\nnyE2NsLj6wId/x23jv3jHvvGPW/3jc+SA0+T9eLiYmRlZTn/HBMTg6ioKOh0Oo8Cm45+4urNROzu\nkSm4e2RKp98nUDBJdY990zr2j3uh2DdSqX1IycvLw9ixY3Hq1CkYjUaXcwwGA9Rq++a9SqXS5esN\nDQ2QSqUehTUAcPWq3iczbG6/cSQKMsajqkqPBltd2xeEILFYhOhoDaqq9LDZhLYvCEHsA/YB4Ps+\nCJVwQKVSNQtnmo4HbelIvX98aqbbvyuzwYQKg8nj99L20ODp+4YAACoqgn8c4H/LrWP/uMe+ca+z\nfeOu3vsssPE0Wb/+E1fAXuQbGho8uk9nP3FlWuge+8Y99k3r2D/uhULfFBUV4e2338Y777zjPGY2\nm9G7d2/s27cPpaWlSEy0b8Ku0+mcy6BSU1Oh0+mcMyp1Oh1SUjwPuwVBgC8fKmWzCbBaw/uHE/YB\n+wBgHwDsg/ZKSUnB5s2bXY7pdDpMmDDBo+s7U+/5d+Ue+6Z17B/32DfuebtvfBbYeJqsuwtxujKB\nB5gWtoZ94x77pnXsH/c60zeB9onrgAEDcOrUKWzfvh2TJk3C/v37UVRUhHfffReXL1/GihUr8MIL\nL+Cbb77Bhx9+iHXr1gEAJk2ahLfeegu5ubmQSqV48803kZ+f7+fvhoiIvC0vLw8mkwmbNm3C1KlT\nsWPHDpSXl2P48OH+bhoRUUDzWWDjabLu+MTVoaKiAtXV1c2WU7nT2U9cmRa6x75xj33TOvaPe6HQ\nN/Hx8XjjjTfwxz/+EUuWLEGfPn3w2muvITU1FUuXLsVzzz2HkSNHQq1WY+HChc4ZNdOmTUN5eTkK\nCgpgNpsxceJE3H///X7+boiIyNvkcjnWr1+PwsJCvPzyy0hOTsbrr7/u8QeyREThymeBjafJ+oQJ\nE3Dvvfdi8uTJuPnmm/Hyyy/j1ltvRUxMjK+aSkRE7ZSdnY333nuv2fHo6GisWrWqxWskEgkWLFiA\nBQsWdHXziIjIz/r164etW7f6uxlEREHFB9s12jmS9Z07d2Lo0KHYvHmzM1lfvHgxFi9eDADo378/\nli5diqeffhp5eXm4cuUKli9f7qtmEhERERERERH5nU+fL+0uWV+yZInLn8ePH+/yqG8iIiIiIiIi\nonDisxk2RERERERERETkGZEgCMG92yURERERERERUYjhDBsiIiIiIiIiogDDwIaIiIiIiIiIKMAw\nsCEiIiIiIiIiCjAMbIiIiIiIiIiIAgwDGyIiIiIiIiKiAMPAhoiIiIiIiIgowDCwISIiIiIiIiIK\nMAxsiIiIiIiIiIgCDAMbIiIiIiIiIqIAE/aBzZkzZ1BQUIDMzEzk5+fj+PHj/m5SQDl8+DCmTJmC\nIUOG4Pbbb8fWrVv93aSAU15ejry8PHz66af+bkrAKCsrw8yZMzF48GDceuut2Lhxo7+bFFCOHj2K\nu+++G4MHD8a4cePwwQcf+LtJ5AbHCI4DDuFe61nXWbuDCWu3e6zpbQv3eu8OxwH3unR8EMKYwWAQ\nRowYIWzZskUwmUzCtm3bhNzcXKGurs7fTQsIVVVVQk5OjvD+++8LVqtVOHXqlJCTkyMcOHDA300L\nKDNmzBD69esn7N27199NCQg2m0341a9+Jbz44ouCyWQSzp8/L+Tk5AhHjhzxd9MCgsViEXJzc4V/\n//vfgiAIwldffSUMGDBAKCkp8XPL6HocIzgONBXOtZ51nbU7mLB2u8ea7plwrvfucBxwr6vHh7Ce\nYXPo0CGIxWJMmzYNMpkMBQUFiIuLQ1FRkb+bFhBKS0sxcuRITJw4EWKxGBkZGbjllltw9OhRfzct\nYPz973+HSqVCQkKCv5sSME6cOIErV67g8ccfh0wmQ1paGrZu3QqtVuvvpgWEmpoaVFRUwGq1QhAE\niEQiyGQySCQSfzeNrsMxguOAQ7jXetZ11u5gwtrtHmt628K93rvDccC9rh4fwjqw0el0SE1NdTmm\n1WpRXFzspxYFlv79++Oll15y/rm6uhqHDx9Gv379/NiqwKHT6fD222+jsLDQ300JKKdPn0ZaWhpe\neuklDBs2DOPGjcOJEycQExPj76YFhJiYGEybNg2PPfYYMjIyMH36dDz77LP8wSAAcYzgOACw1gOs\n6wBrdzBh7XaPNb11rPfucRxwr6vHB6lX3iVI1dfXQ6VSuRxTKpUwGAx+alHgqq2txaxZs5CRkYHR\no0f7uzl+Z7FY8MQTT+Dpp59GdHS0v5sTUKqrq/HFF18gNzcXn376KU6dOoWHHnoISUlJyM7O9nfz\n/M5ms0GpVGLVqlUYPXo0Dh48iN///vfIyMjgD0wBhmOEq3AcB1jr7VjXWbuDCWu3Z8KxpreG9b51\nHAfc6+rxIaxn2KhUqmbF22AwQK1W+6lFgamkpARTp05FVFQU1qxZA7E4rP/ZAADWrl2L/v37Y+TI\nkf5uSsCRy+WIiorCzJkzIZfLnZtvffLJJ/5uWkDYvXs3Tp48iTvvvBNyuRyjRo3CqFGjsH37dn83\nja7DMeKacB0HWOvtWNdZu4MJa3fbwrWmt4b1vnUcB9zr6vEhrP/rTElJgU6nczmm0+nQt29fP7Uo\n8Jw+fRq//vWvMXz4cKxduxZKpdLfTQoIu3btws6dO5GdnY3s7GyUlpbisccew7p16/zdNL/TarWw\nWq2wWq3OY441nQRcvnwZJpPJ5ZhUKuU+CAGIY4RdOI8DrPV2rOus3cGEtbt14VzTW8N63zqOA+51\n+fjgla2Lg5TRaBSGDx8ubNy40WUXeb1e7++mBYSffvpJyM3NFd58801/NyXg3XbbbdxJvlFDQ4Mw\nYsQI4ZVXXhHMZrNw5MgRITMzUzh27Ji/mxYQzp49K2RkZAj//Oc/BZvNJnzxxRdCVlaWcPLkSX83\nja7DMYLjwPXCtdazrrN2BxPWbvdY0z0XrvXeHY4D7nX1+CAShPCOxc6ePYvCwkKcO3cOycnJKCws\nRGZmpr+bFRDeeOMNrFy5stkU0t/+9rdYsGCBn1oVmEaPHo1nn30Wt912m7+bEhAuXryIJUuW4H//\n+x8iIiIwd+5cTJ482d/NChh79+7FqlWrUFJSgsTERDz66KO44447/N0sakG4jxEcB1yFc61nXWft\nDibhXrvdYU33XDjXe3c4DrjXleND2Ac2RERERERERESBJqz3sCEiIiIiIiIiCkQMbIiIiIiIiIiI\nAgwDGyIiIiIiIiKiAMPAhoiIiIiIiIgowDCwISIiIiIiIiIKMAxsiIiIiIiIiIgCDAMbCmpPPfUU\n0tPT3f5avXo1vvjiC6Snp8NoNHZ5ey5duuS89/z58zt8/YULF9o89/HHH3fey5PziYiCFWs9az0R\nhQfWe9Z7ciUSBEHwdyOIOqq2thYGgwEAcOzYMcybNw979+6FXC4HAKjVashkMlRXVyM+Pr7L23Pp\n0iWMGTMGW7ZswU033YRu3bq163qr1YqKigrExsZCIpG0em5tbS10Oh2mTJmCXbt2ITU1tTNNJyIK\nWKz1rPVEFB5Y71nvyZXU3w0g6ozIyEhERkYCAKKiogAAcXFxUCgULuf5oqA3FRMT0+6CDgASicTj\ntkZGRiI2Nrbd9yAiCjas9az1RBQeWO9Z78kVl0RRyLt+2mR6ejp27dqFiRMnYuDAgXjggQdw+fJl\nLFy4EJmZmRg7diwOHTrkvP7HH3/E/PnzkZWVhREjRqCwsBB6vd6jezumQX722We44447MGjQICxY\nsAClpaWYMWMGBg0ahPz8fJw9e9blfMc0yNGjR2PTpk249957cfPNN2PcuHEoKirycg8REQU/1noi\novDAek/hhIENhaUVK1Zg8eLF2Lx5M06fPo1Jkyahf//++Ne//oW+ffvi+eefBwAIgoBHHnkEMpkM\n27Ztw5o1a3D27FksWrSoXfd79dVX8corr2Dt2rX4+OOPMXnyZIwfPx7btm2DUqnEn//851avnTZt\nGnbu3In09HQsWrQIZrO5U98/EVE4YK0nIgoPrPcUqrgkisLSfffdh5ycHABAbm4uSkpK8MADDwAA\n7rnnHsyaNQtWqxVffvkldDod/va3v0EmkwEAli9fjjvvvBNlZWW44YYbPLrf7NmzkZGRAQDo168f\nEhMT8ctf/hIAkJ+fj/Xr17u9duLEiRg/fjwAYM6cOcjPz0dZWRmSkpI69s0TEYUJ1noiovDAek+h\nioENhaXevXs7X6tUKpcCqVQqYbPZYLFYcOHCBdTV1WHo0KHN3kOn03lc1K+/X69evVzuZzKZ3F7b\np08f5+uIiAgAYApPROQB1noiovDAek+hioENhSWp1PWfvljc8upAi8WC3r17t5iSt2ezM0/v1xJH\n+t8UH+5GRNQ21noiovDAek+hinvYELUiNTUVZWVliIyMRHJyMpKTk2GxWPDiiy+irq7O380jIiIv\nYK0nIgoPrPcUbBjYELVi2LBhSE1NxWOPPYbTp0/j1KlTWLhwISorK9GjRw9/N4+IiLyAtZ6IKDyw\n3lOwYWBD1AqxWIy1a9ciIiIC9957Lx544AH06dMHa9as8XfTiIjIS1jriYjCA+s9BRuRwAVzRF5z\n6dIljBkzBrt27UJqamrI3Y+IiFjriYjCBes9+Rtn2BB1gcrKStTU1HTpPWpra1FRUdGl9yAiIvdY\n64mIwgPrPfkLAxuiLjB9+nQ888wzXXqP559/HlOmTOnSexARkXus9URE4YH1nvyFS6KIiIiIiIiI\niAIMZ9gQEREREREREQUYBjZERERERERERAGGgQ0RERERERERUYBhYENEREREREREFGAY2BARERER\nERERBZj/D73wfNvGHj6iAAAAAElFTkSuQmCC\n",
"text/plain": [
""
]
},
"metadata": {},
"output_type": "display_data"
}
],
"source": [
"from ipywidgets import interact\n",
"\n",
"IC = [C0,T0,Tcf]\n",
"\n",
"def sim(Tsetpoint,kp,ki,kd):\n",
" global Tsp, qc\n",
" Tsp = Tsetpoint\n",
" \n",
" # control parameters\n",
" beta = 0\n",
" gamma = 0\n",
"\n",
" # create python list to log results\n",
" log = []\n",
"\n",
" # start simulation\n",
" c,T,Tc = IC\n",
" qc = 150\n",
"\n",
" eP_ = beta*Tsp - T\n",
" eD_ = gamma*Tsp - T\n",
" eD__ = eD_\n",
"\n",
" for t in np.linspace(ti,tf,int((tf-ti)/dt)+1):\n",
" # PID control calculations\n",
" eP = beta*Tsp - T\n",
" eI = Tsp - T\n",
" eD = gamma*Tsp - T\n",
" qc -= kp*(eP - eP_) + ki*dt*eI + kd*(eD - 2*eD_ + eD__)/dt\n",
" qc = sat(qc)\n",
"\n",
" # log data and update state\n",
" log.append([t,c,T,Tc,qc])\n",
" c,T,Tc = odeint(deriv,[c,T,Tc],[t,t+dt])[-1] # start at t, find state at t + dt\n",
" \n",
" # save data for PID calculations\n",
" eD__ = eD_\n",
" eD_ = eD\n",
" eP_ = eP\n",
"\n",
" qplot(log)\n",
" \n",
"interact(sim,Tsetpoint = (360,420),kp = (0,80), ki=(0,160), kd=(0,10));"
]
},
{
"cell_type": "markdown",
"metadata": {},
"source": [
"## Simulation 5. Using the PIDsim Controller Class\n",
"\n",
"From this point on, this notebook describes the development of a PID control class that could be used in more complex simulations. Regard everything after this point as 'try at your own risk'!\n",
"\n",
"Examples of PID Codes in Python:\n",
"\n",
"* [DISCRETE PID CONTROLLER (PYTHON RECIPE)](http://code.activestate.com/recipes/577231-discrete-pid-controller/)\n",
"* [ivPID](https://github.com/ivmech/ivPID)\n",
"* [python-pid_controller](https://gist.github.com/chaosmail/8372717)\n",
"* [PID Controller](https://github.com/chrisspen/pid_controller)\n",
"* [pypid 0.4](https://pypi.python.org/pypi/pypid/)\n",
"* [PIDController](http://cgkit.sourceforge.net/doc2/pidcontroller.html)\n",
"* [PID.py](http://examples.oreilly.com/9780596809577/CH09/PID.py)\n",
"* [APMonitor.com](http://apmonitor.com/do/index.php/Main/NonlinearControl)\n",
"\n",
"Below we demonstrate the use of `PIDsim`, a python module that can be used to implement multiple PID controllers in a single simulation. "
]
},
{
"cell_type": "code",
"execution_count": 8,
"metadata": {},
"outputs": [
{
"data": {
"image/png": "iVBORw0KGgoAAAANSUhEUgAAAtgAAAHvCAYAAABuRKiNAAAABHNCSVQICAgIfAhkiAAAAAlwSFlz\nAAALEgAACxIB0t1+/AAAIABJREFUeJzs3Xl4VFW29/HvqarMCZkI80wYZR4EBERRQGVyalq9otKo\nqG3bgEO3zVVbRW3f2+gFlAYRnJrr0E6gIiiKqAgqMggoCiQgYyAJmSup8f2jkiKBEEKoOpWE3+d5\n+iGpOnXOyrL6ZGXX2nsbXq/Xi4iIiIiIBIQl1AGIiIiIiNQnKrBFRERERAJIBbaIiIiISACpwBYR\nERERCSAV2CIiIiIiAaQCW0REREQkgFRgi8g5aeLEiXTq1Mn/v86dO9OnTx+uu+46vvzyy1CHF3AP\nPPAA/fr1w+FwVPr8+vXr6dSpExs2bKjxNZ599lkuvPDCah//zTff0KlTJ/bu3XvKY+677z6uv/76\nGsckIhIKKrBF5Jx1wQUX8Oabb/Lmm2/y+uuvM3v2bBo0aMAdd9zB9u3bQx1eQI0fP578/Hy++eab\nSp//6KOPaNGiBX379q3xNa6//nrmz59f49eLiNQXKrBF5JyVkJBAr1696NWrF71792bo0KHMmTOH\n2NhY3nzzzVCHF1CDBg2iUaNGfPzxxyc953Q6+eSTTxg3bhyGYdT4Gk2aNKFr165nE6aISL2gAltE\npJzIyEjatGnDwYMHAfjrX//Kn//8Z/70pz/Rs2dPHn74YQD27t3L3XffzYABA+jXrx/Tpk0jIyOj\nwrnWrl3L73//e3r27Mnw4cN54YUXKjy/evVqrrzySrp3784ll1zCkiVLKjy/adMmrrvuOnr37s3A\ngQP5y1/+wrFjxyq8/qqrrqJHjx4MGTKEmTNnUlxcXOnPZbFYGDNmDJ999tlJbSJff/01OTk5jB8/\n3v/YqlWruO666+jVqxc9e/bkuuuuY9OmTf7nr7/+eh5//HFuvPFGevTowcKFC09qEcnJyeGRRx7h\nwgsv5LzzzmPo0KE8/fTTuFyuCtffsGEDY8aMoUePHtx4443s2LGj8v84gMPh4Omnn2bIkCH06NGD\niRMn8uuvv57yeBGRUFCBLSJSjsvl4sCBAzRv3tz/2KpVq4iNjWXevHlcffXVHDx4kAkTJnDkyBGe\neOIJHn30UbZv386NN95IQUEB4CuOb7vtNpo1a8bcuXO55ZZbmDNnDq+++ioAX375JXfddRddu3Zl\n3rx5XHXVVTzxxBP+Ijs/P5877riD5s2bM2/ePB566CHWrl3Lo48+CvgK/HvuuYfzzz+fhQsX8uc/\n/5l33nmHuXPnnvJnO1WbyIcffkivXr1o06YN4Ct47777bvr06cOCBQt46qmnyMvL47777sPj8fhf\n98Ybb9C3b1/mzp3L8OHDT7retGnT+Pbbb3nwwQd58cUXGT9+PIsXL2bp0qUVjps5cybXXHMNs2fP\nxul0cvPNN5OdnV3pz3D//ffz9ttvc9dddzF79mxsNhsTJ0486Y8bEZFQsoU6ABGRUPF6vf7RVI/H\nw+HDh5k/fz5ZWVlce+21FY595JFHiIyMBODJJ58EYPHixcTGxgLQo0cPLrvsMt5++21uueUWXnzx\nRTp16sSzzz4LwIUXXkhGRgabNm3ipptuYs6cOVxwwQX+cw0dOhSXy8XcuXOZMGECu3fvJicnhxtv\nvJHevXsDEBsbS1paGgDbtm3D4XAwefJkGjVqxIABAwgPDz9pdLi8zp0707FjRz7++GMuuugiAOx2\nO59//jn33Xef/7i0tDSuvPJKHnjgAf9jhmEwdepUMjIyaNq0KQCNGzdm2rRplV6rqKgIr9fLzJkz\n6devH+BrU1m9ejU//PAD11xzjf/Yu+++m0mTJgHQs2dPLr74Yt59911uvfXWCufcsWMHK1asYPbs\n2Vx22WUADBkyhJEjR/Lyyy/zl7/85ZQ/u4iImVRgi8g56+OPPz6pJzkpKYnHHnuM7t27+x9r0qSJ\nv7gG+OGHHxgyZIi/uAZo2bIl3bt3Z+PGjdxyyy1s2rSJCRMmVDj3/fffD/iKz23btvHggw9WKIiH\nDBnCv/71L3bu3ElqaioJCQnceeedjBkzhuHDhzN48GCGDRsG+Ar68PBwJkyY4H9+7NixWCxVfzA5\nbtw4FixYgMPhIDw8nNWrV+N0Orniiiv8x0yYMIEJEyZQUFDA7t27SU9P59NPPwV8/dplyka8KxMd\nHc3LL7+M1+vlt99+Iz09nR07dpCdnV3hHAAjRozwf52UlESPHj3YuHHjSef8/vvv/Xkqy5thGAwc\nOJBvv/22yp9bRMRMKrBF5Jw1ZMgQpk6dCvh6lBs0aECLFi1OmuiXnJxc4fu8vLyTHgNfcVjWIpKb\nm0tSUlKl183Ly8Pr9fLkk0/6R7DLO3r0KF27duW1117jueee4+233+a1114jOTmZe++9l2uuuYaW\nLVvy0ksvMX/+fF5++WUWLlxI8+bNeeSRR/xFeGXGjRvHM888wzfffMNFF13Ehx9+yLBhw0hMTPQf\nU1BQwIwZM/jkk0+wWq106NCBZs2aAb5R//I/b1VWrlzJU089xaFDh0hJSaFXr15ERkZWOEdl50lM\nTKzQa14mJycHoNKVTlJSUqqMRUTETCqwReSc1aBBgwoj1WfyuqysrJMez8rK8heisbGxJxWJhw4d\nYt++ff6VNqZPn84FF1xw0nlat24NQMeOHZkzZw4Oh4N169axaNEi/vu//5vBgwfTpEkT+vXrx4sv\nvkhhYSFff/018+fPZ9q0aaxfv57w8PBKY2/cuDEDBgxgxYoV9O3bl6+++opZs2ZVOOaxxx5jy5Yt\nvPbaa/Ts2ZOwsDA+//xzVq1aVe0c7d69m3vvvZeJEycyefJkGjZsCMDVV1990rH5+fkVPg3Izs6u\n9A+YuLg4oqKieO211056LiwsrNqxiYgEmyY5ioicoT59+vD111/7R6sB9u3bx7Zt2+jVqxcAvXr1\nYs2aNRVe9+qrrzJjxgxiY2Pp2LEjBw4coHv37v7/ZWdnM3fuXEpKSli3bh2DBg0iOzub8PBwhg0b\nxr333ovH4+HIkSO8//77XHLJJTidTmJiYhg1ahS33XYbhYWFFeKqzLhx41i9ejWrV68mKirK349d\nZvPmzVx66aX069fPX7iWTYwsP8mxKtu3b8fpdDJlyhR/cX3kyBF27tx50gj22rVr/V9nZGSwZcsW\nf992eX369MFutwNUyNt7773HypUrqxWXiIgZNIItInKGbrnlFt577z3+8Ic/cPvtt+NwOJg9ezaN\nGzf2T9677bbbuOmmm7j//vsZP348u3bt4t///jczZswAfBP7pk2bRkxMDBdeeCH79+/nn//8J+ed\ndx4pKSlERkZiGAb33HMPt912GwAvvPACLVq0oEuXLiQmJpKZmcn06dO57rrrsNvt/Otf/6JPnz6n\nbd0YOXIkjz76KM899xyXX375SaPd3bp1Y/ny5fTq1Yvk5GQ+++wz/+omZQXu6XTt2hWLxcI//vEP\nrrzySv8EUqfTSVFRUYVjZ8+ejcViISEhgblz55KSksJVV1110jl79OjB0KFD+dOf/sTdd99N8+bN\nWbFiBW+88YZ/MqmISG2gEWwRkTPUvHlzlixZQlxcHPfddx+PPvoo3bp14/XXX/e3OvTr14/58+ez\ne/du7rzzTpYsWcJf//pXrrvuOgBGjRrl74W+7bbbmDNnDqNHj2bOnDmArx1i4cKF2Gw27r33XqZO\nnUpMTAyLFi0iLCyMli1b8q9//YuMjAzuvvtuHnzwQTp06OB/fVViY2O59NJL2bt3b4W1r8s8+OCD\n9OzZk0ceeYSpU6eyc+dOXnnlFcLDw9m8eXO1cpSamsoTTzzBhg0buP3223n++ecZNWoUt99+O1u3\nbq0wEv7www+zYMECpk6dSnJyMq+88kqFlpHy5syZw6WXXsqzzz7LlClT2Lx5M7NmzaowSVNEJNQM\n74mf1YmIiIiISI1pBFtEREREJIBUYIuIiIiIBJAKbBERERGRAFKBLSIiIiISQCqwRUREREQCqNau\ng330aH5IrmsYBsnJMWRlFZ60GYIElnJtLuXbPMq1eZRrcynf5lGuzVPdXKekxFX7nBrBPoHF4ku0\nRZkJOuXaXMq3eZRr8yjX5lK+zaNcmycYudZ/NhERERGRAFKBLSIiIiISQCqwRUREREQCSAW2iIiI\niEgAqcA2kdPl5qsfD3LkWFGoQxERERGRIKm1y/TVR6t+2M9/Vu8GYPFfh4c4GhEREREJBo1gm2jd\ntgz/126PJ4SRiIiIiEiwqMA2UZPkaP/Xh7PtIYxERERERIJFBbaJkuIi/F/vP1IQwkhEREREJFhU\nYJvI4zm+/eb+oyqwRUREROojFdgmcrmP913v0wi2iIiISI0NGdKPSy4ZzIgRQxkx4kJGjhzG9Ol3\nk5a2C7vdzqhRw/jii89Oet2BA/sZNmwABw8eCFpsKrBN5CxXYGsEW0REROTsLFz4Cp9++hWffvol\ny5d/Rrt2qdx3358JDw9nxIjLWLHio5Ne89FHy+jb93yaNWsetLi0TJ+J3O7jLSLZeSUUFjuJiQwL\nYUQiIiIiVXO5PWTnFZtyraQGkdisNRv/tdlsXHHFWN5449/k5+czbtxV3H77LRw7dozExEQAPB4P\nK1Z8xNSp9wcy7JNjCerZpYLyI9jgm+jYqVViiKIRERERqZrL7eFvL6wnM9ecArthfCRP3j6wRkV2\nXl4eb7/9Bu3atSchIYGEhARSUzvy6acrmDDhegC+/XYdXq+XwYOHBjr0CqodfWZmJoMGDWL16tUV\nHvd4PEycOJGnn37a/5jX62XWrFkMHDiQ/v37M3PmTNxud+CirqNcrooFtvqwRURERGrujjsmc9ll\nF3HZZRfxX/91LVlZmcyc+f/8z48ff3WFNpGPPlrGmDHjsVqtQY2r2iPYM2bMICcn56THFy9ezIYN\nG+jWrZv/sSVLlvDFF1+wbNkyDMNgypQpLF68mNtuuy0wUddRrhNGsPcezg9RJCIiIiKnZ7NaePL2\ngbW2RWT+/EW0a5d6yucvvXQUc+c+S1raLpKTG7J+/Vr+/Od7AxFqlapVYL/++utERUXRtGnTCo/v\n2LGDd999lxEjRlR4fOnSpdx88800atQIgClTpjB79uxzvsB2lvZgGwZ4vbBldxZujwerRXNNRURE\npHayWS00Sow+/YG1UFRUFCNGjGLFio9ISWlM//4DSElpFPTrnrbATk9P56WXXuKtt97i6quv9j/u\ncDj4y1/+wuOPP85//vOfCq9JS0sjNfX4XxNt27YlPT0dr9eLYRjVCswwDEJRd1osRoV/A6lse/RB\n3ZrwzdbDFNid7Nyfy3ltkwJ+rbogmLmWkynf5lGuzaNcm0v5No9yXT0Wi4HVWnWOrrrqah588AGS\nkpKYPHnKSccHI9dVFtgul4sHHniAGTNmkJCQUOG5WbNmMWTIEPr27XtSgW2324mMjPR/HxUVhcfj\nweFwEBERQXUkJ8dUuxgPhoSEmICf04vv50ltmUjGMTu79+eyJS2boX1bBfxadUkwci2npnybR7k2\nj3JtLuXbPMp11eLjo0lKiq3ymEGD+pOcnEROTg5XXDHilPVlIHNdZYE9b948unTpwrBhwyo8vm7d\nOtavX39SYV0mMjKSkpIS//d2ux2bzVbt4hogK6swZCPYCQkx5OQUVth5MRCKS1wAOB0u+nRoyO79\nuXzz40F+f3G7c7JNJJi5lpMp3+ZRrs2jXJtL+TaPcn1669b9AEB29ukXjVi48BUAjh0rPOm56ub6\ndIV8eVUW2MuXL+fo0aMsX74cgIKCAqZPn47T6SQsLIwLLrgAgOLiYgzDIC0tjQULFtC+fXvS09Pp\n2bMn4GszadeuXbWDAt9KJKFceMTj8VZYtzoQylYRsRgGfTum8J/Vu8kvcrI9/RjntTk320QgOLmW\nU1O+zaNcm0e5NpfybR7l2jyBzHWVBfaKFSsqfD98+HAeeughLr744gqP//WvfyUxMZG//OUvAIwb\nN45FixYxcOBAbDYbCxYsYPz48QEJuC5zlfZg26wGjRKjad04jr0Z+azfdvicLrBFRERE6pOgbDRz\nww03kJmZybXXXovT6WTs2LFMmjQpGJeqU8pGsMuWnxncvQl7M/L5/pcj3DCiI1ER2vdHREREpK47\no4ru888/r/Txf/zjHxW+t1qtTJs2jWnTptU8snqobJm+MJuvwB54XhPeWr0Lh9PDdz9nMKxX81CG\nJyIiIiIBcO7NrAuhso1mykawY6PC6N0hBYCvfzwUsrhEREREJHBUYJvoeIF9fHmYoT19m/fsPpjH\nwcyTZ7aKiIiISN2iAtskbo8Hb+nE1PJbgHZtnUR8bDgAm3dlhiI0EREREQkgFdgmcbmOL/tSvsC2\nWAy6t00GYFtalulxiYiIiEhgqcA2ibO0PQSOT3Is062db4m+nftzsZduRiMiIiIidZPWhTOJq1yB\nbT1hr/uubZIwDHB7vPy89xh9OqaYHZ6IiIhInfPbb3uZN282mzdvwu1206xZc373u98zZsyVLF/+\nAf/4x+PldhI3iI6OZvjwEfzxj3/GZgteGawC2ySuKkawY6PCaN8snl0HctmWlqUCW0REROQ0PB4P\n9957D6NHj+XRR58kLCycLVs28be/3U9sbBwAHTp0YtGi1/yvOXr0CFOn3kVERAR33HF30GJTgW0S\nl7vyHuwy3dolsetALlvTsvF6vRiGcdIxIiIiImZzeVwcK8415VqJkfHYLNUrT3Nzczh06AAjRlxG\nREQkAL179+Wuu+7B6XRW+pqUlEZccMFQ0tJ2BSzmyqjANknZLo5QeYHdvV0y73+VTlZeMQczC2me\nEmtmeCIiIiIncXlcPLb+f8gqPmbK9ZIjE3l44P3VKrITE5Po3bsv06b9kVGjrqB377506XIeY8de\nCcDy5R9UON7j8bBnTxpr1nzOVVf9Lijxl1GBbZLykxzLr4NdpnWTOOJjw8ktcLDx16MqsEVERERO\n45//nMPSpe+wZs1q/v3vlwG48MKLmT79AQB27fqVyy67CACv10tiYhLDh49gwoTrgxqXCmyTuE/T\nImIxDHp3SOGLTQfY+GsmYwe3NTM8ERERkZPYLDYeHnh/rWwRAYiIiGDChBuYMOEGSkpK+PHHzfzr\nX3N56qnHuPDCi0lN7VihB9ssKrBNUtUyfWX6dGzIF5sOsDcjn8xcOw3jo8wKT0RERKRSNouNlOjk\nUIdxks8++4T585/nrbfexzAMIiIi6N9/AA6Hg2eeeZoLL7w4ZLFpHWyTVLVMX5nOrRKJivD9zbNp\np3Z1FBERETmVfv3Ox24vZPbsWRw75lskYv/+fbz99hsMHjw0pLGpwDZJ2SRHm9U45QohNquFnu19\nfyFu+vWoabGJiIiI1DXx8QnMm/cimZlHmTjx91x66RCmTr2LLl3O409/mh7S2NQiYhKXx9eDXVn/\ndXl9Oqaw/qcMftmXQ26hg/iYcDPCExEREalzWrVqw8yZT1f63BVXjOWKK8aaHJGPRrBNcnwEu+qU\nd2+fTESYFa8XNuw4YkZoIiIiIhJAKrBNUjbJ8VQTHMtEhFnp1aEhAN/9nBH0uEREREQksFRgm6Rs\nkmNla2Cf6PwujQDYuT+X7LzioMYlIiIiIoGlAtsk1W0RAejWNtm/msj3ahMRERERqVNUYJukupMc\nwddG0qej2kRERERE6iIV2CY5kxFsgAFdGgOQfiifIzn2oMUlIiIiIoGlAtsk/kmO1ejBBujcOpHY\nqDAAvtcotoiIiEidUe0COzMzk0GDBrF69WoADh8+zF133cWAAQMYPHgwjz/+OA6HAwCv18usWbMY\nOHAg/fv3Z+bMmbjd7uD8BHVE2SRHazVHsG1WC/06pQDw3c/qwxaR+sfr9fLp9/vYqI21RKSeqXaB\nPWPGDHJycvzf33///TRp0oQvv/yS999/n61bt/L8888DsGTJEr744guWLVvG8uXL2bhxI4sXLw58\n9HWIy+3rwT7dMn3lnV/aJrLvSAGHsgqDEpeISKikHcrj9c928ty7W7V7rYjUK9Wq9l5//XWioqJo\n2rQpAA6Hg6ioKO68804iIiJISUlh7NixbNq0CYClS5dy880306hRI1JSUpgyZQrvvfde8H6KOuD4\nMn3VL7A7tkzw7+SoUWwRqW+y80r8X7/40U9kZBeFMBoRkcA57Vbp6enpvPTSS7z11ltcffXVAISH\nh/PCCy9UOG716tV07twZgLS0NFJTU/3PtW3blvT0dLxeL4ZRvR5kwzCwhKBD3GIxKvwbKG7P8RFs\nazX7sK1Wg/O7NuLT7/fz3c8ZXHVh22rnry4IVq6lcsq3eZTr6rGXuMp97WbOOz/y3zf3A2BfRj4d\nWyVgPc0vAuXaXMq3eZRr8wQj11UW2C6XiwceeIAZM2aQkJBQ6TFer5cnnniCtLQ0/ud//gcAu91O\nZGSk/5ioqCg8Hg8Oh4OIiIhqBZacHBPSYjIhISag57OU/pKIiQ4nKSm22q8bObAtn36/n0NZReSV\neGjbLD6gcdUGgc61VE35No9yXTVvueLZajE4lFXE/7y+maxcO/lFTtq3iOeeCb1p1/z09z3l2lzK\nt3mUa/MEMtdVFtjz5s2jS5cuDBs2rNLni4uLeeCBB/jll1947bXXSE5OBiAyMpKSkuMf/dntdmw2\nW7WLa4CsrMKQjWAnJMSQk1OIp3TUORAK7b4JoG6Xm+zsgmq/LiUujOQGkWTlFfPJunR+d3Hq6V9U\nRwQr11I55ds8ynX1HM32zS3p3DqBoT2asfCDn9hzKM///O79uUz/3zU8OLEPHVpUPsijXJtL+TaP\ncm2e6ub6TAZIqyywly9fztGjR1m+fDkABQUFTJ8+nTvvvJMJEyZw6623Eh0dzZtvvllhhLt9+/ak\np6fTs2dPwNdm0q5du2oHBb6R8VAuPOLxeHG7A/eGdpatg22xnPF5+3dpxIpvf2P99gyuGtquXrWJ\nQOBzLVVTvs2jXFetoMg38BAdEcag85qQW+Dg3S/T6JWazPldGvPm5zvJyivhpY928Mik/lXOYVGu\nzaV8m0e5Nk8gc11lgb1ixYoK3w8fPpyHHnqIiy66iJtuuomGDRsyd+5cwsLCKhw3btw4Fi1axMCB\nA7HZbCxYsIDx48cHJOC6yj/J0XbmxfGALo1Z8e1vZOYWk34on3bNGgQ6PBER0xUW+3qwYyJ9v4ou\nG9CKkf1b+vsgE+MiePK1HziQWcjK735j9KA2oQpVROSMnHaSY2U2bdrEd999R0REBOeff77/8a5d\nu7JkyRJuuOEGMjMzufbaa3E6nYwdO5ZJkyYFLOi66Ex3ciyvVeNYGidGkXHMzrrth1Vgi0i9UGh3\nAhATdXyQpvwko/bN4xnWqxlfbD7I0q/30Cw5ht4dU0yPU0TkTJ1Rgf3555/7v/7ll19OeZzVamXa\ntGlMmzat5pHVM87SjxxqUmAbhsEF3Zrw3lfprN9+mAkXp57RetoiIrVR0Qkj2JW55qL2bE3LJiuv\nmOfe3crvh6cyon/LetcqJyL1i6o0k7j962DX7JfC4O5NMfB9pLp5V2YAIxMRCY3C4tIR7MiwUx4T\nExnG3yb2pXXjOLzAG5/v4t+f/Irb4zEpShGRM6cC2yTO0gI7rAYj2ABJDSLp2iYRgK9/PBSwuERE\nQqWgdAQ7uooRbPD1Yv/1v/rQK7UhAKs3HWDRhz/j9Wril4jUTiqwTXJ8kmPNUz64h28nzW3pWRzL\nLznN0SIitZfL7aHE4VsqqnwP9qlEhFu5++ruXNqvBQDrf8rg258yghqjiEhNqcA2iesserDL9OmQ\nQnSEDa8XvtpyMFChiYiYrqz/GiC2ihaR8iwWg+sv6UC3tkkA/PuTX8nOKw5KfCIiZ0MFtklcZ9mD\nDRAeZmVwd98o9potB9WDKCJ1Vln/NZy+RaQ8wzCYdEUXoiNsFJW4WPyRWkVEpPZRgW2S4wX22aX8\not7NADiWX8LmnVlnHZeISCgUlhvBrmqSY2US4yK4cWRHALamZbNi3Z4ARiYicvZUYJvE6fKNsNR0\nkmOZpskxdGntm+z4xab9Zx2XiEgolK2BbRgQGWE949cP6NqYfp18a2Iv+mA7h7OLAhqfiMjZUIFt\nAq/X6x/Btp5lgQ1wce/mAGzfc4xDWYVnfT4REbMdXwM7DEsN1rQ2DIOJozoRHxNOicPNc+9spcTp\nDnSYIiI1ogLbBG7P8f7AsLPowS7Tu2NDkhtEAPDp9/vO+nwiImYr8K+BXaMNhQGIiw5nyvjzsBiw\n70gBr67YoX5sEakVVGCboGz0Gs5umb4yVouFS/q2BGDttsPkFTnO+pwiImYq8q+BfWb91yc6r20S\nN17eBYB12zP4SvsEiEgtoALbBGVL9MHZT3Isc2HPZkSEW3G6PHyx6UBAzikiYpayHuyYqJqPYJe5\n5uIO/k1o/m/Vr2qdE5GQU4FtAqfr+Aj22U5yLBMdaePCHr4VRT77Yb96D0WkTqnONunVZbEY3Dq2\nC/Gx4TicHhYs217hk0MREbOpwDZB+Ru9NQA92GVG9G+B1WKQX+TkS208IyJ1SKF/kuPZj2CDrx/7\n1jFdAfgto4Bla/cE5LwiIjWhAtsE5QvsQI1gAzSMj2JQtyYArPj2twoj5SIitVkgR7DLnNcmyb+V\n+vJ1e0k/lBewc4uInAkV2Cao0IMdgEmO5Y0e2BrD8G08s3arJveISN1QFOAR7DLXDGtP48QoPF4v\nL374E06X2udExHwqsE1QYRWRAI5gAzROimZAl8YAfLhuj0axRaROOD7JMXAj2AARYVYmj+mKYcCh\nrCLe+yo9oOcXEakOFdgmKF/02gLYg11m7OA2GAZk55VoRRERqfW8Xq+/Bzs6wCPYAKnN47lsQCsA\nVn77Gzv35wT8GiIiVVGBbQJ3EEewwbd9+uDuTQHfKHaxwxXwa4iIBEqJ0+3fgCuQPdjlXTmkHc0b\nxuAFXvzwJ+wlui+KiHlUYJvAWdqDbQBWS+BHsAHGDW6DzepbUeST77S7o4jUXmX91xCcEWyAMJuF\nW8d0xWp0nP6XAAAgAElEQVQxOJpTzJJPfw3KdUREKqMC2wSO0jWqw8OsGEZwCuyG8VFc3Lt09vy3\nezmWXxKU64iInK2icqPJ0RHBKbABWjeJ45ph7QH4Ztth1m8/HLRriYiUpwLbBA5XWYEd3HSPHdyG\nmEgbDqeHd9fsDuq1RERqyowR7DIjz29J1zaJALz2yS8czbEH9XoiInAGBXZmZiaDBg1i9erVAOTm\n5vLHP/6Rvn37ctFFF/Gf//zHf6zX62XWrFkMHDiQ/v37M3PmTNzuc3eppBKnrwc73GYN6nVio8K4\ncmg7ANZuO6w1YEWkVirrh7YYBhFhwb0vWgyDyaO7EhsVhr3EzcIPfsLt0WpLIhJc1S6wZ8yYQU7O\n8ZnYDz30ENHR0XzzzTfMmTOHf/7zn2zevBmAJUuW8MUXX7Bs2TKWL1/Oxo0bWbx4ceCjryPKWkQi\nwoP7iwRgWK9mNE2OBuDVlb/g8XhP8woREXOVtYhERQSvba68xLgIJl3RGYBdB3L5QLs8ikiQVavA\nfv3114mKiqJpU99KFYWFhaxatYp77rmHiIgIevTowZgxY3j//fcBWLp0KTfffDONGjUiJSWFKVOm\n8N577wXvp6jl/D3YAd5kpjI2q4UbR3YCYO/hfFZr2T4RqWXs/gI7uO0h5fXukMLFvZsD8ME3e7R0\nn4gE1Wnvbunp6bz00ku89dZbXH311QDs3bsXm81Gy5Yt/ce1bduWTz75BIC0tDRSU1MrPJeeno7X\n6632aIVhGFhC0CFuKV3lwxLA1T6cpcv0RYRbsQZhHewTdWuXxAXdmvDNtsO8++Vu+nVOIalBZNCv\ne6aCkWs5NeXbPMp11YodvkGHmMiws74nnkmurx/RgV/25XAws5CFH/zEY7eeH7RlAusrvbfNo1yb\nJxi5rrLAdrlcPPDAA8yYMYOEhAT/40VFRURGVizYIiMjKS4uBsBut1d4PioqCo/Hg8PhICIiolqB\nJSfHmPLR4akkJMQE7FyG1dcaEhsdTlJSbMDOW5U7ru3Jlt1ZFNqdLFm1i4cnDwhpPqsSyFzL6Snf\n5lGuK+ctvRc1iI0I2D2xurn+6839mf6/X5KZW8wbn+3mvhv71tp7Y22m97Z5lGvzBDLXVRbY8+bN\no0uXLgwbNqzC41FRUZSUVFwGrri4mOhoX+9vZGRkheftdjs2m63axTVAVlZhyEawExJiyMkpDFj/\ncl6+7w8PAy/Z2QUBOWd13HBpBxZ+8BMbfs7ggzW7GNKjqWnXro5g5FpOTfk2j3JdtezSlTzCrJz1\nPfFMcx0faeX3w9uz5NOdfLn5AJ1axte6e2Ntpve2eZRr81Q312cyIFBlgb18+XKOHj3K8uXLASgo\nKGD69OnceuutOJ1ODh48SLNmzQBfK0lZW0j79u1JT0+nZ8+e/ufatWtX7aDAtxJJKBce8Xi8uN2B\neUOXlH4cGma1Buyc1TGwa2O++ymDLbuz+Pcnv9KxRQLJ8bWvVSSQuZbTU77No1xXrrDYCUBUuC1g\n+TmTXA/v04Ifd2ezNS2LV1f8QuvGcTRrqFHCM6H3tnmUa/MEMtdVjhGvWLGCH374gQ0bNrBhwwaa\nNWvGM888wx//+EcuueQSZs2ahd1u58cff+TDDz9k7NixAIwbN45FixZx+PBhMjMzWbBgAePHjw9I\nwHVRiYmriJRnGAY3XdaZmEgb9hIXCz/Yrr+CRSTkikIwybE8wzD4w+guxMeEU+J089y7W7WVuogE\nVI2bMB5//HFcLhfDhg3jnnvu4f777/ePWN9www0MHz6ca6+9ltGjR9OnTx8mTZoUsKDrGjNXETlR\nYlwEt1zeBYBf9+fywTd7TI9BRKQ8e+lGM8HeZKYq8THh3HllN6wWg8PZRSz+6Ge8Xg1AiEhgnNHd\n7fPPP/d/nZCQwOzZsys9zmq1Mm3aNKZNm3Z20dUTJa7SjWaCvKHCqfTtlMJFvZrxxeaDLPs6nTZN\n4uiZ2jAksYiIhHoEu0zHlglMuDiV1z/byQ+/HuXjb3/jioGtQxqTiNQP2irdBI7SHuyIIG+VXpXf\nX9KBVo1j8QIvfLCdQ1mFIYtFRM5tZQV2dIgLbIBL+7VgYNfGALyzZjfb92SHOCIRqQ9UYJsg1CPY\nABFhVv50dQ/ion3bBc99ZytFxeo5FBHzhWKjmVMxDIObL+tMi5QYvF6Y9942Dhw1b7UnEamfVGCb\nwL9VeggLbIDk+EjuKtdzuPCD7XjUcygiJnK5PTicvkGHUPZglxcRbuXua8oGIFw8+58tHMsvOf0L\nRUROQQW2CUI5yfFEnVolcv2lHQDYsjuLNz7bqYk9ImKa8qt11IYR7DKNEqL487U9CbdZyM4r4dm3\ntmhlERGpsdBXfOeAktLRmlCPYJe5uHdzhvXyrV++asN+ln6dHuKIRORcUVSuaK0NPdjltWvWgDvG\nd8MwYP/RAp5/bysutyfUYYlIHaQCO8g8Hq//Bh3KHuzyDMPgxpEd6dcpBYBla/ew4tvfQhyViJwL\nKoxg15IWkfJ6dWjIxJGdAPhpzzHmvbcNp0tFtoicGRXYQeZwHd+OMjyEq4icyGqxcPu48+jWLgmA\nt1bvYvWmAyGOSkTqu/KTq2vbCHaZi3o3Z9zgNgBs3pXJc+9uxekK4dbCIlLn1M67Wz1S1h4CtadF\npIzNauGPV3Xn2be28Ou+HF5b+QsOp5tR57cKdWh1hsfrxe324HT5PqlwuT24PV4MfJ8UGMYJ/4L/\n6zCrBZvNwGqpPX94iQRb2Qh2uM2CzVp73/tXDm2HxTB4/+t0tqZlMeftH7n7mh617j5eW3m9Xtye\nsvuiF6fLg9vjwcB3P4Ry98bSrzHAYhhYLQZhNgtWi+F7XKQOUoF9ArfHTY49l9ySQtwBGLDIKrJD\nmG82uoMicmvhxPRJ49qyYOk29mQU8OZX2zlScIyxF7QN+o3NagWL3R2wXJ8Nr8dLgd1BbpGT3MIS\n8god5BY4yC9yUFTixl7i8v3P4aa4xInD6ful4Q7ABFED/MW2zWrBZrFgtVkIs5Z+bzWwWi2+Y6wW\nrFaDMIuBtbRAsVl9RXqY1YLVZmAzDGw2i+81Ft/xNqtBeJiVhIQoiu0ODAxsFt81LRYDi1H6L75f\ndBbL8T8KfM+Xfm2E6BdeaZ792faClxMeq3Cot/zLKPfQ8dd5T3xNxbOd+J/WWz4G7wlHe73+r72A\n1WKQ4yokN6cIt6dm75G4yCiSYmJr9NpA8nq95DkCt2xdVlEuhJUQGR1Gbkn+WZ8vmPeRYf0b4rLY\n+XDdXrYfOMyst4uYdEUXoiPDAnuh2szrpdjpJrfAQW5hCQV2JyVuL0eyCimwOykucVNU4sTucGMv\ndlHidON2e3G6PSf9f/NMGfg+bQ2zgbX03mizWrDZLNgsvvua755Yev+zGv7nrKX3xkpfU3bvLL0/\nhpUOdNisltLCvuy+67sPWkrvfZbSPwgspYW/774ZvHtjtd7bldwbff+cfH888T7n9VbxutPcH08+\nprJ7dOUxVozpzN8lsRGRJMfGnfHrzGZ4a+kSEkePnv2N90wVu0qY+e0/OVaSa/q1RUTK83oMRqSM\n56qeF4Q0jrmbFrLj2M6QxiAiUsbrMbgoaQwT+gwN2DmtVoOkpFiyswtwu09dFqekVL+wr72fz4WA\nx+uh2F0Lh5hF5JxjWLxsy9wR0hg8Xo+KaxGpVQyLt07UamoRKSc6LIonh87AFV5Mbm4RngBMHE8/\nmMdLH/t+ST58cz9stWAt7Kp4vV4+23iALzcfBCAm0sY1F7UntXl8wK9lsUB8fHTAcl1U7GLv4Tz2\nHM5nz6E8DmfbT/kRZWykjaT4SJLjI0luEEHD+CgSYiOIiwknJsKGxVL/+v4MA2LjIsnOLsTh9Pj7\nI90er++jQq8Xj7fc1x4vHko/RvSUPocXj+f4sZVeB+PEB6r61h9bZUf5ezUrvNDgxK+OP2X4vz/p\nnOUeqHDek85R9o9RyTmOP1dZ7GUfEVssBnGxkRQUFOMp7cmv5Ic8pSU/foQ9ah8Oiqr9mmC7rtPV\ntItvfdbnWb5+D+u3HyG1eQNuuqzzWZ8v0PeRquzNyOeNVTspLHZhGDCiXwsu6N4USy3vE3a5POzP\nLGTPoTzSD+Wx72gBLlfl//8NsxkkN4gkOT6K5PgIkhtEkhgXSYPoMBpEhxEebjUt32Zyezy43V5f\n65/Hg8vlweP14im7/+FbFcyLF68H/3OUu1dSel+sMi+VvFVO9e6xWg2ioyMoKirB4281q969sfx3\n5e8//scq3PxOeF01748nn6fic5Wc2n+PrPzc1fv/UXxkNO1SmlTr2FBSgX2CKFskSQkNyfZU/TFB\ndWVZw/Da4zAMaBXfrE5M2Lh5WDO6NMrg5Y93UHDMzavvHWTMBeGMG9ImoBPyrFaDpITYGufa4/GS\ndiiPzTsz2ZqWxf4jBeUKahvg+yinUWIUbZrE0bpxHK2bxNGqcRyxUedQD2Wpso/AEo3AvLfl1Kr7\nceOpvLM5ATv7cGIPQnQ10zAqieaxTc/6PJaSY3jtdhJsKQE539neR85E89imdExuyZx3fmT/0UJW\nfpnLvt8sTB7dlcS4iKBe+0wdzi7y3xt3Hcg9YalBX29/bFSY797YJM5/j0yOj6zy95SZ+T7Xne19\nREJLBXaQlW0JHB5mrRPFdZnzuzSmdeM4/vX+Nn47UsAH3+zhx7QsbrmsM62bhG5yQbHDxfb0Y2ze\ndZQfd2eRX+Ss8LzFMGjdJI5OrRLo3CqB1Obx59aEJKkXrJ5IABy1qMAOlLJl+mrTLo5nomFCFH+b\n2Jd/f/Ir32w7zE97jvHfL67ndxelcmGvZiEbzXZ7POzan8vmXZls3pVFRvbJn340Soyic6sEOrVK\npEOLeJIbVF1Mi0jN1c07XB1SUrpNel1c2qlxUjQzburLG5/51sjeezifx175nmG9mnPlkLY0iAk3\nJY7svGK27Mpk065MduzNOWlntUaJUfRKbUjXNkl0aBFfZ39xi5Sx+QvsIrxeb70qgsqW6YuuhZvM\nVFdkuI1bx3SlW9sklnz6K4XFLl5d+QtrthzkuuGpdGqVaEocRcUutqVnsXlXJlt3Z1FYXHFr99io\nMHq0T+a8Nkl0apVAUoNIU+ISERXYQecoLbDDa3nv9amE2axMHNWJfp1SeGXlLxw5ZueLTQdYt+0w\nF/Vuxsj+rQL+0WhZ68ePuzPZsiuLfUcqLhFmGJDaPJ5eHRrSK7UhTZKi61UBImL1RAHgNTzYXXai\nw6JDHFHglG2VXh/+EB54XhO6tEni/z79le93HGHv4Xye/r9NdGmdyOhBrenSOjGg9yav18vBrCK2\n7s7ix92Z7Nyfe9IykM0axtAzNZleqQ1p3yy+Xs4nEakL6v4drpYr22imLo5gl9elTRKPTz6fVT/s\n58Nv9mAvcbPyu318+v1+enVoyAXdmtCtbVKNt4PPyi1m5/4ctqZlszUtiwJ7xdaPyHAr3dol0ys1\nme7tkomLNmf0XCQUylpEAPIcBfWqwPaPYNeDAhsgPiacO6/sxsV7j/HG5zv5LaOAn/ce4+e9x2iS\nFM3g7k3o37kRjRJr9t8wr9DB7gO5bEvP5sfdWWTlFVd43mox6NgygZ6pDemVmlzj64hIYNWPO1wt\n5h/BrkXbpNdUmM3K5QNaM7RHM1Zt2MdnP+ynsNjFxl+PsvHXo4SHWejQIoEOLeJpkRJL48Qo4mMj\niAy3YrUYuNxe7CUuMnOLOZJTVPpvMT+lZZGdf/KSO40SouiRmkzP9g3p1CqhVu/6JhJIFQvsfJrE\nNAphNIFV9sdzTB1uEalM59aJPHxLf7bszOTDdXtJP5TH4ewi3lmTxjtr0vz9z60ax9E0OYbEuAii\nI22EWS14vF5KHG6y80rIzLVzNLeYQ5mF7D6Yy9Gc4pOuFRcdRvd2yfRon0y3tkmaZyJSC9WvO1wt\nVOKquz3YpxIbFcaVQ9tx+cDWbNhxhK9+PMTOfTk4nB62p2ezPT27Rue1WS2kNm9Aj/YN6ZmarNYP\nOWdZsOF12TBsLvIc5m+6FSxFxS7sJb57Yn3sB7YYBr07ptC7Ywrph/L4cstBfvjlKAV2J0eO2Tly\nzA4cqtF5WzeJo0d7X1HduklcrV8aUORcpwI7yMqvIlLfRIRZGdy9KYO7NyW30MHW3Vn8ui+HtEN5\nZGRXvUV0RJiVRolRtGrSgJYp0bRvFk+rxnGE1dFedZFA8zrD612BnZ1/fDQ2qUHtWtYu0No2bUDb\npg24cWRHdu3P5ee9x9h9MI/9RwvILXCc8nWR4VYaxkfRKDGKtk3jSG0eT5smDYgIr3+/Q0TqMxXY\nQVZSxyc5Vld8TDhDejRlSA/furYut4ec/BJyixyUONy4PV7CrBYiwq0kNfBtWmCzWbTGp8gpeJ0R\nEFVEvqPg9AfXEdml/cOGAQmx9bvALmO1WOjUKrHCyiIlTje5BSXYS9w4XR4sFoPwMAsJsRHERNr0\nyZ1IPaACO8gcdXiZvrNhs1pomBBFw4SoUIciUid5nb4CNK+kHo1g5/nmWiTERpzTcyp8n+BpMqJI\nfVatO9zy5cu5/PLL6d27N6NHj2bVqlUAZGRkcMcdd9C/f3+GDBnCrFmz8JTuEer1epk1axYDBw6k\nf//+zJw5E7fbHbyfpJaqzy0iIhJETt9KOfWpRaRsBYz63h4iInLaAjs9PZ2//e1vPPHEE2zatIkZ\nM2YwdepUsrOzmTlzJq1atWLdunW8/fbbLF++nGXLlgGwZMkSvvjiC5YtW8by5cvZuHEjixcvDvoP\nVNs4XPVnFRERMU/ZCHZ+PSqwy1pEkuvhBEcRkfJOW/W1bduWtWvX0qdPH1wuF5mZmcTExBAeHs6e\nPXtwu93+UWuLxUJEhO+XwtKlS7n55ptp1KgRKSkpTJkyhffeey+4P00tVJd3chSR0PG3iNSjAjur\ntEWkPq4gIiJSXrV6sGNiYti3bx+jRo3C4/Hw97//ndjYWCZPnsxDDz3E66+/jtvt5qqrruLyyy8H\nIC0tjdTUVP852rZtS3p6erW3/TUMA0sIBn3Ldr0K1O5XztIWkchwK1arJq6UF+hcS9WUb/Ocba4N\nw8Bb2iKS7yzEsHixGObfEA3v8fgtFuOs72HHSlcRaRgfGbD7od7X5lK+zaNcmycYua72JMemTZuy\nZcsWNmzYwF133UXr1q0BmDJlCpMnT+bAgQPccccdvPHGG1x33XXY7XYiI4+PUkRFReHxeHA4HP5R\n7qokJ8eEdCZ1QkJMQM7jKl2qLiE+mqSk2ICcs74JVK6lepRv89Q01z06prDtyzQAPF4PYTEQH2n+\n/cPj9fi/bhAXdVb3MLfHy7HSDaVaN08I+P1Q72tzKd/mUa7NE8hcV7vAttl8hw4aNIiRI0fy0Ucf\n8cEHH/D9998THh5Oamoqt99+u7/AjoyMpKTk+O58drsdm81WreIaICurMGQj2AkJMeTkFOKpYh3n\n6ioq9m0L7HK6yM6uP8ttBUKgcy1VU77Nc7a5HtQlhf+sOT5A8VtGBs3jzL8hli+w8/LtZIfX/B52\nLL8EV+lynBEWAnY/1PvaXMq3eZRr81Q312cyMHDaAnvNmjW89NJLvPzyy/7HnE4nubm5OJ1OnE4n\n4eG+jzKtVitWq6/XuH379qSnp9OzZ0/AN1myXbt21Q7M6/USykVHPB5vQNZmLlumL8xq0VrPpxCo\nXEv1KN/mqWmuoyPCGNSlNd+Vfp9ZmEuT6CaBDa4aPN7jsZ/t++boMbv/64TYiIC/B/W+NpfybR7l\n2jyBzPVph0S6du3Ktm3beP/99/F4PKxZs4Y1a9YwdepUmjRpwtNPP43D4WD//v0sXryY0aNHAzBu\n3DgWLVrE4cOHyczMZMGCBYwfPz4gQdclWkVERGri8v5t8DrDAFi785cQR3P2skvbQ8LDLMREagsG\nEanfTlv1paSkMH/+fF599VX69evH7Nmzef755+ncuTMvvPAC+/fvZ8iQIUycOJErrriCm266CYAb\nbriB4cOHc+211zJ69Gj69OnDpEmTgv4D1SZuj8f/kajWwRaRM9EoMZokWgLwY/735NoLQxzR2cnK\nLV0DOy5SOxWKSL1XrWGEfv368e677570eGpq6inXtrZarUybNo1p06adXYR1WNkmM6Bl+kTkzN3U\naxz/u3U2RlgJi777iOnDJoQ6pBo7vga2NpkRkfpPfQtBVNZ/DRBuU6pF5Mx0bNyMFkY3AHY5NrE3\n62iII6q547s4ag1sEan/VPUFUYHd6f86Wj2HIlIDt/YfB64wDKubed+/7t/Yq645mFUEQKPEqBBH\nIiISfCqwg6hsxMYwfLPmRUTOVKMG8ZyfcBEABeH7eXfLN6ENqAbsJS4ysn0FdusmcSGORkQk+FRg\nB1HZtsAJsRHYrEq1iNTMxH6XEFnSGIDVR1ayPyc7xBGdmd8yjm/33rqxCmwRqf9U9QXR8Uk96jkU\nkZqzWCzc1vv3eN1WCCvhf9e/jMsTwo0CztDew74CO7lBBHHR4SGORkQk+FRgB9HxST1qDxGRs9O5\nSQsGJVwKgD38MPO+XhriiKpvb+kIdiuNXovIOUIFdhBll677mhyvEWwROXsT+19Ckqs9ADuc6/lg\n67chjqh69mb4tkVvo/5rETlHqMAOorIebLWIiEig3D/0JmwlCRgGfHx4KZv2pYU6pCqVONwcyvJt\nkqMJjiJyrlCBHSRuj4djpVsDa91XEQmUBlFRTO0/GZwRGFYXL/70MjszDoY6rFPad6QAr29DW1o3\naRDaYERETKICO0hyCxx4Sn+raARbRAKpbcPG/FeH/yqd9FjM7E0L+S07M9RhVWrP4TwAEmLDiY/R\nBEcROTeowA6SsgmOoK2BRSTwLmjXmfEtfofXY+ANL+T/ffd8rRzJ3rk/F4A2Gr0WkXOICuwgKSuw\nI8OtREVoF0cRCbxRXfowstGV/iL7fzfPZ/P+PaEOy8/t8bA93bdm93ltk0IcjYiIeVRgB0l2uQmO\nhmGEOBoRqa+u7DGI8c1/728XWfjTQtal7Qh1WACkHcyjqMQFQPf2ySGORkTEPCqwg+T4GtjqvxaR\n4BrVpQ83tJsIrjCwOXlt9yu8s3ltqMNia1oWAI2TommUEBXiaEREzKMCO0i0BraImGlI+67c2qVs\ndRE3n2cv5Z+r3wzpjo8/7vYV2N3bqT1ERM4tKrCDJMu/TbomOIqIOXq3bMcD/e4hrMRX0KZ7f+Bv\nnzxHVkGB6bHkFpTwW+kGMz3aqT1ERM4tKrCDwOv1kpmrFhERMV/r5BSeumQ6ya4OABSGH+CRr2ex\ndvfPpsaxeZdv2cBwm4VOrRJMvbaISKipwA6CIzl2ih2+j2VbpMSGOBoROddEhYfz90sn0zNymH+F\nkSXpLzP7y3dwuc1pGVm79TDgm9wYZrOack0RkdpCBXYQ7DmUD0CYzUKzhtEhjkZEzkUWi4XbLxjN\nxHaTsDhiMCxefnV9ywOfPMuuI4eCeu2DmYXsOuBb/3poj2ZBvZaISG2kAjsIynYua9UoFqtFKRaR\n0BnUrjMzh93nbxkpiTjCM1vmsGj9CjweT1Cu+fVWXwGfGBdBN61/LSLnIFV/QVA2gq2dy0SkNoiP\niuGxkbdxUeJYcIVhWN1sLPqc+1c+y68B3v3R5fbwTWmBPbh7EywW7QMgIueeahXYy5cv5/LLL6d3\n796MHj2aVatWAeBwOHj88ccZMGAAAwYMYMaMGTgcDsA30W/WrFkMHDiQ/v37M3PmTNwm9f6Fksfr\nZW+Gr8Bu3SQuxNGIiBz3u95DmXH+vTRwtgKgOCKD//1xLi9881HAlvP79qcM8oqcAAzp3jQg5xQR\nqWtOW2Cnp6fzt7/9jSeeeIJNmzYxY8YMpk6dSnZ2Ns888ww7d+5k5cqVrFy5kl27drF48WIAlixZ\nwhdffMGyZctYvnw5Gzdu9D9Xn2VkF/knOLZpqgJbRGqXZglJPDXq7tLR7HAMq5stxWt4YOUz7Di8\n/6zO7XJ7WPp1OgC9OzSkUaLmoIjIuem0BXbbtm1Zu3Ytffr0weVykZmZSUxMDGFhYbz55ps8/PDD\nJCQkkJCQwJw5cxg7diwAS5cu5eabb6ZRo0akpKQwZcoU3nvvvaD/QKG257Bv9Do8zELTZP1yEZHa\n6Xe9hzJjwL3EO9sAUBJxlDnbnmP+2g9rPJr99dZDZOYWYwBXDW0XuGBFROoYW3UOiomJYd++fYwa\nNQqPx8Pf//53MjIycLvdbNmyhbvuugu73c6YMWOYPn06AGlpaaSmpvrP0bZtW9LT0/F6vRjG6Xvy\nDMMgFPMDy/oFa9o36G8PaRxHeJiWpqrK2eZazozybZ66kuuWSUk8fcUfeXvj16w6vAIjrIStJV/y\nl0928Mf+N9I+pbH/WIvFwGo99c9TVOzig7V7ABhwXmNam/QJXl3JdX2hfJtHuTZPMHJdrQIboGnT\npmzZsoUNGzZw1113MXnyZJxOJ6tXr+btt9+msLCQKVOmEBcX5y+4IyOPb7ISFRWFx+PB4XAQEXH6\n3Q2Tk2OqVYgHS0JCTI1eV7ZzWee2ySQlaQ3s6qhprqVmlG/z1JVc337pZYw5NoDHVrxItiWN4vAj\nzNo4hyGNRviPaRAXdcp7mtfr5YXXNnAsvwSb1cItY7uZfv+rK7muL5Rv8yjX5glkrqtdYNtsvkMH\nDRrEyJEj2bx5Mx6Ph6lTp9KgQQMaNGjApEmTeO2117jrrruIjIykpKTE/3q73Y7NZqtWcQ2QlVUY\nshHshIQYcnIK8Xi8Z/TaAruTX347BkCbRjFkZ5u/PXFdcja5ljOnfJunLuY6kjCeHHUnb/7wJauP\nrACbk6+zPvY/n5dvJzu88nvaZxv2s3aLbzWS6y5JJcqKafe/upjrukz5No9ybZ7q5vpMBg5OW2Cv\nWbOGl156iZdfftn/mNPppE2bNqxdu9a/aghQYZWQ9u3bk56eTs+ePQHfZMl27arfk+f1egnloiMe\nj0kAmCQAACAASURBVBe3+8ze0Ft3Z+H1gtVi0LlV4hm//lxVk1xLzSnf5qmLub6211AG5pzH3PX/\npiD8+KTH/EIH7oSKP4vX62Xld/t4a/UuAHqlNuTi3s1D8jPXxVzXZcq3eZRr8wQy16cdI+7atSvb\ntm3j/fffx+PxsGbNGtasWcP111/PpZdeyjPPPENeXh4ZGRm88sorXHbZZQCMGzeORYsWcfjwYTIz\nM1mwYAHjx48PSNC11dbdWQB0aBFPVES1PxwQEalVWiQk8dTIuxkQOxKvy4bXY+AoiKpwTF6hgxc+\n+MlfXLdt2oA/jO4S0tY+EZHa4rRVYEpKCvPnz+fJJ5/kscceo02bNjz//PO0b9+ep556iqeffpor\nrrgCp9PJlVdeyR/+8AcAbrjhBjIzM7n22mtxOp2MHTuWSZMmBf0HChWP18vW9GwAurdPDnE0IiJn\nx2KxMLH/JXz9jAcXLpbtziAlJonmDWP56seDfPjNHgqLXYBvSb7bx51HhCZ2i4gAYHi93lr5ucPR\no/khua7VapCUFEt2dsEZfUyw93A+j778PQCPTT6fFima4Hg6Nc211IzybZ76lOuvthzk1ZW/4K6k\nLzEy3Mo1w9pzcZ/mWEI0cl2fcl0XKN/mUa7NU91cp6RUf3Uk9TEEyJbdmQAkxkXQvKFm/IpI/TC0\nZzOaNozhhWXbycwtBnzzTAZ3b8r4IW1JjKvexHURkXOJCuwA8Hq9rNueAUCvDg3Vgygi9Upq83j+\nMWUQmbl2Mo7ZaZYcQ3J85OlfKCJyjlKBHQA79+eSkV0EwJDuTUMcjYhI4FksBo0So7X9uYhINYRg\npen656vS9V9bpMTSpok5u5eJiIiISO2kAvssFRW7+H7HEQAu7NlU7SEiIiIi5zgV2Gfp662HcLg8\n2KwWBp7XJNThiIiIiEiIqcA+C/YSFx+t2wPAwPMaExsVFtJ4RERERCT0VGCfhU837CO/yInNajB+\ncNtQhyMiIiIitYAK7BPYS1zYS1ynPS630MGKb38D4OLeLbRklYiIiIgAKrArKLA7mT53LXf/z+fk\nFTpOeZzX6+Wl5T9T7HATGW5l9AWtTYxSRERERGozFdjl2KwGHq+XI8fsLP7oZ061i/zqTQf4cXcW\nANdf2oEG0eFmhikiIiIitZgK7HIiw23814iOAGzamcma0vWty/t57zHe+GwXAP06pWhjGRERERGp\nQAX2CYb2bMqA0uX2Xlv5C5/9sN//3K79ucx5+0dcbg8N4yO56bLOWvdaRERERCrQVuknMAyDP03o\nxcHnv2bfkQKWfPor3/+cQUJcxP9n787DoyrP//G/z+wzSchGwg6SIAHZwg6CQsFdAbWgltoPBUVQ\nq9aFX9Vebh9oq1dFpaIiFK24VL9YF4pWEYroBxREBAQEhUQWIUL2bfY5vz8m58w5Z5bMTCbJTHi/\nrsuSmTlz5uRh+szNPfdzP/jq4GmIIpCVbsJ9vxrOtnxEREREFIQZ7BAy08344/+MxOCCHADA9ydq\nsOM7f3Cdl2XBvTcMR36WtZ2vkoiIiIiSETPYYVjNBvx+5jB888MZ7Pr+DE5X2zFmYBdMLu4Bo4H/\nLiEiIiKi0BhgR6DTCRhZlI+RRfntfSlERERElCKYiiUiIiIiSiAG2ERERERECcQAm4iIiIgogRhg\nExERERElEANsIiIiIqIEYoBNRERERJRAgiiKYntfBBERERFRR8EMNhERERFRAjHAJiIiIiJKIAbY\nREREREQJxACbiIiIiCiBGGATERERESUQA2wiIiIiogRigE1ERERElEAMsImIiIiIEuisDLAPHDiA\nmTNnori4GDNmzMDu3btDHrd+/XpMnToVxcXFWLBgAcrLy9v4SjuGnTt3YtasWRg5ciQuuugivPnm\nmyGPW7BgAYYOHYrhw4fL/1FsVq9ejcGDB6vGcOfOnUHH8b3dcuvWrVON8/DhwzFgwAA89NBDQcfy\nvR2/vXv3YuLEifLtmpoa3H777Rg5ciQmT56MtWvXhn1utHM9BWjHu6ysDLfddhvGjh2LCRMmYPHi\nxXC5XCGfG+38Q37asf72228xcOBA1fitWLEi5HP53o6NcqxPnjwZNHcPGjQIl156acjnxv2+Fs8y\nDodDvOCCC8TXX39ddLlc4tq1a8Vx48aJ9fX1quO+++47ccSIEeLu3btFu90uPvjgg+LNN9/cTled\nuqqrq8XRo0eL69atE71er7hv3z5x9OjR4tatW4OOnThxorh37952uMqO45577hH//ve/RzyG7+3W\nsXXrVnHChAniqVOngh7jezt2Pp9PXLt2rThy5EhxzJgx8v133HGHeN9994kOh0Pcs2ePOGbMGPGb\nb74Jen60cz35hRvvG2+8UXzsscdEh8Mhnj59Wpw1a5b41FNPhTxHNPMPhR/rt956S7zllluafT7f\n29ELN9ZKp0+fFidMmCBu2bIl5OPxvq/Pugz2l19+CZ1Oh9mzZ8NoNGLmzJno3LkztmzZojru3//+\nN6ZOnYphw4bBYrHgvvvuw+eff85MX4xOnjyJSZMmYdq0adDpdBg0aBDGjh2LXbt2qY6rqKhAZWUl\n+vfv305X2jF89913GDhwYMRj+N5OvIaGBtx///149NFH0bVrV9VjfG/HZ8WKFVizZg0WLlwo39fQ\n0ICNGzfizjvvhNlsxtChQ3HVVVfhvffeC3p+tHM9+YUab5fLBavViltvvRVmsxl5eXmYNm0avvnm\nm5DniGb+odBjDfiz0gMGDGj2+XxvRy/cWCs98sgjuPzyy3HhhReGfDze9/VZF2CXlpaisLBQdV/f\nvn1RUlKiuq+kpAT9+vWTb2dnZyMzMxOlpaVtcp0dxcCBA/HXv/5Vvl1TU4OdO3cGTSIHDhxAWloa\nFixYgHHjxuGGG24IO4lTaHa7HaWlpVizZg0mTJiAyy+/HG+//XbQcXxvJ97f//539O/fHxdddFHQ\nY3xvx+eXv/wl3n//fQwZMkS+7+jRozAYDOjVq5d8X6j5G4h+rie/UONtMpmwcuVK5OXlyfdt3rw5\nZBAY7fxDocca8Adyu3btwpQpUzB58mQ88cQTIctx+N6OXrixlnzxxRfYtWsXfv/734d8vCXv67Mu\nwG5sbITValXdZ7FY4HA4VPfZ7XZYLBbVfVarFXa7vdWvsaOqq6vDwoULMWjQIEyZMkX1mNPpRHFx\nMf74xz/is88+w/Tp0zF//nycOXOmna429ZSXl2PkyJH41a9+hc2bN2Px4sV4/PHHg7IafG8nVkND\nA1577TX87ne/C/k439vxyc/PhyAIqvsaGxuD3ruh5m/p2GjmevILNd5KoihiyZIlKCkpwYIFC4Ie\nj3b+ofBjnZ2djSlTpmD9+vV49dVXsX37dvztb38LOo7v7eg1975euXIl5s2bh7S0tJCPt+R9fdYF\n2FarNehN6HA4YLPZVPeFC7q1x1F0jh8/jhtuuAGZmZlYvnw5dDr1W++iiy7CypUrce6558JkMmH2\n7Nno1q0btm/f3k5XnHp69eqF1157DZMmTYLJZMKoUaMwY8YMbNq0SXUc39uJtXHjRnTv3h3FxcUh\nH+d7O3GsViucTqfqvlDzt3RsNHM9Nc/hcOCuu+7C559/jldffRW5ublBx0Q7/1B4K1aswNy5c2Gz\n2dCrVy8sWLAAn3zySdBxfG8nxqlTp/DVV19h5syZYY9pyfv6rAuwCwoKgr4KLy0tVX1lDgCFhYWq\n4yorK1FTUxP0tQw1b//+/bjuuuswceJEPP/880EZKAD46KOP8OGHH6ruczqdMJvNbXWZKW///v1Y\nuXKl6j6n0wmTyaS6j+/txNq8eTMuv/zysI/zvZ04ffr0gdvtxsmTJ+X7Qs3fQPRzPUVWXV2NG2+8\nEdXV1XjrrbdU5TlK0c4/FFpNTQ2eeOIJ1NfXy/eFmyf43k6MzZs3Y8yYMcjJyQl7TEve12ddgD1+\n/Hi4XC68+uqrcLvdePvtt1FeXq5qlQMAV111FTZs2ICdO3fC6XTiqaeewoUXXojs7Ox2uvLUVF5e\njptvvhlz587FAw88EJS5ljQ2NuJPf/oTDh8+DLfbjb///e9wOByYMGFCG19x6rLZbFi+fDk++ugj\n+Hw+fPHFF/jggw9wzTXXqI7jezux9uzZEzZ7DfC9nUjp6emYOnUqli5dCrvdjr1792L9+vWYNm1a\n0LHRzvUUniiKuOOOO9C5c2esXr0aWVlZYY+Ndv6h0DIyMvDJJ59g+fLlcLvdOHr0KFasWIFrr702\n6Fi+txOjubkbaOH7Oua+Ix3Ad999J15//fVicXGxOGPGDLnF00MPPSQ+9NBD8nEffPCBeMkll4jD\nhw8X58+fL5aXl7fXJaesF154Qezfv79YXFys+u+pp54KGu8VK1aIkyZNEocNGyb+6le/Eg8ePNiO\nV56aNm3aJF511VXisGHDxEsuuUT8z3/+I4oi39utxePxiEVFReLhw4dV9/O9nThffvmlqr1WVVWV\neOedd4qjR48WJ02aJK5du1Z+7P333xevuOIK+Xa4uZ7CU473119/Lfbv318cMmSIav6ePXu2KIrB\n4x1u/qHQtO/tH374QZwzZ444YsQI8fzzzxeXLVsm+nw+URT53m4p7ViLoij++te/Ft94442gYxP1\nvhZEURQT9I8BIiIiIqKz3llXIkJERERE1JoYYBMRERERJRADbCIiIiKiBGKATURERESUQAywiYiI\niIgSiAE2EREREVECGdr7AoiIKLL7778f7777btjHx4wZgx07dmDv3r3cIZKIKAmwDzYRUZKrq6uD\nw+EAAHzzzTe444478N///lfertdqtcJutyMvL689L5OIiJowg01ElOQyMjKQkZEBAMjMzAQAdO7c\nWZWtTk9Pb5drIyKiYKzBJiJKcdu3b0dRURGcTicAoKioCB9++CGmTZuGoUOHYt68eTh16hQWLVqE\n4uJiXHLJJfjyyy/l5//888+48847MXz4cFxwwQV49NFH0dDQ0F6/DhFRymOATUTUAS1duhQPP/ww\nXnvtNezfvx/Tp0/HwIED8a9//Qv9+vXDY489BgAQRRG/+93vYDQasXbtWixfvhwHDx7Egw8+2M6/\nARFR6mKJCBFRB/Sb3/wGo0ePBgCMGzcOx48fx7x58wAA119/PRYuXAiv14sdO3agtLQUb7zxBoxG\nIwDgL3/5Cy677DKUlZWha9eu7fY7EBGlKgbYREQdUO/eveWfrVYrevXqJd+2WCzw+XzweDw4cuQI\n6uvrMWbMmKBzlJaWMsAmIooDA2wiog7IYFBP7zpd6IpAj8eD3r17Y9WqVUGPsSsJEVF8WINNRHQW\nKywsRFlZGTIyMtCnTx/06dMHHo8Hjz/+OOrr69v78oiIUhIDbCKis9iECRNQWFiIe+65B/v378e+\nffuwaNEiVFVVIT8/v70vj4goJTHAJiI6i+l0Ojz//PNIT0/HjTfeiHnz5uGcc87B8uXL2/vSiIhS\nFndyJCIiIiJKIGawiYiIiIgSiAE2EREREVECMcAmIiIiIkogBthERERERAnEAJuIiIiIKIEYYBMR\nERERJRADbCIiIiKiBGKATURERESUQAywiYiIiIgSiAE2EREREVECMcAmIiIiIkogBthERERERAnE\nAJuIiIiIKIEYYBMRERERJRADbCIiIiKiBGKATURERESUQAywiYiIiIgSiAE2EREREVECMcAmIkpx\noii29yUQEZECA2wiSmlTpkxBUVERnn322ZCPr1+/HkVFRbjuuusS+rrPPvssJkyYkNBz3n///TFd\np8vlwmOPPYZt27a16HVPnDiBoqIifPbZZ0GPNTQ0oLi4GI8++mjY5//hD3/AxRdf3KJruPDCC/H0\n009Hffx9992HX/3qV2Ef93g8KCoqwtq1a1t0XURE8WCATUQpTxAEbNy4MeRjGzZsaJXXnDVrFlau\nXNkq547W6dOn8cYbb8Dr9bbaa6SlpWHq1Kn45JNP4PP5gh53Op3YuHEjZsyY0aLXWbFiRcSAmYgo\nlTDAJqKUV1xcjIMHD+LEiROq+x0OBz7//HP0798/4a/ZtWtXDBo0KOHnTUYzZsxAeXk5duzYEfTY\np59+ivr6ekyfPr1Fr3Heeeeha9euLToHEVGyYIBNRClvxIgRyM3NDcpif/7558jOzg4KhGtra/HY\nY4/hwgsvxODBgzFx4kT8+c9/htvtBgBs374dRUVF2LVrF2bOnIkhQ4bg0ksvVZ1fWyIilSPMmzcP\nQ4cOxeWXX47//Oc/8uPvvPMOioqK4HQ65fuOHDmCoqIibN++PeTvVVZWhkWLFuH888/HoEGDMGXK\nFLzwwgsA/GUdU6dOBQDMnz8f999/PwB/PfbKlSsxZcoUDBkyBNdee23Q+Q8fPow5c+aguLgYV155\nJfbt2xdxfCdMmIDOnTvjo48+Cnrsgw8+wPDhw9G7d28A/oz2k08+ialTp2Lw4MEYN24cHnjgAdTX\n1wMAjh49iqKiIrz66qu44IILMGbMGBw/fjyoRGTnzp347W9/ixEjRmDIkCGYMWMGNm/eHPT6f/vb\n3zB27FiMHTsWixcvhsPhCPt7/PDDD7jppptQXFyMcePGYcmSJaq/DyKiRGGATUQpT6fTYcqUKdi0\naZPq/g0bNuDSSy8NOv6ee+7Btm3b8MADD2DVqlW49tpr8corr+Ddd99VHXfffffh6quvxosvvoju\n3bvjnnvuQXV1ddjrePzxx9GlSxcsX74cQ4cOxd13340vvvgirt/J5/Ph5ptvxo8//ojFixdj1apV\nOP/88/HMM89g69atyM/Px/LlywEADzzwAG677TYAwNNPP41nn30W119/PZ577jn07dsXN998M/bv\n3w8AqKurw5w5c+B0OvH000/j+uuvxx//+MeI16LX63HllVcGlYnU19fj008/xdVXXy3ft3jxYrz3\n3nu44447sHr1asybNw/r1q3DSy+9pDrnypUr8fDDD+OPf/wjevXqpXrs2LFjmDt3Lrp27YrnnnsO\nzzzzDKxWK+699145UAeAPXv2YOPGjfjTn/6Eu+66C++8807YWvGysjLceOONcDgceOqpp3Dvvffi\n3//+Nx544IFm/iaIiGJnaO8LICJKhIsvvhi33norqqqqkJ2dDbfbjc2bN2PVqlV466235OMcDge8\nXi8ee+wxjBs3DgAwfvx4fPrpp/j6669Viwznz58v1wV369YNl112GbZv3x4yaAf8ZQ5/+ctfAPgX\n7R05cgQvvfQSxo8fH/PvU1ZWhuzsbDz22GMoKCiQr/Pjjz/G119/jQkTJmDgwIEAgIKCAvTu3RvV\n1dV4+eWXcccdd+CWW26Rr+PXv/41nnvuOTz//PN49913UV9fj+effx45OTkA/AsCn3jiiYjXM2PG\nDLzyyivYsWOHPG4bN26Ez+fD5ZdfLh9XXV2NBx98EFdccQUAYOzYsdi5cye+/vpr1flmzZoVdmHk\n4cOHMW7cOPzlL3+BIAgAgLy8PMyaNQsHDx7EqFGjAPj/YbVq1Sp06dIFgD97v2TJEtx7773Izs5W\nnfPll1+GwWDAqlWrYLPZAPjLfG6++WYsXLiwVcqIiOjsxQw2EXUI48ePh9VqlcsIvvjiC1itVhQX\nF6uOs1gsePnllzF27FgcO3YMW7ZswcqVK1FRUSGXiEiGDRsm/yzVB9vt9rDXIAWVkilTpmDXrl1x\n/T7du3fHq6++ij59+qCkpASbNm3C8uXL4fF4gq5TsmfPHrhcLlxwwQXweDzyfxMnTpTLRHbt2oXB\ngwfLwTWAqDqADBo0CIWFhaoykfXr1+MXv/gFMjMz5fuWL1+OK664AidPnsTWrVvx8ssvo6SkJOia\n+/btG/a1pkyZglWrVsHhcGD//v1Yv349/vnPfwKA6jxDhw6Vg2sAmDx5Mnw+H/bs2RN0zq+++gqj\nRo2CyWSSx2Xs2LEwGo1hS3SIiOLFDDYRdQgmkwmTJk3Cxo0bce2112LDhg24+OKL5QyoklRWcPLk\nSXTu3BnFxcUwm81B/aTNZrP8s07nz0eE6qQhycvLU93OyclBfX19xOdE8tZbb+Hpp59GVVUVunXr\nhpEjR8JgMITtey2VryhLNpTsdjtqa2uDsru5ublRXc+MGTOwZs0aPPzww6iursYXX3yBZcuWqY75\n6quv8PDDD6OkpARZWVkYMmQIrFZr0DVHek2Px4PFixfjX//6F3w+HwoKClBUVARA3fNbew7p96qr\nqws6Z3V1Nfbv3x+yjvzMmTPN/OZERLFhgE1EHcbFF1+M+++/Hw0NDfjvf/+Lp556KuiYH3/8Eb//\n/e8xe/ZszJ8/Xw6KZ82a1eLXr6mpUd2uqKhAVlYWdDqdHOgrW+o1NjaGPdeOHTvwyCOP4N5778V1\n110nZ4nPP//8sM/p1KkTAGD16tWqrLLEZDIhMzMTZWVlEa87nGnTpuHpp5/GV199hZKSEqSnp2PS\npEny49XV1bj11lsxadIkrF69Gt27dwcA3HHHHSgvL4/qNQDgueeewwcffIDly5dj/PjxMJvNOHTo\nENavX686rra2VnW7oqICAFTZeUlGRgYuv/xy3HTTTUGPaf9hRETUUiwRIaIO48ILL4Qoinj++efh\n8/kwevTooGMOHDgAt9uNBQsWyIFVeXk5vv/++7gzzZItW7aobm/atEmuF5bqfpXBrbYuWWn37t0w\nm82YP3++HCwfPHgQFRUV8nXq9XrVc4YOHQqDwYCamhoMGTJE/m/r1q345z//Cb1ej9GjR+Pbb79V\nXcfnn38e1e/XvXt3jB49Gps2bcLGjRtxxRVXwGg0yo8fOXIEdXV1mDdvnhxcNzY24ptvvolpt8nd\nu3dj1KhRmDx5svwtgrSZjvLvaO/evapFj5988gmMRiOGDh0adM7hw4ejpKQEgwYNksclLy8PS5cu\nxbFjx6K+NiKiaDCDTUQdRlpaGs4//3y88soruPrqq4MCUAAYMGAA9Ho9Hn/8cfzyl7/E6dOnsWLF\nCjidzoj11dH45JNP8MQTT+D888/HunXr8P3338tdLcaMGQOj0YglS5Zg/vz5KCkpwZo1a8Kea8iQ\nIXA4HHjiiSfwi1/8Aj/++COWL18OQRDk68zIyADgD5B79uyJgoIC3HDDDXj00UdRXl6OoqIi7Nix\nAy+88ALuuusuAMA111yD1atX45ZbbsGdd96J8vLysLtghjJ9+nQ899xzqKiowJ133ql6rLCwEDab\nDcuWLcPcuXNRU1OD1atXo7y8PGRWOZzBgwdjzZo1WLt2LXr37o3t27dj1apVANQ18F6vF7feeisW\nLFiAI0eOYNmyZfjNb36D7OxseDwe1TnnzZuH999/H7fffjtuuOEGuFwuPPvss6iurpbLT4iIEoUZ\nbCLqUC6++GK43e6wC/cKCgrw5z//Gbt378Ytt9yCZ599FlOnTsXChQvx7bfftmhXxFtvvRX79u3D\n7bffjiNHjmDVqlVyNjU7OxvPPPMMTp06hfnz52PdunVB9ctK48ePx6JFi/Dhhx9i/vz5ePnllzFn\nzhxcc8018iK+9PR0zJs3D2+99ZbcBeTBBx/EnDlz8Morr2D+/Pn44IMP8Ic//AELFy4EAFitVrzy\nyivo2rUr7rvvPqxevTriNuhal112GSorK9G9e3fVIlAAyMrKwjPPPIOTJ09iwYIFeOKJJzBo0CA8\n+OCDKCkpCVkbHcrChQtx2WWX4cknn8Ttt9+OrVu3Yvny5ejRowd2794tHzdp0iQMHjwYd911F1as\nWIHf/va3WLRoUchz9u7dG6+99hocDgfuuusuuT3gmjVrQpbTEBG1hCDG8r0dERGFVFRUhEcffZTb\nfRMRETPYRERERESJxACbiIiIiCiBWCJCRERERJRAzGATERERESUQA2wiIiIiogRK2j7YZ85E184p\n0QRBQG5uGioqGmLaGIFix7FuWxzvtsOxbjsc67bF8W47HOu2E+1Y5+VlRH1OZrA1dDr/QOs4Mq2O\nY922ON5th2PddjjWbYvj3XY41m2nNcaaf21ERERERAnEAJuIiIiIKIGiCrA//PBDXH755Rg+fDiu\nvPJKbNy4EQBQU1OD22+/HSNHjsTkyZOxdu1a+TmiKGLp0qUYN24cRo8ejSVLlrRoC2IiIiIiolTQ\n7CLH0tJSPPjgg3jppZcwYsQIbNu2Dbfccgs+++wzPProo7DZbNi2bRsOHTqE+fPn49xzz0VxcTFe\nf/11fPrpp1i3bh0EQcCCBQvw0ksvYf78+W3xexERERERtYtmA+y+ffti69atSEtLg8fjQXl5OdLS\n0mAymbBx40Z8/PHHMJvNGDp0KK666iq89957KC4uxvvvv485c+YgPz8fALBgwQIsW7YsJQJsr0/E\nF/vL0Ds/A11zbHGd46cz9dh9uDzofr1Oh1ED8tA504ryGjt2HjwDr8+HLtk2jCzKgyAIAACX24ut\n355Co9PTot8lHiajHhk2I/r3zEJOJ0ubvz4RdRx2pwdf7i8LOZf17pKBIQW5QfdX1jrw3dEqjB6Q\nD5NR36rX1+Bw46vvTqPB4W72WJNRj042E/r1yERuJudGIgovqjZ9aWlpOH78OC699FL4fD48+uij\nOHbsGAwGA3r16iUf17dvX2zYsAEAUFJSgn79+qkeKy0thSiKchAZSXutnNXpBHx98GeseG8/+nTJ\nwP/ePCau8zz7zrc4XWUP+dj+Hyvx/80ejlc/PoRvSyrl+x/+7SgU9sgEAHzyxXH8a0tJXK+dKAKA\n8/rm4LKxvTGkICeqv7dY6HSC6k9qXRzvtsOxDti06wTeCTOXCQD+evv5yMuyqu5/Y+P32PV9Odwe\nH6aO6hnx/PGOtc8nYu3mw9j49Qm43L6YngsAA/tk45LRvTC8f+eEz43JjO/ttsOxbjutMdZR98Hu\n1q0b9uzZg507d+K2227DTTfdBItF/S94i8UCh8MBALDb7arHrVYrfD4fXC4XzGZzs6+Xm5vWs1e5\nBgAAIABJREFUbpNWxYHTAIA6uws5OelxnaOy1j8O3XLTYLP6h7m6zomKGgfq7W7k5KSjzq7O6Dh9\nkF/vmx8qAAC5mRZkZTQ/XokiioDD6UFFrQNOlxf7Syuxv7QSRX2yMfuSARiuyLInSlZWWkLPR5Fx\nvNsOxxryPJduNaJLrv8bQVEESn6qgQjAJQpB86z0nEa3L+o5ONaxfm/LEXz45TEAgNmkR8/8yK8j\niv5sfGXT3Pjd0Sp8d7QK/XpmYvalAzBqYJezKtDme7vtcKzbTiLHOuoA22DwHzp+/Hhccskl2Ldv\nH5xOp+oYh8MBm80/gVosFtXjdrsdBoMhquAaACoqGtotg+3x+puMe7w+VFbWx3wOr88nn+M3l/bH\noL45AICPth/DPzf+ALfHi8rKerg96kWflVWNqKysR0WNAyUnawAAN105UH5+W3J7fPjmhzPYsOM4\nfjhRg0NHq/DIqi9Q2KMTrrmgAIMTkNHW6QRkZaWhuroBPh+b6Lc2jnfb4VgHVNf5kw0j+nfGTVed\nB8C/CP6mxzfD6xPx8+k69MhWJ2tcbv/cWFvvaHYOjmesz1Tb8ep/DgAARg3Iw9wrBiLdaozquR6v\nD3sOV2DjzuM48GMVDp+owf+u3o6C7p1w7YWJmRuTGd/bbYdj3XaiHetYkq7NBthbtmzByy+/jH/8\n4x/yfW63G71798Znn32GkydPonv37gD8CyKlspDCwkKUlpZi2LBh8mMFBQVRX5goimivpiMer//r\nQp8P8Hpjf1PbnYEL1+uEwDma/vA2nVf7l2h3euD1itj1/RkAgNWsR78emXFdQ0vpBAEj++djxLl5\nOHC0Cu9/XorDP9XgyE+1ePLN3Sjs0QkzJvbFoHNa/mHi84nt8juerTjebYdjDdgd/my0yahXjYXN\nYkBdoxv1dnfQGEm3XW5f1OMX7Vj7fCJWr/8OLrcPnWxG/M+lA2A1GaJ+HQECivt1RnG/zjh0rArv\nfl6K749Xo+Skf27s1yMTV1/QFwP7ZHfoQJvv7bbDsW47iRzrZnPE5513Hvbt24f33nsPPp8PW7Zs\nwZYtW3D99ddj6tSpWLp0Kex2O/bu3Yv169dj2rRpAIDp06dj9erVKCsrQ3l5OV588UXMmDEjIRfd\n2gIBdnyD7PYE6vlMhsACHam2R2w6r/b8zqasze4f/IsjB/fNhUHfvq3KBUHAoHNy8MCNI3Dv9cUo\n7NEJAHDkp1o89dYe/OW1XdhfWsltXIkoJIfLP69ZTOp8js3svx1q8aM0n2i/5UuEdz4rwXdHqwAA\nsy/uH3XmOpSi3tn4w+zhuO+GYvRrWj9z+KcaPPnmbjzxxjc4dKwqIddMRKmn2Qx2Xl4eVqxYgT//\n+c/43//9X5xzzjl47rnnUFhYiMWLF+ORRx7BpEmTYLPZsGjRIjljPXv2bJSXl2PmzJlwu92YNm0a\n5s6d2+q/UCJ4mgJkX5xBo/T1JgCYjIEAWaqdl86rPb/T5YXd6ZEn/+JzO8f1+q1BEAQM6puD887J\nxoEfq/De/5XgyE+1OPxTDZa+5c/azJjYF+ed07GzNkQUG4fLH0BbzepuIDaL/+NHynAreX1SgB37\n4sNIvjxQhg+/PAoAmFTcHaMH5Lf4nIIg4LxzcjCwTzb2l1bi3c9LUXqqFt8fr8YTb3yDwh6d8Ivh\nPTB6QD6MhtbtiEJEySOqGuxRo0bhnXfeCbo/KysLy5YtC/kcvV6Pu+++G3fffXfLrrAduL0tDLDD\nZLCFpghbDrA1nx0utw8/nKiG1ydCEBCyfVV7Uwba+3+sxPufl+LIyUCg3TXHhvGDu2L4uZ3Ro3P7\nLVQlouQglczFksH2iYkPsH8sq8XLHx4EAPTvmYlfX9w/ofOTIAgYXJCLQX1zsPdIBd77v1IcLavD\nkZ9qceSnWvxz4w+YMKQbRg3IR0G3TuwMQdTBRb3I8WwiLVDUBsDRcim+1jSqMthNAbYvTAbb7UVd\no78Xa4bV2KKvLlubIAgY3DcXg87JUQXaZZWNePezErz7WQmyM8wo7JGJPl3SkZdlRX62FXlZVtjM\nBgbeRGcJKYNtMamzt9ZIAbaUwfYmJsCuqXfi2X99C7fHh9xOZtx2zZBWK78TBAHD+nXG0MJc7C+t\nxOZvfsLuw+VocHiw4avj2PDVcaRbjejXIxPndMtAl2wbuuRYkZ9lk7P6RJT6+P/mEKQa7HjripU9\nVc3KGmwpwG46bagAO1y9YrJSBtolp2qxbV8Zvj50BrUNLlTVObHz4GnsPHha9RyzUY/MdBOyM8zI\nz0mDzaRDToYFPfPS0KdrBmyW5P2HBRFFTxRFeU6zajPYEUpEpLnRk6AM9tufHkFVnRMmgw53/HIo\nOqWZEnLeSKSM9uCCXFTWOvDZnpP48sDPOF1lR73djd2Hy4M2I7Oa9chKNyMzzYSsDDOy0s3I7WRB\n985p6N0lHWmcG4lSRmpEcW1MrsGOc5Fj2Ax204/SeaXFjnqdAK9PbAqw/R82ZlNq1eoJgoDC7pko\n7O7/6vVoWR0OHatGyckanKpoxJlqu1w643R7cbrKjtNVdhw6Vq0+D/y7uw0tzMWYgfnokRdfH3Ii\nan8er0+up9ZmsG1mf7AYKoOdyBpsj9eHXU0Lx6++oAC9u2S0+JyxyulkwdUXFGDGxL4oq2zEd0f9\n7f1OnGnA6epGOSljd3phdzbiVEVj0DkEAD3y0jG4IAejivLRt1sGvwkkSmIMsEOQM9hA1DtPKrmb\nJkuDXpCz1kAggy3Kixz991vNBtTb3XC5fYoMdmoF2Eo6QUDfbp3Qt1sn+T5RFFFd78KZajuq652o\nqXehttGFRpcXP1c04OdKOypqHRABHP25Dkd/rsO/t/2Igu6dcNGonhg9IB/69miMTkRxs7sCyQap\nJES+3ZTBbgyRwRYTGGD/cKIG9qYgflRRXovP1xKCIKBbbhq65aZhygj/DpXS3Hi6qhHV9S5U1zvl\nObKqzokzNXZU1johAjhxph4nztTjo+3H0C3XhikjemLi0G4wt/J28kQUOwbYIXgUdX8+UYQ+xgDb\n2ZTBNmlWjOuCFjkGMjv1djecbi+cKVYiEi1BEJCdYUa2YldKvd6/g1tlZT28XhGNDjd+OFGD/T9W\n4utDZ1BV50TJyVqsXHcA/976I2ZOKkTxuWfXtsREqcyhyE4HZ7ClGmx30PO8CazB3tNUhtEjLw2d\nNVuyJ4NQc6NWo8ONIydr8d3RKuw6dAanq+04VdGI1z/5Huu3/YhpE87BhcO6t3tbVyIK6FhRXIJ4\nFE3GfT4g1jlLymAry0MA5SLHpnOLUoDt/2tQ1mCnWolIItgsRgzr1xnD+nXGDVPPxYEfK7Fx5wns\nPVKBUxWNePadb9GvZyaum9wP/XpmtvflElEzHIoMdrgAW7kxlyRRXUREUZT3FSjulzxtT2Nlsxgx\npCAXQwpyMWtyIUpO1WLzrp+w/cDPqGlw4bUN3+PjHcdwzQUFGHve2bVlO1GyYoAdgjaDHSup1tis\nyWALYfpgS/1hXYoa7FQuEUkEXdPiycF9c1F6qhZvf3pErlv882tfY/ygrrjuF4XITA+f9SGi9qUK\nsCOUiGhL8aQkREsD7FMVjThdbQeQ2gG2knK9y/SJffH+5yX4cv/POFPtwMp/H8B/v/kJN17cv11q\nzYkogN8nhaCc1ONZ6CgtcgyfwVa3AZRqE51uLxxNm9RYWFMn69utE+67oRh3XzcMPfPSAABf7C/D\ng6u+xIavjsMbbz9FImpVUu2zIAAmg3o+lDLYPlGUd7GVJGqR454j/ux1J5sRfbt3aubo1JOfZcX8\naYPw2LwxGFro3zfh8IkaPPaPr/DahkNocASX3xBR22CAHYIygx1Pqz6pRERbgy1tNCOdUhTVq+ud\nLkWbPjMDbCVBEDCkIBePzB2NX1/cH1azAXanF29u+gGPvvwVtyQmSkLKtqPasgVlz2ftQkd5q/QW\n1mBLu+IO6pujWnDe0fTMT8fvZw3D72cNRX6WFaII/HfXT3jgxS/x2Z6TcW+aRkTxY4AdgrpEJPbn\nBxY5hslgBy1ylDLYvg67yDFR9Dodpo7sib/cMg4Th3QDAPx0pgFPvPENVq7bj6o6ZztfIRFJwm2T\nDgQy2EAg0y1RZrDj3Y/A4/Xhh+M1AIABvbPjOkeqGVrYGYtvHoNrLyyAyaBDvd2Nf/znIP605muU\nnqpt78sjOqswwA5BublBPP/ylzPYRm0XkcDPPp8I6czSh4/H65M/aNh2KbJOaSbMu3IgHvzNSPTu\n4u+V/eWBn/Hgqi/x0fZjqn8kEVH7CLdNOqBu26fsha2dc5WLzmNxtKxOLj0Z0OfsCLABwGjQ46rz\nz8Gf5o+T2xKWnqrFkld24h//OYi6Rlc7XyHR2YEBdgiqEpEW1GCHy2ADUNUNK3c4q23wT35n+yLH\naPXrkYmH54zGby7pjzSLAU6XF/9v82E88tIOfHmgjPXZRO1IzmCHmM8sJr288FtZIqJd9xJvHfbB\nprKx3E5mdM60xHWOVJabacFt1wzBvdcXo1uuDSKAz/acxAMvfon3/68U9XbWZxO1JgbYIaja9MWR\nPJG6iIRb5Kh9DeXqeum5DLCjp9MJ+MWInvjTLeNw4TB/2cipikasXHcAD678Epu/+QluT3ArMCJq\nXZE2zhIEQdELO0KAHee3UQebdokd0Dv7rG5bN6hvDh6bNwbX/aIfzCY9Gp0evP9/pVj0wjb8v/8e\nZlkdUSthoW8IqhrseDLYYRY5ShvNaF8jdHaHfzWx6mQz4beXD8Sk4h7499YfsftwOc5UO/Dqx4fw\nr0+PYPTAfIwf1BXn9sw86z5wfT4RHp8P9XY3auqdcLl9cHt98HhFeDw+eHw+f9mSYgGuKDb9icCu\nphClf3T6HxcEf6Ak/amD5rbiZ0EI/CNT+ln7mOp80N5WnzvoPvl4yH+/ynNS2wu0HQ09n9ksBjQ4\nPKoabG9QBju6fxx7fT58vvcUPvjiKPQ6ARU1DgBA0VlSfx2JQa/DZWN7Y9ygLvh4xzF8+s1JOF1e\nfLTjGD7+6hgG9c3B+YO7YsS5eUGljR2dKIr+edDra/ov+GevT5r7RPk50vzoAwDlXCnGNzcq5zDp\nMYNBh0aPiNpaO0SfGNfcCCjnQfV90BxHicUoLoSW98GOpkQkcF7tFsIAM9gt0bdbJ9w5cyhOnK7H\nf7YfxfYDp9Ho9GDL7pPYsvskOmdaMKywMwaek40BvbNgsxjb+5LDEkURDpcXdqcHjQ4PGpv+tDul\nn93yfco/7U1/utxeuL0+sImA/4MEqiBfgE7w3xfXB0wzYyoICBp3sbknxfE6baFrrg33/3pEzP/w\nb64rkjT3KUtEtIsaoy0RefKfu3Hgx+BuQgN6Z0X1/LNBVroZ1085F1eOPwcbdx7Hpq9PoMHhwb6S\nSuwrqYTFpMegvjk475wcDDonG/nZtva+5LBEUYTb4wsx/7nl+S9oXlT87HR74fH4gv5BdzYKNTdq\nA/JmRTGMUc1/URxiNRtwy7TzMPCcnOYPbkcMsENorUWOgiLeVgbxoYLps3Enx0TrmZ+O+dMG4ZeT\nCvHlgZ/xxb4y/FTegPIaBzbtOoFNu05AEIBe+enolZeOHnnp6JmXhrxsK7LTzQnJ5PhEEU6XVzHR\nu0MEyeqgWPqQ8B/jTboWW9JkLEBqOxlX2NjmmhLv8MrjmQpXnRyO/VyPIz/VYlDf2D7QIi1yBBCy\nRCQ4g918gO1ye+XgelhhLnQ6Ad/8UI6iXllJuT16e0u3GnH1BQW4Ylwf7D5cjm37yrCvpBIOlxdf\nHzqDrw+dAQDkdDKjd34GeuanoWdeOrpk29A5y4KsrJb/f0cURbg8Ps385w4KigOPB8+d8S6AbU2C\n/D/B/8BOVqk2N7o8Lhw/Xc8AOxWpt0pvpQy2sgY7xIcPM9iJk9PJgivG9cHlY3vj+Ol6fHXwNA4e\nrULpqTr4RBHHfq7HsZ/rg55nMxuQmW6CzWyA2aSH2aiHsenvtKlaAmj6OtDp8cLp8v/ncAf+dLm8\nCZ+qdIIAm8Xg/89sgNUc+DnwpxE2swEmox5GgwC9XgezUYec7DQ0NjghCAKMegEGva7pPwE6nRCy\nzEL+2hHhsxnar0dFUYRPjPF20/PljZjEwPiqzg3pOcqfA38qnxvu61sxxH3xCJfc0ekEpKdbUF/v\nCDGHxJ4tb89vcJ9751uICA58o9HczrTSt0eqRY6alwkVYFfVOZGZZpLL7mrqA50xZk4uRI+8dNTb\n3UFzMKmZjHqMGdgFYwZ2QU29E19/fwb7Sytx8FgV7E4vKmudqKx1YvfhctXzDHodstJNsJoNsJr0\nsJgNMBp0Qe9sEf6SSafLA6fb1zQ3euSWtIlOHghA0HwYuG1U3W826uW5T54HDeo5UfpZr1OXVeia\nomjlXBlufpTmF588z0Q/N+p0AjIzbaiqboDH41MfB2XZnlS6otkpOsI82TRFhngs+L6oxz+qear5\ng5o7j81sQP9eyf/NFAPsENwt7IMtLVQMatOnWuQYeA2jwf9/YOUHGGuwE08QBPTukiFvIdzo8OD7\n49UoOVWLn87U46czDThTbZcD4samLEmi6XVChODYCGtQsKwNmnVxlTTo9QJyctJRWVmv+gdeIkgf\nMO0aCSaR1hzrtqZrmpviSTZIJSKhyuCAQAZbWYPdXBeRvUfK8czavZgwuCtuuuo8AEB1vUN+PCPN\nBMCfpaXoZaabMWVET0wZ0RNenw+lp+pQcrIWJ07X4/iZepwsb5D/LjxeH8prHM2cMXaCANVcGGqe\nDJ4fjYGg2aRPug2F5DUmcfzD2j+P2GCAL+XnkbMRo7gQWtymr6n3qlGbwVYtcgycVxD8fa+VwRwz\n2K3PZjGg+NzOKD63s3yf0+1FVZ0TVXVOVNc7UVPvgsPlr9dzurz+DxipPKLpr1OA/+/PbNLD0pTp\nNpv0sJr8GWTth4LJEF+ATNQe5AA7jmyjFDiHm8/kGuwYuogcOu7vDnL4ZGDjFCmDLQhAehKvqUgV\nep0O/Xpkol+PTPk+n09EbaMLNQ0uuEUBJ07VoMHhhsPlhcPphdsbejGqyRCYG01GPSxN86PZ6L8v\nzWKUM83+1o2cG6ljYIAdQktrsAMZbG2JiOI1FP2ZdTpBbp8EQP5qitqe2ahH1xwbuuYk7+IeorYk\n70Dbkgx2hC4igLpExCtGzmBLwXSjI9DHubqp1VyGzaRKZFDi6HQCstLNyM20+L+d6ZHBrCpRBIzi\nNERRVJVqxBVgR9GmTzkx6QRBVU7CXRyJKFlIO9DGMxdG6oMNhF7kqP3WMDjA9gfTDXaPXHMv3dfJ\nxuw1ESUHBtga2oU88WwEKPVtDc5gh67B1ukEmBXHsv6aiJJFvBlsqZcwEGmRY3ANdnNdRGqadrv1\nNbWwBIDq+kAGm4goGTDA1vBo6v1izdr4fKJcX63NYAtharB1gjprzfprIkoW0jdvsXYRkYJfQL1b\nrZKcwVZ1EYlcg12t6BgiPU8KsDPTGGATUXJggK2h7asZa9bGpdh1LGiRo6I00KvMYGtKRBhgE1Gy\nkALsWJMN9igWbUv3K7PdkbqIeLz+3UglDU112DV1zGATUXJhgK2hzWBrdxVrjlR/DQTXUofbyVEQ\nBGawiSgpxVsiospghyl7U65Lkc4faav02gaX6rGGpgy2tPCxUxprsIkoOTDA1tCuik5kBluIsgbb\nzBpsIkoScoAd4xpHaZMZALCG2SpdrwvMe1JgHVQioshgK8tDgEAnEakPNjPYRJQsGGBrBNVgx/h8\n5YdB0CJH1mATUYqRdrGLNdkgbZMu9YkPRTknyrtwRuiDLXULkTQ4PPCJoiKDzQCbiJIDA2yNoBKR\nWDPYihKRoDZ9YfpgCzpNmz4G2ESUJIQ4A2wpg22OsHmIIoEdyGBHqMGuCSoRcaPR4ZGf24kZbCJK\nEgywNYIWOcZag60oEYmUwdb2wWYGm4iSkT7ORY7NbZMOqNel+MIE2B5ViYg6g93o8KjqstkHm4iS\nBQNsjaASkRhrRCJnsMPUYAuCKhhnH2wiShZSXiDmALuZbdIBzSJHUf2nJHIGWx1gZ7BEhIiSBANs\njaCNZuLMYBv0QtCWveE3mtHUYHMnRyJKEi3tIhIpYaBXfqvXlM0I6iKiqsEOXuRY2+hqeh09d8El\noqTBAFvDG5TBjq8G22gInuhV9YYsESGiFKCLd5GjK94MdqQuIppFjna3nMFmBxEiSiYMsDUSVYNt\nMgQPrapNn0/Tps/ERY5ElHxaupNjxAA7ihrsUCUi2RlmAOoSEfbAJqJkwgBbo6VbpUsZbO0CRyDy\nIkf1To6swSai5BDYyTG250WzyFFdIhI5wPaJohxMd++cBkBa5Ojvhc1t0okomTDA1vBoJncxxkWO\n0oeBdoEjoK3BVu7kyD7YRJScpHkr1l1t7VEsclR+qyeG22imKelRb3fLQXj3XH+A3aCowWaJCBEl\nEwbYGi3OYEslIiEy2EDgw0oqEREEbpVORMlLSjLHXyKSmAy2coFjj7xABpubzBBRMmKArdHirdIj\nLHIEAgsdpcWUUsCtrLu2RPhKlYioLcW7k6O00Uy4bdIB7SJH//m1gbxLCrAbAgscu+bYAAAigGM/\n1wEAcjtZYro+IqLWxEhOo20y2KJcIiJ9wHTLtWFoYS4Meh3yMvlBQUTJIe6dHJ3NZ7BDLnLUzLme\npjnV6QosIM9QbCgjBeB9u2XEdH1ERK2JAbZGcIAd2/PlRY5hMtiCZkW+9AGjEwT8ftaw2F6MiKiV\n6aU+2DHv5Nh8DXYsJSLSn0aDDmlWdccQg16HnvnpMV0fEVFrYomIRtBGMzFG2O5oa7ClEhH+DRBR\nEou3i4g9ihpsZYmItIgyaCfHprlS+tNg0MGmKaMr6NEJBj0nUyJKHpyRNFrcB1vOYIcLsP1/SrXe\nyq9IiYiSTWAnx+hbKnl9PjnjbIlYg618TuQMtkfKYOt1MOh1qnUr/XpmRX1tRERtgQG2hrZERIx1\nkWOENn1AIGMjvY7AAJuIklhgJ8fonyN1EAEAa6w12Jo51+MV4RNFVYkIAKRZAuc9t1d29BdHRNQG\nGGBrBG2VHnMNtv+DxRimRETQlogwviaiJBbPTo5SD2yg+T7YUowdrosI4M9eSyUiRr0UYAfqsM/t\nxQw2ESWXqALsnTt3YtasWRg5ciQuuugivPnmmwCAmpoa3H777Rg5ciQmT56MtWvXys8RRRFLly7F\nuHHjMHr0aCxZsgRerzfcSyQNbYlIrJsrNJvBbvowkTa0ERhhE1ESi2ejGWUGu7m+/npNAB/qddze\nQMmJQZPBNhm5wJGIkk+zXURqampw22234aGHHsKVV16J7777DnPnzkXv3r3x5ptvwmazYdu2bTh0\n6BDmz5+Pc889F8XFxXj99dfx6aefYt26dRAEAQsWLMBLL72E+fPnt8XvFbdWb9MnfZho+mATESUj\nqU46lrlQVSLSTF9/nU4AvKJcgiIF2nqdIP/sDpHBtjVlsM/pmgE9FzgSUZJpdlY6efIkJk2ahGnT\npkGn02HQoEEYO3Ysdu3ahY0bN+LOO++E2WzG0KFDcdVVV+G9994DALz//vuYM2cO8vPzkZeXhwUL\nFuDdd99t9V+opYIWOcbaRaSZNn2BLiJc5EhEyU+ao2IpEXEoSkTMzWSwdZo2gNKca1Lsbuv2+ODx\n+O+XarAH9vHXXY8Z2CXq6yIiaivNZrAHDhyIv/71r/Ltmpoa7Ny5E0VFRTAYDOjVq5f8WN++fbFh\nwwYAQElJCfr166d6rLS0FKIoRrWwTxCEdmlhp/0QEQHo9dEHwVKJiMWkD/m8QD2jT74dy/k7Emks\ndCyTaRMc77bTkcZaan8nitHPhdI3eRaTXg6Iw5FKRESI0OsFSDOwxaSXa7l9ogiPL7DIUa8XcMmY\nXrhgWDe5J3ZHGOtU0JHe28mOY912WmOsY9popq6uDgsXLpSz2GvWrFE9brFY4HA4AAB2ux0WS2BH\nQqvVCp/PB5fLBbPZ3Oxr5eamtUuHDb3mw8BsMSInJ/r6PulrzOwsW8jnSR82oijIt2M5f0eUlZXW\n3pdwVuF4t52OMNbWpgBWH8NcpTNUAABsFkOzz5HKO2w2M3Jy0mFqKimxWQyoqvNvj261maGTFjfa\nTCHP2RHGOpVwvNsOx7rtJHKsow6wjx8/joULF6JXr1545plncOTIETidTtUxDocDNpsNgD/YVj5u\nt9thMBiiCq4BoKKioV0y2I12t/p2gwuVlfVRP9/VVHvodrpDPk9awON0e+TbsZy/I9HpBGRlpaG6\nuiHmUhyKHce77XSksXY3zVVOpyfquaqiqgGAv1SuuedIaZTaWjsqK+vR2OgCANXGMRWVDWhoul/0\n+VTn7EhjnQo43m2HY912oh3rWBKiUQXY+/fvx80334zp06fjD3/4A3Q6Hfr06QO3242TJ0+ie/fu\nAIDS0lK5LKSwsBClpaUYNmyY/FhBQUHUFyaKItqj6Yi0mYF82+eTN4Vpjs8nyhlsvV4I+TwhRA12\ntOfvqHw+8awfg7bE8W47HWGsBQRqsKP9XRodgW3Sm3tOYG8A//mlheZmRQ22w+WVN/HS63Qhz9kR\nxjqVcLzbDse67SRyrJvNEZeXl+Pmm2/G3Llz8cADD0DXlFZOT0/H1KlTsXTpUtjtduzduxfr16/H\ntGnTAADTp0/H6tWrUVZWhvLycrz44ouYMWNGQi66NQVvNBP9c92K4Ly5RY5ebjRDRCkgsJNjDH2w\nXYEa7FjPL3UTMRl0cnbb7fEFbTRDRJTMms1gv/3226isrMQLL7yAF154Qb7/f/7nf7B48WI88sgj\nmDRpEmw2GxYtWiRnrGfPno3y8nLMnDkTbrcb06ZNw9y5c1vvN0mQlmyV7vQEUu5h2/RpM9j8rCCi\nJBbYyTH2Nn2WCLs4SrR9sKXX0ekEGI06uNw+uD1eOfnBAJuIUkGzs9/ChQuxcOHCsI9lVrRRAAAg\nAElEQVQvW7Ys5P16vR5333037r777vivrh14NfsBx/KhIrXoAwBj2K3S1a/DNn1ElMzkOSumPtj+\nEhGrufkMtrTZltymTwyUz5kMerjcPrgUGWwDe14TUQrgTKXRko1mXIoMtjlMliU4g80Am4iSl7yT\nY0x9sGPPYEvJDOVGM9I3gS63lyUiRJRSOFNpBJeIRP9clzKDbQydudFujc4MNhElM50mwxwNKYNt\niSKDHVSD3fQ6gk6Q17K43L5AiQgz2ESUAjhTaQRlsGMpEVEtcoycwQ7cjuHiiIjaWDw7OdpjyGBr\nt2L3hcpge5jBJqLUwplKQ/shkvhFjprbjLCJKIkFSjiif46cwY6ii0jQIke5BjvQqs/p9sktUBlg\nE1Eq4EylEdymL/ZFjnqdAH2Y9iDagJpt+ogomUlzVGwlIv5kgzWaDHZQm77A+hSTUSoRUWSwWSJC\nRCmAM5WGtsF4TDXYTRnscNlrIESJCDPYRJTEtIsQo2GPIYOtrfGWA2xBkEvtXG6vnME2MINNRCmA\nM5VGUAY7li4iTRnscJvMAMEZayawiSiZxbrI0ecT5bkwmkWO2hIRZRcRZYmIhxlsIkohnKk0pC4i\n+jhWzksZ7Eg1gkE12IywiSiJSXNWtBlsqTwEiK5ERJDbAPpvS1OuoFjk6HB55MCbNdhElAo4U2lI\nGWxpYo/la1E5gx2mRR8QXBLCAJuIklmsGWxpgSMQ3yJHOYMtBNr0NToC5zToOWcSUfJjgK0h1WBL\nE3tcNdgRM9iswSai1BFrmz67MoNtjqZNX5idHHUCzE0BeoPDLR8fbpdcIqJkwgBbw6NpBRVPH+xI\nAXbwRjOxXiERUduRAuBoOyo5nLFlsCN2EWmaSxsUGWyWiBBRKuBMpeDziZA+QuQSkXgWOUYqEWEf\nbCJKIYEMc3TH19v92WadIMASRQY7XImITgi06WuwBzLYLBEholTAAFtB2UHEKJeIJHiRI2uwiSiF\nxFoiIgXY6VZDVPObEKFERO6D7VHOzfzYIqLkx5lKwaPogS19NRnLRjNSBtscMYPNjWaIKHXE2lGp\nrrEpwLaZYju/pkRErygRUWINNhGlAgbYCh7FXsBS5iSeRY6R2/RpFznGcIFERG1M0NRIN0fOYFua\nLw8BItRgC6GTFUaWiBBRCmB4p6DcxbFFixwjtunT3GYGm4iSWKw7OcoBdpQZbGlODNrJUVEiosQS\nESJKBZypFNQ12PEscmSbPiLqWLQBcHMCNdjGqI7XLnJUtekzBs+lBu7kSEQpgDOVgjLANsUTYEeR\nwQ5u08cAm4iSlzRHiSIgRjEf1je6AAAZtugCbG2JiGqjGc1catALXLdCRCmBAbaCV7XIsakG2xfu\n6GBxZbD5YUFESUz5LVs0CYe6GDPYOu0iR+VW6Zq5lOUhRJQqOFspKBc5GuPpg+1Rb1ITSlAXEf4N\nEFESU85Z0dRhx1oiIgXYXrkG2z+P6kPUYBtZHkJEKSK6Zd5niax0MwQB6JRmQrrF/+EQW5u+pgw2\nFzkSUQehymA3842eTxRjD7ClEhS5i0jgfm0XEQMz2ESUIhhgK2Slm7Fk/lh0ze+Ej7eVAoitTV80\nW6VzkSMRpZJYSkTsTg+kQ9KjrMGOtMjRpFnkyAw2EaUKBtgaPfPSkZNtk7c0j2ZRjySaRY7cyZGI\nUolyjmpuN8f6xsCW5hmx1mA3ndqn3Cpds6kMa7CJKFVwtgpDF+PuZT5RjCqDrV0Bz41miCiZ6WPI\nYEsLHAEg3RplH+xwXUR0AnQ6QdWWjy36iChVcLYKQzvpN0cKroFmMtiahLUAZrCJKHkpcwLKNSk1\n9c6gb/ikDLZeJ8Bqjm5Lc22JiHROaQG4shc2M9hElCo4W4UhB9hRZrClBY5ArFulM8AmouSlzGBL\nQfCO737G3cu34p+bflAdW2f398BOsxqj7lctaL4tDGSw/fOoMmHBAJuIUgVnqzDk3cui7IMdbQY7\neKOZmC+NiKjNhFrkePBYNQDgu6NVqmOlDiLR1l8DwVuxy4scm15WOZ+yRISIUgVnqzCEGDPYTkUG\nO3IXEc1tRthElMRC9cGurnMCACpqHKoyEalEJNoWfcrzywG2L9BFBADMBpaIEFHq4WwVRqyLHFUZ\n7EgBNruIEFEKUWew/X9W1/sDbIfLC7vTIz8u98COskWf8vw+eaOZpvub5kaWiBBRKuJsFYZ284Pm\nuNzRLnLU7OTIAJuIklioDHZVU4ANAOU1DvnnWDeZAdSLHEVRlANt6X5lL2z2wSaiVMHZKgxtb9bm\nuDxc5EhEHY96J0cRXp8PtQ0u+b6K2kCAXRdHgC2d3ucTVd8YSq+r7IXNnRyJKFVwtgpDOelHQ9pk\nRq/p2xp0Xi5yJKIUol3kWNvghrJyrkKZwW6MfZGjskREuahcSkaYTYoSEWawiShFcLYKI9YabKlN\nX3M1glzkSESpRDlFeX2iXH8tqawN3G5JDbbXJ6oSGoEMNhc5ElHq4VbpYcTaB9sdxTbpQKg2fQyw\niSh5aXdyVJaHAEB5U4mIzyeiQS4RiW4XR0C93iVkiYiRGWwiSj2crcIItI6K7ngpgx2pg4jyvPJt\nZrCJKIkpF2KLvkCLPolUIlLT4IIUHmemRR9gKxc5ehUZ7JCLHJnBJqIUwdkqDCnw1W4FHI4rygx2\ncBeROC6OiKiNqHdy9KGqXp3BrmzKYJ+uapTvy8+2Rn1+5YJyVQZbqsHmIkciSkGcrcIQYl3kGG0N\ntuZhlogQUTLT9sGWarCtZn+FYU2DC26PFz9X2QEAnWxG+bGozq/YaCZkDTZLRIgoBXG2CkOuC0R0\nWWwpg21miQgRdSDaPthSgF3YvZN8f2WtEz83ZbDzs20xnV8fbpFj08uaWSJCRCmIs1UYysA3mioR\naaMZY3MlIlzkSEQpRDlF+UQR1XX+EpECRYBdXuvA6Up/BjuW8hAgsPBbFKPIYDPAJqIUwdkqDJ3m\nQ6U50kYzXORIRB2JIAiqMg4pg52XZUWnpnZ8lTUOuUSkS4wBtmqRYzNdRCLtMUBElEw4W4WhbKcX\nTR12o8MDALA1U3uo3Rqd8TURJTsp2HW6vXKv66wMM3IzLQD826Wfro6vRESZdPB6FV1EBPbBJqLU\nxdkqDFXdYRQZ7EZnU4BtibzBAhc5ElGqkeatKkWLvqx0M7rlpgEAvv7+jFwm1yUntgy28ls8j9cX\ndD8XORJRKuJsFYZq5XwUvbAbHf6sjs0SWwZbe5uIKNlIiYCK2sC26NnpJgwtzAUAnCxvkO/Pz4pv\nkSMAuEME2GYj2/QRUeqJabbau3cvJk6cKN+uqanB7bffjpEjR2Ly5MlYu3at/Jgoili6dCnGjRuH\n0aNHY8mSJfB6vYm78lYWawa7QSoRaSbADq7BjuPiiIjakBQESxlsk0EHq9mAIQW5MOgDc1qGzdjs\nHKilnBM9Hl/Q/aqNZpjBJqIUEdVsJYoi3n77bcybNw9ut1u+/6GHHoLNZsO2bdvwt7/9DU8++SR2\n794NAHj99dfx6aefYt26dfjwww+xa9cuvPTSS63zW7SCWBc5SjXYac0F2CwRIaIUI33TJs1zVrMB\ngiDAajZgYJ8c+bhYO4gA6m8LQ2WwLaZABttsityliYgoWUQVYK9YsQJr1qzBwoUL5fsaGhqwceNG\n3HnnnTCbzRg6dCiuuuoqvPfeewCA999/H3PmzEF+fj7y8vKwYMECvPvuu63zW7QCVZu+ZhY5iqIY\nWOTYXA22tkSEqxyJKMlJGWyHK3hDreH9O8s/x1oeAqiTDh5P8E6OuZ0s+MXwHpg4tFvMHUqIiNpL\nVN/l/fKXv8TChQuxY8cO+b6jR4/CYDCgV69e8n19+/bFhg0bAAAlJSXo16+f6rHS0lKIohhV3bEg\nCO1SPiEF1nrlV5ECoNeHv2a70ytnuTNsxojHamsIjXoh4vEdmTTWbFXYNjjebaejjbWyiwjgL9uQ\n5q2R/fOw5qNDAIBuubaY5zNlsO4TAxnswGsI+O0VA5q9to4y1smO4912ONZtpzXGOqoAOz8/P+i+\nxsZGWCwW1X0WiwUOh38RjN1uVz1utVrh8/ngcrlgNpubfc3c3LR2XQCYlRnIxHTKtCEnQuup05WN\n8s/du3RCTk56+PNWO1W3MzNtEY8/G2RlpbX3JZxVON5tp6OMtZQYkEo4rBajPG/l5KRj3OCu+OrA\nzxhf3CPm+azOFQiqzRaT/HPnzukwGqIvCekoY50qON5th2PddhI51rGtRlGwWq1wOtXBosPhgM3m\nD0QtFovqcbvdDoPBEFVwDQAVFQ3tlsHOykpDfb1dvq+qqgEGMXwrkZ/K6uSfPU43Kivrwx6rPK//\ntiPi8R2ZNNbV1Q1R9RqnluF4t50ON9ZNv4K9qRROAFTz1oLp52HOpUWwWQwxz2d1tYE5sbpG8XN1\nA/RRfAh0uLFOchzvtsOxbjvRjnUsCYS4A+w+ffrA7Xbj5MmT6N69OwCgtLRULgspLCxEaWkphg0b\nJj9WUFAQ9flFUUSyNB1xe3yqDRC06hsDCz8tJn3EY4PWS4qIePzZwOcTz/oxaEsc77bTUcZa+tZU\nrsHW64J+L7Mx8twXDZc7MOmLPqh2dmxORxnrVMHxbjsc67aTyLGOO0ecnp6OqVOnYunSpbDb7di7\ndy/Wr1+PadOmAQCmT5+O1atXo6ysDOXl5XjxxRcxY8aMhFx0W1C16WvmX45Siz6jQdfsV5rcKp2I\nUo00T0kbwSRyR0W9Yk50N7XpEwTuEUBEqS3uDDYALF68GI888ggmTZoEm82GRYsWyRnr2bNno7y8\nHDNnzoTb7ca0adMwd+7chFx0W1D3wY58rLzJTDPbpGvPG+o2EVGy0SYCEhlgh2rTp2figYhSXEwB\n9tixY7F9+3b5dlZWFpYtWxbyWL1ej7vvvht33313y66wnSjj3uba9EW7yQwQ/EHF+JqIkp02EdBa\nAban6atZJh6IKNVxW6wwVFulN1MH2Oj0Z7DTmumBDag3sNG+DhFRMtLOU6ZEBtjKnRybMticF4ko\n1THADkM56Te3zqYlGWxmaogo2QVlsPWJ21FRVSLSVIPNeZGIUh0D7DBiymBHuU06wEWORJR6tDXR\nRmMCFznqmMEmoo6HAXYYsXURaVrkGEWJiHZrdH6OEFGy085TRj1LRIiIImGAHYZyf4PEZrC1t/lB\nQkTJrc26iHjYRYSIOgYG2GHEksFulGuwo1nkyBIRIkotrbrIUXEqt9xFJGGnJyJqFwyww1DXYEc+\nVuqDHVUGO6hNHz9JiCi5tWqbPmWJiIclIkTUMTDADkO90Uykrc/FQBeRKDaa0QbU/BwhomSnDXgN\nCQywBUGQ51u5BpuJByJKcQyww4h2oxmX2wdv0+NRtenTfG5oFz0SESUbbcBrMiSuTR8QKBPhIkci\n6igYYIchCIIcZEfKYEsdRIAoN5phH2wiSjGtuchReX43A2wi6iAYYEcgBb8+X/hjpAWOQLQZbAbY\nRJRaWnORIxCYBz0efzJDz3mRiFIcA+wIpA+VhGawg7qIxHlxRERtJKgPdoIDbKktH0tEiKijYHgX\nQSCDHT7AljLYep0AUxS7m2kDamawiSjZaftSJ3KRI6AoEWEXESLqIBhgRxBNDXajM7DJTDQt94K6\niPCDhIiSXKsvchRYg01EHQsD7AjkDHaEALuyzgkAyLCZojsnFzkSUYrRdjtqrUWObNNHRB0FA+wI\npElfjLDI8WhZHQCgV5f06M6p+eDg5wgRJTttiYhR30qLHL3cKp2IOgYG2BHooigROVpWCwDo0yUj\nhvP6TywI3MmRiJJf0E6OUaw3iYVersHmVulE1DEwwI5AaKaLSF2jCxW1/hKRc7rGEGA3jTq/BiWi\n/7+9e4+NsvrzOP7pQC/Ti5TrqvsDhCKXRaGltoAtlgWC4lKIWkQRYiBq8QIaAwliUAgaMf4g0TTQ\nIP4j1UCKURCQeGORsIIgN7ltVBqWBBArtj96mV7P/lE6zHSmw9A+M+WZvl8JoXOeM2ceTk7OfPn2\nnPPYgc852FZnsH1OEeGrCYC9MYsF0BwAt5bAbl4eIkn92pDBZiMPADvw2eRocQbbd2+Kpc0DQNjd\n+MkonVhrx/QdPHNZFdV1qqhuOgP733rEyxkbfFc2Z8bJYAOwA8+EsiMqSl0szjD7Ph+AuRGAvRFg\nB9D8HeK5ROTiX5Va98UJSVKis+nBMjezPETyzGBbcJMAEGKeAa/VJ4hIvpsau1i8BAUAwo1ZLAB/\nGez/PnLB/XNzBvtmNjg2tevdPgDcyjznqlAE2C2TDfE38RtBALgVEWAHcP1R6U2va+sa9D8nLvrU\n63+zGWxH8ykiBNgAbn1dQpzBbrkkJMFJgA3A3giwA2iZwT545rIqXfVyREVpYvo/JDV98fQP8gzs\nlu2yzhCAHYQ8g90i2ZAYF235ZwBAOJEmCCDKfYpIU4C991jT8pCRg3pq5oRBSoqPVp9kp+Jv8svA\n4d7kaOHNAkCIeD7JMSYMa7ATnATYAOyNADuAlpsc/+9yhSTpvqF91LWLQ9OyBrSp3eZkDUtEANhB\nuJeIsAYbgN2xRCQAzyUidfUNctU2SJK6J8Za0i6bHAHYgfcSkS4hbV8igw3A/giwA/Dc5Hi1qs5d\nnhTfvsmfY/oA2Emoj+nz2eQYRwYbgL0R4gXQnFRpbDT6V1WtuzwpIaZ97fKgGQA24hn/Wv2Y9Kb2\nW2xyJIMNwOYIsANwLxExRv+qbMpgR6n9O9zd52CzyxGADXiuwbb6Mekt25ekBE4RAWBzBNgBeAbY\nV69lsBPjo9sdGDvIYAOwEc9TREKSwfZov4sjKiRBPACEE7NYAM2Tvmm8vgY7Kb59y0Ok64E1p4gA\nsIPQP8nxevsJzmjmRgC2R4AdQPOc75nBvq2dGxyb2mWTIwD78D6mL7SniLDBEUAkYCYLIMpxfYlI\npZUZ7GuBNUtEANhB6DPY13/miD4AkYAcagDNXyreS0SszGATYAO49YX6mD7PDHkCD5kBEAEIsAPw\nt8nxNgsy2BzTB8BOHCF+VLrDI4VNBhtAJCDADsDh8AywQ5DBJr4GYAOec1XXUATYHu1zRB+ASECA\nHYB7k6PHg2asOUXk2t9E2ABsIPQZbM9TRFgiAsD+CLADaJ70XbUNqq1rlGRRBtvBMX0A7CPkmxy9\nThEhgw3A/giwA2ie9Msratxlt7XzMeme7ZLBBmAHoT6mz2uTI8f0AYgABNgBNGeYyytr3WXWHNPH\nJkcA9hEV4lNE5LkGm02OACIAAXYAzRvby64F2I6oKMVbkF1hkyMAOwn1EpGa2gb3zywRARAJCLAD\naP5SaZ78k+KjLck6NzfBGmwAduC1RKSL9V8b1TUeATabHAFEgJAG2KdOnVJeXp5SU1M1ffp0HT16\nNJQfZ7mWAbAVGxwl1mADsBfPxEJMtPVfG67aevfPZLABRIKQBdg1NTWaP3++Hn30UR08eFBz5szR\n888/r8rKylB9pOVaBsBWrL+WPB80Y0lzABBSjpBnsK8H2PE8yRFABAhZgL1//345HA7NmjVL0dHR\nysvLU69evbRnz55QfaTlWgbA1mWwr/1NhA3ABjwetKjoaOtPEan2WIPNvAggEoQsVVBSUqKUlBSv\nsgEDBujs2bNBvT8qKsprUg8X9wkfjijFtPgiSU6MVZcu7Z/8m9vt2tVhSXt25dnXCD36O3wira89\n50JnbBfL563uSbE6d+mqJN1025HW17c6+jt86OvwCUVfhyzArqqqktPp9CqLi4uTy+UK6v09eyZ0\n6CbA5OQEPTh2gP73fJmuVtUp0Rmth7MHqkePxHa3/XDWQP1Z5tJ/WdSe3SUnJ3T0LXQq9Hf4REpf\nJycn6P4RdyjRGaOB/Xpa3v6Cx9P0z09+1n+m/6PNc2Kk9LVd0N/hQ1+Hj5V9HbIA2+l0+gTTLpdL\n8fHxQb3/r78qOyyDnZycoLKySvVI6KrlczO8rl+5UtHuz/j3HnFaOmeUZe3ZlWdfNzaajr6diEd/\nh08k9nV+7n9ICs2c1VXSkqfS2tR+JPb1rYz+Dh/6OnyC7eubSQCELMAeOHCgioqKvMpKSko0derU\noN5vjFFDw43rhUpjo1FDAwM6HOjr8KK/w4e+Dh/6Orzo7/Chr8PHyr4OWY547Nixqq2t1caNG1VX\nV6ctW7aotLRU2dnZofpIAAAAoMOFLMCOiYnRhx9+qB07digzM1NFRUVat25d0EtEAAAAADsK6YGj\nQ4cO1aZNm0L5EQAAAMAthUelAwAAABYiwAYAAAAsFGWMYWsqAAAAYBEy2AAAAICFCLABAAAACxFg\nAwAAABYiwAYAAAAsRIANAAAAWIgAGwAAALAQATYAAABgIQJsAAAAwEKdMsA+deqU8vLylJqaqunT\np+vo0aN+623fvl0TJ05Uamqq8vPzVVpaGuY7jQyHDh3SjBkzlJ6erkmTJmnTpk1+6+Xn52vEiBFK\nS0tz/8HN+eijj3TPPfd49eGhQ4d86jG222/btm1e/ZyWlqahQ4dq2bJlPnUZ2213/PhxZWdnu1+X\nl5frxRdfVHp6usaPH6/i4uJW3xvsXI/rWvb3pUuX9MILL2j06NHKysrSypUrVVtb6/e9wc4/aNKy\nr3/55RcNGzbMq/8KCwv9vpexfXM8+/rChQs+c/fw4cP14IMP+n1vm8e16WRcLpcZN26c+eSTT0xt\nba0pLi42Y8aMMRUVFV71Tp8+bUaNGmWOHj1qqqurzdKlS80zzzzTQXdtX2VlZSYjI8Ns27bNNDQ0\nmBMnTpiMjAyzb98+n7rZ2dnm+PHjHXCXkePVV181GzZsCFiHsR0a+/btM1lZWebixYs+1xjbN6+x\nsdEUFxeb9PR0k5mZ6S5fsGCBWbRokXG5XObYsWMmMzPTHDlyxOf9wc71aNJaf8+ePdusWLHCuFwu\nc/nyZTNjxgyzZs0av20EM/+g9b7evHmzee655274fsZ28Frra0+XL182WVlZZs+ePX6vt3Vcd7oM\n9v79++VwODRr1ixFR0crLy9PvXr10p49e7zqffnll5o4caJGjhypuLg4LVq0SHv37iXTd5MuXLig\nnJwc5ebmyuFwaPjw4Ro9erQOHz7sVe+vv/7SlStXNHjw4A6608hw+vRpDRs2LGAdxrb1KisrtWTJ\nEi1fvly333671zXGdtsUFhbq448/1vz5891llZWV+vbbb7Vw4ULFxsZqxIgRmjp1qr744guf9wc7\n16OJv/6ura2V0+nU888/r9jYWPXu3Vu5ubk6cuSI3zaCmX/gv6+lpqz00KFDb/h+xnbwWutrT2++\n+aamTJmiBx54wO/1to7rThdgl5SUKCUlxatswIABOnv2rFfZ2bNnNWjQIPfr7t27q1u3biopKQnL\nfUaKYcOG6b333nO/Li8v16FDh3wmkVOnTikhIUH5+fkaM2aMnnjiiVYncfhXXV2tkpISffzxx8rK\nytKUKVO0ZcsWn3qMbett2LBBgwcP1qRJk3yuMbbb5rHHHtPWrVt17733usvOnTunrl27qm/fvu4y\nf/O3FPxcjyb++jsmJkbr169X79693WW7d+/2GwQGO//Af19LTYHc4cOHNWHCBI0fP17vvvuu3+U4\njO3gtdbXzX788UcdPnxYr7zyit/r7RnXnS7ArqqqktPp9CqLi4uTy+XyKquurlZcXJxXmdPpVHV1\ndcjvMVJdvXpV8+fP1/DhwzVhwgSvazU1NUpNTdXrr7+uH374QdOmTdOzzz6rP//8s4Pu1n5KS0uV\nnp6uJ598Urt379bKlSu1atUqn6wGY9talZWVKioq0ksvveT3OmO7bfr06aOoqCivsqqqKp+x62/+\nbq4bzFyPJv7625MxRm+99ZbOnj2r/Px8n+vBzj9ova+7d++uCRMmaPv27dq4caMOHDigDz74wKce\nYzt4NxrX69ev17x585SQkOD3envGdacLsJ1Op88gdLlcio+P9yprLehuWQ/BOX/+vJ544gl169ZN\nBQUFcji8h96kSZO0fv163X333YqJidGsWbN0xx136MCBAx10x/bTt29fFRUVKScnRzExMbrvvvs0\nffp0fffdd171GNvW+vbbb3XnnXcqNTXV73XGtnWcTqdqamq8yvzN3811g5nrcWMul0svv/yy9u7d\nq40bN6pnz54+dYKdf9C6wsJCzZ07V/Hx8erbt6/y8/P1zTff+NRjbFvj4sWLOnjwoPLy8lqt055x\n3ekC7IEDB/r8KrykpMTrV+aSlJKS4lXvypUrKi8v9/m1DG7s5MmTevzxx5Wdna21a9f6ZKAkadeu\nXdq5c6dXWU1NjWJjY8N1m7Z38uRJrV+/3quspqZGMTExXmWMbWvt3r1bU6ZMafU6Y9s6/fv3V11d\nnS5cuOAu8zd/S8HP9QisrKxMs2fPVllZmTZv3uy1PMdTsPMP/CsvL9e7776riooKd1lr8wRj2xq7\nd+9WZmamevTo0Wqd9ozrThdgjx07VrW1tdq4caPq6uq0ZcsWlZaWeh2VI0lTp07V119/rUOHDqmm\npkZr1qzRAw88oO7du3fQndtTaWmpnnnmGc2dO1evvfaaT+a6WVVVld5++2399ttvqqur04YNG+Ry\nuZSVlRXmO7av+Ph4FRQUaNeuXWpsbNSPP/6oHTt26JFHHvGqx9i21rFjx1rNXkuMbSslJiZq4sSJ\nWr16taqrq3X8+HFt375dubm5PnWDnevROmOMFixYoF69eumjjz5ScnJyq3WDnX/gX1JSkr755hsV\nFBSorq5O586dU2FhoR599FGfuoxta9xo7pbaOa5v+tyRCHD69Gkzc+ZMk5qaaqZPn+4+4mnZsmVm\n2bJl7no7duwwkydPNmlpaebZZ581paWlHXXLtrVu3TozePBgk5qa6vVnzZo1Pv1dWFhocnJyzMiR\nI82TTz5pzpw504F3bk/fffedmTp1qhk5cqSZPHmy+eqrr4wxjO1Qqa+vN0OGDAl/NoQAAAMsSURB\nVDG//fabVzlj2zr79+/3Ol7r77//NgsXLjQZGRkmJyfHFBcXu69t3brVPPzww+7Xrc31aJ1nf//8\n889m8ODB5t577/Wav2fNmmWM8e3v1uYf+NdybP/666/m6aefNqNGjTL333+/ef/9901jY6MxhrHd\nXi372hhjnnrqKfPpp5/61LVqXEcZY4xF/xkAAAAAOr1Ot0QEAAAACCUCbAAAAMBCBNgAAACAhQiw\nAQAAAAsRYAMAAAAWIsAGAAAALNS1o28AABDYkiVL9Pnnn7d6PTMzUz/99JOOHz/OEyIB4BbAOdgA\ncIu7evWqXC6XJOnIkSNasGCBvv/+e/fjep1Op6qrq9W7d++OvE0AwDVksAHgFpeUlKSkpCRJUrdu\n3SRJvXr18spWJyYmdsi9AQB8sQYbAGzuwIEDGjJkiGpqaiRJQ4YM0c6dO5Wbm6sRI0Zo3rx5unjx\nohYvXqzU1FRNnjxZ+/fvd7//jz/+0MKFC5WWlqZx48Zp+fLlqqys7Kh/DgDYHgE2AESg1atX6403\n3lBRUZFOnjypadOmadiwYfrss880aNAgrVixQpJkjNFLL72k6OhoFRcXq6CgQGfOnNHSpUs7+F8A\nAPbFEhEAiEBz5sxRRkaGJGnMmDE6f/685s2bJ0maOXOm5s+fr4aGBv30008qKSnRp59+qujoaEnS\nO++8o4ceekiXLl3S7bff3mH/BgCwKwJsAIhA/fr1c//sdDrVt29f9+u4uDg1Njaqvr5ev//+uyoq\nKpSZmenTRklJCQE2ALQBATYARKCuXb2nd4fD/4rA+vp69evXTx9++KHPNU4lAYC2YQ02AHRiKSkp\nunTpkpKSktS/f3/1799f9fX1WrVqlSoqKjr69gDAlgiwAaATy8rKUkpKil599VWdPHlSJ06c0OLF\ni/X333+rT58+HX17AGBLBNgA0Ik5HA6tXbtWiYmJmj17tubNm6e77rpLBQUFHX1rAGBbPMkRAAAA\nsBAZbAAAAMBCBNgAAACAhQiwAQAAAAsRYAMAAAAWIsAGAAAALESADQAAAFiIABsAAACwEAE2AAAA\nYCECbAAAAMBC/w/QFRrTSh5vGwAAAABJRU5ErkJggg==\n",
"text/plain": [
""
]
},
"metadata": {},
"output_type": "display_data"
}
],
"source": [
"from PIDsim import PID\n",
"\n",
"# reactor temperature setpoint\n",
"Tsp = 390\n",
"\n",
"# set initial conditions and cooling flow\n",
"IC = [C0,T0,Tcf]\n",
"\n",
"# do simulation at fixed time steps dt\n",
"tstart = 0\n",
"tstop = 8\n",
"tstep = 0.05\n",
"\n",
"# configure controller. Creates a PID object.\n",
"reactorPID = PID(Kp=8,Ki=30,Kd=5,MVrange=(0,300),DirectAction=True)\n",
"\n",
"c,T,Tc = IC # reactor initial conditions\n",
"qc = 150 # initial condition of the MV\n",
"for t in np.arange(tstart,tstop,tstep): # simulate from tstart to tstop\n",
" qc = reactorPID.update(t,Tsp,T,qc) # update manipulated variable\n",
" c,T,Tc = odeint(deriv,[c,T,Tc],[t,t+dt])[-1] # start at t, find state at t + dt\n",
"\n",
"reactorPID.manual() # switch to manual model \n",
"T -= 10 # change process variable by -10 deg\n",
"for t in np.arange(t,t+1,tstep): # simulate for 1 minute\n",
" qc = reactorPID.update(t,Tsp,T,qc) # continue to update, SP tracks PV\n",
" c,T,Tc = odeint(deriv,[c,T,Tc],[t,t+dt])[-1] # start at t, find state at t + dt\n",
"\n",
"reactorPID.auto() # switch back to auto mode\n",
"for t in np.arange(t,t+tstop,tstep): # integrate another tstop minutes\n",
" qc = reactorPID.update(t,Tsp,T,qc) # update MV\n",
" c,T,Tc = odeint(deriv,[c,T,Tc],[t,t+dt])[-1] # start at t, find state at t + dt\n",
" \n",
"# plot controller log\n",
"reactorPID.plot() # plot controller log"
]
},
{
"cell_type": "markdown",
"metadata": {},
"source": [
"### Contents of PIDsim.py"
]
},
{
"cell_type": "code",
"execution_count": 9,
"metadata": {
"collapsed": true
},
"outputs": [],
"source": [
"# %load PIDsim.py\n",
"import matplotlib.pyplot as plt\n",
"import numpy as np\n",
"\n",
"class PID:\n",
" \"\"\" An implementation of a PID control class for use in process control simulations.\n",
" \"\"\"\n",
" def __init__(self, name=None, SP=None, Kp=0.2, Ki=0, Kd=0, beta=1, gamma=0, MVrange=(0,100), DirectAction=False):\n",
" self.name = name\n",
" self.SP = SP\n",
" self.Kp = Kp\n",
" self.Ki = Ki\n",
" self.Kd = Kd\n",
" self.beta = beta\n",
" self.gamma = gamma\n",
" self.MVrange = MVrange\n",
" self.DirectAction = DirectAction\n",
" self._mode = 'inAuto'\n",
" self._log = []\n",
" self._errorP0 = 0\n",
" self._errorD0 = 0\n",
" self._errorD1 = 0\n",
" self._lastT = 0\n",
" self._currT = 0\n",
" \n",
" def auto(self):\n",
" \"\"\"Change to automatic control mode. In automatic control mode the .update()\n",
" method computes new values for the manipulated variable using a velocity algorithm.\n",
" \"\"\"\n",
" self._mode = 'inAuto'\n",
" \n",
" def manual(self):\n",
" \"\"\"Change to manual control mode. In manual mode the setpoint tracks the process \n",
" variable to provide bumpless transfer on return to automatic model.\n",
" \"\"\"\n",
" self._mode = 'inManual'\n",
" \n",
" def _logger(self,t,SP,PV,MV):\n",
" \"\"\"The PID simulator logs values of time (t), setpoint (SP), process variable (PV),\n",
" and manipulated variable (MV) that can be plotted with the .plot() method.\n",
" \"\"\"\n",
" self._log.append([t,SP,PV,MV])\n",
" \n",
" def plot(self):\n",
" \"\"\"Create historical plot of SP,PV, and MV using the controller's internal log file.\n",
" \"\"\"\n",
" dlog = np.asarray(self._log).T\n",
" t,SP,PV,MV = dlog\n",
" plt.subplot(2,1,1)\n",
" plt.plot(t,PV,t,SP)\n",
" plt.title('Process Variable')\n",
" plt.xlabel('Time')\n",
" plt.legend(['PV','SP'])\n",
" plt.subplot(2,1,2)\n",
" plt.plot(t,MV)\n",
" plt.title('Manipulated Variable')\n",
" plt.xlabel('Time')\n",
" plt.tight_layout()\n",
" \n",
" @property\n",
" def beta(self):\n",
" \"\"\"beta is the setpoint weighting for proportional control where the proportional error\n",
" is given by error_proportional = beta*SP - PV. The default value is one.\n",
" \"\"\"\n",
" return self._beta\n",
" \n",
" @beta.setter\n",
" def beta(self,beta):\n",
" self._beta = max(0.0,min(1.0,beta))\n",
" \n",
" @property\n",
" def DirectAction(self):\n",
" \"\"\"DirectAction is a logical variable setting the direction of the control. A True\n",
" value means the controller output MV should increase for PV > SP. If False the controller\n",
" is reverse acting, and ouput MV will increase for SP > PV. IFf the steady state\n",
" process gain is positive then a control will be reverse acting. \n",
" \n",
" The default value is False.\n",
" \"\"\"\n",
" return self._DirectAction\n",
" \n",
" @DirectAction.setter\n",
" def DirectAction(self,DirectAction):\n",
" if DirectAction:\n",
" self._DirectAction = True\n",
" self._action = +1.0\n",
" else:\n",
" self._DirectAction = False\n",
" self._action = -1.0\n",
" \n",
" @property\n",
" def gamma(self):\n",
" \"\"\"gamma is the setpoint weighting for derivative control where the derivative error\n",
" is given by gamma*SP - PV. The default value is zero. \n",
" \"\"\"\n",
" return self._gamma\n",
" \n",
" @gamma.setter\n",
" def gamma(self,gamma):\n",
" self._gamma = max(0.0,min(1.0,gamma))\n",
" \n",
" @property\n",
" def Kp(self):\n",
" \"\"\"Kp is the proportional control gain.\n",
" \"\"\"\n",
" return self._Kp\n",
" \n",
" @Kp.setter\n",
" def Kp(self,Kp):\n",
" self._Kp = Kp\n",
" \n",
" @property\n",
" def Ki(self):\n",
" \"\"\"Ki is the integral control gain.\n",
" \"\"\"\n",
" return self._Ki\n",
" \n",
" @Ki.setter\n",
" def Ki(self,Ki):\n",
" self._Ki = Ki\n",
" \n",
" @property\n",
" def Kd(self):\n",
" \"\"\"Kd is the derivative control gain.\n",
" \"\"\"\n",
" return self._Kd\n",
" \n",
" @Kd.setter\n",
" def Kd(self,Kd):\n",
" self._Kd = Kd\n",
" \n",
" @property\n",
" def MV(self):\n",
" \"\"\"MV is the manipulated (or PID outpout) variable. It is automatically\n",
" restricted to the limits given in MVrange.\n",
" \"\"\"\n",
" return self._MV\n",
" \n",
" @MV.setter\n",
" def MV(self,MV):\n",
" self._MV = max(self._MVmin,min(self._MVmax,MV))\n",
" \n",
" @property\n",
" def MVrange(self):\n",
" \"\"\"range is a tuple specifying the minimum and maximum controller output.\n",
" Default value is (0,100).\n",
" \"\"\"\n",
" return (self._MVmin,self._MVmax)\n",
" \n",
" @MVrange.setter\n",
" def MVrange(self,MVrange):\n",
" self._MVmin = MVrange[0]\n",
" self._MVmax = MVrange[1]\n",
"\n",
" @property\n",
" def SP(self):\n",
" \"\"\"SP is the setpoint for the measured process variable.\n",
" \"\"\"\n",
" return self._SP\n",
" \n",
" @SP.setter\n",
" def SP(self,SP):\n",
" self._SP = SP\n",
" \n",
" @property\n",
" def PV(self):\n",
" \"\"\"PV is the measured process (or control) variable.\n",
" \"\"\"\n",
" return self._PV\n",
" \n",
" @PV.setter\n",
" def PV(self,PV):\n",
" self._PV = PV\n",
"\n",
" def update(self,t,SP,PV,MV):\n",
" self.SP = SP\n",
" self.PV = PV\n",
" self.MV = MV \n",
" if t > self._lastT:\n",
" dt = t - self._lastT\n",
" self._lastT = t\n",
" if self._mode=='inManual':\n",
" self.SP = PV\n",
" self._errorP1 = self._errorP0\n",
" self._errorP0 = self.beta*self.SP - self.PV\n",
" self._errorI0 = self.SP - self.PV \n",
" self._errorD2 = self._errorD1\n",
" self._errorD1 = self._errorD0\n",
" self._errorD0 = self.gamma*self.SP - self.PV\n",
" if self._mode=='inAuto':\n",
" self._deltaMV = self.Kp*(self._errorP0 - self._errorP1) \\\n",
" + self.Ki*dt*self._errorI0 \\\n",
" + self.Kd*(self._errorD0 - 2*self._errorD1 + self._errorD2)/dt\n",
" self.MV -= self._action*self._deltaMV\n",
" self._logger(t,self.SP,self.PV,self.MV)\n",
" return self.MV \n"
]
},
{
"cell_type": "code",
"execution_count": null,
"metadata": {
"collapsed": true
},
"outputs": [],
"source": []
},
{
"cell_type": "markdown",
"metadata": {},
"source": [
"\n",
"< [PID Control - Laboratory](http://nbviewer.jupyter.org/github/jckantor/CBE30338/blob/master/notebooks/04.10-PID-Control.ipynb) | [Contents](toc.ipynb) | [Interactive PID Control Tuning with Ziegler-Nichols](http://nbviewer.jupyter.org/github/jckantor/CBE30338/blob/master/notebooks/04.12-Interactive-PID-Control-Tuning-with-Ziegler--Nichols.ipynb) >
 "
]
}
],
"metadata": {
"kernelspec": {
"display_name": "Python 3",
"language": "python",
"name": "python3"
},
"language_info": {
"codemirror_mode": {
"name": "ipython",
"version": 3
},
"file_extension": ".py",
"mimetype": "text/x-python",
"name": "python",
"nbconvert_exporter": "python",
"pygments_lexer": "ipython3",
"version": "3.7.4"
},
"widgets": {
"state": {
"0593fc1c63274b289c5fff97127efb4d": {
"views": [
{
"cell_index": 15
}
]
}
},
"version": "1.2.0"
}
},
"nbformat": 4,
"nbformat_minor": 2
}
"
]
}
],
"metadata": {
"kernelspec": {
"display_name": "Python 3",
"language": "python",
"name": "python3"
},
"language_info": {
"codemirror_mode": {
"name": "ipython",
"version": 3
},
"file_extension": ".py",
"mimetype": "text/x-python",
"name": "python",
"nbconvert_exporter": "python",
"pygments_lexer": "ipython3",
"version": "3.7.4"
},
"widgets": {
"state": {
"0593fc1c63274b289c5fff97127efb4d": {
"views": [
{
"cell_index": 15
}
]
}
},
"version": "1.2.0"
}
},
"nbformat": 4,
"nbformat_minor": 2
}

